4
Defining Resource-Level-Appropriate Cancer Control
Interventions for cancer, from prevention through palliative care, have been developed largely in high-income countries. In these countries, where cancer is often referred to as “the most feared disease,” the emphasis has always been on maximizing effectiveness and safety. Cost—in terms of money and of other inputs—has been a secondary concern, and other factors that would affect the appropriateness or feasibility of applying an intervention in resource-constrained settings have been considered very little. Specifically, a health care infrastructure sufficient to cover the population, from primary through tertiary care, is assumed. An educated public, reached by at least some basic information about cancer, and access to health care and good nutrition, is taken for granted. Finally, in most high-income countries health care is free or affordable for all or most. Developing cancer control in places where conditions are much different means that all these factors, as well as societal factors, must be considered in deciding on the best approaches. Explicit analysis of effectiveness and costs of alternative approaches and interventions may also help to counter the natural attraction of the medical community and politicians, in low- and middle-income countries (LMCs), as well as in high-income countries, to the newest, high-technology (and expensive) interventions. This is the basic idea behind “resource-level-appropriate” cancer control.
The World Health Organization (WHO), in its 1992 report on guidelines and policies for National Cancer Control Programmes, gave prominence to the idea of tailoring interventions to three scenarios: low, medium, and high resource levels, in a very general sense (WHO, 2002) (Table 4-1). The underlying concepts were enunciated, but not the “how to.” The idea
TABLE 4-1 Priority Actions for National Cancer Control Programs, According to Level of Resources
|
Aspect |
All Countries |
Scenario A: Low Level of Resources |
Scenario B: Medium Level of Resources |
Scenario C: High Level of Resources |
|
National cancer control program |
|
|
|
|
|
Aspect |
All Countries |
Scenario A: Low Level of Resources |
Scenario B: Medium Level of Resources |
Scenario C: High Level of Resources |
|
Prevention |
|
|
|
|
|
Early diagnosis |
|
|
|
|
|
Screening |
|
|
|
|
|
Aspect |
All Countries |
Scenario A: Low Level of Resources |
Scenario B: Medium Level of Resources |
Scenario C: High Level of Resources |
|
Curative therapy |
|
|
|
|
|
Pain relief and palliative care |
|
|
|
|
|
SOURCE: Reprinted, with permission, from WHO (2002). Copyright 2002 by WHO. |
||||
of resource-level-appropriate thinking is intuitively simple, but filling in the details requires systematic analyses of varying complexity. How do policy makers know what specific approach or intervention is likely to succeed under their own circumstances? What is a “comprehensive approach”? What are “necessary resources”? What level of incidence justifies a prevention or early detection program? What WHO laid out was actually an agenda for operations research to develop the information needed to guide complex decisions at the national and local levels in low- and middle-income countries (LMCs), but not the information itself. This agenda is now beginning to be addressed, but it requires significantly more effort.
This chapter discusses the means of generating the resource-level-specific information and guidance needed to put this notion into practice. This is not to suggest that decisions and policy cannot be made and actions taken today. Although the concept of resource appropriateness applies to the entire spectrum of cancer control interventions (as well as to research toward defining and testing resource-level-appropriate interventions), some measures may be appropriate and effective everywhere, regardless of national economic status, and may not require a great deal of resource-level analysis. Taxes on cigarettes and other tobacco products have worked to reduce tobacco use in a wide range of countries, including some LMCs, and every country has the ability to levy taxes. Specific tax laws will vary as will the level of tobacco sales and the types of tobacco smoked or consumed in other ways, which may dictate the details of tax programs, but these must be worked out at the country level. For the types of interventions where this may not be the case, however, there exists a need to develop an evidence-based consensus on resource-level-appropriate interventions and make it readily available to decision makers at all levels in LMCs.
The aim is for countries and individual institutions to make the best decisions for their individual patients and for the populations they serve, to maximize the value of existing inputs, and to create a context for incremental investment. The example of how this objective may begin to be achieved (described in detail in this chapter) is the Breast Health Global Initiative (BHGI), an ongoing international collaboration that has taken the first steps to produce detailed, resource-level-specific guidelines for all relevant aspects of breast cancer, from early detection through palliative care. The next phase of the BHGI, working with LMC partners to develop experience in adapting and applying the guidelines, is just beginning. This involves several layers of decision making, from policy and programmatic decisions that could be taken at the national or subnational level, to the level of the institution determining the mix of services it should offer. Still farther down the line is the challenge of persuading physicians and other health care workers to follow guidelines that have been adopted, and to develop systems that encourage this and that monitor their use and patient
outcomes. The guidelines themselves are a necessary tool in the process, although not an end in themselves.
CONSIDERATIONS FOR RESOURCE-LEVEL APPROPRIATENESS
A number of factors may contribute to defining which interventions are most appropriate in different settings, with cost being important, but not the only consideration. The point should also be made that what is most appropriate is likely to vary within countries and among countries of similar economic status. In particular, the same services may not be available in major urban areas and in rural areas. In some cases, a single option could be best for every low- or middle-income country, but this cannot be assumed. Even where there are choices to be made, however, the number of options in most cases is likely to be limited, making decisions manageable.
Basic factors that may affect decisions about appropriate services include:
-
The monetary cost of the intervention
-
To patients
-
To governments or other payers
-
-
Characteristics of patients and their cancers presenting for treatment
-
Prevalent cancer types
-
Stage distribution of cancers
-
Common co-morbidities and nutritional status
-
Availability of social support for patients during and following treatment
-
-
Characteristics of effective interventions
-
Time course, including total inpatient and outpatient requirements, and follow-up
-
Acute toxicity
-
Long-term effects, including permanent disability, disfigurement, and effect on quality of life
-
Need for and availability of rehabilitation
-
-
Institutional requirements
-
General infrastructure
-
Specific equipment and drugs
-
Infection control measures, including isolation facilities
-
Medical, nursing, technician, and psychosocial personnel needs
-
This chapter draws on the BHGI experience specifically related to guideline development which, at this writing, remains the only available model. However, practitioners in LMCs and those who work with them do implicitly or explicitly weigh alternatives and make “resource-based” deci-
sions in their daily practices. The idea surfaces as well in particular projects and reports. Comprehensive Cervical Cancer Control: A Guide to Essential Practice (WHO, 2006), developed by WHO, is a current example. It is presented as a “how-to” manual for cervical cancer, aimed at LMCs in terms of the range of technologies addressed. It does not simply present the best practices of high-income countries as the only effective approaches, and it recognizes that all possible resources are not available everywhere.
That said, very little of the published evidence base is derived from LMCs. Few clinical trials of cancer interventions have taken place in these countries. The point has been made already that conditions regarding the patient and the environment may be significantly different in LMCs. Thus there is also a need for resource-level-appropriate research on questions of particular importance in LMCs. These could be trials of treatments already in use, to confirm their effectiveness and safety in different populations and under different conditions; questions that are largely relevant only to LMCs (e.g., treatment of Burkitt’s lymphoma or advanced retinoblastoma, both in children); or modified treatment protocols (e.g., “resource-sparing” protocols for radiotherapy) for common cancers.
THE BREAST HEALTH GLOBAL INITIATIVE: A BLUEPRINT FOR DEVELOPING RESOURCE-LEVEL-APPROPRIATE INTERVENTIONS
The BHGI has developed evidence-based, culturally appropriate sets of guidelines that can be used in countries with limited resources—low- and middle-income countries—to improve breast health outcomes. The program is ongoing, co-sponsored by the Fred Hutchinson Cancer Research Center and the Susan G. Komen Breast Cancer Foundation, in collaboration with a number of national and international health organizations, breast health and cancer societies, and nongovernmental organizations (NGOs) (Box 4-1). The BHGI is the brainchild of Dr. Benjamin Anderson, a breast surgeon at the Fred Hutchinson Cancer Center in Seattle, who has led the effort since the beginning.
The first BHGI Global Summit Consensus Conference on International Breast Health Care was held in October 2002 in Seattle (the conference is hereafter referred to as the 2002 Global Summit). The aim of the 2002 Global Summit was to establish breast health guidelines for countries where health care resources are significantly limited (Anderson, 2003). The guidelines were developed using a panel consensus approach with analysis of evidence-based breast cancer research. The panel consisted of breast cancer experts, scientists, and patient advocates from 17 countries and 9 world regions. They were provided with materials prior to the meeting describing the goals of the project as well as literature related to guideline development. Selected panelists prepared presentations relevant to breast
|
BOX 4-1 BHGI Collaborating Organizations American Society for Breast Disease Breast Surgery International Centers for Disease Control and Prevention International Atomic Energy Agency of the United Nations International Network for Cancer Treatment and Research International Society for Nurses in Cancer Care International Society of Breast Pathology International Union Against Cancer Middle East Cancer Consortium National Cancer Institute, Office of International Affairs Pan American Health Organization WHO Programs:
World Society for Breast Health SOURCE: Anderson et al. (2005). |
cancer care in countries with limited health care resources. At the meeting, three panels were formed, on Early Detection, Diagnosis, and Treatment. Each panel was asked to define guidelines for care in their assigned area, using WHO defined criteria for “low-level” and “medium-level” resource countries (WHO, 2002). The resulting BHGI guidelines were published and have been made available in an unrestricted fashion on the Internet for worldwide access (Anderson et al., 2003a; Anderson et al., 2003b; Carlson et al., 2003; Vargas et al., 2003).
The first summit and development of guidelines was a learning process. The next sections of this chapter discuss issues that were important in creating an appropriate context for the guideline effort, and then describe the refined and improved procedures, used in the second summit and to revise the guidelines.
GUIDELINE DEVELOPMENT FOR LIMITED-RESOURCE SETTINGS: SPECIAL CONSIDERATIONS REGARDING BREAST CANCER
To be applicable and effective, practice guidelines must go beyond summarizing the available evidence and prescribing interventions using strictly
quantitative criteria. Social norms and values cannot be ignored in the way practice questions are framed and outcomes chosen, and these may differ among health care systems (Redman, 1996). In the case of breast cancer, gender inequalities that exist between men and women in many societies carry over into health care disparities. Particularly where resources are limited, women may bear more than their share of deprivation in the extent and quality of health services available to them (Gijsbers van Wijk et al., 1996). At the 2002 Global Summit, two axioms were adopted as principles for guideline development:
-
All women have the right to access health care, but considerable challenges exist in implementing breast health care programs when resources are limited.
-
All women have the right to education about breast cancer, but it must be culturally appropriate, and targeted and tailored to the specific population.
In countries with limited resources, most women have advanced or metastatic breast cancer at the time of diagnosis (Pal and Mittal, 2004). Based on evidence-based review and consensus discussion, four observations were made:
-
The more advanced breast cancer is at diagnosis, the poorer the survival and the more resource intensive it is to treat. Efforts to increase early detection can reduce the stage at diagnosis, potentially improving the odds of survival and cure, and enabling simpler and more effective (and more cost-effective) treatment. These efforts are likely to have the greatest overall benefit in terms of both survival and efficient utilization of available resources.
-
Each country must build programs that fit its unique situation.
-
In the low-income countries where it is not yet possible to deliver breast cancer care to women nationwide, the development of cancer centers can be a stepping stone to providing high-quality care to at least some women.
-
Collecting data on breast cancer is imperative for deciding how best to apply resources and for measuring improvements in outcome following programmatic changes.
These observations from the first Global Summit served as the basis of the 2005 BHGI Global Summit Consensus Conference on International Breast Health Care (hereafter referred to as the 2005 Global Summit), where specific recommendations were addressed.
The 2005 Global Summit
The BHGI guidelines were reexamined, revised, and extended at the second Global Summit, held January 12–15, 2005, hosted by the National Cancer Institute in Bethesda, Maryland. Twelve national and international groups and three WHO programs collaborated (Box 4-1). More than 60 experts from 33 countries participated. They represented expertise in screening, pathology and cytology, surgery, medical oncology, radiation oncology, health economics, surveillance, medical ethics, sociology, and advocacy. Participants were organized into four panels:
-
Early Detection and Access to Care
-
Diagnosis and Pathology
-
Cancer Treatment and Allocation of Resources
-
Health Care Systems and Public Policy
Each panel drafted a consensus manuscript summarizing their deliberations and decisions (Anderson et al., 2006a; Eniu et al., 2006; Shyyan et al., 2006; Smith et al., 2006).
Panel Organization and Conference Preparation
Panel co-chairs organized speakers to cover their panels’ topics and drafted the consensus manuscript. Each panel held one full-day meeting for all summit participants, split between plenary sessions and discussion and debate to achieve consensus. Each day began with a presentation by a breast cancer advocate from a limited-resource country sharing the personal experience of facing breast cancer in that country.
Consensus Process
Each panel was asked to stratify the health care interventions relevant to their areas into four levels:
-
“Basic” level—Core resources absolutely necessary for any breast health care system to function. Basic-level resources are typically applied in a single clinical interaction.
-
“Limited” level—Second-tier resources that produce major improvements in outcome. Limited-level resources may involve single or multiple clinical interactions.
-
“Enhanced” level—Third-tier resources that are optional, but important. Enhanced-level resources may produce minor improvements in outcome, but increase the number of therapeutic options and patient choices.
-
“Maximal” level—Resources applied in a modern breast health care practice in countries or settings with high-level resources.
This stratification scheme assumes incremental resource allocation; for example, the limited level assumes that a setting already has all of the resources recommended for the basic level. All interventions available in the basic level are assumed still to be available for use as appropriate at higher levels, and this pattern of building up continues up through the maximal level. Using this scheme, the short-term goal is to move to the next level, and the long-term goal is to move to the enhanced or maximal levels. Of note, multiple resource levels generally co-exist within a country, a region, or even an individual health care facility. For example, a country may have community clinics that provide care at the basic level, regional hospitals that provide care at the limited level, and a national cancer center that provides care at the enhanced or maximal level. Because circumstances vary so widely around the world, decisions about how to plan the overall structure of a national breast program must be made on a country-by-country, region-by-region, and facility-by-facility basis.
Panels were also asked to develop checklists organized by country resource level, with the intention of creating a series of checklists or tables. These checklists describe the strengths, limitations, and necessary resources to apply a given approach in the areas of early detection, diagnosis, treatment, or health care systems and policies. Finally, the panels were asked to identify areas where evidence is lacking and research is needed to better inform future versions of the guidelines.
Manuscript Preparation and Review
Much of the discussion within panels involved creating the tables (Tables 4-2 through 4-8) that stratify interventions according to the four levels. The consensus draft manuscripts were compared centrally for internal consistency in stratification of interventions by a subpanel of co-authors. Differences among recommendations from the panels were reviewed with panel co-chairs and adapted to minimize inconsistencies, when this was possible. However, there were cases of irreconcilable differences, where interventions were definitively stratified in different ways by different panels. In these cases, the panel recommendations were maintained in the tables and the nature of the disagreements was summarized, explained, and discussed in an overview manuscript (Anderson et al., 2006a).
In addition to panel consensus papers, plenary speakers were invited to submit individual manuscripts for publication together with the consensus manuscripts. These papers are more detailed, and on more focused topics, than could be included in the consensus manuscripts, but were vital to an
TABLE 4-2 Early Detection and Access to Care Guidelines
|
Level of Resources |
Detection Method(s) |
Evaluation Goal |
|
Basic |
Breast health awareness (education ± self-examination) Clinical breast examination (CBE) (clinician education) |
Baseline assessment and repeated resurvey |
|
Limited |
Targeted outreach/education encouraging CBE for at-risk groups Diagnostic ultrasound ± diagnostic mammography |
Downstaging of symptomatic disease |
|
Enhanced |
Diagnostic mammography Opportunistic mammographic screening |
Opportunistic screening of asymptomatic women |
|
Maximal |
Population-based mammographic screening Other imaging technologies as appropriate: High-risk groups, unique imaging challenges |
Population-based screening of asymptomatic women |
|
SOURCE: Reprinted, by permission, from Anderson et al. (2006a). Copyright 2006 by the Breast Health Global Initiative, Fred Hutchinson Cancer Research Center. |
||
overall understanding of the guideline recommendations. In the end, some manuscripts on topics of more general interest were referred for journal submission outside the BHGI process. Some were incorporated into guideline consensus articles. Manuscripts with specific merit in support of the guidelines were accepted for publication along with the consensus documents. The combination of consensus and individual manuscripts represents the complete BHGI guideline compendium, which is the final work product of the 2005 Global Summit. It was published in its entirety as a supplement to the January/February 2006 edition of The Breast Journal.
2005 Global Summit Guideline Outcome Summary
The cumulative work of the four panels results in a matrix guideline spanning the spectrum of breast health care from early detection to treatment and palliation of advanced disease, and considers the full spectrum of available resources (Tables 4-3–4-7). A matrix for health care systems and public policy was also developed (Table 4-8).
In most areas, there was good alignment and agreement between consensus panels in the assigned stratification levels. Furthermore, the stratification from basic to maximal levels generally mirrors the evolution of breast cancer diagnosis and treatment that has developed in high-income countries, with a few exceptions, which are described below.
TABLE 4-3 Diagnosis and Pathology Guidelines
|
Level of Resources |
Clinical |
Pathology |
Imaging and Lab Tests |
|
Basic |
History Physical examination Clinical breast examination Surgical biopsy Fine-needle aspiration biopsy |
Interpretation of biopsies Cytology or pathology report describing tumor size, lymph node status, histologic type, tumor grade |
|
|
Limited |
Core needle biopsy Image-guided sampling (ultrasonographic ± mammographic) |
Determination and reporting of ER and PR status |
Diagnostic breast ultrasound ± diagnostic mammography Plain chest radiography Liver ultrasound Blood chemistry profile/CBC |
|
Enhanced |
Preoperative needle localization under mammographic or ultrasound guidance |
Onsite cytopathologist |
Diagnostic mammography Bone scan |
|
Maximal |
Stereotactic biopsy Sentinel node biopsy |
HER-2/neu status IHC staining of sentinel nodes for cytokeratin to detect micrometastases |
CT scan, PET scan, MIBI scan, breast MRI |
|
CBC = complete blood count; CT = computed tomography; ER = estrogen receptor; IHC = immunohistochemistry; MIBI = 99mTc-sestamibi; MRI = magnetic resonance imaging; PET = positron emission tomography; PR = progesterone receptor. SOURCE: Reprinted, by permission, from Anderson et al. (2006a). Copyright 2006 by the Breast Health Global Initiative, Fred Hutchinson Cancer Research Center. |
|||
Introduction of Breast Ultrasound and Diagnostic Mammography in Low-Income Countries
Ideally, diagnostic mammography is a core resource for work-up for all types of breast lesions (NCCN, 2005), and ultrasound is used to examine localized findings from the diagnostic mammogram or clinical breast examination (CBE). Screening breast ultrasound (general survey of the whole breast in clinically asymptomatic women) is generally discouraged because it has a high false-positive rate if strict criteria are followed (Stavros et al., 1995).
In low-resource settings, however, diagnostic ultrasound usually becomes available before diagnostic mammography, for understandable reasons. First, mammography is a highly specialized imaging tool that is con-
TABLE 4-4 Treatment and Allocation of Resources: Stage I Breast Cancer Guidelines
|
Level of Resources |
Local-Regional Treatment |
Systemic Treatment (Adjuvant) |
||
|
Surgery |
Radiation Therapy |
Chemotherapy |
Endocrine Therapy |
|
|
Basic |
Modified radical mastectomy |
|
|
Ovarian ablation |
|
Limited |
Breast-conserving therapya |
Breast-conserving whole-breast irradiation as part of breast-conserving therapy Postmastectomy irradiation of the chest wall and regional nodes for high-risk cases |
Classical CMFb AC, EC, or FACb |
|
|
Enhanced |
|
|
Taxanes |
Aromatase inhibitors LH-RH agonists |
|
Maximal |
Sentinel node biopsy Reconstructive surgery |
|
Growth factors Dose-dense chemotherapy |
|
|
CMF = cyclophosphamide, methotrexate, and 5-fluorouracil; AC = doxorubicin and cyclophosphamide; EC = epirubicin and cyclophosphamide; FAC = 5-fluorouracil, doxorubicin, and cyclophosphamide; LH-RH = luteinizing hormone-releasing hormone. aBreast-conserving therapy requires mammography and reporting of margin status. bRequires blood chemistry profile and complete blood count testing. SOURCE: Reprinted, by permission, from Anderson et al. (2006a). Copyright 2006 by the Breast Health Global Initiative, Fred Hutchinson Cancer Research Center. |
||||
siderably more expensive than ultrasound. Until the recent advent of digital technology—which itself is very expensive—all mammography required the use of X-ray film, which can be a critical barrier in a low-income country (Zotov et al., 2003). Second, many health facilities will not purchase mammography equipment because it is dedicated to the single use of breast imaging. In contrast, ultrasound is commonly available in a wide span of resource settings. It can be used for imaging many parts of the body and it requires no film unless records are needed. Thus, breast ultrasound may be used in settings where mammography is unavailable, simply because the tool exists.
Furthermore, breast ultrasound as an initial diagnostic test may have more utility in low-income countries than it does in high-income countries
TABLE 4-5 Treatment and Allocation of Resources: Stage II Breast Cancer Guidelines
|
Level of Resources |
Local-Regional Treatment |
Systemic Treatment (Adjuvant) |
||
|
Surgery |
Radiation Therapy |
Chemotherapy |
Endocrine Therapy |
|
|
Basic |
Modified radical mastectomy |
Chest wall and regional lymph node irradiation, if available |
Classical CMFa AC, EC, or FACa |
Ovarian ablation Tamoxifen |
|
Limited |
Breast-conserving therapyb |
Breast-conserving whole-breast irradiation as part of breast-conserving therapy Postmastectomy irradiation of the chest wall and regional nodes for high-risk cases |
|
|
|
Enhanced |
|
|
Taxanes |
Aromatase inhibitors LH-RH agonists |
|
Maximal |
Sentinel node biopsy Reconstructive surgery |
|
Growth factors Dose-dense chemotherapy |
|
|
CMF = cyclophosphamide, methotrexate, and 5-fluorouracil; AC = doxorubicin and cyclophosphamide; EC = epirubicin and cyclophosphamide; FAC = 5-fluorouracil, doxorubicin, and cyclophosphamide; LH-RH = luteinizing hormone-releasing hormone. aRequires blood chemistry profile and complete blood count testing. bBreast-conserving therapy requires mammography and reporting of margin status. SOURCE: Reprinted, by permission, from Anderson et al. (2006a). Copyright 2006 by the Breast Health Global Initiative, Fred Hutchinson Cancer Research Center. |
||||
for two reasons. First, patients in these settings commonly present with locally advanced, palpable disease, where breast ultrasound becomes a useful adjunct to CBE to evaluate the extent of disease (Vargas et al., 2003). Second, premenopausal breast cancer is more commonly diagnosed in low-income than high-income countries (Chow and Ho, 2000). The usually dense breast tissue of younger women makes cancers more difficult to image by mammography. For these reasons, the guidelines support the introduction of breast ultrasound before mammography in low-income countries (Table 4-2), which reverses the order generally accepted in high-income countries.
TABLE 4-6 Treatment and Allocation of Resources: Locally Advanced Breast Cancer Guidelines
|
Level of Resources |
Local-Regional Treatment |
Systemic Treatment (Adjuvant) |
||
|
Surgery |
Radiation Therapy |
Chemotherapy |
Endocrine Therapy |
|
|
Basic |
Modified radical mastectomy |
|
Neoadjuvant AC, FAC, or classical CMFa |
Ovarian ablation Tamoxifen |
|
Limited |
|
Postmastectomy irradiation of the chest wall and regional nodes |
|
|
|
Enhanced |
Breast-conserving therapyb |
Breast-conserving whole-breast irradiation |
Taxanes |
Aromatase inhibitors LH-RH agonists |
|
Maximal |
Reconstructive surgery |
|
Growth factors Dose-dense chemotherapy |
|
|
AC = doxorubicin and cyclophosphamide; FAC = 5-fluorouracil, doxorubicin, and cyclophosphamide; CMF = cyclophosphamide, methotrexate, and 5-fluorouracil; EC = epirubicin and cyclophosphamide; LH-RH = luteinizing hormone-releasing hormone. aRequires blood chemistry profile and complete blood count testing. bBreast-conserving therapy requires mammography and reporting of margin status. SOURCE: Reprinted, by permission, from Anderson et al. (2006a). Copyright 2006 by the Breast Health Global Initiative, Fred Hutchinson Cancer Research Center. |
||||
While breast ultrasound is useful for determining the extent of the invasive component of a breast cancer, it will predictably underestimate the extent of some cancers, particularly when they have a large noninvasive component. For example, ductal carcinoma in situ (DCIS, or stage 0 breast cancer) is not seen on breast ultrasound, but can be seen on mammography because it is associated with the deposition of microcalcifications. For this reason, diagnostic mammography (along with good pathology examination) is considered mandatory for breast conservation therapy (Tables 4-4, 4-5, and 4-6) because negative margins are needed with a partial mastectomy, whether it is for invasive or noninvasive disease (Carlson et al., 2000).
Endocrine Therapy and Hormone Receptor Testing
Oral endocrine therapy is among the simplest therapies for breast cancer in women with high tumor estrogen receptor (ER) levels and is recommended at every level, from basic to maximal. If tamoxifen, the standard drug,
TABLE 4-7 Treatment and Allocation of Resources: Metastatic (Stage IV) and Recurrent Breast Cancer Guidelines
|
|
Local-Regional Treatment |
Systemic Treatment (Adjuvant) |
|||
|
Level of Resources |
Surgery |
Radiation Therapy |
Chemotherapy |
Endocrine Therapy |
Supportive and Palliative Therapy |
|
Basic |
Total mastectomy for ipsilateral breast tumor recurrencea |
|
|
Ovarian ablation Tamoxifen |
Nonopioid and opioid analgesics |
|
Limited |
|
Palliative radiation therapy |
Classical CMFb Anthracycline monotherapy or in combinationb |
|
|
|
Enhanced |
|
|
Taxanes Capecitabine Trastuzumab |
Aromatase inhibitors |
Bisphosphonates |
|
Maximal |
|
|
Growth factors Vinorelbine Gemcitabine Carboplatin |
Fulvestrant |
|
|
CMF = cyclophosphamide, methotrexate, and 5-fluorouracil. aRequired resources are the same as those for modified radical mastectomy. bRequires blood chemistry profile and complete blood count testing. SOURCE: Reprinted, by permission, from Anderson et al. (2006a). Copyright 2006 by the Breast Health Global Initiative, Fred Hutchinson Cancer Research Center. |
|||||
is too expensive, surgical or radiation-induced oophorectomy—ovarian ablation—is also effective for premenopausal women. For this reason, the Cancer Treatment and Allocation of Resources panel categorized ovarian ablation and tamoxifen as basic-level resources for all stages of invasive cancer. However, the Diagnosis and Pathology panel designated ER testing as a limited-level resource, reasoning that even in the absence of testing, all patients can be given tamoxifen and/or oophorectomy. With this rationale, ER testing does not meet the formal definition of basic-level resource. This discrepancy between panels was not fully resolved during the 2005 Global Summit.
Treating all patients with endocrine therapy without ER testing means that a large fraction of patients—those without elevated ER levels—will get no benefit. In terms of costs and benefits, limiting hormonal treatments
TABLE 4-8 Health Care Systems and Public Policy Guidelines
|
Level of Resources |
Services |
Facilities |
Recordkeeping |
|
Basic |
Primary care Surgical Oncology Nursing Palliative care |
Health care Surgical Pathology laboratory Pharmacy Outpatient care |
Individual medical records and service-based patient registration |
|
Limited |
Imaging Radiation oncology Peer support Early detection |
Imaging Radiation therapy Clinical information system Health system network |
Facility-based medical records and centralized patient registration Local cancer registry |
|
Enhanced |
Opportunistic screening Cancer follow-up Rehabilitation Group support |
Centralized referral cancer center(s) |
Facility-based follow-up systems |
|
Maximal |
Population-based screening Individual psychosocial care |
Satellite (noncentralized or regional) cancer centers |
National cancer registry |
|
SOURCE: Reprinted, by permission, from Anderson et al. (2006a). Copyright 2006 by the Breast Health Global Initiative, Fred Hutchinson Cancer Research Center. |
|||
to women likely to benefit will save enough to offset, in part, the cost of hormone receptor testing. Thus, regardless of the designation as basic or limited, hormone receptor testing has obvious utility, making it a high-priority test at all levels.
Cytotoxic Chemotherapy and Related Infrastructure
In high-income countries, systemic therapy is usually recommended for all cancers larger than 1 cm, regardless of whether lymph nodes are positive or negative (Carlson et al., 2000). For ER-negative cancers, cytotoxic chemotherapy is needed to reduce the risk of disease recurrence. Chemotherapy is a mainstay of treatment for more advanced cancers. However, because the prognosis for stage I cancer following local therapy (surgery with or without radiotherapy) is already good, chemotherapy increases survival only marginally (Eniu et al., 2006). Thus, the relative utility of cytotoxic chemotherapy, given its cost, can be debated for early-stage, node-negative cancer. To properly reflect this difference in the utility of chemotherapy between early and later stage disease, the Cancer Treatment and Allocation of Resources
TABLE 4-9 Therapy Overview: Modified Radical Mastectomy and Breast-Conserving Therapy
|
Therapy |
Strengths |
Weaknesses |
Required Resources |
|
Modified radical mastectomy |
Effective local treatment Uses surgical techniques widely available Rapid treatment Short posttreatment convalescence Limited long-term complications Radiation therapy can be avoided in some cases |
Loss of body image (mutilation) Negative psychosocial impact Radiation therapy is often still necessary |
Core surgical resources
Postmastectomy irradiation of the chest wall and regional lymph nodes |
|
Breast-conserving therapy |
Equivalent survival to modified radical mastectomy Preservation of body image for the woman Improved quality of life |
Slight increase in the rate of recurrence (in breast) compared with modified radical mastectomy Lower acceptance among less educated people Prolonged treatment course Requires access to a radiation therapy facility |
High-quality breast imaging (mammography and, if available, ultrasound) Core surgical resources (same as for modified radical mastectomy) Pathology for margin assessment Surgical services experienced in the procedure Breast-conserving whole-breast irradiation Geographic accessibility Support systems that allow receipt of radiation therapy over a period of weeks |
|
SOURCE: Reprinted, by permission, from Eniu et al. (2006). Copyright 2006 by the Breast Health Global Initiative, Fred Hutchinson Cancer Research Center. |
|||
panel determined that cytotoxic chemotherapy is a limited-resource therapy for stage I cancer and for metastatic cancer, but is a basic-level resource for patients with stage II or locally advanced cancer.
In a health care system that lacks the infrastructure for providing systemic chemotherapy, stage I, ER positive cancers can be effectively treated and stage IV ER positive cancers can be palliated, but stage II and locally advanced disease can only be palliated at best regardless of ER status. Unfortunately, these more advanced but treatable cancers are the most common presentations in low-income countries. The conclusion, then, is that to provide a reasonable level of breast cancer treatment, the infrastructure
for cytotoxic chemotherapy must be there from the beginning, even though this is considered higher than a basic-level resource for some stages of breast cancer.
THE GLOBAL PARTNERSHIP UNDERLYING THE BHGI
Improving breast health care in a low-resource setting is bound to be complex, requiring the collaboration of multiple sectors, including health care ministries and governmental agencies, NGOs, and public and patient groups (Glassman et al., 1999; Lim, 2002; Mathew et al., 2003; McCabe et al., 1995). The relative contribution of each sector will depend on the country’s governmental structure, the extent of focus on health care and breast cancer, available resources, the strength of the NGO sector, and the ability of patients, survivors, and advocates to act collaboratively.
A strength of the BHGI guideline development process is its collaborative nature, creating partnerships to improve health care in limited-resource countries (Figure 4-1). The guidelines serve as a framework for these organizations to interact and create projects of common interest. The challenges extend beyond the capacity of any one partner or sector to address effectively. This is especially true in a global world where traditional boundaries between what are “public” and “private” responsibilities have become blurred, where civil society organizations have taken on important new roles, and where technology enables the creation of effective networks
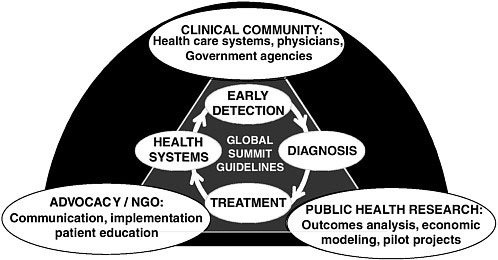
FIGURE 4-1 Linkages among the clinical community, advocacy groups and nongovernmental organizations (NGOs), and the public health research community.
with lowered cost. A next step is to transform the guideline alliance into operational alliances that take the guidelines into practice in low-resource settings.
LESSONS FROM THE BHGI MODEL FOR OTHER CANCERS
The guidelines development process for countries with limited resources tries to offer a practical solution to the implausibility of applying guidelines developed for high-resource countries. In a limited-resource country, many barriers stand between the average patient and the level of care dictated by guidelines applicable to high-resource settings. These include inadequate numbers of trained health care providers; inadequate diagnostic and treatment infrastructure such as pathology, pharmacy, infusion centers, and microbiology laboratories; lack of drugs; lack of radiographic film; and inadequate transportation and communications systems.
Physicians working in a limited-resources environment may be forced to make decisions at odds with their medical knowledge. Despite knowing the optimal management for a patient, less-than-optimal solutions are offered to patients because diagnostic and/or treatment resources are simply lacking. Lack of mammography and radiotherapy facilities, for example, precludes the use of breast-conserving therapy, and unavailability of chemotherapy agents and infrastructure may make it impossible or unsafe to deliver cytotoxic chemotherapy in the adjuvant setting.
Resource limitations frustrate clinicians who are unable to offer “gold standard” treatment to any or only to some patients. This tension is often amplified by the clinicians’ added responsibility for managing inadequate resources from an insufficient cancer program budget. Does a clinician decide to treat 10 patients with an older, less expensive chemotherapy regimen, or to treat 2 patients with a newer, more efficacious, but also more expensive regimen?
To establish a cancer treatment program in a limited-resource setting, key treatment alternatives should be considered, weighing costs, efficacy differences, and the expected availability of resources and personnel to implement the program. Flexibility must be built into recommendations because heterogeneous social, economic, and health system development among countries and often among regions of the same country, make uniform recommendations impractical.
The expert panels asked to develop meaningful, justifiable, and scientifically rigorous guidelines for LMCs requires the same type of multidisciplinary expertise as needed to develop guidelines for resource-rich areas. They must also be willing and competent to estimate the magnitude of relative benefit from the available therapies and to prioritize these therapies with consideration of efficacy, toxicity, and resources, with specific consid-
eration of the conditions in low-resource settings (Anderson et al., 2003a). Recommendations must also respect the social, economic, religious, and political milieu within which they are to be applied. Involvement of social scientists, economists, religious leaders, and politicians may also be appropriate. However, the patient and delivery of evidence-based health care must remain the primary focus.
At every point of recommendation, the panel should consider not only the applicable scientific evidence, but also the implications and barriers to implementing the recommendation. It makes little sense to recommend a therapy that is available at only a few centers in the world. It is also impossible to implement even a cost-effective, simple therapy if it requires special resources that are not available.
Thus, the responsibility of the guideline panels is to develop an inclusive, objective, evidence-based guideline associated with a prioritization schema stratified by available health care resources. Further refinement of the guidelines produced will undoubtedly be necessary when actually applied to be respectful of local geographic, social, political, and religious issues, and to incorporate the actual costs in a given site.
Guideline Implementation
Developing and publishing guidelines is one step, but it does not equal implementation. The practical application of treatment guidelines in a specific site requires an honest evaluation of existing resources and expertise. This assessment must recognize which procedures are available to all patients, as this identifies the associated level of resources, from basic to maximal. To the extent that a sequential strategy can be followed, providing universal access to one level and then moving up, substantial inequity in the use of limited resources is prevented, and the greatest benefit can accrue to the largest number of people.
Once the level of existing resources is defined, an analysis will identify the missing elements to allow completion of that level of resource. Resources can then be allocated strategically to improve the performance of the cancer unit. This strategy can be used at the national level for allocation of resources for breast cancer. It also can help to prevent using scarce resources on the latest technologies or drugs to benefit a few at the expense of basic effective care for much larger numbers.
The checklists developed by the BHGI panels are suitable for use by health ministries or hospital administrators and provide an inventory of the minimal technical and staff requirements. The information can be used to plan the future development of a unit by prioritizing the acquisition of missing elements. The guidelines also can support a clinician’s request for specific equipment and funding for breast cancer treatment. Finally, the
checklists can be used as one input for accreditation of units for breast cancer diagnosis and treatment.
APPLYING THE BHGI MODEL TO OTHER CANCERS
Breast cancer was a good choice for a first effort at guideline development for low-resource areas—for whatever reasons it was actually chosen. It is among the most common cancers everywhere, a variety of effective treatments is available, and breast cancer has been the driver in patient advocacy around the world. It might have been easier to fund this effort than would have been the case for a different cancer, although funding was a challenge, particularly at the beginning (Personal communication, B. Anderson, Director, BHGI June 2006). The success of the BHGI, at least through the guideline development phase, should give potential funders confidence that tackling other cancers can also be successful.
The BHGI has built financial support from the first summit through the upcoming 3rd biennial summit, and no new such undertaking will succeed without adequate support. As a benchmark, the budget for the 2007 global summit, which will support 75 participants from around the world and 5 staff members at a 4-day meeting in Budapest, is US $400,000. An additional US $100,000 is budgeted for a journal supplement with the revised guidelines and supplementary papers, including a writer and production and printing costs. The actual guideline development work is carried out largely uncompensated, by the participants who volunteer their time. In the case of the BHGI, a full-time manager is employed, and the scientific leader has partial salary support for the project. The usual costs of operations (office, equipment, and supplies) also must be covered (Personal communication, L. Sullivan, Program Manager, BHGI, October 2, 2006). These costs will, of course, vary according to the host organizations for other initiatives.
PRIORITIZING CANCERS FOR GUIDELINE DEVELOPMENT IN LIMITED-RESOURCE COUNTRIES
Following the lead of breast cancer, the greatest benefit from additional resource-level-appropriate guidelines would be for relatively common cancers for which treatments are affordable and have proven reasonably successful in some settings. Cancers that are less common but highly curable, with treatments that can be used in a low-resource setting, could also be considered.
No purely objective, scientific method exists for prioritizing cancers for “resource-level-specific” attention. What actually happens will be a result of leaders coming forward for a specific type of cancer, and obtaining support for an international collaborative effort. It would make sense, however, not
to spend a great deal of effort on cancers for which treatment is less successful, but to encourage work on those for which treatment is more successful. Cancers for which treatment is difficult and less likely to succeed would typically not warrant complex guideline development, at least initially. Guidelines for palliative care for the common cancers should be developed, and will be applicable to people dying from all types of cancer.
A major consideration in decisions about offering treatment for cancers in resource-limited settings are characteristics of the interventions themselves—the specific types of surgery or drugs, for example—needed to achieve a good outcome. Short-term toxicity and long-term sequelae are also important considerations for both medical and social reasons. The available choices of interventions, the personnel needed, and other necessary conditions are the heart of future discussions to develop resource-level-appropriate guidelines, as was done by the BHGI. For most cancers, different subtypes can be identified that are more or less amenable to treatment, and the time course and side effect profiles for the treatment choices may differ in ways that are significant medically and practically for patients. These and a host of other details will require careful consideration of every option as it applies to the types of settings that exist in LMCs.
In every case, good cancer treatment will require a multidisciplinary, coordinated team of individuals with training in the specific cancers for which treatment will be offered. One of the benefits of the BHGI-type of initiative is to make explicit the resource needs in all categories. Having this information should assist decision makers, whether at the national or facility level, to decide what can be offered, and what the next steps would be to expand treatment to patients with other types of cancers.
Table 4-10 lists the 10 cancer types responsible for the greatest numbers of deaths in developing countries in 2002. The top four—cancers of the lung, liver, stomach, and esophagus—make up nearly half of the deaths in these countries. Survival from these cancers in the United States, where most people have access to treatment, is relatively poor and has not improved a great deal since the 1970s. This is corroborated by the related (but not identical) statistic of the ratio of mortality to incidence in more developed countries, and its similarity to the same statistic in less developed countries.1 Taking lung cancer, for example, the 5-year survival in the United States was reported as 12 percent in the 1970s. In the late 1990s, it was still only 15 percent. The mortality to incidence ratio was 86 and 88 percent in more and
TABLE 4-10 Selected Cancers: Factors to Consider in Prioritizing Treatment in Developing Countries
|
|
|
5-Year Relative Survival, U.S. 1970s (different years) |
5-Year Relative Survival, U.S. 1995– 2001 |
Deaths in Less Developed Countries, 2002 (GLOBOCAN) |
Mortality/ Incidence Ratio in Less Developed Countries (%) |
Mortality/ Incidence Ratio in More Developed Countries (%) |
|
1. |
Lung |
12 |
15 |
591,000 |
88 |
86 |
|
2. |
Liver |
4 |
9 |
487,000 |
95 |
99 |
|
3. |
Stomach |
15 |
23 |
486,000 |
78 |
68 |
|
4. |
Esophagus |
5 |
15 |
320,000 |
83 |
88 |
|
5. |
Breast |
75 |
88 |
221,000 |
43 |
30 |
|
6. |
Cervix uteri |
70 |
73 |
234,000 |
57 |
47 |
|
7. |
Colon and rectum |
50 |
64 |
214,000 |
60 |
47 |
|
8. |
Leukemia |
34 |
48 |
137,000 |
78 |
68 |
|
9. |
Non-Hodgkin’s lymphoma |
47 |
60 |
97,000 |
66 |
49 |
|
10. |
Prostate |
67 |
100 |
91,000 |
55 |
25 |
|
11. |
Head and neck |
|
|
89,000 |
61 |
42 |
|
|
Oral cavity and pharynx |
54 |
59 |
|
|
|
|
|
Larynx |
66 |
66 |
|
|
|
|
12. |
Hodgkin’s lymphoma |
71 |
85 |
15,000 |
44 |
28 |
|
13. |
Testicular |
79 |
96 |
6,000 |
31 |
9 |
|
SOURCES: IARC (2004); Ries et al. (2006). |
||||||
less developed countries, meaning that there were nearly as many deaths as cases in 2002, regardless of resource levels.
For other cancers down the list of the 10 leading causes of cancer death, the overall survival among U.S. cancer patients is much better, and there are substantial differences between the mortality to incidence ratio between less and more developed countries, suggesting that treatment makes a significant difference in survival. This is true for the following cancers: cancers of the breast, cervix, colon and rectum, prostate, and leukemia and non-Hodgkin’s lymphoma. In the case of leukemias, a substantial increase in survival occurred in the United States from the 1970s through 2000, suggesting that treatment has improved the chances of survival. One caveat in looking at changes in overall survival is that a change can be the result of improved stage distribution (i.e., more cancers detected in early stages), and therefore more amenable to long-term survival or cure.
Head and neck cancers are added to the list because they are relatively common and have a very good prognosis when treated. Hodgkin’s lymphoma and testicular cancer both have very high survival rates and are among the most common cancers in young adults. Treatment of Hodgkin’s lymphoma among older adults is also highly successful.
Table 4-11 looks more closely at the cancers with high survival rates in the United States and relatively large differentials in mortality to incidence ratios between less and more developed countries. It is useful to note that survival is relatively good for both localized and regionally spread cancers for all of these cancer types. This is particularly important for cancers that are more likely to be detected in early stages in more developed countries, either because of screening or because they produce symptoms early, causing people to seek medical attention.
The mortality to incidence ratio has a slightly different interpretation in the case of cervical cancer than for other cancers. This is because most of the effect of screening—which is widespread in the United States and other high-income countries—is to detect precancerous stages rather than early-stage cancers. This dramatically lowers the number of incident cases, so incident cervical cancers in more developed countries are only a small proportion of the cancers that would have occurred (and that do occur in less developed countries) in the absence of screening. The difference in the mortality to incidence ratio between less and more developed countries would be larger if these statistics were more comparable.
Cancers of Children and Young Adults
As is the case with cancers of adults, practitioners in LMCs have formally or informally prioritized cancer types of children and young adults for treatment. Several leaders in pediatric oncology in Asia, Latin America, and the Pacific contacted for this report provided insight into their schema for prioritizing childhood cancers for treatment. Leukemias and lymphomas are the most frequent childhood cancers everywhere, although the specific types and proportions vary around the world. Most also have high cure rates except in advanced stages, and require only chemotherapy. A respondent from Pakistan, in charge of pediatric oncology at a government-owned hospital, provided a table based on his experience (Table 4-12). Tradeoffs are implied in the juxtaposition of prevalence, survival, and cost. The only cancers clearly excluded from treatment are very high-cost, very poor-survival types. Other respondents gave similar priorities, except in some cases retinoblastoma was ranked higher and Wilms’ tumor and neuroblastoma lower. All of these solid tumors, which are the most frequent among children in many countries (Abdullaev et al., 2000; Leal-Leal et al.,
TABLE 4-11 Stage-Specific 5-Year Survival and Main Treatment Modalities for Selected Cancers
|
Cancer Site |
5-Year Relative Survival, U.S. 1995–2001 |
Main Treatment Modalities |
|
Breast (overall) |
88 |
Surgery, radiotherapy, chemotherapy, hormonal therapy |
|
Localized |
98 |
Surgery with or without radiotherapy |
|
Regional |
81 |
Surgery with or without radiotherapy and/or chemotherapy and/or hormonal therapy |
|
Cervix uteri (overall) |
73 |
Radiotherapy, surgery, chemotherapy |
|
Localized |
92 |
Surgery and/or radiotherapy |
|
Regional |
55 |
Radiotherapy or surgery |
|
Colon and rectum (Overall) |
64 |
Surgery, chemotherapy, radiotherapy |
|
Localized |
90 |
Surgery |
|
Regional |
68 |
Surgery and chemotherapy with or without radiotherapy |
|
Non-Hodgkin’s lymphoma (overall) |
60 |
Radiotherapy, chemotherapy |
|
Localized |
73 |
Radiotherapy and/or chemotherapy |
|
Regional |
63 |
Radiotherapy and/or chemotherapy |
|
Head and neck (Overall) |
59–66 |
Radiotherapy, surgery |
|
Larynx |
66 |
|
|
Localized: |
|
Radiotherapy and/or surgery |
|
Oral cavity and pharynx |
82 |
|
|
Larynx |
85 |
|
|
Regional: |
|
Surgery and/or radiotherapy |
|
Oral cavity and pharynx |
51 |
|
|
Larynx |
50 |
|
|
Testicular (overall) |
96 |
Surgery, chemotherapy |
|
Localized |
100 |
Surgery with or without radiotherapy |
|
Regional |
71 |
Surgery and chemotherapy |
|
Prostate (overall) |
100 |
Surgery, radiotherapy |
|
Local/regional |
100 |
Surgery or radiotherapy |
|
Hodgkin’s lymphoma (overall) |
85 |
Chemotherapy, radiotherapy |
|
Local |
90 |
Radiotherapy or chemotherapy |
|
Regional |
90 |
Radiotherapy and/or chemotherapy |
|
Distant |
76 |
Radiotherapy and chemotherapy |
|
SOURCE: National Cancer Institute (2006). |
||
TABLE 4-12 Priority Ranking for Treating Pediatric Cancers in a Pakistani Hospital, with U.S. Survival Rates
|
Disease |
Percentage Distribution of Cancers (%) |
Long-Term Survival (%) |
Cost |
5-Year Relative Survival, U.S., 1985–1994 (%) |
|
Acute lymphoblastic leukemia |
25 |
60 |
Intermediate |
77 |
|
Hodgkin’s lymphoma |
10 |
90 |
Low |
91 |
|
Non-Hodgkin’s lymphoma |
10 |
65 |
Intermediate |
72 |
|
Germ cell tumor |
3 |
80 |
Low |
88 |
|
Wilms’ tumor |
5 |
80 |
Low |
92 |
|
Retinoblastoma |
6 |
70 |
Low |
94 |
|
Rhabdomyosarcoma and neuroblastoma, or NBS (nonmetastatic) |
5 |
30 |
High |
64 (NBS) |
|
Brain tumors: Selected nonmetastatic and low grade |
3 |
50 |
Intermediate |
Various |
|
Osteosarcoma (nonmetastatic) |
3 |
40 |
High |
63 |
|
Would not treat: |
|
|
|
|
|
Acute myelogenous leukemia |
5 |
<10 |
Very high |
41 |
|
Stage IV neuroblastoma and sarcoma |
15 |
<10 |
Very high |
— |
|
Relapsed disease and others |
10 |
<10 |
Very high |
— |
|
SOURCES: Personal communication, M.S. Ashraf, Children Cancer Hospital, Karachi, Pakistan, March 2006; Ries et al. (1999). |
||||
2006) were considered worth treating. Resource-level-specific guidelines would be useful for all the prioritized tumors.
DISCUSSION AND RECOMMENDATION
The BHGI guidelines, as well as any other guidelines produced, are tools that can enable progress in cancer control, but their mere existence is unlikely to result in major changes. The BHGI is entering a new phase, assisting LMCs in putting the guidelines into practice. Feedback from this effort should be useful in moving forward with the BHGI and in new areas. The challenges include making the guidelines and supporting documentation available in languages and formats that are accessible to decision-makers.
Even acknowledging the remaining challenges in putting guidelines into practice, the value of resource-level-appropriate, evidence-based guidelines is clear. Such guidelines should be developed for other priority cancers meeting the criteria discussed in this chapter. As cancer control continues to evolve, with newer drugs, screening methods, and other cutting-edge interventions becoming available, the need will become even greater to
maintain an evidence base of established, effective interventions, especially those that are less costly and less demanding of health care infrastructure than the state-of-the-art in high-income countries.
This report encourages international collaborations to address the priority cancers discussed in this chapter and international funders to support these efforts. The collaborations should be inclusive of all interested parties and should, ideally, be limited to a single collaboration per cancer type or natural grouping of cancers that represents as close to a true global consensus as possible. Each of these efforts should be viewed as a long-term commitment based on sustainable structures that can produce updated guidelines and take on the challenges of implementation.
Leadership and support will be key to the success of further initiatives to develop guidance for other priority cancers. The success of the BHGI thus far is due, in large part, to Dr. Anderson. The existing BHGI model should make it easier for parallel initiatives to get started, but the need for a dedicated leader, willing to devote substantial time to the project, is still a limiting factor. The lion’s share of the technical work of the BHGI—reviewing evidence, preparing papers and guidelines—has been carried out by professionals with no extra compensation. Support is needed, however, for administrative functions (including some personnel costs), travel, and logistics, at a minimum. As is the case with the BHGI, support could come from a number of sources, including public- and private-sector organizations. Sustainability, of both personnel and funding, will be critical to the long-term success of any such initiatives.
RECOMMENDATION 4-1. Resource-level-appropriate guidelines should be developed for the overall management of major cancers for which treatment can make a substantial difference in a meaningful proportion of patients, and for selected pediatric cancers. The BHGI model could be used or others developed. The priority adult cancers for which resource-level-specific guidelines are needed are cervical cancer, colon cancer, and head and neck cancers. Pediatric priority cancers are leukemias and lymphomas. Motivated professionals from high-income countries and LMCs should work together to spearhead these efforts, with financial support from a variety of institutions.
REFERENCES
Abdullaev FI, Rivera-Luna R, Roitenburd-Belacortu V, Espinosa-Aguirre J. 2000. Pattern of childhood cancer mortality in Mexico. Archives of Medical Research 31(5):526–531.
Anderson BO. 2003. Global Summit Consensus Conference on International Breast Health Care: Guidelines for countries with limited resources. Breast Journal 9(Suppl 2):S40–S41.
Anderson BO, Braun S, Carlson RW, Gralow JR, Lagios MD, Lehman C, Schwartsmann G, Vargas HI. 2003a. Overview of breast health care guidelines for countries with limited resources. Breast Journal 9(Suppl 2):S42–S50.
Anderson BO, Braun S, Lim S, Smith RA, Taplin S, Thomas DB, Global Summit Early Detection Panel. 2003b. Early detection of breast cancer in countries with limited resources. Breast Journal 9(Suppl 2):S51–S59.
Anderson BO, Eniu AE, Sullivan L, Carlson RW. 2005 (December). Guidelines for Breast Cancer Detection, Diagnosis and Treatment in Limited Resource Countries as a Framework for Change: The Breast Health Global Initiative. Unpublished.
Anderson BO, Shyyan R, Eniu A, Smith RA, Yip CH, Bese NS, Chow LW, Masood S, Ramsey SD, Carlson RW. 2006a. Breast cancer in limited-resource countries: An overview of the Breast Health Global Initiative 2005 guidelines. Breast Journal 12(Suppl 1):S3–S15.
Anderson BO, Yip CH, Ramsey SD, Bengoa R, Braun S, Fitch M, Groot M, Sancho-Garnier H, Tsu VD, Global Summit Health Care Systems and Public Policy Panel. 2006b. Breast cancer in limited-resource countries: Health care systems and public policy. Breast Journal 12(Suppl 1):S54–S69.
Carlson RW, Anderson BO, Bensinger W, Cox CE, Davidson NE, Edge SB, Farrar WB, Goldstein LJ, Gradishar WJ, Lichter AS, McCormick B, Nabell LM, Reed EC, Silver SM, Smith ML, Somlo G, Theriault R, Ward JH, Winer EP, Wolff A, National Comprehensive Cancer Network. 2000. NCCN Practice Guidelines for Breast Cancer. Oncology (Huntington) 14(11A):33–49.
Carlson RW, Anderson BO, Chopra R, Eniu AE, Jakesz R, Love RR, Masetti R, Schwartsmann G, Global Summit Treatment Panel. 2003. Treatment of breast cancer in countries with limited resources. Breast Journal 9(Suppl 2):S67–S74.
Chow LW, Ho P. 2000. Hormonal receptor determination of 1,052 Chinese breast cancers. Journal of Surgical Oncology 75(3):172–175.
Eniu A, Carlson RW, Aziz Z, Bines J, Hortobagyi GN, Bese NS, Love RR, Vikram B, Kurkure A, Anderson BO, Global Summit Treatment and Allocation of Resources Panel. 2006. Breast cancer in limited-resource countries: Treatment and allocation of resources. Breast Journal 12 (Suppl 1):S38–S53.
Gijsbers van Wijk CM, van Vliet KP, Kolk AM. 1996. Gender perspectives and quality of care:Gender perspectives and quality of care: Towards appropriate and adequate health care for women. Social Science & Medicine 43(5):707–720.
Glassman A, Reich MR, Laserson K, Rojas F. 1999. Political analysis of health reform in the Dominican Republic. Health Policy & Planning 14(2):115–126.
International Agency for Research on Cancer. 2004. GLOBOCAN 2002. Lyon: IARC.
Leal-Leal CA, Rivera-Luna R, Flores-Rojo M, Juarez-Echenique JC, Ordaz JC, Amador-Zarco J. 2006. Survival in extra-orbital metastatic retinoblastoma:treatment results. Clinical & Translational Oncology: Official Publication of the Federation of Spanish Oncology Societies & of the National Cancer Institute of Mexico 8(1):39–44.
Lim GC. 2002. Overview of cancer in Malaysia. Japanese Journal of Clinical Oncology 32(Suppl):S37–S42.
Mathew A, Cowley S, Bliss J, Thistlewood G. 2003. The development of palliative care in national government policy in England, 1986–2000. Palliative Medicine 17(3):270–282.
McCabe MS, Varricchio CG, Padberg R, Simpson N. 1995. Women’s health advocacy: Its growth and development in oncology. Seminars in Oncology Nursing 11(2):137–142.
National Cancer Institute. Cancer Stat Fact Sheets. [Online] Available: http://seer.cancer.gov/statfacts/ [accessed July 2, 2006].
NCCN (National Comprehensive Cancer Network). 2005. The NCCN Breast Cancer Screening and Diagnosis Guidelines. Jenkintown, PA: NCCN.
Pal SK, Mittal B. 2004. Fight against cancer in countries with limited resources: The postgenomic era scenario. Asian Pacific Journal of Cancer Prevention 5(3):328–333.
Redman BK. 1996. Ethical issues in the development and use of guidelines for clinical practice. Journal of Clinical Ethics 7(3):251–256.
Ries LAG, Harkins D, Krapcho M, Mariotto A, Miller BA, Feuer EJ, Clegg L, Eisner MP, Horner MJ, Howlader N, Hayat M, Hankey BF, Edwards BK (eds.). 2006. SEER Cancer Statistics Review, 1975–2003. Bethesda, MD: National Cancer Institute.
Ries LAG, Smith MA, Gurney JG, Linet M, Tamra T, Young JL, Bunin GR. 1999. Cancer Incidence and Survival Among Children and Adolescents: United States SEER Program 1975–1995. Bethesda, MD: National Cancer Institute, SEER Program.
Shyyan R, Masood S, Badwe RA, Errico KM, Liberman L, Ozmen V, Stalsberg H, Vargas H, Vass L, Global Summit Diagnosis and Pathology Panel. 2006. Breast cancer in limited-resource countries: Diagnosis and pathology. Breast Journal 12(Suppl 1):S27–S37.
Smith RA, Caleffi M, Albert US, Chen TH, Duffy SW, Franceschi D, Nystrom L, Global Summit Early Detection and Access to Care Panel. 2006. Breast cancer in limited-resource countries: Early detection and access to care. Breast Journal 12(Suppl 1):S16–S26.
Stavros AT, Thickman D, Rapp CL, Dennis MA, Parker SH, Sisney GA. 1995. Solid breast nodules: Use of sonography to distinguish between benign and malignant lesions. Radiology 196(1):123–134.
Vargas HI, Anderson BO, Chopra R, Lehman CD, Ibarra JA, Masood S, Vass L, Global Summit Diagnosis Panel. 2003. Diagnosis of breast cancer in countries with limited resources. Breast Journal 9(Suppl 2):S60–S66.
WHO (World Health Organization). 2002. National Cancer Control Programmes: Policies and Managerial Guidelines. 2nd ed. Geneva, Switzerland: WHO.
Winn RJ, McClure J. 2003. The NCCN Clinical Practice Guidelines in Oncology: A primer for users. Journal of the National Comprehensive Cancer Network 1(1):5–13.
Zotov V, Shyyan R, PATH Breast Cancer Assistance Program. 2003. Introduction of breast cancer screening in Chernihiv Oblast in the Ukraine: Report of a PATH Breast Cancer Assistance Program experience. Breast Journal 9(Suppl 2):S75–S80.
































