Trimethyltin Encephalopathy
Robert G.Feldman, MD; Roberta F.White, PhD; Ikechukwu I.Eriator, MD
A chemistry student was acutely exposed to vapors of an organotin compound. Seventy-two hours later, he exhibited delirium, spatial disorientation, perseveration, inappropriate affect, and memory defects. Five months later, he experienced episodes of complex partial seizures, which continue to require anticonvulsant medication after 7 years. Trimethyltin was identified in blood and urine samples taken 17 days after the accident; the blood level of trimethyltin was elevated 35 days after exposure. Serial electroencephalograms showed persistent left temporal paroxysmal epileptogenic potentials. Serial neuropsychological tests revealed persistent memory defects, cognitive dysfunction, and dysphoria 4 years after exposure. We review acute, resolving, and long-term residual neurotoxic effects of trimethyltin in man. We describe detailed clinical observations, serial neuropsychological test results, electroencephalographic findings, and exposure data in this patient, confirming the limbic system effects of trimethyltin and relating them to the known histopathologic pattern of this condition. (Arch Neurol. 1993;50:1320–1324)
Toxicologic reports indicate involvement of the central nervous system by alkyltins. Triethyltin intoxication has been used as an experimental model of myelinopathies, neurodegenerative disorders, and cerebral edema, while trimethyltin has been used in the investigation of hippocampal lesions, kainic acid neurotoxicity, and minimal brain dysfunction and to elucidate the cytoarchitecture of the brain and the neuropathologic findings of limbic seizure activity.1,2
Although there are many reports from studies on laboratory animals on the neuropathologic and behavioral changes induced by trimethyltin,3 there have been few reports on such effects in humans4–7 and those that do exist are confounded by exposure to chemicals other than tin, poor exposure data, or incomplete clinical follow up (Table 1).
We describe the acute, resolving, and long-term effects of trimethyltin in a 23-year-old man exposed to this alkyltin through dermal and inhalational routes.
REPORT OF A CASE
A 23-year-old male graduate student, without a significant medical history, was trying to recrystallize an alkyltin compound, bis-trimethyl-stannyl-acetylene, from an ether-based solvent when it accidentally ignited. An explosion and fire resulted in burns (12% first-degree, 2% second-degree, and less than 1% third-degree burns) mainly on the left side of his head, neck, and chest. Although stunned, he had no loss of consciousness. Apart from a renal tubular acidosis (bicarbonate, 17 mmol/L) that resolved after vigorous fluid and electrolyte therapy, other features were essentially neurologic and neurobehavioral.
On admission to the hospital, he was oriented and demonstrated no obvious neurologic deficits. Three days later, he appeared to be confused and disoriented; he
Reprinted with permission from Archives of Neurology 50:1320–4, Copyright 1993, American Medical Society.
Table 1. Human Reports of Trimethyltin Intoxication
|
Source, y |
Clinical Features |
Tests |
Comments |
|
Foxtemps et al,4 1978; case reports (two chemists) |
Headaches, memory defects, pain, loss of vigilance, insomnia, anorexia, confusion, disorientation, seizures (clonic tonic) |
Electroencephalogram normal, theta waves (after secobarbital) |
No urinary, blood, or air monitor or neuropsychiatric test; confounded by dimethyltin or monochioromethane exposure for 3 mo |
|
Brown et al,5 1979; case report (chemist) |
Hyperactivity, insomnia, alternate hyperactivity; and absentmindedness |
None reported |
Not a full case report; duration of exposure not stated; full recovery |
|
Ross et al,6 1981 and 198320; epidemiologic study: compared 12 workers with high exposure with 10 with low exposure |
Alternatting attacks of rage and deep depression, forgetfulness, headaches, loss of Ilbido and motivation, sleep disturbance, disortentation, burns, fatigue, weakness, poor concentration, dim vision, stuttering attacks |
Urine tin levels 20 to 200 parts per billion (ppb); electroencaphalogram, no specific abnormalitites; slow nerve conduction velocity; impaired verbal memory, fine hand-eye coordination, visual motor integration, finger tap speed and learning; emotional disturbances |
Details of neuropsychiatric tests not stated; longest follow-up 2 y and 10 mo; confounded by dimethythand methylchloride; variable outcome on follow-up from personality changes to complete recovery |
|
Rey et al,7 1984; and Besser et al,9 1987; case reports (6 workers) |
Hearing loss, amnesia, disorientation, confabulation, confusion, restlessness, aggressiveness, hyperphagia, seizures, nystagmus, ataxia, neuropathy, blurred vision, disturbed sexual behavior, death |
Urine tin 445 to 1580 ppb (4–8 d after exposure); electroencaphalogram mostly normal; theta activity in fatal case; chest roentgenogram indicated respiratory distress syndrome in most severe cases; autopsy showed necrosis in limbic system and pontine and cerebellar structures |
Confounded by dimethyttin and methylchloride; urine fevels less than 20 ppb 2 mo after exposure; neuropsychiatric testing not reported |
|
Present case; a chemist; accidental severe single exposure |
Disorientation, incongruous affect, memory defects, abnormal cognitive process, complex partial seizures, depression, fatigue, insomnia, amotivation, and indifference |
Urine tin 52 ppb, 17 d after exposure; 10 ppb; 35 d after exposure (normal, <18); serum 13 ppb, 17 d after exposure; 7.4 ppb, 35 d after exposure (normal, < 3.3); electroencephalogram; left paroxysm; temporary theta; magnetic resonance imaging normal; detailed serial neuropsychological assessments over 4-y period |
Acute exposure urine level >52 ppb; neuropsychological testing revealed residual memory impairments; memory and mood complaints; seizures persistent |
could not find the bathroom or remember his visitors. He was disoriented to place and time. He mumbled repetitive statements to himself about the accident. Gait was normal. Tendon reflexes were 2+ equally. He was without pathologic reflexes, and the results of his sensory examination were normal. There was no papilledema. A computed axial tomographic scan was normal. An initial electroencephalogram showed 4- to 5-Hz paroxysmal theta waves over the left temporal area. Asymmetric 3- to 4-Hz delta waves developed on the left side with drowsiness or hyperventilation. Neuropsychological testing revealed significant impairment in new learning of verbal and visuospatial information on the Wechsler Memory Scale-Revised (WMS-R) and performance on the visual-motor and visuospatial subtests of the Wechsler Adult Intelligence Scale-Revised (WAIS-R) were only average. A performance IQ (PIQ) equal to 106 was well below the PIQ of 124 earned in a school assessment 2 years prior to the accident.
Repeated (partial) neuropsychological testing done 10 days after exposure showed some recovery of cognitive function. A PIQ was then identical to premorbid levels. Immediate recall of verbal and visuospatial information on the WMS-R improved, but his delayed recall continued to show forgetfulness (46% on the verbal task and 23% on the visuospatial task).
Urine and serum tin assays done on the 17th day after exposure showed 13 parts per billion (ppb) of trimethyltin (normal, <3.3 ppb). The trimethyltin concentration was still elevated (7.4 ppb) on the 35th day after exposure. The initial urinary trimethyltin level was 52 ppb (normal, <18 ppb), but this had fallen to 10 ppb by the 35th day after exposure (analyses by gas chromatographic mass spectrography, National Medical Services, Willow Grove, Pa).
His memory had not stabilized by the time he was discharged 18 days after exposure. He became lost in his hometown and his parents complained that he was “acting strange” and could not recall information presented to him.
A repeated electroencephalogram 3 months after exposure continued to show excessive scattered slow fre-
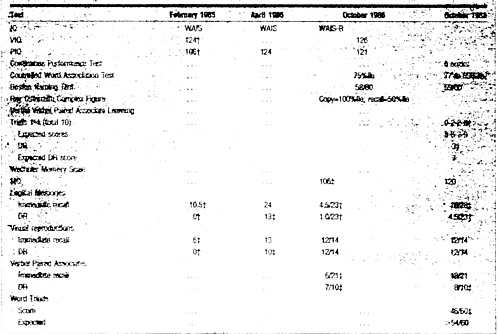
Table 2. Selected Neuropsychological Test Result*
|
 |
|
*IQ indicates intelligence quotient; PIQ, performance IQ; VIQ, very superior IQ; WAIS, Wechsler Adult Intelligence Scale; WAIS-R, WAIS-Revised; DR, delayed recall; and MQ, memory quotient. See White et al36 and White and Proctor37 for a description of the test battery. †Moderately severely impaired for IQ. ‡Mildly impaired for IQ. |
quencies bilaterally, with persistent theta instability over the left hemisphere. When he tried to return to school 4 months after exposure, he could not recall details of his thesis and was unable to continue his work. Five months after exposure, he experienced interrupted awareness on two occasions. His friends described the second episode thus: “his eyes bulged out, he began to shake all over, and he nearly fell out of the chair.”
When seen at 19 months after exposure, he complained of recurrent blackouts of both mild and severe types; the latter was preceded by an aura of visual disturbance, sense of an unpleasant smell, feeling of familiarity, and intense sweating. He also complained of difficulties with sleep, concentration, and recall. Neurologic examination revealed no motor or sensory deficits. Brainstem auditory evoked response, somatosensory evoked potentials, and visual evoked potentials continued to be normal as they had been on discharge. Repeated EEGs continued to show intermittent paroxysmal theta slowing in the left temporal area.
Neuropsychological assessment (including WAIS-R, Ravens Advanced Progressive Matrices, Repeated Motor Programs, Trail Making Test, Wisconsin Card Sorting Test, Hand Grip Strength, Finger Tapping, Grooved Pegboard, Boston Naming Test, Controlled Word List Generation, Writing Sample, Boston Visuospatial Quantitative Battery Constructions, Rey-Osterreith Complex Figure, WMS [Revised], California Verbal Learning Test, Milner Faces, Albert’s Famous Faces, Minnesota Multiphasic Personality Inventory, and Profile of Mood States) at this time revealed very superior IQ scores (Table 2). He performed below expectation on a coding task (WAIS-R Digit Symbol), but most of the other attention, language, and spatial tasks were performed at superior levels. However, memory quotient on the Wechsler Memory Scale (WMS) remained below expectation at 106. Learning of narrative information was well below expectation, with forgetting on delay. Multiple-choice testing of recognition memory was also impaired on this task. He was also impaired in IQ for learning WMS verbal paired associates and lost 2/9 (22%) on delayed recall; learning and recall were well below expectation for IQ on the California Verbal Learning Test (12/16 on Trial 5). Memory for visual designs was within expected limits on immediate and delayed recall. Depression and helplessness were noted on behavioral/ personality inventories.
NEUROPSYCHOLOGICAL ASSESSMENT 43 months after exposure (Table 2) revealed an improved score on the omnibus memory test (WMS Memory Quotient=120). However, he continued to evidence mild memory deficits. Forgetting of verbal (but not visuospatial) information was seen on WMS verbal memory tests, California Verbal Learning Test, and difficult paired associate learning. Performance on a memory test requiring the patient to learn new information despite interference from a distraction task (Peterson-Peterson Word Triads task) was below expectation for his IQ. He also reported symptoms consistent with depressive affect on the Minnesota Multiphasic Personality Inventory. Overall, neuropsychological test findings were considered to be consistent with dysfunction localized to the limbic system, particularly the left mesial temporal (hippocampal) area.
Magnetic resonance imaging of the brain at this time was normal. The electroencephalogram continued to show left temporal theta activity with rare sharp wave and rhythmic discharges.
COMMENT
Trialkyl forms of tin have been associated with severe acute neuropsychologic manifestations in clinical and environmental settings. Of 270 known cases of poisoning following the contamination of an antibiotic by triethyltin, only 10 were said to have recovered completely. Over 100 victims died, while many were left permanently disabled.8 An acute limbic-cerebellar syndrome was reported in six industrial workers who cleaned a tank containing trimethyltin. One died, while two were left permanently disabled.7,9 Two chemists were exposed in a poorly ventilated laboratory and they developed seizures as well as memory and confusional disorders.4 In another report,6 when 22 workers were exposed in a poorly ventilated situation, the four individuals followed up had long-term changes in personality, probably due to irreversible central nervous system changes.
The trimethyltin syndrome has been produced in rats and consists of tremor, hyperactivity, aggression, self-mutilation, and spontaneous seizures. This constellation of behavioral changes probably is attributable to limbic system pathology.10 Higher doses may be associated with perseverations. A consistent observation has been impaired retention in inhibitory avoidance paradigms and long-lasting impairments in acquisition and retention of both food- and shock-motivated responses.3
Histopathologic studies in laboratory animals have shown a preferential vulnerability of the hippocampus. Early pathologic changes have been noted in the pyramidal cells of areas CA2 and CA3 of the hippocampus as well as in the granule cells of the dentate gyrus.5,11,12 Damage also occurs in the amygdala, neocortex, pyriform cortex, entorhinal cortex, and olfactory bulbs. Damaged areas in the forebrain are those associated with learning and memory impairment in animals or dementia and amnestic syndromes in humans.
Our patient manifested overt abnormal neurobehavior 3 days after the accident. This “latent period” has been noted in animal studies10,12,13 as well as in previous human exposure.7,9 A peak brain tin concentration on about the fourth day has been observed in animal experiments,13 though total brain tin levels in such animals may not necessarily correlate with the degree of central nervous system toxicity.14 It is likely that trimethyltin was responsible for this encephalopathy in view of the fact that other organotin compounds undergo biotransformation by dealkylation.15 Trimethyltin is a stable metabolite,16 and trimethyltin was the only form of organotin found in the patient’s blood and urine assays.
Increased urinary levels of trimethyltin are consistent with significant body content of trimethyltin. Review of data from a previous report9 indicates that the maximum urinary levels occur 4 to 10 days after exposure. This patient may, therefore, have had a significantly higher content in the first few days after exposure than that measured in the first urine sample taken 17 days after exposure. The serum level was still elevated 35 days after exposure. This may have been due to high body content and, possibly, to binding of trimethyltin to erythrocytes,2 though the latter has not been clearly documented in primates or man.17 In fact, the lower trimethyltin-hemoglobin binding capacity in mice and humans compared with rats may explain the increased sensitivity of mice and humans to the effects of trimethyltin.18
This case is similar to the other reported human cases in manifesting with memory defects, confusion, incongruous affect, disorientation, seizures, depression, insomnia, poor vigilance, and slow and paroxysmal electroencephalographic records in keeping with a toxic encephalopathy. Perseverations, though observed in animal studies,19 have not been previously documented in humans and were seen rarely in this patient. Hyperactivity, aggression, or alternating attacks of rage and deep depression reported in other human subjects were also not present in this patient.20–22
Classic evidence of focal cerebral dysfunction, with impairments in new learning such as those seen in mesial temporal lobe damage, is present in this patient.23 Interestingly, the memory findings were in the verbal realm and, therefore, suggestive of lateralized left temporal dysfunction. Likewise, his dysphoric mood is typical of mood changes associated with left hemisphere damage.24–27 The lateralization of mood and memory changes to the left cerebral hemisphere is consistent with both the patient’s EEG findings and findings previously reported in patients with left temporal foci excision and seizures not attributable to toxic exposures.28
The only available human autopsy report described a man who died of pulmonary effects secondary to high ex-
posure.7,9 Morphologic alterations in the brain were most prominent in the amygdala, temporal cortex, basal ganglia, and pontine nuclei. Swollen perikarya, loss of Nissl substance, and necrosis of nerve cells were seen. Electron microscopy showed zebra bodies and many vacuoles. Cerebral edema with irreversible cell damage also occurred in the amygdala. The cerebellar cortex showed loss of Purkinje cells.
The exact mechanism of neurotoxic action of trimethyltin remains uncertain. Proposed mechanisms include release of endogenous excitotoxins from the heavy metal-containing pathways of the hippocampus,29 elevated extracellular glutamate levels,30 hyperammonemia,31 decreased γ-aminobutyric acid concentration,32 inhibition of Ca2+-ATPase in a concentration-dependent manner thereby interfering with calcium pump and other cAMP (adenosine 3′:5′-cyclic phosphate)-mediated processes in the brain,33 or a reduction in the hippocampal zinc concentration, possibly leading to mossy fiber disinhibition and subsequent hyperexcitation of the hippocampal electrical circuitry.3,34
Trimethyltin is an intermediate by-product in the production of other more commonly used tin products.3 It, thus, still constitutes an occupational hazard for some groups, especially chemists who often seem unaware of the inherent dangers.35 This article documents the acute and long-term neurotoxic effects of exposure in a young man. The detailed serial electroencephalographic and neuropsychological studies provide a basis for estimating the prognosis in such cases.1
Accepted for publication August 26, 1992.
Reprint requests to Department of Neurology, Boston University School of Medicine, 80 E Concord St, P801A, Boston, MA 02118–2394 (Dr Feldman).
REFERENCE
1. Reiter L, Ruppert P. Behavioral toxicity of trialkyltin compounds: a review. Neurotoxicology. 1984;5:177–186.
2. Hasan Z, Zimmer L, Woolley D. Time course of the effects of TMT on limbic evoked potentials and distribution of tin in blood and brain in the rat. Neurotoxicology. 1984;5:217–244.
3. Walsh T, DeHaven D. Neurotoxicity of the alkyltins. In: Bondy S, Prasad K, eds. Metal Neurotoxicity. Boca Raton, Fla: CRC Press Inc; 1988:87–107.
4. Fortemps E, Amand G, Bomboir A, et al. Trimethyltin poisoning: report of two cases. Int Arch Occup Environ Health. 1978;41:1–6.
5. Brown AW, Aldridge WN, Street BW, Verschoyle RD. The behavioral and neuropathologic sequalae of intoxication by TMT compounds in the rat. Am J Pathol. 1979;97:59–82.
6. Ross WD, Emmett EA, Steiner J, Tureen R. Neurotoxic effects of occupational exposure to organotins. Am J Psychiatry. 1981;138:1092–1095.
7. Rey C, Reinecke H, Besser R. Methyltin intoxication in six men: toxicologic and clinical aspects. Vet Hum Toxicol. 1984;26:121–122.
8. Boyer IJ. Toxicity of dibutyltin, TBT, and other organotin compounds to humans and to experimental animals. Toxicology. 1989;55:252–298.
9. Besser R, Kramer G, Thumler R, Bohl J, Gutmann L. Hopf HC. Acute TMT limbic-cerebellar syndrome. Neurology. 1987;37:945–950.
10. Dyer RS, Walsh TJ, Wonderlin WF, Bercegeay M. The TMT syndrome in rats. Neurobehav Toxicol Teratol. 1982;14:127–133.
11. Chang LW. Neuropathology of trimethyltin: a proposed pathogenic mechanism. Fund Appl Toxicol. 1986;6:217–232.
12. Whitington DL, Woodruff ML, Baisden RH. The time course of TMT-induced fiber and terminal degeneration in hippocampus. Neurotoxicol Teratol. 1989; 11:21–33.
13. Ruppert PH, Walsh TJ, Reiter LW, Dyer RS. TMT induced hyperactivity: time course and pattern. Neurobehav Toxicol Teratol. 1982;4:135–139.
14. Mushak P, Krigman M, Mailman R. Comparative organotin toxicity in the developing rat: somatic and morphological changes and relationship to accumulation of total tin. Neurobehav Toxicol Teratol. 1982;4:209–215.
15. Magos L.Tin. In: Friberg L, Nordberg G, Vouk V, ed. Handbook on the Toxicology of Metals. New York, NY: Elsevier Science Publishing Co Inc; 1986;2: 568–593.
16. Cremer JE. The biochemistry of organotin compounds: the conversion of tetraethyltin into triethyltin in mammals. Biochem J. 1958;68:685–691.
17. Krigman MR, Silverman AP. General toxicology of tin and its organic compounds. Neurotoxicology. 1984;5:129–140.
18. Rose M, Aldridge W. The interaction of triethyltin with components of animal tissues. Biochem J. 1968;106:821–828.
19. Swartzwelder HS, Helpler J, Holahan W, et al. Impaired maze performance in the rat caused by TMT treatment problem solving deficits and perseveration. Neurobehav Toxicol Teratol. 1982;4:169–176.
20. Ross WD, Sholiton MC. Specificity of psychiatric manifestations in relation to neurotoxic chemicals. Acta Psychiatry Scand. 1983;67(suppl 303):100–104.
21. Stoner HB, Barnes JM, Duff JI. Studies on the toxicity of alkyltin compounds. Br J Pharmacol. 1955;10:16–25.
22. Bouldin TW, Goines ND, Bagnell CR, Krigman MR. Pathogenesis of trimethyltin neuronal toxicity: ultrastructural and cytochemical observations. Am J Pathol. 1981;104:237.
23. Luria AR. Higher Cortical Functions in Man. 2nd ed. New York, NY: Basic Books Inc Publishers; 1980.
24. Robinson RG, Kubos KL, Starr LB, Rao K, Price TR. Mood disorders in stroke patients: importance of location of lesion. Brain. 1984;107:81–93.
25. Robinson RG, Lipsey JR, Rao K, Price TR. Two-year longitudinal study of post-stroke and mood disorders: comparison of acute-onset with delayed-onset depression. Am J Psychiatry. 1986;143:1238–1244.
26. Rossi GF, Rosadini G. Experimental analysis of cerebral dominance in man. In: Millikan CH, Darley FL, eds. Brain Mechanisms Underlying Speech and Language. New York, NY: Grune & Stratton; 1967.
27. Terzian H. Behavioral and EEG effects of intracarotid sodium amytal injection. Acta Neurochir. 1964;12:230–239.
28. Novelty RA, Augustine EA, Mattson RH, et al. Selective memory improvement and impairment in temporal lobectomy for epilepsy. Ann Neurol. 1984;15:64–67.
29. Sloviter RS, Doeberitz C, Walsh TJ, Dempster DW. On the role of seizure activity in the hippocampal damage produced by trimethyltin. Brain Res. 1986; 367:169–182.
30. Brodie ME, Opacka-Juffry J, Peterson DW, Brown A. Neurochemical changes in hippocampal and caudate dialysates associated with early trimethyltin neurotoxicity in rats. Neurotoxicology. 1990;11:35–46.
31. Wilson WE, Hudson PM, Kanamatsu T, et al. Trimethyltin induced alterations in brain amino acids, amines and amine metabolites: relationship to hyper-ammonemia. Neurotoxicology. 1986;7:63–74.
32. Mailman R, Krigman MR, Frye GD, Hanin I. Effects of postnatal trimethyltin or triethyltin treatment on CNS catecholamine, GABA and acetylcholine systems in the rat. J Neurochem. 1983;40:1423–1429.
33. Yallapragada P, Vig P, Desaiah D. Differential effects of triorganotins on calmodulin activity. J Toxicol Environ Health. 1990;29:317–327.
34. Chang L, Dyer R. Trimethyltin induced zinc depletion in rat hippocampus. In: Frederickson C, Howell G, eds. Neurobiology of Zinc. New York, NY: Alan R Liss Inc; 1984:275–290.
35. Foncin JF, Gruner JE. Tin neurotoxicity. In: Vinken PJ, Bruyn GW, Cohen MM, Klawans HL, eds. Handbook of Clinical Neurology. Amsterdam, the Netherlands: North Holland Publishing Co; 1979:279–290.
36. White RF, Feldman RG, Travers PH. Neurobehavioral effects of toxicity due to metals, solvents and insecticides. Clin Neuropharmacol. 1990;13:392–412.
37. White RF, Proctor SP. Research and clinical criteria for development of neurobehavioral test batteries. J Occup Med. 1992;34:140–148.





