4
BIOPERSISTENCE OF VITREOUS FIBERS
THE Navy addresses the biopersistence of vitreous fibers in the section of Man-Made Vitreous Fibers titled “Fiber Morphology, Deposition, and Clearance” which presents studies on the deposition and dissolution of various types of fibers. As discussed in detail later in this chapter, the subcommittee found the Navy's discussion of biopersistence, the primary determinant of fiber toxicity, to be inadequate with regard to its review of fiber morphology, deposition, and clearance. Furthermore, the Navy failed to cite many relevant publications, including some that provide important insights into the factors affecting target dose, biological responses to inhaled manufactured vitrous fibers (MVF), and methods for assessing biopersistence. To address some of those information gaps, the subcommittee here defines biopersistence, and how to assess and measure it.
DEFINING BIOPERSISTENCE
Biopersistence with regard to MVF refers to the length of time that an intact fiber remains in the lung and pleura (thorax). As first defined at a 1988 meeting of the International Programme on Chemical Safety (IPCS 1988), the term persistence refers to the ability of a fiber to stay in the biological environment; the prefix “bio” has been added to better delineate the definition. This definition is still applicable, although it has been modified to encompass the idea of translocation to target cells and tissues. The importance of this expanded definition is based on the understanding that the biopersistence of MVF in vulnerable tissues of the
thorax is the primary determinant of its pathogenicity: MVF must remain in the tissues long enough for chronic disease to occur (Berry 1999).
Given the importance of understanding biopersistence when determining protective exposure levels, the subcommittee found the Navy's discussion of the following aspects of biopersistence to be inadequate: critical target region, fiber size in relation to penetration and deposition, and in vivo dissolution of fibers.
In the Navy's discussion of the critical target region, it appears to assume that fibers that do not reach the alveoli are of no concern. However, the subcommittee believes that it is important for the Navy to consider very large (noninhalable) fibers that might be of concern for skin irritation, and thoracic fibers that deposit in the lung conductive airways and theoretically might be responsible for bronchitis and bronchogenic cancer in exposed occupational cohorts (Lippmann 1990a,b, 1993).
With regard to the Navy's discussion of fiber size in relation to penetration and deposition in lung regions, the subcommittee notes that in adults breathing orally, fibers with aerodynamic diameters of about 10 µm (physical diameter, about 3.3 µm) will penetrate only into the thorax, whereas most fibers with aerodynamic diameters less than 4 µm (physical diameter, about 1.3 µm) will penetrate into the deeper, gas-exchange regions of the lung (the alveoli) (Lippmann 1990a, 1993). However, nasal breathing would result in less penetration of fibers into the lung. The Navy's discussion of the influence of fiber length on regional fiber deposition should cite the work of Sussman et al. (1991a, 1991b), who showed that fiber length is an important determinant of fiber deposition in lung conductive airways, particularly for fibers longer than 10 µm. Also, the Navy's brief review of the biological effects of fibers longer than 15 µm should cite the stronger association of lung-cancer incidence with the number concentration of fibers longer than 10 or 20 µm with lung cancer incidence, rather than of fibers longer than 5 µm (Lippmann 1994). The subcommittee suggests that the association of lung cancer with fibers longer than 10 or 20 µm might stem from interception and preferential deposition of fibers longer than 10 µm at bifurcations of large airways. Fiber length might also be associated with disease because longer fibers, in general, are more biopersistent, more toxic to cells, and more mutagenic to chromosomes.
The Navy's review of relevant literature on in vivo fiber dissolution is fairly thorough for work conducted before the middle 1990s but is
insufficiently interpretive. The review should emphasize more-recent work, particularly analyses that have used the available literature to construct and validate predictive models of biological response based on fiber length, diameter, and biopersistence. Some recent extremely relevant papers not cited by the Navy are those of: Collier et al. (1997), Bernstein et al. (1996), Eastes and Hadley (1995, 1996), Kamstrup et al. (1998), Hesterberg et al. (1998b, 1999); McConnell et al. (1999); Zoitos et al. (1997); and Searl et al. (1999). Those papers highlight the critical importance of fiber dissolution and lung biopersistence in determining the extent of the risks of lung fibrosis and cancer. They strongly suggest that risks posed by exposure to MVF are more closely related to lung biopersistence than to the number concentration of long fibers or to the gravimetric concentration of inhalable dust. Therefore, a limit of 1 or 2 fibers per cubic centimeter might be highly appropriate for MVF with a given range of biopersistence, but too conservative for less-biopersistent fibers and insufficiently protective for more-biopersistent fibers.
ASSESSING BIOPERSISTENCE
The primary determinants of biopersistence in the lung are the sites of fiber deposition and the extent of clearance. The dimensions of a fiber determine its initial deposition, which, with its durability in the lung, controls its biopersistence and potential translocation. MVF that deposit in intact conducting airways do not appear to cause a significant pathogenic response, because the mucous coating and rapid clearance via the mucociliary escalator prevents interaction between the MVF and the epithelial lining. Airways that are denuded of an epithelial covering, however, because of confounders such as smoking—could conceivably be vulnerable; fibers might then persist in a location not normally susceptible to fiber-related pathogenicity.
The initial response to MVF in noncompromised lung (such as that of a nonsmoker) is restricted to the alveolar region and is driven by the physicochemical nature of the fiber. Surface properties of fibers can be important for assessing their pathogenicity (Fubini et al. 1998; Kane 1996), but this appears to be of less importance for vitreous (amorphous) fibers than for natural fibers that have a crystalline nature, such as asbestos (Heaney and Banfield 1993).
The typical lung response to a fiber is characterized initially by the
recruitment of neutrophils and resident macrophages, which attempt to phagocytize or destroy the offending material, as the lung would any foreign body. The initial response (within hours) in the lung is inflammatory and, for the most part, is restricted to the gas-exchange area of the lung, the site of initial deposition (for example, the bronchoalveolar junction and proximal alveolar ducts and alveoli) (Brody et al. 1985; Warheit 1994). If the fiber remains in the lung long enough, and if the dose is sufficient, cascade of events can ensue, including clustering of macrophages, interstitial and pleural fibrosis, and in some instances neoplasia.
With regard to fiber dimensions, it has been shown in both rodents and humans that most short fibers (less than 5 µm in length) (Bernstein et al. 1996) and nonfibrous particles (Lehnert et al. 1990; Raabe et al. 1986) deposited in the conductive airways of the lung are removed via the mucociliary apparatus within 24-72 hr. The half-life of particles and short fibers deposited in the gas-exchange region is 60-90 days in rats, and up to 10 times greater in humans, depending on their clinical condition. Several animal studies have demonstrated that the inflammatory response stimulated by particles and short fibers resolves once exposure ceases and the offending material is removed from the lung (Hesterberg et al. 1993; Mast et al. 1995a; McConnell et al. 1994, 1999). Long fibers deposited in the gas-exchange airways require macrophage clearance, whose efficiency is dictated by fiber length. The biological explanation of that observation is that fibers shorter than the diameter of a macrophage (rat,10-13 µm; human,14-21 µm) (Crapo et al. 1983; Krombach et al. 1997; Sebring and Lehnert 1992; Stone et al. 1992) can be phagocytized and removed from the lung. Fibers that are too long to be entirely engulfed by a macrophage cannot be removed and therefore have a greater chance of interacting with the epithelium and tissues under it with resultant pathology.
A fiber must reside in the lung for a sufficient time to elicit a chronic response, such as fibrosis or neoplasia. The residence time of a fiber is dictated by its durability. The durability of an MVF depends on its in vivo solubility, a function of its chemical characteristics, length, and diameter and of the microenvironment of the lung. MVF biodegrade in the lung primarily via dissolution. However, a fiber whose length would normally keep it from being removed efficiently, does not have to be completely dissolved to be removed. For example, if a fiber is weakened at any point in its length, it can break into fragments, which can then be
removed via macrophages. MVF are amorphous and always break transversely in contrast with some crystalline fibers, such as asbestos, which tend to split longitudinally (Bellman et al. 1987; Hesterberg et al. 1998a, 1998b). Therefore, as MVF dissolve or break, their removal from the lung is likely to be expedited and their adverse effects mitigated compared with fibers such as asbestos.
In vitro techniques to determine fiber durability at different pH values and in vivo tests to determine overall biopersistence in the lung directly have been developed. Short fibers that are ingested by alveolar macrophages encounter an acidic pH of 4.5-5 in the phagolysosomes (Lundborg et al. 1995), whereas longer fibers, not phagocytizable by alveolar macrophages, are subjected to the extracellular-fluid pH of about 7.4. Therefore, in vitro tests to measure the leaching of specific fiber constituents into the dissolution medium are generally performed at pH values of both 4.5 and 7.4 to simulate the intracellular and extracellular milieus, respectively (Potter and Mattson 1991). Although acellular in vitro techniques measure only dissolution rates of the fibers, in vivo methods measure the overall retention of fibers in the lung, including dissolution rates and mechanical clearance. A comparison of results from in vitro and in vivo techniques is summarized in Table 4-1 for amosite and crocidolite asbestos and several MVFs. Maxim et al. (1999a) presented similar data showing the in vitro dissolution rates of different vitreous fibers plotted against the retention half-times in the lung after inhalation. They demonstrated that there is excellent correlation (R2≈0.90) between in vitro and in vivo measurements (see Figure 4-1). However, in certain cases, current methods of measuring in vitro dissolution rates might not be good predictors of in vivo behavior of fibers longer than 20 µm, that is, fibers that are not phagocytized by alveolar macrophages. More research is needed to optimize and standardize in vitro dissolution methods.
MEASURING BIOPERSISTENCE
The biopersistence of a given MVF is the result of several physical and biological factors, a combination of which are required for pathogenicity (Barrett et al. 1989). In vivo and in vitro studies have been used to determine biopersistence of MVF; in vivo studies are preferred but are expensive and time-consuming.
TABLE 4-1 In Vitro Solubility and In Vivo Biopersistence of Asbestos and Various Types of MVF in Lung
|
Fiber class |
Fiber type and usea |
Kdisb at pH 7.4 |
Time to dissolvec, days |
WT ½,d days |
Ref. |
|
Asbestos |
Amosite |
<1 |
>5,000 |
~400 |
1 |
|
Asbestos |
Crocidolite |
<1 |
>5,000 |
~800 |
1 |
|
RCF |
High temperature applications |
3 |
~1,700 |
~50 |
1 |
|
Other-RCF substitute |
Magnesium silicate |
>150 |
~35 |
~6 |
2 |
|
Glass |
MMVF 32, specialty applications |
9 |
~550 |
~80 |
1 |
|
Glass |
MMVF 33, filtration |
12 |
~400 |
~50 |
1 |
|
Glass |
MMVF 11, building insulation |
100 |
~50 |
~10 |
1 |
|
Glass |
A, building insulation |
250 |
~20 |
~4 |
1 |
|
Glass |
MMVF 10, building insulation |
300 |
~20 |
~35 |
1 |
|
Glass |
P, building insulation |
>500 |
<10 |
~5 |
1 |
|
Glass |
C, building insulation |
>500 |
<10 |
~4 |
1 |
|
Glass |
B, building insulation |
>500 |
<10 |
~2 |
1 |
|
Rock |
MMVF 21, building insulation |
20 |
~250 |
~60 |
1 |
|
Rock |
MMVF 34e, building insulation |
60 |
~80 |
~5 |
1 |
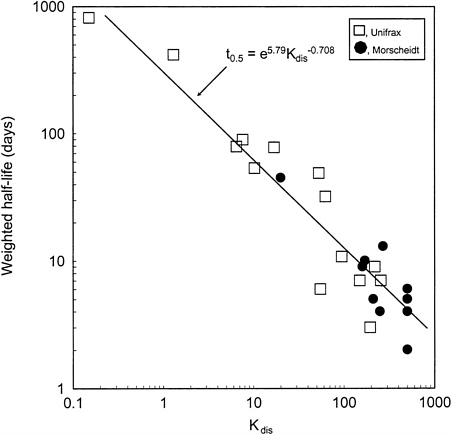
FIGURE 4-1 Relation between in vivo weighted half-life of fibers in short-term inhalation experiments and Kdis. Source: Adapted from Maxim et al. (1999a).
The impact of the aggregate effects of dissolution, breakage, and removal from the lung has been evaluated in vivo. Initial studies to investigate these phenomena were “add-ons” to standard chronic rodent bioassays. In long-term studies, animals (usually rats) were exposed to concentrations of fibers for 2 years; groups of rats were removed from exposure at various times such as 3, 6, 12, or 18 months—and held unexposed for the remainder of the 2 years. The numbers of fibers
remaining in the lungs of the rats exposed for various times were compared (Hesterberg et al. 1993; Mast et al. 1995b; McConnell et al. 1994).
Chronic studies are very costly and time-consuming to conduct, and therefore a simpler method was sought. This has been accomplished through what have been termed "biopersistence" studies (Bellmann et al. 1987). This type of study consists of exposing animals (preferably rats) to a known concentration (number, diameter, and length) of a given fiber type for 6 hr/day for 5 days followed by periodic sacrifices to determine lung fiber burden (Bernstein et al. 1996). Alternatively, rats can be dosed by tracheal instillation with a known amount of fiber in saline, and similar measurements can be made. Although the latter method has been criticized for technical reasons, such as uneven distribution in the lung, it can provide useful comparative information. Findings from both methods are typically presented as weighted clearance half-time (WT½) or as time required to clear 90% of the fibers from the lung (T90) for both WHO fibers1 and fibers longer than 20 µm. Results of these short-term studies have been compared with the results of long-term toxicity studies in animals to establish a WT ½ or T90 and predict what would happen in humans under comparable exposure conditions. The short-term studies have been adopted in Europe as an alternative to long-term carcinogenicity bioassays (EC 1999).
The results of the studies show that various types of MVF differ in their biopersistence (Table 4-1). For instance, Hesterberg et al. (1998a) have reported that the WT½ of MMVF 11 (a standard type of fiberglass insulation) is about 10 days, that of MMVF 33 (a specialty type of fiberglass used for filtration) is about 50 days, and that of amosite asbestos is about 400 days. The T90s of the same fibers were 38, 240, and 2,095 days, respectively. Those findings underscore the importance of biopersistence in explaining the differential pathogenicity of the same MVF, as was seen by McConnell et al. (1999) in a standard lifetime inhalation study in hamsters. McConnell et al. showed that at equivalent exposures, MMVF 10a (similar to MMVF 11) caused only an inflammatory change, MMVF 33 caused a moderate amount of pulmonary and pleural fibrosis and a single mesothelioma, and amosite asbestos caused severe lung and pleural fibrosis and a high incidence of mesotheliomas.
|
1 |
WHO fibers are defined as having a length greater than 5.0 µm, a diameter less than 3.0 µm, and an aspect ratio equal to or greater than 3:1 (WHO 1985). |
The postexposure retention kinetics of deposited fibers can generally be described by a biexponential curve, with a fast phase reflecting fiber clearance from the conducting airways and potential clearance of fiber fragments and a slow phase reflecting—in the case of long fibers (longer than 20 µm)—the elimination of nonphagocytized fibers due to dissolution and breakage. European regulations have adopted the use of a WT½ to describe this clearance behavior (EC 1997) but a numerically calculated half-time may also be used (Miller 1999). However, depending on the fraction of fibers cleared in the fast phase, the fast value could reduce the WT½ to such a degree that the difference between a soluble (low-biopersistence) fiber and a poorly soluble fiber becomes minimal; that is, the power of differentiating between the two might be low. Thus, although the use of WT½ will discriminate different types of fibers, the results could be misleading because both fiber clearance and biopersistence must be considered in disease causation. Therefore, the use of T90 or WT½ values in risk management needs to be evaluated carefully.
In vitro dissolution studies have been proposed as surrogates to predict in vivo solubility of MVF and other fibers (Eastes and Hadley 1996; Zoitos et al. 1997). The rationale is that they are inexpensive and fairly rapid and have been shown to correlate fairly well with the in vivo biopersistence studies discussed above (Eastes et al. 2000). The technique involves placing fibers in something as simple as physiological saline or a solution that mimics what is found in the lung, such as Gamble's solution, and then measuring dissolution of the fiber over time. Dissolution can be measured by determining the dissolution-fluid composition or the weight of the remaining fibers (Potter and Mattson 1991). Such studeis are typically conducted at a pH of 7.4 (the pH of lung fluids), but they can also be performed at a pH of approximately 5 (the pH in a macrophage). The dissolution rate (Kdis) is expressed as nanograms per square centimeter per hour. The results of these studies, like those of in vivo studies, show that MVF vary markedly in solubility within a fiber type and between types (Table 4-1).
Another innovative method that has been used to investigate biopersistence is confocal laser scanning microscopy (CLSM), which allows fibers to be directly visualized in their exact location in the lung and measured in situ without disturbing the lung by fixation or cutting. Rogers et al. (1999) used CLSM to examine portions of lungs from hamsters that had been exposed for 13 weeks, 5 days/wk, 6 hr/day to MVF in a study conducted by McConnell et al. (1999). Rogers et al.
showed that at the end of the exposure period the lengths of MMVF 10a, MMVF 33, and amosite fibers in the lung were reduced by 64.2%, 44.2%, and 8.7%, respectively, in comparison with the original aerosolized-fiber lengths. McConnell et al. (1999), showed that after a single 6-hr exposure in hamsters, almost twice as many WHO amosite fibers and approximately equal numbers of fibers longer than 20 µm were deposited in the lung. The results of CLSM constitute additional evidence that differential shortening occurs in fibers retained in the lung. However, the value of CLSM is limited as it can only be used to visualize to a depth of a few millimeters in the lung, whereas more-centralized locations cannot be visualized without cutting the lung, which would diminish the advantages of the technique. At present, CLSM should be viewed as a research tool rather than as a means of assessing biopersistence definitively.
CONCLUSIONS
The potential hazards posed by a given MVF is directly related to its ability to persist in the lung long enough to cause chronic disease. This persistence has been termed biopersistence. Research has shown that persistence in the lung is directly related to the chemical composition and dimensions of fibers. Biopersistence can be measured with long-term and short-term studies. It has also been proposed that simple in vitro fiber solubility studies can reasonably predict what will happen in vivo. Most of the fibers that are of concern to the Navy have been investigated with one or more of these techniques, and review of the resulting data can be valuable for estimating the hazards potentially associated with the fibers.











