Types of High-Level Radioactive Wastes Formed as a Result of Dry Methods of Spent Fuel Regeneration and Technologies for Their Management
Valentin B. Ivanov
Institute of Geology of Ore Deposits, Petrography, Mineralogy, and Geochemistry
Russian Academy of Sciences
At present for the nuclear industry to adopt new nuclear fuel cycle technologies and for public opinion to be prepared to accept their implementation, it is absolutely essential that all fuel cycle-related problems without exception be resolved, especially those problems involving the management of radioactive wastes. In proposing new approaches to spent fuel regeneration, suppliers of such technologies must ensure that all aspects of the separations process are comprehensively addressed. This sort of approach was taken in the development and testing of dry technologies for spent fuel regeneration. The pyroelectrochemical technology, which is based on the use of salt melts, is the most ready for industrial application, and has been developed and tested on a semi-industrial scale at the Scientific Research Institute of Atomic Reactors (NIIAR).
A top priority in any technology used in the nuclear fuel cycle is its safety, and this means not only nuclear and radiation safety but also general industry safety as well. A safety analysis has shown the fundamental advantages of the pyroelectrochemical regeneration technology in comparison with currently used water-based methods for reprocessing spent nuclear fuel. The basic safety aspects and physical-chemical properties of the process on which it is based are presented in Box 1. This technological process is also notable for the fact that it is self-protected against the unauthorized removal of fissile materials, as it is characterized by the presence of powerful gamma radiation in all its stages. Therefore, an additional requirement for this type of regeneration process is that it should facilitate the separation of the greatest possible quantity of fissile materials with the least possible removal of fission products. Such an approach also minimizes the amount of radioactive materials, and especially radioactive wastes, that are removed from the fuel cycle.
|
BOX 1
|
|
BOX 2 Plutonium oxide with the following isotopic composition:
Am-241 in an amount equaling 2.8 kg per metric ton of plutonium Remainder of fuel load: combined U-238 in dioxide form and 5–10 percent in metal form, U-235 less than 0.2 percent of the total mass of uranium |
To illustrate, I present the following data on the composition of plutonium-based fuel separated from spent fuel from a water-moderated water-cooled power reactor (VVER-440), as well as experimental data obtained as a result of the regeneration of spent fuel removed from a BOR-60 fast reactor, with this material being analyzed for its potential fuel content to be reloaded into a reactor (see Boxes 2, 3, and 4). It is obvious that after regeneration, the fuel contains not only fission products but also minor actinides. These elements do not hinder the phys-
|
BOX 3 Plutonium oxide and uranium oxide with the following isotopic composition:
Am-241 in an amount equaling 5.4 kg per metric ton of plutonium and 0.376 kg per metric ton of uranium; the regenerated uranium output also includes Cm-242 (1.2 g per metric ton) and Cm-244 (14 g per metric ton) Fission products (Curies per gram of PuO2):
Corrosion elements: Mn-54 at 5.9 · 10−5 and Co-60 at 3.2 · 10−6 Curies per gram of PuO2 Cation additives 8.86 mass percent of the mass of PuO2 and by nomenclature Si, Fe, Mg, Cr, Zr, Mo, Gd, Ni, Pb, Ag, Y, La, Tb, Dy, B, Ga, Be, Ca, Ce, Pr, Eu, Ti, Cu, Na, Pd, Nd, Sm, Sc |
||||||||||||||||||||||||||||||||||||
|
BOX 4 Left after regeneration:
Am and Cm in approximately the same quantity per kg as in fuel prepared on the basis of spent fuel from BOR-60 reactors Standard purification coefficients for fission products:
Obviously a substantial part of the fission products and minor actinides remains in the fuel to be reloaded into the active zone of the reactor. |
ics of the reactor’s operation, that is, they may pass through the cycle repeatedly without increasing the volumes and radioactivity of the materials removed. This is a great advantage of this technology, inasmuch as with the use of remote-controlled transport procedures this fulfills the conditions for self-protection of the fuel and reduces the quantity and activity of the radioactive substances removed from the cycle.
Dry regeneration also results in the formation of radioactive wastes. The sources and forms of these wastes are presented in Box 5. These types of wastes are subjected to special processing so that they may be placed either immediately or after a certain holding period into deep geological formations for permanent storage. Thus, enterprises manufacturing regenerated fuel for the new generation of fast neutron reactors will not accumulate radioactive wastes. The technologies and procedures for primary processing of radioactive wastes formed during the pyrochemical regeneration of spent fuel are shown in Box 6.
|
BOX 5
|
|
BOX 6
|
The specific characteristics of high-level radioactive wastes formed during the regeneration of spent fuel with a burn-up rate of 21 percent of the heavy atoms are shown in Table 1. It should be noted that the output volume of contaminated electrolyte, 2.5 kg/kg, is valid only for this particular experiment. In real-life application of the technology the electrolyte undergoes regeneration and the output volume that must be disposed of is on the order of 10 times less.
One method of disposing of the wastes that are created is vitrification. This procedure is carried out directly with the separated product without any intermediate stages. The characteristics of the glass matrices that are produced are presented in Table 2. The high radiation resistance of the matrix and the low rate of leaching are particularly noteworthy. When wastes are placed in a ceramic form, the leaching rate is slightly accelerated, but this process provides a substantially greater degree of thermal stability, which is especially important in the regeneration of spent fuel that has been stored for a relatively short period of time (see Table 3).
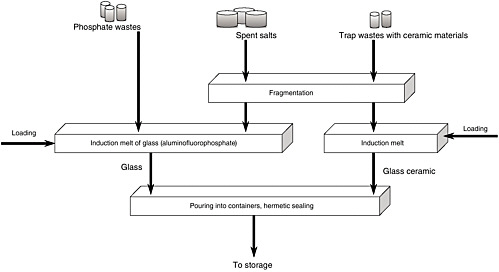
The technology for radioactive waste reprocessing is illustrated in Figure 1. As the schematic clearly shows, the products separated out by the process undergo only two or three operations, and the wastes are then ready for burial.
A full listing of the various types of wastes and their isotopic composition is provided in Table 4. One can see that the bulk of the radioactivity is concentrated in the phosphate precipitate (Ce(Pr)-144), the spent electrolyte, and the steamed salts (Cs-134 and -137). All of these substances are contained in the same chlorinator-electrolyzer device both during the regeneration process and after its completion. They are not transported and consequently do not contaminate any other equipment or hot chambers. All of this is very significant for reducing the volume of radioactive wastes created during the decontamination process.
The fluoride gas technology for regenerating spent nuclear fuel is less ready for industrial application. Nevertheless, a certain amount of research has been completed with regard to the management of radioactive wastes created as a result of this process. In regenerating only uranium irradiated fuel, an average of 86
TABLE 1 Characteristics of Solid High-Level Wastes (Spent Fuel from BOR-60 Reactors with a Burn-up Rate of 21 Percent of Heavy Atoms, Stored for Two Years)
|
Type |
Fuel Output, kg/kg |
Specific Heat, Wt/kg |
Temperature of Spontaneous Combustion, °C |
|
Phosphate precipitate |
0.14 |
14.8 |
40 (180 g) |
|
Electrolyte |
2.5 |
0.95 |
30 (2 kg) |
TABLE 2 Vitrification of High-Level Wastes (HLW) Using the Pyroelectrochemical Process
|
Type of HLW |
Type of Glass Matrix |
Means of Incorporation |
|
Phosphate precipitate |
Pb(PO3)2, NaPO3 |
Vitrification, T-950°C |
|
Spent salt electrolyte |
NaPO3, AlF3, Al2O3 |
Vitrification without conversion of chlorides, T-950°C |
|
Phosphate precipitate plus spent salt electrolyte |
NaPO3, AlF3, Al2O3 |
Joint vitrification without conversion of chlorides, T-950°C |
TABLE 3 Ceramization of High-Level Wastes (HLW) Using the Pyroelectrochemical Process
|
Type of HLW |
Type of Ceramic |
Means of Incorporation |
|
Phosphate precipitate |
Monazite |
Pressing, kilning, T-850°C |
|
Spent salt electrolyte |
Kosnarite (sodium zirconium phosphate—NZP) |
Conversion into NZP from melt or water solution, pressing, kilning, T-950°C |
TABLE 4 Solid Technical Wastes and Products
|
Waste Product |
Mass, kg |
Mn-54 |
Co-60 |
|
UO2-1 product after electrolysis of restored melt |
0.489 |
< 4.4 |
< 1.6 |
|
UO2-2 product after electrolysis of acidified melt |
1.510 |
0.3 |
0.5 |
|
PuO2 product after precipitation crystallization |
0.504 |
2.2 |
0.1 |
|
Phosphate precipitate |
0.442 |
24.8 |
1.3 |
|
Spent electrolyte |
8.114 |
— |
0.1 |
|
Steamed salts |
0.968 |
2.1 |
0.4 |
|
Sublimates |
0.495 |
traces |
traces |
|
Pyrographite materials, filters, fuel rod casings |
20 |
0.8 |
— |
|
Amount of Wastes Incorporated (percent) |
Cs-137 Leaching Rate Over 7 Days, g/cm2/day |
Thermal Stability, °C |
Radiation Resistance |
|
28 |
7 · 10−6 |
400 |
|
|
20 |
7 · 10−6 |
400 |
1 · 107 Gr (γ, β) 1 · 1018 α emitters/g |
|
36 |
4 · 10−6 |
400 |
|
|
Amount of Wastes Incorporated (percent) |
Cs-137 Leaching Rate Over 7 Days, g/cm2/day |
Thermal Stability, °C |
Radiation Stability |
|
100 |
1 · 10−6 |
850 |
5 · 108 grays |
|
30–40 |
3 · 10−6 |
1,000 |
(γ, β) 1 · 1019 α emitters/g |
|
Specific Activity of Nuclides, GBq/kg on June 1, 1995 |
||||||
|
Ru(Rh)-106 |
Sb-125 |
Cs-134 |
Cs-137 |
Ce(Pr)-144 |
Eu-154 |
Eu-155 |
|
40,700 |
362.6 |
0.4 |
4.1 |
196.1 |
2.9 |
4.8 |
|
814 |
35.2 |
0.3 |
3 |
70.3 |
51.8 |
136.9 |
|
4,810 |
77.7 |
0.7 |
7.4 |
1,961 |
21.5 |
74 |
|
151.7 |
229.4 |
16.7 |
170.2 |
96,200 |
888 |
4,440 |
|
2 |
1.1 |
207.2 |
2,072 |
244.2 |
6.3 |
— |
|
777 |
74 |
170 |
1,369 |
2,664 |
21.8 |
144 |
|
48.1 |
484.7 |
26.3 |
263 |
29.6 |
traces |
— |
|
21 |
10.5 |
4.5 |
44.5 |
15.4 |
0.2 |
— |
|
BOX 7 Spent fuel and sorbent materials are first subjected to a dosing procedure and then smelted in a crucible at a temperature of up to 800°C. Then they are poured into stainless steel containers, which are hermetically sealed and inspected. The outside surfaces of the containers are decontaminated. After this process, the wastes are characterized by
|
percent of the activity of the fission products is concentrated in the fluorination residues, whereas in regenerating mixed fuel this figure is only 53 percent, which is explained by the characteristics of the technical process of fluorination (increased temperature, large surplus of fluorine). The remainder of the activity is distributed among the chemical and other sorbents and the various tubes and pipes in the processing equipment. The output of fluorination residues totals about 0.2 kg/kg of spent fuel processed, with these residues showing concentrations of mainly long-lived radionuclides (cerium-144, praseodymium-144, cesium-134 and -137, and strontium-90) that form relatively nonvolatile fluorides.
The stability of the fluorination residues depends on the activity of the fuel being regenerated and increases over time. All of the solid wastes are highly active powders (fluorination residues) or granules (sorbents), and they require special storage technology. All of these materials can be safely stored without any special preparation in stainless steel containers; however, for long-term controlled storage a technology has been developed for the preliminary smelting of solid wastes. Box 7 describes the process for preparing solid wastes with high specific activity and presents information on their fundamental properties.
The release of gas-phase Cs-137 from the wastes increases along with increases in temperature; however, the rate of its volatilization is lower than that for the sorbents and spent fuel wastes separately (that is, the figures go from 1.3 · 10−4 to 1.8 · 10−10 g/cm2 per day). The heat conductivity of the smelted wastes in the temperature range from 100 to 400°C totaled 0.51 to 0.79 Wt/m °C. The temperature of the walls of the stainless steel waste containers must not exceed 300°C in order to avoid notable corrosion of the material of the container casing.
CONCLUSION
-
Research studies and the technologies that have been developed for managing high-level radioactive wastes produced in the regeneration of spent nuclear fuel using dry methods (pyroelectrochemical and gas fluoride technologies) indicate the feasibility of controlled storage of such wastes in simple hermetically sealed vessels for a prolonged period (decades).
-
Radioactive wastes created as a result of pyroelectrochemical regeneration may be placed fairly easily in glass or ceramic forms suitable for permanent burial.
-
A program of research work must be completed to determine the optimal form (for example, chemical composition, type of matrix) for solid high-level radioactive wastes from the standpoint of ensuring the safety of their burial in geological formations. This research is a vital element of the regeneration technologies being developed to facilitate the new closed fuel cycles.