6
Gene Transfer As a Biomedical Tool
Perhaps the most exciting developments in molecular biology have to do with the explosion of information and technology for dealing with biological processes at the level of the genome. A number of workshop presenters have made important progress in this area. At times during the presentations, some possibilities were suggested that would once have been in the domain of science fiction. Before delving into the data-intensive challenges that are of most relevance to mathematical scientists, we discuss two workshop presentations that set the stage.
Eduardo Marbán, of Johns Hopkins University, presented some basic background on the challenges of molecular biology, explaining, for instance, how proteins control all that we do and how DNA controls the proteins. The DNA in the human genome, if unraveled, would be about 6 feet long, and if encoded into phone books would be as high as the Washington Monument. Of this, approximately 99 percent does not code for proteins (although some of it may have other functions), while the remaining 1 percent, which we call genes, contains the blueprints for protein formation.
Genes are sometimes referred to as “wild type” (the predominant forms found in nature) or “mutant” (aberrant forms found in nature or genetically engineered). Genetic engineering or genetic therapy delivers a desired piece of genetic material by first being packaged in a modified virus (viruses being able to insert themselves into cell nuclei; the modification is whatever is necessary to make the virus benign) and injecting them into a subject. These modified viruses (common cold viruses, reengineered AIDS viruses) are specialized for gene transfer and can be very efficient. Genes can then be introduced isogenically (within the same species) or allogenically (from one species to another).
The potential use of gene transfer is unlimited. Some examples are the following:
-
To test hypotheses in biological systems;
-
To treat single-gene deficiency disorders such as ADA deficiency (which results in people without viable immune systems) or hemophilia;
-
To treat a complex disease system, such as that promoting angiogenesis as a treatment for coronary artery disease; and
-
To reengineer cells and tissue at a functional level.
Marbán described a case study in which gene therapy was used to treat cardiac arrhythmia, in particular the problem of atrial fibrillation. (There are many kinds of arrhythmias, which account for more than 2 million deaths per year in the United States and result in the implanting of approximately 255,000 pacemakers per year at about $45,000 per operation.) Current therapies for arrhythmia include drug therapy (which has many side effects) or the implant of pacemakers or defibrillators (which require a lifetime commitment to repeated procedures). Other therapies also carry complications, so it is reasonable to look to gene therapy. Gene therapy avoids the use of implantable hardware, has the potential to treat targeted tissues, and can be reversed. There are still problems to overcome, however. For example, there can be difficulty in delivering the therapy to the appropriate tissue or cell types.
The choice for an initial gene therapy trial aimed at arrhythmia is the genetic modification of the atrial-ventricular (AV) node, a site on the heart that exerts significant control over the heart’s beating. The goal of the treatment is to control and slow the conduction through the AV node, thus decreasing the chance of fibrillation. The genetic approach to treatment is to insert an agent that blocks certain stimulations of the AV node. To test the effectiveness of the gene therapy, an experiment was carried out in swine. The results were successful: The gene therapy limited the pig’s heart rate during atrial fibrillation to 155 beats per minute, whereas the rate during atrial fibrillation for a control subject went up to 190. The role that mathematical scientists will play is in the bioinformatics that enables such gene therapies.
Michael Phelps, of the University of California at Los Angeles, spoke about using a minuscule positron emission tomography (PET) scanner, called a micro-PET scanner, to monitor the progress of a treatment that involves gene therapy. This is a very promising technique to advance the art of gene therapy, and mathematical scientists are needed to address the reconstruction of images from the micro-PET data. The micro-PET scanner provides a tissue concentration measurement, which can be transformed into a metabolic rate by a mathematical model.
Phelps’s lab has built a micro-PET scanner that is used in mice, rats, and monkeys (mice being the typical mammalian model). Their goal is in vivo gene expression measurement,1 and to accomplish that they constructed an RNA probe that is only 13 base pairs in length to carry out in vivo hybridization. The probe carries a PET reporter gene (a gene that the PET scanner can track) and a therapeutic gene (a gene that effects some treatment). The PET reporter gene produces a signal, detectable by a PET scanner, if gene expression occurs—that is, if the therapeutic gene expresses and delivers the therapy. To test the micro-PET technique, Phelps constructed sample probes and injected them into the livers of mice. The level of expression of the therapeutic gene, as detected through the micro-PET technique, was in excellent agreement with the actual level of associated RNA measured during subsequent autopsies.
Figure 6-1 shows the PET tracking of a therapeutic gene that expresses in the liver. Gene expression first increases, as is seen after 2 days, then decreases, and after 2 weeks the immune system terminates the virus. The promoter can be turned on and off with an external switch (tetracycline), as might be needed in some studies. Among other advances, this procedure should allow cancer cells to be tracked over time as they migrate.
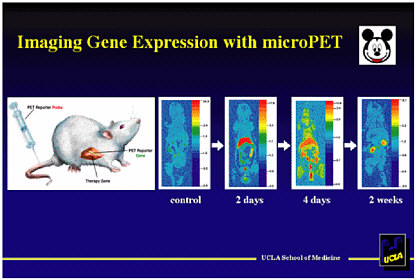
FIGURE 6-1 Imaging gene expression with micro-PET. Figure courtesy of Michael Phelps.
Phelps stressed the importance of going back and forth between humans and mice, because the science is enabled by both. The advantage of the current drug therapy/delivery system is that the drug can be given in truly tiny amounts, and the promoters can be turned on externally to increase the dose.
Several developments are needed on the mathematics/statistics front. At present, Fourier-based reconstruction is used to create images from the micro-PET data, but better methods are needed.



