4
Surveillance and Monitoring
A well-coordinated public health system for the detection and control of emerging infections and antimicrobial resistance requires an effective partnership among health care providers, academic health centers, and the traditional public health system. Effective infectious disease surveillance necessitates that physicians send samples to competent laboratories for confirmation of their diagnoses and that they report the findings through the public health system. Yet, each aspect of infectious disease surveillance is evolving independently and rapidly.
Cost-reduction and reimbursement efforts through managed care emphasize the heavy reliance on empiric care rather than laboratory data. This reduces the use of diagnostic tests, discourages visits to specialists, and shifts care to ambulatory settings, where the capabilities for diagnosis and care are less than those found at inpatient facilities. Yet, standard types of managed care organizations, such as the staff model and preferred provider health maintenance organizations (HMOs), are being bypassed by some large payers in favor of the direct purchase of services from individual physicians and small laboratories. For example, in Minnesota, a state that has been a leader in setting trends in managed care, large buyers of health care (employers, primarily corporations) are dictating coverage. The factors that determine coverage often differ between such groups and the individual consumer or public health groups. One-third of Minnesotans are forced to change health plans on an annual basis, based on their employers' financial situation (Michael Osterholm, State Epidemiologist and Chief, Acute Disease Epidemiology Section, Minnesota Department of Health, Minneapolis, personal communication, March 23, 1998). This situation may signal the end of large, centralized laboratories, a change that could have major implications for disease surveillance as outcome measurement and population-based studies become more difficult. Consequently, there is a particular concern that managed care systems may reduce their levels of compliance in infectious
disease reporting and inadvertently circumvent the requisite comprehensive analysis of pathogenic isolates needed for disease tracking.
Laboratory practices are also changing because fewer tests are conducted to determine new pathogens and levels of susceptibility to antimicrobial agents. Moreover, since large laboratories that are not likely to be in the same state as a patient's HMO conduct much of the laboratory testing, a widespread outbreak might not be detected through reporting by a single HMO. This situation is problematic because HMO patient databases are often uncoordinated and poorly networked, consequently making disease tracking even more complex and difficult.
The uniqueness of each managed care organization and the essential capabilities of private and public microbiological laboratories underscore the importance of working together. The challenge that lies ahead is how to plan a comprehensive infectious disease surveillance system that allows maximum coordination and flexibility for change. The opportunities are many for all parties to become involved in negotiating how the evolving health care system can improve public health.
The presentations described below examine the opportunities and challenges for academic health centers and managed care organizations to work together in the fields of basic and clinical research in addressing issues related to emerging infections.
AN EMPLOYER'S PERSPECTIVE
Presented by Thomas J. Davies, M.P.A., J.D.
Manager of Managed Care, GTE Corporation
Integrated health care delivery systems offer the greatest hope for improving quality while managing costs, including the quality and costs of infectious disease control. Employers as purchasers of health care can play an important role in determining the extent and quality of care for their employees. They can also be instrumental in helping determine how care is administered and used. Many believe that consumers, not employers, have largely driven the growth toward managed care because of their preference for the greater benefits and lower costs of HMOs. Subsequently, employers have responded in a number of ways depending on their size and their perceptions about how they can influence the delivery of health care. Small and midsize companies largely regard their role as helping to pay premiums, that is, to alleviate some of the costs of health care for the employee by arranging and paying for health insurance. Large companies, on the other hand, have unique opportunities to make more of an investment and move toward value-driven purchasing strategies, whereas smaller and midsized firms tend to choose health plans on the basis of cost. Many employers see managed care as their best hope for containing costs while maintaining
quality. Such employers believe that quality health care represents a better value and costs less.
In recent years, 33 large companies on the West Coast of the United States have formed the Pacific Business Group on Health (PBGH) to provide leadership to steer the health care market toward increased quality and accountability. This coalition of businesses strives to improve quality through the administration of their health care programs. The strategy includes encouraging employees to select certain systems of care voluntarily and ensuring that those decisions are based on value. However, employers tend to regard health care in terms of prevention, acute care, and chronic care, not in terms of specific infections, emerging or otherwise. From the point of view of PBGH, too many managed care organizations are only managing costs and are not paying attention to accountability. A larger problem is the lack of timely, accurate, and complete information about employee's health care encounters that can be used appropriately by health plans, individual practitioners, and payers. These data were previously available through the claims information routinely collected by fee-for-service, indemnity-based plans.
MANAGED CARE AND INFECTIOUS DISEASE SURVEILLANCE: OPPORTUNITIES FOR COLLABORATION
Presented by Denise Koo, M.D., M.P.H.
Director, Division of Public Health Surveillance and Informatics, Centers for Disease Control and Prevention
Public health surveillance and monitoring activities are a cornerstone of public health practice, providing information essential for assessing public health status, monitoring trends, suggesting public health priorities, and evaluating the effectiveness of public health programs. In one of these monitoring functions, the Centers for Disease Control and Prevention (CDC) manages a system of national surveillance of notifiable diseases in coordination with state health departments and publishes data on a weekly basis.
When considering how public health surveillance may be conducted through managed care organizations, there is great potential for many benefits and opportunities to be gained through partnerships with managed care. Because the improved health of a population is a primary goal of managed care organizations and public health agencies, both will benefit by sharing data and working together. There are, however, several impediments to conducting surveillance through managed care organizations.
First, there is a lack of understanding on the part of health care providers about the roles of state and local public health departments in infectious disease surveillance. In general, few health care providers understand the importance of public health surveillance, the role of the provider as a source of data, and the role of the health department in response to infectious diseases. Some of this
lack of understanding stems from the fact that public health agencies may not always provide feedback to providers on how the data are used and may not make the data available to the public (or at least to providers or potential users of the data). For example, public health systems can provide surveillance data about the incidence of a particular disease in the community at large. These data can help raise or lower the threshold of clinical suspicion for that condition and encourage early detection and appropriate treatment of that infectious disease. Public health systems, as part of the response to cases of infectious disease, can also facilitate the tracking of contacts who have been exposed to a disease and subsequent administration of prophylactic treatment of contacts, regardless of whether the original case patient is enrolled in the same HMO. in general, managed care systems provide an excellent opportunity for public health agencies to clarify their role in infectious disease surveillance and response and to build partnerships to ensure communication concerning cases of disease and opportunities for prevention of the spread of disease. Public health officials need to disseminate infectious disease surveillance data and reports to health care providers in a timely, readily accessible, and useful manner. One available mechanism for the expeditious dissemination of this information is the Internet.
An additional impediment to public health surveillance is proprietary concerns about patient data. Many managed care organizations regard their consumer data as proprietary. They fear that their data will be used to measure their performance against the performances of other managed care organizations, particularly concerning items not entirely under their control (such as disease incidence). Public health agencies need to reassure managed care organizations that public health surveillance is not a regulatory function and that the purpose of conducting disease surveillance is to monitor the status of the public's health and to identify opportunities for improving community health status.
A disincentive to managed care organizations is duplication of data entry. To make the best use of the potential wealth of data in managed care information systems, public health agencies need to work with managed care organizations to ensure efficient, direct, and confidential reporting of data from managed care laboratory data systems to public health agencies for surveillance purposes. Additionally, public health agencies need to devise mechanisms to obtain for surveillance purposes other relevant data from managed care inpatient and outpatient information systems, such as enrollment and encounter records. Some states are already conducting pilot studies precisely directed to the achievement of such goals.
Some public health officials worry that fewer diagnostic tests will be performed in the managed care setting because of cost-cutting requirements (for example, tests for self-limited illnesses like diarrhea) leading to the decreased availability of information about emerging infectious diseases. There is concern not only about whether diseases or conditions will be detected, but there is also concern that the data captured in managed care information systems for such patients either will be insufficient for determination of the etiology of a disease or will be inaccurate. Some public health officials are concerned that the data
gathered by managed care organizations are weighted toward economic considerations (i.e., for reimbursement purposes) and might not contain medical information necessary for surveillance purposes. Some pilot studies conducted by managed care organizations and public health agencies are trying to determine the quality, accuracy, and availability of data regarding infectious diseases. These studies are showing that diagnostic testing may actually be no less prevalent among providers associated with managed care organizations than among other providers. If it is determined that diagnostic testing is not used because of cost considerations, partnerships between public health laboratories and managed care organizations could be negotiated to ensure that the testing needed for public health purposes is completed. To achieve this goal, public health agencies and managed care organizations need to work with medical information specialists as well as others to develop standards for computer-based patient records to ensure that such records will be useful for population-based health.
Finally, apprehension about the sharing of electronic data records with outsiders, including public health officials, is widespread among health care providers inside and outside of managed care. However, to accomplish their jobs effectively, public health agencies routinely have access to sensitive personal data, such as sexually transmitted disease contacts or sexual or other risk factors for disease. To date, public health agencies have an excellent record of accomplishment in safeguarding patient confidentiality and using these data only for public health purposes. However, fears about confidentiality have been used as an argument against the sharing of electronic medical data even for public health purposes. These concerns underscore the importance of privacy legislation pending before the U.S. Congress.
The field of public health is increasingly interested in expanding the amount and type of data used for surveillance to monitor old, new, or emerging public health problems. Public health officials often struggle to reconcile data about cases of infectious diseases reported by distinct sources and are seeking means of obtaining and linking the data already available electronically. The administrative simplification portion of the 1996 Health Insurance Portability and Accountability Act, which requires the potential use of standards developed by industry, provides additional incentives to work toward these solutions. Public health agencies must include managed care organizations as partners in this effort.
COLLABORATIVE SURVEILLANCE EFFORTS AND MONITORING OF DATA
Presented by Richard Platt, M.D.
Director of Research, Harvard Pilgrim Health Care
HMOs and public health agencies are natural partners in the development of community-based surveillance systems. Harvard Pilgrim Health Care, a mixed
model HMO with a membership of approximately 1.4 million people, has four ongoing collaborations with CDC, and two of these include the Massachusetts Department of Public Health. The first three partnerships involve antibiotic use, sexually transmitted disease surveillance, and tuberculosis surveillance, respectively. The fourth partnership, described below, focuses on the epidemiology of nosocomial infections. Its goal is to create a population-based, integrated program that addresses the major problems of these infections in whole populations.
TABLE 4-1 New Problems and New Solutions
|
Problems |
Solutions |
|
• Increased risk because of involvement of acutely ill patients and riskier interventions • Examples of factors that result in increased risk include: placement • More antibiotic-resistant pathogens • Receipt of care outside of acute-care hospitals • Resource constraints |
• Information systems provide better data more efficiently in an intensive care unit, immunosuppression, receipt of implanted prosthetic material, and extensive surgery • Integrated delivery systems and HMOs have data on delivery of care and outcomes in multiple locations |
|
SOURCE: Table compiled from presentation by Richard Platt at workshop. |
|
Nosocomial infections are a growing concern because they involve more acutely ill patients and riskier interventions and are caused by a growing number of antibiotic-resistant pathogens. Moreover, they are increasingly likely to be initiated in the outpatient setting, which constrains control and prevention efforts. For example, many surgical operations are performed in an ambulatory-care setting, and a larger fraction of postoperative care for all patients is administered outside of acute care hospitals. In addition, resources available for traditional infection surveillance are increasingly constrained. At the same time, HMOs use information systems that provide better data more efficiently than was previously possible. Furthermore, integrated delivery systems contain data on the delivery and outcomes of care in several locations within the system, allowing analysis of care across the continuum (Table 4-1).
Recently, Harvard Pilgrim Health Care used the information from its staff model program to study surgical site infections (SSIs). Analysis of data from 5,572 surgical procedures showed that less than half a percent of these patients developed infections before discharge (Figure 4-1). About 84 percent of infections that occurred became patent after discharge from the hospital. However, most procedures involved no postoperative stay, and few of the patients who
developed infections returned to the hospital where the surgery was performed for treatment. Comparisons of different postdischarge surveillance systems, primarily surgeons' and patients' questionnaires, illustrate that both questionnaires missed most infections. More surprising, the majority of infections reported by surgeons were not due to postoperative SSIs. Infections were detected through automated HMO records, and these records detected 75 percent of infections with a predictive value of 50 percent. The HMO's record linkage capabilities, including records from the hospital, ambulatory-care data, and pharmacy dispensing data, substantially outperformed both of the measures (surgeons' and patients' questionnaires) described above. Automated HMO data records supplemented by a limited record review (6 percent of records) resulted in a sensitivity of detection of 90 percent of infections and a predictive value of 100 percent (Figure 4-2).
On the basis of these findings and under a cooperative agreement with CDC's Hospital Infections Program, Harvard Pilgrim Health Care is collaborating with 11 hospitals and the three largest HMOs in eastern Massachusetts to assess the broader applicability of these methods. All of these participants realize the need to serve the public's health. The members of these HMOs (a total of 2.8 million members) account for 90 percent of the managed care enrollees in the region, which amounts to 36 percent of the region's residents. Together, they perform 70 percent of the coronary bypass surgeries and 40 to 50 percent of the breast surgeries and cesarean deliveries, the procedures on which this effort focuses.
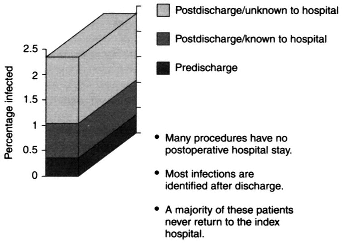
Figure 4-1 Limits of hospital-based SSI surveillance.
Data are for 5,572 procedures. Source: Sands et al., 1996.
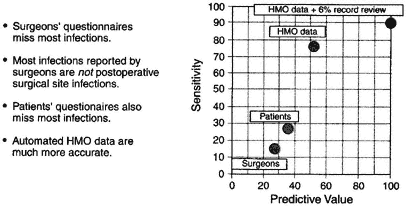
Figure 4-2 Postdischarge SSI surveillance. Source: Sands et al., 1996, 1999.
The goal of such collaboration is to identify SSIs by using automated inpatient and population-based ambulatory-care data, to characterize antibiotic-resistant pathogens, and to relate the rates of infection and antibiotic resistance to the appropriateness of preoperative antibiotic prophylaxis. A secondary goal is to assess and control other types of infection and, to the extent possible, extend this collaborative model to other types of problems. Although this approach was developed in a research-oriented HMO, it should be widely applicable in other settings and useful for a variety of other surveillance problems. Future activities under this agreement may include monitoring of other nosocomial infections, including neonatal infections and implanted central venous catheter infections.
Ultimately, these and similar collaborative efforts should serve as models for surveillance systems that work best when public health agencies and all of the participants involved in the care of individuals work together. These Harvard Pilgrim Health Care studies have proven that HMO data can help solve an existing problem—in this case, the inability to perform effective postdischarge surveillance for nosocomial infections—and can thereby reduce unnecessary efforts by hospital personnel.
LATTER DAY SAINTS HOSPITAL'S SYSTEM
Presented by John P. Burke, M.D.
Chief, Department of Clinical Epidemiology, Latter Day Saints Hospital, and Professor of Medicine, University of Utah School of Medicine
Experiences from the Latter Day Saints (LDS) Hospital in Salt Lake City, Utah, suggest that the best surveillance system operates in real time, providing
immediate responses that guide providers in performing interventions that are based on practice guidelines. The LDS Hospital has a highly advanced information system that maintains each patient's medication records and warns the pharmacy of potential drug interactions or allergic reactions. It also suggests medications for specific patients on the basis of prescription guidelines and current indicators such as white cell count, fever, and results of microbiological cultures and roentgenograms. In the case of emerging infections, the system also alerts the physician to infections caused by multiple-drug-resistant organisms or to a prescription that is at odds with what would normally be prescribed.
The goal of the system is flexible, informed decision making. The system includes bedside monitors that provide monographs on formulary antibiotics, as well as each nursing unit's history of nosocomial infections and the susceptibility patterns of the strains that caused those infections. An outpatient model provides guidance for the treatment of otitis media, pharyngitis, and urinary tract infections based on the cost of antimicrobial agents adjusted for patient-specific factors such as height, weight, and renal function. All of this is designed to make the system easy to use and thus promote its use; physicians, for example, find that it has greatly simplified the task of prescribing antibiotics.
Analysis of practice over the past 11 years at LDS Hospital indicates that the proportion of patients receiving antibiotics has increased from 32 to 54 percent, which may represent LDS Hospital's rising role as the main referral center for a 23-hospital system. However, the number of patients receiving antibiotics does not adequately reflect the antibiotic exposure of this population. Further analysis shows that the number of defined daily doses per 1,000 patient-days has decreased by 40 percent and that the cost of medication declined from $122 to $52 per antibiotic-treated patient over the same period. In addition, the incidence of adverse drug events due to antibiotics has declined by 60 to 70 percent, and there has been little change in the levels of antibiotic resistance. The research question being pursued is whether surveillance and monitoring have affected antibiotic resistance. The hope is that patient-specific use of information will stabilize antibiotic resistance and lead to a more sophisticated type of informed decision making, one that will control costs as well as prevent the emergence or reemergence of antibiotic resistance.
Laboratory-Based Reporting and Managing Encounter-Level Data
Presented by Richard Dixon, M.D., FACP
Medical Director, National Independent Practice Association Coalition
If surveillance for emerging and changing infection trends is to be effective, diagnoses must be accurate, reported in a standardized manner, and monitored. Sources of information on infections include voluntary reports of clinical diag-
noses by clinicians, reports of laboratory results, and diagnoses recorded in administrative data sets. Diagnoses recorded in administrative data sets include descriptions of diagnoses and procedures performed during individual patient encounters that are usually submitted to obtain payment for the services rendered. However, none of these sources is complete or accurate. Physicians are notoriously lax in reporting even those conditions required by law or regulation. Because administrative data sets primarily involve financial transactions, they commonly fail to capture critical features of illnesses. Fortunately, not every infection needs to be reported; common infections require that only a small but consistent portion of cases be recognized and reported for surveillance to be effective. On the other hand, conditions that occur infrequently require highly efficient capture of diagnoses if changes in their patterns are to be recognized. For each monitored condition, there is a threshold of accuracy and completeness of reporting. Surveillance and monitoring will fail if that threshold is not met.
The efficiency of infectious disease surveillance will likely be affected by the dramatic changes that are occurring in the U.S. health care system. Efforts both to reduce costs and to stay within predetermined budgets are having the greatest effects. Care that was formerly provided within or by hospitals and other institutions is being shifted to ambulatory-care sites where administrative data are of lower quality. Patients with complex cases of disease, who were formerly treated in university and other referral centers, are increasingly being treated in community-based facilities where laboratories are likely to have fewer resources and lower capacities. Visits to highly trained specialists are being discouraged. Empirical treatment based on symptoms is being promoted to reduce the use of costly diagnostic procedures, especially when treatments can be prescribed without precise diagnoses, such as the use of broad-spectrum antimicrobial agents to cover a broad range of potential pathogens.
These changes are often blamed on managed care. It must be recognized, however, that these changes are likely to occur under any model of care delivery in which cost containment is emphasized and that they do not simply reflect the policies of managed care organizations. In fact, many of the gaps in diagnostic information antedated managed care. Since administrative data have mainly been used to reimburse clinicians and hospitals, there has been no incentive to report observations unrelated to reimbursements. In the past decade, hospitals have been forced to document the accuracies of their submitted claims, but the focus has been on the detection of excessive or fraudulent billings, not on the identification of underreported diseases.
Even less attention has been paid to the accuracy or completeness of ambulatory-care data other than to uncover fraud or other inappropriate billing practices. An extensive effort is being conducted in California to improve the completeness, accuracy, and availability of clinical information so that outcomes and accountability can be better assessed. This involves the California Information
Exchange (CALINX) project, a joint effort of California's purchasers (led by the Pacific Business Group on Health), hospitals and provider organizations (led by the National Independent Practice Association Coalition), and health plans (led by the California Association of Health Plans).* As part of CALINX, the Completeness and Accuracy of Managed-Care Administrative Data Sets (CAMAS) project is being conducted. The CAMAS project will assess the accuracy and completeness of the information reported to managed care organizations by physicians under contract to those organizations. It is suspected that only a small portion of important information about clinical encounters is actually ever reported to HMOs, and if those suspicions were true, it would be unrealistic to expect HMOs to provide data that they themselves do not have.
In theory, managed care organizations should place more value on the data used to make diagnoses. A principal tool used to reduce costs is effective management of care and the provision of the right treatment, at the right time, to the right people. Thus, better diagnostic information should reduce the use of expensive treatments or at least permit more focused and, often, less expensive therapy. Moreover, the National Committee for Quality Assurance (NCQA) is placing a greater emphasis on outcomes in making accreditation decisions about managed care organizations and will likely begin to accredit provider organizations. Both of these NCQA initiatives should improve the quality of diagnoses on which surveillance is based. The Health Insurance Portability and Accountability Act of 1996 requires the federal government to set data standards, which will affect both laboratory and clinical data. Some believe, however, that the absence of standards is not the major impediment to obtaining good public health data. Increasing concerns about privacy are an important obstacle. An even greater obstacle may be the absence of a ''business case" for reporting surveillance data. For example, to report more complete and accurate diagnostic codes, providers will need improved information systems. They also need to change their patterns of work to improve the amount and precision of information collected and reported. These changes are costly, but providers do not believe that they will be rewarded for their investments in terms of increased revenues, improved efficiency, or a better ability to manage patients.
Effective surveillance will entail more than regulations requiring reporting of recognized infections and more than standards for the ways in which those infections are diagnosed and reported. It will require good data to populate those reports. Better information systems are important, but the greatest need entails substantial changes in provider incentives to work up cases of disease and report on them. At present, those incentives call for doing less; the incentives should reward better diagnoses and reports.
|
* |
Additional information about CALINX may be obtained from its web site: www.calinx.org. |
LABORATORY-BASED REPORTING ISSUES
Presented by Robert Rubin, M.D.
President and Chief Operating Officer, The Lewin Group
There may be a structural barrier between infectious disease surveillance and managed care organizations. In theory, private-sector laboratories report unusual infections and forward the isolates responsible for those infections to public health officials, who then verify the diagnosis and perform special studies of esoteric or low-volume infections. A study conducted by the Lewin Group, public health laboratories, and Health System Change on behalf of the U.S. Department of Health and Human Services evaluated public health laboratories in light of the changes in the health care system. Survey results from that study counter the commonly held belief that managed care has worsened the infrastructure of public health and microbiology laboratories. Only 43 percent of public health laboratory directors agreed with this widely held opinion, whereas 47 percent thought that managed care has no impact on public health efforts and 10 percent pointed to positive effects from managed care.
Under managed care, however, there is a tendency to treat patients empirically on the basis of symptoms rather than to perform laboratory tests and use the results of those tests as a basis for specific diagnoses. The emphasis on cost-efficiency may also be a disincentive to submission of reports and isolates. In addition, many HMOs have comprehensive contracts with large, national laboratories, with the Laboratory Corporation of America, Quest Diagnostics, and SmithKline Beecham Clinical Laboratories controlling 43 percent of the clinical testing in the United States (Sachs, 1997). Because of the interstate nature of their business, those laboratory personnel may not always be aware of state and local reporting requirements.
Several initiatives are under way to address these issues. The state of Washington's public health laboratory has entered into an agreement with Group Health Cooperative of Puget Sound to create the Clinical Laboratory Advisory Council to improve the laboratory delivery system and to open communications with all stakeholders. Representatives from public and private laboratories work through advisory groups that develop recommendations on a range of laboratory issues, from integration to practice guidelines to licensure. CDC has funded a few inventive pilot projects, including a study of the transmission of laboratory test results between managed care and the state public health laboratory in Washington State.
Public health laboratories, however, have not made the best use of the potential advantages of working with managed care. With limited resources being the obvious deterrent, many public health laboratories view managed care as a potential competitor. In addition, surveillance and monitoring issues, as well as the general topic of emerging infections, are seldom on the table when states seek to contract out services for their Medicaid populations. This may be shortsighted,
because good-quality surveillance and care may reduce long-term costs for all stakeholders. Such efforts at dialogue may facilitate improvements in public health laboratories' attempts to preserve vital elements of their infrastructures.
LEGAL CONCERNS
Presented by René Bowser, J.D.
Fellow, Georgetown University Law Center
Federal and state government responsibility for public health is certainly a necessity but is not a sufficient condition for maximization of public health. Indeed, unilateral actions by governments in fully promoting and protecting the health of the community are limited. For example, governments are not nearly as closely associated with individuals and their health care as managed care organizations are. From this perspective, managed care is better positioned to observe personal choices, obtain information on rates of illness and injury from specific causes, and identify health risks in the community. Furthermore, these organizations generally possess greater expertise in providing cost-effective personal medical services and clinical preventive services than government and are likely to be more flexible in their approaches to solving public health problems. Yet, despite the advantages that managed care organizations bring to public health, few incentives exist for them to provide communitywide public health services.
A deliberative effort should be made to secure managed care's cooperation in expanding public health activities, including surveillance and monitoring. This can be accomplished through well-considered delegations of public health activities, incentives, and regulations. Most local health departments lack the personnel, laboratories, and information systems need to identify and effectively respond to the great variety of health risks facing populations. At the same time, many managed care organizations are developing the capacity to identify clusters of diseases and injuries within the enrolled population and, through investigation, determine their causes. This information is critical for public health policy development and community health assessment. Ideally, surveillance should be a coordinated process in which the health department sets the agenda for surveillance, epidemiological, and statistical studies; contracts with a managed care plan to provide the information; uses the findings to assess health risks; and allocates resources to those events that pose the greatest risks.
Contracting with managed care organizations to identify clusters of diseases in the enrolled population is a desirable strategy. Not only does it identify health risks in the community, but it also provides the managed care plan with information that could serve as the basis for its own prevention strategies to avoid future treatment costs. In addition, managed care can assist in the early detection and control of emerging microbial threats through data collection and analysis. If managed care plans routinely reported the community's health status to offi-
cials at public health departments, such disease clusters could be reported before a widespread epidemic occurred. Even with the onset of an epidemic, public health officials can take advantage of managed care's cost-effectiveness by contracting with a health plan for the clinical testing and treatment of large numbers of individuals at risk of particular infections.
There are no insurmountable legal obstacles to these activities. Delegation of public health activities raises a constitutional issue only when government power is exercised by a private entity that deprives a person of life, liberty, or power under the behest of the state. Data collection, analysis, reporting, and specimen testing are constitutionally permissible since no significant liberty or property rights are violated. In contrast, monitoring and prevention activities such as contact tracing, mandatory testing, and compulsory quarantine and isolation are constitutionally suspect when performed by the private sector, and therefore, it is important that public health departments retain these functions.
SUMMARY OF CHALLENGES AND OPPORTUNITIES
Jonathan R. Davis, Ph.D., Editor
Disease surveillance and monitoring activities are a cornerstone of public health practice. These activities provide information crucial not only for assessment of the status of public health, monitoring of health trends, and recommendation of priorities but also for evaluation of the effectiveness of public health programs. A partnership between public health surveillance activities and managed care systems has many potential benefits since a primary goal of both is the health of a population. However, as identified in the workshop discussions, several impediments must be overcome before surveillance through managed care can be conducted.
Understanding Professional Roles in Surveillance and Monitoring
Relatively few health care providers fully understand the importance of public health surveillance, the role of the provider as a source of data, and the role of public health departments in responding to infectious diseases. The lack of understanding of the health department's role in surveillance may have originated either from the absence of feedback from public health agencies indicating how data are used or from a lack of availability of the data to the public, providers, or potential users of those data. Workshop participants asserted that managed care systems must provide an opportunity for public health agencies not only to explain their roles in surveillance and response but also to establish partnerships as a way of ensuring bilateral communication on cases of infectious diseases and opportunities for preventing the spread of infectious diseases.
Health care providers need to understand their role as an important source of data in infectious disease control and the role of the health department in responding to an infectious disease. Managed care systems provide an opportunity for public health departments to explain their role in surveillance for infectious diseases and their responses to infectious diseases. Managed care organizations also have the opportunity to establish partnerships with public health agencies to prevent the spread of infectious diseases.
Ensuring Availability of Data
The possibility that fewer diagnostic tests will be performed in managed care systems as a result of cost-reduction requirements could lead to a decrease in the amount of information on emerging infectious diseases needed by public health officials. The trend in managed care toward chronic disease management rather than infectious disease control may hamper surveillance efforts as less value is placed on emerging infections. This concern deserves greater attention and more data are required to assess the impact of managed care on infectious disease control.
Pilot studies involving managed care organizations and the public health system could determine the quality, accuracy, and availability of data on infectious diseases. Partnerships between public health laboratories and managed care organizations could ensure that the necessary testing is completed and not neglected. Standards for a computer-based patient record will ensure the usefulness of patient records for population-based health.
Promoting Sharing of Data
Certain misperceptions and obstacles must be addressed if pilot studies are to go forward. Public health agencies are concerned that some managed care organizations treat patient data in a proprietary way and are thus unwilling to share those data, which may subsequently impede surveillance efforts. Fears about violation of confidentiality can prohibit sharing of electronic medical data, with the potential for hampering surveillance efforts. Data sharing between public health agencies and managed care systems should be encouraged but should also be attentive to protection of patient confidentiality. To accomplish this, managed care organizations need to be reassured that public health surveillance is not a regulatory function but rather a mechanism for monitoring the status of the public's health and identifying opportunities for improving community health status.
Even when sharing of data is accomplished, the incompatibility of computer networks among managed care organizations makes the sharing of information difficult, resulting in a lack of timely, accurate, and complete information on health care encounters. Compatible model systems should be developed so that
managed care systems' computers can effectively and quickly communicate locally as well as nationwide for assessment of disease threats. In addition, collaborative information systems such as population-based integrated programs could survey and monitor nosocomial infections. To avoid duplication of effort and maximize efficiency, a workshop speaker proposed that public health agencies need to work with managed care organizations to ensure that direct reporting is conducted from their laboratory data systems to public health agencies for surveillance purposes.
Tracking Nosocomial Infections
The increasing use of ambulatory-care settings for surgical operations and postoperative care may hinder the ability to adequately track and monitor nosocomial infections because of the decreased disease reporting capabilities of such settings. The lack of reporting ability may constrain the ability to swiftly disseminate information, which is critical, for example, to decrease the incidence of adverse events from antibiotics and, therefore, for surveillance and control efforts.
Collaborative information systems, such as those used by hospitals and HMOs, can provide a means of addressing the problems associated with surveillance and monitoring of nosocomial infections. Integrated population-based programs can address such infections in the context of changes in health care delivery. For example, although all hospitals and HMOs now have information systems that provide better data more efficiently than was previously possible, hospital integrated delivery systems and HMOs have data on the type of care delivered and outcomes at the various locations within their systems, thereby allowing analyses of care across the continuum. A collaborative effort between hospitals and HMOs can therefore help identify and address nosocomial infections. To maximize surveillance and monitoring efforts, however, public health agencies and other health care professionals involved in the care of individuals need to undertake similar collaborative efforts.
In addition to concerns regarding decreased reporting of nosocomial infections because of substantial resource constraints and because ambulatory-care settings are increasingly being used for surgery and care, workshop participants expressed apprehension regarding the timeliness of reporting of such infections as well as antibiotic resistance. Workshop participants indicated that the ability to disseminate information swiftly is critical to surveillance and control efforts. For instance, not only are postoperative surgical wound infections very costly, but they are also the most frequent type of hospital-acquired infections. Besides decreasing the incidence of such infections, a real-time surveillance mechanism may also serve as a means for decreasing the incidence of adverse drug events from the use of antibiotics.
Real-time surveillance capabilities could monitor adverse drug events and nosocomial infections, which would lead to immediate applications and interventions based on clinical practice guidelines. Artificial intelligence techniques can
also be used to detect markers that could then help identify specialist groups that are needed for patient consultations and help prevent hospital-acquired infections.
Already, bedside computers are providing monographs for formulary antibiotics in 1- and 5-year antibiograms that determine the susceptibility patterns of isolates that cause hospital-acquired infections. Computerization of patients' medication histories can provide physicians with the means to check for previous allergies. In addition, the data available online would remain accessible and would prevent pharmacists from accidentally dispensing medications that could result in allergic reactions. Surveillance efforts could be further enhanced by such methods by identifying patients who are infected or colonized with multi-drug-resistant organisms or patients whose antibiotic prescriptions do not coincide with prescriptions that would otherwise be recommended. An approach that emphasizes patient-specific information can stabilize antibiotic resistance patterns and can lead to a more sophisticated type of informed decision making that would be amenable not only to reducing costs but also to preventing the further emergence of antimicrobial resistance.
Accurate Reporting of Encounter-Level Data
Effective surveillance for emerging infections and varying infection trends requires reporting of an accurate diagnosis in a standardized manner and requires that the trends be monitored on a continuous basis. Although fairly common infections require only that a small but consistent portion of cases be recognized and reported for effective surveillance, conditions that occur infrequently require highly efficient capture of a diagnosis so that changes in their patterns can be recognized. Workshop participants expressed concern that sources of information for encounter-level data are typically incomplete or inaccurate. Hospitals as well as ambulatory-care settings have not traditionally focused on the identification of gaps in diagnostic information. Because administrative data have primarily been used to reimburse clinicians and hospitals, there has been no incentive to report observations unrelated to reimbursement; rather, the focus has been on the detection of excessive or fraudulent billings and not on assessment of the accuracy or completeness of data.
Effective surveillance will require more than just the implementation of good regulations for the reporting of recognized infections and the use of standards for the diagnosis of those infections and reporting on those infections. It will also require good encounter-level data for those reports. Although better information systems are important, substantial changes in provider incentives are needed to encourage clinicians to diagnose diseases and actually report those diagnoses. Those incentives are set up to generate the capture and transmission of fewer data. The system needs to be revamped so that new incentives are implemented to reward better diagnoses and reporting. Additionally, efforts need to be initiated to teach individuals how to perform coding correctly so that accurate diagnoses can be reported. By improving the quality of the diagnoses on which
surveillance is based, better diagnostic information should also reduce the use of expensive treatments as well as allow the use of more focused and less expensive therapy. Such care management can maximize the efforts to provide the right treatment to the right people at the right time and lead the way for an effective disease management program.
Overcoming Structural Barriers
Workshop participants expressed concern that there is a fundamental structural barrier between surveillance and practice in managed care. They felt that managed care has negatively affected public health laboratories and the microbiology laboratory infrastructure for a number of reasons, some of which are listed here. First, because managed care focuses on efficiency, there is generally a disincentive for disease reporting and for submission of isolates recovered from patients with emerging diseases. Second, many managed care systems typically have comprehensive contracts with large national laboratories; for instance, three laboratories (Laboratory Corporation of America, Quest Diagnostics, and SmithKline Beecham Clinical Laboratories) control 43 percent of the clinical testing in the United States today. Because of the interstate nature of this type of business, laboratory personnel may not always be aware of the state and local reporting requirements, which may exacerbate the problems of identification and monitoring of diseases. Third, there is a tendency for managed care organizations to provide empirical treatment, which results in reporting problems. Fourth, public-sector laboratory information systems have lagged behind private-sector systems in leveraging potential advantages for managed care. Finally, as people are placed into Medicaid managed care environments, discussions and contract terms generally do not contain provider expectations about surveillance monitoring and their effects on emerging infections.
Workshop participants identified a number of initiatives that addressed the barriers between surveillance and managed care practices. These opportunities are identified below, but they are by no means all inclusive. With integrated patient databases, there is a potential for seamless communication between laboratories, managed care organizations, and public health officials. Such communication could help dissolve the structural barriers between surveillance and practice in managed care organizations and subsequently facilitate the exchange of information.
Additionally, the large national laboratories that are used extensively by managed care systems need to implement better internal guidance policies to help identify which states require the reporting of which infectious diseases. Workshop discussions suggested the need to establish incentives for disease reporting and for isolate submission within managed care organization's policies and regulations; the lack of such incentives jeopardizes surveillance and monitoring efforts. Treatment based on scientific observations rather than on empirical knowledge must also be emphasized as a rule of thumb, especially in ambu-
latory-care settings. Furthermore, because public health laboratories have not leveraged the potential advantages of working with managed care organizations, states need to increase the available resources in their respective public health laboratories to maximize the potential advantages for managed care systems. Finally, when states seek to contract out services for their Medicaid populations, such contracts must incorporate terminology that will take into account the need for surveillance and monitoring as well as emerging infection issues.
Summary
There is concern that managed care may have a negative effect on infectious disease surveillance and monitoring. For example, a feature of managed care is to shift patient care from inpatient to outpatient settings, where there are reduced diagnostic and patient management capabilities. Another concern is that managed care relies heavily on empirical diagnoses rather than laboratory data, which may further reduce disease reporting as well as the forwarding of pathogens for detailed analysis and epidemiological investigation. Consequently, public health laboratory practices may have to adjust their role in the overall health care system because fewer tests are available to identify new pathogens and their susceptibilities to therapeutic agents. This is coupled with fact that testing is being conducted primarily by three large commercial laboratories that frequently are not in the same state as the contracting HMO. Thus, it seems unlikely that the next outbreak will be detected through a single HMO, making the outbreak more difficult to track.
Given that the current system of managed care is evolving rapidly and that the level of heterogeneity is increasing among HMOs, there is concern that the extensive patient databases maintained by HMOs may remain uncoordinated and poorly networked. Such an outlook may complicate the present tasks of infectious disease surveillance and monitoring and may make it difficult to plan for future surveillance systems. Models of sophisticated, integrated data systems at the local level and of coordinated action to standardize databases and reporting at the regional level may be extended to other levels and specific diseases. Action at the state level may also be needed to improve the ability of the public health system to handle the information and samples provided by HMOs. There appears to be no legal impediment to privatizing or contracting out at least some of the functions of this public health surveillance system. These issues could be examined in a future workshop to examine the capacities of public health systems to respond to emerging infections.



















