2
Behavioral Models and the Resources at Risk
Behavioral Models for Spills of Nonfloating Oils
Based on an understanding of the physical and chemical properties of nonfloating oils (mostly from observations of past spills), Behavioral models have been developed (Michel et al., 1995). These models are descriptive, qualitative predictions of how oils with a density near or higher than the density of the receiving water might behave. The key factors that determine the behavior of spilled nonfloating oils are: water density, current speed, and the potential for interaction with sand.
Water Density
If the ratio of the density of oil to the density of the receiving water is greater than 1.0, the oil will not float. If it less than 1.0, the oil will float. If it is within a few percent of 1.0, then the oil is much more likely to become submerged by wave action. Figure 2-1 shows the relationship between the density and salinity of the water for a fixed temperature. The density is also shown in terms of the API (American Petroleum Institute) gravity. Oils with higher densities than the receiving water (above the line) will sink; oils with lower densities that the receiving water (below the line) will initially float.
Current Speed
If current speeds are greater than 0.1 m/s, nonfloating oils will be suspended in the water column. If the currents are very slow, oils heavier than the receiving water will sink to the bottom (Nielsen, 1992).
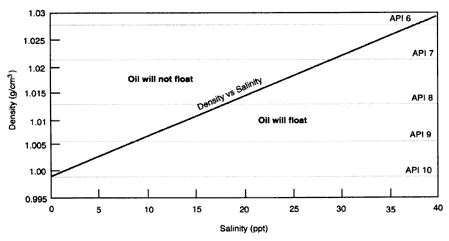
Figure 2-1
The relationship between water density and salinity at a temperature of 15°C. The density is also shown in API gravity units (right vertical axis).
Potential for Interaction with Sand
When floating oil is mixed with 2 to 3 percent sand, it becomes heavier than water and sinks (Michel and Galt, 1995). The density of sand grains is much higher than the density of silt or clay particles. Figure 2-2 is a schematic illustration showing the relationships among these factors and how they affect the short-term behavior of nonfloating oils. The density of oil relative to the receiving water is important only in determining whether the oil will initially float. Significant currents can keep heavier-than-water oil suspended in the water column. Any oil still on the surface or suspended in the water column will still sink if it mixes with sand also suspended in the water column. The models in Figures 2-3 and 2-4 illustrate combinations of factors that influence the behavior of nonfloating oils.
Oil Lighter than Water, Low Sand Interaction
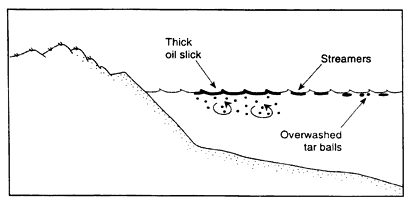
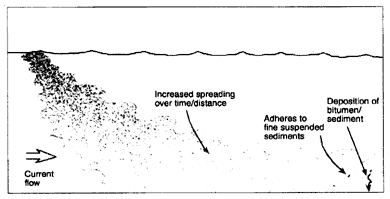
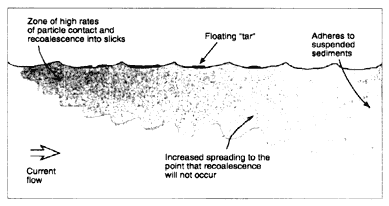
If the oil-to-water density ratio is less than 1.0, the oil will initially float. At 15°C, oils with an API gravity above 6.5 (Figure 2-1) will still be lighter than seawater with a salinity of 35 parts per thousand. These oils will float, at first in contiguous slicks that may quickly (often within a few kilometers) break up into widely scattered fields of large mats and tar balls that can spread over large distances and become reconcentrated again in convergence zones (Figure 2-3a).
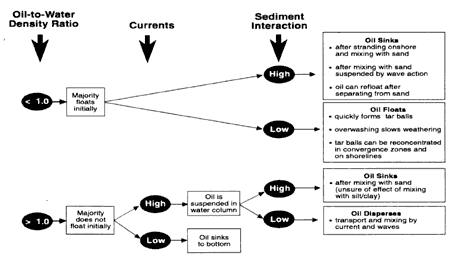
Figure 2-2
Behavior of spilled nonfloating oils.
Because of the higher viscosities of heavy oils, the tar balls are more persistent than for spills of light and medium oils. More important, however, as the density of the oil approaches that of the water, these tar balls tend to become ''overwashed'' by wave action making them very difficult to track and slowing most weathering processes (e.g., evaporation or formation of a "skin") (Lee et al., 1989). Furthermore, if oil emulsifies, the emulsion can contain 50 to 80 percent water making the density of the oil even closer to the density of the water. Evaporation of emulsified oils is slow, and, unless they interact with sediment, they will remain floating. When tar balls are eventually stranded, sometimes hundreds of kilometers away from the original spill site, the oil can still be relatively fresh and have a significant impact on the water surface and shoreline resources (see Box 2-1). Because the oil still floats, this type of spill is not considered further in this report. After the evaporative loss of the lighter fraction, particularly of the cutter stock in bunker fuels, the remainder might sink, but this has been observed at only one spill (Lee et al., 1992; Michel and Galt, 1995).
Oil Lighter than Water, High Sand Interaction
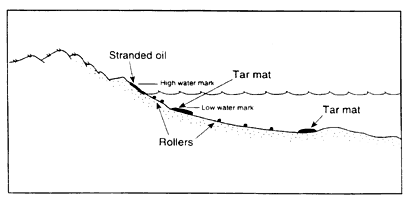
Spilled oil that is lighter than the receiving water can still sink, either by becoming stranded on sand beaches or by mixing with sand in the surf zone. In several spills, such as the IXTOC I (Gundlach et al., 1981), Alvenus (Alejandro and Buri, 1987), and Haven (Martinelli et al., 1995), heavy oils floated initially
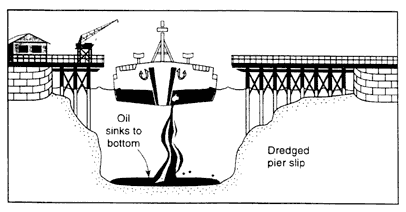
Figure 2-3d
Oil-to-water density > 1.0; low currents; majority of oil sinks.
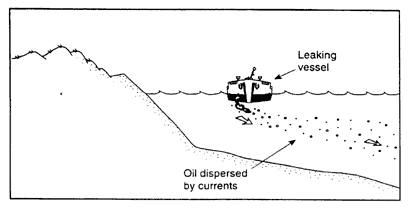
Figure 2-3e
Oil-to-water density > 1.0; high currents; oil disperses in water column.
and became stranded on sand beaches but then were eroded from the beaches and sank, forming tar mats between nearshore bars. In these cases, the oil was too viscous to penetrate the sand; instead, the sand coated the oil layers and/or mixed with the viscous oil as it was eroded from the beaches by wave action. The oil/sand mixture contained only a few percent sand and was deposited at the toe of the beach just offshore (Figure 2-3b). The distribution of sunken oil/sand tar mats was highly variable, ranging from thick, continuous deposits tens of meters long to small widely scattered tar balls. If there was current activity, especially generated by waves breaking on the shore, the oil/sand mixture formed cigar-shaped "rollers" that were scattered on the bottom or accumulated into mats in the
|
BOX 2-1 The Nestucca Spill The Nestucca spill in December 1988 released 5,500 barrels of heavy marine fuel oil with an API gravity of 12.1 three kilometers off Grays Harbor, Washington. The spilled oil quickly formed tar balls that moved below the water surface (i.e., were overwashed by waves) and could not be tracked visually. Two weeks later, oil unexpectedly came ashore along the coast of Vancouver Island, Canada, 175 kilometers north of the release site, contaminating 150 kilometers of shoreline (NOAA, 1992). The oil had a significant effect on the large number of marine birds wintering in the area. Of the 10,300 birds collected, about 9,300 were either already dead or died in treatment centers. Many more were believed to have died but were never collected. |
troughs of offshore bars. These rollers picked up more sand and shell fragments as they moved, making them heavier.
Experts have long been concerned that oil spilled in turbulent waters with heavy loads of suspended silt and clay (i.e., glacial meltwater, such as upper Cook Inlet and the Yukon River) would mix with the sediments and sink (Kirstein et al., 1985). Laboratory studies have shown that oil mixed with water with heavy suspended sediment loads does adhere to the sediments, with concentrations up to 0.1 gram of oil per gram of solid (McCourt and Shier, 1998). However, this process is likely to result in the deposition of oiled sediments rather than the transport of bulk oil to the bottom.
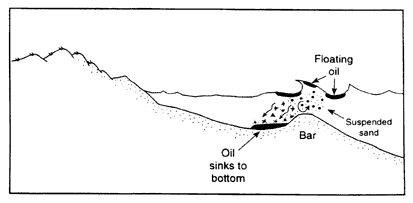
During the Tampa Bay and Morris J. Berman spills (Box 2-2), response teams observed that floating oil sank by mixing with sand in nearshore waters, without coming into contact with intertidal sediments on the shoreline (Figure 2-3c). If a floating slick of heavy oil drifts into shallow water along an exposed shoreline, it is more likely to be mixed into the water column by wave turbulence. If the bottom is sandy, the sand may be suspended in the water column by waves and could mix with the oil. The suspended sand concentrations in breaking waves is commonly 300 to 500 mg/L and can easily reach 5,000 mg/L (Kana, 1979), compared to typical concentrations of fine-grained suspended sediment of 20 mg/ L in estuaries and nearshore waters. Because the specific gravity of quartz is 2.65, it only takes 2 to 3 percent sand by weight mixed into oil to make it heavier than seawater. Again, high viscosity is an important factor, because viscous oils tend to form large tar balls (rather than small droplets) that pick up sand. The oil/sand mixture can be carried by long shore currents and deposited in relatively sheltered areas where it can form extensive, thick layers of oil/sand on the bottom.
|
BOX 2-2 The Morris J. Berman Spill On January 7, 1994, the Morris J. Berman barge ran aground just offshore San Juan, Puerto Rico, releasing about 18,000 barrels of heavy fuel oil (API gravity of 9.5). Although much of the oil floated, response teams reported finding oil on the bottom within the first 24 hours, and eventually mats of submerged oil were found in both offshore areas and on the landward side of nearshore reefs. Most of the sunken mats were within 1 or 2 kilometers of the vessel, although one site was 110 kilometers from the release site. The oil adhered to rocky surfaces and coated seagrass beds (Burns et al., 1995). It was later determined that most of the oil on the bottom had sunk without coming into contact with the shore (Michel et al., 1995). The oil contained a few percent sand and could readily refloat In seawater and recontaminate the adjacent shoreline once it was separated from the sand. Three different methods were used to remove the oil: diver-directed vacuuming of the more liquid oil; manual pickup by divers of the more viscous patches; and dredging of large deposits in a small bay (Burns et al., 1995; Ploen, 1995). |
Oil Heavier than Water, Low Currents
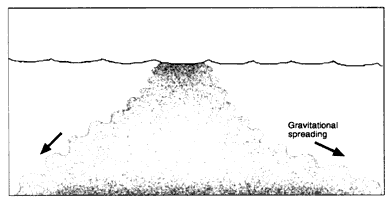
If the density of the oil is higher than the density of the receiving water, some of the oil can form a sheen, but the majority does not float. As the oil mixes into the water column, it forms small droplets, ranging in size from approximately 0.5 microns to several millimeters. If the water column is strongly stratified, some of the oil droplets may accumulate on the pycnocline, provided that they are less than the underlying water. If current speeds are low, oil that is more dense than the water sinks and accumulates on the bottom (Figure 2-3d). Direct sinking in low-flow areas was observed after the Sansinena oil spill (see Box 2-3) while it was docked at a pier (Hutchison and Simonsen, 1979), and the Mobiloil spill (in the lee of the grounded vessel) (Kennedy and Baca, 1984).
Suspended oil can sink when the oil is transported into low-flow areas similar to the way fine-grained sediments are deposited in estuaries during slack periods of the tide. However, oil droplets can be readily remobilized by tidal currents, so long-term accumulation is likely only in areas where wave-generated, tidal, or riverine currents have little effect. Examples of such areas include abandoned channels, dredged channels or pits, depressions adjacent to piers caused by "propeller wash" of anchoring vessels, dead-end canals, and the lee side of natural and man-made structures. If the oil does accumulate on the bottom, the oil droplets recoalesce into pools of liquid oil that can be tens of centimeters thick. Evaporation and photo-oxidation of sunken oil are much slower than for floating oil slicks, and the oil tends to remain as a liquid on the bottom. Dissolution from thick mats is slow (Lee et al., 1989). Observations of spills have shown that this
|
BOX 2-3 The Sansinena Spill On December 17, 1976, the tanker SS Sansinena exploded while loading fuel in Los Angeles harbor, releasing more than 33,000 barrels of bunker fuel oil (API gravity 7.9 to 8.8). Approximately 200 barrels floated, but the majority of the oil sank. Divers reported large pools of oil up to three meters deep on the harbor bottom, where the oil had settled into depressions (Hutchison and Simonsen, 1979). Initial recovery was by diver-directed vacuum removal and separation in tanks mounted on a barge, but this method was abandoned because of the great difficulty of moving the suction head along the uneven bottom. Next, diver-guided hydraulic pumps were used on thick accumulations close to the pier. Specially designed pumping units consisting of a prime mover and hydraulic pumps on a barge were then used to collect oil from outer depressions. Nearly 16,000 barrels were recovered during the initial recovery operations. Eventually, a suction head and pump device was designed on site for recovery of the large quantities of oil still remaining on the bottom. This device had to be operated according to directions from a diver because some of the oil pools had become silted over and even had marine life living in the silt, making the oil difficult to locate. During the next 90 days, 10,300 barrels were recovered from the harbor bottom. Over a 16-month period, 33,000 barrels, nearly all of the spilled volume, were recovered. |
type of oil does not initially adhere to or mix with large amounts of fine-grained sediments under water.
Oil Heavier than Water, High Currents
If currents are greater than about 0.1 m/s, oil droplets stay suspended in the water column and disperse (Figure 2-3e). In rivers and most nearshore coastal settings, the oil is not likely to accumulate on the bottom because the currents are strong enough to keep it suspended in the water column. For example, little or no oil accumulation on the bottom was observed after heavy-oil spills in the Columbia River (Kennedy and Baca, 1984), the Mississippi River near Vicksburg (Weems et al., 1997) and in Puget Sound (Yaroch and Reiter, 1989). However, even in strong currents, heavy oils can accumulate in sheltered areas. For example, after about 4,760 barrels of slurry oil were spilled into the Mississippi River, nearly 50 percent of the oil was recovered from the bottom, but only from the lee created when the leaking barges were pushed at a 45-degree angle against the river bank (Weems et al., 1997). No other significant amounts of oil were found in extensive surveys. The oil was not expected to adhere initially to debris or
vegetation as it mixed into the water column because fresh oil generally does not stick to water-wet surfaces.
Spills of Emulsified Fuels
Emulsified fuels (anthropogenic fuels manufactured by mixing water and surfactants with liquid oils or solid hydrocarbon products) behave very differently. Because only one small accidental spill of emulsified fuel has been reported (Sommerville et al., 1997), our understanding of the behavior of these oils is based mostly on research conducted specifically with OrimulsionTM, an emulsified fuel manufactured from bitumen produced in Venezuela. Laboratory and field experiments on emulsified oils have been conducted in Canada (Jokuty et al., 1995), the United States (Deis et al., 1997; Ostazeski et al., 1997), Venezuela, and Europe (Sommerville et al., 1997). In freshwater, the surfactant in emulsified fuels will maintain its effectiveness over longer periods of time, preventing recoalescence of the bitumen particles. In low-flow conditions (Figure 2-4a), the spilled oil will settle to the bottom of the water column. In these quiescent conditions, the oil has little potential for mixing with sediment, except in the long term by bioturbation.
In freshwater with currents, the predispersed bitumen particles will slowly descend to the bottom down current (Figure 2-4b), and the surfactant will remain effective for a limited period of time, preventing recoalescence of the particles. The eventual fate of the bitumen particles is uncertain, particularly in terms of interaction with fine-grained sediments. Because the bitumen particles are highly adhesive, it is likely that they will adhere to suspended sediments and eventually be deposited in low-flow zones.
In saltwater, the emulsified oils will initially form clouds of dispersed particles in the upper 1 or 2 meters of the water column (Figure 2-4c). Laboratory and field tests have shown that surfactants quickly lose their effectiveness in saltwater. In areas with high bitumen concentrations, the particles can recoalesce and rise to the surface, forming tarry slicks. In wave-tank experiments, the tar coated the glass sides of the wave tank (Jokuty et al., 1995). However, in open water, the particles would disperse. Therefore, options for containing and recovering spilled emulsified oils quickly decrease over time.
Refloating Mechanisms
Sunken oil can refloat, creating significant problems for spill-response teams and a chronic source of exposure. In the Morris J. Berman spill, months after the spill large quantities of liquid oil refloated, recontaminating beaches and exposing resources in the water-column and on the surface to oil after the bulk of the floating oil had been recovered. There are three mechanisms for refloating oil: (1) still-buoyant oil can separate from the sand; (2) wave-generated currents can
loosen and resuspend pieces of buoyant oil from the bottom; and (3) increases in water and/or oil temperature can make the oil less viscous and/or more buoyant.
Spill-response teams often assume that oil refloats because of a short-term change in temperature (e.g., in the afternoon when the water warms up). However, it is unlikely that short-term changes in temperature can cause oil-sand mixtures to sink in one situation and float in another because differential oil and water expansion coefficients are very small. Hence, oil on the bottom and the overlying water expand at about the same rate, and relative changes in density are small.
A more likely mechanism for refloating sunken oil is the physical separation of the sediment from oil (Michel and Galt, 1995). If one dumps sand into a can of motor oil, the sand falls through the oil by gravity and forms a layer of sand on the bottom. Settling rates through more viscous oils would be longer but could be increased in the field by wave motion and other physical processes. In the Morris J. Berman spill, buoyant oil droplets were observed breaking off layers of oil on the bottom, somewhat like droplets being released from the heated mass in lava lamps. These droplets are believed to have been formed as the still-buoyant oil became less viscous during the daytime heating of the water, allowing the oil to separate from the sand and droplets to break away from the submerged oil by wave action generated by the land-sea breezes.
Potential Effects of Nonfloating-Oil Spills
When a floating oil is spilled, response teams typically have to recover oil slicks, clean up oil stranded on the shoreline, and recover and treat animals along the shoreline and in the water. Their focus is on the water surface and shoreline, the so-called "bath-tub ring." Life forms in the water column and benthic habitats are usually considered to be at less risk of exposure and injury from floating oil slicks than from nonfloating oils. Table 2-1 compares the predicted impacts of nonfloating-oil spills and floating-oil spills on shoreline and benthic habitats, major assemblages of fish and wildlife, and human-use resources. Spills of nonfloating oils are expected to have less impact on shoreline habitats because smaller amounts of oil are likely to be stranded and cleanup activities are likely to be less disruptive (Scholz et al., 1994). Any oil that is stranded, however, is likely to be very persistent because of the slow natural removal rates for heavy, adhesive oils. Nonfloating oils are less likely to penetrate porous sediments or wetland vegetation because of their high viscosities and adhesiveness (Harper et al., 1995).
Impacts on water-surface resources are also expected to be lower from spills of nonfloating oils because of the significant reduction in the amount of oil on the water surface. If the oil refloats, it could be a chronic source of exposure to both water-surface and shoreline resources, but the risk is likely to be limited to areas adjacent to sunken oil deposits (NOAA, 1995).
All water-column and benthic habitats are at increased risks from spills of
TABLE 2-1 Relative Changes in the Resources at Risk from Spills of Nonfloating Oils Compared to Floating Oils
nonfloating oils (Scholz et al., 1994). Oils that quickly sink or are suspended in the water column have greater impacts on organisms in the water column because more of the water-soluble fraction of the oil dissolves rather than evaporates. Oil on the surface is primarily weathered by evaporation to the atmosphere and, to a lesser degree, to the water column by dissolution. Oils suspended in the water column or deposited on the bottom are less likely to evaporate but more likely to dissolve, although the water-soluble fraction of heavy oils is usually very low. Consequently, the water column can have higher concentrations of toxic fractions from nonfloating oils than from floating oils. Dissolution tends to be a slower process than evaporation (Lee et al., 1989, p.37), thus increasing potential exposure times. In the Morris J. Berman spill in Puerto Rico, divers observed dead fish, living fish with lesions and tumors, and many lethargic territorial fish in nearshore waters adjacent to the spill site (Vincente, 1994). Mobile species may be able to move to uncontaminated areas, thus reducing their exposure.
Nonfloating oils are often high in polynuclear aromatic hydrocarbons (PAHs), which are the primary source of both acute and chronic toxicity to aquatic organisms. Naphthalene compounds (two-ringed aromatics) have been shown to be more toxic than lightweight aromatics, such as benzene and toluene (Anderson et al., 1987). In terms of the water-soluble fraction, bunker C is as toxic as diesel oil (Markarian et al., 1993). Thus, even though heavy residual oils are not usually considered to be acutely toxic to fish (NOAA and API, 1995), oils that are mixed into the water column without weathering by evaporation on the water surface first may have a higher fraction that dissolves and, therefore, may be more acutely toxic to organisms in the water column.