8
Analyzing Health Risks
ASSESSMENT OF RISKS POSED BY A BIOLOGICAL HAZARD
Dose–response analysis for microorganisms and for chemicals differs substantially in several ways. First, illness can result from the exposure to low numbers (up to tens or hundreds) of microorganisms. Illness attendant to exposure to chemical agents, even at low doses, involves exposure to substantially larger numbers of molecules (thousands or more). Also, even within a population exposed to small quantities of microorganisms, there can be large differences in the number of organisms to which individuals in a population are actually exposed (Figure 8-1). Hence, the intrinsic sampling variability—the fact that there can be significant differences in the actual numbers of organisms to which each person is exposed, even in a group exposed under the same average conditions—must be considered in the assessment of exposure and dose–response with microorganisms. Chemical risk assessment can ignore that phenomenon.
Figure 8-1 shows that at an average inhaled dose of 0.01 organism, there is a 1% probability of inhaling one organism (1 person in 100 exposed persons will inhale 1 organism). There is a somewhat smaller than 0.01% chance of inhaling 2 organisms. If the average inhaled dose is 3 organisms, there is about a 0.02% chance of inhaling 10 organisms. This shows that even if the average inhaled dose is low, there can still be a small fraction of the exposed population that will actually inhale an amount much larger.
The second difference is in the potential consequences of exposure to a single microorganism compared with exposure to one chemical particle. Plausible biological information indicates that one microorganism can cause harm in
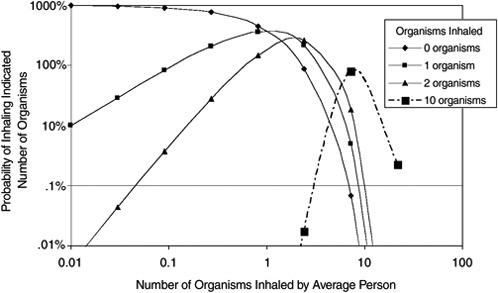
FIGURE 8-1 Probability of inhaling different numbers of organisms as a function of the average inhaled dose in a population (based on Poisson statistics).
the form of specific disease in specific subgroups of the human population (Rubin, 1987), whereas many particles of a chemical agent would be necessary to provoke an effect—depending on the chemical agent and the mode of action. In the case of microbial agents, an ingested, inhaled, or absorbed microorganism can multiply within the body of a susceptible human, to produce sufficient microorganisms in vivo that illness can result. However, the ability of the microorganisms to multiply does not imply that the exposure to a single organism will always produce illness, because the organism could be killed by defense processes—for example, the acidity of the gastrointestinal tract or the action of the immune system—before it can reproduce in sufficient numbers to cause an effect. That said, a single organism has the biological potential to produce an effect if a sufficient number of progeny are produced and survive. The ability of one organism to produce such an effect is strongly dependent on the susceptibility of the host, although susceptibility varies extensively in the human population, which consists of the young, the old, the healthy, and the immunocompromised.
The third difference is that the microbial exposure of one individual also can have a subsequent effect in the broader population (including persons who are not exposed directly). Some diseases are transmissible from person to person and can be spread even via asymptomatic individuals. Therefore, interpersonal contact can cause others to become ill. The degree of secondary spread depends on the organism’s infectivity and its excretion pattern and on the behavior of infected persons.
A dose–response relationship describes the quantitative dependency of the proportion of the population that experiences an adverse effect (infection, illness, or death) caused by the average dose of microorganisms that the population receives (ingests, inhales, or contacts). Individual “points” on the relationship can be useful to describe the potency of the infectious agent; for example, the ID50 and LD50 denote the dose needed to infect or kill half of the population exposed, respectively. Those terms also are called the “median infectious dose” or “median lethal dose.”
Unfortunately, summary tabulations of infectious agents often use the terms “infectious dose” or “lethal dose,” without modifying adjectives. The result has been the misapprehension that doses below the ID50 and LD50 are without adverse effect. That interpretation is incorrect, and, for example, to ascertain the dose required to infect 1% of a population (which might be termed the ID1), one would need to examine the dose–response relationship at low doses (and risks).
DOSE-RESPONSE: PRINCIPLES AND UNCERTAINTIES
Exposure to infectious organisms has been described with a dose–response construct at least since the 1950’s. Wells (1955) described the process of inhalation infection as involving the statistics of inhalation of “quanta of infection,” and he acknowledged an approximate exponential dose-response relationship by analogy with the most-probable-number bacterial assay. Riley and O’ Grady (1961) elaborated on Wells’s concept. They recognized that there may be various intrinsic and extrinsic modifiers of the infectivity of organisms.
All of those researchers’ ideas about dose–response relationships have been used in industrial hygiene applications, inlcuding the analysis of tuberculosis risk (Nicas, 1996).
Although there are several dose-response models for infection or disease produced by pathogens, only one has been used with a Category A agent, and in that case the research involved animal hosts. Other dose-response models have not been published for other Category A agents, but there are data available that could be used to infer doses and responses for those agents (Table 8-1).
Since the early 1980s, a paradigm has emerged for describing risk attributable to exposure to microorganisms. Standard risk assessment techniques (dose–response assessment, exposure assessment) are used to estimate risk from pathogenic microorganisms (Haas, 1986; Haas et al., 1999a).
The two most successful dose-response models are the exponential model and the beta-Poisson model. Both models are derived from the following assumptions:
-
A single organism surviving to colonize at a target site is sufficient to initiate the infectious disease process in some individuals.
-
The probability that any ingested organism survives to colonization is independent among all organisms that are actually inhaled or ingested.
TABLE 8-1 List of Published Dose-Response Studies (or Data from which Dose–Response Could be Inferred) for Microbial Infection or Illness
|
Bacteria |
|
|
|
|
Salmonella |
(Fazil 1996; Holcombet al., 1999; Havelaar et al., 2000) |
|
Shigella |
(Crockett et al., 1996) |
|
|
Enteropathogenic E coli |
(Haas et al., 1999) |
|
|
E. coli O157:H7 (animals) |
(Haas et al., 2000) |
|
|
Vibrio |
(Haas et al., 1999) |
|
|
Campylobacter |
(Medema 1996) |
|
|
Listeria (animals) |
(Haas 1999) |
|
|
Bacillus anthracis (animals) |
(Haas 2002) |
|
|
Francisella tularensis (animals) |
(Oyston 2004) |
|
|
Viruses |
|
|
|
|
Rotavirus |
(Haas et al., 1993; Gerba et al., 1996) |
|
ECHO |
(Haas et al., 1999) |
|
|
Coxsackie |
(Haas et al., 1999) |
|
|
Adenoviruses |
(Haase et al., 1999) |
|
|
Ebola (animals) |
(Johnson et al., 1995) |
|
|
Lassavirus (animals) |
(Stephenson et al., 1984) |
|
|
Smallpox |
(Wehrle et al., 1970) |
|
|
Protozoans |
|
|
|
|
Giardia (cysts) |
(Rose et al., 1991) |
|
Cryptosporidium (oocysts) |
(Haas et al., 1996; Messner et al., 2001; Teunis et al., 2002) |
|
In the case of the exponential model, the survival probability is constant among all microorganisms. For the beta-Poisson model, the survival probabilities vary according to a beta probability distribution. The survival probability (in the case of the exponential) or the survival probability distribution (in the case of the beta-Poisson) accounts for all factors, including host immunity, that act as forces to reduce the ability of a retained organism to successfully colonize to the extent necessary to induce infection or disease. Both models predict that the risk at low doses is a linear function of dose. In several situations, outbreak data have been consistent with risks extrapolated from human volunteer trials (Crockett et al., 1996; Haas and Rose, 1994; Rose et al., 1991).
Dose–response modeling has been used extensively to posit the risk associated with ingestion (Cassin et al., 1998; Crockett et al., 1996; Gale et al., 1998; Haas et al., 1996; Medema et al., 1996) and dermal exposures (Gibson et al., 1999). Risks from inhalation have been modeled for bioaerosol emissions from sludge disposal operations (Dowd et al., 2000). A somewhat parallel model has been developed in the industrial hygiene literature in which an exponential model
is used to assess risk (Riley and O’Grady, 1961). Nicas (1996) modified that model to incorporate interhost variability, which effectively produces a set of models similar to the beta-Poisson model.
Animal Models
Although dose–response assessments have been developed for human subjects, such data are unlikely to be widely applicable. Some pathogens are so dangerous (for example, hepatitis A virus) that ethics rules prohibit their use in dose–response studies with human subjects. The effect of susceptibility on outcome would be difficult to ascertain from animal data alone—again because of ethical rules for the use of human subjects. Hence, the use of animal data for developing dose–response relationships and for ascertaining the influences of various modifying factors on outcome is clearly desirable. Yet there are no good animal models for some of the Category A organisms.
In the regulatory toxicology of chemical agents, animal model data are frequently relied on as a source of dose–response, or potency, information. The principles for the extrapolation to humans of results obtained in animal studies are well developed for chemical carcinogens (U.S. Interagency Staff Group on Carcinogenesis, 1986). However, in studying exposure to biological agents, scientists have less experience extrapolating from animal models to humans. A few studies have been done, but it is not necessarily straightforward to extrapolate from those examples to other agents.
Two studies have revealed promise for the use of animal dose–response data to develop human dose–response information (Haas et al., 1999b; 2000). The studies involve Listeria monocytogenes and Escherichia coli O157:H7. The disease rate during several human outbreaks examined (for those organisms) was consistent with the estimated exposure and with the use of the animal dose-response information without interspecies correction factors. The amount of information would be likely to expand as additional animal trials and validations are performed.
In the case of inhalation of Bacillus anthracis spores, primate data are consistent with an exponential dose–response relationship (Haas, 2002). Data appear to be available to perform dose–response analyses for other agents of concern, and such analyses should be done to develop dose–response relationships.
Relative Susceptibility of Different Subpopulations in Risk Analysis
As a matter of public policy, the guidelines for safe reoccupation of facilities contaminated with harmful biological agents should protect not only the general population but also sensitive or susceptible subpopulations. However, what constitutes more susceptible has not been rigorously defined. In 2000, a working group (Balbus et al., 2000) gave the following definition:
Susceptibility is a capacity characterizable by a set of intrinsic and extrinsic factors that modify the impacts of a specific exposure upon risks/severity of outcomes in an individual or population.
By that definition, susceptible subpopulations could include the immunocompromised (including HIV-infected people and patients in treatment with immunosuppresive drugs), pregnant women, the elderly, and children (Gerba et al., 1996). In addition, it also could include persons with limited access to health care or with concomitant factors, such as diet, tobacco use, or use of illicit drugs, which could enhance their risk or susceptibility to infection.
However, there is no validated way to incorporate altered susceptibility into a risk assessment for infectious microorganisms. That will require the use of animal models to assess dose–response shifts associated with various modifiers to susceptibility. There could remain unresolvable uncertainties about which of the particular modifiers might be most appropriate, especially where good animal models are lacking for an agent.
“Thresholds” for Microbial Dose–Response Analysis
In assessing risks attributable to exposure to microorganisms, it has frequently been asserted that there exists a threshold (minimum infectious dose) below which there is no risk to a population. Such a concept is not consistent with the current understanding of microbial risk assessment, and the issue is addressed directly in this section. The no-risk concept originated from the fact that in trials (either animal or human), low doses of microorganisms often produced no adverse effects in exposed subjects.
Several terms should be precisely defined. A population exposed to microorganisms—for example, by inhaling contaminated air—is exposed to a dose for which an average can be calculated. For example, people who pass through a contaminated airport might be exposed to an average of 0.15 organisms (of a particular type) during their transit. However, microorganisms are individual particles at low number density1 so that “average dose” means that some proportion of the population, perhaps most of it, would be exposed to zero organisms, and some proportion of the population would be exposed to 1, 2, or higher numbers of organisms.
The relationship between the average dose to a population and the proportion of that population that exhibits a particular adverse effect is called the dose–response relationship. The relationship between the actual number of organisms
to which a subpopulation is exposed and the proportion of people in that exposed population to the actual number of people who experience the adverse effect is called the conditional dose-response relationship (Haas et al., 2002).
For in vitro quantification of organisms—such as by colony or plaque counting, most probable number, or TCID50 (the dose that produces infection in 50% of the tissue culture samples exposed) methods—the underlying assumption for data interpretation is that a single microorganism is sufficient to generate a colony, plaque, or infected tube or lawn of cells. In other words, it is not necessary for more than one organism to be present for such infection to occur. However, it is recognized that the probability that any organism will actually initiate such an infection is not 100%. In specific terms, the conditional dose–response relationship is nonzero for an actual exposure to one organism in vitro. The same set of assumptions underlies the dose–response relationship used for assessing risk attributable to animal or human exposure to pathogens. Therefore, long-standing observations on microbial behavior have supported the use of population dose-response models without a threshold; that is, a dose below which no risk to a population exists (or a nonzero intercept on the dose scale).
All animal and human exposure data that have been subjected to dose-response analysis are consistent with models in which the dose intercept is zero and the value of the conditional dose–response relationship for one organism is non-zero. Hence, the concept of “minimum infectious dose” is consistent with the data, and there are no data for which a “threshold” is necessary (in the form of a nonzero intercept on the dose scale).
In animal and human trials, experiments are conducted with a small number of subjects. There are many reasons this is necessary, but one consequence is that it is harder to make robust conclusions about what would happen if larger numbers of subjects were exposed to a particular pathogen. For example, in a case where 10 nonhuman primates are studied it could appear that the pathogen has no effect (no positive reactions). However, if the same study were done using 100 nonhuman primates, several animals might exhibit an effect. That concept is shown in Figure 8-2. The probability of finding no affected subjects out of 10 as a function of the true proportion of positives in the population (the population risk) is shown. If the population risk were 0.1, there would still be a probability of 0.3 of finding zero positive subjects among 10. Hence, it would not be prudent to assert that a finding of no positives in a small sample would show that there is minimal risk in a large population. That concept explains how there could be situations in which it appears that there is a threshold dose below which there is no risk of exposure, when in fact there are not sufficient data available to conclusively establish that threshold.
Studies on the infection of laboratory animals with several infectious agents, as reviewed by Rubin (1987), provide strong experimental support for a nonthreshold methodology. The potential variations in agents of the same species and among potential human hosts (in immune status, for example) strongly sug-
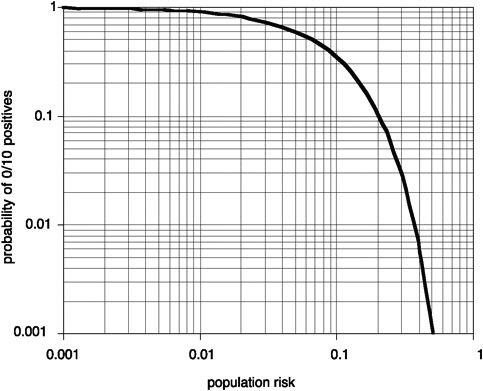
FIGURE 8-2 Relationship between the population risk and the probability of getting no positive subjects among 10 exposed (based on binomial probability).
gest that the limited information currently available should be interpreted cautiously. Although the nonthreshold model implies that there is no threshold below which an agent poses zero risk, it indicates that the probability of infection is extremely low.
In contrast, a threshold model implies a definitive threshold below which no infection would occur. So the nonthreshold model is a more cautious and more appropriate approach than is the threshold model in some circumstances. Therefore, the committee believes that prudent public health protection requires the continued application of nonthreshold approaches to the assessment of microbial dose response—that is, it is not possible to calculate a threshold for environmental contamination with B. anthracis spores (or other pathogens or toxins) below which there would be zero risk of disease. Microbial dose–response models can be divided into the categories of mechanistic and the empirical. Mechanistic models have been derived by considering the individual particle nature of microorganisms and the variability induced by small numbers that can be present in an individual exposure (for example, as noted in Figure 8-1).
The exponential and beta-Poisson models are examples of the class of mechanistic models that are derived by assuming that one organism, if it survives to colonize, is sufficient to initiate the infection and disease process. Alternative models can be derived on the basis that more than one organism is required to survive to colonize. For example, if the same assumptions as the exponential (identical and independent survival probabilities, random distribution of organisms) are used, except that the number of surviving organisms that successfully colonize must be at least kmin (>1), then the dose–response model becomes (Haas et al., 1999a)
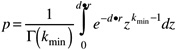
where d is the average dose, r is the individual survival probability, and p is the risk attributable to the exposure. This equation is also that of the gamma probability distribution. For all such “threshold” models (with kmin>1), the slope of the dose-response relationship at the median (ID50, LD50) is steeper than it is in the non-threshold models. The available data on infectious organisms are inconsistent with dose–response relationships that have steeper slopes than the nonthreshold models at the median, and therefore the concept of a threshold is not supported by available data.
Determination of Cleanup
The determination of the amount of cleanup required, and the processes used to achieve it, must consider both quantitative (risk assessment) and qualitative (stakeholder input) factors. Risk assessment can be used to inform the decision-making process in the specific context of cleanup of a facility after an act of bioterrorism.
The process can be described by a series of graphs. Figure 8-3 illustrates a relationship between the environmental concentration in a specific medium (such as surface and air) and the exposure of a specific population (such as workers, commuters, transient populations). Point A on Figure 8-3 represents the environmental concentration measured before decontamination (for example, organisms m-3 in air, or organisms m-2 on surfaces) on the x-axis, and the exposure (number of organisms) on the y-axis. The relationship must be developed by modeling exposure patterns and transport processes for potentially exposed individuals.
Point B of Figure 8-3 represents the environmental concentration and resulting exposure that can result from remediation by a specific decontamination strategy. The relationship can be nonlinear. By relating the resulting exposure after decontamination B to the dose–response relationship, the residual risk remaining after decontamination can be estimated. This is shown in Figure 8-4.
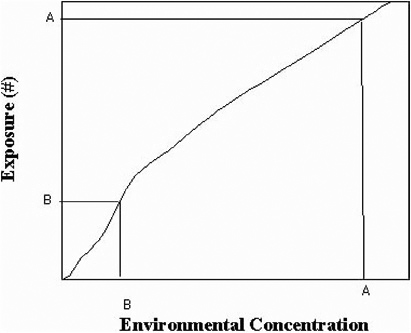
FIGURE 8-3 Relationship between environmental concentration versus exposure.
Therefore, the reduction in the environmental concentration (from A to B in Figure 8-3) results in a reduction in risk from A to B in Figure 8-4.
Figures 8-3 and 8-4 can be ascertained only with some level of uncertainty and variability. A partial list of sources of uncertainty and variability is given in Table 8-2. The uncertainties and variability would lead to the development of risk assessments in which confidence limits (or distributions) for each of the inputs are used to derive a confidence limit (or distribution) for the risk and/or the use of an empirical “safety factor.”
Figures 8-3 and 8-4 can be used by a risk manager to assess the risk attributable to reducing environmental concentration to a target amount (going from Figure 8-3 to Figure 8-4). Alternatively, if a target risk is specified, the resulting dose can be computed via Figure 8-4 and the resulting environmental concentration can be computed from Figure 8-3. An illustration of the latter procedure is given in Box 8-1.
Close examination shows that there are several numerical inputs in Box 8-1: dose–response parameter, breathing rate, duration, transference ratio. Each has a specific uncertainty and variability, so the final estimate of surface and air concentrations corresponding to the risk level (or, conversely, the risk resulting from a given set of surface and air concentrations) itself has a certain confidence distribution.
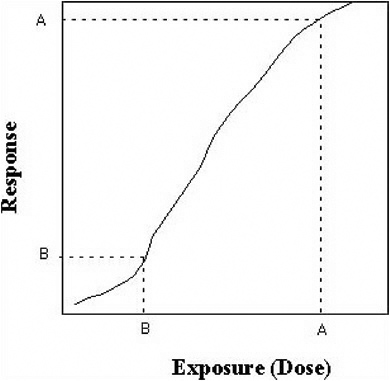
FIGURE 8-4 Schematic exposure (dose) response.
Appendix E presents a detailed probabilistic application of risk analysis in which the propagation of uncertainty and variability is set forth. For the American Media, Inc., building in Boca Raton, Florida, the example also shows that the computation of risk from inhalation of B. anthracis provides an estimate of disease impact that is consistent with the observed number of cases. The example in Appendix E shows that risk assessment and epidemiology are complementary pursuits, and that the results of epidemiological investigation can (with information on exposure) help support the computations of a risk assessment.
FINDINGS AND RECOMMENDATIONS
Finding 8-1
The concept of a “threshold” below which no risk to a population exists for a microbial dose response is not supported by currently available data. Nonthreshold dose–response models present a more cautious approach that has been found appropriate for describing human response to a diversity of infectious agents via ingestion, inhalation, and other routes of exposure. Dose–response data for most of the pathogens of concern (biological agents) are incomplete or have not been critically analyzed in the open literature.
TABLE 8-2 Some of the Uncertain and Variable Factors in Risk Assessment
|
|
Uncertainty |
Variability |
|
Concentration/exposure relationship |
|
|
|
Exposure/response relationship |
|
|
|
an |
|
|
BOX 8-1 The risk resulting from contamination of a room with spores of Bacillus anthracis must be assessed. The concentration in the air comes from spores that are resuspended from a surface. Surface concentrations also are measured. The following is assumed:
|
Recommendation 8-1
Available dose–response data for pathogens of concern should be analyzed by nonthreshold dose–response models.
Finding 8-2
Because minimal publicly available data exist on which to base human dose–response relationships for the critical pathogens, animal data must be used. How-
ever, our understanding of interspecies extrapolation of dose–response relationships for infectious agents from animals to humans is low.
Recommendation 8-2
Targeted research to help inform decision making on extrapolation of dose–response data between species for the pathogens of concern should be conducted. That research might use several species of organisms or use animal and human tissues to reach conclusions that are relevant for human exposures. With the increasing difficulty of performing primate studies, it will become more important to develop in vitro techniques that can be used to develop dose-response information.
REFERENCES
Balbus, J., R. Parkin, and M. Embrey. 2000. Susceptibility in microbial risk assessment: definitions and research needs. Environmental Health Perspectives 108(9): 901-905.
Cassin, M.H., A.M. Lammerding, E.C.D. Todd, W. Ross, and R.S. McColl. 1998. Quantitative risk assessment for Escherichia coli O157:H7 in ground beef hamburgers. International Journal of Food Microbiology 41: 21-44.
Crockett, C.S., C.N. Haas, A. Fazil, J.B. Rose, and C.P. Gerba. 1996. Prevalence of shigellosis in the U.S.: consistency with dose-response information. International Journal of Food Microbiology 30(1-2): 87-100.
Dowd, S.E., C.P. Gerba, I.L. Pepper, and S.D. Pillai. 2000. Airborne pathogens in association with biosolids: mathematical modeling and risk assessment. Journal of Environmental Quality 29: 343-348.
Druett, H.A., D.W. Henderson, L. Packman, and S. Peacock. 1953. Studies on respiratory infection. I. The influence of particle size on respiratory infection with anthrax spores. Journal of Hygiene (London) 51(3): 359-371.
Fazil, A.M. 1996. A Quantitative Risk Assessment Model for Salmonella. Philadelphia, Pennsylvania: Environmental Studies Institute, Drexel University.
Gale, P., C. Young, and D. Oakes. 1998. A review: development of a risk assessment for BSE in the aquatic environment. Journal of Applied Microbiology 84(4): 467-477.
Gerba, C.P., J.B. Rose, C.N. Haas, and K.D. Crabtree. 1996. Waterborne rotavirus: a risk assessment. Water Research 30(12): 2929-2940.
Gibson, L.L., J.B. Rose, and C.N. Haas. 1999. Use of quantitative microbial risk assessment for evaluation of the benefits of laundry sanitation. American Journal of Infection Control 27(6): S34-39.
Haas, C.N. 1986. Wastewater disinfection and infectious disease risks. CRC Critical Reviews in Environmental Control 17(1): 1-20.
Haas, C.N. 2002. On the risk of mortality to primates exposed to anthrax spores. Risk Analysis 22(2): 189-193.
Haas, C.N., and J.B. Rose. 1994. Reconciliation of Microbial Risk Models and Outbreak Epidemiology: The Case of the Milwaukee Outbreak. Annual Conference—American Water Works Association, New York.
Haas, C.N., J.B. Rose, C.P. Gerba, and S. Regli. 1993. Risk assessment of virus in drinking water. Risk Analysis 13(5): 545-552.
Haas, C.N., C.S. Crockett, J.B. Rose, C.P. Gerba, and A. Fazil. 1996. Infectivity of Cryptosporidium parvum oocysts. Journal of the American Water Works Association 88(9): 131-136.
Haas, C.N., J.B. Rose, and C.P. Gerba. 1999a. Quantitative Microbial Risk Assessment. New York: John Wiley.
Haas, C.N., A. Thayyar-Madabusi, J.B. Rose, and C.P. Gerba. 1999b. Development and validation of dose-response relationship for Listeria monocytogenes. Quantitative Microbiology 1(1): 89-102.
Haas, C.N., A. Thayyar-Madabusi, J.B. Rose, and C.P. Gerba. 2000. Development of a dose-response relationship for Escherichia coli O157:H7. International Journal of Food Microbiology 56(2-3): 153-159.
Havelaar, A.H., J. Garssen, et al. (2000). Dose Response Relationships for Gastrointestinal Pathogens in an Animal Model. Bilthoven, NL, RIVM.
Holcomb, D.L., M.A. Smith, G.O. Ware, Y.C. Hung, R.E. Brackett, and M.P. Doyle. 1999. Comparison of six dose-response models for use with food-borne pathogens. Risk Analysis 19(6): 1091-1100.
Johnson, E., N. Jaax, J. White, and P. Jahrling. 1995. Lethal experimental infections of rhesus monkeys by aerosolized ebola virus. International Journal of Experimental Pathology 76: 227-236.
Medema, G.J., P.F.M. Teunis, A.H. Havelaar, and C.N. Haas. 1996. Assessment of the dose-response relationship of Campylobacter jejuni. International Journal of Food Microbiology 30(1-2): 101-112.
Messner, M.J., C.L. Chappell, and P.C. Okhuysen. 2001. Risk assessment for Cryptosporidium: a hierarchical Bayesian analysis of human dose-response data. Water Research 35(16): 3934-3940.
Nicas, M. 1996. An analytical framework for relating dose, risk and incidence: an application to occupational tuberculosis infection. Risk Analysis 16(4): 527-538.
Oyston, P.C.F., A. Sjostad, and R.W. Titball. 2004. Tularemia: bioterrorism defense renews interest in Franciscella tularensis. Nature Reviews Microbiology 2: 967-978.
Riley, R.L., and F. O’Grady. 1961. Airborne Infection: Transmission and Control. New York: Macmillan.
Rose, J.B., C.N. Haas, and S. Regli. 1991. Risk assessment and the control of waterborne giardiasis. American Journal of Public Health 81: 709-713.
Rubin, L.G. 1987. Bacterial colonization and infection resulting from multiplication of a single organism. Reviews of Infectious Diseases 9(1): 488-493.
Stephenson, E.H., E.W. Larson, and J.W. Dominik. 1984. Effect of environmental factors on aerosol-induced Lassa virus infection. Journal of Medical Virology 14: 295-303.
Teunis, P.F.M., C.L. Chappell, and P.C. Okhuysen. 2002. Cryptosporidium dose response studies: variation between hosts. Risk Analysis 22(3): 475-485.
U.S. Interagency Staff Group on Carcinogenesis. 1986. Chemical carcinogens: a review of the science and its associated principles. Environmental Health Perspectives 67: 201-282.
Wells, W.F. 1955. Airborne Contagion and Air Hygiene. Cambridge, MA: Harvard University Press.
Wherle, P.F., J.J. Posch, K.H. Richter, and D.A. Henderson. 1970. An airborne outbreak of smallpox in a German hospital and its significance with respect to other recent outbreaks. Bulletin World Health Organization 43: 2230-2251.















