11
Sexually Transmitted Diseases and HIV Infection
In Sub-Saharan Africa, sexually transmitted diseases (STDs) are among the most common reasons that adults, as a group, seek health care (Hira et al., 1992b; Meheus et al., 1990; Zimbabwe, 1989). Yet this statistic refers primarily to males. Most women with STDs will not seek medical care at all, or will only present late for treatment, when complications have already developed, complications that have devastating physical, psychological, and social consequences, particularly for women and their children (Carty et al., 1972; Latif, 1981). These consequences for individuals, in turn, have serious repercussions for the societies of which they are part.
Human immunodeficiency virus (HIV) infection is considered by many to be the most serious STD because of its multiple debilitating manifestations, its high fatality rate, and the severe stigma and discrimination that surround it in communities around the globe. In Sub-Saharan Africa, where the infection is acquired sexually in most cases, HIV seriously affects, and will spread among, the very same women and men who already have very high rates of other STDs.
Table 11-1 depicts the gender-related burden of HIV infection and the STDs reviewed in this chapter. As the table indicates, both are of unique significance to female health because of their capacity for transmission from mother to offspring during pregnancy and birth and, in the case of HIV infection, through breast milk. In contrast to HIV infection, which in Sub-Saharan Africa is a disease of both women and men, the other STDs disproportionately affect women, who bear 80 percent of the disability-adjusted life years (DALYs) lost to these diseases (World Bank, 1993). In addition, compared with HIV infection, which has stimulated extensive research, including research related to the women of Sub-Saharan Africa (de Bruyn, 1992; Mane et al., 1994; Mann et al., 1992; Orubuloye et al., 1993; Temmerman et al., 1994; Ulin, 1992; WHO, 1993, 1994), relatively little attention has been paid to the other STDs. We therefore choose to consider HIV and other STDs together, and to place greater emphasis on non-HIV STDs.
This chapter first provides a conceptual framework for thinking about STDs and HIV infection. It then highlights the gender differences that make all STDs a critical women's health concern and examines special issues that are unique to the different stages of women's lives. Next, the chapter describes the magnitude of the problem in Sub-Saharan Africa, analyzing existing data, explores the geographic distribution and clustering of five common STDs, including HIV infection. The chapter proceeds to an examination of societal factors that may influence STD and HIV prevalence, and closes with a discussion of implications for research, policy, and programs.
TABLE 11-1 HIV Infection and Other Sexually Transmitted Diseases in Sub-Saharan Africa: Gender-Related Burden
|
Disorder |
Exclusive to Females |
Greater for Females than for Males |
Burden for Females and Males Comparable, but of Particular Significance for Females |
|
HIV infection |
Xa |
||
|
Other STDs |
Xa |
||
|
NOTE: Significance is defined here as having impact on health that, for any reason—biological, reproductive, sociocultural, or economic —is different in its implications for females than for males. a Both HIV infection and other STDs have the capacity to be passed on from an infected mother to her offspring through congenital, perinatal, or neonatal transmission. |
|||
OVERVIEW
STDs, Including HIV Infection, and their Complications
One of the simplest and most useful ways to classify STDs is based on their two most common syndromes: genital discharge and genital lesions. STDs such as gonorrhea, chlamydia, and trichomoniasis cause genital discharge. Others cause genital lesions that can be further categorized as ulcerative diseases such as syphilis, chancroid, and genital herpes, or as nonulcerative diseases such as genital warts. In addition, a few STDs, such as human immunodeficiency virus (HIV) infection, do not fit this syndromic classification. The final stage of HIV infection, acquired immunodeficiency syndrome (AIDS), develops after a variable period of time, with onset generally following initial infection within approximately 10 years, during which it remains largely asymptomatic. The symptomatic stages of AIDS are characterized by the onset of opportunistic infections or cancers, some of which, such as cervical cancer, affect the genital tract.
This chapter refers to studies from Africa whenever they are available. When African data do not exist, or when comparison is appropriate, data are presented from other countries, particularly Sweden, which has accumulated a substantial and unique body of data on STDs in women.
Complications of STDs
Untreated or inappropriately treated, STDs may lead to severe complications, which account for most of their morbidity. Such complications occur most frequently in populations that lack ready access to effective treatment. Furthermore, both the anatomy of the female genital tract —prone to ascending infections—and reproductive events and related medical procedures—childbirth, stillbirth, abortion, uterine curettage, and intrauterine device insertion—are additional risk factors for STD complications particular to women (Meheus et al., 1990). The main STD complications include pelvic inflammatory disease (PID) and its sequelae, and the most important of these are impaired fertility and adverse pregnancy outcomes, enhanced HIV transmission, and cervical cancer.
The clinical manifestations associated with HIV infection depend on several factors, including the pathogens to which an individual has been exposed; the susceptibility of the host; possibly, the strain of HIV; and access to medical care. Most studies have not highlighted significant gender differences in disease progression. In the United States, however, one study indicated that, compared with men, HIV-infected women were at increased risk of death, which may reflect differential access to health care or different socioeconomic status and social support for women compared with men (Melnick et al., 1994). In Africa, little information is available on the natural history of HIV infection, particularly among women. The clinical picture of HIV infection in Sub-Saharan Africa
TABLE 11-2 Sexually Transmitted Diseases in Women
|
Potential Complications |
|||||
|
Syndrome |
Etiology |
Infertility/Ectopic Pregnancya |
Adverse Pregnancy Outcomea |
Cervical Cancera |
Enhanced HIVa Transmission |
|
Discharge |
Chlamydia |
X |
X |
X |
|
|
Gonorrhea |
X |
X |
X |
||
|
Trichomonas |
X |
X |
|||
|
Lesions |
|||||
|
Genital ulcers |
|||||
|
Syphilis |
X |
X |
|||
|
Chancroid |
X |
||||
|
Herpes |
X |
X |
|||
|
Other |
|||||
|
HPV infection and genital warts |
X |
X |
|||
|
a Potentially fatal. |
|||||
appears to be dominated by dermatological and gastrointestinal manifestations and by tuberculosis (De Cock et al., 1993; Kreiss and Castro, 1990).
A recent study strongly suggests that a sexually transmitted herpes virus causes Kaposi's sarcoma, one of the clinical manifestation of AIDS (Chang, 1994). In Africa, while this cancer has always been endemic, predominantly affecting men, with the spread of HIV its incidence has risen notably, and it is affecting an increasing number of women. In Zambia, the proportion of women among persons with Kaposi's sarcoma increased from 9 percent in 1983 to 30 percent in 1990–1992 (Ebrahim et al., 1993).
The onset of AIDS may be delayed by antiretroviral drugs. Because of their high cost, however, it is unlikely that these drugs will become widely available in the near future in Sub-Saharan Africa. Most important, several AIDS-indicative diseases may be prevented. Indeed, some opportunistic infections can be prevented by the use of chemoprophylaxis, and invasive cervical cancer can be prevented by proper recognition and treatment of cervical dysplasia or early cervical cancer.
The syndromic classification of STDs described above also suggests the kinds of complications that may occur. For example, fertility impairment is primarily associated with the genital discharge syndromes, and both genital discharges and genital ulcers may be prodromal to poor pregnancy outcomes and enhanced risk of sexual HIV transmission (Table 11-2).
In men, STD complications do not have the frequency, severity, or consequences they have in women. They result mainly from urethral infections, and include urethral strictures and epididymitis, an inflammation of the excretory duct of the testicle and the leading cause of male infertility in parts of Sub-Saharan Africa (Berger, 1990).
Pelvic Inflammatory Disease and Its Sequelae In women, pathogens that cause genital discharge, such as gonococci and chlamydiae, tend to rise into the upper genital tract and cause pelvic inflammatory disease. As many as 10 to 20 percent of women with untreated gonorrhea or chlamydia develop PID (Hook and Handsfield, 1990; Weström and Mårdh, 1984), the most common of the complications of sexually transmitted diseases.
Although the acute clinical manifestations of pelvic inflammatory disease may be quite severe, their public health significance resides primarily in their long-term sequelae. The partial or total occlusion of the fallopian tubes that can ensue from ID produces harsh and irreversible sequelae: infertility, ectopic pregnancy, chronic pelvic pain, and recurrent infection. Both gonococcal and chlamydial PID may be associated with those sequelae, but manifest differently. PIDs of chlamydial origin generally produce less severe symptoms than those of gonococcal cause, and they appear to produce more tubal damage and correspondingly higher rates of infertility
and ectopic pregnancy. In part, this may be caused by the more indolent character of chlamydia, which makes women with chlamydial PID less likely to seek timely treatment than women with gonococcal PID (Hillis et al., 1993).
Rates of infertility consequent to a pelvic inflammatory disease are high, even in developed countries. In a landmark study of Swedish women treated for one episode of PID, 11.4 percent became involuntarily infertile, with the risk of infertility roughly doubling with each subsequent episode; 23.1 percent of women became infertile after two episodes of PID; and 54.3 percent became infertile after three or more episodes (Weström and Mårdh, 1990). In the absence of timely, effective treatment, which is the normative circumstance in Sub-Saharan Africa, this risk becomes even greater: according to data from the preantibiotic era, 45–75 percent of women who had suffered from PID became infertile (Weström and Mårdh, 1990). Delay in PID treatment is a critical risk factor for impaired fertility; women who wait to seek health care for more than two days after onset of symptoms have a threefold increase in risk of infertility or ectopic pregnancy compared with those who seek care promptly. This risk is highest for women with chlamydial infection: 17.8 percent of those who delayed seeking health care developed impaired fertility; none of those who sought prompt treatment became so afflicted (Hillis et al., 1993).
Ectopic pregnancy is caused by partial occlusion of the fallopian tubes, in which the damaged tube hampers the passage of the fertilized ovum into the uterus. Absent emergency care, it is frequently fatal. In Sweden, women with a history of any pelvic infection have a risk of ectopic pregnancy 6 to 10 times greater than women with no such history, and the first conception following an infection leads to ectopic pregnancy in almost 10 percent of women (Weström et al., 1992). In Africa, studies from Zimbabwe and Gabon implicate both gonorrhea and chlamydia in the development of ectopic pregnancy (De Muylder et al., 1990; Ville et al., 1991), although the Gabon study shows a stronger association between chlamydia and ectopic pregnancy, which is consistent with the pattern of association suggested by the Swedish data.
While chronic pelvic pain has neither the emotional significance of infertility nor the fatal character of ectopic pregnancy, it is the most disabling of all PID sequelae, often so insidious and severe that it interferes with daily activities. It affects 15 to 18 percent of all women who have had a pelvic inflammatory disease and is often associated with infertility (Weström, 1980). In industrialized countries, up to one-third of women who have had a single episode of PID develop recurrent infections because of inadequate treatment, increased vulnerability of damaged tubes, repeated exposure to sex partners who remain untreated, or unaltered risky behaviors (Weström and Mårdh, 1990). This risk, as is the case for all the other sequelae of PID, may be much greater in Sub-Saharan Africa simply because of the pervasive lack of access to effective care.
Adverse Pregnancy Outcomes Adverse pregnancy outcomes, which include congenital or perinatal infection, low birthweight, and fetal death, are a major public health problem in Sub-Saharan Africa, and linked to both genital discharge syndromes and genital ulcer diseases. When STDs occur during pregnancy, a mother may pass the infection to her child, either during pregnancy or at the time of delivery Thus, not only the pregnant woman herself but her entire family is affected, so that she becomes the unwitting, critical link between horizontal and vertical transmission.
Congenital infections occur during pregnancy; perinatal infections occur around the time of birth. Either may result in transient illness, permanent disability, or neonatal death. In Sub-Saharan Africa, the most common and best-documented congenital or perinatal infections caused by STDs include ophthalmia neonatorum, congenital syphilis, and congenital HIV infection. Ophthalmia neonatorum, an eye infection that develops within the first month of life, is one of the most common infections of newborns related to maternal STDs, and may be caused by gonorrhea, chlamydia, or both. Easily preventable by instillation of medication in the eye at birth, untreated it may result in blindness. In a Nairobi hospital where ocular prophylaxis at birth had been discontinued, ophthalmia neonatorum occurred in 42 percent of infants whose mothers were infected with gonorrhea, and in 31 percent of infants whose mothers had chlamydia (Laga et al., 1986).
Congenital syphilis is a very serious condition, often debilitating when it is not fatal. Approximately one-third of women with syphilis are delivered of live infants with syphilis, who then go on to suffer the ravages of that disease; untreated, the disease can proceed to fatality (Schultz et al., 1990). HIV-infected babies have an even
worse prognosis, since no good therapeutic options are yet available. In a Rwanda study, 38 percent of HIV-infected infants had died by the age of 6 months (Lepage and Van de Perre, 1988a,b).
HIV-infected mothers have a risk of transmitting HIV to their babies ranging from 13 to 45 percent, with a tendency toward higher rates in Africa (Blanche et al., 1989; Hira et al., 1989; Lallemant et al., 1994; Lepage et al., 1993; Newell, 1992; Ryder et al., 1989; St. Louis et al., 1993; Tovo et al., 1988). One reason that has been postulated for the difference in transmission rates between countries is that, compared with those from industrialized countries, African HIV-infected women are generally in poor health because of poor nutritional status and frequent infections, and thus are at greater risk of transmutting HIV to their infants (Lepage et al., 1993). HIV may be transmitted during pregnancy, during birth, or through breast milk, but the relative contribution of these different modes of transmission to infection is still unclear in most African countries. A collaborative French-American study showed that the uptake of zidovudine by HIV-infected pregnant women reduces the risk of mother-to-child transmission by approximately one-third (Connor et al., 1994). The complexity of the regimen used in this study and the cost of the drug, however, make it unlikely that this kind of therapy will become available to most of the HIV-infected women of Sub-Saharan Africa in the near future.
Low birthweight, defined as live birth below 2,500 grams, occurs because of intrauterine growth retardation, premature delivery, or both. Many factors, both infectious and noninfectious—such as diet, smoking, and hypertension—may cause low birthweight. The relative contribution of each of these factors remains to be determined, and undoubtedly varies substantially according to their local prevalence. In developing countries, up to 30 percent of infants are below 2,500 grams at birth, compared with only 2 to 10 percent of infants in industrialized countries (Barnes, 1979). Because 70 percent of mortality and morbidity during the first month of life occurs among low birthweight babies (Barnes, 1979), STDs would appear to be an important factor in child survival.
Syphilis and genital herpes appear to be associated with both intrauterine growth retardation and premature delivery, while gonorrhea, chlamydia, and trichomoniasis induce premature delivery without affecting intrauterine growth (Brunham et al., 1990a; Cotch, 1990). In a study in Nairobi, Elliot and colleagues suggested that treatment of maternal gonorrhea may reduce prematurity rates by 14 percent (Elliot et al., 1990). Studies from Kenya, Rwanda, Zaire, and Zambia have identified a significant association between HIV infection and low birthweight (Braddick et al., 1990; Bulterys et al., 1991; Hira et al., 1989; Ryder et al., 1989; Temmerman et al., 1992b), which appears to derive from intrauterine growth retardation.
Fetal death, occurring either before 20 weeks of gestation (spontaneous abortion), or at or after 20 weeks of gestation (stillbirth), also appears to be associated with several types of sexually transmitted infections. Studies from industrialized countries suggest that spontaneous abortion is more frequently associated with gonorrhea or genital herpes, with stillbirth more often linked with chlamydia or syphilis (Brunham et al., 1990a; Cooper-Poole, 1986; Mtimavalye and Belsey, 1987). Schulz and his colleagues have estimated that at least 50 percent of women with untreated syphilis may lose the fetus they are carrying (Schulz et al., 1990). In Zambia, pregnant women with untreated syphilis had a risk of bearing a stillborn child 28 times higher than pregnant women without syphilis (Watts et al., 1984). In another Zambian study, 42 percent of stillbirths were attributed to syphilis (Ratnam et al., 1982), and in Ethiopia, data suggest that 5 percent of all pregnancies are lost to that disease (Bishaw et al., 1983).
Enhanced HIV Tranmission The relationships between HIV infection and other STDs are complex, intriguing, and in large measure speculative. STDs may enhance HIV transmission either by increasing the infectivity of HIV-infected persons or by increasing the susceptibility to HIV infection of non-HIV-infected persons. Several studies, mostly from Africa, indicate that both genital ulcers and genital discharge syndromes enhance the risk of HIV transmission (Cameron et al., 1989; Laga, 1990; Laga et al., 1990; Plummer et al., 1991; Wasserheit, 1992). HIV also may alter the natural course of other STDs, including their response to treatment. In Zimbabwe and Kenya, HIV-infected persons have an increased risk of treatment failure for chancroid (Latif, 1989; MacDonald et al., 1989). All of these interactions are worrisome. If co-infection with HIV prolongs or increases the infectivity of individuals with genital ulcers, and if genital ulcers facilitate transmission of HIV infection, then at the community level the two infections have the potential to greatly amplify each other (Wasserheit, 1992).
Cervical Cancer Cervical cancer is a major public health problem throughout the world, and it is the most
common cancer in Africa (Parkin et al., 1988; see also Chapter 7). It is strongly associated with several types of human papillomavirus (HPV), the etiologic agent of genital warts (WHO, 1987) and increasingly recognized as important human carcinogens.
HIV-infected women are at increased risk for cervical dysplasia, a precursor lesion for cervical cancer (Laga et al., 1992; Schafer et al., 1991; Wright et al., 1994a). Furthermore, HIV infection may adversely alter the course and treatment of cervical dysplasia and cancer (Frutcher et al., 1992; Klein et al., 1992; Maiman et al., 1990; Rellihan et al., 1990; Schwartz et al., 1991; Wright et al., 1994b). Compared with women of unknown HIV serostatus, HIV-infected women have a ninefold greater risk of recurrent or persistent cervical intraepithelial neoplasia following loop electrosurgical excision (Wright et al., 1994b).
If detected and treated early, cervical cancer is almost always curable; in the absence of early treatment, it is almost always fatal. In Sub-Saharan Africa, many women die from cervical cancer because their disease is only diagnosed at an advanced, incurable stage.
Gender Differentials in STDs and HIV Infection
Gender is linked to biological and behavioral factors that contribute to crucial differences in the acquisition, course, and consequences of STDs. Compared with men, women are less likely to have control over the circumstances of their sexual activity. If they have sex with an infected partner, they are more likely to acquire an STD, including HIV infection; if they have an STD or HIV infection, they are less likely to seek health care; if they seek health care, they are less likely to be treated effectively, are more likely to develop complications, and, finally, frequently pay a heavier social toll for STDs, including HIV and their sequelae. These individual factors operate against a backdrop of such societal features as the low-status of women and the high ratios of males to females in urban areas. These societal factors clearly influence sexual behavior and are discussed in greater detail below.
Control Over the Circumstances of Sexual Intercourse
Gender typically influences economic and social status and, in most societies, women's roles are defined primarily by their sexual and reproductive relationships with men, relationships that place severe limitations on the extent of women's control of their own sexuality (Aral, 1990). Compared with men, in most societies women also have fewer options about when, where, how, and with whom they will have sex.
Acquisition of STDs and HIV Infection and Development of Complications
Gender affects the efficiency of transmission of some STDs. Transmission of STD pathogens that produce discharge or are present in genital secretions—such as gonococci, chlamydiae, trichomonads, and HIV—appears to be more efficient from male to female than vice versa, at least partly because of the prolonged exposure to organisms when infected ejaculate is retained in the vagina. In contrast, the transmission of syphilis, chancroid, and genital herpes, all of which cause genital ulcers, differs little by gender. This may be because transmission of these STDs depends on small breaks in the genital skin that probably occur in both sexes during vigorous coitus. Sexual behaviors may also synergize with biological factors to promote STD transmission. For example, the practice of ''dry sex" (use of intravaginal desiccants to increase friction during coitus) in some parts of Sub-Saharan Africa—such as Malawi, Zaire, and Zambia (Brown et al., 1992; Dallabetta et al., 1990; Nyirenda, 1992)—frequently causes breaks in the vaginal surface (Brown et al., 1992), which may enhance the risk of acquiring HIV infection and other STDs. Finally, unavailability of female-controlled barrier methods of birth control restricts women's ability to protect themselves against all STDs, including HIV infection.
Health-Seeking Behavior
Another gender-related consequence of STDs is the exacerbation of their sequelae brought about by differences in health-seeking behavior. Although in some industrialized countries women are more likely than men to
seek health care and subsequently receive appropriate treatment, this is rarely the case in Sub-Saharan Africa (Ehrhardt and Wasserheit, 1991). First, women may not suspect that they have an STD. Indeed, women with STDs are more frequently asymptomatic than men: 50 to 80 percent of women infected with gonorrhea are asymptomatic (Jones and Wasserheit, 1991), compared with 20 to 40 percent of men (Rothenberg and Potterat, 1990). In addition, when STD symptoms are present in women, they are often subtle, and may resemble changes that normally occur during the menstrual cycle, such as increased vaginal discharge, lower abdominal pain, and vaginal bleeding. Finally, cultural factors may influence the way women perceive the significance of their symptoms. For example, in regions where women are circumcised, practice "dry sex," or where STDs are widespread, pain during coitus, irregular vaginal bleeding, or pelvic pain may be considered the norm.
Another major reason for gender differentials in seeking care for STDs is the female-specific, stigmatizing character of STDs, including HIV infection. Women face very strong sociocultural barriers to STD care, which are frequently fueled by the judgmental attitudes of health care providers. Finally, financial barriers for accessing care for HIV and other STDs may be stronger for women than for men, because women are frequently either of lower socioeconomic status than men or do not have independent financial resources.
Detection and Treatment of STDs
Even if they do seek health care for STDs, women are less likely than men to receive correct diagnosis and adequate treatment, for several reasons. First, most health care providers have limited expertise in STD management in women; they also share a common misconception that STDs affect only promiscuous women and prostitutes. Second, as discussed earlier, because STD signs and symptoms in women are often subtle and nonspecific, clinical diagnosis is not reliable. Third, in many Sub-Saharan African settings, laboratory tests are not available, even though it is women who would benefit most from laboratory tests because of the poor predictive value of female STD syndromes, particularly for vaginal discharge. Finally, the increased antimicrobial resistance of many pathogens to older antibiotics such as penicillin requires that new, more expensive drugs be used. Once again, women may be disproportionately affected: most sexually active women in Sub-Saharan Africa are either pregnant or breastfeeding at any given time, yet antibiotics that are effective and safe under those conditions may be neither available nor affordable.
The Social Implications of STDs and HIV Infection
Both STDs and their complications have far greater social significance for women than they do for men. Uncomplicated STDs typically cause personal embarrassment and domestic conflicts, but whoever is responsible for bringing a given infection into the relationship, it is the woman who is typically blamed and who endures the most serious consequences; these may include violence, divorce, and social ostracism. The complications of STDs, especially impaired fertility, are even more devastating. Although male infertility scientifically explains about one-third of all infertility, women are customarily blamed when a couple cannot have children. In parts of Africa, it is not uncommon for the female of an infertile couple to look for another male to make her pregnant (Rob et al., 1987), out of fear of being rejected by her husband and in-laws and becoming a social outcast. As a result, many infertile men may never become aware of their infertility. For women, childlessness, whether through infertility or poor pregnancy outcome, can be a major tragedy. Not only does it cause personal pain for the women themselves, but it becomes a paramount infirmity in a society that values women primarily for their ability to produce healthy offspring. Infertile women may be divorced by the same husbands who infected them with the STD that produced the infertility. The same is true of HIV-infected women, who are at risk of abandonment by the very men who transmitted the infection (de Bruyn, 1992). Ostracized by society, divorced women have few survival options other than prostitution, which increases STD transmission and the risk of infertility in the larger community, creating a vicious cycle of disease; misery; and, increasingly, death.
In this way, STDs and their complications affect individuals, communities, and, eventually, whole societies. In some areas, notably the so-called "infertility belt," which extends from Gabon in the west of Africa to southwestern Sudan in the east, STDs have had a dramatic impact on fertility rates. Brunham estimates in a mathematical
model that a 20 percent gonorrhea prevalence in sexually active adults may produce up to a 50 percent reduction in net population growth, an estimate consistent with fertility levels actually observed in some parts of Uganda (Brunham et al., 1991). The consequences of HIV infection are even more striking. In another mathematical model, Anderson predicts that in some Sub-Saharan African countries, HIV infection may reverse demographic growth from positive to negative over a few decades (Anderson et al., 1988).
Yet another dramatic—and paradoxical—effect of STDs on society is their potential for jeopardizing family planning programs. STDs may decrease acceptance and continuation of contraceptive methods in two ways: directly, by creating the perception of a contraceptive side effect, and indirectly, by creating a fear of fertility impairment in the face of complications of STDs. Numerous studies in Bangladesh have found that the most common reason given for discontinuing a contraceptive method is the perception of a method-associated side effect (Akbar et al., 1981; Bhatia, 1982; Jain and Sivin, 1977; Rob et al., 1987). Similar findings have turned up all over the globe and can be extrapolated comfortably to other settings. Absent accurate diagnosis and effective education and therapy, it is far easier for a woman to blame a vaginal discharge on her current contraceptive method than to confront the possibility of having been infected by her husband—who is then, by definition, unfaithful. The net result is that the woman stops using her contraceptive method in the mistaken belief that if caused the unrelated infection (Wasserheit, 1989).
The indirect impact of STDs on family planning is equally important. Several authors have postulated that high levels of infertility or frequent adverse pregnancy outcomes might result in a compensatory decrease in acceptance of family planning methods (O'Reilly, 1986; Rosenberg et al., 1986). In societies that value children highly, couples are unlikely to undertake voluntary fertility regulation unless they are confident that they will be able to raise as many healthy children as they desire (Wasserheit, 1989).
STDs and HIV Infection Throughout the Female Life Span
The relative contribution of different risk factors for STDs, including HIV infection, varies with age. Increased vulnerability to STDs may be the result of biological or behavioral factors, and these may go in the same or in the opposite direction. In adolescence, for example, biological and behavioral factors act concurrently to increase dramatically the risk of HIV infection and other STDs and their complications. In later years, however, these factors may have different weights. In the adult, biological factors tend to decrease in importance and to be outweighed by behavioral factors. In the elderly, while physiological changes may facilitate the transmission of HIV and other STDs, behavioral factors tend to be protective (Ehrhardt and Wasserheit, 1991).
The striking changes that occur in cellular morphology in the vagina and cervix over the female life span have a direct effect on susceptibility to STDs and on the spectrum of infections found at each stage. During the first few weeks of life, the vagina and cervix of the newborn are lined by squamous epithelial thick cells that are relatively resistant to infection. From one month until menarche, changes in this lining increase susceptibility to STDs, especially to chlamydia and gonorrhea. Then, beginning at puberty, under hormonal influence, the cervico-vagina vault will again be covered by a thicker layer of squamous cells that are more resistant to some infections. The junction between squamous cells and the columnal cells that are the site of attachment for chlamydial and gonococcal infection is called the zone of ectopy. In the adolescent, the zone of ectopy is found on the surface of the cervix, where it is particularly susceptible to chlamydia, gonorrhea, and HIV. In young adulthood, the zone will usually migrate from the surface of the cervix to a less-exposed position in the cervical canal, reducing susceptibility to those infections (Cohen et al., 1985).
Sexuality and sexual behavior are forbidden subjects in many societies. As a result, real data on these topics are quite limited, particularly in developing countries. Most of our knowledge about sexual behavior comes from studies in industrialized countries, and it may not be applicable to Sub-Saharan Africa. In addition, sexual behavior in Sub-Saharan Africa is likely to vary a great deal across its many cultures and countries. At the same time, biological factors are more likely to be similar across cultures. This section enlarges upon the key biological and behavioral risk factors that are unique to each stage of the female life span.
Infancy, Childhood, and Adolescence
Infancy During infancy, STDs, including HIV infection, are rarely transmitted sexually unless sexual abuse has occurred. Instead, they result from intrauterine or perinatal transmission of maternal infections. Behavioral risk factors for congenital infections therefore concern the infant's mother.
The consequences of congenital and perinatal infections associated with STDs and HIV infection include, at the extreme, blindness and other physical disabilities, mental retardation, and death. All the morbidities may have continuous, severe consequences throughout childhood, adulthood, and senescence. In addition, congenital infections affect not only the baby, but also its mother, who has to care for the sick or disabled child, perhaps for her remaining lifetime. Also, the children born to HIV-infected mothers, whether infected themselves or not, will, if they outlive their parents, become orphans, representing an extra burden for the older members of the community, often the grandmothers.
Childhood STDs and HIV infection during childhood are relatively rare, and result almost entirely from sexual abuse. Sexual activities during childhood are exploratory and sporadic; while such sexual play between children of the same age is normative, sexual behavior between adults and children is not. Sexual abuse of children by strangers or by family members has received very little attention in Sub-Saharan Africa, and may well merit more. There are no studies of the region that would permit evaluation of the extent of emotional injury and long-term impact on the subsequent psychosexual development of sexually abused children or the magnitude of the problem. Studies from industrialized countries indicate that the extent of trauma varies and that the effects are nonspecific and may influence later sexual, emotional, or substance abuse behavior (Browne and Finkelhor, 1986). Sexual abuse of children may be more likely than rape of adult women to result in STD and HIV transmission because penetration is more likely to be traumatic in children.
Adolescence This is the period when behavioral and biological factors combine to maximize the risk for STDs and HIV infection. As noted, during puberty and adolescence, several physiological changes occur in the female genital tract that result in a particularly high-risk. In addition, adolescence is a period of life in which many women have their first sexual encounter, and it is the time when sexual behavior patterns become established. Furthermore, when the initial sexual encounter is traumatic, it may be a high-risk event for male-to-female HIV transmission (Bouvet et al., 1989). Age at sexual debut (time of first sexual intercourse) and sexual activities during adolescence follow secular trends and also vary greatly according to culture. Studies indicate that an earlier age of sexual debut is associated with higher total numbers of sex partners over a lifetime. In Ethiopia, where child marriage is practiced, half of 2,111 women surveyed had experienced their first sexual intercourse before the menarche (Duncan et al., 1990). That same early sexual activity was associated with an increased prevalence of STDs, PID, and cervical cancer later in life. The study also indicates that the younger the age at first marriage or first coitus, the shorter the duration of the marriage and the greater the likelihood of divorce (Duncan et al., 1990).
The extent of sexual assault in Sub-Saharan Africa is unknown. As in other parts of the world, rapes are rarely reported (see also Chapters 2 and 8). Instead, the incident is often settled between the families involved through compensation, in cash or goods, by the perpetrator to the family of the woman raped. There is evidence that rape is a serious problem in parts of Sub-Saharan Africa. In Kenya, when 71 teenage girls were raped during one night in the dormitory of a boarding school, a Kenyan newspaper described the incident as a common occurrence, and as sanctioned by the principal and his staff (Heise et al., 1994). There is a good chance that sexual abuse is a menace for African women throughout their lives. In South Africa, as one example, the incidence of rape appears to exceed rates in the United States, with an annual incidence of 34 rapes per 1,000 women, compared with 18 per 1,000 women in the United States (Heise et al., 1994).
Finally, the limited health knowledge and constrained health-seeking behavior of adolescents compound the impact of physiological factors on STD and HIV infection morbidity. Many adolescents are unable to recognize or to understand the significance of STD or HIV symptoms and have little or no independent access to health care, or are afraid to utilize existing services.
Adulthood In adulthood, behavioral factors dominate the risk for STDs and HIV infection and are characteristically tied to the sex partner's sexual behavior. The physiological changes that affect the risk for STDs, including HIV infection, during the adult reproductive years are related primarily to the menstrual cycle, pregnancy, and contraceptive use.
The menstrual cycle seems to play a key role in the risk of PID. Symptomatic gonococcal and chlamydial PID appear to occur more frequently during the first week of the menstrual cycle, probably because of the retrograde flow of menstrual blood from the uterus into the fallopian tubes. During pregnancy, although the lower genital tract, particularly the cervix, may be more susceptible to infections, the upper genital tract appears to be protected from infections by gestational anatomical changes.
Sex during menses has been hypothesized, although not shown, to be a risk factor for female-to-male transmission (De Vincenzi, 1994). Although there is no evidence thus far to suggest that pregnancy aggravates the course of HIV infection, several studies have documented the occurrence of serious HIV-related infections during pregnancy (Selwyn and Antoniello, 1993).
Contraceptives may play a prominent part in altering the risk of HIV and other STDs during the reproductive years, and even beyond. Barrier contraceptive methods such as condoms and diaphragms serve as a mechanical barrier to pathogens. Oral contraceptive pills seem to increase risk of cervical chlamydial infection, but appear to decrease the frequency and severity of PID. Users of intrauterine devices have increased risk of developing PID, particularly during the first four months following insertion, and this risk varies by geographic area; Africa has the highest rates of all the world's regions (Farley et al., 1992).
Behavioral risk factors for STDs include sexual and health-seeking behavior. Because men tend to have more partners and engage in riskier sexual behavior than women, women's risk for STDs, including HIV infection, is more closely related to their husbands' sexual behavior than their own. Nonsexual transmission of STD pathogens present in the blood—such as HIV, treponemes (which cause syphilis), and hepatitis B virus—may be a concern for health workers, particularly for midwives and traditional birth attendants, who are usually women and who frequently come into contact with abundant quantities of blood.
The lifelong impacts of STDs and their long-term sequelae are tremendous, and were described earlier. Obviously, the earlier in life those sequelae occur, the longer women have to bear their consequences. In addition, women may be more susceptible to such consequences, notably the psychological and social repercussions, in part because they are affected by HIV and other STDs at an earlier age than men.
Maturity and Senescence As people become older, their sexual activity often decreases as a result of physiologic changes and societal influences. During senescence, increased biological risk for STDs compensates for decreased behavioral risk.
The physiologic changes that occur in women include atrophic changes in the vagina, with thinning of the epithelium, reduced lubrication, and narrowing and shortening of the vaginal canal. A vagina that is dry can be injured more easily by penile penetration (Holmes, 1990; Mooradian and Greiff, 1990), placing a woman at greater risk for transmission of HIV infection and other STDs from an infectected partner.
Sexual behavior at older ages is determined by sexual interest, which is influenced, in turn, by the hormonal environment and sociocultural factors. The level of sexual activity among older people, particularly women, varies greatly across cultures. While sexual activity is socially acceptable or desirable for older men, it may not be so for older women. In Sub-Saharan Africa, for example, the limited available evidence suggests that many women cease to be sexually active at menopause.
A different, yet major aspect of the burden of the HIV epidemic on women, including older women, is the care of the sick. Indeed, it is women who—as health care workers, community members, or family members—provide most of the care for people with chronic illnesses, including HIV infection and AIDS (Richardson, 1989).
MAGNITUDE OF THE PROBLEM
Morbidity
This section provides general information on the extent of the problem of sexually transmitted diseases, including HIV infection, in Sub-Saharan Africa and estimates the prevalence of five common STDs across the countries of the region.
There are noteworthy differences in the magnitude of the STD burden between the industrialized and the developing countries. First, sexually transmitted diseases including HIV infection are much more prevalent in most developing countries, particularly in Sub-Saharan Africa, than in the majority of industrialized countries. Second, developing countries, again particularly those in Sub-Saharan Africa, have a higher incidence of complications of STDs than industrialized countries experience, and these complications are often more severe. Third, different kinds of STDs predominate in developing and industrialized countries. For example, the proportion of STDs characterized by genital ulcers is greater in developing countries than in industrialized countries; in Sub-Saharan Africa, chancroid and syphilis account for the majority of genital ulcers, while in industrialized countries, herpes is the main cause of those lesions (Meheus et al., 1990). Finally, while in industrialized countries HIV infection is often transmitted through homosexual contact and injecting drug use, and affects predominantly males, in Sub-Saharan Africa, HIV infection is almost exclusively transmitted heterosexually and affects both women and men equally, except for substantial mother-to-child transmission.
The following statistics highlight the severity of the problem in Sub-Saharan Africa.
-
STDs are among the top five reasons for clinic attendance in many Sub-Saharan African countries (Meheus, 1990). For example, STDs account for 13 percent of adult outpatient visits in Zimbabwe (Zimbabwe, 1989), and for 5 to 10 percent of outpatient visits in Zambia (Hira et al., 1992b). In some countries, PID is the most common diagnosis among women attending STD clinics. In Zimbabwe, 47 percent of women attending an STD clinic had a pelvic inflammatory disease (Latif, 1981), and in Ethiopia at least 39 percent of gynecological outpatients had signs or symptoms suggestive of PID (Perine et al., 1980). Even though many women with PID are never diagnosed and the majority are not hospitalized (Carty et al., 1972), it is one of the leading causes of gynecological admission (Brown and Cruickshank, 1976; De Muylder, 1989; Perine et al., 1980; Ratnam et al., 1980b; Zacarias and Aral, 1985).
-
In many countries of Sub-Saharan Africa, where the prevalence of syphilis among pregnant women is at least 10 percent, Schulz and colleagues estimate that 5 to 8 percent of all pregnancies that last beyond 12 weeks will fail to produce a healthy infant because of congenital syphilis, or syphilis-related fetal death or infant death (Schulz et al., 1987).
-
In some Sub-Saharan African countries—for example, Cameroon, Ethiopia, and Kenya—as many as 4 to 6 percent of newborn babies develop gonococcal ophthalmia, a condition that, untreated, leads to blindness (Galega et al., 1984; Lepage and Van de Perre, 1988a; Muhe and Tafari, 1986).
-
In Sub-Saharan Africa, HIV infection has not only reached high levels of prevalence—overall, affecting one in 40 adults, and in certain cities as many as 30 percent of pregnant women (see Appendix, Table 11-8)—but is also rapidly spreading, particularly in urban areas and among selected subgroups, such as prostitutes (Nkowame, 1991).
-
In several African cities, AIDS is the leading cause of death among young women and men.
Prevalence
Estimates of the prevalence of HIV infection and other STDs among women in Sub-Saharan Africa must be made cautiously. Although more information on STDs is available in Sub-Saharan Africa than in other parts of the developing world, the quantity, quality, and heterogeneity of data across countries and populations limit the ability to precisely define the extent of STD morbidity in the region. There is virtually no surveillance system in Sub-Saharan
Africa to assess incidence, and most available data derive from prevalence studies in family planning, antenatal, and STD clinics. For certain diseases and syndromes—gonorrhea, chlamydia, trichomoniasis, syphilis, and HIV infection—data are relatively abundant, while for others—chancroid, genital ulcers, PID, cervical cancer, adverse outcomes of pregnancy, and ectopic pregnancy—data are patchy at best.
Despite these constraints, it is possible to estimate the burden of STDs across Sub-Saharan African countries by examining five common diseases: gonorrhea, chlamydia, trichomoniasis, syphilis, and HIV infection. Prevalence data have been compiled for each disease by country, for low-risk, high-risk, and very-high-risk populations. The three populations were defined as follows: (1) low-risk populations were women attending antenatal clinic or family planning clinics, or women sampled in community-based studies who are probably fairly representative of the general population; (2) high-risk populations were women attending STD clinics, or women with symptoms suggestive of STDs who were attending other clinical facilities; and (3) very-high-risk populations were women engaged in prostitution. To obtain sufficient data for prevalence estimates that could be considered moderately robust, the authors reviewed studies from the past 4 years for HIV, and for the past 20 years for the other STDs.
For all STDs except HIV infection, disease-country-population-specific prevalence was estimated by taking the median value when data from more than one study were available for a given country; the assumption was that a single study represented the country prevalence in countries that had only one disease-population-specific study. For HIV1 infection, because the number of studies varies tremendously across countries and across regions within countries, a slightly different approach was used to avoid biasing estimates toward countries or regions with the highest number of studies. To estimate the population-specific HIV prevalences by country, the authors first calculated population-specific median HIV prevalences by region within a given country, and then took the median of those medians for each country. For all five diseases, to estimate the disease-population-specific prevalences for Sub-Saharan Africa, the median of all the country-specific median prevalences was taken.
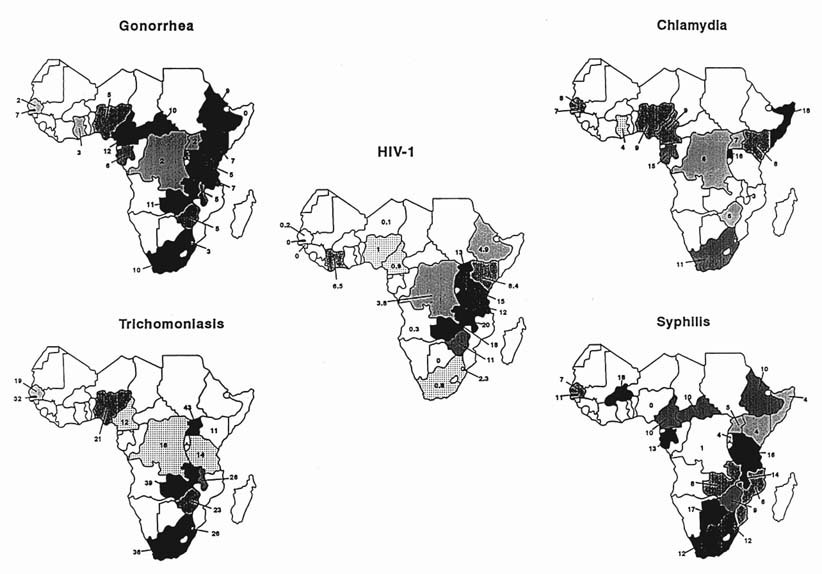
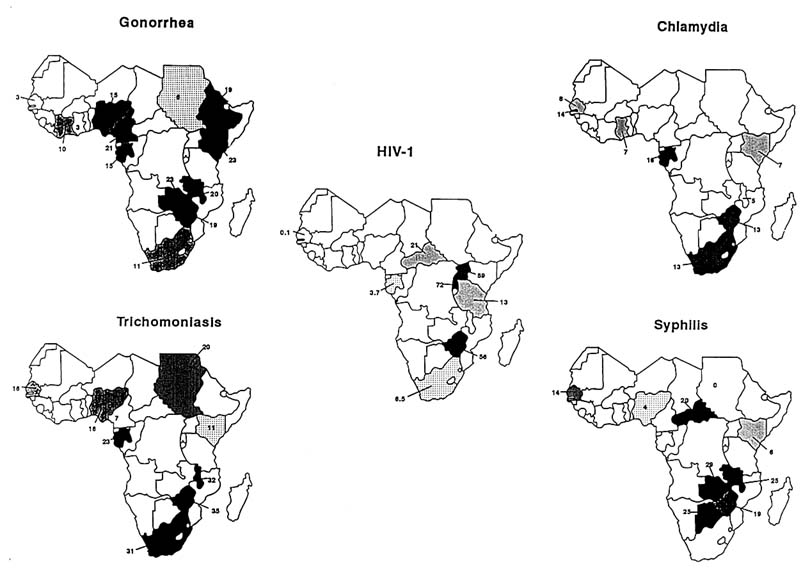
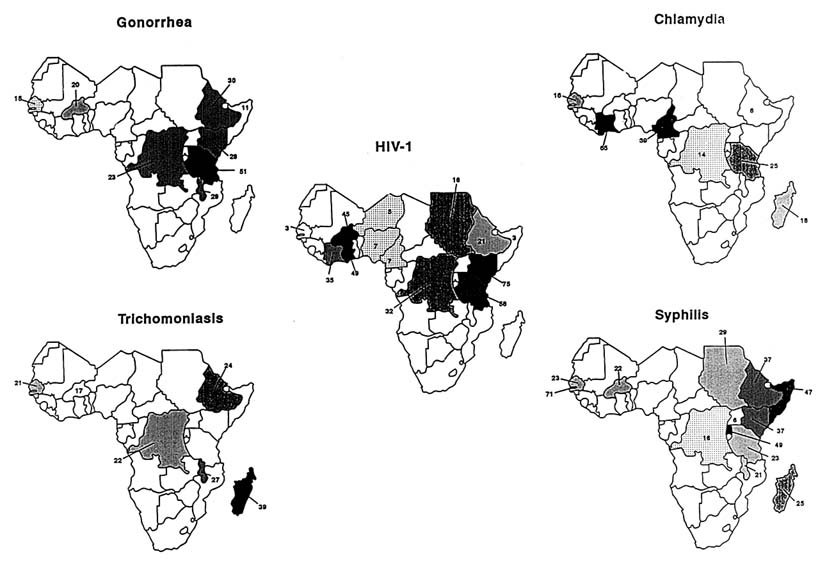
The individual study prevalences and the median country prevalences reveal that: (1) the amount of data varies greatly between countries; (2) most studies concern HIV infection; there are relatively few data on other STDs; (3) there are large variations in disease prevalence, within and among countries; and (4) prostitutes constitute a major reservoir of STDs. Furthermore, the prevalences of STDs, including HIV infection, are generally higher in Sub-Saharan Africa than in industrialized countries, even among low-risk populations. In addition, in most Sub-Saharan African countries, as in the industrialized countries, chlamydia is more prevalent than gonorrhea in populations considered at low-risk (Appendix Tables 11-6, 7, and 8 and Tables 11-3 and 11-4, below).
Table 11-4 shows the median STD and HIV prevalences for Sub-Saharan Africa. Because of the heterogeneity among countries, these figures are necessarily crude, and almost meaningless when considered as absolute quantities. Nevertheless, they are useful in assessing the relative importance of "core groups" in the transmission of the five STDs in the region as a whole. Core groups are defined as groups of individuals who are highly sexually active, and thus contribute disproportionately to disease transmission in a given population (Yorke et al., 1978). Core groups usually include prostitutes, their clients, and other individuals who have multiple sex partners, such as those in the military and long-distance truck drivers. The ratio of STD prevalence in prostitutes to the STD prevalence in low-risk populations can be derived from Table 11-4, which shows diminishing rates per 100 women for HIV infection (7), gonorrhea (5.2), syphilis (2.5), chlamydia (2.3), and trichomoniasis (1). These descending values suggest that core groups in Sub-Saharan Africa play a critical role in the transmission of HIV infection and gonorrhea, but are less important in the transmission of syphilis and chlamydia, and have little influence in the transmission of trichomoniasis. The role of the core group inferred through this analysis of STD is consistent with what has been postulated elsewhere, with the exception of syphilis, which elsewhere has been highly associated with core groups. This discrepancy may be brought about by the measurement of syphilis by syphilis serology in the data being used here, a measure that indicates either a past or a current infection. In addition, while compared with the general population, core groups may have a much higher prevalence of current infectious syphilis, their prevalence of positive syphilis serology may not be significantly greater.
STD and HIV prevalence among high-risk groups is significantly correlated with STD and HIV prevalence in low-risk groups (p < 0.05), using Pearson's correlation coefficient. Data on persons attending STD clinics must be examined with caution, however, because various health services in different countries may attract very heterogeneous populations that may not be comparable across countries.
TABLE 11-3 Median STD Prevalences (median prevalence per 100 women)
|
Low-Risk Population |
High-Risk Population |
Very-High-Risk Population |
|||||||||||||
|
Country |
GC |
CT |
TV |
TP |
HIV |
GC |
CT |
TV |
TP |
HIV |
GC |
CT |
TV |
TP |
HIV |
|
Angola |
0.3 |
||||||||||||||
|
Botswana |
17 |
0 |
25 |
||||||||||||
|
Burkina Faso |
18 |
20 |
17 |
22 |
45 |
||||||||||
|
Central African |
10 |
10 |
20 |
21 |
|||||||||||
|
Republic |
|||||||||||||||
|
Cameroon |
12 |
9 |
12 |
10 |
0.9 |
21 |
7 |
39 |
7.1 |
||||||
|
Côte d'Ivoire |
6.5 |
10 |
65 |
35 |
|||||||||||
|
Ethiopia |
9 |
10 |
4.9 |
19 |
30 |
6 |
24 |
37 |
21 |
||||||
|
Guinea Bissau |
0 |
||||||||||||||
|
Gabon |
6 |
15 |
13 |
15 |
16 |
23 |
3.7 |
||||||||
|
Gambia, The |
7 |
7 |
32 |
11 |
0 |
14 |
71 |
||||||||
|
Ghana |
3 |
4 |
3 |
7 |
49 |
||||||||||
|
Kenya |
7 |
8 |
11 |
4 |
8.4 |
23 |
7 |
11 |
6 |
28 |
37 |
75 |
|||
|
Madagascar |
18 |
39 |
25 |
||||||||||||
|
Malawi |
5 |
3 |
26 |
14 |
20 |
20 |
5 |
32 |
25 |
29 |
27 |
21 |
|||
|
Mozambique |
6 |
||||||||||||||
|
Niger |
0.1 |
4.9 |
|||||||||||||
|
Nigeria |
5 |
9 |
21 |
0 |
1 |
15 |
16 |
4 |
6.5 |
||||||
|
Rwanda |
5 |
16 |
4 |
15 |
72 |
49 |
|||||||||
|
Senegal |
2 |
8 |
19 |
7 |
0.2 |
3 |
8 |
16 |
14 |
0.1 |
15 |
16 |
21 |
23 |
3.4 |
|
Somalia |
0 |
18 |
10 |
11 |
47 |
3 |
|||||||||
|
South Africa |
10 |
1 |
36 |
12 |
0.8 |
11 |
13 |
31 |
6.5 |
||||||
|
Sudan |
6 |
20 |
0 |
29 |
16 |
||||||||||
|
Swaziland |
3 |
26 |
12 |
2.3 |
|||||||||||
|
Tanzania |
7 |
14 |
16 |
12 |
13 |
51 |
25 |
23 |
58 |
||||||
|
Uganda |
2 |
7 |
43 |
5 |
13 |
59 |
6 |
||||||||
|
Zaïre |
2 |
6 |
16 |
1 |
3.8 |
23 |
14 |
22 |
16 |
32 |
|||||
|
Zambia |
11 |
39 |
8 |
18 |
23 |
29 |
|||||||||
|
Zimbabwe |
5 |
6 |
23 |
9 |
11 |
19 |
13 |
35 |
19 |
65 |
|||||
|
NOTE: GC, gonorrhea; CT, chlamydia; TV, trichomoniasis; TP, syphilis; HIV, HIV infection type 1. Low-risk population includes women attending antenatal or family planning clinics or women sampled in community-based studies. High-risk population includes women attending STD clinics, or women with symptoms suggestive of STDs. Very-high-risk population includes women engaged in prostitution. Median prevalences are calculated using data from the Appendix. |
|||||||||||||||
TABLE 11-4 Median STD Prevalences in Sub-Saharan Africa (per 100 women)
|
Disease |
|||||
|
Population |
Gonorrhea |
Chlamydia |
Trichomoniasis |
Syphilis |
HIV Infection |
|
Low-risk |
5 |
8 |
23 |
10 |
3 |
|
High-risk |
15 |
11 |
20 |
19 |
17 |
|
Very-high-risk |
26 |
18 |
23 |
25 |
21 |
|
NOTE: Low-risk population includes women visiting antenatal or family planning clinics or women sampled in community-based studies. High-risk population includes women attending STD clinics or women with symptoms suggestive of STDs. Very-high-risk population includes women engaged in prostitution. Median prevalences are calculated using data from Table 11-3. |
|||||
A mapping of country prevalences (Figures 11-1, 11-2, and 11-3) reveals a clustering of diseases: highest prevalences occur in eastern and southern Africa, and in a limited region of central Africa that includes Cameroon and the Central African Republic. The pattern of gonorrhea prevalences resembles that of syphilis, although the correlation between the prevalences of these two diseases was not statistically significant. The highest prevalence of HIV infection occurs in East Africa and in the northern part of southern Africa. The very high prevalence of STDs in other regions, particularly in southern Africa, in Cameroon, and the Central African Republic, suggests that those regions have the potential for very rapid spread of HIV. Nevertheless, caution is advisable in predicting where the next wave of the HIV epidemic will strike, since there are almost no data from several large parts of Sub-Saharan Africa, notably from many countries of western Africa.
Mortality
The burden of diseases has traditionally been measured in mortality. As health interventions evolve and disease patterns shift, other indicators, such as morbidity and quality of life, must also be used to measure disease impact and to set health priorities. Indeed, measuring the burden of diseases solely by mortality may mask the extent of a problem. The sexually transmitted diseases are an excellent example: the disease burden derived from STDs is primarily the product of frequent and severe morbidity rather than mortality. Nevertheless, of the six major STD complications, four (ectopic pregnancy, adverse pregnancy outcomes, cervical cancer, and enhanced HIV transmission) are potentially fatal, and HIV infection itself has an extremely high mortality rate, and it is now a major cause of death among young adults in some regions of Africa. In Abidjan, Côte d'Ivoire, AIDS is the leading cause of death in adult men, and in a study in Rwanda, approximately 90 percent of mortality among childbearing urban women is attributed to HIV infection (De Cock et al., 1990; Linden et al., 1992).
Economic Impact
The cost attributable to a disease is the sum of direct costs (drugs, hospitalization, transportation, provider payments) and indirect costs (loss of productivity and physical, psychological, and social impacts) (Drummond et al., 1989). Indirect costs are extremely difficult to quantify, primarily because estimating physical, psychological, and social costs requires culture-specific value judgments. They are, however, crucial.
The general level of health care provided in a country corresponds largely, though not totally, to its level of economic development. In the case of STD care, for instance, laboratory tests (even inexpensive microscopic examinations) are unavailable in most Sub-Saharan African countries. It is crucial, then, to take opportunity costs into consideration, as well as the actual number of dollars spent, when estimating costs and comparing them across countries.
Although few data are available on the direct costs of STDs, the magnitude and complexity of that group of diseases, including the frequent need for hospitalization, indicates that they are very high. In addition, costs of
case management of STDs have markedly increased with the emergence of antimicrobial resistance and correspondingly poor response to STD treatment in HIV-infected persons. Poor countries now have to spend their scarce foreign currency to import costly antibiotics. In addition to the cost of Western medicine, people in Sub-Saharan Africa spend a considerable amount of money on traditional medicine to treat STDs and their sequelae.
The largest economic burden of STDs is not primarily the direct financial cost of treating diseases, but rather its indirect costs. Over and Piot have estimated the cost of the STD burden (including HIV infection) in healthy life-years lost per capita each year relative to other diseases in Sub-Saharan Africa. They found that in urban areas with high prevalences,2 STDs account for up to 17 percent of productive healthy life-years lost and are second only to measles in their disease burden (Over and Piot, 1991). Because this model omits consideration of the devastating psychological and social costs of infertility in women, the total economic burden of STDs in Sub-Saharan Africa is higher. Treatment for infertility in Sub-Saharan Africa is very costly and failure rates are high, but what may appear to be an irrational expenditure of scarce resources is, instead, hard evidence of the high psychological and social costs of infertility and, by extension, the diseases that cause it.
SOCIETAL DETERMINANTS OF STDS AND HIV INFECTION
Several societal factors have been postulated as determinants in the transmission of STDs, including HIV infection: population-age composition, sex ratio, urbanization, population mobility, war and social unrest, sexual norms, women's social status, and the availability of health (Aral, 1990; Brunham and Embree, 1990). Clearly such factors are interrelated and will change, independently and in association, in the course of socioeconomic development. The impact of each factor on STDs and HIV infection will therefore be time-dependent: the effect of one factor may be contingent on changes in the other factors.
Population-Age Composition
The population of Sub-Saharan Africa is characterized by a youthful age composition and fertility rates that are higher than in any other major region of the world (Cohen, 1993). In parts of the developing world, the marked decline in childhood mortality resulting from advances in medical sciences, childhood immunization, and improved sanitation has not been paralleled by a comparable decline in natality. This is particularly true for Sub-Saharan Africa, where birth rates have not changed since 1950 (Caldwell and Caldwell, 1990), although this figure masks considerable variation among regions and individual countries (Cohen, 1993).
The age composition of a community can influence STD and HIV rates in several ways. First, a broad-based age pyramid, dominated by young people, implies a large and increasing proportion of the population in its sexually active years. Second, all STDs are significantly age-related, with the highest STD and HIV rates occurring in young adults. Third, such a population structure may hamper the ability of the older generation to guide the younger generation's sexual norms and behaviors. Fourth, the relative scarcity of older men may increase promiscuity between a few older men and many younger women, given that sexual union between older men and younger women is almost universally accepted and practiced (Aral, 1990).
Urbanization, Sex Ratio, and Population Mobility
Rapid urbanization is a cardinal demographic feature of virtually all developing countries. In Africa, that phenomenon rests on the base of the region's urban centers, which were created during the colonial period. In many of these centers, only male workers were allowed, and the numerous waves of migration since independence have consisted mainly of young men looking for work (Larsen, 1989). Agriculture and mining have generated dormitory townships where men stay temporarily, sometimes for months, without going back to their homes, leaving wives and families in their rural villages. These cumulative processes have created a marked disequilibrium between the numbers of men and women in the region's urban settlements. In Nairobi and Harare, as just two examples, there are 50 to 80 percent more men than women. Social movement between status groups is another important factor that promotes anonymity, loosens societal structure, and, in turn, increases risky sexual behavior.
Population mobility is not solely a phenomenon brought about by urbanization and industry. In traditional societies, migration is often seasonal and is associated with herding and agriculture. In Botswana, for instance, Tswanas may have as many as four homes to accommodate seasonal migrations. In many areas, particularly in urban centers, the relative scarcity of women, combined with population mobility, poverty, and social inequities, creates an environment all too conducive to prostitution, thereby swelling the high-risk core group.
War and Social Unrest
War and social unrest not only engender geographic and social mobility, but add extreme levels of mental and physical violence. In Mozambique, between 1975 and 1989, an estimated 1.6 million people (10 percent of the population) were displaced by the frequent RENAMO (Mozambique National Resistance) attacks on rural populations (Cliff, 1991). Gersony reports that 15 percent of refugees reported systematic rape of civilian women by RENAMO combatants, and in areas controlled by RENAMO (Gersony, 1988):
[a] function of the young girls and women is to provide sex for the combatants…these women are required to submit to sexual demands, in effect to be raped, on a frequent, sustained basis.… One of the frequent refugee complaints (verified by medical workers in some of the refugee camps) is the level of infection with venereal diseases which this practice proliferates. Severe beating is inflicted on young girls and women who resist sexual demands.
Sexual Norms, Women's Social Status, and Patterns of Sexual Behavior
Perhaps because sexuality is such a fundamental element of human life, it is complex, sensitive, and somewhat hidden, so that the gap between proscribed sexual norms and sexual practices can be quite wide (Holm's and Aral, 1991). They are undoubtedly interrelated, nevertheless, and both are influenced by the status of women.
Larsen (1989) has described two patterns of sexual behavior in Sub-Saharan Africa. Schematically, the first pattern characterizes societies of patrilineal descent, in which women tend to be socioeconomically dependent on men, and widows are inherited by their husbands' kin. In such a situation women have little control over their lives and very few sex partners, while men have sex with several women, usually prostitutes, who themselves have sex with many men. The second pattern characterizes matrilineal societies, in which women are comparatively more independent and both men and women have multiple sexual partnerships. Prevalence of HIV and other STDs tends to be higher in the patrilineal than in the matrilineal societies (Larsen, 1989).
Availability and Utilization of Health Services
Availability of health services influences the prevalence of STDs by reducing the size of the pool of infected persons through treatment. The principal impact of treatment is on the bacterial STDs, and while this may have a substantial indirect impact on the spread of HIV, any direct impact on the virus itself is lacking. In addition, unless there are routine screening programs that can identify infected individuals, and perhaps curtail risky behavior, availability of health services primarily influences diseases that are symptomatic. And, although in some industrialized countries women tend to be more likely than men to seek health care and thus receive appropriate treatment (Ehrhardt and Wasserheit, 1991), this is rarely the case in Sub-Saharan Africa. Furthermore, care for STDs appears to be an exception, for a variety of reasons.
CONCLUSIONS
STDs and HIV infection in women are among the most challenging problems facing the international health community. As Table 11-5 indicates, STDs and HIV infection can occur at any age, but the importance of both behavioral and biologic risk factors for these diseases ebbs and flows across the life span in ways that have important implications for both research and intervention. Because both of these kinds of factors peak during
TABLE 11-5 Risks of Sexually Transmitted Diseases and HIV Infection among Females in Sub-Saharan Africa by Stage of the Life Span and Type of Risk Factor
|
In Utero/ Infancy |
Childhood (ages 1–14) |
Adolescence/Young Adulthood (ages 15–24) |
Mature Adulthood (ages 25–49) |
Postmenopause (age 50+) |
|
|
Risk of exposure (importance of behavioral risk factors) |
Low-medium |
Low |
High |
High |
Low |
|
Risk of acquisition if exposed (importance of biologic risk factors) |
High |
High |
High |
Medium |
High |
|
NOTE: Because STDs and HIV infection can be acquired at any point during the life span, this table, unlike those in Chapters 3 to 10, presents the comparative risks of acquisition of, and exposure to, STDs and HIV infection. Also, the life span age categories defined here differ slightly from those of the other chapter tables, because they represent age groupings with similar risks of acquisition and exposure. |
|||||
adolescence and young adulthood, this period constitutes the stage of highest risk in the life span of women. Both STDs and HIV infection occur most frequently in populations that are difficult to reach, and they are intertwined with sexuality and gender role, which in turn are determined by underlying socioeconomic factors. Those issues may be difficult to confront. Failure to identify and address these socioeconomic factors, however, narrows the ability to effectively prevent and control all STDs, including HIV infection. Limited data also suggest that the socioeconomic costs of HIV infection and other STDs are substantial. From a programmatic perspective, because of the impacts of STDs on reproductive health and HIV transmission, it is likely that interventions to prevent and control STDs in addition to HIV infection will synergistically promote family planning, child survival, and safe motherhood, and have a profoundly positive impact on the health of females across their entire life span.
RESEARCH NEEDS
This section highlights selected research issues of high priority in the reduction of STD/HIV morbidity among women in Sub-Saharan Africa and, in some cases, among women worldwide. Some of this research, particularly that involving basic science, may best be done in industrialized countries. Research done in developing countries must be relevant to the needs of those countries and carried out by local scientists in collaboration with local policy planners. Research involving both developing and industrialized countries should be collaborative and strongly encouraged, since it is in the common interest of both sets of nations.
Operational Research
Operational research is one of the most crucial areas of research for the improvement of public health. It concerns the translation of knowledge into action and it integrates information from a variety of disciplines: psychology, sociology, anthropology, epidemiology, clinical medicine, microbiology, and administration. Operational questions that are critical for reducing sexually transmitted diseases in women include the following:
-
How can we effectively integrate STD control with other relevant programs such as AIDS prevention, family planning, and antenatal care into a comprehensive health care package that is more accessible and acceptable to women?
-
What characteristics of health care systems would allow them to better serve the needs of women?
-
How should counseling, testing, and partner notification for both HIV and other STDs be done in settings where resources are scarce and where such activities may lead to violence against women? In the absence of routine screening for women, the only way to identify asymptomatic women with STDs may be through their male sex partners.
-
How should programs be designed to reach groups at high-risk for STDs, including HIV infection, such as prostitutes, their clients, and long-distance truck drivers?
-
How can traditional practitioners such as traditional birth attendants be involved in the prevention of HIV infection and other STDs?
Technology Development
Two technological issues are fundamentally important for women. The first is the development of noninvasive, simple, rapid, and affordable diagnostic tests for STDs (particularly for gonorrhea and chlamydia) that would permit the identification of women with asymptomatic infections. The second is the development of female-controlled prevention methods, such as safe intravaginal microbicidal agents, that would allow women to protect themselves against STDs, including HIV infection, and unwanted pregnancy.
Behavioral Research
The design of interventions to bring about behavioral change requires that people's behavioral patterns be understood, in this case the sexual and health-care-seeking behaviors that are the major determinants of STDs, including HIV infection. It is also important to understand reproductive decisions as another essential determinant of sexual behavior that may be influenced dramatically by the mortality associated with HIV infection. Research questions that may be relevant to understanding those behaviors and developing interventions to control HIV and other STDs include the following:
-
What are the factors that determine sexual, health-seeking, and reproductive behaviors?
-
What are the factors that determine women's social status?
-
How does women's social status influence sexual, health-seeking, and reproductive behaviors?
Intervention trials, with comparison groups, to assist women in building the skills and self-esteem to reject unsafe sexual behavior should be developed. Special effort should be directed toward adolescent girls to help them avoid becoming infected with HIV and other STDs, recognizing the unique physiological and psychological factors that influence the spread of these diseases during adolescence.
Individual behaviors clearly are determined by societal factors. The influence of each of those factors and the interactions among them must be defined if individual behavior is to be well understood. This chapter attempted to explore, at the country level, the associations among such societal factors as urbanization, women's status, access to health services, and STD/HIV prevalence. The scarcity and poor quality of data and the lack of good indicators impeded that exploration in many cases. Methodologies for measurement of societal indicators, data collection, and data analysis that are appropriate to the sensitive context and dimensions of STDs must be developed.
Clinical Research and Epidemiology
-
The clinical and epidemiological predictors of STDs in women must be refined to develop better management algorithms for asymptomatic as well as symptomatic infections.
-
The relative importance of and the gender differences in HIV-associated diseases should be determined to assist in the development of simple management algorithms for opportunistic infections and neoplasms.
-
The natural history of subclinical PID must be better described to improve detection.
-
Both the impact of STDs, including HIV infection, on adverse pregnancy outcomes and the influence of pregnancy on the course of STDs need to be better understood to provide adequate care to women of reproductive age.
-
Simple, cheap treatment regimens for HIV-infected pregnant women to reduce perinatal transmission should be developed.
NOTES
1. There are two types of HIV: HIV-1 and HIV-2. In these analyses, only HIV-1 was considered because it is more uniformly distributed across Sub-Saharan Africa.
2. High urban prevalence is defined as: HIV > 10 percent and gonorrhea > 5 percent among sexually active adults; syphilis > 10 percent among pregnant women.
REFERENCES
Adjorlolo, G. 1992. Natural history of HIV-2. Intl. Conf. AIDS 8:session 50.
Ahmed, H., K. Omar, Y. Adan, M. Guled, L. Grillner, and S. Bygdeman. 1991. Syphilis and human immunodeficiency virus seroconversion during a 6-month follow-up of female prostitutes in Mogadishu, Somalia. Intl. J. Stud. AIDS 2(2):119–123.
Aissu, T., M. C. Raviglione, J. P. Narain, et al. 1992. Monitoring HIV-associated tuberculosis in Uganda: Seroprevalence and clinical features. Intl. Conf. AIDS 8 (poster PoC 4023).
Akbar, J., J. Chakraborty, N. Jahan, et al. 1981. Dynamics of depo medroxyprogesterone acetate (DMPA) use effectiveness in the Matlab Family Planning Health Services Project. Paper presented at Bangladesh Fertility Research Programme, Seventh Annual Contributors' Conference, Dacca.
Akinsete, I., O. S. Ayelari, Y. Olurinde, and A. S. Akanmu. 1991. The pattern of HIV infection at the Lagos University Teaching Hospital (LUTH) Lagos, Nigeria, from January 1988–December 1990. Intl. Conf. AIDS 7(1):374 (abstract no. M.C. 3306).
Aladesanmi, A. F. K., G. Mumtaz, and D. C. W. Mabey. 1989. Prevalence of cervical chlamydial infection in antenatal clinic attenders in Lagos, Nigeria. Genitour. Med. 65:130.
Anderson, R. M., R. M. May, and A. R. McLean. 1988. Possible demographic consequences of AIDS in developing countries Nature 4:1087–1093.
Aral, S. O. 1990. Sexual behavior as a risk factor for sexually transmitted disease Pp. 185–198 in Reproductive Tract Infections: Global Impact and Priorities for Women's Reproductive Health A. Germaine, K. K. Holmes, P. Piot, and J. Wasserheit, eds. New York: Plenum.
Arya, O., H. Nsanzumuhire, and S. Taber. 1973. Clinical, cultural, and demographic aspects of gonorrhoea in a rural community in Uganda. Bull. WHO 49:587–595.
Arya, O. P., S. R. Taber, and H. Nsanze. 1980. Gonnorrhea and female infertility in rural Uganda. Am. J. Obstet. Gynecol. 138:929–932.
Asiimwe, G., G. Tembo, W. Naamara, et al. 1992. AIDS surveillance report: June 1992. Ministry of Health, AIDS Control Programme Surveillance Unit, Entebbe, Uganda. Photocopy.
Ballard, R. C., and H. Fehler. 1986. Chlamydial infections of the eye and genital tract in Southern Africa S. Afr. Med. J. Oct.(Suppl.):76–79.
Ballard, R. C., H. G. Fehler, M. O. Duncan, et al. 1981. Urethritis and associated infections in Johannesburg: the role of Chlamydia trachomatis. S. Afr. J. Sex. Trans. Dis. 1:24–26.
Ballard, R. C., et al. 1986. Chlamydial infections of the eye and genital tract in developing societies. Pp. 479–486 in Chlamydial Infections (Proceedings of the Sixth International Symposium on Human Chlamydial Infections), D. Oriel, et al., eds. Cambridge, U.K.: Cambridge University Press.
Barnes, T. E. C. 1979. Obstetrics in the Third World with particular reference to field research into delivery of maternal care to the community. Pp. 109–136 in Recent Advances in Obstetrics and Gynaecology, J. Stallworthy, and G. Bourne, eds. New York: Churchill Livingstone.
Barongo, L. R., J. B. Rugemalila, R. M. Gabone, et al. 1992. The epidemiology of HIV infection in adolescents of Kagera region Intl. Conf. AIDS 8 (poster PoC 4197).
Beaujan, G., and I. Willems. 1990. Prevalence of Chlamydia trachomatis infection in pregnant women in Zaire. Genitour. Med. 66:124–126.
Belec, P. L., G. Gresenguet, M. C. Georges-Courbot, A. Villon, M. H. Martin, and A. J. Georges. 1989. Seroepidemiologic study of several sexually transmitted diseases (including HIV infection) in a rural zone of the Central African Republic. Bull. Soc. Pathol. Exot. Filiales 81(4):692–698.
Bello, C. S. S., O. Y. Elegba, and J. D. Dada. 1983. Sexually transmitted diseases in northern Nigeria. Five years' experience in a university teaching hospital clinic. Br. J. Vener. Dis. 59:202–205.
Bentsi, C., C. Klufio, P. Perine, et al. 1985. Genital infections with Chlamydia trachomatis and Neisseria gonorrhoeae in Ghanaian women. Genitour. Med. 61(1):48–50.
Berger, R. E. 1990. Acute epididymitis. Pp. 641–651 in Sexually Transmitted Diseases, 2nd ed., K. K. Holmes, P. A. Mårdh, P. F. Sparling, et al., eds. New York: McGraw-Hill.
Bhatia, S. 1982. Contraceptive intentions and subsequent behavior in rural Bangladesh Stud. Fam. Plan. 13:24–31.
Bishaw, T., N. Tafari, M. Zewdie, et al. 1983. Prevention of congenital syphilis. Pp 148–153 in Proceedings of the Third African Regional Conference on Sexually Transmitted Diseases, H. Nsanze, R. H. Widy-Wirski, and R. H. Ellison, eds. Basel: Ciba Geigy.
Blanche S., C. Rouzioux, M-L. Guihart Moscato, et al. 1989. A prospective study of infants born to women seropositive for human immunodeficiency virus type 1. N. Engl. J. Med. 320:1643–1678.
Borgdorff, M. W., L. R. Barongo, C. N. Van Jaarsveld, et al. 1992. Sentinel surveillance: How representative are blood donors? Intl. Conf. AIDS 8 (poster PoC 4450).
Bottiger, B., I. B. Palme, J. L. da Costa, L. F. Dias, and G. Biberfeld. 1988. Prevalence of HIV-1 and HIV-2/HTLV-IV infections in Luanda and Cabinda, Angola. J. AIDS 1(1):8–12.
Bouvet E., I. De Vincenzi, R. Ancelle, and F. Vachon. 1989. Defloration as risk factor for heterosexual HIV transmission. Lancet i: 615.
Braddick, M. R., J. K. Kreiss, J. E. Embree, et al. 1990. Impacts of maternal HIV infection on obstetrical and early neonatal outcome. J. AIDS 4:1001–1004.
Brown, McL., and J. G. Cruickshank. 1976. Aetiological factors in pelvic inflammatory disease in urban blacks in Rhodesia S. Afr. Med. J. 50:1342–1344.
Brown, R. C., J. E. Brown, and B. A. Okako. 1992. Vaginal inflammation in Africa. N. Engl. J. Med. 20:572.
Browne, A., and D. Finkelhor. 1986. Impact of child sexual abuse: a review of the research. Psychol. Bull. 99:66–77.
Brunham, R. C., and J. E. Embree. 1990. Sexually transmitted diseases: current and future dimensions of the problem in the Third World. Pp. 35–58 in Reproductive Tract Infections: Global Impact and Priorities for Women's Reproductive Health, A. Germaine, K. K. Holmes, P. Piot, and J. Wasserheit, eds. New York: Plenum.
Brunham, R. C., K. K. Holmes, and J. E. Embree. 1990a. Sexually transmitted diseases in pregnancy. Pp. 771–801 in Sexually Transmitted Diseases, K. K. Holmes, P. A. Mårdh, P. F. Sparling, et. al., eds. New York: McGraw-Hill.
Brunham, R., M. Laga, J. Simonsen, et al. 1990b. The prevalence of Chlamydia trachomatis infection among mothers of children with trachoma. Am. J. Epidemiol. 132(5):946–952.
Brunham, R. C., G. P. Garnett, J. Swinton, and R. M. Anderson. 1991. Gonococcal infection and human fertility in Sub-Saharan Africa. Proc. R. Soc. London. Ser. B 246(1316):173–177.
Bulterys, M., A. Chao, A. Saah, et al. 1990. Risk factors for HIV-1 seropositivity among rural and urban pregnant women in Rwanda. Intl. Conf. AIDS 6(1):269 (abstract No. Th.C. 576).
Bulterys, M., A. Chao, J. B. Kurawige, et al. 1991, Maternal HIV infection and intrauterine growth; a prospective cohort study in Butare, Rwanda. Intl. Conf. AIDS 7 (abstract WC 3234).
Burans, J. P., E. Fox, M. A. Omar, et al. 1990. HIV infection surveillance in Mogadishu, Somalia. E. Afr. Med. J. 67(7):466–472.
Burney, P. 1976. Some aspects of sexually transmitted disease in Swaziland. Brit. J. Vener. Dis. 52:412–414.
Bwayo, J. J., M. Otido, D. Oduor, et al. 1992. Regular clients of female sex workers: condom use and risk of HIV-1 infection. Intl. Conf. AIDS 8 (poster PoC 4182).
Caldwell, J. C., and P. Caldwell. 1990. High fertility in Sub-Saharan Africa. Sci. Am. 262(5):118–125.
Cameron, D. W., L. J. D'Costa, G. M. Maitha, et al. 1989. Female to male transmission of human immunodeficiency virus type 1: risk factors for seroconversion in men. Lancet 2:403–407.
Carswell, J. W., et al. 1987. HIV infection in healthy persons in Uganda. AIDS 1(4):223–227.
Carty, M. J., M. Nzioki, and A. R. Verhagen. 1972. The role of gonococcus in acute pelvic inflammatory disease in Nairobi E. Afr. Med. J. 49(5):376–379.
Chang, Y., E. Cesarman, M. S. Pessin, et al. 1994. Identification of herpes-virus-like DNA sequences in AIDS-associated Karposi's sarcoma. Science 226:1865–1869.
Chao, A., P. Habimana, M. Bulterys, et al. 1992. Oral contraceptive use, cigarette smoking, age at first sexual intercourse, and HIV infection among Rwandan women. Intl. Conf. AIDS 8 (poster PoC 4338).
Chiphangwi, J., N. G. Liomba, P. Miotti, G. Dallabetta, E. Ndovi, and A. J. Saah. 1989. Serial seroprevalence studies and estimates of incidence of HIV-1 antibody in pregnant women in Malawi. Intl. Conf. AIDS 5:1023 (abstract no. G.507).
Chiphangwi, J., G. Dallabetta, A. Saah, G. Liomba, and P. Miotti. 1990. Risk factors for HIV-1 infection in pregnant women in Malawi. Intl. Conf. AIDS 6(1):158 (abstract no. Th.C. 98).
Cliff, J. 1991. The war on women in Mozambique. Health consequences of South African destabilization, economic crisis, and structural adjustment. Pp. 15–33 in Women and Health in Africa, M. Turshen, ed. Trenton, N.J.: Africa World Press.
Cohen, B. 1993. Fertility levels, differentials and trends. Pp. 8–67 in Demographic Change in Sub-Saharan Africa. Washington, D.C.: National Academy Press.
Cohen, M. S., J. R. Black, R. A. Proctor, and P. F. Sparling. 1985. Host defense and the vaginal mucosa. Scand. J. Nephrol. 86:13–22.
Connor E. M., R. S. Sperling, and R. Gelber. 1994. Reduction of maternal-infant transmission of human immunodeficiency virus type 1 with zidovudine treatment. N. Engl. J. Med. 331(18):1173–1180.
Cooper-Poole, B. 1986. Prevalence of syphilis in Mbeya, Tanzania. The validity of the VDRL as a screening test. E. Afr. Med. J. 63:646–650.
Corwin, A., J. Olson, M. Omar, A. Razaki, and D. Watts. 1991. Human immunodeficiency viral type 1 infection among prostitutes in Somalia. Intl. Conf. AIDS 7(1):306 (abstract no. M.C. 3032).
Cotch, M. F. 1990. Carriage of Trichomonas vaginalis (TV) is associated with adverse pregnancy outcome. Interscience Conference on Antimicrobial Agents and Chemotherapy 30:199 (abstract 681).
Dabis, F., F. Lepage, A. Serufilira, F. Nsengumuremyi, P. Msellati, and P. Van de Perre. 1989. Transmission du virus VIH-1 de la mère à l'enfant en Afrique Centrale: une étude de cohorte à Kigali, Rwanda—caractérisation épidémiologique à l'inclusion. Conférence internationale sur les implications du SIDA pour la mère et l'enfant. Paris (Résumé B11).
Dada, A. J., J. Ajayi, O. Ransome-Kuti, G. Williams, E. Abebe, A. Levin, T. Quinn, and W. Blattner. 1991. Prevalence survey of HIV-1 and HIV-2 infections in female prostitutes in Lagos State, Nigeria. Intl. Conf. AIDS 7(1):371 (abstract no. M.C. 3295).
Dallabetta, G., et al. 1990. Vaginal tightening agents as risk factors for acquisition of HIV. Intl. Conf. AIDS 6(1):268 (abstract no. Th.C. 574).
Dallabetta, G., P. Miotti, G. Liomba, et al. 1992. Evaluation of clinical and laboratory markers of cervicitis in STD patients in Malawi. Intl. Conf. AIDS 8(1):29 (abstract TuB 0510).
Damiba, A., S. Vermund, and K. Kelley. 1990. Prevalence of gonorrhoea, syphilis and trichomoniasis in prostitutes in Burkina Faso. E. Afr. Med. J. 67 (7 Suppl 2A):473–477.
D'Costa, L., F. Plummer, I. Bowmer, et al. 1985. Prostitutes are a major reservoir of sexually transmitted diseases in Nairobi, Kenya. Sex Trans. Dis. 12(2):64–67.
de Bruyn, M. 1992. Women and AIDS in developing countries. Soc. Sci. Med. 34(3):249–262.
De Clercq, A. 1982. Problèmes en obstétrique et gynécologie. Pp. 627–656 in Santé et maladies au Rwanda, A. Meheus, S. Butera, W. Eylenbosch, G. Gatera, M. Kivits, and I. Musafili, eds. Bruxelles: Administration Générale de la Coopération au Développement.
De Cock, K., B. Barrere, L. Diaby, et al. 1990. AIDS—the leading cause of adult death in the West African city of Abidjan, Ivory Coast. Science 249:793–796.
De Cock K. M., S. B. Lucas, S. Lucas, J. Agness, A. Kadio, and H. D. Gayle. 1993. Clinical research, prophylaxis, and care for HIV disease in Africa Am. J. Public Health. 83(10):1385–1389.
Delacolette, C., K. Kihemu, C. Delacolette-Lebrun, and R. Habyambere. 1986. Prévalence et sensibilité aux antibiotiques de l'infection à Neisseria gonorrhea en milieu rural au Kivu, Zaïre. Ann. Soc. Belge Méd. Trop. 66:87–90.
Del Mistro, A., J. Chotard, A. J. Hall, et al. 1992. HIV-1 and HIV-2 seroprevalence rates in mother-child pairs living in The Gambia (West Africa). J. AIDS 5(1):19–24.
De Muylder, X. 1989. Standardised management of PID in a developing country. Genitour. Med. 65:281–283.
De Muylder, X., M. Laga, C. Tennstedt, E. Van Dyck, G. Aelbers, and P. Piot. 1990. The role of Neisseria gonorrhea and Chlamydia trachomatis in pelvic inflammatory disease and its sequelae in Zimbabwe. J. Inf. Dis. 162:501–505.
De Schampheleire, I., et al. 1990. Maladies sexuellement transmissibles dans la population féminine à Pikine, Sénégal. Ann. Soc. Belge Med. Trop. 70: 227–235.
De Schryver, A., and A. Meheus. 1989. International travel and sexually transmitted diseases. Rapp. Trim. Stat. Sanit. Mond. 42:90–99.
De Vincenzi, I. 1994. A longitudinal study of human immunodeficiency virus transmission by heterosexual partners. N. Engl. J. Med. 331:341–346.
Diaw, I., T. Siby, I. Thior, M. Traore, L. Dabo, M. Ndaw, A. Diaw, C. Langley, N. Kiviat, I. Ndoye, et. al. 1992. HIV and STD infections among newly registered prostitutes in Dakar Intl. Conf. AIDS 8(2):C300 (abstract no. PoC 4333).
Didier, B., N. Lorenz, L. H. Ouedraogo, Y. Zina, P. Barth, and T. Rehle. 1990. KAP-study and HIV-seroprevalence among prostitutes in rural Burkina Faso. Intl. Conf. AIDS 6(1):146 (abstract no. Th.D. 52).
Dietrich, M., A. A. Hoosen, J. Moodley, and S. Moodley. 1992. Urogenital tract infections in pregnancy at King Edward VIII Hospital, Durban, South Africa, Genitour. Med. 68(1):39–41.
Dosso, M., H. Faye, Y. Diakite-Harding, H. Aissi, J. Aka, and M. Duchassin 1986. Aspects épidémiologiques et prévalence de Neisseria gonorrhea dans les infections génitales à Abidjan: analyze de 1742 prélèvements. Bull. Soc. Path. Ex. 79:130–139.
Drescher, C., T. Elkins, O. Adkeo, et al. 1988. The incidence of urogenital Chlamydia trachomatis infections among patients in Kumasi, Ghana. Intl. J. Gynecol. Obstet. 27:381–383.
Drummond, M. F., G. L. Stoddart, and G. W. Torrance. 1989. Methods for the Economic Evaluation of Health Care Programmes. Oxford, U.K.: Oxford University Press.
Duncan, M., G. Tibaux, A. Pelzer, et al. 1990. First coitus before menarche and risk of sexually transmitted disease Lancet 335:338–340.
Dupont, A., D. Schrijvers, E. Delaporte, et al. 1988. Séroprévalence de la syphilis dans les populations urbaines et semi-rurales du Gabon. Bull. Soc. Path. Ex. 81:699–704.
Ebrahim, S. H., M. R. Sunkutu, and N. Mwansa. 1993. Epidemiological and clinical features of Kaposi's sarcoma in adults in Zambia. Intl. Conf. AIDS (abstract WS-B15-1).
Ehrhardt, A. A., and J. N. Wasserheit. 1991. Effects of age and gender on sexual behavior. Pp. 97–121 in Research Issues in Human Behavior and Sexually Transmitted Diseases in the AIDS Era, J. N. Wasserheit, S. O. Aral, K. K. Holmes, and P. J. Hitchcock. eds. Washington, D.C.: American Society for Microbiology.
Elliot, B., R. Brunham, M. Laga, et al. 1990. Maternal gonococcal infection as a preventable risk factor for low birthweight. J. Infect. Dis. 161:531–536.
Engels, H., A. Nyongo, M. Temmerman, W. G. V. Quint, E. Van Marck, and W. J. Eylenbosch. 1992. Cervical cancer screening and detection of genital HPV infection and chlamydial infection by PCR in different groups of Kenyan women Ann. Soc. Belge Med. Trop. 72:53–62.
Fakeya, R. 1986. Antitreponemal antibodies among antenatal patients at the University of Ilorin Teaching Hospital. Afr. J. Sex Trans. Dis. 1:9–10.
Farley, T. M., M. J. Rosenberg, P. J. Rowe, J. H. Chen, and O. Meirkik. 1992. Intrauterine devices and pelvic inflammatory disease: an international perspective. Lancet 339:785–788.
Fassassi-Jarretou, A., A. Mabignath-Sall, B. Narraido, et al. 1991. Impacts de Chlamydia trachomatis sur les femmes enceintes au Gabon. Bull. Soc. Pathol. Exot. Filiales 84(5):620–626.
Feleke, W., M. Yusuf, H. Tesfay, A. Abraham, and G. Carosi. 1992. Prevalence of syphilis among pregnant women attending urban and semi-urban health centers in Ethiopia. Intl. Conf. AIDS 8(2):B148 (abstract PoB 3371).
François-Gerard, C., J. Nkurunziza, C. De Clerq, L. Stouffs, and D. Sondag. 1992. Seroprevalence of HIV, HBV and HCV in Rwanda. Intl. Conf. AIDS 8(2):C249 (abstract no. PoC 4027).
Frank, O. 1983. Infertility in Sub-Saharan Africa. Center for Policy Studies Working Paper 97. New York: The Population Council.
Friedmann, P., and D. Wright. 1977. Observations on syphilis in Addis Ababa. 2. Prevalence and natural history. Br. J. Vener. Dis. 53:276–280.
Fruchter, R., M. Maiman, E. Serur, and S. Cuthill. 1992. Cervical intraepithelial neoplasia in HIV-infected women. Intl. Conf. AIDS 8(1) (abstract MoB 0057).
Galega, F., D. Heymann, and B. Nasah. 1984. Gonococcal ophthalmia neonatorum: the case for prophylaxis in tropical Africa. Bull. WHO 62:95–98.
Gaye-Diallo, A., F. Denis, S. Mboup, et al. 1992. Co-infections HIV/HTLV-1 in Senegal. Intl. Conf. AIDS 8 (poster PoC 4028).
Gersony, R. 1988. Summary of Mozambican refugee accounts of principally conflict-related experience in Mozambique. Washington, D.C.: Bureau for Refugee Persons, U.S. Department of State.
Geyid, A., H. S. Tesfaye, A. Abraha, et al. 1991. A study on STD pathogens in sex workers in Addis Ababa, Ethiopia. Intl. Conf. AIDS 7(2):301 (abstract No W.C. 3023).
Gichangi, P., M. Temmerman, A. Mohamed, J. Ndinya-Achola, F. Plummer, and P. Piot. 1992. Rapid increase in HIV-1 infection and syphilis between 1989 and 1991 in pregnant women in Nairobi, Kenya. Intl. Conf. AIDS 8(2):pC 249 (abstract PoC 4029).
Gini, P., W. Chukudebelu, and A. Njoku-Obi. 1989. Antenatal screening for syphilis at the University of Nigeria Teaching Hospital, Enugu, Nigeria, A six-year survey. Intl. J. Gynecol. Obstet. 29(4):321–324.
Goeman, J., A. Meheus, P. Piot, et al. 1991. L'épidémiologie des maladies sexuellement transmissibles dans les pays en développement à l'ère du SIDA. Ann. Soc. Belge Med. Trop. 71: 81–113.
Green, S. D., J. K. Mokolo, M. Nganzi, A. G. Davies, I. R. Hardy, D. J. Jackson, E. B. Klee, R. A. Elton, P. D. Hargreaves, and W. A. Cutting. 1992. Seroprevalence and determinants of HIV-1 infection in pregnancy in rural Zaire. Intl. Conf. AIDS 8(2):C247 (abstract no. PoC 4016).
Gresenguet, G., L. Belec, P. M. V. Martin, et al. 1991. Séroprévalence de l'infection à VIH-1 au sein des consultants de la clinique des maladies sexuellement transmissibles de Bangui, Centrafrique. Bull. Soc. Path. Ex. 84(3):240–246.
Guiness, L., S. Sibandze, E. McGrath, and A. Cornelis. 1988. Influence on antenatal screening on perinatal mortality caused by syphilis in Swaziland. Genitour. Med. 64:294–297.
Heise, L., A. Rulkes, C. H. Watts, and A. B. Zwi. 1994. Violence against women: a neglected public health issue in less-developed countries. Soc. Sci. Med. 39(9):1165–1179.
Hellmann, N. S., P. Nsubuga, E. K. Mbidde, and S. Desmond-Hellmann. 1990. Specific heterosexual risk behaviors and HIV seropositivity in a Uganda STD clinic. Intl. Conf. AIDS 6(1):269 (abstract no. Th.C. 578).
Hillis, S. D., R. Joesoef, P. A. Marchbanks, J. N. Wasserheit, W. Cates, and L. Westrom. 1993. Delayed care for pelvic inflammatory disease as a risk factor for impaired fertility. Am. J. Obstet. Gynecol. 168:1503–1509.
Hira, P. R. 1977. Observations on trichomonas vaginalis infections in Zambia. J. Hyg. Epidemiol. Microbiol. Immunol. 2:215–224.
Hira, S. 1984. Epidemiology of maternal and congenital syphilis in Lusaka and Copperbelt Provinces of Zambia Lusaka: Republic of Zambia.
Hira, S., C. Bhat, A. Ratnam, C. Chintu, and R. Mulenga. 1982. Congenital syphilis in Lusaka. II. Incidence at birth and potential risk among hospital deliveries. E. Afr. Med. J. 59(5):306–310.
Hira, S. K., J. Kamanga, G. J. Bhat, et al. 1989. Perinatal transmission of HIV-1 in Zambia. Br. Med. J. 299:1250–1252.
Hira, S. K., et al. 1990. Syphilis intervention in pregnancy: Zambian demonstration project Genitour. Med. 66:159–164.
Hira, S., S. Godwin, J. Kamanga, and G. Mukelabai. 1992a. Efficacy of spermicide use and condom use by HIV-discordant couples in Zambia. Intl. Conf. AIDS 8(1) (abstract no. WeC 1085).
Hira, S., R. Sunkutu, and P. Perine. 1992b. Zambian National STD Control Program: The model program in Africa Intl. Conf. AIDS 8(2):B229 (abstract No. PoB 3829).
Hitti, J., C. K. Walker, P. S. J. Nsubuga, et al. 1992. Oral contraceptive use and HIV infection. Intl. Conf. AIDS 8 (poster PoC 4309).
Holmes, K. K. 1990. Lower genital tract infections in women: cystitis, urethritis, vulvovaginitis, and cervicitis. Pp 527–545 in Sexually Transmitted Diseases, K. K. Holmes, P. A. Mårdh, P. F. Sparling, et. al., eds. New York: McGraw-Hill.
Holmes, K. K., and S. O. Aral. 1991. Behavioral interventions in developing countries. Pp. 318–344 in Research Issues in Human Behavior and Sexually Transmitted Diseases in the AIDS Era, J. N. Wasserheit, S. O. Aral, K. K. Holmes, and P. J. Hitchcock, eds. Washington D.C.: American Society for Microbiology.
Hook, E. W., and H. H. Handsfield. 1990. Gonococcal infections in the adult. Pp. 149–165 in Sexually Transmitted Diseases, 2nd ed., K. K. Holmes, P. A. Mårdh, P. F. Sparling, et al., eds. New York: McGraw-Hill.
Hoosen, A. A., S. M. Ross, M. J. Mulla, and M. Patel. 1981. The incidence of selected vaginal infections among pregnant urban Blacks. S. Afr. Med. J. 59:827–829.
Hopcraft, M., A. Verhagen, S. Ngigi, and A. Haga. 1973. Genital infections in developing countries: experience in a family planning clinic. Bull. WHO. 48:581–586.
Hudson, C., A. Hennis, P. Kataaha, et al. 1988. Risk factors for the spread of AIDS in rural Africa: evidence from a comparative seroepidemiological survey of AIDS, hepatitis B and syphilis in southwestern Uganda. AIDS 2(4):255–260.
Iquatt, B., and A. Sawhney. 1988. Symptomless gonorrhoea in women in Maiduguri (northeastern Nigeria) Genitour. Med. 64:349.
Ismail, S., H. Ahmed, M. Jama, et al. 1990. Syphilis, gonorrhoea and genital chlamydial infection in a Somali village. Genitour. Med. 66(2):70–75.
Jain, A. K., and I. Sivin. 1977. Life-table analysis of IUDs: problems and recommendations. Stud. Fam. Plan. 8:24–47.
Jama, H., et al. 1987. Syphilis in women of reproductive age in Mogadishu, Somalia: Serological survey. Genitour. Med. 63:326–328.
Jones, R. B., and J. N. Wasserheit. 1991. Introduction to the biology and natural history of sexually transmitted diseases. Pp. 11–37 in Research Issues in Human Behavior and Sexually Transmitted Diseases in the AIDS Era, J. N. Wasserheit, S. O. Aral, K. K. Holmes, and P. J. Hitchcock, eds. Washington D.C.: American Society for Microbiology.
Kanyama, I. 1991. Sentinel surveillance of HIV infection in Northern Zambia. Intl. Conf. AIDS 7(1):373 (abstract no. M.C. 3301).
Kapiga, S., D. J. Hunter, J. F. Shao, et al. 1992. Contraceptive practice and HIV-1 infection among family planning clients in Dar es Salaam, Tanzania. Intl. Conf. AIDS 8 (poster PoC 4343).
Kaptue, L., et al. 1990. Setting up a sentinel surveillance system for HIV infection in Cameroon Intl. Conf. AIDS 6(abstract F.C. 597).
Kaptue, L., L. Zekeng, S. Djoumessi, M. Monny-Lobe, D. Nichols, and R. Debuysscher. 1991. HIV and chlamydia infections among prostitutes in Younde, Cameroon Genitour. Med. 67:143–145.
Karita, E., P. Van de Perre, A. Nziyumvira, W. Martinez, J. Nyiraminani, E. Fox, and J.B. Butera. 1992. HIV seroprevalence among STD patients in Kigali, Rwanda, during the four-year period 1988–1991. Intl. Conf. AIDS 8(2):C323 (abstract no. PoC 4468).
Kitabu, M. Z., G. M. Maitha, J. N. Mungai, et al. 1992. Trends and seroprevalence of HIV amongst four population groups in Nairobi in the period 1989–1991. Intl. Conf. AIDS 8 (poster PoC 4018).
Kivuvu, Mayimona, B. Malele, N. Nzila, A. Manoka, et al. 1990. Syphilis among HIV+ and HIV - prostitutes in Kinshasa: prevalence and serologic response to treatment. Fifth Intl. Conf. AIDS in Africa. Kinshasa (abstract TPC 7).
Klieg, R. S., A. Adachi, I. Fleming, I, G. Y. F. Ho, and R. Burk. 1992. A prospective study of genital neoplasia and human papillomovirus (HPV) in HIV-infected women (abstract). Intl. Conf. AIDS 8(1) (abstract TuB 0527).
Kreiss, J. K., and K. G. Castro. 1990. Special considerations for managing suspected human immunodeficiency virus infection and AIDS in patients from developing countries. J. Infect. Dis. 162:955–960.
Kreiss, J., D. Koech, F. Plummer, et al. 1986. AIDS virus infection in Nairobi prostitutes. N. Engl. J. Med. 314(7):414–418.
Laga, M., et al. 1988. Prophylaxis of gonococcal and chlamydial ophthalmia neonatorum: A comparison of silver nitrate and tetracycline. N. Engl. J. Med. 318: 653–657.
Laga, M. 1990. Human immunodeficiency virus infection prevention: The need for complementary STD control. Pp. 131–144 in Reproductive Tract Infections: Global Impact and Priorities for Women's Reproductive Health, A. Germaine, K. K. Holmes, P. Piot, and J. Wasserheit, eds. New York: Plenum.
Laga, M., F. Plummer, H. Nzanze, et al. 1986. Epidemiology of ophthalmia neonatorum in Kenya. Lancet 2:1145–1149.
Laga, M., N. Nzila, A. T. Manoka, et al. 1989. High prevalence and incidence of HIV and other sexually transmitted diseases (STD) among 801 Kinshasa prostitutes. Intl. Conf. AIDS 5:74 (abstract Th.A.O. 21).
Laga, M., N. Nzila, A. T. Manoka, et al. 1990. Nonulcerative sexually transmitted diseases (STD) as risk factors for HIV transmission. Intl. Conf. AIDS 6(abstract Th.C. 97).
Laga, M., J. P. Icenogle, R. Marsella, et al. 1992. Genital papilloma virus infection and cervical dysplasia—opportunistic complications of HIV infection. Intl. J. Cancer. 50:45–48.
Lallemant M., A. Baillou, S. Lallemant-Le Coeur, et al. 1994. Maternal antibody response at delivery and perinatal transmission of human immunodeficiency virus type 1 in African women. Lancet 343:1001–1005.
Larsen, A. 1989. Social context of human immunodeficiency virus transmission in Africa: Historical and cultural bases of East and Central African sexual relations. Rev. Inf. Dis. 11:716–731.
Latif, A. 1981. Sexually transmitted disease in clinic patients in Salisbury, Zimbabwe Br. J. Vener. Dis. 57:181–183.
Latif, A. S. 1989. Epidemiology and control of chancroid. Eighth ISSTDR (abstract 66).
Latif, A., S. Bvumbe, J. Muongerwa, E. Paraiwa, and W. Chikosi. 1984. Sexually transmitted diseases in pregnant women in Harare, Zimbabwe Afr. J. Sex. Trans. Dis. January–March: 21–23.
Laukamm-Josten, U., D. Ocheng, B. Mwizarubi, C. Mwaijonga, R. Swai, and M. Trupin. 1992. HIV and syphilis seroprevalence and risk factors in truckstops and nearby communities in Tanzania. Intl. Conf. AIDS 8(2):C272 (abstract PoC 4162).
Leclerc, A., E. Frost, M. Collet, J. Goeman, and L. Bedjabaga. 1988. Urogenital Chlamydia trachomatis in Gabon: an unrecognised epidemic. Genitour. Med. 64:308–311.
Lepage, P., and P. Van de Perre. 1988a. Clinical manifestation of AIDS in infants and children. Bailliere's Clin. Trop. Med. Commun. Dis. 3(1):89–101.
Lepage, P., and P. Van de Perre. 1988b. Strategies in the identification and control of HIV-infected women in Africa. Pp. 215–255 in AIDS in Children, Adolescents, and Heterosexual Adults, A. J. Schinazi-Nahmias, ed. New York: Elsevier.
Lepage, P., P. Van de Perre, P. Msellatl, et al. 1993. Mother-to-child transmission of human immunodeficiency virus type 1 (HIV-1) and its determinants: a cohort study in Kigali, Rwanda Am. J. Epidemiol. 137(6):589–599.
Liljestrand, J., S. Bergström, F. Nieuwenhuis, and B. Hederstedt. 1985. Syphilis in pregnant women in Mozambique. Genitour. Med. 61(6):355–358.
Linden, C. P., S. Allen, A. Serufilira, et al. 1992. Predictors of mortality among HIV-infected women in Kigali, Rwanda Ann. Intern. Med. 116:320–328.
Luyeye, M., et al. 1990. Prevalence et facteurs de risque pour les MST chez les femmes enceintes dans les soins de santé primaires à Kinshasa. Fifth Int. Conf. AIDS in Africa. Kinshasa (abstract T.P.C. 8).
Mabey, D. C., et al. 1984. Sexually transmitted diseases among randomly selected attenders at an antenatal clinic in The Gambia. Br. J. Vener. Dis. 60:331–336.
Mabey, D. 1986. Syphilis in Sub-Saharan Africa. Afr. J. Sex. Trans. Dis. 6:2(2):61–64.
Mabey, D., and H. Whittle. 1982. Genital and neonatal chlamydial infection in a trachoma endemic area Lancet 2(93):300–301.
MacDonald, K. S., W. Cameron, L. J. D'Costa, et al. 1989. Evaluation of fleroxacin (RO-23-6240) as single-oral-dose therapy of culture-proven chancroid in Nairobi, Kenya. Antimicrob. Ag. Chemother. 33:612–614.
Maiman, M., R. G. Fruchter, E. Serur, J. C. Remy, G. Feuer, and J. Boyce. 1990. Human immunodeficiency virus infection and cervical neoplasia. Gynecol. Oncol. 38:377–382.
Mandara, N., S. Takulias, J. Kanyawana, and F. Mhalu. 1980. Asymptomatic gonorrhoea in women attending family planning clinics in Dares-Salaam, Tanzania: results of a pilot study. Trop. Geogr. Med. 32:329–332.
Mane, P., G. R. Gupta, and E. Weiss. 1994. Effective communication between partners: AIDS and risk reduction for women. AIDS 8 (Suppl.):S325–S331.
Mann, J. M., N. Nzilambi, P. Piot, N. Bosenge, M. Kalala, H. Francis, R. C. Colebunders, P. K. Azila, J. W. Curran, and T. C. Quinn. 1988. HIV infection and associated risk factors in female prostitutes in Kinshasa, Zaire. AIDS 2(4):249–254.
Mann, J. M., D. J. M. Tarantola, and T. W. Netter, eds. 1992. AIDS in the World. Cambridge, Mass.: Harvard University Press.
Manneh, K. K., R. S. Njie, R. Sarr, et al. 1992. HIV status of antenatal women reporting for routine blood test at the Royal Victoria Hospital. Intl. Conf. AIDS 8 (poster PoC 4015).
Manoka, A., M. Laga, M. Kivuvu, et al. 1992. Syphilis among HIV+ and HIV- prostitutes in Kinshasa: Prevalence and serologic response to treatment. Intl. Conf. AIDS 6(3):102 (abstract SB 27).
Martin, D. H., L. Koutsky, D. A. Eschenbach, et al. 1982. Prematurity and perinatal mortality in pregnancies complicated by maternal Chlamydia trachomatis infections. J. Am. Med. Assoc. 247:1585.
Mason, P. R., et al. 1983. Epidemiology and clinical diagnosis of Trichomonas vaginalis infection in Zimbabwe. Centr. Afr. J. Med. 29(3):53–56.
Mason, P., D. Katzenstein, T. Chimbira, and L. Mtimavalye. 1989. Vaginal flora of women admitted to hospital with signs of sepsis following normal delivery, cesarean section or abortion. The Puerperal Sepsis Study Group. Centr. Afr. J. Med. 35(3):344–351.
Mason, P., et al. 1990. Genital infections in women attending a genito-urinary clinic in Harare, Zimbabwe, Genitour. Med. 66(3):178–181.
Mason, P. R., L. Gwanzura, and F. Le Bacq. 1992. Correlation between positive syphilis serology and HIV infection in Zimbabwe. Intl. Conf. AIDS 8 (poster 4303).
Matasha, E., J. Changalucha, H. Grosskurth, et al. 1992. Commercial sexual workers intervention programme: A pilot project in northern Tanzania: operational data and intermediate results. Intl. Conf. AIDS 8(2): pD 495 (abstract PoD 5639).
Mboup, L., et al. 1991. Surveillance sentinelle des infections à VIH. Bull. Epidémiol. VIH:3.
M'Boup, S. 1992. Natural history of HIV-2. Intl. Conf. AIDS 8(session 50).
Mboya, T. O. 1992. Traditional behavior and AIDS: Analysis and change through community participation. Intl. Conf. AIDS 8 (poster PoD 5697).
Mbugua, G., G. Gachihi, S. Adaw, F. Mueke, K. Mandalya, and E. Mupate. 1989. HIV seroprevalence survey among high-risk females at Mombasa, Kenya Intl. Conf. AIDS 5:1014 (abstract W.G.P. 27).
Mbugua, G., L. Muthami, J. Kitama, S. Oogo, and P. Waiyaki. 1991. Rising trend of HIV infection among antenatal mothers in a Kenyan rural area. Intl. Conf. AIDS 7(2):366 (abstract W.C. 3283).
McCallum, M. et al. 1973. A survey of selected vaginal flora in Malawian women. Centr. Afr. J. Med. 19(8):176–178.
McCarthy, M. C., and A. E. El Hag. 1990. HIV-I infection in Juba, southern Sudan. Intl. Conf. AIDS 6(2):232 (abstract no. F.C. 605).
McCarthy, M. C., J. P. Burans, N. T. Constantine, et al. 1989. Hepatitis B and HIV in Sudan: a serosurvey for hepatitis B and human immunodeficiency virus antibodies among sexually active heterosexuals Am. J. Trop. Med. Hyg. 41(6):726–731.
Mefane, C., and M. Toung-Mve. 1987. Syphilis chez la femme enceinte à Libreville (Gabon). Bull. Soc. Path. Ex. 80(2):162–167.
Meheus, A. 1990. Women's health: importance of reproductive tract infections, pelvic inflammatory disease and cervical cancer. Pp. 61–91 in Reproductive Tract Infections: Global Impact and Priorities for Women's Reproductive Health, A. Germaine, K. K. Holmes, P. Piot, and J. Wasserheit, eds. New York: Plenum.
Meheus, A., W. Eylenbosch, and A. Ndibwami. 1975. Serological evidence for syphilis in different population groups in Rwanda. Trop. Geogr. Med. 27(2):165–168.
Meheus, A., F. Friedman, E. Van Dyck, and T. Guyver. 1980. Genital infections in prenatal and family planning attendants in Swaziland. E. Afr. Med. J. 57(3):212–217.
Meheus, A., K. F. Schulz, and W. Cates. 1990. Development of prevention and control programs for sexually transmitted diseases in developing countries. Pp. 1041–1046 in Sexually Transmitted Diseases, K. K. Holmes, P. A. Mårdh, P. F. Sparling, et al., eds. New York: McGraw-Hill.
Melnick, S. L., R. Sherer, T. A. Louis, et al. 1994. Survival and disease progression according to gender of patients with HIV infection. JAMA 272(24):1915–1921.
Minkoff, H. L., C. Henderson, H. Mendez, et al. 1990. Pregnancy outcomes among mothers infected with human immunodeficiency virus and uninfected control subjects. Am. J. Obstet. Gynecol. 163:1598–1604.
Mirza, N. B., et al. 1983. Microbiology of vaginal discharge in Nairobi, Kenya. Br. J. Vener. Dis. 59:186–188.
Mlisana, K., S. Monokoane, A. Hoosen, J. Moodley, M. Adhikari, and L. Taylor 1992. Syphilis in the ''unbooked" pregnant women. S. Afr. Med. J. 82(1):18–20.
Mofenson, L. M., P. Stratton, and A. Willoughby. 1992. HIV in pregnancy and effects on the fetus and infant. Year Book Obstet. Gynecol.:xiii-xlii.
Mokwa, K., V. Batter, F. Behets, et al. 1991. Prevalence of sexually transmitted diseases (STD) in childbearing women in Kinshasa, Zaire, associated with HIV infection. Intl. Conf. AIDS 7(2):358 (abstract No. W.C. 3251).
Mooradian, A. D., and V. Greiff. 1990. Sexuality in older women. Arch. Intern. Med. 150:1033–1038.
Mouden, J. C., C. Genin, J. C. Gruel, and P. Coulanges. 1988. Cyto-microbiological and serological study for the screening of sexually transmissible diseases in Malagasy multiparous women. Arch. Inst. Pasteur Madagascar 54(1):217–228.
Mtimavalye, L., and M. Belsey. 1987. Infertility and sexually transmitted diseases: major problems in maternal and child health and family planning. International Conference on Better Health for Women and Children through Family Planning, Nairobi, 5–9 October.
Muhe, L., and N. Tafari. 1986. Is there a critical time for prophylaxis against neonatal ophthalmia? Genitour. Med. 62:356–357.
Mungai, J., G. Maitha, M. Kitabu, et al. 1992. Prevalence of HIV and other STD's in three populations in Nairobi for the year 1991. Intl. Conf. AIDS 8(2):C362 (abstract PoC 4714).
NACP/MOH (Ethiopia). 1992. Surveillance and research activities on HIV/AIDS: Activities accomplished so far in Ethiopia, 1984–91. Ethiopia NACP/MOH data. Photocopy.
Nasah, B., R. Nguematcha, M. Eyong, et al. 1980. Gonorrhea, trichomonas, and candida among gravid and non-gravid women in Cameroon. Intl. J. Gynecol. Obstet. 18:48.
Naucler, A., S. Anderson, P. Albino, et al. 1992. Association between HTLV-1 and HIV-2 infections in Bissau, Guinea-Bissau AIDS 6(5):510–511.
Neequaye, A. R., J. A. A. Mingle, G. Ankra-Badu, et al. 1987. Human immune deficiency virus infections in Ghana. AIDS and Associated Cancers in Africa, Second International Symposium, Naples: 85–93.
Ndour-Sarr, A. N., D. S. Ba, T. Ndoye, A. Gueye, T. Siby, C. S. Boye, R. Child, A. Diouf, N. Mbaye, F. Kebe, et al. 1992. HIV infection in pregnant women in Dakar. Intl. Conf. AIDS 8(2):C285 (abstract no. PoC 4241).
Ndumbe, P., A. Andela, J. Nkemnkeng-Asong, E. Watonsi, and Nyambi. 1992. Prevalence of infections affecting the child among pregnant women in Yaounde, Cameroon. Med. Microbiol. Immunol. 181(3):127–130.
Newell, M. L. 1992. Risk factors for mother-to-child transmission of HIV-1. Lancet 339:1007–1012.
Nkowame, B. 1991. Prevalence and incidence of HIV infection in Africa: a review of data published in 1990. AIDS 5 (Suppl. 1):S7–16.
Nkya, W. M. M. M., W. P. Howlett, C. Assenga, et al. 1987. Seroepidemiology of HIV-1 infection in the Kilimanjaro region. AIDS and Associated Cancers in Africa, Second International Symposium, Naples: 61–65.
Nkya, W. M., S. H. Gillespie, W. Howlett, J. Elford, C. Nyamuryekunge, C. Assenga, and B. Nyombi. 1991. Sexually transmitted diseases in prostitutes in Moshi and Arusha, Northern Tanzania. Intl. J. STD AIDS 2(6):432–435.
Nkya, W. M., W. P. Howlett, K. I. Klepp, B. Nyombi, and C. Assenga. 1992. HIV-cohort study of self-proclaimed prostitutes in Moshi and Arusha towns, northern Tanzania. Intl. Conf. AIDS 8(2):D496 (abstract no. PoD 5645).
Nsanze, H., S. Waigwa, N. Mirza, et al. 1982. Chlamydial infections in selected populations in Kenya. Pp. 421–424 in Chlamydial Infections, P. A. Mårdh, et. al., eds. New York: Elsevier.
Nsofor, B. I., C. S. Bello, and C. G. Ekwempu. 1989. Sexually transmitted diseases among women attending a family planning clinic in Zaria, Nigeria. Intl. J. Gynaecol. Obstet. 28(4):365–367.
Nsubuga, P., F. Miremba, S. Kalibbala, et al. 1992. Reported sexual behavior and sexually transmitted infection prevalence among women attending a prenatal clinic in Kampala, Uganda. Intl. Conf. AIDS 8(2):C346 (abstract PoC 4619).
Nyirenda, M. J. 1992. A study of the behavioral aspects of dry sex practice in Lusaka urban Intl. Conf. AIDS 8(2):D461 (abstract No. PoD 5448).
O'Farrell, N., A. A. Hoosen, A. B. Kharsany, and J. van den Ende. 1989. Sexually transmitted pathogen in pregnant women in a rural South African community. Genitour. Med. 65(4):276–280.
O'Farrell, N., I. Windsor, and P. Becker. 1990. Risk factors for HIV-I amongst STD clinic attenders in Durban, South Africa. Intl. Conf. AIDS 6(2):232 (abstract no. F.C. 604).
Ogbonna, C., I. Ogbonna, A. Ogbonna, and J. Anosike. 1991. Studies on the incidence of Trichomonas vaginalis amongst pregnant women in Jos area of Plateau State, Nigeria. Angew. Parasitol. 32(4):198–204.
Okepere, E. E., E. E. Obaseiki-Ebor, and S. M. Oyaide. 1987. Type of intra-uterine contraceptive device (IUCD) used and the incidence of symptomatic Neisseria gonorrhoeae. Afr. J. Sex Trans. Dis. 3:7–8.
Ole-Kingori, N., K. I. Klepp, K. S. Mnyika, et al. 1992. Population-based HIV-screening in Arusha, Tanzania: A pilot study Intl. Conf. AIDS 8 (poster PoC 4031).
Omanga, U., F. Fendler, M. Bamba, M. Sulu, and B. Mikanga. 1989. Sero-épidémiologie de la syphilis congénitale à Kinshasa, Zaïre. Ann. Soc. Belg. Med. Trop. 69(4): 313–318.
Omer, E. E., et al. 1980. Study of sexually transmitted disease in Sudanese women Trop. Doctor 10:99–102.
Omer, E. E., R. D. Catterall, M. H. Ali, H. A. El-Naeem, and H. H. Erwa. 1985a. Vaginal trichomoniasis at a sexually transmitted disease clinic at Khartoum. Trop. Doctor 15:170–172.
Omer, E., T. Forsey, S. Darougar, M. Ali, and H. El-Naeem. 1985b. Seroepidemiological survey of chlamydial genital infections in Khartoum, Sudan. Genitour. Med. 61:261–263.
O'Reilly, K. R. 1986. Sexual behavior, perceptions of infertility and family planning in sub-Saharan Africa. Afr. J. Sex Trans. Dis. 2:47–49.
Orubuloye I. O., J. C. Caldwell, and P. Caldwell. 1993. African women's control over their sexuality in an era of AIDS. Soc. Sci. Med.:37(7):859–872.
Osoba, A., and A. Onifade. 1987. Venereal diseases among pregnant women in Nigeria. W. Afr. Med. J. 11(1):23–25.
Ouattara, S. A., D. Diallo, M. Meite, et al. 1988. Epidémiologie des infections par les virus de l'immunodéficience humaine VIH-1 et VIH-2 en Côte d'Ivoire. Med. Trop. 48(4):375–379.
Ousseini, H., J. L. Pecarrere, D. Meynard, et al. 1991. Evolution de la séroprévalence des infections à VIH-1 et VIH-2 à l'Hàpital National de Niamey, Niger. Bull. Soc. Path. Ex. 84(3):235–239.
Over, M., and P. Piot. 1991. HIV infections and sexually transmitted diseases. Pp. 455–527 in Disease Control Priorities in Developing Countries, D. T. Jamison, W. H. Mosley, A. R. Measham, and J. L. Bobadilla, eds. Washington, D.C.: World Bank.
Parkin, D. M., E. Läärä, and C. S. Muir. 1988. Estimates of the worldwide frequency of sixteen major cancers in 1980. Intl. J. Cancer 41:184–197.
Perine, P. 1983. Congenital syphilis in Ethiopia. Med. J. Zambia 17(1):12–14.
Perine, P., M. Duncan, D. Krauss, et al. 1980. Pelvic inflammatory disease and puerperal sepsis in Ethiopia. Am. J. Obstet. Gynecol. 138:969–977.
Piot, P., and M. Laga. 1990. STD control in developing countries. Pp. 281–295 in Reproductive Tract Infections: Global Impact and Priorities for Women's Reproductive Health, A. Germaine, K. K. Holmes, P. Piot, and J. Wasserheit, eds. New York: Plenum.
Plummer, F., M. Laga, R. Brunham, et al. 1987. Postpartum upper genital tract infections in Nairobi, Kenya: Epidemiology, etiology, and risk factors. J. Infect. Dis. 156(1):92–98.
Plummer, F. A., D. W. Cameron, N. Simonsen, et al. 1991. Co-factors in male-female transmission of human immunodeficiency virus type 1. J. Infect. Dis. 163:233–239.
Quarcoopome, C. O. 1983. Opthalmia neonatorum: problems of prophylaxis and treatment in Africa WHO PBL/ON/83-1:1–4. Geneva.
Ratnam, A. V., S. Din, T. Chatterjee. 1980a. Sexually transmitted diseases in pregnant women. Med. J. Zambia 14:75–78.
Ratnam, A. V., S. N. Din, and T. K. Chatterjee. 1980b. Gonococcal infection in women with pelvic inflammatory disease in Lusaka, Zambia. Am. J. Obstet. Gynecol. 138:965–968.
Ratnam, A. V., S. Din, S. Hira, et al. 1982. Syphilis in pregnant women in Zambia. Br. J. Vener. Dis. 58:355–358.
Rellihan, M. A., D. P. Dooley, T. W. Burke, M. E. Berkland, and R. N. Longfield. 1990. Rapidly progressing cervical cancer in a patient with human immunodeficiency virus infection. Gynecol. Oncol. 36:435–438.
Retel-Laurentin, A. 1973. Fecondité et syphilis dans la région de la Volta Noire. Population 28:793–815.
Richardson D. 1989. Women and the AIDS Crisis, 2nd ed. London: Pandora.
Rob, U., J. F. Phillips, J. Chakraborty, et al. 1987. The use effectiveness of the Copper-T-200 in Matlab, Bangladesh. Intl. J. Obstet. Gynecol. 25:315.
Ronald, A., and S. O. Aral. 1990. Assessment and prioritization of actions to prevent and control reproductive tract infections in the Third World. Pp. 199–225 in Reproductive Tract Infections: Global Impact and Priorities for Women's Reproductive Health, A. Germaine, K. K. Holmes, P. Piot, and J. Wasserheit, eds. New York: Plenum.
Rosenberg, M. J., K. F. Schulz, and N. Burton. 1986. Sexually transmitted diseases in Sub-Saharan Africa. Lancet 849:152–153.
Rothenberg, R. B., and J. J. Potterat. 1990. Strategies for management of sex partners. Pp. 1081–1086 in Sexually Transmitted Diseases, K. K. Holmes, P. A. Mårdh, P. F. Sparling, et. al., eds. New York: McGraw-Hill.
Rushwan, H. 1980. Etiologic factors in pelvic inflammatory disease in Sudanese women Am. J. Obstet. Gynecol. 138:877–879.
Ryder, R. W., W. Nsa, S. E. Hassig, et al. 1989. Perinatal transmission of the human immunodeficiency virus type 1 to infants of seropositive women in Zaire. N. Engl. J. Med. 320:1637–1642.
Salehe, O., G. Riedner, Y. Hemed, H. Kazimbazi, T. Rehle, and F. von Sonnenburg. 1992. HIV infection among STD patients and correlated conditions in Mbeya, Tanzania. Int. Conf. AIDS 8(2):C362 (abstract no. PoC 4715).
Samb Noone, D., F. Van Der Veen, M. Sene, A. Thiam, L. Van De Velde, and D. Diouf. 1992. Sentinel surveillance of STDs, and its implications for AIDS control in Senegal. Int. Conf. AIDS 8(2):C299 (abstract PoC 4328).
Sangare, A., G. Leonard, M. Verdier, et al. 1988. Comparison of C. trachomatis, HIV, HTLV-I. Prevalence in Ivory Coast populations. Abstract No FP33, IIIe International Conference on AIDS and Associated Cancers in Africa, Arusha, Tanzania.
Schafer, A., W. Friedmann, M. Mielke, B. Schwartlander, and M. A. Koch. 1991. The increased frequency of cervical dysplasia-neoplasia in women infected with the human immunodeficiency virus is related to the degree of immunosuppression. Am. J. Obstet. Gynecol. 164:593–599.
Schulz, K. F., W. Cates, and P. R. O'Mara. 1987. Pregnancy loss, infant death, and suffering: legacy of syphilis and gonorrhea in Africa. Genitour. Med. 63:320–325.
Schulz, K. F., J. M. Schulte, and S. M. Berman. 1990. Maternal health and child survival: Opportunities to protect both women and children from the adverse consequences of reproductive tract infections. Pp. 145–182 in Reproductive Tract Infections: Global Impact and Priorities for Women's Reproductive Health, A. Germaine, K. K. Holmes, P. Piot, and J. Wasserheit, eds. New York: Plenum.
Schrijvers, D., E. Delaporte, M. Peeters, et al. 1988. Role of sexually transmissible pathogens in transmitting HIV-1. Genitour. Med. 64:(6)395–396.
Schwartz, L. B., M. L. Carcangiu, L. Bradham, and P. E. Schwartz. 1991. Rapidly progressing squamous carcinoma of the cervix coexisting with human immunodeficiency virus infection: clinical opinion. Gynecol. Oncol. 41:255–258.
Scott, D. A., A. L. Corwin, N. T. Constantine, M. A. Omar, G. Guled, M. Yusef, C. R. Roberts, and D. M. Watts. 1991. Low prevalence of human immunodeficiency virus-1 (HIV-1), HIV-2, and human T-cell lymphotropic virus-1 infection in Somalia. Am. J. Trop. Med. Hyg. 45(6):653–659.
Selwyn, P. A., E. E. Schoenbaum, K. Davenny, et al. 1989. Prospective study of human immunodeficiency virus infection and pregnancy outcomes in intravenous drug users. J. Am. Med. Assoc. 261:1289–1294.
Selwyn, P. A., and P. Antoniello. 1993. Reproductive decision making among women with HIV infection. Pp. 174–185 in HIV Infection in Women, M. A. Johnson and F. D. Johnstone, eds. London: Churchill Livingstone.
Sende, P., T. Abong, and G. Garrigue. 1986. Chlamydia trachomatis: étude de la prévalence chez la femme enceinte, la femme inféconde, et l'homme souffrant d'une uréthrite chronique. Afr. J. Sex. Trans. Dis. 2:72–74.
Senkoro, K., J. Newell, H. Grosskurth, et al. 1992. Syphilis and STD syndromes in Northern Tanzania: Prevalences, risk factors and association with the seroprevalence of HIV. Intl. Conf. AIDS 8 (poster PoC 4329).
Senyonyi, I. B. 1987. Prevalence des infections gynécologiques à Chlamydia trachomatis et à Neisseria gonorrhoeae chez 199 femmes examinées en consultation prénatales ou en consultation pour planning familial à Kigali et à Kabgayi. Rev. Med. Rwandaise 19:85–89.
Shao, J., S. Kapiga, D. Hunter, G. Lwihula, J. Mtui, and E. Mbena. 1992. Sexually transmitted diseases (STD) and HIV-1 infection among family planning clients in Dar-es-Salaam, Tanzania. Intl. Conf. AIDS 8(2):pC 300 (abstract PoC 4330).
Sheller, J., N. Pedersen, and B. Kvinesdal, et. al. 1990. HIV infection, syphilis og genitale lesion i Maun, Botswana. Ugesk Laeger. 152(20):1441–1443.
Sibailly, T. S., G. Adjorlolo, H. Gayle, et al. 1992. Prospective study to compare HIV-1 and HIV-2 perinatal transmission in Abidjan, Côte d'Ivoire. Intl. Conf. AIDS 8 (abstract WeC 1065).
South Africa, RSA Department of National Health and Population Development 1991. AIDS in South Africa: Status on World AIDS Day 1991. Epidemiolog. Conf. 18(11:229–249).
South Africa, RSA Department of National Health and Population Development 1992. Second National Survey of Women Attending Antenatal Clinics, South Africa, October/November 1991. Epidemiol. Conf. 19(5) 80–92.
St. Louis, M. E., M. Kamenga, C. Brown, et al. 1993. Risk for perinatal HIV-1 transmission according to maternal immunologic, virologic, and placental factors. JAMA 269(22): 2853–2859.
Tafari, N., D. Zewdi, B. Gebrehiwot, and T. Kebede. 1990. The simultaneous occurrence of perinatal HIV-1 and syphilis in Central Ethiopia: implications for control measures. Intl. Conf. AIDS 6(2):446 (abstract no 3176).
Tembo, G., J. Twa-Twa, G. Assimwe, et al. 1991. AIDS surveillance report: December 1991. Ministry of Health, AIDS Control Programme Surveillance Unit, Entebbe, Uganda. Photocopy.
Temmerman, M., F. Mohamed Ali, J. Ndinya-Achola, S. Moses, F. Plummer, and P. Piot. 1992a. Rapid increase of both HIV-1 infection and syphilis among pregnant women in Nairobi, Kenya. AIDS 6:1181–1185.
Temmerman, M., T. K'Oduol, F. A. Plummer, J. O. Ndinya-Achola, and P. Piot. 1992b. Maternal HIV infection as a risk factor for adverse obstetrical outcome Intl. Conf. AIDS 8(2) pC 283 (abstract PoC 4232).
Temmerman, M., E. N. Chomba, and P. Piot. 1994. HIV-1 and reproductive health in Africa. Intl. J. Obstet. 44:107–112.
Tovo, P. A., M. de Martino, G. Caramia, and the Italian Register for HIV Infection in Children. 1988. Epidemiology, clinical features, and prognostic factors of paediatric HIV infection. Lancet 2:1042–1045.
Twa-Twa, J., G. Tembo, G. Assimwe, et al. 1991. AIDS surveillance report (first and second quarter) for the year 1991. Ministry of Health, AIDS Control Programme Surveillance Unit, Entebbe, Uganda. Photocopy.
Ulin, P. R. 1992. African women and AIDS: negotiating behavioral change. Soc. Sci. Med. 34(1):63–73.
UNDP (United Nations Development Programme). 1992. Human Development Report 1992. New York: Oxford University Press.
Ursi, J., E. van Dyck, C. van Houtte, et al. 1981. Syphilis in Swaziland. A serological survey. Br. J. Vener. Dis. 57:95–99.
Van de Perre, P., N. Clumeck, M. Steens, G. Zissis, M. Carael, R. Lagasse, S. De Wit, T. Lafontaine, P. De Mol, and J. P. Butzler. 1987. Seroepidemiological study of sexually transmitted diseases and hepatitis B in African promiscuous heterosexuals in relation to HTLV-III infection Eur. J. Epidemiol. 3(1):14-8.
van Rensburg, H., and J. Odendaal. 1992. The prevalence of potential pathogenic micro-organisms in the endocervix of pregnant women at Tygerberg Hospital. S. Afr. Med. J. 81(3):156–157.
Venter, A., J. Pettifor, F. Exposto, and M. Sefuba. 1989. Congenital syphilis—who is at risk? A prevalence study at Baragwanath Hospital, Johannesburg, 1985–1986. S. Afr. Med. J. 76(3):93–95.
Ville, Y., M. Leruez, E. Glowaczower, J. N. Roberston, and M. E. Ward. 1991. The role of Chlamydia trachomatis and Neisseria gonorrhea in the aetiology of ectopic pregnancy in Gabon. Br. J. Obstet. Gynaecol. 98(12):1260–1266.
Vink, G., and J. Moodley. 1989. Gonorrhea in black women attending a gynaecological outpatient department S. Afr. Med. J. 58(2):901–902.
Walker, U., and W. Hofler. 1989. Prevalence of Chlamydia trachomatis in pregnant women and infertility cases in Abeokuta, Nigeria. Trop. Med. Parasitol. 40(1):77–78.
Wasserheit, J. N. 1989. The significance and scope of reproductive tract infections among Third World women. Intl. J. Gynecol. Obstet. (Suppl.) 3:145–168.
Wasserheit, J. N. 1992. Epidemiological synergy: interrelationships between human immunodeficiency virus infection and other sexually transmitted diseases. Sex. Trans. Dis. 19:61–77.
Wasserheit, J. N., and K. K. Holmes. 1990. Reproductive tract infections: Challenge for international health policy, programs, and research. Pp. 7–33 in Reproductive Tract Infections: Global Impact and Priorities for Women's Reproductive Health, A. Germaine, K. K. Holmes, P. Piot, and J. Wasserheit, eds. New York: Plenum.
Watson, P. 1985. The use of screening tests for sexually transmitted diseases in a Third World community—a feasibility study in Malawi. Eur. J. Sex. Trans. Dis. 2:63–65.
Watts, T., S. Larsen, and S. Brown. 1984. A case-control study of stillbirths at a teaching hospital in Zambia, 1979–80: Serological investigations for selected infectious agents Bull. WHO 62:803–808.
Weissenberger, R., A. Robertson, S. Holland, et al. 1977. The incidence of gonorrhea in urban Rhodesian black women. S. Fr. Med. J. 52(28):1119–20.
Welcomed, N., A. Mahaffey, and J. Van de Ended. 1986. Prevalence of Neisseria gonorrhea infection in patients attending an antenuptial clinic. S. Fr. Med. J. 69(1):32–34.
Weasels, P. H., G. J. Viljoen, N. F. Marais, J. A. de Beer, M. Smith, and A. Gericke. 1991. The prevalence, risks, and management of Chlamydia trachomatis infections in fertile and infertile patients from the high socioeconomic bracket of the South African population. Fertil. Steril. 56(3):485–488.
Weström, L. 1980. Incidence, prevalence and trends of acute pelvic inflammatory disease and its consequences in industrialized countries. Am. J. Obstet. Gynecol. 138:880–892.
Weström, L., and P. A. Mårdh. 1984. Salpingitis. Pp. 615–632 in Sexually Transmitted Diseases, K. K. Holmes, P. A. Mårdh, P. F. Sparling, et al., eds. New York: McGraw-Hill.
Weström, L., and P. A. Mårdh. 1990. Acute pelvic inflammatory disease (PID). Pp. 593–613 in Sexually Transmitted Diseases, K. K. Holmes, P. A. Mårdh, P. F. Sparling, et al., eds. New York: McGraw-Hill.
Weström, L., R. Joesoef, G. Reynolds, A. Hagdu, and S. Thompson. 1992. Pelvic inflammatory disease and fertility: A cohort of 1844 women with laparoscopically verified disease and 657 control women with normal laparoscopic results. Sex. Trans. Dis. July–August:185–192.
Whiteside, A. 1991. HIV infection and AIDS in Zimbabwe: An assessment. Southern Africa Foundation for Economic Research. Economic Research Unit, University of Natal.
Whiteside, A. 1992. An evaluation of the likely impact of AIDS on the Mananga medical service subscribing companies. Draft report: 1–12. Photocopy.
Widy-Wirski, R., and J. D'Costa. 1980. Maladies transmises par voie sexuelle dans une population rurale en Centrafrique. Pp. 651–656 in Rapport Final, 13e Confírence Technique. Yaounde, Cameroon: OCEAC.
World Bank. 1992. World development indicators. Pp. 209–308 in World Development Report 1992: Development and Environment. New York: Oxford University Press.
World Bank. 1993. World Development Report 1993: Investing in Health. New York: Oxford University Press.
WHO (World Health Organization). 1987. Genital human papillomavirus infections and cancer: Memorandum from a WHO meeting Bull. WHO 65(6):817–827.
WHO (World Health Organization). 1993. Report of a Consultation on Women and HIV/AIDS. WHO/GPA/DIR/94.1. Geneva.
WHO (World Health Organization). 1994. Women and AIDS: Agenda for Action. WHO/GPA/DIR/94.4. Geneva.
Wright, T. C., T. V. Ellerbrock, M. A. Chiasson, N. Van Devanter, and S. Xiao-Wei. 1994a. Cervical intraepithelial neoplasia in women infected with human immunodeficiency virus: prevalence, risk factors, and validity of Papanicolaou smears Obstet Gynecol. 84:591–597.
Wright, T. C., J. Koulos, F. Schnoll, J. Swanbeck, T. V. Ellerbrock, M. A. Chiasson, and R. M. Richart. 1994b. Cervical intraepithelial neoplasia in women infected with the human immunodeficiency virus: outcome after loop electrosurgical excision Gyn. Oncol. 55:253–258.
Yorke, J. A., H. W., Hethcote, and A. Nold. 1978. Dynamics and control of the transmission of gonorrhea. Sex. Trans. Dis. 5:51–56.
Yvert, F., J. Riou, E. Frost, and B. Ivanoff. 1984. Les infections gonococciques au Gabon (Haut-Ogooué). Pathol. Biol. 32(2):80–84.
Zacarias, F. R., and S. O. Aral. 1985. STD control in less developed countries: The time is now. Intl. J. Epidmiol. 14:505–509.
Zekeng, L., P. Barth, R. Salla, L. Kaptue, B. Schmidt-Ehry, and T. Rehle. 1990. Seroepidemiological study of sexually transmitted agents (HIV, hepatitis B virus, T. pallidum) among sentinel populations in the northwest and southwest provinces, Cameroon. Int. Conf. AIDS 6(2):230 (abstract FC 598).
Zekeng, L., R. Salla, L. Kaptue, et al., 1992. HIV-2 infection in Cameroon: No evidence of indigenous cases. J. AIDS 5(3):319–320.
Zimbabwe, Central Statistical Office, Ministry of Health. 1989. Annual Report. Harare.
APPENDIX
TABLE 11-6 Genital Discharge Syndrome
|
Country |
Year |
Population |
N |
Ref. |
GC |
CT |
TV |
|
Low-risk population |
|||||||
|
Cameroon |
1977 |
PP |
296 |
87 |
14 |
||
|
1980 |
AN (asymptomatic) |
110 |
180 |
14 |
15 |
||
|
1980 |
FP (asymptomatic) |
53 |
180 |
2 |
8 |
||
|
1980 |
PP (asymptomatic) |
296 |
180 |
10 |
|||
|
1986a |
AN |
234 |
9 |
||||
|
Central African Republic |
1980 |
AN |
268 |
10 |
|||
|
Ethiopia |
1973–75 |
PP (asymptomatic) |
200 |
209 |
9 |
||
|
Gabon |
1981 |
AN |
530 |
270 |
6 |
||
|
1988 |
PP (asymptomatic) |
598 |
137 |
10 |
|||
|
1987–89 |
AN |
2,305 |
82 |
19 |
|||
|
Gambia, The |
1982 |
AN |
87 |
145 |
7 |
7 |
|
|
1982 |
AN (asymptomatic) |
100 |
144 |
7 |
7 |
32 |
|
|
Ghana |
1983 |
AN |
213 |
3 |
|||
|
1988a |
AN (asymptomatic) |
110 |
74 |
4 |
|||
|
Kenya |
1973 |
FP |
200 |
111 |
18 |
26 |
|
|
1981 |
AN |
54 |
188 |
0 |
6 |
||
|
1981 |
FP |
57 |
188 |
17 |
4 |
||
|
1984 |
PP |
728 |
131 |
7 |
23 |
||
|
1984 |
PP |
845 |
212 |
7 |
21 |
||
|
1985 |
PP |
175 |
99 |
4 |
17 |
||
|
1985–86 |
PP |
2,732 |
127 |
6 |
9 |
||
|
1985–87 |
Trachoma area |
168 |
35 |
4 |
|||
|
1988–89 |
AN |
549 |
161 |
3 |
2 |
||
|
1990 |
AN |
269 |
242 |
7 |
|||
|
1990 |
FP |
80 |
6 |
11 |
|||
|
1992 |
AN |
5,674 |
178 |
10 |
|||
|
Malawi |
1973 |
AN |
100 |
162 |
19 |
||
|
1990 |
AN |
6,483 |
57 |
5 |
3 |
32 |
|
|
Nigeria |
1971 |
AN |
208 |
203 |
3 |
21 |
|
|
1971–75 |
Gyn (IUD users) |
282 |
256 |
5 |
27 |
||
|
1971–75 |
Asymptomatic housewife |
130 |
256 |
5 |
15 |
||
|
1983–85 |
FP |
500 |
196 |
4 |
|||
|
1988 |
AN |
80 |
253 |
11 |
|||
|
1988 |
Infertile |
20 |
253 |
15 |
|||
|
1989a |
FP |
150 |
189 |
0–8 |
0–8 |
||
|
1989a |
No FP |
50 |
189 |
6 |
4 |
||
|
1989a |
AN |
46 |
6 |
6 |
|||
|
1991a |
AN (rural) |
250 |
195 |
25 |
|||
|
1991a |
AN (urban) |
250 |
195 |
38 |
|||
|
Rwanda |
1987 |
AN + FP |
199 |
236 |
5 |
||
|
1987 |
AN + FP |
198 |
236 |
16 |
|||
|
Senegal |
1983 |
AN |
213 |
19 |
|||
|
1987 |
AN |
236 |
176 |
2 |
|||
|
1990 |
511 |
93 |
30 |
||||
|
Senegal |
1987 |
AN |
200 |
68 |
2 |
7 |
|
|
1989–92 |
AN |
781 |
227 |
1 |
8 |
16 |
|
|
Somalia |
1987 |
All village |
200 |
115 |
0 |
18 |
|
|
South Africa |
1980a |
AN |
105 |
110 |
10 |
23 |
|
|
1980a |
AN |
80 |
110 |
90 |
|||
|
1981 |
AN (asymptomatic) |
1,200 |
260 |
11 |
|||
|
1983 |
AN |
213 |
10 |
||||
|
1986 |
AN |
88 |
12 |
13 |
|
Country |
Year |
Population |
N |
Ref. |
GC |
CT |
TV |
|
Low-risk population |
|||||||
|
South Africa |
1986a |
AN |
231 |
14 |
1 |
||
|
1986a |
FP |
62 |
14 |
16 |
|||
|
1987 |
AN |
193 |
193 |
6 |
11 |
49 |
|
|
1991a |
AN (white, high SES) |
41 |
261 |
7 |
|||
|
1992a |
AN (asymptomatic) |
170 |
72 |
4 |
5 |
||
|
1992a |
AN |
206 |
249 |
7 |
11 |
||
|
Swaziland |
1973 |
OPD (all women) |
179 |
39 |
29 |
||
|
1973 |
AN |
215 |
39 |
50 |
|||
|
1978 |
AN |
51 |
167 |
4 |
23 |
||
|
1978 |
FP |
52 |
167 |
2 |
15 |
||
|
Tanzania |
1978 |
FP (asymptomatic) |
341 |
147 |
8 |
||
|
1983 |
AN |
213 |
8 |
||||
|
1986a |
AN |
69 |
6 |
||||
|
1991–92 |
AN |
2,009 |
237 |
5 |
14 |
||
|
Uganda |
1971–72 |
Community (Ankole) |
168 |
9 |
2 |
||
|
1971–72 |
Community (Teso) |
295 |
9 |
22 |
|||
|
1980 |
Low fertil. district |
336 |
10 |
18 |
|||
|
1980 |
High fertil. district |
246 |
10 |
2 |
|||
|
1992a |
AN |
450 |
101 |
1 |
7 |
43 |
|
|
Zaire |
1985 |
AN |
208 |
64 |
2 |
4 |
|
|
1988 |
AN |
101 |
17 |
9 |
|||
|
1990 |
AN |
701 |
142 |
2 |
6 |
17 |
|
|
1991 |
AN |
1,875 |
174 |
1 |
5 |
16 |
|
|
Zambia |
1977a |
AN |
218 |
102 |
39 |
||
|
1979 |
AN (asymptomatic) |
161 |
216 |
11 |
|||
|
Zimbabwe |
1976 |
AN |
50 |
259 |
2 |
||
|
1976 |
FP |
100 |
259 |
12 |
|||
|
1983 |
AN |
199 |
154 |
31 |
|||
|
1986–87 |
PP (asymptomatic) |
111 |
155 |
5 |
6 |
14 |
|
|
High-risk population |
|||||||
|
Cameroon |
1980 |
AN (symptomatic) |
610 |
180 |
15 |
21 |
|
|
1980 |
FP (symptomatic) |
81 |
180 |
21 |
7 |
||
|
1980 |
PP (symptomatic) |
42 |
180 |
21 |
2 |
||
|
Côte d'Ivoire |
1986a |
Symptoms |
73 |
10 |
|||
|
Ethiopia |
1973–75 |
PP (puerperal sepsis) |
67 |
209 |
28 |
||
|
1973–75 |
PID (outpatient) |
100 |
209 |
19 |
|||
|
1973–75 |
PID (hospital) |
46 |
209 |
15 |
|||
|
Gabon |
1980–82 |
Symptomatic (vaginal D/C) |
261 |
270 |
15 |
23 |
|
|
1988a |
Symptomatic (vaginal D/C) |
252 |
137 |
18 |
|||
|
1988a |
Symptomatic (LAP) |
265 |
137 |
14 |
|||
|
Gambia, The |
1982 |
Gyn (symptomatic) |
23 |
145 |
14 |
||
|
Ghana |
1985 |
Gyn |
162 |
20 |
3 |
5 |
|
|
1985 |
Gyn (LAP) |
40 |
20 |
0 |
10 |
||
|
1985 |
PP (hospitalized) |
148 |
20 |
3 |
|||
|
1985 |
PP (hospitalized) |
39 |
20 |
8 |
|||
|
Kenya |
1981 |
STD (symptomatic) |
58 |
188 |
23 |
7 |
|
|
1982 |
STD (vaginal D/C) |
122 |
171 |
26 |
34 |
||
|
1982 |
STD (vaginal D/C) |
58 |
171 |
7 |
|||
|
1985 |
PP (preterm) |
166 |
99 |
11 |
13 |
||
|
1992 |
STD |
80 |
4 |
11 |
|||
|
1992 |
STD+FP |
212 |
80 |
11 |
|
Country |
Year |
Population |
N |
Ref. |
GC |
CT |
TV |
|
High-risk population |
|||||||
|
Malawi |
1992 |
STD |
505 |
58 |
15 |
26 |
|
|
1992 |
STD |
175 |
58 |
5 |
|||
|
1990 |
STD |
255 |
57 |
25 |
36 |
||
|
Nigeria |
1971–75 |
Gyn (symptomatic) |
228 |
256 |
17 |
19 |
|
|
1977–81 |
STD |
435 |
19 |
13 |
11 |
||
|
1988a |
Gyn |
120 |
114 |
15 |
|||
|
Senegal |
1990 |
Gyn |
250 |
68 |
4 |
8 |
|
|
1989–92 |
Gyn |
795 |
227 |
2 |
8 |
16 |
|
|
South Africa |
1980 |
STD |
135 |
19 |
13 |
||
|
1980a |
AN (symptomatic) |
127 |
110 |
9 |
31 |
||
|
1980a |
Gyn |
100 |
252 |
11 |
|||
|
1986a |
STD |
135 |
14 |
13 |
|||
|
Sudan |
1976 |
Gyn (symptomatic) |
147 |
224 |
1 |
12 |
|
|
1977 |
STD (symptomatic) |
613 |
199 |
7 |
20 |
||
|
1976–78 |
STD (vaginal D/C) |
132 |
200 |
4 |
|||
|
1976–78 |
STD (vaginal D/C) |
144 |
200 |
||||
|
1976–78 |
STD (vaginal D/C) |
114 |
200 |
20 |
|||
|
1985a |
STD |
404 |
201 |
8 |
|||
|
1985a |
STD |
613 |
199 |
20 |
|||
|
Zambia |
1977a |
Gyn |
518 |
102 |
31 |
||
|
1979 |
STD (nonpregnant) |
1,000 |
216 |
19 |
|||
|
1979 |
STD (pregnant) |
170 |
216 |
23 |
|||
|
1979 |
PID (hospitalized) |
100 |
216 |
46 |
|||
|
Zimbabwe |
1976 |
Gyn |
118 |
259 |
11 |
||
|
1979–80 |
STD |
234 |
200 |
23 |
|||
|
1982 |
STD (pregnant) |
160 |
133 |
26 |
38 |
||
|
1983 |
STD |
156 |
154 |
37 |
|||
|
1986–87 |
PP (puerperal sepsis) |
95 |
155 |
19 |
16 |
12 |
|
|
1990a |
STD |
100 |
153 |
19 |
8 |
32 |
|
|
Very-high-risk population |
|||||||
|
Burkina Faso |
1990 |
CSW |
127 |
59 |
20 |
17 |
|
|
Cameroon |
1991a |
CSW |
168 |
122 |
39 |
||
|
Côte d'Ivoire |
1988a |
CSW |
228 |
65 |
|||
|
Ethiopia |
1991a |
CSW |
282 |
90 |
30 |
24 |
|
|
1992 |
CSW |
46 |
28 |
30 |
6 |
||
|
Kenya |
1981 |
CSW (upper SES) |
60 |
16 |
|||
|
1981 |
CSW (middle SES) |
60 |
28 |
||||
|
1981 |
CSW (lower SES) |
60 |
46 |
||||
|
Madagascar |
1987 |
CSW |
298 |
175 |
39 |
||
|
1987 |
CSW |
40 |
175 |
18 |
|||
|
Malawi |
1990 |
CSW (STD) |
274 |
57 |
29 |
27 |
|
|
Senegal |
1989–92 |
CSW |
397 |
227 |
13 |
17 |
18 |
|
1989–92 |
CSW |
653 |
70 |
16 |
14 |
24 |
|
|
Somalia |
1990a |
CSW |
89 |
38 |
11 |
||
|
Tanzania |
1991a |
CSW |
106 |
185 |
51 |
||
|
1991a |
CSW |
47 |
185 |
25 |
|||
|
1992a |
CSW |
70 |
156 |
||||
|
Zaire |
1989a |
CSW |
801 |
130 |
23 |
14 |
22 |
|
NOTE: GC, gonorrhea; CT, chlamydia; TV, trichomoniasis; AN, antenatal; PP, postpartum; FP, family planning; LAP, lower abdominal pain; D/C, discharge; CSW, commercial sex worker; SES, socioeconomic status. a No study date. |
|||||||
TABLE 11-7 Syphilis
|
Country |
Year |
Population |
N |
Ref. |
Non-T |
Trep |
Non-T + Trep |
GUD |
|
Low-risk population |
||||||||
|
Botswana |
1985–87 |
AN |
113 |
238 |
17 |
3 |
||
|
Burkina Faso |
1973a |
Village (Karba) |
208 |
217 |
18 |
|||
|
1973a |
Village (Dedougo) |
239 |
217 |
32 |
||||
|
1973a |
Village (Moko) |
133 |
217 |
8 |
||||
|
Cameroon |
1989–90 |
AN |
544 |
184 |
16 |
|||
|
1990a |
AN |
608 |
272 |
24 |
||||
|
1990a |
AN |
900 |
121 |
10 |
||||
|
Central African Republic |
1980 |
AN |
268 |
10 |
||||
|
Ethiopia |
1974–75 |
AN |
2,717 |
208 |
18 |
17 |
||
|
1977a |
AN |
337 |
86 |
13 |
11 |
11 |
||
|
1990a |
AN |
1,719 |
240 |
7 |
||||
|
1992a |
AN |
100 |
83 |
9 |
||||
|
Gabon |
1983 |
AN |
527 |
165 |
12 |
|||
|
1984 |
AN |
715 |
165 |
13 |
||||
|
1985 |
AN |
623 |
165 |
14 |
||||
|
1986 |
Urban |
211 |
77 |
15 |
||||
|
1987 |
Semi-rural |
277 |
77 |
9 |
||||
|
Gambia, The |
1986a |
AN (rural) |
100 |
143 |
15 |
11 |
||
|
1986a |
AN |
238 |
143 |
26 |
13 |
|||
|
1982 |
AN (urban) |
100 |
144 |
9 |
1 |
1 |
||
|
Kenya |
1984 |
PP |
1,013 |
131 |
11 |
4 |
||
|
1985 |
PP |
133 |
99 |
5 |
||||
|
1988–89 |
AN |
549 |
161 |
4 |
||||
|
1989–91 |
AN |
4,883 |
91 |
4 |
||||
|
1992a |
AN |
5,674 |
178 |
3 |
||||
|
Malawi |
1985a |
AN |
182 |
257 |
18 |
14 |
||
|
1990 |
AN |
6,483 |
57 |
13 |
7 |
|||
|
Mozambique |
1985 |
AN 10 districts |
1,468 |
140 |
8 |
6 |
||
|
Nigeria |
1971 |
AN |
208 |
203 |
0 |
|||
|
1979–84 |
AN |
29,083 |
92 |
3 |
0.4 |
|||
|
1986a |
AN |
81 |
2 |
|||||
|
1989a |
FP |
150 |
189 |
15 |
||||
|
1989a |
Controls (non-FP) |
50 |
189 |
12 |
||||
|
Rwanda |
1972 |
AN |
862 |
166 |
3 |
|||
|
1972 |
Students |
153 |
166 |
1 |
||||
|
1980 |
AN |
2,321 |
62 |
4 |
||||
|
1987a |
Asympt. blood donor |
33 |
248 |
15 |
6 |
|||
|
1989a |
AN |
54 |
4 |
4 |
||||
|
Senegal |
1989–92 |
AN |
781 |
227 |
6 |
|||
|
1990 |
AN |
511 |
93 |
8 |
||||
|
Somalia |
1985–86 |
AN |
67 |
117 |
3 |
3 |
3 |
|
|
1987 |
Village |
200 |
115 |
23 |
16 |
|||
|
South Africa |
1981 |
AN |
1,200 |
260 |
21 |
|||
|
1985–86 |
AN |
9,071 |
250 |
3 |
||||
|
1987 |
AN |
193 |
193 |
12 |
||||
|
1992a |
AN (asymptomatic) |
170 |
72 |
8 |
||||
|
South Africa |
1992a |
AN (unbooked) |
114 |
172 |
31 |
|||
|
Swaziland |
1978 |
FP |
52 |
167 |
6 |
6 |
||
|
1978 |
AN |
51 |
167 |
14 |
2 |
|||
|
1981a |
AN |
90 |
247 |
10 |
33 |
|||
|
1986 |
PP (asymptomatic) |
283 |
96 |
13 |
||||
|
Tanzania |
1983–84 |
AN |
5,430 |
51 |
19 |
16 |
||
|
1991–92 |
AN |
2,009 |
237 |
3 |
||||
|
1992a |
Community |
115 |
136 |
16 |
|
Country |
Year |
Population |
N |
Ref. |
Non-T |
Trep |
Non-T +Trep |
GUD |
|
Low-risk population |
||||||||
|
Uganda |
1986 |
Outpatients |
24 |
112 |
0 |
8 |
0 |
|
|
1986 |
AN |
1,011 |
43 |
26 |
||||
|
1992a |
AN |
405 |
190 |
10 |
||||
|
Zaire |
1988 |
PP |
314 |
198 |
2 |
|||
|
1990 |
AN |
701 |
142 |
1 |
||||
|
1991 |
AN |
1,857 |
174 |
2 |
1 |
2 |
||
|
Zambia |
1979–80 |
PP (asympt. live birth) |
261 |
258 |
29 |
4 |
||
|
1980a |
AN |
163 |
215 |
18 |
||||
|
1981 |
PP |
464 |
104 |
7 |
||||
|
1982a |
AN |
202 |
214 |
14 |
13 |
13 |
||
|
1984a |
AN |
101 |
13 |
|||||
|
1985–86 |
AN |
5,007 |
103 |
8 |
||||
|
Zimbabwe |
1985–87 |
PP (asymptomatic) |
104 |
67 |
19 |
9 |
||
|
High-risk population |
||||||||
|
Botswana |
1985–87 |
AN |
175 |
238 |
25 |
16 |
||
|
Central African Republic |
1988a |
Hosp. |
30 |
18 |
20 |
|||
|
Kenya |
1985 |
PP (pretiorum) |
157 |
79 |
6 |
|||
|
Malawi |
1992a |
STD |
505 |
58 |
25 |
|||
|
Nigeria |
1977–81 |
STD |
435 |
19 |
4 |
|||
|
Senegal |
1989–92 |
Gyn |
796 |
227 |
14 |
|||
|
Sudan |
1976–78 |
STD (vaginal D/C) |
152 |
200 |
8 |
0 |
||
|
Zambia |
1979–80 |
PP (stillbirth) |
262 |
258 |
54 |
29 |
||
|
Zimbabwe |
1982 |
STD (pregnant) |
160 |
133 |
4 |
|||
|
1990 |
STD |
100 |
153 |
44 |
33 |
|||
|
Very-high-risk population |
||||||||
|
Burkina Faso |
1990 |
CSW |
127 |
59 |
22 |
|||
|
Ethiopia |
1991a |
CSW |
203 |
90 |
37 |
|||
|
Gambia, The |
1986a |
CSW |
31 |
143 |
74 |
71 |
||
|
Kenya |
1981 |
CSW (upper SES) |
71 |
60 |
37 |
2 |
||
|
1981 |
CSW (middle SES) |
51 |
60 |
31 |
6 |
|||
|
1981 |
CSW (lower SES) |
71 |
60 |
53 |
6 |
|||
|
1985 |
CSW (low SES) |
64 |
126 |
55 |
42 |
|||
|
1989a |
CSW |
366 |
161 |
22 |
||||
|
Madagascar |
1987 |
CSW |
298 |
175 |
25 |
|||
|
Malawi |
1990 |
CSW (STD) |
274 |
57 |
21 |
|||
|
Rwanda |
1973 |
CSW |
43 |
166 |
30 |
|||
|
1987a |
CSW |
33 |
248 |
58 |
82 |
|||
|
Senegal |
1989–92 |
CSW |
653 |
70 |
6 |
31 |
||
|
1989–92 |
CSW |
397 |
227 |
40 |
||||
|
Somalia |
1985–86 |
CSW |
85 |
117 |
45 |
|||
|
1989 |
CSW |
57 |
232 |
51 |
||||
|
1990 |
CSW |
155 |
2 |
69 |
47 |
|||
|
1990a |
CSW |
89 |
38 |
28 |
||||
|
Sudan |
1987 |
CSW |
202 |
163 |
29 |
|||
|
Tanzania |
1992a |
CSW |
70 |
156 |
18 |
16 |
||
|
1991a |
CSW |
106 |
185 |
27 |
74 |
|||
|
Uganda |
1986 |
Bar girls |
36 |
112 |
46 |
6 |
||
|
Zaire |
1989a |
CSW |
801 |
150 |
28 |
16 |
7 |
|
|
1990a |
CSW |
1,233 |
125 |
28 |
16 |
7 |
||
|
NOTE: Non-T, non-treponemal test; Trep, treponemal test; Non-T+Trep, positive non-treponemal test confirmed by treponemal test in most cases; GUD, genital ulcer disease; D/C, discharge; AN, antenatal; PP, postpartum; FP, family planning; CSW, commercial sex worker; SES, socioeconomic status. a No study date. |
||||||||
TABLE 11-8 HIV-1 Infection
|
Country |
Year |
Population |
N |
Ref. |
HIV-1 |
|
Low-risk population |
|||||
|
Angola |
1986 |
AN (Luanda) |
357 |
25 |
0.3 |
|
Botswana |
1985–87 |
AN (Maun) |
113 |
121 |
0 |
|
Cameroon |
1987–90 |
AN (4 cities/towns) |
2,417 |
273 |
0.9 |
|
Côte d'Ivoire |
1992 |
AN (Abdidjan) |
12,750 |
239 |
9.5 |
|
1992 |
15–49 (Abidjan) |
10,136 |
1 |
9.4 |
|
|
1991 |
15–49 (Abidjan) |
3,833 |
1 |
8.7 |
|
|
1987 |
AN |
246 |
204 |
3.6 |
|
|
Ethiopia |
1991 |
AN (Diredawa) |
360 |
179 |
6.9 |
|
1991 |
AN (Metu) |
322 |
179 |
2.8 |
|
|
Gambia, The |
1990 |
AN (Banjul) |
1,057 |
149 |
0.3 |
|
1989 |
Vil (Gunjur) |
98 |
65 |
0 |
|
|
1988 |
Vil (Essau) |
96 |
65 |
0 |
|
|
1988 |
Vil (Kankunku) |
103 |
65 |
0 |
|
|
1988 |
Vil (Badjakunda) |
105 |
65 |
0 |
|
|
1988 |
Vil (Brikama) |
109 |
65 |
0 |
|
|
1989 |
Vil (Sibanor) |
108 |
65 |
0 |
|
|
1989 |
Vil (Diabugu) |
97 |
65 |
0 |
|
|
1989 |
Vil (Farafeni) |
99 |
65 |
0 |
|
|
1988 |
Vil (Gambisara) |
98 |
65 |
0 |
|
|
Guinea Bissau |
1989–90 |
AN (Bissau) |
272 |
181 |
0 |
|
Kenya |
1991 |
AN (national) |
4,599 |
159 |
10.1 |
|
1990 |
AN (national) |
7,232 |
159 |
6.6 |
|
|
Malawi |
1990a |
AN |
1,482 |
47 |
21.6 |
|
1988 |
AN |
247 |
48 |
19.0 |
|
|
Niger |
1987–88 |
AN (Niamey) |
1,477 |
205 |
0.1 |
|
Nigeria |
1988–90 |
AN (Lagos) |
500 |
5 |
1.0 |
|
Rwanda |
1992 |
AN (Kigali) |
128 |
84 |
31.3 |
|
1989 |
AN (urban) |
164 |
37 |
20.1 |
|
|
1989–91 |
AN (Butare) |
5,288 |
46 |
9.8 |
|
|
1989 |
AN (rural) |
193 |
37 |
4.2 |
|
|
Senegal |
1990 |
AN (Dakar Reg) |
182 |
157 |
1.2 |
|
1990 |
AN (Kaolak Reg) |
496 |
157 |
0.4 |
|
|
1991 |
AN (Dakar Reg) |
299 |
157 |
0.3 |
|
|
1992 |
AN (remote areas) |
781 |
227 |
0.3 |
|
|
1990 |
AN (Ziguinchor) |
825 |
157 |
0.2 |
|
|
1992 |
AN (various areas) |
2,120 |
88 |
0.2 |
|
|
1991–92 |
AN (Dakar) |
4,698 |
183 |
0.2 |
|
|
1991 |
AN (Kaolak Reg) |
240 |
157 |
0 |
|
|
1991 |
AN (Ziguinchor) |
715 |
157 |
0 |
|
|
1990 |
AN (St Louis) |
603 |
157 |
0 |
|
|
1991 |
AN (St Louis) |
418 |
157 |
0 |
|
|
South Africa |
1991 |
AN (national) |
17,155 |
157 |
0 |
|
1991 |
AN (national) |
1,576 |
157 |
0.6 |
|
|
Swaziland |
1990 |
AN |
266 |
2.3 |
|
|
Tanzania |
1991 |
All 15–54 (Arusha) |
148 |
197 |
12.8 |
|
1991–92 |
FP (Dar es Salaam) |
2,009 |
237 |
12.5 |
|
|
1992 |
AN (Mwanza region) |
24 |
11.6 |
||
|
Tanzania |
1991–92 |
FP (Dar es Salaam) |
2,285 |
120 |
11.5 |
|
1992 |
Rural (10–19) Kagera |
717 |
16 |
3.2 |
|
|
1992 |
All (Mwanza town) |
590 |
156 |
1.9 |
|
|
1987 |
AN (Moshi town) |
180 |
187 |
1.1 |
|
|
Uganda |
1992 |
AN (Aber) |
239 |
11 |
17.2 |
|
1990–91 |
AN (Hoima) |
346 |
3 |
13.0 |
|
|
1989 |
AN (Jinja) |
11 |
24.9 |
|
Country |
Year |
Population |
N |
Ref. |
HIV-1 |
|
Low-risk population |
|||||
|
Uganda |
1992 |
AN (Jinja) |
296 |
11 |
24.7 |
|
1991 |
AN (Jinja) |
300 |
11 |
22.0 |
|
|
1990–91 |
AN (Jinja) |
1,485 |
3 |
18.0 |
|
|
1990 |
AN (Jinja) |
11 |
15.8 |
||
|
1990–91 |
AN (Kabale) |
791 |
3 |
12.5 |
|
|
1990 |
AN (Kampala) |
628 |
245 |
36.6 |
|
|
1991 |
AN (Kampala) |
512 |
245 |
29.7 |
|
|
1992 |
AN (Kampala) |
285 |
11 |
29.5 |
|
|
1992 |
AN (Kampala) |
269 |
11 |
29.4 |
|
|
1989 |
AN (Kampala) |
2,017 |
245 |
29.0 |
|
|
1991 |
AN (Kampala) |
266 |
11 |
27.8 |
|
|
1990 |
AN (Kampala) |
426 |
245 |
27.0 |
|
|
1991 |
AN (Kampala) |
305 |
11 |
26.9 |
|
|
1988 |
AN (Kampala) |
88 |
245 |
25.0 |
|
|
1989 |
AN (Kampala) |
498 |
245 |
24.5 |
|
|
1992 |
AN (Kilembe) |
257 |
11 |
25.3 |
|
|
1992 |
AN (Mbale) |
255 |
11 |
17.3 |
|
|
1991 |
AN (Mbale) |
264 |
11 |
12.1 |
|
|
1991 |
AN (Mbale) |
241 |
12.0 |
||
|
1990 |
AN (Mbale) |
11 |
11.0 |
||
|
1989 |
AN (Mbale) |
11 |
9.0 |
||
|
1992 |
AN (Mbarara) |
205 |
11 |
30.2 |
|
|
1991 |
AN (Mbarara) |
217 |
11 |
24.4 |
|
|
1989 |
AN (Mbarara) |
1,017 |
245 |
21.6 |
|
|
1990–91 |
AN (Mbarara) |
1,098 |
3 |
20.6 |
|
|
1991 |
AN (Moyo) |
241 |
12.8 |
||
|
1990 |
AN (Moyo) |
241 |
11.0 |
||
|
1989 |
AN (Moyo) |
241 |
3.3 |
||
|
1989 |
AN (Moyo) |
615 |
245 |
3.3 |
|
|
1992 |
AN (Mutolere) |
11 |
5.6 |
||
|
1991 |
AN (Mutolere) |
11 |
4.1 |
||
|
1992 |
AN (Palisa) |
236 |
11 |
7.6 |
|
|
1991 |
AN (Potal) |
689 |
11 |
23.1 |
|
|
1992 |
AN (Tororo) |
222 |
11 |
13.1 |
|
|
1991 |
AN (Tororo) |
257 |
11 |
12.8 |
|
|
1990 |
AN (Tororo) |
11 |
4.1 |
||
|
Zaire |
1988–91 |
An (Kimpese) |
9,129 |
194 |
3.8 |
|
Zambia |
1991a |
AN (Solwezi—periurban) |
119 |
30.0 |
|
|
1991a |
AN (Kabompo—rural) |
119 |
20.0 |
||
|
1991a |
AN (Mukinge—rural) |
119 |
16.0 |
||
|
1991a |
AN (Mwinilunga—rural) |
119 |
9.0 |
||
|
Zimbabwe |
1989 |
AN (Mash. W) |
295 |
267 |
20.0 |
|
1989 |
AN (Mat. N) |
289 |
267 |
11.1 |
|
|
1989 |
AN (Midlands) |
298 |
267 |
7.7 |
|
|
High-risk population |
|||||
|
Central African Republic |
1989 |
STD (Bangui) |
76 |
95 |
21.0 |
|
Gabon |
1988 |
STD (Southeast) |
734 |
231 |
3.7 |
|
Rwanda |
1990 |
STD (Kigali) |
123 |
75.0 |
|
|
1989 |
STD (Kigali) |
123 |
74.0 |
||
|
1988 |
STD (Kigali) |
123 |
70.0 |
||
|
1991 |
STD (Kigali) |
123 |
69.0 |
||
|
Senegal |
1992 |
Gyn (remote areas) |
795 |
227 |
0.1 |
|
Country |
Year |
Population |
N |
Ref. |
HIV-1 |
|
High-risk population |
|||||
|
South Africa |
1990a |
STD (Durban) |
937 |
194 |
3.2 |
|
1991 |
STD (J'burg Black) |
1,626 |
223 |
9.8 |
|
|
Tanzania |
1991–92 |
STD (Mbeya) |
299 |
226 |
48.8 |
|
1992 |
STD (urban,w/syph) |
235 |
21.0 |
||
|
1992 |
STD (urban,w/out syph) |
235 |
14.0 |
||
|
1992 |
STD (north,w/syph) |
235 |
9.5 |
||
|
1992 |
STD (north,w/out syph) |
235 |
8.9 |
||
|
1992 |
STD (rural,w/syph) |
235 |
4.5 |
||
|
1992 |
STD (rural,w/out syph) |
235 |
3.0 |
||
|
Uganda |
1990a |
STD (Kampala) |
96 |
98 |
60.5 |
|
1989–91 |
STD (Kampala, 20–29) |
107 |
59.0 |
||
|
1989–91 |
STD (Kampala, 30–39) |
107 |
55.0 |
||
|
1989–91 |
STD (Kampala, 10–19) |
107 |
45.0 |
||
|
1989–91 |
STD (Kampala, 40–49) |
107 |
40.0 |
||
|
1989–91 |
STD (Kampala, 50+) |
107 |
20.0 |
||
|
Zimbabwe |
1991 |
STD (Karoi) |
72 |
152 |
65.3 |
|
Very-high-risk population |
|||||
|
Burkina Faso |
1990a |
CSW (rural) |
38 |
88 |
44.7 |
|
Cameroon |
1988 |
CSW (Younde) |
168 |
122 |
7.1 |
|
Côte d'Ivoire |
1987 |
CSW |
210 |
204 |
35.3 |
|
Ethiopia |
1990 |
Bar girls (Adis) |
858 |
179 |
42.0 |
|
1989 |
Bar girls (Adis) |
966 |
179 |
22.0 |
|
|
1989 |
CSW (Adaitu) |
110 |
179 |
41.8 |
|
|
1988 |
CSW (Adaitu) |
116 |
179 |
32.8 |
|
|
1990 |
CSW (Adis) |
1,225 |
179 |
54.2 |
|
|
1989 |
CSW (Adis) |
2,663 |
179 |
24.3 |
|
|
1988 |
CSW (Adis) |
330 |
179 |
19.4 |
|
|
1988 |
CSW (Arbaminch) |
255 |
179 |
8.2 |
|
|
1989 |
CSW (Asmara) |
379 |
179 |
5.8 |
|
|
1988 |
CSW (Asmara) |
389 |
179 |
2.3 |
|
|
1988 |
CSW (Asseb) |
352 |
179 |
31.5 |
|
|
1988 |
CSW (Assela) |
326 |
179 |
12.9 |
|
|
1988 |
CSW (Awasa) |
260 |
179 |
15.4 |
|
|
1991 |
CSW (Baherdar) |
366 |
179 |
69.4 |
|
|
1990 |
CSW (Baherdar) |
362 |
179 |
55.0 |
|
|
1989 |
CSW (Baherdar) |
353 |
179 |
48.2 |
|
|
Ethiopia |
1988 |
CSW (Baherdar) |
324 |
179 |
35.8 |
|
1988 |
CSW (Bobi) |
213 |
179 |
12.2 |
|
|
1988 |
CSW (Dessie) |
312 |
179 |
38.1 |
|
|
1990 |
CSW (Diredawa) |
366 |
179 |
48.1 |
|
|
1989 |
CSW (Diredawa) |
361 |
179 |
30.5 |
|
|
1988 |
CSW (Diredawa) |
361 |
179 |
18.0 |
|
|
1988 |
CSW (Gewane) |
119 |
179 |
30.3 |
|
|
1988 |
CSW (Gonder) |
367 |
179 |
14.7 |
|
|
1988 |
CSW (Jimma) |
309 |
179 |
9.7 |
|
|
1988 |
CSW (Keren) |
361 |
179 |
2.5 |
|
|
1988 |
CSW (Massawa) |
318 |
179 |
1.3 |
|
|
1988 |
CSW (Mekele) |
363 |
179 |
24.2 |
|
|
1988 |
CSW (Meteka) |
79 |
179 |
17.9 |
|
|
1990 |
CSW (Metu) |
165 |
179 |
36.4 |
|
|
1989 |
CSW (Metu) |
240 |
179 |
12.5 |
|
|
1988 |
CSW (Metu) |
262 |
179 |
5.3 |
|
|
1988 |
CSW (Moyale) |
99 |
179 |
16.2 |
|
Country |
Year |
Population |
N |
Ref. |
HIV-1 |
|
Very-high-risk population |
|||||
|
Ethiopia |
1991 |
CSW (Nazareth) |
328 |
179 |
65.6 |
|
1990 |
CSW (Nazareth) |
315 |
179 |
52.4 |
|
|
1989 |
CSW (Nazareth) |
354 |
179 |
31.1 |
|
|
1988 |
CSW (Nazareth) |
333 |
179 |
19.5 |
|
|
1989 |
CSW (Nekemt) |
294 |
179 |
31.3 |
|
|
1988 |
CSW (Nekemt) |
274 |
179 |
15.3 |
|
|
1988 |
CSW (Shashemene) |
325 |
179 |
19.4 |
|
|
Ghana |
1986–87 |
CSW (return from Côte d'Ivoire) |
151 |
182 |
49.0 |
|
Kenya |
1991 |
CSW (Nairobi) |
490 |
40 |
85.9 |
|
1991 |
CSW (Nairobi) |
524 |
124 |
75.2 |
|
|
1990 |
CSW (Nairobi) |
981 |
124 |
73.8 |
|
|
1989 |
CSW (Nairobi) |
656 |
124 |
67.2 |
|
|
Niger |
1987–89 |
CSW (Niamey) |
610 |
205 |
4.9 |
|
Nigeria |
1990 |
CSW (Lagos) |
546 |
55 |
10.3 |
|
1988–90 |
CSW (Lagos) |
117 |
5 |
2.6 |
|
|
Senegal |
1985–92 |
CSW (Dakar) |
1,364 |
158 |
4.7 |
|
1990 |
CSW (Dakar) |
471 |
157 |
4.5 |
|
|
1991 |
CSW (Dakar) |
665 |
157 |
4.4 |
|
|
1989–91 |
CSW (Dakar,Regis) |
653 |
96 |
4.4 |
|
|
1985–92 |
CSW (Dakar,Regis) |
2,216 |
158 |
4.3 |
|
|
1991 |
CSW (Kaolack) |
113 |
157 |
11.5 |
|
|
1990 |
CSW (Kaolack) |
106 |
157 |
4.7 |
|
|
1990 |
CSW (St Louis) |
74 |
157 |
2.7 |
|
|
1991 |
CSW (St Louis) |
69 |
157 |
0 |
|
|
1992 |
CSW (various area) |
1,342 |
88 |
2.4 |
|
|
1991 |
CSW (Ziguinchor) |
138 |
157 |
1.4 |
|
|
1990 |
CSW (Ziguinchor) |
194 |
157 |
0.5 |
|
|
1992 |
CSW (remote) |
397 |
227 |
13.8 |
|
|
Somalia |
1990 |
CSW (Mogad, Merca, Ch) |
303 |
52 |
3.0 |
|
Sudan |
1989 |
CSW (Juba) |
50 |
164 |
16.0 |
|
Tanzania |
1987–89 |
CSW (Moshi—Arusha) |
212 |
186 |
72.2 |
|
1987 |
CSW (Kili) |
52 |
187 |
57.7 |
|
|
1992 |
CSW (Niwanza town) |
103 |
156 |
39.8 |
|
|
Zaire |
1988 |
CSW (Kin) |
801 |
130 |
37.2 |
|
1988a |
CSW (Kin) |
377 |
148 |
26.8 |
|
|
a No study date. NOTE: AN, antenatal; FP, family planning; Vil, village; w/syph, with syphilis; w/out syph, without syphilis; CSW, commercial sex worker. |
|||||