4
Detecting Cancer Early
“Because early detection remains the best guarantee for successful treatment, the Coalition will develop a statewide screening and early detection network of public and private healthcare providers so that every Georgian will have access to cancer screenings.”
Strategic Plan for the Georgia Cancer Coalition, 2001
“In summary, one of the safest, simplest, and most cost-effective ways to reduce cancer morbidity and mortality is to raise the screening rates for selected cancers. There is considerable consensus among experts about high quality screening practices.”
National Healthcare Quality Report,
Agency for Healthcare Research and Quality, 2003
The early detection of cancer refers to the use of screening tests to identify cancer or premalignant disease in persons without signs or symptoms of the disease. It is well established that finding breast cancer and colorectal cancer at an early stage and promptly beginning appropriate treatment—before symptoms develop—improves health outcomes and saves lives (IOM, 2003; NCI, 2004a).
Unfortunately, though, what is known about the potential of early cancer detection is not reflected in current practice (Roetzheim et al., 1999; Breen et al., 2001).
Regular mammography screening for early detection of breast cancer may be a routine in many women’s lives, but far too many women are still
|
BOX 4-1 Use of Cancer Screening Interventions
Cancer Stage at Diagnosis
|
not screened as recommended (Coughlin et al., 2004).1 Large proportions of low-income and uninsured women are especially at risk of not being screened for breast cancer (Rao et al., 2004; Taplin et al., 2004). Rates of colorectal cancer screening fall far short of recommended levels across the board (Swan et al., 2003). If the Georgia Cancer Coalition (GCC) raises breast and colorectal cancer screening rates in Georgia to recommended levels, the state is likely to experience significant declines in cancer-related morbidity and mortality (assuming newly identified cases are promptly followed with appropriate treatment).
The Institute of Medicine (IOM) committee recommends that Georgia adopt five quality indicators to assess the state’s progress in achieving the full potential of early cancer detection (Box 4-1). Two of the indicators would be used to monitor cancer screening rates and three would track cancer stage at diagnosis to gauge the impact of improvements in cancer screening and other means of early detection.
The five recommended quality measures pertaining to cancer early detection are discussed further below, along with the rationale for their selection. For each measure discussed, there is a section providing a brief explanation of the evidence underlying the measure (the “consensus on care”) and a description of what is known about the gap between the evidence and current practice (“knowledge vs. practice”). Potential data sources for measures in the early detection domain are briefly discussed, as
|
1 |
See Chapter 7, Crosscutting Issues in Assessing the Quality of Cancer Care, for a discussion about using quality indicators to address disparities in the behaviors and conditions that lead to cancer, as well as in the incidence, diagnosis, treatment, and outcomes of cancer. |
well. Summaries of each recommended quality measure in the early detection domain appear at the end of the chapter.
RECOMMENDED MEASURES FOR TRACKING THE QUALITY OF EARLY CANCER DETECTION
Use of Cancer Screening Interventions
The IOM committee recommends two quality measures to monitor use of screening services for breast cancer and colorectal cancer:
-
Measure 4-1—Breast cancer screening rate—the proportion of women aged 52 to 69 with one or more mammograms in the past 2 years.
-
Measure 4-2—Colorectal cancer screening rate—the proportion of adults aged 52 to 80 who have received either a fecal occult blood test within the past year, flexible sigmoidoscopy within the past 5 years, colonoscopy within the past 10 years, or double-contrast barium enema within the past 5 years.
Lung and prostate cancer screening measures are not recommended. A cost-effective screening method for lung cancer has yet to be developed (Mahadevia et al., 2003). Prostate screening is controversial. Although the prostate-specific antigen (PSA) screening test detects very early cancers, some of these cancers might not cause any harm if left untreated and, if treated, might lead to impotence and incontinence (Harris and Lohr, 2002; USPSTF, 2002d; Clark et al., 2003). PSA screening is also associated with false positive results and unnecessary anxiety, biopsies, and follow-up diagnostic procedures (Sirovich et al., 2003).
Breast Cancer Screening Rate
Early detection of breast cancer saves women’s lives (Fletcher and Elmore, 2003; IOM, 2005; NCI, 2004b). Strong evidence from numerous randomized clinical trials has shown that routine mammography screening, by detecting cancers sufficiently early for curative treatment, cuts the risk of death from breast cancer by as much as 30 percent (USPSTF, 2002b; IOM, 2003).
In Georgia, breast cancer is the most commonly diagnosed cancer and the second leading cause of cancer death for women—4,953 women were diagnosed with breast cancer and 996 women died of the disease in 2000 (NCI and CDC, 2004; GDPH, 2004b). The American Cancer Society estimates that 6,080 new cases of breast cancer will be diagnosed in Georgia in 2004 (ACS, 2004).
The IOM committee recommends that Georgia monitor mammography rates among women aged 50 to 69, with the goal of increasing the rate statewide. The choice of this age group is a pragmatic one. The evidence supporting regular mammography is strongest for women aged 50 to 69 compared with either younger or older women (USPSTF, 2002b). The measure focuses on a starting age of 52, rather than age 50, because it will be applied retrospectively and should allow for the full 2 years to receive recommended screening. This age group is also targeted by the National Committee for Quality Assurance through its HEDIS program (NCQA, 2004). HEDIS, the Health Plan Employer Data and Information Set, is one of the most widely-used, standardized approaches for assessing the quality of health plans, especially with respect to preventive services.2
Consensus on care. Clinical guidelines with respect to the age at which women should begin regular mammography screening vary. One reason is that the risk of developing breast cancer is age-related. Breast cancer is rare among younger women but incidence increases steadily beginning at age 40 and continues to rise until it peaks in women aged 70 to 79. Among Georgia women under age 40, for example, only 14 per 100,000 were diagnosed with breast cancer from 1999 to 2000 (ACS et al., 2003) (Figure 4-1). During the same period, there were 270 breast cancer cases per 100,000 women aged 50 to 59 and 402 breast cancer cases per 100,000 women aged 70 to 79.
Although there is a strong consensus that all women should receive mammograms every 1 to 2 years beginning at age 50, some groups urge that screening begin at age 40 (IOM, 2003). Earlier screening is advised for those with an increased risk for the disease. The U.S. Preventive Services Task Force (USPSTF) has concluded that the evidence is strongest for women aged 50 to 69, although many studies also indicate a mortality benefit for women aged 40 to 49 (USPSTF, 2002b). Most groups have not issued specific recommendations for women older than age 70 because so few studies have included this age group (Fletcher and Elmore, 2003). Only two randomized trials have enrolled women over age 69, and no trials have enrolled women over age 74 (Humphrey et al., 2002; USPSTF, 2002b; Fletcher and Elmore, 2003).
Knowledge vs. practice. A substantial proportion of Georgia women report having had a mammogram in the past 2 years—81.8 percent of women aged 55 to 64 and 77.5 percent of women over age 64 (Martin et al., 2004). Nevertheless, two vulnerable groups of women—those with lower incomes
|
2 |
See Appendix A for additional information on HEDIS. |
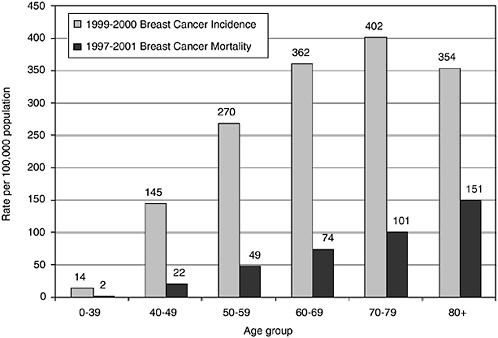
FIGURE 4-1 Breast cancer incidence and mortality in Georgia, by age group, 1997-2001.
NOTE: Rates are annualized and age-adjusted to the 2000 U.S. standard population.
SOURCE: ACS, 2003.
and those who report that they did not have health insurance or they were unable to see a doctor “because of cost” in the previous year—are significantly less likely to be screened. In 2002, as shown in Table 4-1, mammography screening rates for women aged 40 and older with incomes under $25,000 were 15 percentage points lower than the rates for women with incomes of at least $35,000 (65.9 percent or lower vs. 81.7 percent or higher) (Martin et al., 2004). Lacking health insurance and difficulties with medical care costs were particularly significant barriers to getting a mammogram. Almost half of Georgia women aged 40 and older (i.e., 49.7 percent) who cited these difficulties with access to care also reported not having been screened for breast cancer.
Colorectal Cancer Screening Rate
Screening for colorectal cancer substantially improves one’s chances of surviving the disease. It has also been credited with preventing the disease, because when precancerous (referred to as adenomatous) polyps are
TABLE 4-1 Breast Cancer Screening Rate Among Women over Age 40 in Georgia, by Income and Access to Medical Care, 2002
|
Population of Women |
Mammogram in the Past 2 Years (%) |
|
All women |
75.5 |
|
By income group |
|
|
—Less than $15,000 |
63.8 |
|
—$15,000 to $24,999 |
65.9 |
|
—$25,000 to $34,999 |
72.8 |
|
—$35,000 to $49,999 |
81.7 |
|
—$50,000 to $74,999 |
82.2 |
|
—$75,000 or more |
85.6 |
|
By access to medical care |
|
|
—No health insurance or unable to see a doctor “because of cost” |
49.7 |
|
—All others |
79.5 |
|
SOURCE: Martin, et al. 2004. |
|
detected during screening, they can be removed during the procedure (Mandel et al., 2000; Winawer et al., 2003). More than 80 percent of colorectal cancers begin as adenomatous polyps (USPSTF, 2002c).
Colorectal cancer is the second leading cause of cancer death for males and females in the United States. Almost 3,300 Georgians were diagnosed with and 1,260 died of the disease in 2000 (Landis et al., 2004). The risk of developing colorectal cancer increases with age, rising sharply beginning at age 50 (Figure 4-2).
The IOM committee recommends that Georgia monitor colorectal screening rates for all adults aged 50 to 80. The measure focuses on adults starting at age 52, rather than age 50, to allow for the first 2-year period during which recommended screening should occur.
Consensus on care. USPSTF and most other guidelines recommend that, starting at age 50, all people should be periodically screened for colorectal cancer using one of the available options (USPSTF, 2002b; Winawer et al., 2003; IOM, 2003). As shown in Box 4-2, several types of procedures with different frequency are recommended for colorectal cancer screening: the home fecal occult blood test (FOBT), flexible sigmoidoscopy, the combination of home FOBT and flexible sigmoidoscopy, colonoscopy, and double-
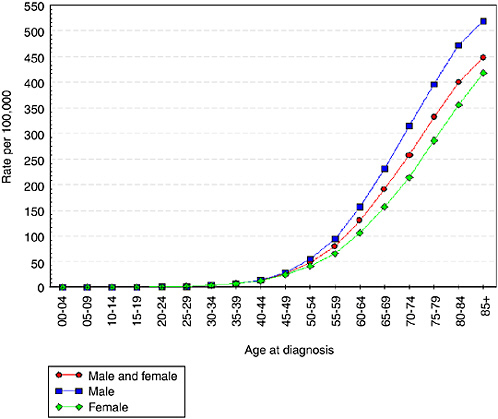
FIGURE 4-2 Incidence of colorectal cancer by age at diagnosis and sex, United States, 1997-2001.
SOURCE: NCI SEER, 2004.
contrast barium enema (USPSTF, 2002c). USPSTF has concluded that while there is insufficient evidence to determine which particular screening strategy is best, colorectal cancer screening is likely to be cost-effective regardless of the type of screening method.
Knowledge vs. practice. Colorectal cancer screening rates in Georgia fall far short of recommended levels (Figure 4-3). In 2002, 47 percent of men and 51 percent of women, over aged 50, reported ever having a sigmoidoscopy or colonoscopy (Martin et al., 2004). An even smaller proportion of adults were screened via an FOBT. In 2001, about 32 percent of all Georgia adults, over age 50, said they had an FOBT in the past 2 years (Martin et al., 2003). Lacking health insurance and difficulties with medical care costs were important barriers to being screened either by FOBT (21.5 percent) or by sigmoidoscopy/colonoscopy (34.3 percent) (Table 4-2).
|
BOX 4-2 Four types of tests can be used to detect premalignant polyps and early-stage colorectal cancers, as discussed below. Fecal Occult Blood Test (FOBT) An FOBT is used to find occult (i.e., hidden) blood in the stool. Blood vessels at the surface of colorectal polyps, adenomas, or cancers often release a small amount of blood into the stool. For an FOBT, a small sample of stool is applied to a chemically treated card; then a chemical developer solution is added. If the card changes color, there is blood in the stool. Blood in the stool can be caused by cancer, but it may also be due to a number of conditions including hemorrhoids, anal fissures, polyps, peptic ulcers, and ulcerative colitis. Recommended annually. Flexible Sigmoidoscopy Sigmoidoscopy enables the physician to view the lining of the large intestine from the rectum through the last part of the colon, called the sigmoid colon. A sigmoidoscope is a slender, flexible, hollow, lighted 2-foot tube. It is inserted through the rectum into the lower part of the colon and transmits an image via a tiny video camera. Recommended every 5 years. Colonoscopy A colonoscope is a longer version of a sigmoidoscope that provides a complete view of the colon. Colonoscopy not only detects but also prevents colorectal cancer because, during the screening procedure, any identified premalignant polyp can be removed. Recommended every 10 years. Double-Contrast Barium Enema A double-contrast (or air-contrast) barium enema is an X-ray examination of the colon and rectum. Using a tube inserted into the rectum, the colon is filled with a contrast material containing barium and is then drained out, leaving only a thin layer of barium on the wall of the colon. Next, the colon is filled with air, providing a detailed X-ray view of the inner surface of the colon including any small polyps, colorectal cancer, or inflammation. Recommended every 5 years. SOURCE: USPSTF, 2002b. |

FIGURE 4-3 Percentage of adults aged 50 and older in Georgia who ever had a colonoscopy or sigmoidoscopy, by sex, 1993-2002.
SOURCE: GDPH, 2004a.
TABLE 4-2 Colorectal Cancer Screening Rate Among Adults over Age 50 in Georgia, by Age and Access to Medical Care, 2001
TABLE 4-3 Survival of Breast Cancer and Colorectal Cancer in the United States, by Stage at Diagnosis, 1995-2000
Cancer Stage at Diagnosis
Cancer stage describes the extent and severity of an individual’s cancer (NCI, 2005).3 If the cancer has spread, the stage describes how far it has spread from the original site to other parts of the body. As illustrated in Table 4-3, cancer stage at diagnosis determines an individual’s prognosis and chance for cure (Compton, 2003). With expansion of breast and colorectal cancer screening, diagnosis of these two cancers should increasingly occur at an earlier, more treatable stage.
In the past, tumor registries typically recorded cancer stage using three different staging systems: (1) Tumor, Regional Lymph Nodes, and Distant Metastasis method (commonly referred to as TNM); (2) Surveillance, Epidemiology, and End Results (SEER) Summary Stage; and (3) SEER Extent of Disease. Each system had a different purpose, data set, and algorithm for translating medical record information into a coded stage. More recently, a unified data set referred to as Collaborative Stage has come into use. Collaborative staging was developed to meet the needs of the three staging systems by combining and standardizing the information needed to assign the stage (AJCC, 2004). NAACCR and SEER registries (including Georgia’s registries) have been required to follow the collaborative method for cases diagnosed from January 2004 forward. Nevertheless, the other staging systems can be derived from the collaborative data elements and they continue to be useful for epidemiological and longitudinal studies.
The IOM committee recommends that GCC adopt the following three quality measures for tracking cancer stage at diagnosis:
-
Measure 4-3—Early-stage breast cancer diagnosis—the proportion of new breast cancer cases that are diagnosed at an early stage
-
Measure 4-4—Advanced-stage breast cancer diagnosis—the number of newly diagnosed advanced-stage breast cancers per 100,000 women aged 40 and older
-
Measure 4-5—Advanced-stage colorectal cancer diagnosis—the number of newly diagnosed advanced-stage colorectal cancer cases per 100,000 adults aged 50 and older
The terms “early stage” and “advanced stage” refer to the SEER summary stage (Box 4-3). SEER summary staging is based on how a cancer grows (Young et al., 2001). Early-stage cancers are noninvasive “in situ” and “localized” tumors that have not spread beyond the organ of origin.
|
BOX 4-3 The SEER summary stages are described below. Early-Stage Cancers
Advanced-Stage Cancers
SOURCE: Young et al., 2001. |
Advanced-stage cancers include “regional” tumors that extend beyond the organ of origin and “distant” cancers in which tumor cells have spread to other parts of the body remote from the original tumor.
Monitoring stage at diagnosis for lung and prostate cancer is not recommended. As noted earlier in the chapter, a cost-effective method for detecting early lung cancer is not currently available (Mahadevia et al., 2003). Although PSA screening detects very early prostate cancers, not treating some of these cancers might be harmless while treating them can lead to serious complications (Harris and Lohr, 2002; USPSTF, 2002d; Clark et al., 2003).
Breast Cancer Stage at Diagnosis
Early-stage breast cancer diagnosis. The first recommended measure related to cancer stage at diagnosis, as noted above, tracks the overall proportion of new breast cancer cases that are at an early, treatable stage.
Consensus on care. Breast cancer is a progressive disease and early detection is life-saving because there are effective treatments for early breast cancer (USPSTF, 2002b; IOM, 2003; NCCN, 2004). Early detection also means that patients have a greater choice of treatments, including the option for breast conserving surgery (Freedman et al., 2003). The available treatment options for advanced-stage breast cancer are much less successful in saving women’s lives.
Knowledge vs. practice. Early-stage breast cancer was relatively rare before the advent of widespread mammography screening, but it has climbed since the 1970s (Fletcher and Elmore, 2003). The most recent data available for Georgia indicate that more than two-thirds of the state’s breast cancer cases (68.5 percent) are diagnosed at an early stage (Table 4-4) (GCCR, 2004). Yet, the likelihood of early-stage diagnosis is considerably
TABLE 4-4 Early- and Advanced-Stage Breast and Colorectal Cancers at Diagnosis in Georgia as a Percentage of Total Cases, 1999-2000
lower among certain groups including African-American women, women who are uninsured, and women who live in high-poverty areas (Li et al., 2003; ACS, 2003; Ghafoor et al., 2003; Gwyn et al., 2004; Ward et al., 2004).
Advanced-stage breast cancer diagnosis. The second recommended quality measure related to cancer stage at diagnosis, as noted above, tracks the incidence of advanced-stage breast cancer (calculated as the number of newly diagnosed advanced-stage breast cancers per 100,000 women aged 40 and older).
Consensus on care. The proportion of women who survive 5 years after a breast cancer diagnosis dramatically declines with advanced-stage disease; from 97.5 percent for localized cases, to 80.4 percent for regional-stage cases, and down to 25.5 percent for distant-stage cases (Table 4-3 above) (Ries et al., 2004). In the previous chapter, the committee recommended that GCC track the overall incidence rates of breast, cervical, and colorectal cancers to evaluate the success of Georgia’s prevention interventions. In this chapter, the committee recommends that GCC monitor another dimension of cancer incidence—the incidence of advanced-stage disease—which should decline as the state draws more women into breast cancer screening and other early breast cancer detection programs.
Knowledge vs. practice. Stage-specific breast cancer incidence rates are not available for Georgia. Nationally, there was little change in the incidence of advanced-stage breast cancer during the 1980s to 1990s (Ghafoor et al., 2003). Yet there are marked differences in the incidence of advanced-stage breast cancer by age. In 2000, for example, there were 125.8 cases per 100,000 women aged 40 to 64 compared with 204.9 cases per 100,000 women aged 65 and older (Table 4-5) (AHRQ, 2003). Although the incidence of breast cancer is highest among white women, the rate of advanced-
TABLE 4-5 Incidence of Advanced-Stage Breast and Colorectal Cancers by Age, in the United States, 2000
|
Type of Cancer |
Age at Diagnosis |
Incidence (per 100,000) |
|
Breast cancer |
Aged 40 and older |
149.7 |
|
40 to 64 |
125.8 |
|
|
65 and older |
204.9 |
|
|
Colorectal cancer |
Aged 50 and older |
95.6 |
|
50 to 64 |
43.4 |
|
|
65 and older |
157.1 |
|
|
SOURCE: AHRQ, National Healthcare Quality Report, 2003. |
||
stage breast cancer is consistently higher among African-American women (ACS, 2003).
Advanced-Stage Colorectal Cancer Diagnosis
The incidence of advanced-stage colorectal cancer will be an indicator of Georgia’s success in ensuring that adults are screened as recommended. If the state succeeds in significantly increasing routine colorectal cancer screening, the incidence of advanced-stage colorectal cancer in Georgia will decline (USPSTF, 2002c; IOM, 2003).
Consensus on care. Colorectal cancer is most likely to be successfully treated if diagnosed early (NCCN, 2003). If diagnosed with early-stage colorectal cancer, adults have an 89.9 percent survival rate at 5 years compared to 67.3 percent for regional-stage colorectal cancer and only 9.6 percent for distant-stage colorectal cancer (Table 4-3) (Ries et al., 2004). There is also strong evidence that if premalignant polyps are removed during the screening procedure, the patient is far less likely to ever develop colorectal cancer (USPSTF, 2002c).
Knowledge vs. practice. Almost 60 percent of Georgians with colorectal cancer are diagnosed with advanced-stage disease (Table 4-4 above) (Martin et al., 2004). From 1999-2000, 7.1 percent of colorectal cancer cases were in situ, 34.2 percent were localized, 41.6 percent were regional, and 17.1 percent were distant. U.S. incidence of advanced-stage colorectal cancer was 95.6 per 100,000 adults aged 50 and older in 2000 (Table 4-5 above) (AHRQ, 2003).
DATA SOURCES
The data sources for the measures presented in this chapter, with one exception, are already currently available in Georgia (Table 4-6). Georgia should expand the prostate cancer screening section of the annual Behavioral Risk Factor Surveillance Survey to include questions regarding men’s awareness of the risks and benefits of the PSA test. Further information about the data sources is presented in Chapter 2, Concepts, Methods, and Data Sources and Appendixes A and B.
SUMMARY
In this chapter, the IOM committee has recommended six cancer quality indicators that GCC should use to gauge its progress in promoting early detection of cancer. There is sound scientific evidence showing that finding
TABLE 4-6 Potential Data Sources for Recommended Measures of the Quality of Cancer Early Detection in Georgiaa
|
Quality Measure |
Potential Georgia-Based Data Sources |
Potential National Data Sources for Benchmarking and Comparison |
|||||||
|
GCCR and Georgia SEER |
Georgia BRFSS |
Georgia SEER/Medicare |
BRFSS |
HEDIS |
HP 2010 |
SEER |
SEER/ Medicare |
NHQR |
|
|
Mammography screening |
|
● |
|
● |
● |
● |
|
● |
● |
|
Colorectal cancer screening |
|
● |
|
● |
● |
● |
|
● |
● |
|
Early-stage breast cancer diagnosis |
● |
|
● |
|
|||||
|
Advanced-stage breast cancer diagnosis |
● |
|
|
|
|
● |
● |
|
● |
|
Advanced-stage colorectal cancer diagnosis |
● |
|
|
|
|
● |
● |
|
● |
|
aSee Chapter 2, Concepts, Methods, and Data Sources, and Appendixes A and B for descriptions of data sources. NOTE: BRFSS = Behavioral Risk Factor Surveillance System; GCCR = Georgia Comprehensive Cancer Registry; HEDIS = Health Employer Data Information Set; HP 2010 = Healthy People 2010; NHQR = National Healthcare Quality Report; SEER = Surveillance, Epidemiology, and End Results. The symbol ● indicates data are currently available. The symbol |
|||||||||
breast cancer and colorectal cancer at an early stage, and beginning treatment early, improves health outcomes and saves lives (IOM, 2003). Unfortunately, what is known about the potential of early detection is not reflected in current practice, especially among many men and women who are poor or uninsured. If GCC succeeds in raising breast and colorectal cancer screening rates to recommended levels, with prompt, appropriate treatment, Georgia is likely to experience significant declines in cancer-related morbidity and mortality. In the shorter term, the state’s progress will be evident in a declining incidence of advanced-stage breast and colorectal cancer.
QUALITY MEASURE SPECIFICATIONS: DETECTING CANCER EARLY
The following section contains summary descriptions of the quality indicators presented in this chapter. These quality indicators were drawn from a variety of clinical practice setting organizations, federal programs, provider groups, and other sources. See Appendix A for descriptions of each of these organizations, including their classification schemes for grading clinical recommendations and characterizing evidence.
|
Measure 4-1. |
Breast Cancer Screening Rate |
|
Measure 4-2. |
Colorectal Cancer Screening Rate |
|
Measure 4-3. |
Early-Stage Breast Cancer Diagnosis |
|
Measure 4-4. |
Advanced-Stage Breast Cancer Diagnosis |
|
Measure 4-5. |
Advanced-Stage Colorectal Cancer Diagnosis |
MEASURE 4-1: DETECTING CANCER EARLY—Breast Cancer Screening Rate
|
Description |
Breast cancer screening rate |
|
Source |
Health Plan Employer Data and Information Set (HEDIS); U.S. Preventive Services Task Force (USPSTF) |
|
Consensus on care |
Clinical guidelines vary with respect to the age at which women should begin regular mammography screening. Although there is a strong consensus that all women should receive mammograms at least every 2 years beginning at age 50, some groups urge that screening begin at age 40 and be repeated annually. Earlier screening is advised for those with an increased risk for the disease. Most groups have not issued specific recommendations for women older than age 70 because so few studies have included this age group. USPSTF recommends screening mammography every 1-2 years for women aged 40 and older but concludes that the evidence is strongest for women aged 50 to 69 (Category B recommendation). Evidence from randomized controlled trials indicates that screening mammography reduces the risk of death from breast cancer for women aged 50 and older. |
|
Knowledge vs. practice |
Most women over age 54 in Georgia report having had a mammogram in the past 2 years. In 2002, 81.8 percent of women aged 55 to 64 and 77.5 percent of women aged 65 and older in Georgia had a mammogram in the past 2 years. However, mammography rates among low-income women (<$15,000 per year) and women with no access to medical care were lower; around 66 percent and 50 percent, respectively, for women aged 40 and older. |
|
Approach to calculating the measure |
|
|
Numerator |
Number of women aged 52 to 69 with one or more mammograms in the past 2 years |
|
Denominator |
Number of women aged 52 to 69 |
|
Potential data source(s) |
Behavioral Risk Factor Surveillance System; Medicare claims and enrollment files; census data; Medicaid claims and enrollment files |
|
Comments |
Although monitoring should begin at age 50, the measure starts at age 52 because it will be applied retrospectively and should allow for the full 2 years to receive recommended screening. |
|
Limitations |
— |
|
Potential benchmark source(s) |
BRFSS; Breast Cancer Surveillance Consortium; National Committee on Quality Assurance/HEDIS reports; Surveillance, Epidemiology, and End Results/Medicare dataset |
|
Key references |
Martin LM, et al. 2004. Georgia Behavioral Risk Factor Surveillance System, 2002 Report. Atlanta, GA: Georgia Department of Human Resources. Publication Number DPH04/158HW. NCQA. 2002. The State of Health Care Quality: 2002. Washington, DC: NCQA. USPSTF. 2002. Guide to Clinical Preventive Services. Third ed. Rockville, MD: AHRQ. |
MEASURE 4-2: DETECTING CANCER EARLY—Colorectal Cancer Screening Rate
|
Description |
Colorectal cancer screening rate |
|
Source |
Health Plan Employer Data and Information Set (HEDIS) |
|
Consensus on care |
Recent studies show an association between colorectal cancer screening and decreased incidence of colorectal cancer. There is also evidence that screening may reduce colorectal cancer mortality. Screening for colorectal cancer is strongly recommended for men and women aged 50 and older (U.S. Preventive Services Task Force category A recommendation). There are several screening options but insufficient evidence on which strategy is best: (1) annual fecal occult blood test (FOBT); (2) flexible sigmoidoscopy every 5 years; (3) annual FOBT plus flexible sigmoidoscopy every 5 years; (4) double-contrast barium enema; and (5) colonoscopy every 10 years. |
|
Knowledge vs. practice |
Screening for colorectal cancer lags far behind screening for other cancers. In 2002, an estimated 49.2 percent of Georgia adults reported having had a sigmoidoscopy or colonoscopy. |
|
Approach to calculating the measure |
|
|
Numerator |
Number of adults aged 52 to 80 who have received either a FOBT within the past year, flexible sigmoidoscopy within the past 5 years, colonoscopy within the past 10 years, or double-contrast barium enema within the past 5 years |
|
Denominator |
Number of adults aged 52 to 80 |
|
Potential data source(s) |
Behavioral Risk Factor Surveillance System (BRFSS); Medicare and Medicaid claims and enrollment files; commercial datasets |
|
Comments |
Although monitoring should begin at age 50, the measure starts at age 52 because it will be applied retrospectively and should allow for the full 2 years to receive recommended screening |
|
Limitations |
HEDIS measures are routinely collected by National Committee for Quality Assurance—accredited managed care organizations; however, HMO enrollment in Georgia is relatively minor outside the Atlanta metropolitan area and minimal among the Medicare population in general. |
|
Potential benchmark source(s) |
HEDIS; BRFSS; Surveillance, Epidemiology and End Results/Medicare dataset |
|
Key references |
CDC. 2003. Behavioral Risk Factor Surveillance System, Prevalence Data, Georgia 2002 Colorectal Cancer Screening. [Online] Available: http://www.apps.nccd.cdc.gov/brfss [accessed November 2003]. GDPH. 2004. OASIS Web Query—Death Statistics. [Online] http://oasis.state.ga.us/webquery/death.html [accessed April 2004]. NCQA. 2002. The State of Health Care Quality: 2002. Washington, DC: NCQA. Schneider E, et al. 2003. Screening for Colorectal Cancer. Draft HEDIS measure. USPSTF. 2002. Guide to Clinical Preventive Services. 3rd edition. Rockville, MD: AHRQ. |
MEASURE 4-3: DETECTING CANCER EARLY—Early-Stage Breast Cancer Diagnosis
|
Description |
Proportion of breast cancer cases diagnosed at an early stage |
|
Source |
Vermont Cancer Center |
|
Consensus on care |
Screening mammography reduces mortality from breast cancer because it detects cancers at an early stage. Five-year relative survival of early-stage, localized breast cancer is 97.5 percent. In contrast, 5-year relative survival of regional-stage breast cancer is 80.4 percent; distant stage is 25.5 percent. Increased breast cancer screening should ultimately increase the proportion of breast cancer cases diagnosed at an early stage. |
|
Knowledge vs. practice |
In Georgia, from 1999-2000, 68.5 percent of breast cancers were diagnosed at an early stage; 31.5 percent at an advanced stage. |
|
Approach to calculating the measure |
|
|
Numerator |
Number of new breast cancer cases diagnosed at an early stage (see comments below) |
|
Denominator |
Number of new breast cancer cases |
|
Potential data source(s) |
Surveillance, Epidemiology, and End Results Program (SEER), Georgia Comprehensive Cancer Registry |
|
Comments |
Early stage refers to SEER summary stage of in situ or localized disease. In situ stage refers to a neoplasm that has not invaded the supporting structure of the organ on which it arose. Localized stage refers to a neoplasm that is limited to the organ of origin; it has not spread beyond that organ. |
|
Limitations |
— |
|
Potential benchmark source(s) |
SEER; U.S. Cancer Statistics publications |
|
Key references |
GCCR. 2004. Georgia Cancer Cases by Stage at Diagnosis, 1999-2000. Unpublished data. IOM. 2003. Fulfilling the Potential of Cancer Prevention and Early Detection. Washington, DC: The National Academies Press. Martin LM, et al. 2004. Georgia Behavioral Risk Factor Surveillance System, 2002 Report. Georgia Department of Human Resources. Publication Number DPH04/158HW. Ries LAG, et al. 2004. SEER Cancer Statistics Review 1975-2001. Bethesda, MD: National Cancer Institute. Available: http://seer.cancer.gov/csr/1975_2001/results_merged/topic_survival.pdf. U.S. DHHS. 2000. Healthy People 2010: Understanding and Improving Health. Chapter 3 Cancer. 2nd edition. Washington, DC: U.S. GPO. USPSTF. 2002. Guide to Clinical Preventive Services. 3rd edition. Rockville, MD: AHRQ. |
MEASURE 4-4: DETECTING CANCER EARLY—Advanced-Stage Breast Cancer Diagnosis
|
Description |
Incidence of advanced-stage breast cancer |
|
Source |
National Healthcare Quality Report; Surveillance, Epidemiology, and End Results Program (SEER) |
|
Consensus on care |
Detection of tumors at an early stage significantly reduces mortality. Five-year relative survival of early-stage, localized breast cancer is 97.5 percent. In contrast, 5-year relative survival of regional-stage breast cancer is 80.4 percent; distant stage is 25.5 percent. Increased breast cancer screening should ultimately reduce the incidence of advanced-stage breast cancer. |
|
Knowledge vs. practice |
The U.S. incidence of advanced-stage breast cancer was 149.7 per 100,000 for women aged 40 and older in 2000. In Georgia, 31.5 percent of breast cancers were diagnosed at an advanced stage from 1999-2000. |
|
Approach to calculating the measure |
|
|
Numerator |
Number of females, aged 40 and older, with new breast cancer diagnosed at advanced-stage (see comments below) |
|
Denominator |
Number of females, aged 40 and older |
|
Potential data source(s) |
SEER; Georgia Comprehensive Cancer Registry |
|
Comments |
Incidence rate = (New advanced-stage breast cancers diagnosed among women aged 40 and older) ÷ (Female population aged 40 and older) x 100,000. Estimate should be age-adjusted to allow comparisons. |
|
|
Advanced stage refers to SEER summary stage of regional or distant disease. Regional stage refers to a neoplasm that has extended beyond the limits of the organ of origin (i.e., into surrounding organs or tissues or into regional lymph nodes). Distant stage refers to a neoplasm that has spread to parts of the body remote from the primary tumor. |
|
Limitations |
— |
|
Potential benchmark source(s) |
National Healthcare Quality Report; Healthy People 2010; U.S. Cancer Statistics publications |
|
Key references |
AHRQ. 2003. National Healthcare Quality Report. Rockville, MD: U.S. DHHS. GCCR. 2004. Georgia Cancer Cases by Stage at Diagnosis, 1999-2000. Unpublished data. IOM. 2003. Fulfilling the Potential of Cancer Prevention and Early Detection. Washington, DC: The National Academies Press. Ries LAG, et al. 2004. SEER Cancer Statistics Review 1975-2001. Bethesda, MD: National Cancer Institute. Available: http://seer.cancer.gov/csr/1975_2001/results_merged/topic_survival.pdf U.S. DHHS. 2000. Healthy People 2010: Understanding and Improving Health. Chapter 3 Cancer. 2nd edition. Washington, DC: U.S. GPO. USPSTF. 2002. Guide to Clinical Preventive Services. 3rd edition. Rockville, MD: AHRQ. |
MEASURE 4-5: DETECTING CANCER EARLY—Advanced-Stage Colorectal Cancer Diagnosis
|
Description |
Incidence of advanced-stage colorectal cancer |
|
Source |
National Healthcare Quality Report; Surveillance, Epidemiology, and End Results Program (SEER) |
|
Consensus on care |
Colorectal cancer screening detects premalignant polyps and early stage cancers. It can also guide removal of premalignant polyps thus preventing cancer from developing. Detection of tumors at an early stage significantly reduces mortality. Five-year relative survival of localized-stage colorectal cancer is 89.9 percent. In contrast, 5-year relative survival of regional-stage colorectal cancer is 67.3 percent; 9.6 percent, distant stage. Increased colorectal cancer screening should ultimately reduce the incidence of advanced-stage colorectal cancer. |
|
Knowledge vs. practice |
The U.S. incidence of advanced-stage colorectal cancer was 95.6 per 100,000 for adults aged 50 and older in 2000. In Georgia, 58.7 percent of colorectal cancers were diagnosed at an advanced stage from 1999-2000. |
|
Approach to calculating the measure |
|
|
Numerator |
Number of adults, aged 50 and older, with new colorectal cancer diagnosed at an advanced stage (see comments below) |
|
Denominator |
Adult population aged 50 and older |
|
Potential data source(s) |
Georgia Comprehensive Cancer Registry, SEER |
|
Comments |
Incidence rate = (New advanced-stage colorectal cancers diagnosed among adults aged 50 and older) ÷ (Adult population aged 50 and older) × 100,000. Estimate should be age-adjusted to allow comparisons. |
|
|
Advanced stage refers to SEER summary stage of regional or distant disease. Regional stage refers to a neoplasm that has extended beyond the limits of the organ of origin (i.e., into surrounding organs or tissues or into regional lymph nodes). Distant stage refers to a neoplasm that has spread to parts of the body remote from the primary tumor. |
|
Limitations |
— |
|
Potential benchmark source(s) |
National Healthcare Quality Report; Georgia Comprehensive Cancer Registry; SEER; U.S. Cancer Statistics publications |
|
Key references |
AHRQ. 2003. National Healthcare Quality Report. Rockville, MD: U.S. DHHS. GCCR. 2004. Georgia Cancer Cases by Stage at Diagnosis, 1999-2000. Unpublished data. Martin LM, et al. 2003. Georgia Behavioral Risk Factor Surveillance System, 2001 Report. Atlanta, GA: Georgia Department of Human Resources, Publication Number DPH03-069HW. NCI. 2004. SEER Cancer Statistics Review, 1975-2001. Bethesda, MD: National Cancer Institute. Available: http://seer.cancer.gov/csr/1975_2001/results_merged/topic_survival.pdf. U.S. DHHS. 2000. Healthy People 2010: Understanding and Improving Health. 2nd edition. Chapter 3 Cancer. Washington, DC: U.S. GPO. USPSTF. 2002. Guide to Clinical Preventive Services. 3rd edition. Rockville, MD: AHRQ. |
REFERENCES
ACS (American Cancer Society). 2003. Breast Cancer Facts & Figures 2003. Atlanta, GA: ACS.
——. 2004. Cancer Facts & Figures 2004. Atlanta, GA: ACS.
ACS, University of Georgia Cooperative Extension Service, Georgia Department of Human Resources. 2003. Breast Cancer. [Online] Available: http://www.fcs.uga.edu/pubs/ppt/PPT-43.ppt [accessed July 23, 2004].
AHRQ (Agency for Healthcare Research and Quality). 2003. National Healthcare Quality Report. Rockville, MD: U.S. DHHS.
AJCC (American Joint Committee on Cancer). 2004. Collaborative Staging: Introduction. [Online] Available: http://www.cancerstaging.org/cstage/cstageintro.html [accessed January 13, 2005].
Breen N, Wagener DK, Brown ML, Davis WW, Ballard-Barbash R. 2001. Progress in cancer screening over a decade: results of cancer screening from the 1987, 1992, and 1998 National Health Interview Surveys. J Natl Cancer Inst. 93(22): 1704-13.
CDC. 2003. Behavioral Risk Factor Surveillance System, Prevalence Data, Georgia 2002 Colorectal Cancer Screening. [Online] Available: http://www.apps.nccd.cdc.gov/brfss [accessed November 2003].
Clark JA, Inui TS, Silliman RA, Bokhour BG, Krasnow SH, Robinson RA, Spaulding M, Talcott JA. 2003. Patients’ perceptions of quality of life after treatment for early prostate cancer. J Clin Oncol. 21(20): 3777-84.
Compton CC. 2003. Colorectal carcinoma: diagnostic, prognostic, and molecular features. Mod Pathol. 16(4): 376-88.
Coughlin SS, Uhler RJ, Bobo JK, Caplan L. 2004. Breast cancer screening practices among women in the United States, 2000. Cancer Causes Control. 15(2): 159-70.
Fletcher SW, Elmore JG. 2003. Clinical practice. Mammographic screening for breast cancer. N Engl J Med. 348(17): 1672-80.
Freedman GM, Anderson PR, Goldstein LJ, Hanlon AL, Cianfrocca ME, Millenson MM, von Mehren M, Torosian MH, Boraas MC, Nicolaou N, Patchefsky AS, Evers K. 2003. Routine mammography is associated with earlier stage disease and greater eligibility for breast conservation in breast carcinoma patients age 40 years and older. Cancer. 98(5): 918-25.
GCC (Georgia Cancer Coalition). 2001. Strategic Plan. Atlanta, GA: GCC.
GCCR (Georgia Comprehensive Cancer Registry). 2004. Georgia Cancer Cases by Stage at Diagnosis, 1999-2000. Unpublished Data.
GDPH (Georgia Division of Public Health). 2004a. Georgia Behavioral Risk Factor Surveillance System: Colorectal Cancer Screening. [Online] Available: http://health.state.ga.us/epi/brfss/colorectal.asp [accessed November 26, 2004].
——. 2004b. OASIS Web Query—Death Statistics. [Online] Available: http://oasis.state.ga.us/webquery/death.html [accessed April 2004].
Ghafoor A, Jemal A, Ward E, Cokkinides V, Smith R, Thun M. 2003. Trends in breast cancer by race and ethnicity. CA Cancer J Clin. 53(6): 342-55.
Gwyn K, Bondy ML, Cohen DS, Lund MJ, Liff JM, Flagg EW, Brinton LA, Eley JW, Coates RJ. 2004. Racial differences in diagnosis, treatment, and clinical delays in a population-based study of patients with newly diagnosed breast carcinoma. Cancer. 100(8): 1595-604.
Harris R, Lohr KN. 2002. Screening for prostate cancer: an update of the evidence for the U.S. Preventive Services Task Force. Ann Intern Med. 137(11): 917-29.
Humphrey LL, Helfand M, Chan BK, Woolf SH. 2002. Breast cancer screening: a summary of the evidence for the U.S. Preventive Services Task Force. Ann Intern Med. 137(5 Part 1): 347-60.
IOM (Institute of Medicine). 2003. Fulfilling the Potential of Cancer Prevention and Early Detection. Curry S, Byers T, Hewitt M, Editors. Washington, DC: The National Academies Press.
——. 2005. Saving Women’s Lives: Strategies for Improving Breast Cancer Detection and Diagnosis. Joy JE, Penhoet EE, Petitti DB, Editors. Washington, DC: The National Academies Press.
Landis SH, Steiner CB, Bayakly AR, McNamara C, Powell KE. 2004. Georgia Cancer Data Report, 2000. Atlanta, GA: Georgia Department of Human Resources, Division of Public Health, Cancer Control Section, and the American Cancer Society, Southeast Division.
Li CI, Malone KE, Daling JR. 2003. Differences in breast cancer stage, treatment, and survival by race and ethnicity. Arch Intern Med. 163(1): 49-56.
Mahadevia PJ, Fleisher LA, Frick KD, Eng J, Goodman SN, Powe NR. 2003. Lung cancer screening with helical computed tomography in older adult smokers: a decision and cost-effectiveness analysis. JAMA. 289(3): 313-22.
Martin LM, Chowdhury PP, Powell KE, Clanton J. 2003. Georgia Behavioral Risk Factor Surveillance System, 2001 Report. Atlanta, GA: Georgia Department of Human Resources, Division of Public Health, Chronic Disease, Injury, and Environmental Epidemiology Section.
——. 2004. Georgia Behavioral Risk Factor Surveillance System, 2002 Report. Atlanta, GA: Georgia Department of Human Resources, Division of Public Health, Chronic Disease, Injury, and Environmental Epidemiology Section.
NCCN (National Comprehensive Cancer Network). 2003. Clinical Practice Guidelines in Oncology-v.1.2003. Colorectal Screening. [Online] Available: http://www.nccn.org/professionals/physician_gls/PDF/colorectal_screening.pdf [accessed 2004].
——. 2004. Clinical Practice Guidelines in Oncology-v.1.2004. Breast Cancer Screening and Diagnosis Guidelines. [Online] Available: http://www.nccn.org/professionals/physician_gls/PDF/breast-screening.pdf [accessed 2004].
NCI (National Cancer Institute). 2004a. Cancer Progress Report—2003 Update. [Online] Available: http://progressreport.cancer.gov/ [accessed July 30, 2004].
——. 2004b. Cancer Progress Report Update—2003. Early Detection: Breast Cancer Screening. [Online] Available: http://progressreport.cancer.gov/doc.asp?pid=1&did=21&chid=10&coid=24&mid=vpco [accessed July 29, 2004].
——. 2005. Introduction to Collaborative Staging. [Online] Available: http://training.seer.cancer.gov/module_collab_stage/00_cs_home.html [accessed February 1, 2005].
NCI SEER (National Cancer Institute, Surveillance, Epidemiology, and End Results). 2004. Incidence: Colon and Rectum Cancer. SEER*Stat Databases. [Online] Available: http://seer.cancer.gov/faststats/html/inc_colorect.html [accessed February 28, 2005].
NCI and CDC (National Cancer Institute and Centers for Disease Control and Prevention). 2004. State Cancer Profiles. [Online] Available: http://statecancerprofiles.cancer.gov/incidencerates/incidencerates.html [accessed July 9, 2004].
NCQA (National Committee for Quality Assurance). 2002. The State of Health Care Quality: 2002. Washington, DC: NCQA.
——. 2004. The Health Plan Employer Data and Information Set (HEDIS). [Online] Available: http://www.ncqa.org/Programs/HEDIS/index.htm [accessed December 3, 2004].
Rao RS, Graubard BI, Breen N, Gastwirth JL. 2004. Understanding the factors underlying disparities in cancer screening rates using the Peters-Belson approach: results from the 1998 National Health Interview Survey. Med Care. 42(8): 789-800.
Ries LAG, Eisner MP, Kosary CL, Hankey BF, Miller BA, Clegg L, Mariotto A, Feuer EJ, Edwards BK, Editors. 2004. SEER Cancer Statistics Review, 1975-2001. Bethesda, MD: NCI.
Roetzheim RG, Pal N, Tennant C, Voti L, Ayanian JZ, Schwabe A, Krischer JP. 1999. Effects of health insurance and race on early detection of cancer. J Natl Cancer Inst. 91(16): 1409-15.
Schneider E, McGlynn E, and Nadal M. 2003. Screening for Colorectal Cancer. Draft HEDIS measure.
Sirovich BE, Schwartz LM, Woloshin S. 2003. Screening men for prostate and colorectal cancer in the United States: does practice reflect the evidence? JAMA. 289(11): 1414-20.
Swan J, Breen N, Coates RJ, Rimer BK, Lee NC. 2003. Progress in cancer screening practices in the United States: results from the 2000 National Health Interview Survey. Cancer. 97(6): 1528-40.
Taplin SH, Ichikawa L, Yood MU, Manos MM, Geiger AM, Weinmann S, Gilbert J, Mouchawar J, Leyden WA, Altaras R, Beverly RK, Casso D, Westbrook EO, Bischoff K, Zapka JG, Barlow WE. 2004. Reason for late-stage breast cancer: absence of screening or detection, or breakdown in follow-up? J Natl Cancer Inst. 96(20): 1518-27.
U.S. DHHS (U.S. Department of Health and Human Services). 2000. Healthy People 2010: Understanding and Improving Health. 2nd edition. Washington, DC: U.S. Government Printing Office.
USPSTF (U.S. Preventive Services Task Force). 2002a. Guide to Clinical Preventive Services. 3rd edition. Rockville, MD: AHRQ.
——. 2002b. Screening for Breast Cancer: Recommendations and Rationale. Rockville, MD: U.S. DHHS, AHRQ.
——. 2002c. Screening for Colorectal Cancer: Recommendations and Rationale. Rockville, MD: U.S. DHHS, AHRQ.
——. 2002d. Screening for Prostate Cancer: Recommendations and Rationale. Rockville, MD: U.S. DHHS, AHRQ.
Ward E, Jemal A, Cokkinides V, Singh GK, Cardinez C, Ghafoor A, Thun M. 2004. Cancer disparities by race/ethnicity and socioeconomic status. CA Cancer J Clin. 54(2): 78-93.
Winawer S, Fletcher R, Rex D, Bond J, Burt R, Ferrucci J, Ganiats T, Levin T, Woolf S, Johnson D, Kirk L, Litin S, Simmang C. 2003. Colorectal cancer screening and surveillance: clinical guidelines and rationale-update based on new evidence. Gastroenterology. 124(2): 544-60.
Young JL, Roffers SD, Ries LAG, Fritz AG, Hurlbut AA, Editors. 2001. SEER Summary Staging Manual—2000: Codes and Coding Instructions. Bethesda, MD: NCI





























