APPENDIX B
Screening for Skin Cancer
Mark Helfand, M.D., M.P.H.,* Susan Mahon, M.P.H., and Karen Eden, Ph.D.
INTRODUCTION
In the United States in 1999, approximately one million new cases of basal cell and squamous cell carcinoma, and about 44,000 new cases of malignant melanoma, are expected to be diagnosed.1 Malignant melanoma is often lethal, and its incidence in the United States has increased rapidly over the past two decades. Nonmelanoma skin cancer is seldom lethal but, if advanced, can cause severe disfigurement and morbidity.
Advanced melanoma and invasive squamous cell carcinoma of the skin occur most often in the elderly, especially elderly men. Early detection and treatment of melanoma might reduce mortality, while early detection and treatment of squamous cell carcinoma and basal cell carcinoma might prevent major disfigurement, reduce the need for expensive reconstructive surgery, and to a lesser extent, prevent mortality.
In this paper, we examine published data on the effectiveness of screening for skin cancer by a physician. Specifically, we examine the accuracy of the tests used for screening, the diagnostic yield of screening in the general population, and evidence that treatment of cancers found by screening improves outcomes.
We use the term “screening” to denote a systematic effort to detect unsuspected disease by performing a total-body skin examination or by assessing the risk for skin cancer in all patients seen in the primary care setting. We did not
This evidence review was developed for the Institute of Medicine and the U.S. Preventive Services Task Force and was reviewed and approved by both groups. This paper may differ slightly in format from the version that will be released by the Task Force (expected early in 2000).
examine the effect of skin surveillance on children or on patients with familial syndromes that confer a high risk of melanoma.
We also did not examine the value of routine diagnosis and treatment of skin cancer in clinical practice. In everyday primary care, the clinician sees the skin of every patient’s face and, in many, the extremities, chest, and back. Clinicians almost universally agree that incidental discovery of a suspicious skin lesion should prompt an evaluation, including a skin biopsy and a thorough inspection of the skin. The data we reviewed about screening do not address the value of attention to the skin as part of conscientious clinical care.
Other strategies to prevent skin cancer, such as promotion and counseling to reduce risky health behaviors and skin self-examination, are not addressed in this review. However, many studies combine screening with health promotion programs, and screening may itself contribute to primary prevention, since it provides the physician with an opportunity to increase awareness of skin cancer and to demonstrate examination techniques that patients can apply themselves.
EPIDEMIOLOGY AND BURDEN OF SUFFERING
Melanoma
In the United States, the lifetime risk of being diagnosed with melanoma is 1.74 percent in white men and 1.28 percent in white women. The lifetime risk of dying of melanoma is 0.36 percent in men and 0.21 percent in women. Between 1973 and 1995, the incidence of melanoma in the United States increased about 4 percent per year, from 5.7 per 100,000 in 1973 to 13.3 per 100,000 in 1995, according to data from the Surveillance, Epidemiology, and End Results program (SEER) of the National Cancer Institute.2 By comparison, the overall rate in Queensland, Australia is 55 per 100,000.
The elderly and, in particular, elderly men bear a disproportionate burden of morbidity and mortality from melanoma. As shown in Figure B-1, older men have the highest incidence of invasive melanoma. In 1995, the age-adjusted incidence rate was 68.7 per 100,000 in white men over age 65 and 30.6 per 100,000 in white women over 65. Men over 65 years of age, who constitute 5.2 percent of the U.S. population, have 22 percent of newly diagnosed malignant melanomas each year; women over 65 who constitute 7.4 percent of the population, have 14 percent.
Melanoma in the elderly is not only more common, but also more lethal than in younger populations. In Australia, where for many years public education about melanoma has been intense, 75 percent of “thick” (>3 mm) melanoma lesions and 75 percent of deaths occur in people over 50 years of age; 50 percent of deaths occur in men over 50.3 Similarly, in the United States, about 50 percent of deaths from melanoma are in men over 50 years of age.2 Figure B-2
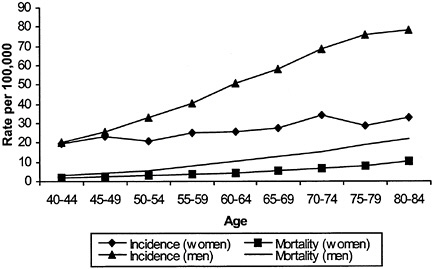
FIGURE B-1 Melanoma age-specific incidence and mortality.
shows that melanoma in the elderly, particularly elderly men, is more likely to be detected in advanced stages. For men in their 40s, for example, there are six times as many cancers diagnosed as deaths; for men in their 70s, there are about four cancers diagnosed per death. Some experts argue that the elderly, particularly elderly men, may have lower “skin awareness” and lower rates of skin self-examination, resulting in higher rates of advanced melanoma.4
Overall mortality from melanoma has increased. Between 1973 and 1995, overall mortality rates for melanoma increased by 1.3 percent per year, from 1.6 per 100,000 in 1973 to 2.2 per 100,000 in 1995. Nearly all of the increase was in white men (2.2 to 3.6 percent), especially older white men. Five-year survival for melanoma has improved to 88 percent currently from 80 percent 20 years ago. During this time, the rate of diagnosis of “early” or thin melanoma increased sharply, but so did the incidence of thicker (>3 mm) melanomas.5
Changes over time in ascertainment, diagnostic criteria, self-examination, and registry procedures make it difficult to draw reliable inferences about the effectiveness of early detection from epidemiologic data.5–7 In an analysis of trends in Australia and New Zealand, Burton and colleagues noted that although there has been a huge increase in the incidence of very thin melanomas, the incidence of thick melanomas has increased as well.8 Some experts interpret this to mean that increased surveillance in the population may detect a relatively unaggressive, unimportant type of thin melanoma.7–10 Increased detection of these very thin, nonmetastasizing melanomas would increase the incidence and five-year survival rates for melanoma but would have little impact on mortality. However, in contrast to prostate and thyroid cancers, in which a large
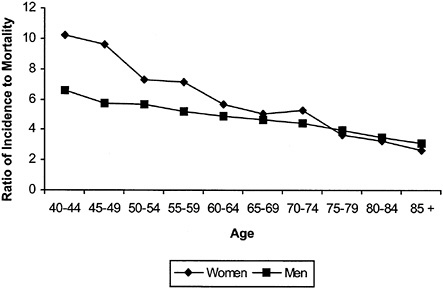
FIGURE B-2 Ratio of incidence to mortality by age.
reservoir of unaggressive cancers are known to exist, longitudinal studies of melanoma have not established the frequency or existence of histologically malignant, but behaviorally benign, melanoma in the general population.
Nonmelanoma Skin Cancer
Rates of nonmelanoma skin cancer in the United States are difficult to determine, since these cancers are not typically tracked by cancer registries. Cancer registries in Denmark and Canada do include nonmelanoma skin cancers. In British Columbia, Canada, the age-standardized incidence rate for basal cell cancer in men was 70.7 per 100,000 in 1973, increasing to 120.4 per 100,000 in 1987.11 In women, basal cell cancer incidence increased from 61.5 to 92.2 per 100,000 over the same period. Squamous cell cancer incidence rose from 16.6 to 31.2 per 100,000 in men and from 9.4 to 16.9 per 100,000 in women.
Population-based surveys show substantial variation among geographic areas. In Queensland, Australia in the early 1990s, the age-adjusted incidence rates of basal cell carcinoma among men and women were 2,074 and 1,579 per 100,000 per year, respectively.12 Squamous cell carcinoma occurred at half the rate of basal cell carcinoma among men and at about one-third the rate among women. In Geraldton, Western Australia, the estimated incidence of basal cell carcinoma was 1,335 per 100,000 in men and 817 per 100,000 in women.13 In that community, the prevalence of nonmelanoma skin cancer in men and women
under 65 years of age was 7 percent in men and 4.7 percent in women; approximately 90 percent of these were basal cell cancers. A survey of one large health plan in Albuquerque, New Mexico, which was not population based, found age-standardized basal cell cancer rates of 1,073 per 100,000 in non-Hispanic white men and 415 per 100,000 in non-Hispanic white women. Squamous cell cancer rates were 214 and 50 per 100,000 for non-Hispanic white men and women, respectively.
Rates are much lower in other U.S. studies. A population-based study in Rochester, Minnesota, covering the years 1976–1984 found that age-standardized incidence rates per year of basal cell cancer were 175 per 100,000 in men and 124 per 100,000 in women.14,15 Rates of squamous cell cancer were 63.1 per 100,000 in men and 22.5 per 100,000 in women. Rates of both basal cell cancer and squamous cell cancer increased with advancing age. A population-based study of NMSC in New Hampshire suggests that incidence rates are increasing. This study looked at incidence rates for two time periods, 1979–1980 and 1993–1994.16 In men, the age-adjusted incidence of basal cell cancer increased from 170 per 100,000 in 1979–1980 to 310 per 100,000 in 1993–1994; and in women basal cell cancer incidence rose from 91 to 166 per 100,000 over the same period. For squamous cell cancer, incidence rates in men rose from 29 to 97 per 100,000 over these periods, and in women, squamous cell cancer incidence rose from 7 to 32 per 100,000.
NATURAL HISTORY, DIAGNOSIS, AND STAGING OF SKIN CANCER
Melanoma
There are four major subtypes of melanoma: superficial spreading, nodular, lentigo maligna melanoma, and acral lentiginous melanoma.17 Superficial spreading melanoma, the most common subtype in whites, is usually diagnosed at an early (thin) stage before there is a high risk of metastasis. Nodular melanoma is the second most common subtype in whites. Nodular melanomas are difficult for patients to find and are usually diagnosed in a more advanced stage.18 The natural history of nodular melanoma is controversial.19 The prevailing view is that nodular melanoma is characterized by rapid, early vertical growth and lack of an identifiable radial growth phase.
To determine which skin lesions are suspicious for melanoma, some clinicians in the United States use the ABCD checklist for detecting melanoma.20 With this system, pigmented lesions are classified as suspicious for melanoma if they have an asymmetric shape; an irregular border that is scalloped, uneven, or ragged; varied color; or a diameter larger than 6 mm. Some add a fifth criterion (ABCDE) for elevation or enlargement. Some clinicians in the United Kingdom use a seven-point checklist that includes change in mole size, shape,
and color; crusting or bleeding; sensory change; and a mole greater than 7 mm in diameter.20
Once a lesion suspected to be cancer is identified, one of several biopsy techniques is employed to obtain tissue for analysis. The pathological diagnosis of suspicious pigmented lesions can be difficult, especially for borderline and in situ neoplasms. In one recent study, four histopathologists evaluated 140 slides and classified each lesion as “melanoma” or “other pigmented lesion”; they were in agreement on diagnoses for 74 percent (kappa=.61) of the slides.21 Similarly, when eight expert pathologists (recruited based on publications and reputations) classified 37 slides as “benign,” “malignant,” or “indeterminate,” they had complete agreement, or only one discordant, on 62 percent (kappa= .50) of the cases.22
Stage is the most important prognostic factor in melanoma. The American Joint Commission on Cancer Classification, which is based on the TNM (tumor, node, metastasis) system, describes the stages from I to IV. Stage I is a primary tumor less than 1.5 mm in thickness with no regional lymph node metastases; Stage II is a primary tumor 1.5–4.0 mm in thickness with no regional lymph node metastases; Stage III is any primary tumor with regional lymph node metastases or in-transit metastases; and Stage IV is any primary tumor with distant metastases.23 According to SEER data through 1995, five-year relative survival rates for localized, regional, and distant disease were 96 percent, 59 percent, and 12 percent, respectively.2
The thickness of the primary tumor is the strongest predictor of prognosis. To measure thickness of a melanoma, the pathologist uses a device called a “micrometer,” similar to a small ruler under the microscope. This technique is called the Breslow measurement.24 In general, melanomas less than 1 mm in depth have a very small chance of metastasizing. Five-year survival for those with melanomas between 1.5 mm and 4 mm is approximately 70 percent, and for those with melanomas thicker than 4 mm, it is about 45 percent. Thickness of the melanoma also guides the choice of therapy.
Nonmelanoma Skin Cancers
Basal cell carcinoma and squamous cell carcinoma are the most common forms of skin cancer. Despite their very high incidence they account for less than 0.1 percent of cancer deaths. There are several morphological types of basal cell cancer, such as nodular, ulcerative, and plaque-like but regardless of type, metastasis is very rare. Basal cell carcinoma can be locally destructive and frequently recurs.
Squamous cell cancers usually occur on chronically sun-exposed areas of the skin, especially the face, ears, or backs of the hands. Squamous cell cancer has the potential to metastasize and may account for up to 20 percent of deaths
from skin cancer. A large primary tumor (>2 cm) is associated with an increased risk of metastasis.
Most studies of the natural history of nonmelanoma skin cancer have been done in selected patients who have an elevated risk due to environmental exposures, such as Psorolen plus ultraviolet-A (UV-A) radiation for psoriasis.25,26 Patients with these exposures may constitute a substantial proportion of all patients who die of metastatic squamous cell cancer.27 Very elderly men are also overrepresented among patients who die of squamous cell cancer. While there is strong suspicion on clinical grounds that advanced locally invasive or metastatic nonmelanoma skin cancers result from medical neglect, careful studies of the rate of progress of nonmelanoma skin cancers in the elderly are lacking.
RECOMMENDATIONS OF SECOND TASK FORCE AND OTHERS
Current recommendations of professional societies regarding screening for skin cancer vary. The American Cancer Society recommends skin examination every three years for people between 20 and 40 years of age and yearly for anyone over 40. The American College of Preventive Medicine and the U.S. Preventive Services Task Force28 recommend total body skin examination in high-risk individuals who see a physician for other reasons, but they do not recommend routine screening. All of these organizations advise some form of public or patient education to change behaviors that may increase the risk of skin cancer and increase the likelihood of early self-detection.
ANALYTIC FRAMEWORK AND KEY QUESTIONS
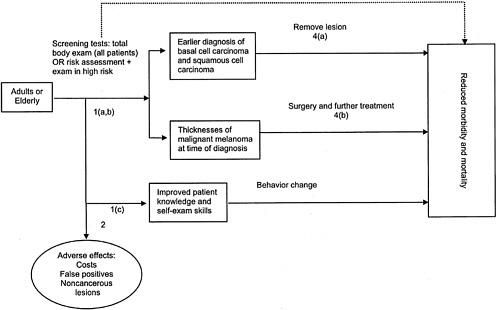
Before the consequences of screening can be estimated, a necessary first step is to formulate the screening problem by specifying the population that screening is intended to reach; the screening tests, follow-up tests, and treatments that will be used; and the types of outcomes that will be affected by screening.
The analytic framework in Figure B-3 shows the interventions, intermediate outcome measures, and health outcome measures we examined. The accompanying key questions (Figure B-4) correspond to the numbered arrows in the analytic framework and articulate the main questions that guided our literature review and that are addressed in the results section of this appendix.
We studied screening in the general adult population and in the elderly, the group with the highest prevalence of and mortality from skin cancer. We have included studies of both mass-screening and case-finding programs to detect and treat melanoma and nonmelanoma skin cancer in the general population. In mass-screening programs, self-selected individuals respond to an invitation to undergo a total-body skin examination. Those with suspicious skin lesions are
|
Accuracy of Screening Arrow 1a
Consequences of Screening Arrow 1b
Arrow 1c What is the effect of screening on patients’ skin knowledge and self-care behavior? Arrow 2 What are the adverse effects of screening? Arrow 3 Is there direct evidence that screening for skin cancers leads to reduced morbidity and mortality? Effectiveness of Early Treatment Arrow 4a Does treatment of non-melanoma skin cancer found by screening reduce morbidity and mortality? Arrow 4b Does treatment of malignant melanoma found by screening reduce morbidity and mortality? |
FIGURE B-4 Key questions.
referred to their primary care physician or to a specialist for further evaluation. In case-finding programs, a total-body skin examination is offered to individuals who see a primary care physician for other reasons. A total-body skin examination may be offered to every individual or to selected individuals considered at high risk of skin cancer. Because the great majority of Medicare beneficiaries do see a physician each year, case-finding in physicians’ offices might reach high-risk patients, especially elderly men, who are less likely than other groups to respond to an invitation to be screened in a mass screening program.
We sought studies of the accuracy of two methods of screening for skin cancer (Arrow 1a): (1) performing a total-body skin examination in all patients seen in the primary care setting, and (2) assessing the risk for skin cancer in all patients, followed by a total-body skin examination in those found to be at high risk. The primary aim of these strategies is earlier detection of melanoma, for which an examination confined to areas not covered by clothing is likely to miss a high proportion of potentially lethal cancers.29 To assess the accuracy of these methods, both for melanoma and for nonmelanoma skin cancer, we sought studies that used these initial tests to screen in the general population or in the elderly and then confirmed positive screening test results with skin biopsy results.
We examined the consequences of screening on detection of squamous cell carcinoma, basal cell carcinoma, and malignant melanoma (Arrow 1b). Specifically, we examined how often patients are found to have skin cancer, how often suspected skin cancer is confirmed by biopsy, and at what stage cancer is found.
In addition to early detection, screening itself might confer a potential benefit by improving patients’ knowledge and self-examination skills. We therefore sought evidence about the effect of screening on patients’ health beliefs and practices regarding skin cancer prevention (Arrow 1c). We also considered the adverse effects of screening, including the frequency and consequences of false positive examinations or biopsies and the diagnosis of noncancerous lesions that may not require treatment (Arrow 2).
In considering outcomes, we sought, but did not find, direct evidence from controlled studies of the effect of screening on health outcomes (Arrow 3, dotted line) such as mortality and quality of life. In the absence of randomized trials of screening, these links may be made by studies of the association between delay of diagnosis and the outcome of cancer or of the outcomes of screened versus nonscreened populations.
Note that we did not examine the effectiveness or adverse consequences of various treatments for skin cancer, but rather investigated the evidence that detection of earlier cancers by screening in the general population is associated with reduced mortality and morbidity.
METHODS
Literature Review
To find relevant articles on screening for skin cancer, we searched the MEDLINE database for papers published in 1994 or later, using the search string in Addendum B-1 (see page 214). We conducted monthly updates during the course of the project. We included studies if they contained data on yield of screening, screening tests, risk factors, risk assessment, effectiveness of early detection, or cost-effectiveness.
Two reviewers evaluated abstracts for inclusion. The reviewers also searched the reference lists of relevant reviews. Of 54 included studies, 5 contained data on accuracy of screening tests, 24 contained data on yield of screening, 8 contained data on stage or thickness of lesions found through screening, 11 addressed risk assessment, and 7 addressed the effectiveness of early detection (some studies addressed more than one topic) (see Addendum B-2, page 215). We retrieved the full text of these articles and abstracted the data as described below. In addition, we retrieved the full text of 47 studies of various risk factors for skin cancer. We read these articles but did not systematically abstract them.
We identified the most important studies from before 1994 from the Guide to Clinical Preventive Services, Second Edition;28 from high-quality reviews published in 1994 and 1996;30,31 from reference lists of recent studies; and from experts. We found that many relevant publications from before 1994 were preliminary results of programs discussed in greater detail in more recent publications.
Data Extraction and Synthesis
We abstracted the following descriptive information from full-text, published studies of screening and recorded it in an electronic database: study type (mass screening, population based, case finding, other); setting (hospital, community, specialty clinic, primary care, other); population (percent white, age), recruitment (volunteers, invitation, random sampling), screening test (total-body skin examination, partial skin examination, lesion-specific examination, other); examiner (dermatologist, primary care physician, other); advertising targeted at high-risk groups or not targeted; reported risk factors of participants; and procedure for referring patients found to have a positive screen.
We also abstracted the number and probability of the following events from each study: referrals for skin examination; compliance with referral; suspected basal cell cancers, squamous cell cancers, actinic keratoses, and melanoma; confirmed melanoma and melanoma in situ; negative screening examinations; biopsies performed; the number with confirmed melanoma and number with suspicious melanoma; and the number with confirmed melanoma and number of all
suspicious lesions. When available, the type, stage, and thickness of lesions found through screening were also recorded.
For studies that reported test performance, we also recorded the definition of a suspicious lesion, the “gold standard” determination of disease, the number of true positive, false positive, true negative, and false negative test results. To analyze data from these studies, we defined sensitivity as the proportion of people with a histologic diagnosis of skin cancer who had a positive test result—that is, a suspicious lesion on examination. Specificity was defined as the proportion of people who did not have skin cancer who had no suspicious lesions detected during the skin exam.
The positive predictive value (PV+) was computed in two ways to account for noncompliance in studies. The lower bound (Low PV+) of the predictive value was computed by dividing the number of patients with confirmed skin cancer by the number of patients who were diagnosed with a suspicious lesion. The upper bound (High PV+) was computed by dividing the number of patients with confirmed skin cancer by the number of patients who had biopsies. If the study provided sufficient detail, we calculated the PV+ of examination for each type of skin cancer. Most studies, however, did not report results in sufficient detail; for these, we combined the results for different types of skin cancer.
We calculated likelihood ratios (LRs) for each study. The LR for a positive test was calculated using the formula
LR=[High PV/(1−High PV)]/[p(cancer)/(1−p(cancer)],
where p(cancer) is the observed prevalence of disease, estimated as p(cancer)= (number of true positives+number of false negatives)/(number of patients screened).32
This formula was derived from the odds ratio form of Bayes’ theorem.33 The advantage of using the odds ratio form is that LR can be computed in studies that reported findings only for patients with positive tests. The computation is based on the following,
Posttest odds=pretest odds×likelihood ratio.
In computing the LRs, we used the High PV+, which included only those patients who went on for biopsy. We assumed that the High PV+ was more representative of screening in a primary care setting. These patients would be more likely to follow through with a biopsy than those attending a mass screening.
In studies that did not measure the false negative rate, we assumed that there were no false negative results. The observed prevalence was computed by dividing the number of patients with true positive results by the number of screened patients. If there were, in fact, patients in these studies with false nega-
tive results, the observed prevalence was underreported. Since computation of the LR depends on an accurate measure of prevalence, LRs computed with an underreported prevalence are inflated. We performed a sensitivity analysis to record the effect on the estimate of LR when the number of false negatives and the prevalence were varied over a reasonable range of values.
RESULTS
Accuracy of Screening Tests
Before considering the consequences of screening, we raised and attempted to answer the following questions about the accuracy of the screening tests most commonly used in detecting skin cancers. The numbers and letters in parentheses relate to the arrows in the analytic framework (Figure B-3) and the related key questions (Figure B-4).
How accurate is total-body skin examination in the detection of skin cancer? (Arrow 1a, Q1)
Screening Studies
Table B-1 summarizes five recent prospective studies of the accuracy of skin examination in screening programs. In all of the studies the participants were self-selected individuals who responded to an advertisement that may have emphasized skin cancer risk factors. In some studies, total-body skin examinations were performed on all participants; in others, the examination focused on specific lesions identified by the patient. In all but one study,34 the skin exams were conducted by dermatologists.
In these studies, a positive screening test result was defined as the clinical diagnosis of a suspicious skin lesion. Histologic diagnosis on skin biopsy was the gold standard for determination of disease. In four of the studies, skin examination was considered positive if the lesion was suspicious of any skin cancer.34–38 In these studies, from 4.2 to 28.4 percent of subjects had suspicious lesions, and from 1 percent to 6 percent proved to have skin cancer. Basal cell cancer accounted for 74–93 percent of the confirmed skin cancer cases; melanoma, 7–27 percent; and squamous cell cancer, 0–4 percent of the skin cancer cases. This suggests that basal cell cancers contributed heavily to the summary estimates of the accuracy of skin examination reported in these studies.
Work-up bias was present in all of these studies—that is, only suspicious lesions were biopsied. This design permits measurement of the positive predictive value of screening, but not of the false negative rate. In Table B-1, High PV+ estimates (as defined in the methods section, above) for all types of skin cancer
values ranged from 0.30 to 0.58, indicating that between 30 and 58 percent of patients found to have a suspicious lesion were eventually diagnosed to have skin cancer on biopsy.34–38 The variation in High PV+ may be related to variation in the prevalence of disease, which in turn could be related to the type of patients recruited (high risk or not targeted).
The Low PV+ (also defined in the methods section) takes into account the effect of noncompliance with the recommendation to have a biopsy done. The two smallest studies34,37 had great differences between the lower and upper estimates of predictive value. In both of these small studies the percentage of patients with suspicious lesions was high (21 percent34 and 28 percent37), but the percentage of those patients who went on for biopsy was low (36 percent37 and 65 percent34). The clinicians in these studies appeared to have less rigid criteria for diagnosing skin cancer from skin exams than did clinicians in the other, larger studies, since they were four times as likely to find a suspicious lesion. Low PV+ results may better represent the actual impact of mass screening, in which the patient must comply with referral to a physician for biopsy in order for screening to affect follow-up and treatment.
The last study shown in Table B-1 focuses on detection of melanoma in self-selected individuals.39 The study demonstrated that dermatologists found lesions suspicious for melanoma in a very small proportion of individuals. In this study, 282,555 members of the general public were recruited for free examinations without regard to risk factors for skin cancer. Clinical suspicion was classified as “suspected melanoma” or “rule-out melanoma.” Only 0.3 percent (n =763) of the participants had a clinical diagnosis of suspected melanoma at the skin examination. Of the 679 patients who went on for biopsies, 130 patients had melanoma (PV+=0.191). Although the use of a lower cutoff, rule-out melanoma, identified an additional 234 patients with melanoma, an additional 2,316 patients without melanoma were biopsied and the predictive value was 0.09. Interestingly, compliance with biopsy was significantly lower for participants given a diagnosis of rule-out melanoma—0.69, compared to 0.89 for patients with a diagnosis of suspected melanoma.
Although data are sparse, the sensitivity of a complete skin examination performed by a dermatologist is thought to be high. One of the studies in Table B-1 provided an indirect measure of sensitivity and specificity through registry data.40 In this study, performed in the Netherlands, 1,551 of 1,763 participants who had negative screening examinations consented to be followed through two population-based cancer registries for 42 months. Similarly, 87 of 93 patients with positive tests were also followed. Fifteen patients with negative screening results (about 1 percent) appeared in at least one of the cancer registries to have skin cancer. Review of medical records revealed that 12 of the patients had new lesions, while 3 patients had documented lesions that had been misdiagnosed. One patient had a basal cell carcinoma on his back that had been diagnosed as a common nevus. A second patient had a squamous cell carcinoma on his wrist
TABLE B-1 Skin Cancer Screening Accuracy
|
Suspicious Lesions |
Probability of Cancer |
BCC/ Skin Cancer (%) |
MM/ Skin Cancer (%) |
SCC/ Skin Cancer (%) Positive |
Predictive Value |
Biopsy Ratea |
Likelihood Ratiob |
||
|
n |
% |
Low |
High |
||||||
|
93 |
4.7 |
0.031 |
85.1 |
12.8 |
2.1 |
0.51 |
0.54 |
0.935 |
37.32 |
|
51 |
20.6 |
0.057 |
92.9 |
7.1 |
0.0 |
0.27 |
0.42 |
0.647 |
12.26 |
|
173 |
4.2 |
0.011 |
73.5 |
26.5 |
0.0 |
0.28 |
0.30 |
0.912 |
37.95 |
|
132 |
28.4 |
0.060 |
85.2 |
11.1 |
3.7 |
0.21 |
0.58 |
0.364 |
21.80 |
|
763 |
0.3 |
0.001 |
|
0.17 |
0.19 |
0.890 |
183.57 |
||
|
3,695 |
1.3 |
0.001 |
0.06 |
0.09 |
0.690 |
78.33 |
|||
that was recorded as a seborrheic keratosis. The third patient had a basal cell carcinoma on the forehead originally diagnosed as an actinic keratosis. Overall sensitivity of the initial examination was 0.940 and specificity was 0.975. For a patient with a negative initial skin examination, the probability of having no skin cancer on follow-up was 0.998.
While this follow-up study40 provides the best available data on sensitivity and specificity in a screening setting, a caution should be raised about generalizing this study to screening in a primary care setting. The examiners were dermatologists. Additionally, it unclear how many lesions diagnosed as precursor lesions were, in fact, skin cancer. Patients with precursor lesions (n=111) were excluded from the study.
Since positive predictive values are highly dependent on the probability of skin cancer in each study, positive likelihood ratios were computed. The positive LRs in Table B-1 ranged from 12 to 37 for patients screened for all types of skin cancer.34,35,37,40,41 Thus, according to the conditional probability definition of LR, p(positive test|skin cancer)/p(positive test|no skin cancer), patients with suspicious lesions for any type of skin cancer were 12 to 38 times more likely to have skin cancer than to not have skin cancer. When screened specifically for melanoma, patients with a suspected melanoma diagnosis were 184 times more likely to have melanoma than to not have melanoma.39 Patients with a rule-out melanoma diagnosis were 78 times more likely to have melanoma than to not have melanoma.
Most LR results shown in Table B-1 must be considered with caution. With the exception of one study,40 the computation of LRs assumed that there were no false negative results. In this study, 3 of 50 skin cancer patients had false negative results, suggesting that skin exams have a false negative rate of 6 percent.40 When the LR was recomputed in this study and the false negative rate was assumed to be 0 percent, the LR increased from 37.32 (using false negative rate of 6 percent) to 39.78 (0 percent false negative rate). Similarly, if the false negative rate was actually 10 percent, the LR for this study would be 35.84. If it were assumed to be 0 percent, the computed LR would be 39.78. Taken to an extreme, if the examiner missed one of every five patients with skin cancer (a false negative rate of 20 percent), the computed LR would be overestimated by 27 percent. However, if the false negative rate found in the follow-up study40 can be generalized to the studies that did not identify false negatives,34,37,39,41 the inflation may be approximately 6 percent.
In summary, estimates of accuracy are based on a handful of mass screening, cross-sectional studies. All studies suffer from work-up bias. However, one study40 attempted to reduce this bias by following all patients with negative tests to determine how many patients with skin cancer were missed. Also, all studies except one34 provide accuracy measures for dermatologists in mass screening settings but not for primary care physicians. Accuracy for dermatologists
screening patients is thought to be high, with a sensitivity of 0.94 and a specificity of 0.975.40
Non-Screening Studies
Several studies have examined the accuracy of nondermatologists’ assessments of photographs of skin lesions or of preselected patients with lesions, using the histologic diagnosis as the reference standard. One recent review summarized studies that used color slides (rather than actual patients) to test physicians’ accuracy in predicting the histologic diagnosis (mostly NMSC).42 When these studies were combined, dermatologists performed better (93 percent correct) than family medicine attending physicians (70 percent correct) and internal medicine attending physicians (52 percent correct). Another recent review found that in studies that used photographs or selected patients with known lesions, use of the ABCD(E) or seven-point checklist had a sensitivity of 50–97 percent and a specificity of 96–99 percent for the histologic diagnosis of skin cancer.20 Nondermatologists’ examinations were less sensitive than examinations performed by dermatologists. Many of these studies were small and used convenience samples of attending physicians at academic medical centers. More importantly, these studies did not examine the accuracy of a total-body skin examination or the ability of physicians to efficiently identify suspicious lesions in the setting of a screening program.
One well-designed prospective study of the accuracy of total-body skin examination found that skin cancer specialists’ decisions about biopsy were more sensitive and much more specific than those of general practitioners (GPs).43 Four skin cancer specialists and 63 randomly selected general practitioners in Australia performed total-body skin examinations on 109 selected patients, 43 of whom had suspicious pigmented lesions diagnosed previously by a skin specialist. The sensitivity of total-body skin examination for detecting suspicious lesions was 0.72 for the GPs, versus 0.97 for the four skin cancer specialists. The positive predictive value for the GPs was 0.39. Of the 43 patients with suspicious lesions, 12 (28 percent) had melanomas. While the GPs’ diagnoses were highly sensitive for melanomas (0.97), they classified about 11 benign lesions as suspicious for each melanoma. For the four dermatologists, the ratio was 2.1 benign lesions to 1 melanoma. Because the proportion of patients who had suspicious lesions (and melanoma) was much higher in this study than would occur in unselected patients, the positive predictive value of primary care physicians’ examinations would be lower in an actual screening study.
How accurate are risk-assessment tools as a screening test for skin cancer? (Arrow 1a, Q2)
Another screening strategy is to use a questionnaire or interview to identify a group of high-risk patients. In this strategy, only high-risk patients would have total-body skin examinations. Potentially, such a strategy could reduce the cost of screening because fewer total-body skin examinations and biopsies would be required to diagnose patients with skin cancer. The association of demographic, behavioral, and clinical factors with the risk of skin cancer has been well studied, and studies have established that physicians and patients can reliably measure some of these factors. As discussed below, however, the validity of formal risk assessment tools to screen unselected patients in primary care has not been established.
Risk Factors for Nonmelanoma Skin Cancer
A past history of skin cancer, or of actinic keratoses (AK), and white race are the strongest risk factors for nonmelanoma skin cancer. Among whites who have no prior history of skin cancer or AK, sun exposure is the most important risk factor for nonmelanoma skin cancer. Cumulative sun exposure and possibly intermittent intense sun exposure,44 total time spent outdoors,45 geographic area of residence,46 and lifetime number of severe sunburns46 have all been shown to be associated with higher risk of NMSC. Other risk factors include past history of NMSC,47 light or red hair,45,46 propensity to sunburn,46,47 and family history of skin cancer.
Risk Factors for Melanoma
A high count of common moles larger than 2 mm and the presence of atypical moles are risk factors for melanoma.48–51 In these studies, the risk of malignant melanoma was at least three times as high in patients who had 50–99 common moles as in patients with fewer than 10 moles. Similarly, the likelihood of melanoma increased several times (odds rate [OR] ranged 1.6–7.3) for patients with one to four atypical moles compared to patients with no atypical moles.
Other risk factors for melanoma are red or light hair (OR ranged 1.4–3.5); a few (OR 1.9) or many actinic lentigines (OR 3.5); very heavy sun exposure (OR 2.63); reported growth of a mole (OR 2.3); skin that does not tan easily (OR 1.98); a family history of melanoma (OR 1.81); light eye color (OR ranged 1.55–1.60); and light skin color, types 1 or 2 (OR ranged 1.40–1.42).48–50,52,53 The validity of some risk factors, such as hair color and sun exposure, is lower in the elderly.48,54
The relation of sun exposure to melanoma is complex. Since the second edition of the U.S. Preventive Services Task Force’s Guide to Clinical Preventive Services was published in 1996, a meta-analysis of case-control studies found that “intermittent” sun exposure was associated with increased risk of
melanoma, while heavy occupational exposure was found to be slightly protective.55 This meta-analysis included 23 case-control studies on “intermittent” sun exposure, 20 occupational sun exposure case-control studies, and 21 case-control studies on sunburn, all completed by 1992. Intermittent exposure was defined as recreational and vacation sun exposure. Patients with the highest level of intermittent exposure had nearly twice the risk of melanoma as those with the lowest “intermittent” exposure (OR 1.71; 95 percent confidence interval [CI] 1.54–1.90). Patients with heavy occupational exposure were at slightly decreased risk compared to those with low occupational exposure (OR 0.86; 95 percent CI 0.77–0.96). Patients with histories of sunburn (usually a result of intermittent sun exposure) were at increased risk for melanoma (OR 1.91) compared to those without sunburn history. Since the meta-analysis was published, an analysis of body site distribution among incident melanoma cases in British Columbia found that intermittent sun exposure was a risk factor for melanoma in individuals less than 50 years of age.56 A recent case-control study also supported the hypothesis that intermittent ultraviolet exposure is a risk factor for melanoma in younger individuals who are susceptible to burning.57
Reliability of Measures of Risk
To be useful as practical tools for classifying patients into risk groups, risk factors must be reliably assessed by patients or physicians. As noted above, the two strongest risk factors for melanoma are the presence of atypical moles and a large number of common moles.
Several studies have examined the reliability of mole counts by patients, interviewers, and dermatologists. In these studies, having a trained interviewer or the patient count the moles on the arm was not useful as an indicator of total-body mole count,58–60 but patients’ counts of moles on the trunk or total body were more reliable.59–61 In a large, population-based prospective study, 670 Swedish women completed a melanoma risk-assessment questionnaire twice (one to years apart). The test-retest reliability of a mole count by the patient was high (kappa=.52–.83).59 In a work site screening study, 104 of 125 employees correctly placed themselves in high- or low-risk melanoma categories based on count of total-body moles larger than 5 mm.61 Of 104 lower-risk patients (with fewer than six large moles), 92 correctly assessed themselves when compared to a dermatology fellow’s assessment. However, only 12 of 21 high-risk patients (more than six large moles) assessed themselves correctly. This suggests that patients are able to screen themselves for large-mole-count risk with specificity of 0.88 and sensitivity of 0.57. Women tended to overcount moles and men tended to undercount them.
There is less information on the reliability of self-report of other risk factors. In the Swedish study described above,59 response agreement was good for
questions related to hair color (kappa=.77) and freckles (kappa=.83), but only fair for the number of raised nevi on the left arm (none, 1–3 or >3) (kappa=.40) and sunburn history (kappa=.54). In another study, the agreement between patients’ self-appraisal of skin characteristics and clinical skin examinations by a physician was reflected in kappa values of .67 for freckles and .43 for atypical nevi.62
Use of Risk Assessment in Practice
The ideal study to measure the accuracy of risk assessment tools would assign risk levels for patients in a primary care setting, perform total-body skin examinations on patients classified as high and low risk, and then monitor the patients regularly to determine what proportion of incident melanomas occurred in the high-risk group. In fact, no longitudinal studies of the use of a risk assessment tool in primary care practice have been reported.
Although it was not done in a primary care setting, a large, prospective study validated the use of an initial count of atypical moles in predicting the incidence of melanoma over five years.63 In that study, 3,889 employees a the Lawrence Livermore National Laboratory had total-body skin examinations performed by a dermatology fellow specializing in melanoma. Atypical moles were diagnosed clinically using previously defined criteria:64 “ill-defined border; irregular border; irregularly distributed pigmentation; a diameter more than 5 mm; erythema (blanchable in lesion or at edge); and accentuated skin markings.” Seven percent of the subjects were in the highest-risk category—that is, had few to many moles that met five or more of these criteria. This highest-risk group accounted for 56 percent (five of nine) of the subjects who developed melanoma over five years. By contrast, 64 percent of the patients were in the lowest-risk category—had no atypical moles. This lowest-risk group accounted for 11 percent (one out of nine) of the patients who developed melanoma.
Two recent cross-sectional studies have examined the reliability and practicality of classifying primary care patients into risk groups using a standardized, self-administered instrument.60,62 One of these62,65 applied a melanoma risk assessment questionnaire66 in 16 randomly selected group practices in Cheshire, United Kingdom. Patients were asked about freckling propensity, number of moles, existence of large moles with irregular borders or colors, and history of sunburn. Their responses were compared with the results of a physical examination by a physician.
Although this study did not track the incidence of melanoma over time, it did provide data on the proportion of primary care patients who would be classified as high risk. A total of 3,105 patients completed the questionnaire. According to their responses, patients were then placed into the following risk groups: “marginally increased risk” (49.2 percent); “increased risk” (26.6 percent);
“very increased risk” (4.4 percent); and “worrying high risk” (4.3 percent). Most patients in the two highest-risk groups were not aware of their high-risk status.
In summary, several recent case-control studies confirm earlier evidence that patients with atypical moles and/or many (>50) common moles are at increased risk for melanoma. Evidence suggests that patients can count the number of moles 5 mm or larger in reasonable agreement with physicians. No prospective evidence is available linking risk assessment by limited physical examination with incidence of melanoma, but one well-done prospective study demonstrated that this strategy could identify a relatively small (<10 percent) group of primary care patients for more thorough evaluation.
Consequences of Screening
We examined the consequences of screening described in 24 recent reports of screening programs (Table B-2).34,35,37,39–41,63,67–85 In these studies, we examined (1) how often skin cancer is suspected in individuals who are screened; (2) how often melanoma and nonmelanoma skin cancers are diagnosed; (3) how often referrals for follow-up and biopsies are performed; and (4) how the type of skin examination, examiner, and compliance affect the yield of screening. This information is summarized in Table B-2. We also examined the distribution of thickness and stage of lesions found through screening versus usual care (Table B-3) and the adverse effects of screening.
There are limitations in using data from these studies to draw conclusions about screening in a case-finding setting. The majority of recently published studies were in a mass-screening setting. To be effective in reducing morbidity and mortality, screening should reach those who are at high risk both for developing skin cancer and for presenting with thicker lesions in the absence of screening. People who attend mass screening are a self-selected group, tend to be more skin aware, and may come because they are worried about a lesion that they have already discovered and for which they would have sought medical attention anyway. While some mass screening programs tend to attract a relatively high-risk group,68,69 others do not, and very high-risk individuals, particularly ill elderly individuals, may be underrepresented.86,87
How often does screening detect suspicious lesions? (Arrow 1b, Q1)
Rates of suspected melanoma in mass screening, case finding, and population-based screening ranged from 0 to 8 per 100 people screened, with the most common findings between 1 and 3 per 100. Most studies found from 2 to 10 suspected nonmelanoma skin cancers per 100 screened. In some populations, the rate of suspected basal or squamous cell carcinoma was much lower (4 per 10,000 in a Japanese study67) or higher (21 per 100 screened in surfers).73 Studies did not report the prevalence of nonmelanoma skin cancer by age, and
TABLE B-2 Studies of Screening for Skin Cancer
|
Author, Year |
Population/Setting/ Recruitment |
Screening Test, Examiner |
Media Target; Reported Risk Factors |
Referral Procedure (pt’s PCP, pt’s dermatologist, study PCP/derm, reminder, no reminder) |
Referral (p) |
|
Mass Screening |
|
||||
|
Jonna, 1998 |
464 volunteers, hospital, US; 72% <65, 94% white, 43% male |
TSE, dermatologists |
Targeted high risk |
Pt’s PCP or dermatologist, no reminder |
|
|
Limpert, 1995 |
247 volunteers, PCP clinic, US; mean age 53.5 (range 4–84), 100% white, 38% male |
TSE, family physician |
Not targeted |
Pt’s PCP, pt’s dermatologist, or study PCP, reminder |
|
|
Rampen, 1995 |
1,961 volunteers, hospital, Netherlands |
TSE or partial skin exam, dermatologists |
Targeted high risk |
Not specified |
0.1 |
|
McGee, 1994 |
279 volunteers, New Zealand; 41% 40–59 y/o, 29% ≥60 y/o |
Not specified, general medical practitioners |
42% came because of a “worrying mark,” 25% fair skin, 53% h/o severe sunburn, 22% fair or red hair, 8% personal h/o sc, 20% family h/o sc |
Pt’s PCP, no reminder |
0.2 |
|
Koh, 1996 |
282,555 volunteers, hospitals, US |
Not specified (need Koh, 90) |
Not targeted |
Pt’s PCP or dermatologist, no reminder |
|
|
de Rooij, 1995 |
2,463 volunteers, hospital, Netherlands; 53% >50 y/o |
Lesion specific TSE |
Not targeted |
Pt’s PCP, no reminder |
|
|
de Rooij, 1997 |
4,146 volunteers, hospital, Netherlands, 34% >50 y/o |
Lesion specific TSE |
Targeted high risk |
Pt’s PCP, no reminder |
0.12 |
|
Katris, 1996 |
3,379 volunteers, hospital and community, Australia; 35% ≥50 y/o, 16% ≥60 y/o |
TSE |
Targeted high risk |
Not specified |
|
|
Katris, 1998 |
256 volunteers, hospital and community, Australia |
TSE, nurses, and plastic surgeons |
Target high risk |
Not specified |
|
|
Rivers, 1995 |
1,681 volunteers, community, Canada; 16% ≥65 y/o |
TSE partial skin exam lesion specific, dermatologists |
Beachgoers; 33% had 2 or more risk factors (blond or red hair, blue or green eyes, propensity to sunburn) |
Pt’s PCP |
|
|
Results (p) |
||||||||
|
Suspected MM |
Confirmed MM |
Confirmed Melanoma in Situ |
Suspected BCC/SCC |
Suspected AK |
Negative Screen |
Biopsy |
MM Biopsy Suspected MM only |
MM Biopsy all Suspicious Lesions |
|
0.08 |
0.002 |
0.002 |
0.21 |
|
0.42 |
0.21 |
0.15 |
0.04 |
|
|
0.004 |
|
|
|
0.57 |
0.18 |
|
0.02 |
|
0.003 |
0.003 |
0.001 |
0.02 |
|
0.9 |
|
||
|
0.03 |
|
|
0.08 |
|
||||
|
0.02 |
0.001 |
0.001 |
|
|
|
|
0.11 |
|
|
0.01 |
0.003 |
0.001 |
0.04 |
0.06 |
|
|||
|
0.02 |
0.001 |
0.002 |
0.02 |
0.02 |
|
0.08 |
0.17 |
0.03 |
|
0.02 |
0.08 |
0.038 |
0.13 |
|
0.83 |
|
0.08 |
|
|
0.05 |
|
|
Surgeon: .10 Nurse: .07 |
Nurse: .03 |
Surgeon: .70 Nurse: .60 |
|
||
|
0.005 |
|
|||||||
|
Author, Year |
Population/Setting/ Recruitment |
Screening Test, Examiner |
Media Target; Reported Risk Factors |
Referral Procedure (pt’s PCP, pt’s dermatologist, study PCP/derm, reminder, no reminder) |
Referral (p) |
|
Dozier, 1997 |
1) 49 volunteers, community, US; mean age 29.7 2) 53 volunteers, hospital, US; mean age 35.4 |
1) partial skin exam 2) TSE dermatologists |
1) surfers 2) not targeted |
1) Pt’s PCP, reminder 2) Pt’s PCP, no reminder |
|
|
Population Based Screening |
|||||
|
Harvey, 1995, 1996 |
560 random population sample, community, UK; 100% ≥60 y/o |
Partial skin exam, dermatologists |
NA |
Pt’s PCP, reminder |
0.02 |
|
Ichihashi, 1995 |
4,736 consecutive attendees at regional health exam, Japan |
Partial skin exam, dermatologists |
NA |
Study dermatologist, no reminder |
|
|
Tornberg, 1996 |
1,654 random sample, hospital, Sweden; 100% 40–54 y/o |
Not specified, nurses, dermatologist, oncologist |
NA |
Study dermatologist, no reminder |
0.05 |
|
Bergenmar, 1997 |
501 random sample, hospital, Sweden; 100% 40–54 y/o |
NA |
NA |
|
|
|
Casefinding |
|||||
|
Ruskiewicz 1998 |
1,000 consecutive patients, optometrist office, US; mean age 66.3 (range 35–96) |
Partial skin exam, optometrist |
NA; .096 had prior dx of SC or AK |
Dermatologist |
|
|
Whited, 1997 |
190 consecutive patients, pcp and specialty clinic, US |
Partial skin exam, dermatologists, internists, physician assistants |
NA |
|
|
|
Worksite Screening |
|||||
|
Friedman, 1995 |
421 hospital employees, identified as high risk and invited, US; mean age 41 (SD 10.6) |
Not specified, dermatologists |
Targeted high risk |
Pt’s dermatologist or study dermatologist |
0.32 |
|
Schneider |
3,889 laboratory employees, 9% of 20–24 y/o employees, 56% of 70 y/o and older employees |
Not specified, dermatologists |
NA; participants classified according to no. of atypical moles: 64% none, 29% possible or probable, 7% clear pattern of marked atypical moles |
Study dermatologist |
|
|
Results (p) |
||||||||
|
Suspected MM |
Confirmed MM |
Confirmed Melanoma in Situ |
Suspected BCC/SCC |
Suspected AK |
Negative Screen |
Biopsy |
MM Biopsy Suspected MM only |
MM Biopsy all Suspicious Lesions |
|
1) 0.0 2) 0.0 |
|
|
1) .16 2) .02 |
1) .41 2) .15 |
|
|||
|
0.004 |
|
|
0.02 |
0.23 |
0.75 |
|
||
|
0 |
|
|
0.0004 |
0.01 |
|
|||
|
0.09 |
0 |
0 |
0 |
0 |
0.91 |
0.04 |
0 |
|
|
0 |
|
|
0.1 |
0.003 |
0.9 |
|
|
|
|
|
0.31 |
|
0 |
|||||
|
0.002 |
|
|
0.02 |
0.09 |
|
|||
|
|
0.002 |
|
||||||
TABLE B-3 Thickness of Malignant Melanoma Lesions Found in Screening Studies
|
N (p) |
||||||||
|
Study |
0.5–1 mm |
1.0–1.5 mm |
1.5–2 mm |
2–2.5 mm |
2.5–3 mm |
3–3.5 mm |
3.5–4 mm |
>4 mm |
|
De Rooij |
12 (.92) |
|
1 (.08) |
|
||||
|
Herd |
82 (.76) |
26 (.24) |
||||||
|
Katris |
4 (1.0) |
0 (0) |
||||||
|
Jonna |
1 (1.0) |
0 (0) |
||||||
|
Schneider |
9 (1.0) |
0 (0) |
||||||
|
Koh |
180 (.87) |
22 (.11) |
|
4 (.02) |
||||
|
Marghoob |
10 (1.0) |
0 (0) |
|
|||||
|
Van der Spek |
1,451 (.67) |
506 (.23) |
|
|
206 (.10) |
|
||
we found no clear difference in prevalence between studies of mostly older individuals and studies in younger individuals.
Suspected actinic keratosis was generally the most frequent finding in these studies, but rates were variable. The highest rates were 41 per 100 screened in the surfers,73 23 per 100 in a population sample of people over age 59,73–75 and 15 per 100 in a small mass-screening study.73
How often does screening result in a diagnosis of cancer? (Arrow 1b, Q2 and Q3)
Rates of confirmed melanoma and melanoma in situ were consistently in the range of 1 to 4 per 1,000 people screened, with two exceptions. An Australian study that targeted high-risk people70 had a rate of eight confirmed melanomas per 100 people screened. The other, a population-based study in Sweden,77 had no confirmed melanomas of 152 suspected melanomas in 1,654 people screened.
Eight studies in Table B-2 reported the number of histologically confirmed nonmelanoma skin cancers. The prevalence varied widely, from 0.05 of people screened to 0.0004, with most reporting between 0.01 and 0.05.
How often does screening lead to referral for follow-up and biopsy? (Arrow 1b, Q4)
Among all the studies that we reviewed, rates of referral for follow-up care of suspicious lesions ranged from 2 to 34 per 100 people screened. Two studies reported rates of compliance with a recommendation to see a physician for follow-up. In one mass-screening study,41 95 percent of people complied with recommended follow-up, and in a work site program,81 45 percent complied.
As for biopsies, five studies reported the number of biopsies performed as a result of screening.34,37,41,77,80 In these studies, from 4 to 31 biopsies per 100 people screened were performed. Among patients with suspected melanoma, from 0 to 17 percent had a final diagnosis of melanoma. Among all patients who underwent a biopsy, 0 to 0.04 proved to have a melanoma.
How do characteristics of the screening program affect the yield of screening? (Arrow 1b, Q5)
Certain characteristics of a screening program could affect compliance with follow-up recommendations and, ultimately, with the yield of screening. We consider three such characteristics below: (1) the type of skin examination, (2) the recruitment strategy, and (3) the procedure for referring patients for follow-up.
Type of Skin Examination
Examiners conducted either total-body skin examination,34,37,70,71,73,82,83 partial skin examination (e.g., only above the waist or on sun-exposed areas),73– 76,79,80 examination only of lesions the participant was worried about,35,41 or some combination of these (see Table B-2). Overall, compared with partial skin examination or examination of lesions the patient was worried about, the use of total-body skin examination did not appear to increase the rate of confirmed melanomas. One study88 specifically addressed the question, In self-selected patients who have noticed a skin lesion, does total-body skin examination increase the likelihood of finding skin cancers? In that study, 2,910 of 4,146 (70 percent) people screened complained of at least one skin lesion. When these lesions were examined, 13 melanomas and 44 nonmelanoma skin cancers were diagnosed on biopsy. For those patients who originally came in with specific lesions, an additional total-body skin examination was offered. For the 1,356 patients who went on for a total-body skin examination, no malignant melanomas and three basal cell carcinomas were identified. This finding raises doubts about the benefits of conducting total-body skin examinations on everyone rather than lesion-specific examinations.
Recruitment Strategy
In 12 papers, one of which reported on two studies, screening involved a media campaign to encourage individuals to seek a skin examination (mass screening) (see Table B-2); in seven of these programs, the media campaign targeted individuals with suspicious lesions on self-examination or risk factors for melanoma, while six were not targeted. We found no systematic relationship between these characteristics and the proportion of individuals eventually diagnosed with cancer. However, most studies did not report sufficient information to determine how well targeting succeeded in recruiting a high-risk population, and the descriptions of the study samples were not adequate to exclude differences in baseline risk factors between studies as the main reason for observed differences in results.
Procedure for Referring Patients for Follow-Up
In most studies the patient was instructed to see a primary care clinician or dermatologist for follow-up of suspicious lesions. In some studies, the patient’s physician was contacted directly, or the patient was sent to a study dermatologist. Some studies used reminders such as letters to the patient.
Although not depicted in Table B-2, we collected information on compliance rates for screening, follow-up, and biopsy when possible. Three population-based studies invited a target group of people to be screened and reported
response rates.36,74,75,77,89 In these studies, between 60 and 70 percent of those invited attended screening. One work site screening program that identified and invited high-risk people reported a lower response rate of 19 percent.80 The mean age of this sample was somewhat lower than that in the population-based studies.
Compared to usual care, how much earlier does screening detect skin cancers and precancerous lesions? (Arrow 1b, Q6)
Eight studies reported the thickness of melanoma lesions found through screening (Table B-3). In four studies,37,63,70,83 all detected melanoma lesions were 1.0 mm or thinner, and in a fifth,41 92 percent were 1.0 mm or thinner. In three other studies that used 1.5 mm as the cutoff, the proportion of melanomas 1.5 mm or less was 67–87 percent.39,82,84
No study of screening directly followed an unscreened population to compare the distribution of thickness or stage of melanomas detected. Nonetheless, the proportion of thin melanomas is clearly higher in screening programs than in usual care. In an analysis of SEER data from 1992 to 1994, 57 percent of melanomas were thinner than 0.76 mm, 23 percent were 0.76–1.5 mm, 15 percent were 1.51–3.99 mm, and 5 percent were 4.0 mm or thicker.39 The SEER registry routinely reports the TNM stage, but not the thickness, of melanomas at the time of diagnosis. From 1989 to 1994, 81 percent of melanomas detected through usual care were localized, 9 percent regional, 4 percent distant, and 6 percent unstaged. In population-based studies, moreover, the incidence of melanoma detected by screening is higher than base rates, and the increase is almost entirely attributable to thin melanomas.
What is the effect of screening on patients’ skin knowledge and self-care behavior? (Arrow 1c)?
Advocates of screening note that having a total-body skin examination could improve morbidity and mortality indirectly by promoting skin awareness and sun protection measures. In a follow-up study to the American Academy of Dermatology’s Melanoma/Skin Cancer Screening Programs (see reference to Koh, 1996, in Table B-1), 1,049 participants who had skin lesions were surveyed about their skin health behaviors two months after undergoing a total-body skin examination. Among the 643 respondents, the proportion of individuals who regularly checked their skin increased from 60 to 84 percent after screening.90
What are the adverse effects of screening? (Arrow 2)
Total-body skin examination is noninvasive. In one study of self-selected participants found by screening to have skin lesions, patient satisfaction was high (81 percent), and only a small proportion of patients reported embarrassment or discomfort as a result of screening (4.8 percent).90
False positive skin examination results might also be considered an adverse effect. In any screening program, most lesions referred for biopsy because of clinical suspicion of skin cancer are false positives. There are no studies by which to judge the extent of harm, if any, related to these tests.
Misdiagnosis is another potential adverse effect of screening. It is known to occur, but no studies have been done on its rate of occurrence. The diagnosis of melanoma has a serious emotional and financial impact, and even when the melanoma is very thin and has an excellent prognosis, obtaining insurance can be very difficult.7 Critics worry that if screening becomes widespread, pathologists may set the threshold low for diagnosing borderline lesions as melanoma, since the risk to the patient and the potential legal cost to the pathologists for missing melanoma are overwhelming.9 The effects on diagnostic criteria of widespread screening are hard to predict, but uncertainty about these effects should be considered in weighing a recommendation to screen.
Some experts consider diagnosis of common, nonmalignant skin lesions found incidentally in screening to be a costly adverse effect of screening. Screening detects large numbers of benign skin conditions, especially seborrheic keratoses, which are very common in the elderly. Detection of these lesions could be considered an “adverse effect” of screening if it leads to additional biopsies and unnecessary or expensive procedures. While this has been shown to occur in usual care,91 none of the studies of screening examined the rate at which this occurred.
Is there direct evidence that screening for skin cancers leads to reduced morbidity and mortality? (Arrow 3)
No randomized trials or case-control studies of screening for skin cancer have been completed. Well-done, frequently cited observational studies of the relationship between early detection and mortality have been done,92 but in such studies the effect of promoting primary prevention and self-examination cannot be distinguished from that of routine screening in patients seeing the physician for unrelated reasons.86 The lack of data reflects the lack of population-based programs that focus on routine total-body skin examination by a physician.
The absence of randomized trials is also not surprising since melanoma is relatively rare in the general population. A recent review by J.M.Elwood examined the options for conducting a randomized trial of screening in detail.31 Elwood calculated that to have a 90 percent chance of detecting a one-third reduction in mortality, a trial of screening with total-body skin examination in the general population aged 45–69 would require 400,000 subjects in each group.
Put differently, about 21,000 people would have to be screened to prevent one death.
An alternative would be to conduct a trial in patients classified as high risk by a risk assessment questionnaire. Using this approach, Elwood assumed that 7 percent of the population would be classified as high risk; 35 percent of all melanomas occur in this high-risk group; 60 percent of patients complete the questionnaire; and 80 percent of the high-risk patients would comply with total-body skin examination. He calculated that to have a 90 percent chance of detecting a one-third reduction in mortality, 6 million questionnaires would have to be administered to enroll 100,000 high-risk subjects in each group.
In fact, a trial involving 600,000 subjects has begun in Australia and is expected to require nine more years to complete.
Effectiveness of Early Treatment
Screening in a population is justified if there is evidence that early detection and treatment improve outcomes such as mortality and quality of life. Other issues to be considered include consequences of false negative and false positive tests, acceptability of the test, and the risks of screening and of treatment.
Does treatment of nonmelanoma skin cancer found by screening reduce morbidity and mortality? (Arrow 4a)
Early treatment of basal and squamous cell carcinoma might reduce morbidity and disfigurement, but no studies have evaluated whether screening improves the outcomes of these cancers. Basic information, such as the proportion of Medicare patients with nonmelanoma skin cancer who suffer disfigurement or death, is lacking. If we assume that 20 percent of squamous cell cancers in the elderly are either lethal or disfiguring, this assumes a prevalence of 0.025 for men and women combined and that screening would reduce this by half, about 400 patients would have to be screened to prevent one lethal or disfiguring case. These calculations, while theoretical, do suggest that screening is potentially beneficial and that a trial of early detection in the elderly should examine outcomes in nonmelanoma skin cancer rather than just melanoma.
Does treatment of melanoma found by screening reduce morbidity and mortality? (Arrow 4b)
Well-designed observational studies can provide persuasive information about the effect of early detection on mortality. For some cancers, notably colon cancer, observational studies make a convincing case for the effectiveness of early detection, even in the absence of randomized controlled trials. Such a con-
clusion must be based on data that link actions taken as a result of screening to health outcomes.
In the absence of randomized trials and case-control studies of screening or of early treatment, the inference that earlier treatment as a result of screening improves health outcomes must rely on three lines of indirect evidence: (1) a case-control study in which skin self-examination reduces the incidence of lethal melanoma; (2) comparison of the stages of cancers and mortality found in screening to those found in usual practice; and (3) evidence from studies of the consequences of delay in diagnosis. These are summarized below.
Case-Control Study of Self-Examination
While there are no case-control studies of screening, one case-control study has examined the effect of skin self-examination on mortality from melanoma.49 In this study, 650 incident cases of melanoma in 1987–1989 were identified through the Connecticut Tumor Registry and compared with randomly selected, age- and sex-matched controls. After five years of follow-up, cases were classified as “lethal” if the individual died or had distant metastases.
A structured questionnaire was used to assess skin self-examination attitudes and behavior. The definition of skin self-examination used in this study was, “did you ever (in your life) carefully examine your own skin? By this I mean actually check surfaces of your skin deliberately and purposely?” Based on their responses to this and related questions, 13 percent of the cases and 17.5 percent of control subjects were classified as careful or rigorous examiners, and an additional 57.4 percent of cases and 66.7 percent of controls were classified as casual examiners. The questionnaire also assessed potential confounding factors, such as risk factors for skin cancer, but did not assess general health behaviors such as diet, exercise, and medical care-seeking behavior that might affect the risk of cancer and the likelihood of early detection.
The investigators performed two multivariate analyses: one for primary prevention and one for secondary prevention. In the first analysis, after adjustment for sun exposure, skin color, the number of nevi, and other risk factors, skin self-examination was negatively associated with incidence of melanoma (OR 0.66; CI 0.44–0.99).
In the second analysis, after adjustment for confounding risk factors, skin self-examination was associated with a reduced risk of lethal melanoma (OR 0.37; CI 0.16–0.84). Survival analysis comparing patients who practiced skin self-examination with those who did not suggested that after an average of 5.4 years, self-examination was associated with a lower probability of lethal melanoma. The authors noted that the shape of the survival curves—the curve for the self-examination group plateaus after three years, while survival continues to decrease up to five years in the patients who do not practice self-examination—ooffers some reassurance that the observed benefit is due to actual improvement
offers some reassurance that the observed benefit is due to actual improvement in survival rather than to lead-time bias.
As noted by the authors, this case-control study provides suggestive, rather than definitive, evidence for the effectiveness of skin self-examination. More direct evidence is needed to link self-examination behaviors to specific actions that could reduce the incidence or lethality of melanoma. To prevent melanoma, self-examination on the part of the patient must lead to actions—such as identification of a suspicious lesion, self-referral to a physician, earlier treatment of precancerous lesions, and health behavior changes—to prevent the development of new melanomas. While the study indicates that patients who practiced self-examination had undergone more biopsies than those who had not, it does not report the frequency of these intermediate steps or whether their frequency was different enough from that of other patients to explain the observed differences in outcome.
Apart from concerns about the strength of the study design, how relevant is a study of skin self-examination to screening by primary care providers? If skin self-examination prevents death from melanoma, it may be more likely that examination by a physician could also prevent deaths, especially if examination by a physician promotes more accurate self-examination. In fact, case finding by a physician might be expected to be more effective because it reaches patients, especially elderly men, who are at high risk and are the least likely to practice self-examination effectively87 or to respond to an invitation or health promotion campaign. However, self-examination occurs much more frequently (monthly, on average, in the case-control study) than screening by a physician and can note findings—in particular, changes in size, border, or color of lesions—that cannot be recognized easily by infrequent examinations. Nevertheless, this case-control study provides the strongest available evidence that early detection of melanoma reduces mortality.
Comparing Stages of Cancers and Mortality Found in Screening to Those Found in Usual Practice
Advocates cite the results of public information campaigns in Australia and the United Kingdom as evidence of the potential benefits of screening. In Australia, public information campaigns have promoted sun protection behaviors and early detection for more than 15 years. Melanoma mortality, which had increased for decades, reached a plateau in 1985 and, in recent years, has fallen slightly.93 It is thought that this trend is related to skin health promotion activities, including primary prevention and self-examination, but because it is not a prominent feature of these campaigns, it is not possible to determine what role, if any, screening by physicians has played.
In the United Kingdom, registry data were used to compare rates of invasive melanoma before and after public information campaigns to promote early detection of skin cancers. In the West of Scotland, a community-based skin health promotion campaign compared melanoma thickness and mortality before and after implementing a public information campaign and rapid referral system in 1985.92 The number and proportion of thin melanomas diagnosed yearly in the population increased immediately afterward. The response to the public information campaign was stronger in women than in men. In women, within 2 years, the rate of diagnosis of thick melanomas (>3.5 mm) began to decrease and, within 5 years, sustained decreases in thick melanomas and in melanoma mortality were observed. In men, the rate of diagnosis of thick melanomas did not change and the melanoma mortality rate rose. As in Australia, the role of screening by physicians in these results is not clear.
A subsequent implementation of a similar program in seven British districts failed to replicate these results.82,94,95 The incidence rates of both thin and thick melanomas increased during the public information campaign (1987–1989) and have remained higher than before the program began.
In contrast to these health promotion efforts, mass-screening programs cannot be evaluated using population-based registries. Mass screening increases the proportion of melanomas detected in an “early” stage (see Table B-3), but the significance of this finding is unclear. Survival is strongly related to lesion thickness at the time of resection, but it is difficult to know the extent to which comparison of the distribution of the stage of cancers found by screening to those found in usual care is affected by lead-time bias or length bias. The natural history of melanoma, in particular the significance of the many additional thin melanomas found in screened populations, is another source of uncertainty.
Retrospective Studies of the Consequences of Delay in Diagnosis
The argument for screening would be strengthened if evidence pointed to a consistent relationship between delay of diagnosis and the thickness of melanoma. Nine case series examined the causes and consequences of apparent delay in the diagnosis of melanoma. The two largest studies, one from Scotland and one from Australia, found no relation between delay in diagnosis and tumor thickness.18,82 The Australian study found that male sex, nodular melanoma, and location on the head and neck (but not delay) were associated with thick melanoma.
Five studies, which were performed in specialty clinics, observed patients with melanoma of the hand, foot, eye, penis, or nailbeds.96–100 In these studies, misdiagnosis was a common cause of delay in treatment. Effects of delay on tumor thickness or survival were reported in three of the studies, and the results were inconsistent. In a study of 83 patients with acral melanomas, 17 of 33 subungual melanomas and 10 of 50 palmoplantar melanomas were clinically misdi-
agnosed by physicians.96 Misdiagnosis caused a median delay of 12 months in the diagnosis of palmoplantar melanomas and 18 months in the diagnosis of subungual melanomas. Delay in diagnosis was associated with increased tumor thickness, more advanced stage at time of melanoma diagnosis, and a lower estimated five-year survival rate (15.4 percent versus 68.9 percent for palmoplantar; 68.5 percent versus 90.9 percent for subungual). In another series of 140 patients with melanoma of the foot, delay in diagnosis had no effect on clinical outcome.100 Another series of 102 consecutive melanoma patients found no relationship between delay in diagnosis and tumor thickness.101
Two recent case series from specialized clinics in major referral centers reported that lesions detected by physicians were thinner than those detected by patients.102,103 In one of these, 24 of 102 consecutive patients had physician-detected melanomas; the median thickness was 0.23 mm versus 0.9 mm in self-detected melanomas. Eleven of the 24 physician-detected melanomas were in situ. In the other study, 172 of 590 consecutive patients had physician-detected melanomas; these were significantly thinner, but the difference was not as striking (0.9 mm versus 1.3 mm).
The latter study103 also carefully examined the relationship between melanoma thickness and delay, either to seek medical advice or after seeking medical care, and concluded that poor prognosis was due to rapidly growing tumors rather than delays. They examined four intervals: (1) the time patients first noticed a lesion and the time they considered it to be suspicious; (2) from then until they saw a physician; (3) from then until the physician proposed removal; and (4) from then until surgical resection. In the 418 patients who had self-detected cancers, the first, third, and fourth intervals were not associated with tumor thickness. Not surprisingly, once a lesion was recognized as being dangerous, patients with thicker lesions sought medical attention more quickly (i.e., for the second interval, thinner lesions had the longest delay). In a subgroup of 247 patients who reported a delay of less than 5 years between the time they noticed the lesion and the time they believed it to be dangerous, the time between noticing a lesion and considering it dangerous was longest for lesions 1.5–2.99 mm thick, but was shorter for lesions thicker and thinner than this.
Cost-Effectiveness Studies of Screening for Skin Cancer
A cost-effectiveness analysis of screening for malignant melanoma is currently in press.104 The analysis found that the average projected discounted life expectancy was 15.0963 without screening versus 15.0975 with screening. This difference is equivalent to an increase of about nine hours per person screened or 337 days for each person with melanoma.
If a screening examination by a dermatologist is assumed to cost $30, the incremental cost-effectiveness (CE) ratio was $29,170 per year of life saved. The CE ratio was unexpectedly low because, in the model, savings from pre-
vention of late-stage melanomas offset most of the costs of screening. Thus, the key assumptions in the model, affecting the calculation of both effectiveness and cost, were that the proportion of late-stage melanomas would decrease from 6.1 percent without screening to 1.1 percent with screening. Similarly, the model assumed that invasive cancers would decrease from 70.3 to 58.1 percent and melanomas thicker than 1.5 mm would decrease from 20.1 to 12.6 percent of invasive melanomas. These assumptions are based on comparison of cross-sectional data on the stages of melanoma in individuals who attended the American Academy of Dermatology’s mass screening programs with data on usual care from the SEER registry.
SUMMARY
Table B-4 (see page 210) summarizes the literature review by describing the evidence for each link in the analytic framework (see Figures B-3 and B-4). The quality of the evidence at each link ranged from poor to fair and is explained in Table B-4.
The case for screening is based on the assumption that melanoma and other skin cancers have a long latency period during which they can be treated with a high rate of success. Another assumption is that early detection prevents progression of early-stage cancers to advanced, lethal stages. These assumptions are reasonable, but studies of early detection have not focused on screening and have not adequately linked it with reduced incidence of invasive disease.
Despite these information gaps, skin cancer screening, perhaps using a risk assessment technique to identify high-risk patients who are seeing the physician for other reasons, is the most promising strategy for addressing the excess burden of disease in older individuals. This group has substantial morbidity and mortality from skin cancer. By themselves, primary prevention efforts and promotion of self-examination seem unlikely to change these rates substantially. While the efficacy of screening has not been established, the screening procedures themselves are noninvasive, and the follow-up test, skin biopsy, has low morbidity.
In this situation, indirect evidence of efficacy might be sufficient to decide that the potential benefits of screening outweigh the potential harms. To better estimate the benefit of earlier detection, we need to know how much earlier, compared to usual care, screening will detect cancers that would eventually have become aggressive. Population-based studies using concurrent comparisons could provide better information about the consequences of delay and how much they can be reduced by screening. No studies of screening have measured these parameters.
Gaps in our knowledge of the progression to thick melanoma in the elderly should also be addressed. Better information about the natural history of thin melanomas and about the history and patterns of growth of melanoma in the
TABLE B-4 Summary of Evidence for Screening for Skin Cancer
|
Linkage in Analytic Framework |
Evidence Code |
Quality of Evidence |
|
1a. Accuracy of total body skin examination: evidence that total body skin examination can detect skin cancer. |
II-2 |
Fair: The accuracy of a total body skin examination by primary care physicians in unselected patients may be low. Reliability of pathologic diagnosis in community practice in the U.S. is not clear. |
|
1b. Accuracy of risk-assessment: evidence that a questionnaire or interview, followed by exam in selected patients, can detect skin cancer. |
II-2 |
Fair: Mole counts and other factors predict elevated risk over time, but no study has determined the accuracy of risk stratification followed by total body skin examination in selected patients as a screening method. |
|
2. Adverse effects of screening: evidence that screening causes significant harms. |
III |
Poor: most postulated adverse effects have not been evaluated in studies. |
|
3. Effectiveness of early detection: evidence that persons detected through screening have better outcomes than those who are not screened. |
II-3 |
Poor: There are no studies that directly link screening to lower mortality and morbidity. Most well-done population-based studies concern promotion of self-care behaviors such as self-examination rather than universal screening. |
|
4a. Effectiveness of treatment of nonmelanoma skin cancer found by screening. |
III |
Poor: The hypothesis that early detection by screening could reduce mortality and morbidity is plausible but has not been examined in studies. |
|
4b. Effectiveness of treatment of melanoma found by screening. |
II-1, III |
Fair: There are no controlled studies of treatment in patients found by screening to have thin melanomas, but epidemiologic studies, studies of skin health behaviors, and studies of factors associated with advanced melanoma suggest that elderly men are at high risk and are unlikely to benefit from health promotion efforts. |
|
|
|
Studies of delay in diagnosis have conflicting results, and the ability of screening to reach individuals at high risk and to find aggressive tumors while they are still curable have not been established. |
|
*I=randomized controlled trial; II-1=controlled trial without randomization; II-2=cohort or case-control analytic studies; II-3=multiple time series, dramatic uncontrolled experiments; II-1=controlled trial without randomization; II-2=cohort or case-control analytic studies; II-3=multiple time series, dramatic uncontrolled experiments; and III opinions of respected authorities, descriptive epidemiology. |
||
ADDENDUM B-1 STRATEGY FOR SKIN CANCER SEARCH
-
Skin neoplasms
-
Exp mass screening
-
genetic screening
-
mass chest x-ray
-
multiphasic screening
-
vision screening
-
mandatory screening
-
-
Screen$.tw. (Text word taken from title and abstract of article)
-
Exp physical examination
-
self-examination
-
skinfold thickness
-
-
Exp neoplasms metastasis
-
lymphatic metastasis
-
neoplasm circulating cells
-
neoplasm seeding
-
neoplasms, unknown primary
-
-
Neoplasm recurrence, local
-
Recurrence
-
Exp morbidity
-
incidence
-
prevalence
-
-
Exp sensitivity and specificity
-
predictive value of tests
-
ROC curve
-
-
2 or 3 or 4 or 5 or 6 or 7 or 8 or 9
-
Skin neoplasms/mo (mortality)
-
Skin neoplasms/ep (epidemiology)
-
10 or 11 or 12
-
1 and 13
-
Limit 14 to human
-
Limit 15 to english language
-
Looked at english abstracts for foreign language articles
REFERENCES
1. Landis SH, Murray T, Bolden S, Wingo PA. Cancer statistics, 1999. CA Cancer Jr Clin. 1999; 49:8.
2. Ries LAG, Kosary CL, Hankey BF, Miller BA, Edwards BK. SEER Cancer Statistics Review, 1973–95. Bethesda, MD: National Cancer Institute, 1998.
3. Jelfs P, Coates M, Giles G. Cancer in Australia 1989–1990 (with projections to 1995). Canberra, Australian Institute of Health and Welfare; 1996.
4. Kelly J. Melanoma in the elderly—A neglected public health challenge. Med J Aust 1998; 169:403–404.
5. Dennis LK. Analysis of the melanoma epidemic, both apparent and real: data from the 1973 through 1994 surveillance, epidemiology, and end results program registry. Arch Dermatol, 1999; 135:275–280.
6. Berwick M, Halpern A. Melanoma epidemiology. Curr Opin Oncol. 1997; 9:178–182.
7. Swerlick RA, Chen S. The melanoma epidemic: More apparent than real? Mayo Clin Proc 1997; 72:559–564.
8. Burton R, Armstrong B. Recent incidence trends imply a nonmetastasizing form of invasive melanoma. Melanoma Res 1994; 4:107–113.
9. Swerlick RA, Chen S. The melanoma epidemic. Is increased surveillance the solution or the problem? Arch Dermatol 1996; 132:881–884.
10. Burton R. An analysis of a melanoma epidemic. Int J Cancer 1995; 55:765–770.
11. Gallagher RP, Hill GB, Bajdik CD, et al. Sunlight exposure, pigmentation factors, and risk of nonmelanocytic skin cancer. II. Squamous cell carcinoma. Arch Dermatol 1995; 131:164–169.
12. Green A, Battistutta D, Hart V, Leslie D, Weedon D. Skin cancer in a subtropical Australian population: Incidence and lack of association with occupation. The Nambour Study Group. Am J Epidemiol 1996; 144:1034–1040.
13. Kricker A, English DR, Randell PL, et al. Skin cancer in Geraldton, Western Australia: A survey of incidence and prevalence. Med J Aust 1990; 152:399–407.
14. Chuang TY, Popescu A, Su WP, Chute CG. Basal cell carcinoma. A population-based incidence study in Rochester, Minnesota. J Am Acad Dermatol 1990; 22:413–417.
15. Chuang TY, Popescu NA, Su WP, Chute CG. Squamous cell carcinoma. A population-based incidence study in Rochester, Minnesota. Arch Dermatol 1990; 126:185–188.
16. Karagas MR, Greenberg ER, Spencer SK, Stukel TA, Mott LA. Increase in incidence rates of basal cell and squamous cell skin cancer in New Hampshire, USA. New Hampshire Skin Cancer Study Group. Int J Cancer 1999; 81:555–559.
17. McGovern V, Cochran A, Van der Esch E. The classification of malignant melanoma, its histological reporting and registration: Revision of the 1972 Syndey classification. Pathology 1986; 18:12–21.
18. Hersey P, Sillar RW, Howe CG, et al. Factors related to the presentation of patients with thick primary melanomas. Med J Aust 1991; 154:583–587.
19. Heenan PJ, Yu L, English DR. Melanoma in the elderly—A neglected public health challenge [letter]. Med J Aust 1999; 170:394–395.
20. Whited JD, Grichnik JM. Does this patient have a mole or a melanoma? JAMA 1998; 279:696–701.
21. Corona R, Mele A, Amini M, et al. Interobserver variability on the histopathologic diagnosis of cutaneous melanoma and other pigmented skin lesions. J Clin Oncol 1996; 14:1218–1223.
22. Farmer ER, Gonin R, Hanna MP. Discordance in the histopathologic diagnosis of melanoma and melanocytic nevi between expert pathologists. Hum Pathol 1996; 27:528–531.
23. Gershenwald JE, Buzaid AC, Ross MI. Classification and staging of melanoma. [Review] [114 refs]. Hematology—Oncology Clinics of North Am 1998; 12:737–765.
24. Breslow A. Thickness, cross-sectional areas and depth of invasion in the prognosis of cutaneous melanoma. Ann of Surg 1970; 172:902–908.
25. Preston DS, Stern RS. Nonmelanoma cancers of the skin. N Engl J Med 1992; 327:1649–1662.
26. Stern RS, Lunder EJ. Risk of squamous cell carcinoma and methoxsalen (psoralen) and UV-A radiation (PUVA). A meta-analysis. Arch Dermatol 1998; 134:1582–1585.
27. Osterlind A, Hjalgrim H, Kulinsky B, Frentz G. Skin cancer as a cause of death in Denmark. Br J Dermatol 1991; 125:580–582.
28. Screening for Skin Cancer (Including Counseling to Prevent Skin Cancer). DiGuiseppi C, Atkins D, Woolf SH, Kamerow DB, (eds.) Guide to Clinical Preventive Services, 2nd edition. Baltimore: Williams & Wilkins 1996:pp. 141–152.
29. Rigel DS, Friedman RJ, Kopf AW, et al. Importance of complete cutaneous examination for the detection of malignant melanoma. J Am Acad Dermatol 1986; 14:857–860.
30. Elwood JM. Screening for melanoma. Miller AB, (ed.) Advances in Cancer Screening. Boston: Kluwer Academic Publishers, 1996:pp. 129–146.
31. Elwood JM. Screening for melanoma and options for its evaluation. J Med Screen 1994; 1:22–38.
32. Sackett DL, Haynes RB, Guyatte GH, Tugwell P. Clinical Epidemiology, A Basic Science for Clinical Medicine, 2nd Edition Boston: Little, Brown and Company, 1991.
33. Sox HC, Blatt MA, Higgins MC, Marton KI. Medical Decision Making. Boston: Butterworth-Heinemann 1988.
34. Limpert GH. Skin-cancer screening: A three-year experience that paid for itself. J Fam Pract 1995; 40:471–475.
35. de Rooij MJ, Rampen FH, Schouten LJ, Neumann HA. Skin cancer screening focusing on melanoma yields more selective attendance. Arch Dermatol 1995; 131:422–425.
36. de Rooij MJ, Rampen FH, Schouten LJ, Neumann HA. Factors influencing participation among melanoma screening attenders. Acta Derm Venereol 1997; 77:467–470.
37. Jonna BP, Delfino RJ, Newman WG, Tope WD. Positive predictive value for presumptive diagnoses of skin cancer and compliance with follow-up among patients attending a community screening program. Prev Med 1998; 27:611–616.
38. Rampen RH. Point-counterpoint. Mass population skin cancer screening is not worthwhile. J Cutan Med Surg 1998; 2:128–129.
39. Koh HK, Norton LA, Geller AC, et al. Evaluation of the American Academy of Dermatology’s National Skin Cancer Early Detection and Screening Program. J Am Acad Derm 1996; 34:971–978.
40. Rampen FH, Casparie-van Velsen JI, van Huystee BE, Kiemeney LA, Schouten LJ. False-negative findings in skin cancer and melanoma screening. J Am Acad Dermatol 1995; 33:59–63.
41. de Rooij MJ, Rampen FH, Schouten LJ, Neumann HA. Volunteer melanoma screenings. Follow-up, compliance, and outcome. Dermatol Surg 1997; 23:197–201.
42. Federman DG, Concato J, Kirsner RS. Comparison of dermatologic diagnoses by primary care practitioners and dermatologists. A review of the literature. Arch Fam Med 1999; 8:170–172.
43. Burton RC, Howe C, Adamson L, et al. General practitioner screening for melanoma: Sensitivity, specificity, and effect of training. J Med Screen 1998; 5:156–161.
44. Gallagher RP, Ma B, McLean DI, et al. Trends in basal cell carcinoma, squamous cell carcinoma, and melanoma of the skin from 1973 through 1987. J Am Acad Dermatol 1990; 23:413–421.
45. Gamble JF, Lerman SE, Holder WR, Nicolich MJ, Yarborough CM. Physician-based case-control study of non-melanoma skin cancer in Baytown, Texas. Occup Med 1996; 46:186–196.
46. Grodstein F, Speizer FE, Hunter DJ. A prospective study of incident squamous cell carcinoma of the skin in the Nurses’ Health Study. J Natl Cancer Inst 1995; 87:1061–1066.
47. Kaldor J, Shugg D, Young B, Dwyer T, Wang YG. Non-melanoma skin cancer: Ten years of cancer-registry-based surveillance. Int J Cancer 1993; 53:886–891.
48. Bataille V, Bishop JA, Sasieni P, et al. Risk of cutaneous melanoma in relation to the numbers, types and sites of naevi: A case-control study. Br J Cancer 1996; 73:1605–1611.
49. Berwick M, Begg CB, Fine JA, Roush GC, Barnhill RL. Screening for cutaneous melanoma by skin self-examination. J Natl Cancer Inst 1996; 88:17–23.
50. Garbe C, Buttner P, Weiss J, et al. Risk factors for developing cutaneous melanoma and criteria for identifying persons at risk: Multicenter case-control study of the Central Malignant Melanoma Registry of the German Dermatological Society. J Invest Derm 1994; 102:695–699.
51. Tucker MA, Halpern A, Holly EA, et al. Clinically recognized dysplastic nevi. A central risk factor for cutaneous melanoma. JAMA 1997; 277:1439–1444.
52. English DR, Armstrong BK. Identifying people at high risk of cutaneous malignant melanoma: Results from a case-control study in Western Australia. Br Med J (Clin Res Ed) 1988; 296:1285–1288.
53. Marrett LD, King WD, Walter SD, From L. Use of host factors to identify people at high risk for cutaneous malignant melanoma. Can Med Assoc J 1992; 147:445–453.
54. Bliss JM, Ford D, Swerdlow AJ, et al. Risk of cutaneous melanoma associated with pigmentation characteristics and freckling: Systematic overview of 10 case-control studies. The International Melanoma Analysis Group (IMAGE). Int J Cancer 1995; 62:367–376.
55. Elwood JM, Jopson J. Melanoma and sun exposure: An overview of published studies. Int J Cancer 1997; 73:198–203.
56. Elwood JM, Gallagher RP. Body site distribution of cutaneous malignant melanoma in relationship to patterns of sun exposure. Int J Cancer 1998; 78:276–280.
57. Walter SD, King WD, Marrett LD. Association of cutaneous malignant melanoma with intermittent exposure to ultraviolet radiation: Results of a case-control study in Ontario, Canada. Int J Epidemiol 1999; 28:418–427.
58. Byles JE, Hennrikus D, Sanson-Fisher R, Hersey P. Reliability of naevus counts in identifying individuals at high risk of malignant melanoma. Br J Dermatol 1994; 130:51–56.
59. Westerdahl J, Anderson H, Olsson H, Ingvar C. Reproducibility of a self-administered questionnaire for assessment of melanoma risk. Int J Epidemiol 1996; 25:245–251.
60. Little P, Keefe M, White J. Self screening for risk of melanoma: Validity of self mole counting by patients in a single general practice. BMJ 1995; 310:912–916.
61. Lawson DD, Moore DH, 2nd, Schneider JS, Sagebiel RW. Nevus counting as a risk factor for melanoma: Comparison of self-count with count by physician. J Am Acad Dermatol 1994; 31:438–444.
62. Jackson A, Wilkinson C, Ranger M, Pill R, August P. Can primary prevention or selective screening for melanoma be more precisely targeted through general practice? A prospective study to validate a self administered risk score. BMJ 1998; 316:34–38; discussion 38–39.
63. Schneider J.S., Moore DH, 2nd, Sagebiel RW. Risk factors for melanoma incidence in prospective follow-up. The importance of atypical (dysplastic) nevi. Arch Dermatol 1994; 130:1002–1007.
64. Kelly JW, Crutcher WA, Sagebiel RW. Clinical diagnosis of dysplastic melanocytic nevi. A clinicopathologic correlation. J Am Acad Dermatol 1986; 14:1044–1052.
65. Jackson A, Wilkinson C, Pill R. Moles and melanomas—who’s at risk, who knows, and who cares? A strategy to inform those at high risk. Br J Gen Pract 1999; 49:199–203.
66. MacKie RM, Freudenberger T, Aitchison TC. Personal risk-factor chart for cutaneous melanoma. Lancet 1989; 2:487–490.
67. Engelberg D, Gallagher RP, Rivers JK. Follow-up and evaluation of skin cancer screening in British Columbia. J Am Acad Dermatol 1999; 41:37–42.
68. McGee R, Elwood M, Williams S, Lowry F. Who comes to skin checks? NZ Med J 1994; 107:58–60.
69. Hourani LL, LaFleur B. Predictors of gender differences in sunscreen use and screening outcome among skin cancer screening participants. J Behav Med 1995; 18:461–477.
70. Katris P, Crock JG, Gray BN. Research note: The Lions Cancer Institute and the Western Australian Society of Plastic Surgeons skin cancer screening programme. Aust NZ J Surg 1996; 66:101–104.
71. Katris P, Donovan RJ, Gray BN. Nurses screening for skin cancer: An observation study. Aust NZ J Pub Health 1998; 22:381–383.
72. Rivers JK, Gallagher RP. Public education projects in skin cancer. Experience of the Canadian Dermatology Association. Cancer 1995; 75:661–666.
73. Dozier S, Wagner RF Jr, Black SA, Terracina J. Beachfront screening for skin cancer in Texas Gulf coast surfers. South Med J 1997; 90:55–58.
74. Harvey I, Frankel S, Marks R, Shalom D, Nolan-Farrell M. Non-melanoma skin cancer and solar keratoses. I. Methods and descriptive results of the South Wales Skin Cancer Study. Br J Cancer 1996; 74:1302–1307.
75. Harvey I, Frankel S, Marks R, Shalom D, Nolan-Farrell M. Non-melanoma skin cancer and solar keratoses II Analytical results of the South Wales Skin Cancer Study. Br J Cancer 1996; 74:1308–1312.
76. Ichihashi M, Naruse K, Harada S, et al. Trends in nonmelanoma skin cancer in Japan. Recent Results Cancer Res 1995; 139:263–273.
77. Tornberg S, Mansson-Brahme E, Linden D, et al. Screening for cutaneous malignant melanoma: A feasibility study. J Med Screen 1996; 3:211–215.
78. Bergenmar M, Tornberg S, Brandberg Y. Factors related to non-attendance in a population based melanoma screening program. Psycho-Oncology 1997; 6:218–226.
79. Ruskiewicz J. Skin cancer and actinic keratoses. J Am Optom Assoc 1998; 69:229–235.
80. Whited JD, Hall RP, Simel DL, Horner RD. Primary care clinicians’ performance for detecting actinic keratoses and skin cancer. Arch Int Med 1997; 157:985–990.
81. Friedman LC, Webb JA, Bruce S, Weinberg AD, Cooper HP. Skin cancer prevention and early detection intentions and behavior. Am J Prev Med 1995; 11:59–65.
82. Herd RM, Cooper EJ, Hunter JA, et al. Cutaneous malignant melanoma. Publicity, screening clinics and survival—The Edinburgh experience 1982–90. Br J Dermatol 1995; 132:563–570.
83. Marghoob AA, Slade J, Salopek TG, Kopf AW, Bart RS, Rigel DS. Basal cell and squamous cell carcinomas are important risk factors for cutaneous malignant melanoma. Screening implications. Cancer 1995; 75:707–714.
84. van der Spek-Keijser LM, van der Rhee HJ, Toth G, Van Westering R, Bruijn JA, Coebergh JW. Site, histological type, and thickness of primary cutaneous malignant melanoma in western Netherlands since 1980. Br J Dermatol 1997; 136:565–571.
85. Veierod MB, Thelle DS, Laake P. Diet and risk of cutaneous malignant melanoma: A prospective study of 50,757 Norwegian men and women. Int J Cancer 1997; 71:600–604.
86. Koh HK, Geller AC. Public health interventions for melanoma. Prevention, early detection, and education. Hematol Oncol Clin North Am 1998; 12:903–928.
87. Hanrahan PF, Hersey P, D’Este CA. Factors involved in presentation of older people with thick melanoma. Med J Aust 1998; 169:410–414.
88. De Rooij MJ, Rampen FH, Schouten LJ, Neumann HA. Total skin examination during screening for malignant melanoma does not increase the detection rate. Br J Dermatol 1996; 135:42–45.
89. Dhir A, Orengo I, Bruce S, Kolbusz RV, Alford E, Goldberg L. Basal cell carcinoma on the scalp of an Indian patient. Dermatol Surg 1995; 21:247–250.
90. Geller AC, Halpern AC, Sun T, et al. Participant satisfaction and value in American Academy of Dermatology and American Cancer Society skin cancer screening programs in Massachusetts. J Am Acad Dermatol 1999; 40:563–566.
91. Stern RS, Boudreaux C, Arndt KA. Diagnostic accuracy and appropriateness of care for seborrheic keratoses. A pilot study of an approach to quality assurance for cutaneous surgery. JAMA 1991; 265:74–77.
92. MacKie RM, Hole D. Audit of public education campaign to encourage earlier detection of malignant melanoma. BMJ 1992; 304:1012–1015.
93. Giles GG, Armstrong BK, Burton RC, Staples MP, Thursfield VJ. Has mortality from melanoma stopped rising in Australia? Analysis of trends between 1931 and 1994. BMJ 1996; 312:1121–1125.
94. Melia J, Cooper EJ, Frost T, et al. Cancer Research Campaign health education programme to promote the early detection of cutaneous malignant melanoma. II. Characteristics and incidence of melanoma. Br J Dermatol 1995; 132:414–421.
95. Melia J. Early detection of cutaneous malignant melanoma in Britain. Int J Epidemiol 1995; 24:S39–44.
96. Metzger S, Ellwanger U, Stroebel W, Schiebel U, Rassner G, Fierlbeck G. Extent and consequences of physician delay in the diagnosis of acral melanoma. Melanoma Res 1998; 8:181–186.
97. Warso M, Gray T, Gonzalez M. Melanoma of the hand. J Hand Surg 1997; 22:354–360.
98. Larsson K, Shaw H, Thompson J, Harman R, McCarthy W. Primary mucosal and glans penis melanomas: The Sydney Melanoma Unit experience. Aust NZ J Surg 1999; 69:121–126.
99. Holden R, Damato B. Preventable delays in the treatment of intraocular melanoma in the UK. Eye 1996; 10:127–129.
100. Bennett DR, Wasson D, MacArthur JD, McMillen MA. The effect of misdiagnosis and delay in diagnosis on clinical outcome in melanomas of the foot. J Am Coll Surg 1994; 179:279–284.
101. Baccard M, Chevret S, Chemaly P, Morel P. Delay in diagnosing melanoma. A prospective study in 102 patients. Annal Dermatol Venereol 1997; 124:601–606.
102. Epstein DS, Lange JR, Gruber SB, Mofid M, Koch SE. Is physician detection associated with thinner melanomas? JAMA 1999; 281:640–643.
103. Richard MA, Grob JJ, Avril MF, et al. Melanoma and tumor thickness: Challenges of early diagnosis. Arch Dermatol 1999; 135:269–274.
104. Freedberg K. Screening for melanoma: A cost-effectiveness analysis. J Am Acad Dermatol 1999; 41:738–745.