CHAPTER 13
ANALYSIS OF ALTERNATIVE EMISSIONS CONTROL STRATEGIES
(Chapter 13 was written by D.Warner North and M.W.Merkhofer under the general supervision of the committee, which reviewed the work at several stages and suggested modifications that have been incorporated. While every committee member has not necessarily read and agreed to every detailed statement contained within, the committee believes that the material is of sufficient merit and relevance to be included in this report.)
INTRODUCTION AND SCOPE
Sulfur oxide and particulate emissions have adverse consequences for human health and welfare, but the means for controlling these emissions entail considerable expense. This section of the report presents a quantitative framework for comparing alternative strategies for emissions control from stationary sources. The primary focus of attention will be on emissions from coal fired steam electric power plants in the eastern United States. The method of approach to be used in this section is easily adapted to other sources and other regions of the country.
The Clean Air Amendments of 1970 required that by July 1, 1975 specified levels of ambient air quality for sulfur oxides must be met. On the basis of these ambient standards the State Implementation Plans (SIP) were developed that set sulfur oxide emission limitations. The national primary ambient air quality standard for sulfur oxide is 80 ug/m3 for the annual arithmetic mean and 365 ug/m3 for a maximum 24-hour concentration. These values were selected to protect human health, with a margin of
safety. No separate standard presently exists for suspended sulfate levels.
When the Clean Air Act was passed and during the period when State Implementation Plans were being developed, there was still no obvious indication that natural gas would be in very short supply for industrial and utility users or that the U.S. would be unable to rely on imported oil to supply a fuel that was lower in sulfur than the indigenous coals that were being burned. Many utilities converted their coal-fired facilities to low sulfur oil or gas as quickly as they were able, and by 1974 23,600 MW of capacity was burning oil (although these facilities may be reconverted to burn coal). The emerging energy shortages culminated with the Arab embargo of oil to the U.S. in October 1973. This event in turn has motivated an energy policy that puts increased emphasis on reducing oil imports to a level low enough that the U.S. economy can continue to function satisfactorily even if the imports are again embargoed.
The shift to low sulfur fuels was made because it appeared to the electric utility industry to be the best way to meet the new sulfur oxide standards. Tall stacks and intermittent control systems facilitate compliance with sulfur dioxide ambient standards, but they do not reduce the total amount of sulfur oxides released into the atmosphere. The commercial feasibility of stack scrubbing devices, the other viable option for reducing sulfur oxide emissions, has been a matter of sharp dispute between the utility industry and the Environmental Protection Agency (EPA). It is obvious that the sulfur oxide levels that were to be achieved by July 1, 1975 (under the terms of the Clean Air Amendments of 1970) cannot be met now even if stack scrubbing technology were ready to be used routinely at
power plants, because of the time required to build and install the scrubbing devices.
During the last decade, emissions and ambient sulfur dioxide concentrations in urban areas have decreased, while nationwide emissions from electric power plants have nearly doubled. Ambient concentrations of sulfate particles in urban areas have not decreased but have remained almost constant from 1957 to 1970. This persistence of high urban sulfate levels despite the decline in urban sulfur oxide emissions may be the result of the increased emissions from remotely located electric power plants. Sulfate levels approaching the level of urban concentrations have been observed in rural areas of the Northeast with no local sources of sulfur oxide emissions and very low sulfur dioxide ambient levels.1
Sulfur oxide and particulate emissions from power plants may pose a serious health hazard. Sulfur oxides and suspended particulate matter may act to impair health by a variety of possible mechanisms following inhalation and retention in the human respiratory tract. It will require further investigation to elucidate these mechanisms. In past epidemiological studies devoted to examining the health effects of air pollution, the pollution parameters of sulfur dioxide concentration and total suspended particulate matter concentration have been utilized for correlation with effects. These parameters are probably only indicators of the toxic potential of the pollution mix and not causal agents. Thus, the particulate phase is known not to be a single agent, but a complex mixture of particles of different size, shape, density and chemical composition. The CHESS studies suggested that particulate sulfates, rather than either of the above two parameters, may be a better indicator of the toxic potential of the polluted atmosphere.2
Laboratory studies which utilized animals have also suggested that certain particulate sulfates (Zn(NH4)SO4, ZnSO4 and H2SO4) are potent bronchoconstrictors, far more effective than sulfur dioxide in air at concentrations comparable to the particulate sulfate concentration. The bronchoconstriction capacity
of the particulate matter increased as particle size decreased in the guinea pig assay method, which utilized pulmonary flow resistance as the indicator of toxic potential.
For the above reasons, and with full recognition of the large uncertainties which still remain to be resolved, this analysis of the social costs of sulfur oxides and particulate pollution will focus on the impact of control strategies for particulate sulfate concentrations in air. The uncertainties in this approach are not only associated with the effects of sulfates on health and the ecosystem, but with the measured and predicted concentrations of sulfate in air; analytical methods to determine particulate sulfates in air are not yet reliable. As will be seen, the range of uncertainty on many of the factors in the analysis is therefore large.
ALTERNATIVES FOR EMISSIONS CONTROL
This portion of the report will examine the costs for various control methods that might be adopted, together with the reduction in emissions that each method might achieve3. The analysis will address the choice among alternatives that can be implemented by 1980. Promising technologies are under development to remove sulfur before or during combustion, and to improve efficiency, thereby reducing the quantity of fuel needed to generate a given quantity of electricity. However, these technologies cannot be implemented on a large scale until 1985 or later, and the costs may not be significantly lower than the technologies presently available to remove sulfur from stack gases (see Chapter 10). Therefore, the analysis will focus on alternatives that are presently available.
The presently available alternatives for controlling sulfur oxide and particulate emissions are the following:
Tall Stacks and Intermittent Control
A stack height of the order of 100 to 300 meters may be sufficient (depending to some extent on the quantity of sulfur oxides emitted) to disperse the plume of effluent gases over a wide area, permitting ambient concentrations to be held below the levels established as standards. Under some meterological conditions high concentrations that violate standards may develop. A meteorological monitoring system is used to anticipate the outset of these conditions, and an intermittent control is then exercised to reduce emissions by shifting to a cleaner fuel or by reducing the levels of operation of the plants. The net result is that the total quantity of emissions may be reduced slightly or not at all, but ambient concentration in violation of standards may be avoided (see Chapter 12).
Coal Preparation
By pulverizing the coal and washing it prior to combustion it is possible to remove much of the physically bound portion of the sulfur and a large fraction of the ash. The coal washing process is relatively inexpensive, but some of the energy content of the coal is lost (see Chapter 10).
Shifting to Low Sulfur Coal
Much of the coal burned by utilities in the eastern United States has a sulfur content of 2–6 percent by weight. Some eastern coal is available with a sulfur content below 1 percent, but its extent is limited, and much of it is held for metallurgical applications. Low sulfur eastern coal commands a substantial price premium, which might increase if there were additional demand for low sulfur fuel. Low sulfur western coal is abundant, but the mining and transportation capability does not currently exist to provide it in large quantity to the eastern United states. Because of the low BTU
and high ash content compared to the eastern coals, it is generally not possible to burn western coal in a boiler designed for eastern coal without extensive retrofitting or derating of the plant capacity (see Chapter 10).
Flue Gas Desulfurization (FGD)
A number of technologies are under development for scrubbing pollutants from the effluent gases before they are released from the stack. The lime scrubbing process appears to be the best developed technology for coal fired power plants. It permits removal of the order of 90 percent of the sulfur oxide, plus much of the fine particulate matter. Both capital cost and operation costs of flue gas desulfurization are high, but it is the most effective means of removing sulfur oxides and other pollutants from the emissions into the atmosphere (see Chapter 11).
Demand Modification
Since sulfur oxides and other pollutant emissions from power plants are a by-product of electricity generation, one alternative for reducing these emissions is to reduce demand growth for electric power. The relationship between growth and emission levels is not a simple one, however. Demand is allocated among plants in an electric power system so as to meet demand with acceptable reliability at the least total cost. In practice, the newest and most efficient plants are used almost continuously to meet the base load, while the oldest plants are used to meet the peak loads and to furnish reserve capacity. The allocation of demand for electricity from an electrical system is summarized by the total energy demand made on each plant. This total demand is usually expressed by the loading (load factor) for the plant: the equivalent number of hours (percentage of time) the plant must operate during the year at its rated capacity in order to provide that amount of energy. A slowing of
demand growth that leads utilities to delay construction on new plants with low emissions levels may have little effect on total pollutant emissions from the system, if older plants with high emissions continue to be used at high load factors (see Chapter 8).
Nuclear power plants provide an economical means of producing base load power without emitting any sulfur oxides or particulates. By accelerating the construction of nuclear power plants, the loading, and consequently, the sulfur oxide emissions from coal fired plants can be reduced. (Of course, there are other environmental problems associated with nuclear power that should be assessed in considering it as an alternative to coal fired plants.)
A continued national effort toward domestic self-sufficiency may result in shifts from oil and gas to coal as a fuel for electric power generation. This effort may involve shifting from oil or gas to coal for many existing power stations that have the capability to burn coal, conversion to coal burning capability for fossil steam plants now being planned or under construction, and higher loading for existing coal fired plants as oil or gas fired plants are taken out of operation or reduced in loading.
METHODOLOGY
The approach to be taken in comparing alternative control strategies is to assess their economic impact on the costs associated with generating electricity and their effect in reducing emissions. A judgment must then be made to evaluate this tradeoff: What increment in increased electricity costs is justified by a given level of emission reduction? We shall assess the benefits of emission reduction by modeling the effect of the emissions on ambient air quality levels and on the deposition of pollutants, then modeling the effects on human health, materials damage, ecological changes, and aesthetic degradation. Through an assessment of costs and benefits the analysis can provide guidance to policymakers in assimilating the complex array of factors that
impact on the tradeoff between the consequences of the pollution and the costs associated with emissions control.
Considerable uncertainty may characterize the costs of the alternatives, the emissions reductions that may be achieved, the relationship between emissions and ambient air quality levels, and the health, materials damage, and environmental consequences of given levels of pollutant concentration. The degree to which these uncertainties will impact on the decision among alternative control strategies may be identified by sensitivity analysis. If changing a factor within its range of uncertainty will change the preferred decision alternative, it will be useful to quantify the uncertainty by assessing a probability distribution over the range of values the uncertain factor could assume. The value of resolving the uncertainty can then be computed from the decision context (see Howard 1966, 1968; North 1968, and Tribus 1969 on the use of probability in decision analysis; see Spetzler and Stael von Holstein 1972 on methods to encode probability distributions). The scope of the present report does not permit an extensive application of these methods. The approach will be illustrated on the most important uncertainties; the analysis could be expanded to include other uncertainties.
In the context of a public policy question such as controlling emissions from power plants, the assessment of overall costs and benefits may need to address issues of equity and of distribution: different people may receive the benefit than those who pay the costs. Cost-benefit analysis of public policy decisions usually assumes implicitly that the parties to the decision may be persuaded to make their choice on the basis of maximizing the overall net benefits to society. The question of how to implement the socially optimal alternative may well be the most difficult aspect of the problem. While a cost benefit analysis may be useful in identifying the best alternative from society’s point of view, considerable further effort may be needed to determine what is the best alternative for practical and effective
public policy. The public policy decision makers must understand how the various concerned private parties to the decision will react to a new policy initiative, and they must choose with care the means by which the private parties are to be motivated to act in society’s best interest.
There are two ways that a private party may be motivated to act in the public interest when it is at variance with his own immediate objectives: (1) his decision alternatives may be limited by regulations or standards imposed on him by public authority (2) his values may be shifted toward the overall values of the society by economic means: incentives, taxes, penalties, fees; or by non-economic means such as persuasion that his action will gain him the good will (or enmity) of his fellows. The legalistic approach places the responsibility for planning on the public authority, which must assimilate a complex array of economic and technical factors in order to establish the standard. Once established, a standard is difficult to change. If new information indicates that the standard is not appropriate, the planning exercise must be redone and the concerned private and public parties convinced that the change in standards is justified.
The use of economic incentives has been advocated by virtually every economist who has written on pollution, but it has rarely been used as a way of controlling emissions4. It has the advantage of flexibility: by delegating the social cost of the pollution as a direct cost to the private party making the decision, the public authority provides him with the incentive to make decisions that are optimal from the viewpoint of the public authority. Planning is therefore decentralized, and the detailed knowledge possessed by the private parties can be used to improve the decision process. Flexibility is much easier to achieve: If the public authority determines that levels of pollution are too high, it raises the cost associated with emissions, providing an incentive to the private parties to reduce them.
THE EMISSIONS CONTROL DECISION FOR A REPRESENTATIVE ELECTRIC POWER PLANT
The public policy decision on emissions control will involve setting standards or implementing a system of incentives and fees. The actual resource allocations to implement an emissions control alternative will be made by the electric utility. The decision problem on emissions control is ultimately whether the owners of a power plant shall modify their operations by such means as installing a flue gas desulfurization (FGD) process, switching to a low sulfur content fuel, or installing a taller stack and intermittent control system. The adoption of the emission control strategy will result in higher costs to the owners of the power plant, and these higher costs will generally be passed on as higher prices to the consumers of electricity. The benefits from adopting the emissions control strategy come from the change in amount (and timing) of emissions of sulfur oxide and other materials into the atmosphere that may adversely affect human health, cause damage to other living organisms or material property, and result in effects, such as visibility reduction, that are aesthetically undesirable. A decision between alternative strategies requires a balancing of the additional cost imposed on the generation of electric power against the value of emissions reduction.
The analysis will focus on sulfur oxide emissions from coal fired steam power plants in the northeastern United States. As described in other sections of this report, most of the sulfur is emitted as sulfur dioxide rather than as sulfur trioxide or sulfate, but subsequent atmospheric chemical processes may oxidize the sulfur dioxide to sulfuric acid aerosol and suspended particles of ammonium sulfate and other sulfate salts. Recent epidemiological data have indicated that these sulfates may give rise to serious and widespread health effects (EPA 1974). Damage to material property from atmospheric sulfur oxides has been estimated to cause hundreds of millions of dollars in annual losses (Waddell 1974). “Acid rain” resulting
from atmospheric sulfur oxides may lead to retarded growth in forests, deleterious effects on lakes and streams, damage to agricultural crops, and damage to building materials, statues and other material property (see Section 1 of Part Two). Aesthetic effects from degraded visibility may be another substantial problem. While sufur dioxide is invisible, sulfate particles do absorb and scatter visible light5. Degraded visibility in areas with high real estate or environmental values is a substantial public concern that should be appropriately reflected in the values associated with sulfur emissions (Randall et al. 1974).
There are many distinctions that must be made between power plants in different locations. Ideally, a detailed model for assessing costs and benefits should be developed at each power plant for which a decision on emissions control is to be made. This scale of effort is not possible in the present study. We shall not attempt to address in detail the decision at a particular plant, but rather do illustrative calculations that are chosen to be representative of different types of plants and different locations in the northeastern United States. Specifically, we shall consider the following as representative cases:
-
An existing coal fired plant in a remote rural location.
-
An existing plant capable of burning coal in an urban or near-urban location.
-
A new coal fired plant in a remote rural location.
A fourth category, a new coal-fired plant in an urban or near-urban location could be added, but for this category the decision would seem relatively obvious: an efficient sulfur removal system would almost surely be required under existing state and local air quality regulations. Federal New Source Performance Standards (NSPS) will require flue gas desulfurization on new coal-fired plants beginning in 19756. (This requirement also holds true for remotely sited plants, but since some utilities have alleged that application of these standards to such plants serves little or
no social purpose, consideration of that case was included in this analysis.)
The approach in carrying out the cost-benefit analysis will be to evaluate alternative strategies for representative power plants in each category by assessing the economic costs of electricity generation and the costs associated with sulfur oxide pollution effects. Because of the suspected hazards of sulfate, the emphasis will be on regional effects caused by long range pollutant transport, rather than local violations of air quality standards in the immediate vicinity of the plant. The intention is to assess the marginal benefit of pollution control strategies for this plant as it affects regional ambient levels of sulfur dioxide, sulfate, and acid rain, and to compare the consequences associated with these levels with the marginal increases the strategy will impose on the cost of electricity generation. Both the consequences of the sulfur oxide emissions and the cost of abatement methods will vary, depending on local and regional conditions. These local and regional factors should be taken into account in the decision among alternative emissions control strategies.
AN OVERVIEW OF THE ASSESSMENT OF COSTS AND BENEFITS FOR A REPRESENTATIVE PLANT
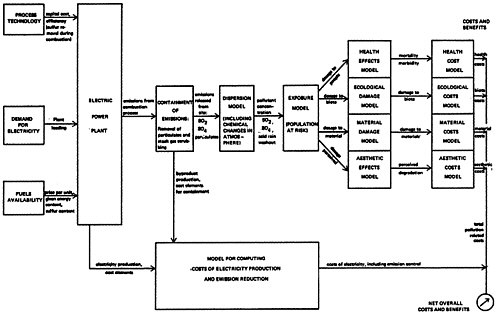
Figure 13–1 presents an overview of a model for assessing costs and benefits, which will be applied for each alternative strategy for pollution control. The framework is a general one that could be applied to any stationary source, but the concern here is a coal fired steam electric plant. This plant meets demand from the power system for electricity: The effect of demand on an individual plant is summarized by its loading, the number of hours the plant is operated.
In steam power plants the sulfur content of the fuel is oxidized in the combustion process and mixes with other combustion products that are exhausted from the boiler. The heating value and sulfur content of the fuel and the efficiency of the power plant determine the
amount of sulfur oxides produced per kilowatt hour (kwh) of electricity generated. Sulfur oxide emissions from the combustion process may be removed by stack gas scrubbing devices, and particulates may be removed by the scrubing devices or electrostatic precipitators. The costs of these technologies are assessed and included in the cost of electricity generation.
The price of the fuel, e.g., the number of dollars per ton of coal, varies by its energy content and its sulfur content. For example, a coal with 24 million BTU’s heating value per ton and 3 percent sulfur content (by weight), is assumed for this analysis to be available at $24/ton delivered to the power station. Lower sulfur content coal will generally be more expensive: for example $32/ton for coal with 0.9 percent sulfur and the same heating value. The supply of low sulfur coal is limited, and a large shift from high sulfur to low sulfur coals for power plant fuel would result in an increase in their relative prices. The supply-demand-price relationship may be critical in the evaluation of an overall national policy intended to cause a shift to lower sulfur content coal, but changes in the price relationship will not be discussed further. If the price increase is sufficiently high, the situation becomes equivalent to not having low sulfur coal available. We shall examine how the choice among alternative strategies is affected if eastern low sulfur coal is not available.
From the fuel prices, capital costs, plant efficiency, and loading, the costs of electricity generation may be computed. We can then compare alternative strategies for emission control on the basis of two numbers: the cost per kilowatt-hour of electricity generated, and the sulfur oxide emissions produced per kilowatt-hour of electricity7. For example, a representative existing power plant burning 3 percent sulfur coal produces electricity for a cost of 17.2 mills per kilowatt-hour and emits 0.026 lbs of sulfur per kilowatt-hour generated. If higher priced (eastern) low sulfur (0.9 percent) coal is used, the cost of electricity increases to 20.6 mills per kilowatt-hour, but
emissions are reduced to 0.0078 lbs of sulfur per kilowatt hour generated.
A choice between these two alternatives implies assessing the tradeoff between reducing the sulfur emissions and increasing the cost of electricity. A simple way in which to assess the tradeoff is by placing a value on each pound of sulfur emission, and then examining the total cost of electricity generation plus the cost attributed to sulfur emissions. We shall carry out this calculation on an incremental or marginal basis for the individual power plant, rather than national implementation of the emission control alternatives. That is, we shall assess the benefits from reducing emissions by a relatively small amount relative to existing pollution levels, and compare them to the costs based on existing price levels for the emission control alternatives. Benefits from emissions reduction may depend on ambient levels, and costs for emission control alternatives may depend on the demand for them. For example, if health effects from ambient sulfate were significant only above a threshold level, then the pollution cost per pound of sulfur emitted would drop substantially once ambient sulfate levels are reduced below this threshold level. Likewise, increased use of low sulfur coal to reduce power plant sulfur oxide emissions will cause the price premium over high sulfur coal to rise. In our calculations we assume only small changes in pollution levels (and costs) as a result of implementing an alternative strategy at a particular power plant. As a consequence, the pollution cost per pound of sulfur is assumed to remain constant for this power plant regardless of the amount of sulfur emissions removed. However, the pollution cost per pound of sulfur removed will depend on many factors (such as ambient pollution levels) that differ for different plants in different locations. The benefits of a control strategy per pound of sulfur removed may be substantially different at different power plants.
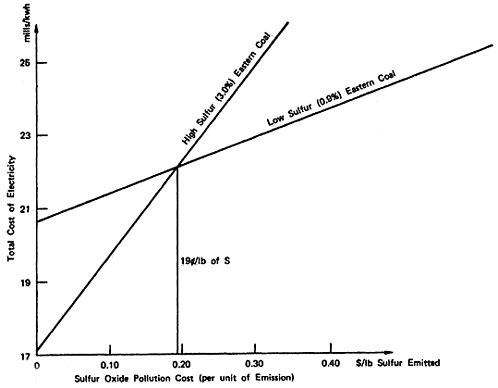
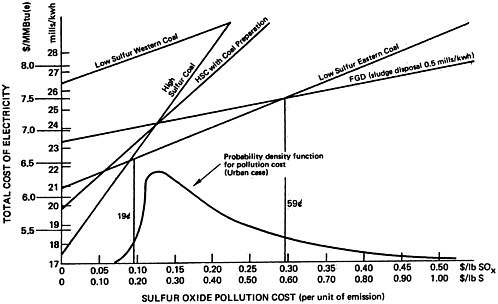
Figure 13–2 shows the calculation of a representative existing power plant that can burn 3 percent sulfur coal or a more expensive
0.9 percent sulfur coal. If no cost is attributed to sulfur emission the total cost for the two alternatives is 17.2 and 20.6 mills per kilowatt hour as before. Suppose instead a cost of $0.30 is attributed to each pound of sulfur emitted into the atmosphere. Then the total cost for generating a kilowatt-hour using high sulfur coal is 25 mills per kilowatt-hour, 17.2 for the cost of producing the electricity plus 0.026×300 mills (=$.30) for the cost attributed to the sulfur emissions. The total cost of generating a kilowatt-hour using low sulfur coal is 23 mills/kwh, 20.6 for the cost of producing the electricity plus 0.0078×300 mills (=$.30) for the sulfur emissions. The lowest total cost is now achieved using low sulfur coal, whereas when emissions were assigned a zero cost, high sulfur coal resulted in the lowest total cost. The total cost for producing electricity plus the cost of the emissions associated with producing the electricity is shown plotted against the cost assigned to a pound of sulfur emissions in Figure 13–2 for each of the alternatives. It may be seen that the graph of total cost for each alternative is a straight line, since the cost increases linearly with the pollution cost per unit emitted. The slope of the line is given by the pounds of sulfur emitted per kilowatt-hour generated. The point at which the two lines cross gives the cost per pound of sulfur emitted ($.19) at which the total costs of the alternatives are equal: If the cost attributed to sulfur emission is greater than 19 cents per pound of sulfur, the best alternative is the low sulfur coal; and if the cost attributed to sulfur emissions is less than 19 cents per pound of sulfur, the best alternative is the high sulfur coal. A judgment that the low sulfur coal alternative is better than the high sulfur coal alternative is equivalent to a judgment that the social cost of sulfur emissions are judged to exceed 19 cents per pound of sulfur emitted, for this is the equivalent increase in the price of electricity that must be accepted in return for the lower emission levels.
Similarly, a judgment to use 3 percent sulfur coal in this plant instead of the more expensive
low sulfur coal implies a judgment that the social cost of sulfur emissions is less than 19 cents per pound of sulfur emitted; the reduction in emissions from a switch to low sulfur coal is judged not worth the increased electricity generation costs.
If the dollar value of the consequences of sulfur emissions could be accurately assessed, it would be a simple matter to carry out this type calculation to determine what emissions control alternative is best in the sense of providing a kilowatt-hour of electricity at the least total cost to society8. But the consequences of the emissions are complex and uncertain, and the assessment process is a difficult one. However, it must be kept in mind that any decision to impose a particular alternative technology, or a requirement that emissions shall be held below a specified level, implies a value judgment on the cost of pollution. A decision among emissions control alternatives is required: either the existing fuels and control technology will be used, or a switch will be made to another type of fuel and/or emission control technology. Since this decision will implicitly require judgments on the value of reducing emissions, the assessment process cannot be avoided. The value of emissions reduction must be assessed, either explicitly or implicitly.
An explicit method for assigning values to the social cost of pollution is represented by the right hand side of Figure 13–1. The left hand side may be regarded as a model for calculating the emissions released into the environment and electricity generating costs for a representative electric power plant. The boxes shown on the right hand side represent the models used to evaluate the social costs to be assessed on the emissions from that power plant:
-
A Dispersion Model to relate sulfur oxide and particulate emissions to ambient concentrations of sulfur dioxide, sulfate, and particulates, and to acid rain washout.
-
An Exposure Model of the population, biota, and material property that may be impacted by the pollutant
-
concentrations. The output of this stage is the dosage of pollutant received.
-
Models for the effect of a given dosage to a given population on human health, on vegetation and other ecological systems, on material property, and consequences that are aesthetically undesirable, such as visible smog. The output of this stage is a description of the physical consequences of the pollutant concentration: for example, morbidity and mortality, reduced growth in vegetation, eroding of galvanized steel, and reduction in visibility.
-
The last stage is the assessment of values on these physical consequences of pollution so that they may be compared with the costs of emissions control strategies. Values assigned to health, vegetative damage, materials damage, and aesthetic degradation become the basis for valuing the effects caused by a given level of emissions, and the model structure can be used to compute a value per pound of pollutant emitted for the evaluation of alternative strategies as illustrated in Figure 13–2.
The need for making a decision motivates the need for the value assessment process. The models for the emission to ambient relationship, the effect of ambient air quality on human health, the effect of acid rain on vegetation, etc., are summaries of the information available as a basis for decision making. There may be substantial uncertainties in some of the factors or relationships used in these models, and there may be disagreements on the valuing, for example, of health effects. In carrying through the assessment process, attention and effort should be focused on those uncertainties and value judgments that affect the decision.
Now that the methodology has been described, we turn our attention to the calculation of the electricity generation costs and emissions levels for representative plants, which will be summarized in a series of diagrams constructed
in the same manner as Figure 13–2. We then develope dispersion models for the emissions to ambient relationship for these representative plants. Finally, models for the effects on health, ecological systems, material property, and aesthetics are used to evaluate the consequences of pollution and assess a social cost per unit of emissions. Important uncertainties on pollution consequences are reflected in a probability distribution over a range of values for this pollution cost, and the impact of the uncertainty on the emissions control decision is examined. Since resolving the uncertainty leads to an improvement in the decision compared to a choice made on the basis of presently available information, a value of resolving the uncertainty will be calculated.
CALCULATION OF TOTAL SOCIAL COSTS FOR VARIOUS SULFUR EMISSION REDUCTION ALTERNATIVES
Calculations are now presented for the electricity generation costs and emission levels that might be expected if various decision alternatives are chosen. The alternatives considered are:
-
High sulfur coal, perhaps with tall stacks and intermittent control.
-
Removal of sulfur from fuel before combustion (coal preparation)
-
Flue gas desulfurization (FGD)
-
Shift to low sulfur fuel (eastern or western low sulfur coal)
Since the best strategy for controlling sulfur emissions may differ for different plants, three representative cases are considered:
-
An existing coal fired plant in a remote non-urban location.
-
A coal fired plant planned for construction in the near future in a remote non-urban location.
-
An oil burning plant, originally designed to burn coal, which may be reconverted to coal. This plant is presumed to be located in an urban area of the East Coast.
Detailed assumptions for the specific plants are summarized in Table 13–1. Since an additional alternative might be to construct a nuclear plant instead of a new coal fired plant, data for a base load nuclear plant is also included in the table for a point of reference.
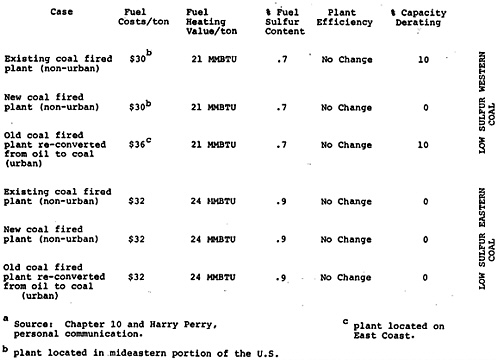
Tables 13–2, 13–3, and 13–4 give the cost and sulfur emissions data assumptions made in the evaluation of each alternative. Coal preparation is assumed to require a small capital investment and to result in coal heating value losses of the order of 10 percent. The reduction in sulfur content achievable by coal cleaning depends on the particular process used. Conventional techniques now available will reduce the sulfur content of high sulfur coal by approximately 40 percent10. Flue gas desulfurization, represented by the lime scrubbing process, is expensive: there is a sizeable capital cost, additional operating costs, a loss of plant capacity, and an associated energy loss. The price of low sulfur western coal depends critically on transportation costs. A higher price is assumed for plants located on the East Coast as opposed to the mideastern portion of the country. For older plants with boilers not designed for lower BTU coal, capacity derating will result from the burning of the lower heating value western coal.
Use of tall stacks and intermittent control is equated to the base case, which we henceforth denote by “high sulfur coal”. Such techniques may be useful in meeting ambient standards for sulfur dioxide in the immediate vicinity of the plant, but it is not anticipated that their costs or contributions to the overall reduction in sulfur oxide emissions would be significant relative to those of the other strategies being considered11. An efficient particulate removal system is also assumed. The importance of particulates in oxidizing sulfur dioxide to sulfates will be discussed in the next section, below.
Capital charges, operating and maintenance, and fuel costs for each plant type under each alternative were summed to obtain an estimated production cost per kilowatt-hour of electri-
TABLE 13–1
Economic and Technical Factors for Representative Power Plants.a
|
|
TECHNOLOGY |
FUEL |
ECONOMICS |
||||||||
|
CASE |
Plant Size (MW) |
Average Load (hrs/yr) |
Load Factors % |
Efficiency % |
Fuel |
Sulfur Content % |
MMBTU/ton |
Cost including trans. |
Capital Cost ($/kw) |
Capital Amortization % |
Operating Cost (mills/kwh) |
|
Existing coal fired plant (non-urban) |
620 |
6000 |
69 |
33 |
Coal |
3 |
24 |
$24/ton $1.00/MMBTU |
250 |
14 |
1.0 |
|
New coal fired plant (non-urban) |
612 |
7000 |
80 |
38 |
Coal |
3 |
24 |
$24/ton $1.00/MMBTU |
500 |
17 |
.5 |
|
Old coal fired plant re-converted from oil to coal (urban)b |
620 |
6000 |
69 |
33 |
Coal |
3 |
24 |
$24/ton $1.00/MMBTU |
261e |
14 |
1.0 |
|
Nuclear plant |
1000 |
7000 |
80 |
32 |
U |
- |
- |
$.18/MMBTU |
800 |
17 |
.4 |
|
a Sources: Chapter 10 and personal communications, Harry Perry. b plant assumed to be located on East Coast. d plant is assumed to be equiped with a particulate collection system. e includes $7/kw conversion charges plus $4/kw to upgrade the particulate collection system. The latter is not charged if FGD is employed. |
|||||||||||
TABLE 13–2
Coal Preparation Cost Factorsa
|
CASE |
Capital Costs |
Added Fuel Costs/tonb |
% Energy Loss |
% Reduction Sulfur Content |
|
Existing coal fired plant (non-urban) |
$6/ton yr. |
$2.25 |
10 |
40 |
|
New coal fired plant (non-urban) |
$6/ton yr. |
$2.25 |
10 |
40 |
|
Old coal fired plant re-converted from oil to coal (urban) |
$6/ton yr. |
$2.25 |
10 |
40 |
|
a Source: Chapter 10. b Includes amortization of capital costs. |
||||
TABLE 13–3
Flue Gas Desulfurization Cost Factorsa
|
CASE |
Capital Costs/kw |
% Amortizationb |
Operating Costs/kwhc |
Sludge Disposal Cost/kwhd |
% Capacity Deratinge |
% Energy Consumed |
% Reduction of Sulfur Oxides |
|
Existing coal fired plant (non-urban) |
$125 |
17 |
0.6m |
0.5m |
6 |
6 |
90 |
|
New coal fired plant (non-urban) |
$100 |
17 |
0.5m |
0.3m |
6 |
6 |
90 |
|
Old coal fired plant reconverted from oil to coal (urban) |
$125 |
17 |
0.6m |
0.9m |
6 |
6 |
90 |
|
a Costs based on lime scrubbing. Source: Chapter 11 and personal communications, Leigh Short. b Harry Perry, personal communication. c labor, maintenance, and supplies cost. Capital amortization, sludge disposal, and energy loss included separately. d Ponding of sludge is assumed for the non-urban site. For the urban sites it is assumed that sludge will have to be disposed of by other means. e capacity valued at $500/kw. |
|||||||
city. The sulfur emissions in pounds of sulfur per kilowatt-hour of electricity were also calculated. Table 13–5 summarizes these results. Details of the cost calculation for the base case (high sulfur coal) and for flue gas desulfurization are given in Appendix 13-D.
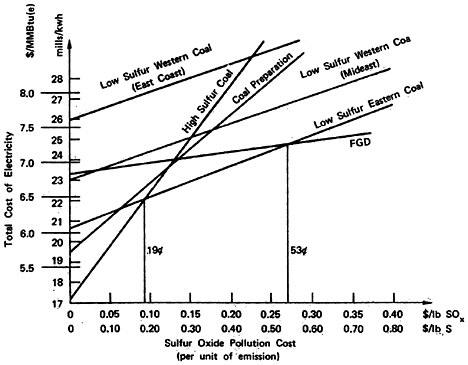
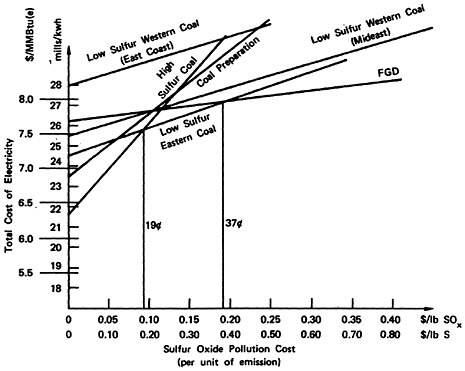
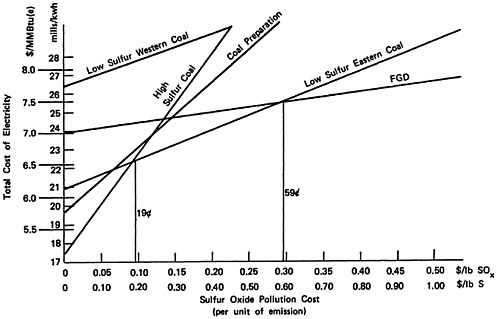
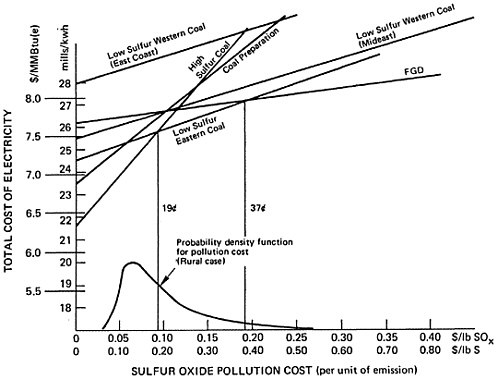
Total social cost will be the production cost plus the product of sulfur emissions and pollution cost per unit of sulfur emission. For this reason, the plots of total social cost versus pollution cost, illustrated in Figures 13–3, 13–4, and 13–5, are straight lines. We may easily identify the most desirable emission reduction alternative as that straight line relationship that, for a given assignment for pollution costs, yields the lowest value of total social costs. Inspection of the figures show that for pollution costs below 19 cents per pound of sulfur emitted the best alternative is to burn high sulfur coal. As pollution costs are increased above 19 cents per pound of sulfur emitted the best alternative becomes eastern low sulfur coal. We note that there is little difference in the location of this crossover point among the three cases. For the cost elements given in Tables 13–1 through 13–4 the alternatives of coal preparation and switching to western low sulfur coal do not give as large a marginal reduction in sulfur emissions compared to their marginal cost. For pollution costs above 19 cents per pound, the eastern low sulfur coal provides an additional value from reducing sulfur emissions more than sufficient to offset the 33 percent premium in fuel costs, which is reflected in the cost of electrical generation.
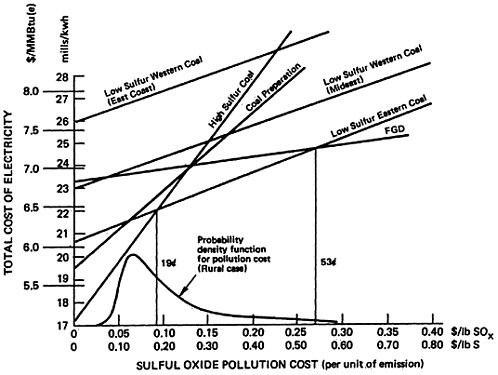
As pollution costs are raised still higher, the flue gas desulfurization (FGD) alternative becomes the best alternative. Although it is the most expensive (excepting low sulfur western coal) the FGD alternative permits overall emissions reductions approaching 90 percent wheras low sulfur eastern coal gives only a 70 percent reduction. The added cost for the additional sulfur removal may be substantial, so that the crossover points where FGD drops below low sulfur eastern coal are high: 53 cents per pound of sulfur for the retrofit non-urban plant
(Figure 13–3), 37 cents for the new non-urban plant (Figure 13–4), and 59 cents for the retrofit urban plant (Figure 13–5). The latter case includes a cost of 0.9 mills/kwh to sludge disposal, which corresponds to about $4/ton of sludge removed. At this cost level recycling FGD systems such as Wellman Lord or magnesium oxide should be almost competitive. However, there is less experience in applying these systems to high sulfur coal than is the case for lime scrubbing.
The economics of sludge disposal or material recycling may be a dominant consideration for FGD installations in urban areas. Where sludge can be ponded, the sludge disposal costs are moderate, of the order of 0.15 to 1.0 mills/kwh (see Chapter 11). However, for existing urban or suburban power plant locations ponding of sludge may not be feasible because of the absence of a suitable site. If sludge must be disposed of by hauling it off, the costs are estimated to be much higher: in the range of 0.6 to 1.5 mills/kwh, or possibly even higher (Leigh Short, personal communication). We have taken 0.9 mills as a nominal value for the representative urban plant calculation. The sludge material is about two thirds water, even after settling, so the volume to be removed is of the order of 50–100 percent of the coal needed to fuel the plant. Given the environmental restrictions and aesthetic considerations that may apply in some urban sites, lime or limestone scrubbing may be more expensive because of sludge disposal. For these sites the use of low sulfur coal or other scrubbing technologies will be indicated.
Low sulfur eastern coal is available only in limited supply. For some power plants it may not be available except at a price premium well in excess of the 33 percent we have assumed in the calculations above, especially if large numbers of other plants have shifted from high sulfur to low sulfur coals. In this case the line on Figures 13–3, 13–4 and 13–5 for low sulfur eastern coal will be shifted up to where it lies above high sulfur coal, coal preparation, and FGD for all values of pollution cost. With low sulfur coal no longer a
competitive alternative the values of pollution cost at which flue gas desulfurization becomes advisable drop sharply. For an existing plant in a remote rural location, lime scrubbing is preferred to burning high sulfur coal if the pollution cost per pound of sulfur emitted is above 26 cents (Figure 13–3). If coal preparation is considered as the alternative to scrubbing, then the cost per pound of sulfur at which scrubbing becomes preferred is 27 cents. For a new plant (Figure 13–4) the value is substantially lower: above 23 cents per pound of sulfur removed lime scrubbing will be the preferred alternative in the absence of available low sulfur eastern coal. Coal preparation is not quite marginally competitive: the marginal cost of sulfur removal for a new plant is about 27 cents per pound of sulfur removed, and this alternative permits only 33 percent sulfur removal (including the effect of energy losses), compared to about 89 percent for lime scrubbing. For plants located in mideastern as opposed to east coast locations, western low sulfur coal may be a competitive alternative if it is available at a cost somewhat lower than as assumed in Table 13–4.
Retrofit installations of flue gas desulfurization systems on oil burning plants in urban areas may involve higher costs for sludge disposal. If low sulfur coal is available, the pollution cost justifying lime scrubbing instead of low sulfur eastern coal is 59 cents per pound of sulfur removed. If low sulfur coal is not available, the crossover point drops to 29 cents per pound (where lime scrubbing is preferred to coal preparation) or 28 cents per pound if the gas desulfurization is compared to burning high sulfur coal (Figure 13–5).
The values of the crossover points are sensitive to the emissions levels and costs of electrcity given in Table 13–5. If different values are used the crossover points will change. The crossover between low sulfur coal and flue gas desulfurization is particularly sensitive; a change of 1 mill per kilowatt-hour in the cost of flue gas desulfurization chnages the crossover point by 23.2 cents for a new plant, and 19.8 cents for a retrofit
TABLE 13–5
Electricity Production Costs and Sulfur Emissions for Three Representative Power Plants
|
|
CASE |
|||||
|
ALTERNATIVE |
EXISTING COAL FIRED PLANT (non-urban) |
NEW COAL FIRED PLANT (non-urban) |
OLD COAL FIRED PLANT RE-CONVERTED FROM OIL TO COAL (urban) |
|||
|
|
Cost per unit of electricity (mills/kwh) |
Sulfur Emitted (×10−3 lbs/kwh) |
Cost per unit of electricity (mills/kwh) |
Sulfur Emitted (×10−3 lbs/kwh) |
Cost per unit of electricity (mills/kwh) |
Sulfur Emitted (×10−3 lbs/kwh) |
|
High sulfur coal with tall stacks and intermittent control |
17.2 |
25.9 |
21.6 |
22.5 |
17.5 |
25.9 |
|
Coal preparation |
19.4 |
17.2 |
23.6 |
15.0 |
19.7 |
17.2 |
|
Flue gas desulfurization |
23.3 |
2.75 |
26.2 |
2.4 |
23.9 |
2.75 |
|
Switch to low sulfur Western coal |
23.0 (mideastern) 26.0 (East Coast) |
6.9 |
25.5 (mideastern) 28.0 (East Coast) |
6.0 |
26.3 |
6.9 |
|
Switch to low sulfur Eastern coal |
20.6 |
7.8 |
24.6 |
6.7 |
20.9 |
7.8 |
|
Nuclear |
- |
- |
New Nuclear Plant 21.7 |
0 |
- |
- |
installation. If the comparison is between high sulfur coal and flue gas desulfurization, the sensitivity is rather low: a change of 1 mill per kilowatt-hour in the cost of flue gas desulfurization causes a change in the crossover point by 5.0 cents for a new plant and 4.3 cents for a retrofit installation. The crossover point between high sulfur and low sulfur coal changes by 6.3 cents for a 1 mill increase in the cost for low sulfur coal for a new plant, and 5.5 cents for an existing plant. These are both equivalent to 0.57 cents increase in the crossover point for a 1 cent per million BTU change in the price differential of low sulfur coal over high sulfur coal.
THE RELATION BETWEEN SULFUR OXIDE EMISSIONS FROM A SINGLE POWER PLANT AND AMBIENT INCREASES IN SULFUR DIOXIDE AND SULFATE LEVELS
The relationship between the emissions of sulfur oxide and ambient levels of sulfur dioxide and sulfate is not well understood at present (see Part Two, Section 1). The major source of difficulty lies in the lack of knowledge on the oxidation rate of sulfur dioxide to sulfate. There are a variety of chemical mechanisms by which the oxidation reaction can occur. In clean air, photooxidation of sulfur dioxide may proceed rather slowly: smog chamber experiments indicate rates of the order of 0.1 to 0.2 percent per hour (Bufalini 1971). Impurities act to catalyze the oxidation reaction at a much faster rate: photo-induced radicals, soot or metallic oxide particulates, and various organics or oxidants may lead to oxidation rates of the order of 5–10 percent per hour. Rates of this magnitude have been calculated for observations of sulfate in the photochemical smog of the Los Angeles Basin (Roberts and Friedlander 1974), and similar rates are consistent with recent airborne measurements of sulfur oxides in England (Smith and Jeffrey 1975). Rates in excess of 5 percent per hour
have been observed in direct measurements of sulfate levels in plumes from electric power plant and smelter plumes (Gartrell 1963, Weber 1970, Newman et al. 1975a). However, recent measurements of a power plant plume from a coal fired plant with an efficient electrostatic precepitation system (99.5 percent by weight particulate removal) indicate a lower rate, below 5 percent cumulative oxidation during the period (about two hours) for which the plume could be observed before sulfate levels were too close to background for accurate measurement (Newman 1975b).12
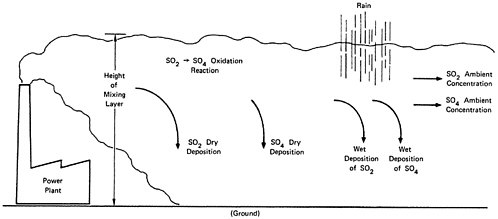
Sulfur dioxide may leave the atmosphere by being adsorbed on vegetation, soil, or water before it is oxidized to sulfate. The rate of sulfur dioxide removal has been extensively studied in England. The rate of removal of a volume of gas by a surface area is generally expressed as a deposition velocity (volume per unit time divided by area has the dimensions of velocity). A deposition velocity of 0.8 cm/sec was judged appropriate as an average value for the English countryside (Garland 1974). The deposition rate for removal of sulfates (in the absence of precipitation) has also been studied, but there is less agreement for this figure. The OECD study (OECD 1974) is using 0.4 cm/sec for sulfate deposition, although the other data indicates that this value may be high.13
Precipitation is relatively efficient in removing sulfate particles from the air. There is evidence that high relative humidities that accompany precipitation increase the oxidation rate of sulfur dioxide to form sulfate, and some sulfur dioxide is removed directly by being absorbed into the water droplets. European data indicate that rain will remove nearly all the sulfur oxide from the air within 60–100/km of the point of emission (Miller and DePena 1973, Hogstrom 1973a,b; Brosset 1973). The resulting precipitation is highly acidic, and this acid rain may have deleterious effects on vegetation, fisheries, and materials (see Chapter 7).
To carry through our analysis we need to assess the effect of emissions from a power plant in changing the ambient levels of sulfur dioxide and sulfates in downwind areas. Our
concern is with average annual levels, not peak concentrations, and with large areas relatively remote from the plant location. Motivation for this emphasis comes from the health and material damage assessments to follow: It is believed that very low level increases in ambient sulfate levels may cause significant health effects and damage to material property.
In the absence of available quantitative relationships between sulfur oxide emissions and ambient sulfate levels, we have developed a highly simplistic model for the emissions to ambient relationship14. The details of this model are given in Appendix 13-A. The model is not intended as a predictive device, but rather a summary of the limited available understanding of the relationships involved. An important purpose of the model is to clarify the need for further data and research on the emissions to ambient relationships. For decisions on emissions control alternatives for specific power plants, this model is clearly inadequate. Data on sulfur dioxide and sulfate levels and detailed information on regional meterology could be acquired at a modest cost and this information should be used as the basis for a much more extensive analysis than we have been able to carry out in this study.
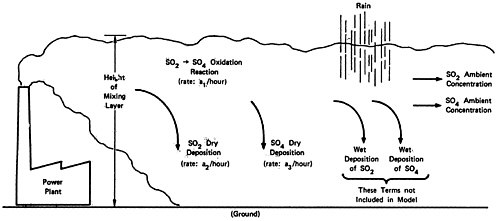
The model is based on the following assumptions:
In a period shortly following the emission, the sulfur oxides become uniformly distributed from the ground to a mixing layer height. The height of the plume then remains constant.
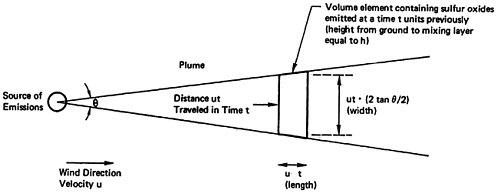
The emissions are uniformly distributed over an arc of constant size, so that the width of the plume expands in direct proportion to the time since emission, or (with constant wind velocity) in direct proportion to the distance downwind traveled by the plume.
We are assuming that the emissions travel down the plume uniformly distributed in a “box” whose length is the distance traveled by the wind per unit time15, whose height is the height of the mixing layer, and whose width is the distance perpendicular to the wind direction subtended by an angle of constant size. Thus, the width of the box grows in direct proportion
to time, and the concentration of pollutants decreases inversely with time.
We make the following assumptions about the chemical reactions of the sulfur oxides.
Sulfur dioxide to sulfate oxidation takes place according to a first order rate reaction. The rate may change considerably between comparatively clean rural air and pollutant-laiden urban air.
Sulfur dioxide is removed to the ground at a constant deposition velocity, beginning at the time of emission. (This assumption will overestimate the amount of sulfur dioxide removal to ground for a plume from a tall stack that travels many miles before touching ground, and it will underestimate the sulfur dioxide removal from a shorter stack where the plume is in contact with the ground for substantial time before the plume is dispersed uniformly up to the inversion or mixing layer height).
Suspended sulfate is removed to the ground at a constant deposition velocity, beginning with the time of emission.
Precipitation removal can be ignored in calculating reaction rates. In rainy weather it is reasonable to assume that virtually all of the ambient sulfate is removed over a distance of the order of 100 km. We shall assume later that a fraction of the sulfate is removed by precipitation between the time of its formation and the time it reaches the area when ambient levels are to be measured.
The relationships in the model are shown in Figures 13–6 and 13–7. Figure 13–6 indicates the chemical changes and removal mechanisms involved and Figure 13–7 shows the geometry assumed.
We shall use the model to examine two representative situations:
-
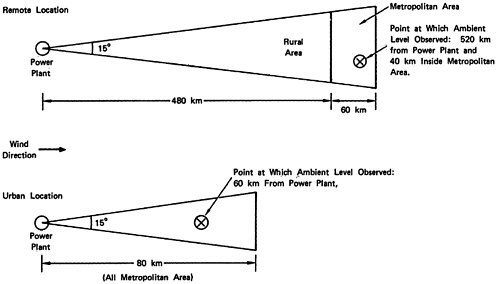
a plant located in a remote rural area, approximately 500 km upwind of a major metropolitan area,
-
a plant located in a metropolitan area, with urban settlement extending 40–80 km downwind from the plant.
We shall use the first to examine an existing coal fired plant and a new coal fired plant in a remote location, and the second to
examine an existing plant in an urban location that could be reconverted from oil to coal.
We use the following values as inputs to the calculation:
-
The wind is a constant 20 kilometers/ hour.
-
The angle subtended by the plume is 15°.
-
The height of the mixing layer above ground is 1,000 meters.
-
The depostion velocity of sulfur dioxide to the ground is 0.8 cm/sec, giving a removal rate of 2.88 percent per hour with a mixing layer height of 1,000 meters above ground.
-
The deposition velocity of SO4 to the ground is 0.4 cm/sec, giving a removal rate of 1.44 percent per hour with a mixing layer height of 1,000 meters.
-
The oxidation rate of sulfur dioxide to form sulfates is 0.5 percent per hour in rural air outside of a metropolitan area, and 5.0 percent when the air has passed over a metropolitan area and contains particulates, oxidants, and hydrocarbons from urban emission sources.
-
All sulfur oxides emitted from the power plant are emitted as sulfur dioxide rather than as sulfates. Assuming 1 to 2 percent of SO4 is emitted as sulfate, the error introduced by this approximation is negligible.
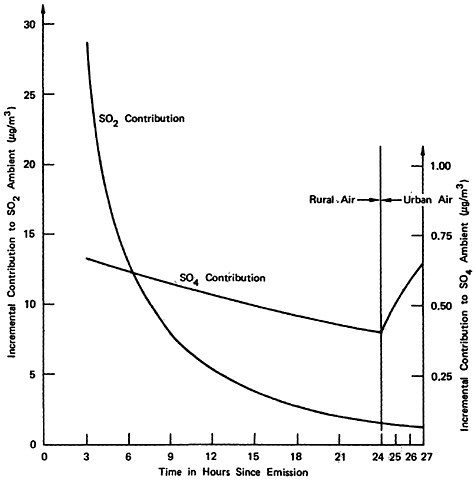
We now examine the solution obtained for these numbers. (The equations will be found in Appendix 13-A.) First, let us examine the case of a remotely located power plant of about 600 MW, burning 3 percent sulfur coal. Its average emission level (including the effect of plant loading) is computed to be 104 kilograms of sulfur dioxide per hour. Table 13–6 shows the marginal addition to the ambient concentrations of sulfur dioxide and sulfate from this plant, assuming that the pollution does not encounter pollutant laden metropolitan air (oxidation rate of SO2 to SO4:0.5 percent/hour).
TABLE 13–6
Incremental Contributions to Ambient Levels of Sulfur Dioxide and Sulfate Resulting from the Emissions of a Single 600 MW Power Plant, Rural Air (Oxidation Rate of 0.5 percent per hour Assumed).
|
Time, hours since emission |
3 |
6 |
12 |
18 |
24 |
50 |
|
Distance from plant, km |
60 |
120 |
240 |
360 |
480 |
1000 |
|
Increase in SO2 concentration (μg/m3) |
28.6 |
12.9 |
5.27 |
2.87 |
1.76 |
0.35 |
|
Increase in SO4 concentration (μg/m3) |
0.66 |
0.62 |
0.53 |
0.46 |
0.40 |
0.22 |
Now we repeat the calculation assuming that the emissions occur in pollutant laden urban air, for which an oxidation rate of 5.0 percent per hour is assumed (see Table 13–7).
We now give the results of calculations for the representative situations. First, let us examine the situation in which the power plant is remotely located, but the emissions encounter polluted urban air in a metropolitan area 480 kilometers (300 miles) downwind after 24 hours. At this time we assume the oxidation rate changes from 0.5 percent to 5 percent per hour. We compute the incremental contributions to ambient sulfur dioxide and sulfate levels as shown in Table 13–8 and plotted in Figure 13–8.
A detailed assessment of health, materials, damage, and other consequences would include the spatial variation in the ambient levels of sulfur dioxide and sulfate. We shall avoid this level of detail and use representative single values for the incremental contribution to ambient sulfur dioxide and sulfate levels resulting from the emissions of the power plant. For the remotely located plant, we take as representative for computing pollution consequences the values after two hours of oxidation in urban air (following 24 hours in rural air to give a total of 26 hours since emission from the power plant). For the urban plant, we take as representative the values three hours after emission, assuming, assuming oxidation in urban air during this time (see Table 13–9).
Of the effect of changes in the inputs, the most uncertain is the oxidation rate. A sensitivity analysis is given in Table 13–10.
The dominant effect of the oxidation rates in determining the ambient sulfate concentration shows up strongly in Table 13–10: Depending on whether we use high or low values, we get about an eightfold change in the contribution to ambient sulfate levels from the power plant. Note we would have even higher sulfate levels from the remotely located plant if we had used a uniform 2 percent per hour oxidation rate for both urban and rural air. (A forthcoming paper by Eliassen and Saltbones [1975] uses a trajectory model with conversion assumptions similar to ours to examine sulfate observations
TABLE 13–7
Incremental Contributions to Ambient Levels of Sulfur Dioxide and Sulfate Resulting from the Emissions of a Single 600 MW Power Plant, Urban Air (Oxidation Rate of 5.0 percent per Hour Assumed).
|
Time, hours since emission |
3 |
6 |
12 |
18 |
24 |
50 |
|
Distance from plant, km |
60 |
120 |
240 |
360 |
480 |
1000 |
|
Increase in SO2 concentration (μg/m3) |
25.0 |
9.9 |
3.1 |
1.3 |
0.6 |
0.04 |
|
Increase in SO4 concentration (μg/m3) |
6.2 |
5.4 |
4.2 |
3.3 |
2.6 |
1.03 |
TABLE 13–8
Incremental Contributions to Ambient levels of SO2 and Sulfate from the Emissions of a Single Power Plant: Representative Calculation for 600 MW Plant 300 miles Upwind of Urban Area.
|
Time, hours since emission |
3 |
6 |
12 |
18 |
24 |
25 |
26 |
27 |
|
Distance from plant, km |
60 |
120 |
240 |
360 |
480 |
500 |
520 |
540 |
|
Oxidation rate, % per hour |
0.5 |
0.5 |
0.5 |
0.5 |
5.0 |
5.0 |
5.0 |
5.0 |
|
Increase in SO2 concentration (μg/m3) |
28.6 |
12.9 |
5.27 |
2.87 |
1.76 |
1.56 |
1.39 |
1.23 |
|
Increase in SO4 concentration (μg/m3) |
0.66 |
0.62 |
0.53 |
0.46 |
0.40 |
0.50 |
0.58 |
0.65 |
TABLE 13–9 Emissions to Ambient Calculation for Representatives Power Plants (Emissions Rate Assumed is 104 Kilograms of Sulfur Dioxide per Hour)
|
|
Increase in Ambient Concentration, μg/m3 |
Location of Measurement |
Oxidation Rate Assumption |
|
|
Case |
SO2 |
Sulfate |
||
|
Remotely located plant (Existing or New Plant) |
1.39 |
0.58 |
Urban Area 26 Hours (540 km) Downwind |
24 hours at 0.5%/hr. then 2 hours at 5%/hr. |
|
Urban Plant (Existing) |
25.0 |
6.2 |
Urban area 3 hours (60 km) Downwind |
3 hours at 5%/hr. |
TABLE 13–10
Sensitivity Analysis, Emissions to Ambient Relationship
|
|
Remote Plant, 480 Km from (after 26 hours) |
Urban Plant (after 3 hours) |
||
|
SO2 conc, μg/m3 |
SO4 conc, μg/m3 |
SO2 conc, μg/m3 |
SO4 conc, μg/m3 |
|
|
Nominal Values |
1.39 |
0.58 |
25.0 |
6.20 |
|
Low Oxidation Rate |
|
|||
|
0.1% per hour in rural air 1.0% per hour in urban air |
1.6 |
0.13 |
28.2 |
1.32 |
|
High Oxidation Rate |
|
|||
|
1.0% per hour in rural air 10.0% per hour in urban air |
1.1 |
1.06 |
21.5 |
11.6 |
|
With Constant 2% Oxidation Rate |
1.03 |
1.30 |
27.3 |
2.59 |
|
SO2 Deposition Rate |
|
|||
|
Low: 0.4 cm/sec |
2.0 |
0.74 |
26.1 |
6.33 |
|
High: 1.6 cm/sec |
0.7 |
0.38 |
22.9 |
5.95 |
in Northern Europe and to compute rates for sulfur dioxide and SO4 oxidation. The mean of their computed rate values is 0.8 percent per hour. This rate gives essentially the same answer for the incremental increase in ambient sulfate as our representative rural plant calcuation using a rate of 0.5 percent for 24 hours and 5 percent for two hours.)
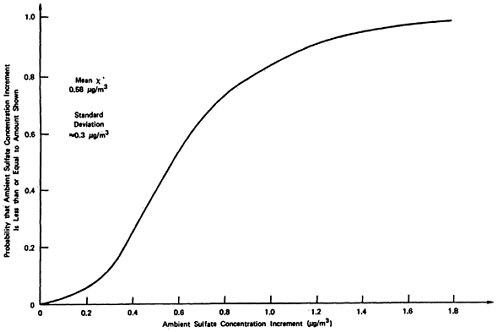
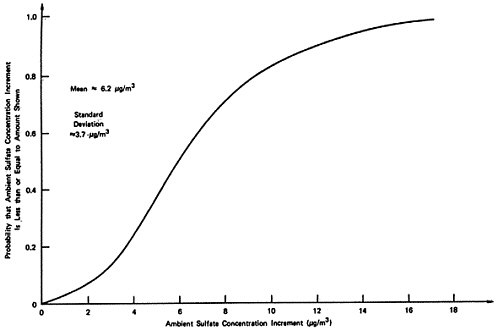
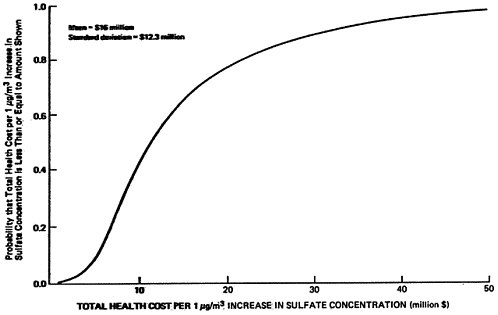
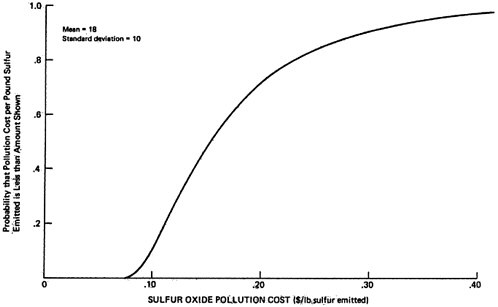
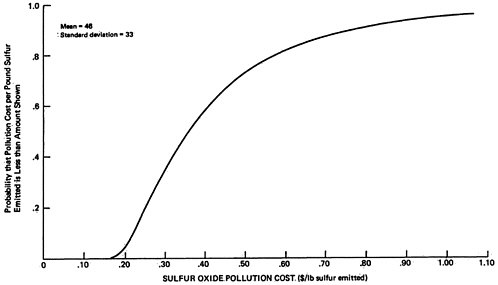
The sensitivity values shown in Table 13–10 have been chosen rather subjectively by the authors as representing a set of reasonable extreme values. As a rough approximation for assessing the uncertainties involved, we will assume that each set represents approximately the 5 percent and 95 percent points on a cumulative probability distribution assigned to the quantity (e.g., the probability is judged to be 90 percent that the uncertain quantity would lie in the interval between the low and high values used in the sensitivity analysis, rather than outside the interval). In addition, uncertainties are assumed independent, except for the rural and urban oxidation rates which are assumed totally dependent (e.g., if one is high then the other will be high also, and visa versa). A sketch of the resulting probability distribution on ambient sulfate levels is given in Figures 13–9 and 13–10. These curves are meant to illustrate the great uncertainty on the incremental change in ambient sulfate levels resulting from the emissions from a power plant located upwind from an urban area in which there are substantial health and material property values at risk. More refined models, formal probability assessment procedures, and formal probability processing could be used to improve the degree to which these curves summarize present knowledge on the emissions to ambient relationship for sulfates. The curves shown may be taken as rough summaries of the present state of knowledge, and they are subject to revision as further information is obtained.
RECONCILING THE MODEL FOR AMBIENT INCREASES FROM A SINGLE PLANT TO EMISSIONS DATA AND AMBIENT SULFUR OXIDE LEVELS FOR THE NORTHEASTERN UNITED STATES
The calculations in the last section give us an indication of the relationship that may exist between sulfur oxide emissions from a single power plant and the ambient levels of sulfur dioxide and sulfate in downwind areas. The relationship must be regarded as tenuous, and subject to substantial change when regional models of sulfur oxide transport become available. Nonetheless, the objective set forth for this portion of the report is to carry out an analysis of alternative strategies based on the limited information presently available. We will therefore attempt to use the preceding calculations, after suitably modifying them to account for observed sulfate levels, as a basis for assessing the social cost per pound of sulfur emitted. The framework for this calculation is easily adapted to improved models relating power plant emissions to observed ambient sulfate levels, where such improved models are developed. The model used in the last section (and described in more detail in Appendix 13-A) is essentially a “back-of-the-envelope” calculation carried out by the authors when we were apprised that there was no model for long range sulfur oxide transport available for our use in a cost-benefit analysis. We believe that an essential step in a cost benefit analysis of alternative abatement strategies must be a quantitative relationship between the emission level and the pollutant concentrations at the location where the pollutants cause adverse consequences.
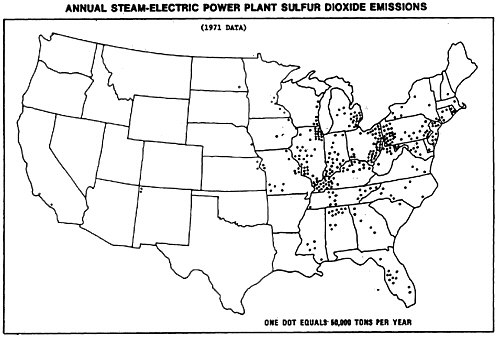
In order to assess the degree that our representative cases of the previous section were representative of the power plants presently emitting sulfur oxide, we compiled the map shown in Figure 13–10. The data on power plant emissions were obtained from Federal Power Commission records for 1972 and dots were placed in the Air Quality Regions corresponding to plant locations. Each dot corresponds to power plant emissions of 50,000 tons of sulfur dioxide per year. For the representative plant calculations of the last section we assumed emissions of 10,000 kilograms of sulfur dioxide per hour, or about 96,500 tons of sulfur dioxide per year. This quantity corresponds to just
less than two dots on the map. One dot is quivalent then to about 300 MW of capacity burning 3 percent sulfur coal. Lists of large power plants burning coal and oil fired plants that might be reconverted to burn coal are given in Appendix 13-B.
Let us examine the map of Figure 13–10a. We note a large concentration (about 70 dots) in Eastern Ohio—Western Pennsylvania—Maryland—West Virginia area. This amount corresponds to more than 10 percent of the total sulfur oxide emissions in the nation. Another large concentration is found in the Southern Illinois-Indiana-Kentucky area.
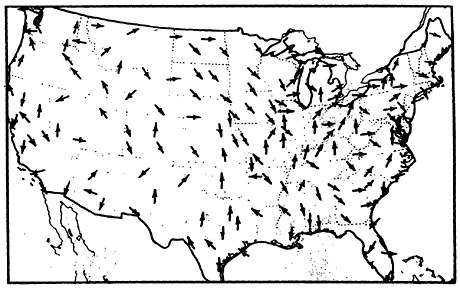
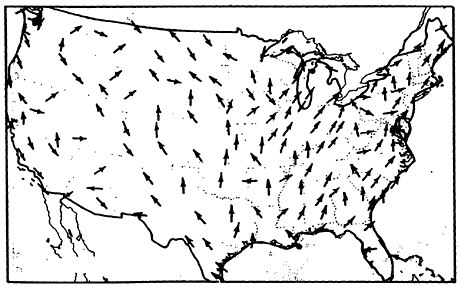
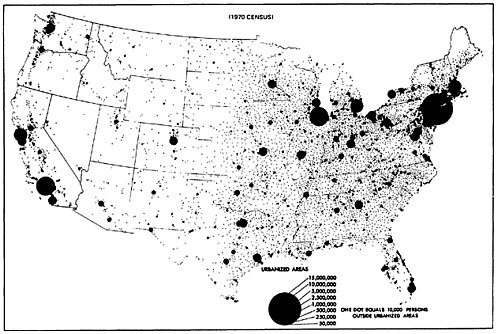
A look at the surface wind maps of Figures 13–11 and 13–12 indicates the direction of the wind, and Figure 13–13 shows us the location of the population concentrations. The implication of these maps is grim: The sulfur oxides emitted by these rural power plants tend to blow into the populous urban Northeast corridor.
Let us focus our attention on those 70 dots that represent the concentration of power plants in the Pennsylvania-Ohio border area about 500 km upwind of Metropolitan New York. Recall that for the representative rural plant we calculated a sulfate concentration of 0.66 ug/m3 (after 3 hours or 60 km), 0.40 ug/m3 (after 24 hours or 480 km) and then an increase to 0.58 ug/m3 after 2 hours exposure to urban air. The observed data of sulfate levels for rural locations in Pennsylvania and Virginia is an annual average level of about 9 ug/m3 (see Appendix 13-C). In these locations there are no local sources of sulfur oxide emissions, and observed sulfur dioxide levels are very low, of the order of ten micrograms per cubic meter. The average annual sulfate levels observed in greater New York are in the range of 12–20 ug/m3. While 12.5 ug/m3 is the average for urban areas in the Eastern United States, the urban areas of New York, Pennsylvania, New Jersey and New England are somewhat higher, in the range of 16 ug/m3 (see Appendix 13-C).
We must reconcile these observations with the ambient increases computed from the model in the last section. To do this, two additional factors must be considered that were not
incorporated in the model: (1) variations in wind direction, and (2) loss due to wet deposition of sulfur dioxide and sulfate.
Whereas 15° may be an appropriate value for the angle subtended by the plume on a particular day, during the year the wind will vary in direction and the sulfur oxide emissions will be distributed over a much wider sector. We shall assume that during the year the variations in wind direction cause the sulfur oxide to be distributed over a sector of 45°, three times the 15° sector we assumed for the power plant plume. This will reduce average annual sulfate (and sulfur dioxide) ambient increments to one third the values shown in Table 13–9. To take account of wet deposition losses from rain, we will assume that 25 percent of the sulfur dioxide and sulfate is removed by precipitation prior to observation16. This will further reduce the average annual ambient increments by 25 percent. The average annual ambient increments contributed by a single representative plant would then be 1/4 the values shown in Table 13–9. The adjusted annual average increments are shown in Table 13–11.
The 70 dots in the Pennsylvania/Ohio Border-West Virginia area on the map, Figure 13–10, indicate the equivalent of 36 representative plants of 600 MW burning 3 percent sulfur coal. Using the values of Table 13–11 (and the sulfate value for 12 hours from Table 13–6 adjusted for precipitation and plume width) we would then expect to see an increase of 36×0.25×0.53= 4.8 ug/m3 in rural areas about half way to New York and an increase of 36×0.25×0.58=5.2 ug/m3 in the urban New York area. If we assume a background level of sulfates from other natural and remote man-made sources of 4–5 ug/m3 and a contribution from local sources near New York of about 3–10 ug/m3 of sulfate, we see that the numbers are in general agreement with observations. Obviously, the assumptions (1) that the sulfate resulting from emissions in the Pennsylvania/Ohio border-West Virginia area is distributed over a 45° sector, and (2) that 25 percent of the sulfate is removed by rain within 12 to 24 hours can and should be subjected to considerable refinement using meteorological
TABLE 13–11
Summary of Judgment for Average Annual Ambient Increases of Sulfur Oxide and Population at Risk Assumptions for Representative Power Plant Case Analyses
|
Representative Power Plant with Emissions of 104 kg of SO2/hour or 96,500 tons of SO2 per year |
Ambient Increase from Plant Emissions for a 15° Angle Plume, from Table 13–9 |
Average loss from Wet Deposition of Sulfur Oxides |
Average Annual Ambient Increases from Plant Emissions Assuming Distribution over a 45° Sector |
Population at Risk Assumed with 45° Sector for Representative Case |
||
|
|
SO2 (μg/m3) |
Sulfate (μg/m3) |
|
SO2 (μg/m3) |
Sulfate (μg/m3) |
|
|
Remote Location (Ambient Increase measured 26 hours or 520 km downwind) |
1.39 |
0.58 |
25% |
0.35 |
0.145 |
50 million |
|
Urban Location (Ambient Increase measured 3 hours or 60 km downwind) |
25.0 |
6.2 |
10% |
7.5 |
1.86 |
11.5 million |
information for the area on air trajectories and precipitation frequencies.
It may be useful to note the following implication for the numbers above: If sulfur oxide emissions from power plants on the Pennsylvania/Ohio border-West Virginia area were doubled, we would expect an increase in rural sulfate levels from 9 to 13.8 ug/m3, an increase of about 50 percent; and an increase in the urban sulfate level in the Boston-New York-Washington D.C. urban complex from about 16 ug/m3 to 21.2 ug/m3, an increase of about 30 percent. These numbers are of course extremely rough calculations based on many simplifying assumptions, not the least of which is the assumption of first order rate reactions. If oxidation is limited by the availability of pollutants that act as catalysts, then the increase in urban sulfates from a doubling of sulfur oxide emissions would be less. An overall estimate for the increase in urban sulfate in the Boston-New York-Washington urban corridor from doubling power plant emissions in the Ohio-Western Pennsylvania-West Virginia area might be taken to be in the range of 15–45 percent17.
Let us now consider the population at risk for a plant in the Pennsylvania/Ohio border West Virginia area. If we assume the emissions to be distributed over a 45° arc in the direction of the prevailing winds, the area affected will include the New England states. New York, Pennsylvania, New Jersey, Maryland, Delaware, and the District of Columbia. The combined population of these states in 54 million (1970 census) of which 47 million live in urban areas18. A substantial population in Southeastern Canada may also be affected by the emissions. We shall summarize these considerations by assuming a population at risk of 50 million persons impacted by the increase in sulfate levels from a representative rural plant located in the area approximately 500 km west of the Boston-Washington metropolitan corridor.
We will make similar assumptions for the urban location representative plant. Let us assume that on an average annual basis, the
emissions from the power plant are distributed through a 45° arc in the direction of the prevailing winds. Let us further assume that about 10 percent of the sulfate is removed by precipitation prior to impacting on the population at risk (since the time assumed between emission and measurement is 3 hours for this case instead of 26 hours, we would expect less loss from rain). The net computation of average annual ambient increments is equivalent to multiplying the values in Table 13–9 by 0.3: a reduction of a factor of 3 caused by variations in wind direction, and a further reduction of 10 percent caused by washout and rainout from precipitation. For the population impacted by the emissions from the urban location representative plant, we will rather arbitrarily take 11.5 million, the population of the greater New York Metropolitan area. The calculated values for SOx levels for the urban location case are summarized in Table 13–11.
ESTIMATED HEALTH COSTS OF ELEVATED AMBIENT LEVELS OF SULFUR OXIDES
A comprehensive evaluation of a pollution reduction strategy must compare the dollar costs of implementing that strategy with the health and other costs sacrificed by not implementing it. In terms of our present problem, this means that we must convert a given level of sulfur oxide pollution to a dollar health cost. Placing a dollar value on morbidity and mortality is a difficult task, one that we all, understandably, are reluctant to undertake. However, if the comparisons of monetary and health costs are not made explicitly in the analysis, they will be made implicitly elsewhere. The importance of the assessment of the health effects of sulfur oxide emissions dictates their explicit consideration in our analysis (see Part One).
Our discussion in this section will consist of (1) a review of some of the health effects which appear to be associated with sulfur oxide air pollution, (2) an illustration of the sort of quantitative model which is
needed for the evaluation of pollution reduction strategies, (3) an application of that model which will indicate the sensitivity of health effects to changes in the level of sulfate pollution, and (4) a method for converting specific health effects to aggregated health costs for society.
The Relationship Between Adverse Health Effects and Ambient Levels of Sulfur Oxides
In order to choose logically among sulfur oxide pollution reduction strategies we need to have some understanding of the dose response relationship between adverse health effects and ambient levels of sulfur oxides. Unfortunately, this relationship is not accurately known at present.
In general, epidemiological studies have indicated an association between ambient sulfur dioxide levels and adverse human health effects, yet controlled human exposure studies show that considerably higher than existing sulfur dioxide levels are incapable of producing an acute response in man. The apparent paradox is explainable by the possible toxicity of sulfur dioxide by-products present in polluted air. This contention is supported by recent studies carried out as part of the Community Health and Environmental Surveillance System (CHESS 1974) which indicated that health effects seem to be more closely associated with exposure to suspended atmospheric sulfate than with exposure to other pollutants.
The mechanisms by which sulfur oxides and suspended particulate matter may act to impair health are not well understood. The fine sulfate particles, particulrly acid sulfate aerosols, are believed to irritate the membranes lining the respiratory tract, making breathing more difficult and impeding the normal protective and self-cleansing mechanisms of the lung. There is some data indicating that the bronchoconstricting potency of particulate sulfates is related both to physical properties of the particles, such as size19 and to chemical
properties, such as pH (Lewis et al. 1972, Amdur 1969). The biological reactivity of the particles also seems to depend on such atmospheric conditions as humidity and temperature and there may be important synergistic effects with other atmospheric agents (McJilton and Frank 1973). The most effective control strategy for sulfur oxides will most likely depend critically on the exact nature of the sulfate-biologic response relationship in man. Therefore, it would appear that there is a very high value to clarifying these biological issues before any substantial resource commitment is made to a particular method for atmospheric ambient level sulfate control.
Adverse Health Effects Attributable to Elevated Levels of Atmospheric Suspended Sulfates.
The stated objective of the epidemiologic studies of the Environmental Protection Agency’s Community Health and Environmental Surveillance System (CHESS) program is to establish data to aid the development of dose-response relationships between short-term and long term pollutant exposures and adverse health effects. As stated above, more research is required before any definite quantitative relationship may be established. However, progress is being made in this area, and it is useful to demonstrate the manner in which dose-response curves should be utilized in the context of the sulfur oxide pollution control decision problem. In the next section a hill present possible forms for the dose-response curves for some of the pollutant-disease associations that have been observed in recent epidemiological studies. A description and assessment of studies linking various adverse health effects with sulfur oxide pollution appears in Chapter 4.
Short term exposures to elevated levels of sulfur oxides, especially acid-sulfate aerosols, seem to aggravate asthma and pre-existing heart and lung disorders. Results of a study of asthmatics in the New York area showed higher rates of asthmatic attacks, which are
characterized by marked hyperconstriction of the trachea and bronchi leading to sneezing and gasping for breath, on days with the highest suspended sulfate levels. The average threshold for observed aggravation by sulfates was calculated as 7.3 ug/m3 on warmer days with a minimum temperature above 50 F and 11.9 ug/m3 on warmer days with a minimum temperature lying between 30 and 50 F (Finklea et al. 1974). A study of cardiopulmonary patients in the New York Metropolitan area found elevated levels of suspended sulfates to be consistently associated with symptom aggravation, particularly shortness of breath, cough, and increased production of phlegm (Goldberg et al. 1974).
Elevated short term exposures to acid sulfate aerosols have frequently been shown to accompany perceptable increases in daily mortality. Older individuals with pre-existing heart and lung disease are particularly at risk. However, it is not known whether severe episoede of air pollution tend to shorten only the life span of the severely ill, or if the life span of a much larger group of the population is affected (see the section on chronic bronchitis and emphysema in Part One).
Epidemiological studies appear, in general, to indicate that repeated short term peak exposures or annual average elevations of suspended sulfates accompany excess acute lower respiratory disease in children, excess risk for chronic respiratory disease symptoms in adults, and decreased ventilatory function in children. The Salt Lake Basin studies of the CHESS program indicated, for example, an increased incidence of lower respiratory infection, croup, and bronchitis in children living in communities with higher annual average levels of suspended sulfates. Similarly, chronic bronchitis, which was characterized by the presence of cough and phlegm on most days for at least three months each year, was found to have a higher incidence rate among adults living in the more polluted communities (Finklea 1974, Galke 1974 a,b).
Dose-Response Relationship between Atmospheric Sulfate Level and Health Effects
Summarizing, epidemiological studies seem to indicate that short term exposures to elevated levels of sulfur oxides
-
aggravate pre-existing heart and lung disorders in elderly patients,
-
aggravate asthma,
-
perceptably increase daily mortality.
Repeated short-term exposures or elevations of annual average exposures seem to
-
increase the incidence of lower respiratory disease in children,
-
increase the risk for chronic respiratory disease in adults.
These effects were observed in community studies where levels of sulfur dioxide, acid sulfate aerosols, and suspended particulate matter were usually but not always simultaneously elevated. Accurate measurement of total suspended sulfate was in many cases not available. The data, therefore, is seriously limited by the lack of accurate dose information. Similar problems are involved in the evaluation to the response side of the dose response relationship. The studies often differ in their locale and their methods of ascertainment of illness. Different analytical and statistical approaches have been utilized to evaluate the available data, making it difficult to compare studies performed by different investigators.
Despite these difficulties, and in reconition of them, an attempt has been made at a preliminary quantification of these health effects in the form of dose-response curves20. As a crude approximation let us assume that each of the above health effects is characterized by a linear threshold relationship. The approximation is that no adverse health effect results below some threshold level of suspended sulfate concentration. Above this level, the percent of cases of the adverse health effect which may be attributed to the sulfate (the “percent excess” is assumed to increase linearly
with concentration. By plotting the results of various epidemiological studies on a graph relating the percent excess of health effects to levels of suspended sulfates, the “best fit” for such a linear-threshold relationship may be obtained. The results of such a curve fitting exercise and the studies upon which they were based are summarized in Table 13–12.
Obviously there is a good deal of arbitrariness in the definition of these curves. Consequently, the analysis that follows is meant more to illustrate method than to accurately quantify health costs. The uncertainty in the magnitude of the health effects from a given increase in ambient sulfate is of the order of a factor of 20, as will be described below.
Health Effects of Suspended Sulfates in the New York Metropolitan Area
Once dose-response curves have been agreed upon, they may be combined with expected frequency distributions of suspended sulfate concentrations to obtain an estimation of the health effects expected from an increase in the current ambient levels of suspended sulfates.
We illustrate the reasoning with an analysis of the health effects of suspended sulfates in the New York Metropolitan area. Total population for this area and the population of various age groups (of interest as populations at risk) are summarized on page 604.
TABLE 13–12 “Best Judgment” Dose-Response Functions.*
|
|
BEST JUDGMENT THRESHOLD FUNCTION |
||
|
ADVERSE HEALTH EFFECT |
EXPOSURE DURATION |
THRESHOLD (μg/m3) |
SLOPE** |
|
INCREASED DAILY MORTALITY |
24 HOURS OR LONGER |
25 |
.252 |
|
AGGRAVATION OF HEART AND LUNG DISEASE |
24 HOURS OR LONGER |
9 |
1.41 |
|
AGGRAVATION OF ASTHMA |
24 HOURS OR LONGER |
6 |
3.35 |
|
EXCESS LOWER RESPIRATORY DISEASE IN CHILDREN |
Up to 10 YEARS |
13 |
7.69 |
|
EXCESS RISK FOR CHRONIC RESP. DISEASE IN ADULTS*** |
Up to 10 YEARS |
12 |
11.1 |
|
*These dose response relationships were developed in an unpublished study for the U.S. Environmental Protection Agency. The “best judgment threshold functions” represent subjective approximations to data, not precise mathematical fits. The studies upon which the estimated were based are as follows: Mortality; Lindeberg (1968), Martin and Bradley (1960), Lawther (1963), Glasser and Greenburg (1965) Brasser et al. (1967), Watanabe and Kaneko (1971), Nose and Nose (1970) Buechley et al. (1973). Aggravation of heart and lung disease; Carnow et al. (1970), Goldberg et al. (1974). Aggravation of asthma; French, Sugita et al. (1970), Finklea et al. (1974a), Finklea et al. (1974c). Excess lower respiratory disease in children; Nelson et al. (1974) Finklea et al. (1974b), Douglas and Waller (1966), Lunn at al. (1967), Love et al. (1974), Hammer (1974). Excess chronic respiratory disease; Burn and Pemberton (1974), Goldberg et al. (1974), House et al. (1973), Hayes et al. (1974), Yashiso (1968), House (1974), Galke and House (1974a), Galke and House (1974b). **Change in percent excess over base rate for population, per μg/m3 change in suspended sulfate level. ***For chronic respiratory disease, difficulties with available data necessitated the unit of measurement to be excess risk rather than direct incidence of illness. Actually, in its originally calculated form, separate dose response functions were assessed for cigarette smokers and nonsmokers. The function described in the table is a weighted linear average based upon the average prevalence of cigarette smoking in the adult population at risk. |
|||
NEW YORK METROPOLITAN AREA20
-
Population 11.5 million
2.12 million children age 0–13
7.27 million adults
1.24 million elderly over age 65
Daily concentrations of ambient suspended sulfate concentrations are assumed to follow a lognormal frequency distribution with an annual average of 16 ug/m3 and a standard deviation of 5.6 ug/m3. This assumption is consistent with the New York studies of the CHESS report. The objective of the analysis will be to answer the following two questions:
-
What are the health effects of a 1 ug/m3 increase in the annual average level of suspended sulfate concentration?
-
Suppose the concentration for the top 1 percent peak days increases by 10 ug/m3.
What are the health effects?
The answers to these questions will tell us something about the sensitivity of the health effects to changes in levels of sulfate concentration. This information, in turn, should help us to evaluate the health impacts of various emission reduction alternatives.
For increased daily mortality, aggravation from heart and lung disease, and asthmatic attacks, which are affected by daily levels of suspended sulfates, the expected effect of 1 ug/m3 increase in sulfate levels may be estimated by calculating the expected number of cases of each health effect under current ambient concentrations and under current ambient levels plus 1 ug/m3 sulfate concentration. More precisely, if f (x;m, σ) represents the (normalized) frequency distribution21 for 24 hour sulfate concentrations and y(x) is the dose-resonse curve, the additional percent of adverse health cases due the unit increase in sulfate concentrations is given by
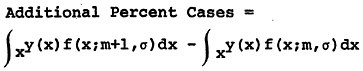
Lower respiratory disease in children and chronic respiratory disease in adults are more closely related to yearly average levels and so the percent excess cases may be read directly from the annual average dose-response curves. Comparing, category by category, the percent excess cases with the total number of observed cases, estimates of the number of cases attributable to the suspended sulfate concentration are obtained.
The results of such calculations are summarized in Table 13–13. Since annual average concentrations may differ from 16 ug/m3, depending upon exact location, the calculations were also performed assuming annual average concentrations of 12.5 and 20 ug/m3. The last column of Table 13–13 gives the expected number of additional cases resulting from a 1 ug/m3 annual average sulfate concentration increase.
The effects of an increase of 10 ug/m3 in the top 1 percent days of peak concentration may be obtained by altering the shape of the 24 hour sulfate concentration frequency distribution. Specifically, shifting the upper 1 percent “tail” of the distribution by 10 ug/m3 will give us the desired mathematical expression of the effect. The results are given in Table 13–14, the last column of which again gives the additional number of cases to be expected from the concentration increase. Of the five health effects, only premature mortality, heart and lung disease aggravation, and asthmatic attacks are significantly affected by short term elevations of sulfate concentrations. Due to the assumed linearity of the dose-response curves above threshhold levels, these entries are independent of average concentration. However, for high levels of concentrations, nonlinearities become increasingly important and may imply significant deviations from these values. Nevertheless, the indication is clear that, with the exception of premature mortality, a 1 ug/m3 average increase is considerably more significant in terms of average health effects than is an occasional 10 ug/m3 increase on high pollution days. This result is of interest for the comparison of control measures which are directed to annual average concentration levels
TABLE 13–13
Estimates of Adverse Health Effects Attributable to a 1 μg/m3 Increase of Average Suspended Sulfates in the New York Metropolitan Area
|
HEALTH EFFECT |
AVERAGE NUMBER OF CASES* (MILLION) |
CASES ATTRIBUTABLE TO SUSPENDED SO4 CONCENTRATION |
|||||
|
AT AVERAGE CONCENTRATION |
AT AVERAGE CONCENTRATION +1 μg/m3 |
||||||
|
μg/m3 |
PERCENT OF CASES |
NUMBER OF CASES |
PERCENT OF CASES |
NUMBER OF CASES |
ADDITIONAL EXPECTED CASES |
||
|
INCREASED DAILY MORTALITY (premature deaths per yr.) |
|
12.5 |
.0418 |
49.4 |
.0512 |
60.5 |
11.1 |
|
.118 million Deaths/year |
16.0 |
.0839 |
99.2 |
.1031 |
121.9 |
22.7 |
|
|
|
20.0 |
.1919 |
226.9 |
.2350 |
277.9 |
51.0 |
|
|
AGGRAVATION FROM HEART AND LUNG DISEASE (million person-days per yr.) |
|
12.5 |
5.829 |
1.422 |
6.902 |
1.684 |
.262 |
|
24.4 |
16.0 |
9.907 |
2.417 |
11.569 |
2.823 |
.406 |
|
|
|
20.0 |
15.137 |
3.693 |
16.903 |
4.124 |
.431 |
|
|
|
12.5 |
22.29 |
.5595 |
25.50 |
.6401 |
.0806 |
|
|
ASTHMATIC ATTACKS (millions per year) |
2.51 |
16.0 |
33.48 |
.8404 |
36.83 |
.9245 |
.0841 |
|
|
20.0 |
46.88 |
1.1768 |
50.23 |
1.2609 |
.0841 |
|
|
LOWER RESPIRATORY DISEASE IN CHILDREN (thousands of cases per year) |
|
12.5 |
0 |
0 |
3.815 |
4.85 |
4.85 |
|
127 |
16.0 |
23.04 |
29.2 |
30.73 |
39.10 |
9.90 |
|
|
|
20.0 |
53.80 |
68.5 |
61.49 |
78.20 |
9.90 |
|
|
CHRONIC RESPIRATORY DISEASE SYMPTOMS (millions of cases, point prevalence) |
|
12.5 |
7.75 |
.0284 |
18.85 |
.0690 |
.0406 |
|
.366 |
16.0 |
46.60 |
.1707 |
57.70 |
.2113 |
.0406 |
|
|
|
20.0 |
91.00 |
.3333 |
102.10 |
.3739 |
.0406 |
|
TABLE 13–14
Estimates of Adverse Health Effects of a 10 μg/m3 Increase in Suspended Sulfate on the Top 1 percent Peak Concentration Days
|
HEALTH EFFECT |
AVERAGE NUMBER OF CASES* |
CASES ATTRIBUTABLE TO SO4 WITH INCREASE |
ADDITIONAL CASES DUE TO INCREASE |
|
|
PERCENT OF CASES |
NUMBER OF CASES |
|||
|
INCREASED DAILY MORTALITY (Premature deaths total per year) |
.118 million deaths a year |
.0483 |
57.2 |
7.70 |
|
AGGRAVATION FROM HEART AND LUNG DISEASE (Million man-days per year) |
24.4 |
5.865 |
1.431 |
.00895 |
|
ASTHMATIC ATTACKS (Millions per year) |
2.51 |
22.38 |
.5617 |
.000439 |
|
*See footnote for Table 13–13. |
||||
versus intermittent controls which are designed only to reduce sulfate concentrations on days of extremely high ambient levels.
Estimation of Health Costs
To complete our estimation of the health costs resulting from increases in the ambient levels of suspended sulfates in the New York Metropolitan area we need to assign estimates of the costs to society appropriate to each incidence of an adverse health effect. The nominal dollar values which we have chosen to employ are summarized below.
ESTIMATION OF PER CASE HEALTH COSTS
|
One Premature Death |
= |
$30,000 |
|
One Day Aggravation From Heart and Lung Disease Symptoms |
= |
$20 |
|
One Asthma Attack |
= |
$10 |
|
One Case Child’s Lower Respiratory Disease |
= |
$75 |
|
One Case Chronic Respiratory Disease |
= |
$250 |
Ideally, these values should be assigned so as to reflect what society would (or should) just be willing to pay in dollars to prevent the losses sustained when a typical individual suffers one of the possible health effects (see Lave and Seskin 1970). We believe that medical cost and lost productivity should not be the only basis for assessing these values, as was done in their paper). Since there may be considerable disagreement on what the dollar assignments should be, we will need to check the sensitivity of our results to these values.
Although expressing health losses in terms of the dollar is convenient for our purposes,
other common units of measurement are possible. One other popular numeraire for estimating health damages is the “equivalent day of restricted activity (EDRA)” (Jacoby et al. 1973:189). The Public Health Service defines a day of restricted activity as one on which a person substantially reduces the amount of activity normal for that day because of a specific illness or injury (PHS 1971:53). If we assume a person-day of restricted activity to imply health costs of between $1 and $10 (Jacoby et al. 1973, NAS 1974: volume 4), then our nominal health costs are equivalent to the following assessment of EDRA’s:
|
one day aggravation from heart and lung disease |
= |
2–20 EDRA’s |
|
one asthma attack |
= |
1–10 EDRA’s |
|
one case child’s lower respiratory disease |
= |
7.5–75 EDRA’s |
|
one case chronic respiratory disease |
= |
25–250 EDRA’s |
These ranges compare well with the estimations of Jacoby et al. (1973: Table 8–4). For premature death we have used a value of $30,000. Most of the deaths occur among chronically ill elderly people, and the amount by which their lives are reduced may be only a matter of days or weeks. It is not known whether sulfate levels have any correlation with a general reduction in life expectancy for persons of average age and health. Rather, the effect observed is a statistical increase in number of deaths recorded on days of high pollution levels versus days of low pollution levels. For this reason we have used a value of $30,000 per life rather than the value of $200,000 used in highway safety and other applications for the value of life for a representative individual in the population (NAS 1974). Sensitivity analysis indicates that our results are not dependent on the value assigned to premature death.
Multiplication of per-case health costs times the number of cases given in Table 13–13 yields the societal health costs resulting from various levels of suspended sulfate concentration. Our sample values yield the health costs shown in Table 13–15. Observe that the major contribution to health costs from a unit increase in sulfate level appears to be due to
TABLE 13–15
Estimation of Health Costs for Various Levels of Ambient Suspended Sulfate (Population at Risk=11.5 million)
|
Health Effect |
Annual Average Ambient Sulfate Concentration, μg/m3 |
Health Effect, (number) |
Health Cost (million $) |
Additional Change in Sulfate Concentration Increased 1 μg/m3 |
|
|
|
effect (number) |
cost (million $1) |
|||
|
Excess Mortality (number of deaths) per year |
12.5 |
49 |
1.5 |
11 |
0.33 |
|
16.0 |
99 |
3.0 |
23 |
0.68 |
|
|
20.0 |
227 |
6.8 |
51 |
1.53 |
|
|
Aggravation of Heart-Lung Disease Symptoms (million person-days of aggravation per day) |
12.5 |
1.4 |
28.4 |
0.26 |
5.2 |
|
16.0 |
2.4 |
48.3 |
0.41 |
8.1 |
|
|
20.0 |
3.7 |
73.9 |
0.43 |
8.6 |
|
|
Asthmatic Attacks (thousands of attacks) per year |
12.5 |
560 |
5.6 |
80 |
0.81 |
|
16.0 |
840 |
8.4 |
84 |
0.84 |
|
|
20.0 |
1180 |
11.8 |
84 |
0.84 |
|
|
Lower Respiratory Disease in Children (thousands of cases per year) |
12.5 |
0 |
0 |
4.9 |
0.36 |
|
16.0 |
29 |
2.2 |
9.9 |
0.74 |
|
|
20.0 |
69 |
5.1 |
9.9 |
0.74 |
|
|
Chronic Respiratory Disease (thousands of cases, point prevalence) |
12.5 |
28.4 |
7.1 |
40.6 |
10.2 |
|
16.0 |
171 |
42.6 |
40.6 |
10.2 |
|
|
20.0 |
333 |
83.3 |
40.6 |
10.2 |
|
|
12.5 |
|
42.6 |
|
16.9 |
|
|
Total Cost |
16.0 |
|
104.5 |
|
20.5 |
|
20.0 |
|
180.9 |
|
21.9 |
|
the expected increase of 40,600 cases of added risk for chronic respiratory disease, valued at $250 per case, and 262 thousand additional person-days of aggravated heart lung disease symptoms, valued at $20 each.
Sensitivity of Health Costs
We now investigate the sensitivity of our estimate of the health costs of a 1 ug/m3 increase in average sulfate concentration to various assumptions used in the analysis. The calculations are in principle identical to those performed in the section on the relationship of sulfur dioxide emissions to ambient sulfate levels. Rather than fixing each quantity in the analysis at a nominal value, we vary that quantity over a range of possible values and observe the effect on the output value, which in this case is total health cost. The results of the analysis appear in Table 13–16.
The sensitivity values shown in Table 13–16 have been chosen somewhat arbitrarily as representing a set of reasonable extreme values. The low and high values for the change in incidence of each health effect, per unit change of ambient sulfate, were taken to be 10 percent and 200 percent of the nominal values respectively. This judgment was communicated to us by representatives from the Assembly of Life Sciences Committee that authored for Part one of this report.
We see from the sensitivity results that health costs of a 1 ug/m3 increase in ambient sulfate are most sensitive to (in order of magnitude) the estimated health cost per case of chronic respiratory disease, the health cost per day of aggravation from heart and lung disease symptoms, the number of additional cases of chronic respiratory disease, and the additional number of days of aggravation from heart and lung disease symptoms.
TABLE 13–16
Sensitivity Analysis: Total Health Cost of a 1 μg/m3 Increase in Average Suspended Sulfate Concentration in the New York Metropolitan Area
|
|
|
Total Health Cost per μg/m3 SO4 (million $) |
|
Nominal values |
20.5 |
|
|
Additional premature deaths |
|
|
|
low: |
2.3 |
19.9 |
|
high: |
46 |
21.2 |
|
Additional days of aggravated heart and lung disease |
|
|
|
low: |
0.041 mil./yr. |
13.2 |
|
high: |
0.82 mil./yr. |
28.6 |
|
Additional asthmatic attacks |
|
|
|
low: |
8.4 thous./yr. |
19.7 |
|
high: |
168 thous./yr. |
21.3 |
|
Additional cases lower respiratory disease in children |
|
|
|
low: |
.99 thous./yr. |
19.8 |
|
high: |
19.8 thous./yr. |
21.2 |
|
Additional cases chronic respiratory disease |
|
|
|
low: |
4.06 thous./yr. |
11.3 |
|
high: |
81.2 thous./yr. |
30.7 |
|
Cost per premature death |
|
|
|
low: |
$3,000 |
19.9 |
|
high: |
$120,000 |
22.5 |
|
Cost per day aggravation from heart and lung disease |
|
|
|
low: |
$2 |
13.2 |
|
high: |
$80 |
44.8 |
|
Cost per asthma attack |
|
|
|
low: |
$1 |
19.7 |
|
high: |
$40 |
23.0 |
|
Cost per case child’s lower respiratory disease |
|
|
|
low: |
$7.50 |
19.8 |
|
high: |
$300 |
22.7 |
|
Cost per case chronic respiratory disease |
|
|
|
low: |
$25 |
11.3 |
|
high: |
$1000 |
51.1 |
Uncertainty in Assessed Health Costs
The indication from the sensitivity analysis is that the most critical quantities in the assessment of the total health cost of a 1 ug/m3 increase in suspended sulfate concentration are the number of additional cases of chronic respiratory disease, and the number of additional person-days of aggravated heart-lung disease symptoms22. As a rough approximation we shall neglect all uncertainties in health costs but those arising from these two most critical variables. To proceed with a quantitative description of the uncertainty we shall assume that the range of uncertainty in the dose-response relationship (of 10 percent to 200 percent of the values given in Table 13–12) can be treated as having a probability of 90 percent. The uncertainties in the number of additional cases of chronic respiratory disease and the number of additional days of aggravation from heart and lung disease symptoms, are judged to be such that there is only once chance in ten that each of these quantities lies outside the interval defined by the low and high values used in the sensitivity analysis. Specifically, we assume that there is only a 5 percent chance that the additional number of cases of chronic respiratory disease from a 1 ug/m3 increase in SO4 is less than 4060, and a 5 percent chance that this number is greater than 81,200.23 Further, these uncertainties are assumed to be totally dependent (e.g. if the additional number of cases of chronic respiratory disease is high then the number of additional days aggravation from heart and lung disease will also be high, and visa versa). Similarly, we assume that there is a 5 percent chance that the number of additional days of aggravation from heart and lung disease is less than 41,000, and a 5 percent chance that this number exceeds 820,000. Finally, in order to facilitate the calculations, we shall assume the probability distribution characterizing the resulting uncertainty in the total health cost is a lognormal distribution. The shape of this distribution makes it a good approximation, however, greater accuracy could be obtained by
formally assessing the subjective distributions of experts in the area of health effects. Procedures for such probability assessments do exist (Spetzler and Stael von Holstein 1972).
Using the nominal values of $250 per case of chronic respiratory disease and $20 per person day of aggravated heart-lung disease symptoms and the uncertainty on the dose-response relationship for these two health effects as characterized above, we can compute a probability distribution on the total health cost for a 1 ug/m3 increase in average annual ambient sulfate concentration. As has been the case previously, the calculation is done for an average ambient sulfate level of 16 ug/m3 and a population at risk of 11.5 million persons. This probability distribution shows a 5 percent chance that the total health cost will lie above $39 million and 5 percent chance that the total health cost will lie below $4 million, with a 90 percent chance that the total health cost will lie between these values. A lognormal distribution with these properties has an arithmetic mean of $16 million and a standard deviation of $12.3 million24. This broad distribution characterizes the great uncertainty on the magnitude of the health effects caused by suspended sulfate. The distribution is plotted as Figure 13–14.
SULFUR OXIDE DAMAGE TO MATERIALS
A number of studies have indicated that sulfur oxide air pollution accelerates the degradation of many materials (Waddell 1974). Spence and Haynie conclude that sulfur dioxide plays an important role in the chemical deterioration of modern day exterior paints. Based upon a corrosion study of metal structures, Fink et al. (1971) estimate that the accelerated corrosion of zinc by sulfur oxides accounts for most of the damage to metals. They estimate that more than 90 percent of the national economic costs of air pollution corrosion are due to the effect of sulfur oxides on zinc. As part of an economic evaluation of the damages of air pollution, based primarily on a review of
the literature, Waddell (1974) assesses the national SOx-related material damage costs in 1970 to lie between $.4 billion and $.8 billion. These figures are arrived at by summing past estimates of damages to specific material categories. Consequently, there may be some double counting, and it is possible that some significant categories of materials have not been taken into account.
A Simple Materials Damage Model
Although the total national sulfur oxide pollution damage is a useful number, it does not help us much if we are trying to judge between various pollution reduction strategies. What we really need to know is the sensitivity of pollution damage to pollution levels. Therefore, our objective in this section will be to estimate the reduction in pollution damage which will occur for a given reduction in ambient sulfur oxide concentration.
Unfortunately, little is known about pollution material damage dose-response relationships. In fact, the mechanism by which pollution contributes to the degradation of concrete and metal building materials, paints, and fibers, which account for most of the estimated damage, is not entirely clear. Rates of deterioration appear to be functions of sulfate accumulation implying that there is no ambient sulfate level below which no effect will occur25. The relative significance for damage of the various atmospheric sulfur compounds are not known.
We shall summarize our available information into a crude dose-response model. Assume that most material damage occurs in Northeastern urban areas and let us take as the average Northeastern urban concentrations 45 ug/m3 sulfur dioxide (Gillette 1974) and 16 ug/m3 suspended sulfate (see Appendix 13-C). The model consists of a linear approximation to material costs about these concentrations. Let f be the fraction of the estimated materials damage cost which is due to exposure to sulfur dioxide and let the remaining fraction 1-f, be the result of
exposure to suspended sulfates. A doubling of sulfur oxide pollution levels is assumed to roughly double costs (see Appendix 13-C). The net change in urban material damage costs, denoted MD, for a given change k1 in average sulfur dioxide concentration and a given change k2 in average sulfate concentration, is then given by
Following the method of analysis in the section preceding on health costs, we wish to estimate the effect of a 1 ug/m3 increase in ambient sulfate concentration will have on material damage costs. In the absence of more detailed information we shall proceed as follows. We begin with an estimate of $600 million for annual damage caused by sulfur oxides to material property, which applies to the nation as a whole (Waddell 1974: a high value of $800 million and a low value of $400 million are given in addition to a “best” value of $600 million). We shall assume that 90 percent of this damage occurs in the northeastern United States, 60 percent in the New England states, New York, Pennsylvania, Maryland, New Jersey, Delaware, and Greater Washington, D.C. and 30 percent elsewhere east of the Mississippi river. In the above mentioned states we shall assume that the materials damage from sulfur oxides for an area is proportional to its population; most of the population in these states is in urban areas and sulfur oxide damage to materials such as paints and metals will occur primarily in urban areas. We assume as earlier that the population at risk for the remotely located plant (assumed in the Pennsylvania/Ohio border-West Virginia area) is 50 million, and that total sulfur oxide materials damage in area affected by this plant (the states mentioned above) is 60 percent of $600 million or $360 million dollars per year. For the urban representative plant, the population at risk is assumed to be 11.5 million, so
the total sulfur oxide materials damage is assumed to be (11.5/50)×60 percent×600=$83 million per year.
The calculated values for the change in materials damage per 1 ug/m3 change in ambient sulfur dioxide and sulfate levels for the representative case analysis are given in Table 13–17. In the absence of better information we assume that fifty percent of the damage is caused by sulfur dioxide and 50 percent by sulfate. The numbers of Table 13–17 must be regarded as extremely crude estimates.
A review of the information on material property damage by a member of the Committee suggests that Waddell’s estimate for material property damage caused by sulfur oxides might be low by a factor of four or five (see Appendix 13-E). The implication of this increase in material property damage will be addressed in the concluding section.
AESTHETIC COSTS
Damages to the aesthetic quality of the environment by air pollution are a major public concern. Aesthetic costs include the reduction of long distance visibility and light transmittal of the atmosphere, noxious odors, soiling, and the deterioration of materials of historic or artistic significance.
Public perception seems to be that the magnitude of these costs are substantial. A study by the New Mexico State University Agricultural Experiment Station recently assessed the aesthetic environmental damage of the Four Corners Power Plant at roughly $25 million per year in 1970. Respondents to questionnaires used in this study considered reduced visibility to be the most serious aesthetic effect (Randall et al. 1974).
The visibility-restricting air pollutants are particulates and nitrogen dioxide. The extent of visibility restriction caused by suspended particulate depends upon the chemical composition of the particle, its size, shape, and concentration. It is likely that sulfates may significantly contribute to visibility
TABLE 13–17
Materials Damage Increment from a 1 μg/m3 Change in Ambient SOx Levels
|
Fraction of Damage Caused by |
Change in Materials Damage for North Central and New England States* per 1 μg/m3 change in |
Change in Materials Damage for New York Metropolitan Area** per 1 μg/m3 change in |
||
|
SO2 |
SO2 (million $) |
Sulfate (million $) |
SO2 (million $) |
Sulfate (million $) |
|
1.0 |
6.0 |
0 |
1.4 |
0 |
|
0.5 |
3.0 |
11.3 |
0.7 |
2.6 |
|
0 |
0 |
22.5 |
0 |
5.2 |
|
*Maine, Vermont, New Hampshire, Rhode Island, Massachusetts, Connecticut, New York, Pennsylvania, New Jersey, Delaware, Maryland, Washington, D.C. Population at risk approximately 50 million. **Population at risk 11.5 million. |
||||
reduction by increasing the particulate loading (see Chapter 5). Oxides of sulfur are also thought to be responsible for the accelerated decay of art and statuary (Waddell 1974).
One method that has been used to estimate aesthetic costs of air pollution is by market studies of real estate values (see Waddell 1974). Most investigators agree that costs associated with organoleptic effects (effects pertaining to sight and smell) as well as soiling-caused cleaning and maintenance expenditures are capitalized in real estate market values (Waddell 1974:27). By applying classical least squares regression analysis, the existence of a relationship between air pollution and property values can be tested. In most applications a statistically significant inverse relationship between air pollution and property values (or rents) has been identified.
There are at least two major criticisms of the property value approach. First, for the method to have validity the housing market must adequately internalize aesthetic pollution costs into its pricing structure. It may be argued that this is not the case. Second, all significant multi-collinearities between the pollution variables and other explanatory variables for property values must be removed. Since many unfavorable neighborhood attributes will likely be correlated with air pollution (e.g., automobile congestion) there is considerable danger of overstating the pollution coefficients.
Waddell gives an estimate of $5.8 billion as the annual national cost of aesthetic and soiling effects, of which 50 percent is allocated to sulfur oxides and 50 percent is allocated to particulates (Waddell [1974], Table 13–21). The allocation seems misplaced if visibility and odors are the major factors. Photochemical smog and particulates may account for much of the local visibility effects and local concentrations of industrial emissions are responsible for most of the noxious odors. Sulfur dioxide is not detectable by its odor in concentrations below 80 ug/m3.
Suspended sulfates may play a significant role in reducing visibility on a regional scale
(see Chapter 5). These visibility effects due to sulfate deserve further investigation. Sulfate particles are concentrated in the submicron size range that causes the largest visibility reduction affects. Other fine particulates may also be important. Electrostatic precipitators remove only about half of the submicron particles, and flue gas desulfurization should remove a substantially larger proportion. The effect on aesthetics of sulfur oxide emissions from power plants may be most pronounced in rural areas with high recreational or aesthetic values, such as national parks. If most particulates are removed by efficient electrostatic precipitators, it is not likely that appreciable sulfate oxidation will occur until the plume is dispersed, so the reduction in visibility may be small in the neighborhood of the plant.
The aesthetic effects of sulfur oxide emissions may be substantial for some particular power plants. It is difficult to assess values to the aesthetic effects on a regional basis. We will retain an aesthetics cost term, and for a value we will arbitrarily take 20 percent of Waddell’s 5.8 billion per year figure for aesthetic damages, $1.16 billion as being due to SO4, and prorate this over 33.9 million tons of sulfur dioxide, or 33.9×109 pounds of sulfur, emitted per year. The resulting value of 3.4 cents per pound of sulfur emissions is too low to be of major significance compared to health effects, but we shall keep this in the analysis for sensitivity since in a specific instance it might be considerably larger.
ACID RAIN: EFFECTS ON SOILS, FORESTS, AND FISHERIES
Acid rain resulting from sulfur oxide and nitrogen oxide emissions is now widespread over Northern Europe and the northeastern United States. (See further discussion in Chapter 7.) The consequences may include acidification of soils, loss of growth in forests, death of fish from low pH in lakes and streams, and damage to
outdoor statues and building facades. These effects are difficult to assess in economic terms, but they do not seem significant compared to the potential health effects discussed above. In Chapter 7 the first three effects are discussed, and it it is roughly estimated that each might involve incremental costs of the order of 1 cent per pound of sulfur emitted with the sulfur emissions levels that might be achieved by 1980 in the absence of control. However, not all of the costs would be felt immediately, and direct evidence for the extent of the damage is limited.
We will therefore choose arbitrarily a value for the overall costs associated with acid rain of $500 million annually, which is equivalent to about 1.5 cents per pound of sulfur emitted (with 1970 emission levels). As with aesthetic effects this value will be too low to be of significance compared to the potential health effects. Sensitivity analysis will indicate that the costs ascribed to aesthetics and acid rain have little effect on the overall result for the total pollution cost per pound of sulfur emitted.
EVALUATION OF SULFUR OXIDE POLLUTION COSTS FOR THE REPRESENTATIVE CASES
We shall now summarize our assessment of the costs associated with sulfur oxide emissions from the types of plants we have taken as representative, and compare these to generating costs and emission quantities to identify the best strategy. It is worth reiterating that the calculations rest on very limited information, and many assumptions have been made rather arbitrarily in order to carry these calculations out. The purpose of the analysis is not to achieve numerical answers, but by using a quantitative framework to achieve and understanding of the complex issues involved. An analysis of this kind does not resolve uncertainty; it may be useful in determining the importance of uncertainties in the context of a decision. As we shall see, the uncertainty on the pollution cost to be attributed to a pound
of sulfur emission is large, and it would be worth a great deal to resolve this uncertainty in the context of decisions among the alternative strategies for sulfur oxide emissions control on power plants.
Remotely Located Plant
We have assumed for this case that the population at risk of 50 million, roughly that of the North Central and New England states. The plant is assumed to be located in the Western Pennsylvania-Ohio-West Virginia area, about 500 kilometers upwind of the urban concentrations in the Northeast corridor. We have assumed an oxidation rate of 0.5 percent per hour for the 24 hours when the plume is traveling through rural air, then 5 percent per hour for two hours after the plume has encountered a pollutant-laden urban air mass. Assuming that over a year the emissions are dispersed over a 45° sector and 25 percent is removed by rain before reaching the urban area, we compute an increase in average annual ambient concentrations of 0.145 ug/m3 of sulfate and 0.35 ug/m3 of sulfur dioxide, as resulting from 104 kilograms per hour of SOx emissions. This level of emissions equals 96.5 million pounds of sulfur emitted per year. It corresponds to the emissions from 620 MW of generating capacity with an efficiency of 33 percent burning 3 percent sulfur coal at a load factor of 6000 hours per year (representative existing plant), or 612 MW of generating capacity with an efficiency of 38 percent burning 3 percent sulfur coal at a load factor of 7000 hours per year (representative new plant).
The nominal value for the pollution cost described to these emissions is computed in Table 13–18. The total emission cost is seen to be $20.3 million, or $0.21 per pound of sulfur emitted. We can place these costs on a mills per kilowatt hour basis by dividing them by the numbers of kilowatt hours produced annually. The existing plant produces 3.72×109 kwh per year, so the pollution cost computes out to about 5.5 mills/kwh. The more efficient new
TABLE 13–18
Cost of Sulfur Oxide Emissions Representative Calculation for Remote Plant Emitting 10,000 kg of SOx Per Hour (96.5×106 Pounds of Sulfur Per Year)
|
Costs computed based on 0.145 μg/m3 ambient increase in sulfate and 0.35 μg/m3 ambient increase in sulfur dioxide in metropolitan areas with a population of 50 million |
|
|
Health effects (computed at ambient level of 16 μg/m3) |
|
|
25,600 cases of chronic respiratory disease×$250 per case |
$6.4 million |
|
256,000 person-days of aggravated heart-lung disease symptoms×$20 |
5.1 |
|
53,000 asthma attacks×$10 each |
0.5 |
|
6,200 cases of children’s respiratory disease×$75 |
.5 |
|
14 premature deaths×$30,000 |
.4 |
|
Total Health Costs |
12.9 |
|
Materials damage |
|
|
$11.3 million per μg/m3 of SO4×0.145 |
1.6 |
|
$3.0 million per μg/m3 of SO2×0.35 |
1.1 |
|
Aesthetics ($0.034×96.5×106 lbs) |
3.3 |
|
Acid Rain ($0.015×96.5×106 lbs) |
1.4 |
|
Total Emissions Costs |
$20.3 million |
|
Emissions Cost Per Pound of Sulfur |
21¢ |
plant produces 4.28×109 kwh in producing the same amount of sulfur emission, so the pollution cost computes out to 4.7 mills/kwh. The reader may wish to verify on Figures 13–3 and 13–4 this correspondence: Adding a pollution cost of 21 cent per pound of sulfur emitted raises the total social cost of generating electricity in an existing plant burning high sulfur coal from 17.2 mills per kilowatt hour to 22.7 mills per kilowatt hour, and similarly from 21.6 mills per kilowatt hour to 27.4 mills per kilowatt hour for a new plant. For both cases we see that a better alternative is to burn low sulfur eastern coal. The reduction in sulfur pollution costs achieved by switching to a low sulfur coal slightly more than offset the higher fuel price for this fuel. The increase needed before flue gas desulfurization becomes the best alternative is the order of a factor of two: to 53 cents per pound of sulfur for an existing rural plant, to 59 cents per pound for an existing urban plant, and to 37 cents per pound for a new plant.
The pollution cost is very sensitive to many of the input values assumed for the analysis. A list of sensitivity calculations is given in Table 13–19. The two most important health effects are chronic respiratory disease and aggravation of symptoms for persons suffering from chronic heart or lung disease conditions. Both the costs ascribed to these health effects and the dose-response relation for increases in ambient sulfate concentration may cause changes of the order of a factor of two on the pollution cost value. Oxidation rates are also very important: For example, a constant oxidation rate of 2 percent for both rural and urban air causes the pollution cost to rise to 39.4 cents, nearly a 100 percent increase.
Examination of the sensitivity analysis indicates that changes in input values such as oxidation rate, health costs, and the dose response relation for health effects could lead to a pollution cost above 37 cents, at which point flue gas desulfurization becomes the preferred alternative for a new plant, and possibly even above 53 cents, the value at which flue gas desulfurization becomes the preferred
TABLE 13–19
Selected Sensitivity Analysis Pollution Cost Per Pound of Emitted Sulfur for Representative Remote Plant
|
Nominal value |
21¢ |
|
Cost of chronic respiratory disease increased to $1000/case (nominal value=$250/case) |
40.9¢ |
|
Cost per person-day of aggravated heart-lung disease symptoms increased to $80/day (nominal value=$20/day) |
36.9¢ |
|
Cost per premature death increased to $200,000 (nominal value=$30,000) |
23.5¢ |
|
All health costs increased by a factor of four |
61.1¢ |
|
All health costs decreased by a factor of ten |
9.0¢ |
|
Incidence of chronic respiratory disease per μg/m3 of SO4 increased by a factor of two |
27.7¢ |
|
Incidence of chronic respiratory disease per μg/m3 of SO4 decreased by a factor of ten |
15.1¢ |
|
Dose response for aggravated heart-lung disease symptoms increased by a factor of two |
26.3¢ |
|
Dose response for all health effects increased by a factor of two |
34.4¢ |
|
Dose response for all health effects decreased by a factor of ten |
9.0¢ |
|
Materials damage increased by a factor of four |
29.4¢ |
|
Materials damage decreased by a factor of four |
18.9¢ |
|
Aesthetics costs increased by a factor of four |
30.9¢ |
|
Acid Rain costs increased by a factor of four |
25.5¢ |
|
Oxidation Rates assumed doubled to 1% per hour in rural air and 10% per hour in urban air |
33.2¢ |
|
Low Oxidation rates 0.1% per hour in rural air and 1.0% per hour in urban air |
9.5¢ |
|
Oxidation rate constant at 2% per hour |
39.4¢ |
alternative for a retrofitted (rural) plant in our representative plant calculations. The uncertainty is such that although low sulfur eastern coal looks like the best alternative for the representative remote plant, neither burning high sulfur coal nor flue gas desulfurization can be ruled out. With better information to resolve the uncertainty, either could become the preferred alternative.
Urban Plant
The population at risk here is assumed to be 11.5 million, the population of the greater New York City area. The plant is assumed to be located about sixty kilometers upwind of the city, and an oxidation rate of 5.0 percent per hour has been assumed for sulfur dioxide in this metropolitan area. The increase in average annual ambient sulfate levels is computed on the assumption that the emissions are dispersed over a 45° sector and that 10 percent of the sulfur is removed by rain before impacting on the population or values at risk. As for the remotely located plant, the level of emissions assumed is 96.5 million pounds of sulfur per year, or an average of 104 kilograms of sulfur dioxide per hour.
The nominal value for the pollution cost ascribed to these emissions is computed in Table 13–20. The total annual emission cost is $53.1 million, or $0.55 per pound of sulfur emitted.
Looking at the total social cost calculations for Figure 13–5, we see that this value lies well above the crossover point of 19 cents at which low sulfur coal is preferred to the alternative of burning high sulfur coal, and well below the crossover point of $1.00 per pound of sulfur where flue gas desulfurization becomes the least cost alternative.
Another point to be mentioned here is that reduction of other particulate emissions accomplished by flue gas desulfurization has not been included in this analysis. Reduction in emissions of other toxic particulates, such as trace metals, may be an important social benefit for the flue gas desulfurization alternative as
TABLE 13–20
Cost of Sulfur Oxide Emissions Representative Calculation for Urban Plant Emitting 10,000 kg of SOx per hour (96.5×106 pounds of sulfur per year)
|
Costs computed based on 1.86 μg/m3 increase in sulfate and 7.5 μg/m3 increase in sulfur dioxide concentrations in metropolitan areas with population of 11.5 million. |
|
|
Health effects from increase: (computed at ambient level of 16 μg/m3) |
|
|
75,500 cases of chronic respiratory disease×$250 per case |
$18.9 million |
|
755,000 person days of aggravated heart-lung disease symptom×$20 per day |
15.1 |
|
156,000 asthma attacks at $10 each |
1.6 |
|
18,400 cases of children’s lower respiratory disease at $75 |
1.4 |
|
42 premature deaths at $30,000 |
1.3 |
|
Total Health Costs |
$38.3 million |
|
Materials damage Materials damage |
|
|
2.6 million per μg/m3 of SO4×1.86 |
4.8 |
|
0.7 million per μg/m3 of SO2×7.5 |
5.3 |
|
Aesthetics ($0.034×96.5×106 lbs) |
3.3 |
|
Acid Rain etc. ($0.015×96.5×106 lbs) |
1.4 |
|
Total Emissions Costs |
$53.1 million |
|
Emissions Costs per Pound of Sulfur |
55¢ |
TABLE 13–21
Selected Sensitivity Analysis Pollution Cost per Pound of Emitted Sulfur for Representative Urban Plant
|
Nominal value |
55¢ |
|
Cost of chronic respiratory disease increased to $1000/case (nominal value=$250/case) |
113.8¢ |
|
Cost per person-day of aggravated heart-lung disease symptoms increased to $80/day (nominal value=$20/day) |
102.0¢ |
|
Cost per premature death increased to $200/000 (nominal=$30,000) |
62.4¢ |
|
All health costs increased by a factor of four |
174¢ |
|
All health costs decreased by a factor of ten |
19.3¢ |
|
Incidence of chronic respiratory disease per μg/m3 of SO4 increased by a factor of two |
74.6¢ |
|
Incidence of chronic respiratory disease per μg/m3 of SO4 decreased by a factor of ten |
37.4¢ |
|
Dose response for aggravated heart-lung disease symptoms increased by a factor of two |
70.7¢ |
|
Dose response for all health effects increased by a factor of two |
94.7¢ |
|
Dose response for all health effects decreased by a factor of ten |
19.3¢ |
|
Materials damage increased by a factor of four |
86.4¢ |
|
Materials damage decreased by a factor of four |
47.2¢ |
opposed to the use of low sulfur eastern coal. This consideration ought to be included in making the decision on emissions control alternatives for a particular power plant, especially one that is located in a metropolitan area.
As in the case of the rural plant we have carried out sensitivity analysis to indicate how the pollution cost per pound of sulfur would change if different inputs were assumed for the calculation. The results, shown in Table 13–21, show that the pollution cost per pound of sulfur emitted could differ by a factor of two or more if different assumptions were used for the cost attributed to the most significant health effects (chronic respiratory disease and aggravation of heart-lung disease symptoms), the increased prevalence of these effects per unit increase in average ambient sulfate concentration, and the oxidation rate for sulfur dioxide to form sulfate.
UNCERTAINTY ON POLLUTION COSTS AND THE VALUE OF RESOLVING THIS UNCERTAINTY
In previous sections we have used sensitivity values as a basis for sketching probability distributions to represent the uncertainty on the ambient increase in urban sulfate resulting from the emissions of a single power plant (Figures 13–9 and 13–10) and the health effects of a given ambient sulfate increase on incidence of chronic respiratory disease and aggravation of heart-lung disease symptoms (Figure 13–14). The product of these two uncertain factors is the dominant term in establishing the pollution cost per pound of sulfur emitted; other terms such as materials damage, aesthetics, and effects caused by acid rain are judged to be small by comparison. Thus, the probability distribution on the product provides a first cut assessment of the overall uncertainty on pollution cost.
An overall probability distribution on pollution cost per pound of sulfur is given in Figure 13–15 for the representative remote
plant, and Figure 13–16 for the representative urban plant.
These curves should be regarded as sketches indicating the present state of information. They are computed from the probability distribution on the emissions to ambient relationship (Figures 13–9 and 13–10) and the probability distribution on health effects (Figure 13–14), by assuming that each of these distributions could be approximated as lognormal. Since the product of lognormal distributions is lognormal, the the distributions shown in Figures 13–15 and 13–16 are of the lognormal family. It should be noted that only uncertainties in the emissions to ambient relation and the magnitude of the health effects listed in Table 13–12 are included in these distributions. Other health effects, property damage, visibility reduction, and possible climatic changes all would introduce additional uncertainty that would serve to broaden these probability distributions.
It might be noted also that values to health effects (e.g., $250 per case of chronic respiratory disease) have not been treated as uncertain. Changes in these values would change the width of the probability distributions. For example, if health value were increased by a factor of four, the probability distribution in Figure 13–15 would spread from about 8 cents to about $1.36 instead of 40 cents.
Let us recall the pollution costs corresponding to the cross-over points between the alternative strategies which are shown in Figures 13–3, 13–4, and 13–5. For the remote rural plant the probability that the pollution cost would be below 19 cents, were the uncertainties in the health effects and emission to ambient relationship resolved, is about 65 percent. The probability that the cost would be above 37 cents is 5 percent and above 53 cents, of the order of one percent. We can similarly read from Figure 13–16 the corresponding values for the probability that the pollution cost will be above 59 cents, the crossover pont for flue gas desulfurization, case where sludge could not be ponded sludge disposal about 19 percent. It
should be recalled that these probabilities are very rough assessments.
The Figures 13–17, 13–18, and 13–19 are copies of Figures 13–3, 13–4, and 13–5, showing the total social cost per kilowatt hour for the representative plant as a function of the cost per pound of sulfur emitted. We show the probability distribution on pollution cost plotted as a probability density function. The height of the curve is the likelihood of various values of the pollution cost. The area under the curve between any two values for the pollution cost corresponds to the probability that the pollution cost would lie in that interval were the uncertainty to be resolved.
The Value of Resolving Uncertainty
Perhaps the most important consequence of the decision analysis formulation is that we can now place a value on what it would be worth to resolve the uncertainty. The value of resolving uncertainty is the difference between the value of the decision situation where information will be made available to resolve the uncertainty before the decision is made, and the value of the decision situation where the decision must be made in the face of uncertainty. (The concept of the value of information is a basic and rather subtle idea in modern decision theory. See for example Howard (1966, 1968), North (1968), Raiffa (1961, 1969), Tribus (1969) for a detailed explanation of the concept and the computation). To illustrate this concept, let us pose a hypothetical question. Suppose a clairvoyant were available who knew the emissions to ambient relationship, and the health effects of sulfate. From his information we would be able to compute the pollution cost of sulfur and resolve the uncertainty shown in Figures 13–15 and 13–16. In the context of the decision between alternative strategies, how much would we pay him for this information? He could help us avoid expensive mistakes. For example, if we had planned to use low sulfur Eastern coal in an existing rural plant but he then told us the pollution cost was only 10 cent
per pound, we would switch to high sulfur coal and save for an existing plant, 20.6+.78− (17.2+2.59)=1.59 mills per kilowatt hour, or about $6 million a year in social cost for a 620 MW plant running 6000 hours per year.
We know what we would have done without the information, and given the information, we can compute how much we would save by switching the decision. We compute an expected value over the probability distribution on pollution cost of the savings we would get by switching the decision from the one we would have made without the clairvoyant’s information to resolve the uncertainty. The probability distribution on pollution cost is of course exactly the same as a probability distribution on what the clairvoyant will tell us.
For the existing rural plant, the decision is close between burning high sulfur and low sulfur Eastern coal. It is unlikely but possible that resolution of the uncertainty would indicate that flue gas desulfurization is the best alternative. The value of resolving the uncertainty in this situation is about 0.53 mills/kwh, or $2 million a year for the plant. The new rural plant has a similar answer. It is a bit more likely that resolving the uncertainty will result in a switch to flue gas desulfurization, but the result of the calculation is about the same, .48 mills/kwh or $2 million a year for a new 612 MW plant operating 7000 hours a year. If low sulfur eastern coal is not available at the rural plant location the value of resolving uncertainty drops by about one half, to about $1.2 million per year for an existing plant and $1.6 million for a new plant.
For the urban plant the decision is very close between low sulfur coal and flue gas desulfurization. With sludge disposal cost at 0.9 mills/kwh for F&D for the urban site, the value of resolving the uncertainty on pollution cost per pound is about 0.25 mills/kwh, or $900,000 per year for an existing plant of 620 MW running 6000 hours per year.
Judging from the list of large coal burning plants in Appendix 13-B and the sulfur emissions data in Figure 13–10, there are at least the
equivalent of about one hundred of our representative rural plants (60,000 MW) burning coal of the order of 3 percent sulfur in the Northeastern region of the United States. In addition there are about 60,000 MW of new coal plants planned or under construction, and the order of 20,000 MW of oil fired plants that might be converted to coal. If we scale up the result of the value of information calculation according to these numbers, we reach a value of about $250 million per year as a rough estimate of what it might be worth to resolve uncertainty about the emissions to ambient relationship and health consequences of sulfate.
Relation to the costs of a Research Program to Resolve Uncertainty on Health Effects and the Emission to Ambient Relationship
Some estimates for the costs and time to obtain results in resolving the uncertainties on the health effects of sulfates and the emissions to ambient relationship are contained in a paper by David P.Rall, of EPA, which is in the Volume 2 of the National Academy of Sciences report on Automobile Emissions 1974 p. 427–431. The estimated cost for the research program recommended in this document is approximately $8–10 million per year, with significant results expected from most of the program elements in two to five years. The program funding is about equally divided between research on monitoring and understanding sulfate in the atmosphere, and achieving a better understanding of the health effects of sulfates. In the light of the preceding calculation, which shows a value of the information the order of twenty-five times higher than the cost of carrying out this research, such a research program would seem like an excellent idea.
CONCLUSIONS AND OBSERVATIONS
The analysis of this section is intended as a means for organizing and placing in perspective the information made available in
this study to serve as a basis for decisions on sulfur oxide control. This task is an ambitious one, and the authors are well aware of the limited way in which they were able to deal with various issues under limitations of time and resources. We regard the analysis as a framework and not a finished product, and we hope that diligent review and criticism of our work will provide a better basis for decisions.
The major thrust of the analysis has been to relate the consequences of emissions levels to the costs imposed on electric power generation. We carried out calculations for three representative power plants in which we examined the effect of alternative strategies on the sum of the cost of electricity plus the social costs attributable to sulfur oxide emissions. Whereas the calculation of electricity cost is relatively straightforward, the social cost of sulfur oxide emission is both uncertain and judgmental, so its assessment is difficult. Yet a decision on emissions control strategy requires that this assessment be made, explicitly or implicitly. We have shown a way of carrying through the assessment explicitly, and we have illustrated how the preferred alternative for each representative plant changes with the assessment of pollution cost and with changes in the added costs of electricity imposed by that alternative.
In Table 13–22 we have summarized the main analytical results for the representative plant calculations. We show only the alternatives of burning high sulfur coal, switching to low sulfur eastern coal, or installing flue gas desulfurization. (Coal preparation and use of western low sulfur coal were found in these calculations to be somewhat inferior in sulfur removal for the additional cost, but it should be cautioned that for some power plant situations one of these alternatives could be preferred.) The values of pollution cost corresponding to the crossover points between high sulfur coal, low sulfur coal if available, and flue gas desulfurization are shown in the first section, and below we summarize the change in the crossover point per increment of additional cost in the abatement alternatives.
TABLE 13–22
Summary of Major Results for Representative Plants
|
Crossover values of pollution cost (cents per pound of sulfur) at which alternative strategy changes: |
|||
|
|
Switch from high sulfur to eastern low sulfur coal |
Switch to FGD |
|
|
with low sulfur coal available |
without low sulfur coal available |
||
|
new plant (rural) |
19 |
37 |
23 |
|
existing plant (rural) |
19 |
53 |
26 |
|
oil plant to be reconverted (urban) to coal |
19 |
59 |
28 |
|
Sensitivities: Change in crossover values for a 1 mill/kwh increment in capital cost of FGD (1 cent/MMBTU increment in price premium for eastern low sulfur coal) |
|||
|
new plant (rural) |
(0.57) |
23 |
5.0 |
|
existing plant (rural) |
(0.57) |
20 |
4.3 |
|
oil plant reconverted (urban) |
(0.57) |
20 |
4.3 |
|
Pollution costs per pound of sulfur emitted |
|||
|
|
Normal value |
Point on Probability Distribution* |
||
|
|
5% |
Mean |
95% |
|
|
rural case (new and existing) |
21 |
9 |
18 |
37 |
|
urban case (oil plant reconverted) |
55 |
21 |
46 |
100 |
|
Expected Value of Resolving Uncertainty*, in mills/kwh (million $/year each representative plant) |
||||
|
|
If low sulfur coal |
|
|
|
Available |
Not available |
|
new plant (rural) |
0.48 (2.1) |
0.38 (1.6) |
|
existing plant (rural) |
0.53 (2.0) |
0.34 (1.2) |
|
oil plant reconverted (urban) |
0.19 (0.9) |
0.40 (1.5) |
|
*The probability distributions (Figures 13–15, 13–16) are a rough summary of judgment regarding uncertainties in the emissions to ambient relationship and in a number of cases adverse health effects given in Table 13–12. Other uncertainties have not been included in these distributions or the value of information calculation. |
||
Assessment of the pollution cost was found to depend most critically upon the sulfur dioxide-ambient sulfate relationship, the dose-response relation between health effects and ambient sulfate, and the social cost ascribed to the health effects. Material property damage was the next most important factor, and the other effects were judged to have less importance. Calculation for the representative cases was carried out using nominal values for these input factors, and uncertainty from the emissions-to-ambient and dose-response relations was characterized by probability distributions. These probability distributions were then used to calculate the expected value of resolving the uncertainty before the choice of emissions control strategy is made.
High Sulfur Coal: A Poor Alternative for the Urban Representative Case
The results for the urban case analysis indicate that burning high sulfur coal in a plant close upwind of a major metropolitan area is a poor strategy relative to low sulfur eastern coal or flue gas desulfurization. With a population at risk of 11.5 million (e.g., Metropolitan New York) and health values of $250 per case of chronic respiratory disease, $20 per day of aggravated heart lung disease symptoms, the nominal pollution cost was computed at 55 cents, far above the cross-over points of 19 cents (from high sulfur coal to low sulfur coal) and 28 cents (from high sulfur coal to FGD, assuming low sulfur coal is not available). Resolving the uncertainty on emissions to ambient and dose-response is very unlikely to bring the pollution cost below 19 cents, although a significant chance (29 percent) is computed that this resolution would bring the cost below 28 cents.
Would it be worth waiting to resolve this uncertainty before acting to abate the emissions? The following argument indicates that the waiting is not advisable. Let us consider the case where low sulfur coal is not available, and assume the decision is between
high sulfur coal and flue gas desulfurization. Suppose the decision is delayed a year, during which high sulfur coal is burned: the expected net social cost of this choice (pollution cost plus electricity cost) is about 4 mills/kwh, or about $15 million/year. The expected value of resolving the uncertainty is 0.4 mills/kwh, a tenth as much. Even discounting this value forward for the remaining life of the plant, the expected improvement in the decision does not compensate for the loss incurred by a year of delay. The argument for continued use of high sulfur coal if low sulfur coal is available is even weaker: the cost of waiting is high and the chances are very low that further information would show that sulfur removal was not worth at least 19 cents per pound.
Other Comparisons Among Strategies: The High Value of Further Information
The size of the uncertainties and the importance of judgment on health values make it difficult to draw additional implications in comparing other strategy alternatives. For the urban case low sulfur eastern coal and flue gas desulfurization are close, and for the rural case the best strategies appear to be low and high sulfur coal. The difference between these alternatives is slight, and the value of resolving uncertainty is large. An estimate for the country as a whole based on the representative plant calculations is $250 million per year to resolve the uncertainty on the sulfur oxide emissions to ambient sulfate relationship and the dose response relation for health effects. This indicates the great need for near term research results in these areas.
The values assigned to health effects play an equally important role in the calculations. If the values for chronic respiratory disease of $250 per case and $20 per day for aggravated heart-lung disease symptoms are increased a factor of four, the range of pollution costs is shifted up to where flue gas desulfurization appears desirable for both the urban and rural case. If further information confirms oxidation
rates and dose response relationships of approximately the present estimates, the decisions will hinge largely on these difficult value judgments: how society shall make tradeoffs between reduction in these health effects and reduction in the cost of electricity. In this situation equity and distributional issues would undoubtedly assume much greater importance. On the other hand, further information may indicate that the consequences of sulfates on human health are sufficiently great (or small) so that the decisions are much less sensitive to these value judgments.
Other Needed Information
Appendix 13-F contains a brief review of the materials damage literature, with the implication that the materials damage value used in this analysis may be low by as much as a factor of four or five. One implication of this change would be to make emissions abatement for the urban case justified on the basis of alleviating material property damage alone. It should be recalled, however, that many of the assumptions used in the materials damage calculation were extremely arbitrary, and clearly more clarification on the materials damage issue would be very welcome.
There are a number of important potential consequences of sulfur oxide emissions that were not included in the analysis: the possibility of more serious health effects such as cancer or emphysema, climatic changes induced by sulfates, or catastrophic effects on living systems from high levels of acid rain. These areas should receive substantial research attention, and information on these areas should be included in subsequent analysis on sulfur oxide emissions control strategy. Information that suflates contribute substantially to serious respiratory disease could motivate a strategy of very high emissions reduction wherever possible. Since the cost of additional land permitting a scrubber to be added at a later time is low, perhaps in the range of a few thousand dollars
annualized cost per year, it would seem prudent for all new electric coal burning power stations to allow for this possibility.
Using the Methodology on a Case by Case Basis
The variation in circumstances from one power plant to another makes it highly advisable that the decision among alternative strategies be examined on a case by case basis. A possible problem in this approach arises from the economic and air quality relationships between the plants. The main economic relation is the limited near term supply of low sulfur eastern coal. If there is more demand for it, its price premium will increase relative to high sulfur coal until it is effectively no longer available. We have avoided this difficulty by examining the two extreme cases, low sulfur coal available at about the present price premium, and not available at all. The relation through air quality is that if a large number of plants install emissions abatement, the pollution damage per pound of sulfur may decrease below the crossover point to install abatement. Given the present uncertainties this relation does not seem a serious concern: there is little evidence to indicate a sharp threshold for damages as emissions (or ambient levels) are changed.
We believe that an important use for the type of methdology illustrated in this section is to determine where limited resources (such as flue gas desulfurization construction capacity, available low sulfur coal) can do the most to alleviate pollution damage in the near term. This approach may be most helpful in setting priorities among types of plants regionally or among several plants in one locality, and it could be extended to case by case analysis of a number of power plants in a large region.
FOOTNOTES
|
1 |
Ambient levels of sulfur dioxide and suspended sulfates and their relation to power plants and other emission sources are discussed in Section 1 of Part Two. |
|
2 |
Further discussion on the relationship between health effects and ambient levels of sulfur oxides and particulates is found in Part 1. |
|
3 |
Since other pollutants act as catalysts to accelerate the oxication of sulfur dioxide to sulfate, a possible additional control strategy to reduce ambient sulfate levels is to reduce the emissions of these other pollutants. In some local situations this approach might be very effective. For example, the difference in observed oxidation rates in plumes (Gartrell 1963, (Newman et al. 1975a,b) may be largely due to the presence of particulates, especially trace metals such as vanadium. For the northeastern region of the U.S. taken as a whole the effectiveness of this approach appears much less promising. The chemistry of the oxidation is complex and not well understood, and there are many materials believed to act as catalysts. Some of these may be impossible or extremely expensive to eliminate from the atmosphere: for example, terpenes and other hydrocarbons given off naturally by vegetation, soot and smoke particles, and photochemical smog resulting from automobile emissions. While further investigation of a catalyst control approach appears warranted, the best approach for reducing sulfate levels appears to be reduction of the quantity of sulfur oxide emitted into the atmosphere. We shall not include a catalyst control alternative among the strategies considered in the analysis. |
|
4 |
An exception is the toxic materials sections (307 and 311) of the Federal Water Pollution Control Act Ammendments of 1972, Public Law 92500, 92 Congress, S. 2770, October 18, 1972, specifying a fine schedule depending on the amount spilled for toxic substances |
|
|
should be assessed and if significant included in the cost of coal preparation. Other fine particulates beside sulfate may have substantial health effects, but there is virtually no detailed information now available on these effects. In the absence of more information sulfate is used as an indicator of the toxic potential of participates. It should be noted that FGD results in a reduction of about 50 percent in fine particulate emissions over the levels obtainable with particulate removal technologies alone, and switching to low sulfur coal might actually increase fine particulate emissions from a power plant unless particulate removal technologies are carefully optimized for the new coal. Some credit for lowering fine particulate emissions should be given to FGD relative to the low sulfur coal alternative. In view of the great uncertainties in the health effects area we have not attempted to assess the magnitude of this credit. |
|
8 |
While the discussion has dealt with sulfur oxide pollution, effects of other pollutants and any other important externalities should be included in the assessment of the least total cost to society. See footnote7. |
|
9 |
Pollution externalities associated with nuclear power have not been included in the cost of the nuclear alternative. The magnitude of nuclear externalities should be assessed and included in a comparison between nuclear and coal fired generation. Health effects in the mining and processing of fuels may be as significant as the pollutants released from nuclear and coal fired plants. See, for example, Sagan (1972), (1974). |
|
10 |
More advanced techniques of coal preparation are discussed in Chapter 10. These techniques are considerably more expensive for the additional amount of sulfur removed. |
|
11 |
As stated earlier, the focus of this analysis is the hazards from low levels of sulfur dioxide and sulfate, and not the reduction in violations for the sulfur dioxide primary ambient standard. We assume that such violations will be eliminated either by using tall stacks and intermittent control, |
|
|
transport of sulfur oxides include Zeedijk and Velds (1973) and Rodhe (1972). |
|
15 |
Strictly speaking, the emissions are not uniformly distributed along the length unless the unit of time is small. See Appendix A for more precise description of the model. |
|
16 |
About 40 percent of the emitted sulfur oxide is returned as sulfate in rain, from calculations in Chapter 10. |
|
17 |
These results are in general agreement with the results given in Part Two Section 1: the 10–40 percent increase was predicted for urban sulfate levels (for the nation as a whole) as a consequence of doubling sulfur oxide emissions from all power plants throughout the country. |
|
18 |
Source: 1973 Statistical Abstract, using 1970 census data. 47 million persons lived in standard metropolitan stastical areas in the eleven states plus the Greater Washington, D.C. area. |
|
19 |
Limited data have shown that, within the range of .3 to 2.5 um, the smaller the particle size the greater the irritant potency. Reference: Amdur (1960). |
|
20 |
The dose-response relations that we are about to describe were developed in an unpublished study for the U.S. Environmental Protection Agency. Our use of these particular curves is not meant to indicate an endorsement. Rather, we present them as highly simplified illustrations of a set of possible relationships that have not yet been firmly established. Judgment communicated to us by representatives from the Assemble of Life Sciences Committee responsible for Part one of this report was that these numbers were not unreasonable, and that they could be considered the best estimates of the dose response relationships available at this time. Source: Statistical Abstract of the U.S., 1972. The populations listed for the various age groups are estimations based upon the total New York Metropolitan Population (Table 20, pg 20) and the age distribution for New York State (Table 36, pg 31). |
|
|
reason, while a threshold may not exist for physical damage, economic loss from physical damage may exhibit such a threshold. Threshold effects may be introduced when converting physical material damage to economic loss. Our analysis ignores this effect, possibly causing us to underestimate marginal material damage costs. More accurate dose-response information ought to be applied in the analysis as it becomes available. |
APPENDIX 13-A
A MODEL RELATING SULFUR OXIDE EMISSIONS TO AMBIENT SULFATE LEVELS
Appendix 13-A presents the mathematical relationships and calculations for the emissions to ambient sulfate model. As described in the text, the model is a simple “box” model in which the width of the box grows proportionally with time. SO2 removal, SO2 oxidation to sulfate, and sulfate removal all proceed according to a first order rate reaction. The geometry of the model is illustrated in Figures App. 13-A1 and App. 13-A2.
The geometric relationships in the model are as follows. We assume a constant wind velocity of u kilometers per hour, and assume that the plume subtends an angle θ in the direction downwind of the plant. That direction is assumed to remain constant. In a small time increment Δt a quantity Δx of SO2 is emitted. We assume this to remain uniformly distributed between ground and the mixing layer at height h above ground, across a width (perpendicular to the direction of the wind) of w(t)=2ut tan (θ/2), and through a length (parallel to the direction of the wind) of uΔt. Then the assumption for the box model is that at time t the concentration of sulfur dioxide per unit volume, Yo, is uniform within this box, which has a volume of
(1)
where A(t) is the rectangular area in the crosswind vertical plane. If we assume an emissions rate Δx/Δt=q then the concentration of SO2
from the power plant at a time t after emission (a distance of ut downwind), assuming no oxidation or removal has occurred, is given by the quantity emitted Δx=qΔt divided by the volume A(t) uΔt of the volume element:
(2)
We assume that there are no further sources of SO2, and we ignore any SO2 present before the air reached the power plant. Our concern is just the SO2 emitted from the power plant, and Yo is the incremental SO2 concentration from that power plant, to be added to the ambient levels from other sources. Likewise, in the discussion below, we treat incremental quantities of SO2 and sulfate resulting from the emissions of the power plant source.
Let us consider the volume element ΔV(t)=A(t)uΔt to contain an amount of sulfur dioxide Y(t) and an amount of sulfate Z(t). No new sulfur oxide enters this volume element after it leaves the power plant, and sulfur dioxide and sulfate are assumed to be removed only through dry deposition at given deposition velocities. The sulfur oxides are assumed to be uniformly distributed throughout the volume element ΔV(t) at all times. We now write the differential equations for the changes in the quantities contained in ΔV(t). Then from equation (1) we can compute the concentrations of SO2 and SO4 by dividing the quantities by the volume ΔV(t) in the same manner as for equation (2).
Let us first take the oxidation reaction. Of the quantity Y(t) of SO2 contained in ΔV(t), a proportion of a1 is lost through oxidation to sulfate per unit of time:
(3)
A loss also occurs through dry deposition. Of
the quantity Y(t) of SO2 contained in the volume element, a proportion is lost per unit time equal to v1 (the dry deposition velocity for SO2) times the area of contact with the ground, which is the uΔt×2ut tan(θ/2), times the concentration, which is Y(t)/huΔt×2ut tan(θ /2)). The proportion of SO2 lost through dry deposition per unit time is then

(4)
We shall define v1/h=a2, the rate of loss due to dry deposition of SO2. Since loss through oxidation and loss through dry deposition are the only mechanisms by which the quantity of SO2 in the volume element is changed, we combine (3) and (4) into a single differential equation for Y(t), the quantity of SO2 in the volume element:
(5)
This equation has the solution
(6)
where Yo is the initial quantity of SO2 in the volume element. By dividing the quantity Y(t) by the volume of the element A(t) uΔt we obtain
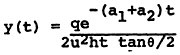
(7)
We go through a similar reasoning process to arrive at a solution for the sulfate concentration. If oxidation of one gram of SO2 produces k grams of sulfate ion, then the quantity of sulfate Z(t) in the volume element is given by
(8)
since a1Y(t) grams of SO2 are oxidized per unit time in the volume element at time t (from equation(3)). We also must account for the loss of sulfate through dry deposition. The quantity lost from the volume element per unit time is the dry deposition velocity (v2) times the lower surface of the volume element, the area in contact with the ground, times the concentration of sulfate in the volume element. The area of contact with the ground is (as before) uΔt×2ut tan(θ/2), and the concentration of sulfate is the quantity Z(t) of sulfate in the volume element divided by the volume of the element: Z(t)/huΔt×2ut tan(θ/2)). The proportion of sulfate from the volume element lost per unit of time is then
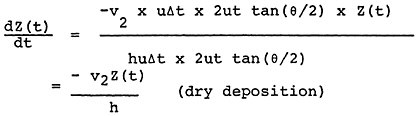
(9)
We shall define v2/h=a3, the rate of loss due to dry deposition of sulfate. Since formation through oxidation and loss from dry deposition are the only means by which the quantity of sulfate in the volume element is changed, we can combine (8) and (9) into a single differential equation for the quantity of sulfate Z(t):
(10)
The solution to this differential equation is given by
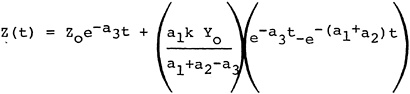
(11)
where Zo and Yo are initial quantities of sulfate and SO2, respectively, in the volume element. As with sulfur dioxide, we can divide the quantity Z(t) by the size of the volume element to obtain the concentration z(t). If the initial quantity of sulfate Zo is zero,
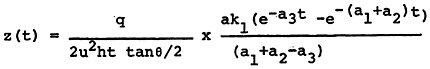
(12)
It is also possible to solve these equations (6) and (11) for rates that change discontinuously from one time period to another. One computes the solution for the first time period, uses this to establish the quantities Yo and Zo as initial conditions for the second time period, then one solves equations (6) and (11) for the second time period. Of course, when the solutions are expressed as concentrations these must be computed by dividing the quantities of SO2 and sulfate by the volume ΔV(t) of the volume element, and this volume is increasing in direct proportion to time.
In solving this model to obtain concentration of SO2 and sulfate we shall use the following values for the inputs:
-
The wind is a constant 20 kilometers/ hour.
-
The angle subtended by the plume is 15°.
-
The height of the mixing layer above ground is 1000 meters.
-
The dry deposition velocity for SO2 to the ground is 0.8cm/sec, giving a loss rate a2 of 2.88 per cent per hour with a mixing layer height of 1000 meters.
-
The dry deposition velocity of SO4 to the ground is 0.4cm/sec, giving a loss rate a3 of 1.44 percent/hour with a
-
mixing layer height of 1000 meters above ground.
-
The oxidation rate a1 of SO2 to form sulfate is 0.5 percent per hour in rural air outside of a metropolitan area, and 5.0 percent per hour when the air has passed over a metropolitan area and contains particulates, oxidants, and hydrocarbons from urban emission sources.
-
We neglect the 1–2 percent of sulfur oxide emitted initially as sulfate and consider that all of the 104 kilograms per hour of sulfur dioxide is emitted as SO2. This assumption is of course easily relaxed, but since the initial sulfate is small compared to the sulfate formed subsequently by the oxidation, we may neglect it in the calculation without introducing a significant error.
Calculations are given in Table 13–6 of the main text for incremental increases in the ambient concentrations of SO2 and sulfate over time for an oxidation rate of 0.5 percent per hour and in Table 13–7 of the text for an oxidation rate of 5 percent per hour. Table App. 13-A1 shows the results if a 2 percent oxidation rate is used.
Table App. 13-A1
Incremental Contributions to Ambient Levels of SO2 and Sulfate Resulting from the Emissions of a Single 600 MW Power Plant (Oxidation Rate of 2.0 percent per hour assumed.) hour assumed.)
|
Time, hours since emission |
3 |
6 |
12 |
18 |
24 |
50 |
|
Distance from plant, km |
60 |
120 |
240 |
360 |
480 |
1000 |
|
Increase in SO2 concentration (μg/m3) |
27.3 |
11.8 |
4.4 |
2.2 |
1.23 |
0.17 |
|
Increase in SO4 concentration (μg/m3) |
2.59 |
2.36 |
1.96 |
1.64 |
1.37 |
0.66 |
Table 13–8 of the main text and Figure 13–8 give results for the case in which an oxidation rate of 0.5 per hour is assumed for 24 hours (480 km) then the rate increases to 5 as the sulfur dioxide oxidation reaction is catalyzed by the presence of urban pollutants. Tables App. 13-A2, 3, and 4 show the effect of varying the time (distance) between the plant and the metropolitan area. The values after two hours (40 km) of exposure to urban air are shown in boxes for comparison to the values of 1.39ug/m3 of SO2 and 0.58ug/m3 of SO4 for the ambient increments from Table 13–8.
Table App. 13-A2
Incremental Contributions to Ambient Levels of SO2 and Sulfate from a Power Plant in a Remote Location (104 kg/hour SO2 emissions, 120 km from plant to city).
|
Time, hours since emission |
3 |
6 |
7 |
8 |
9 |
|
Distance from plant, km |
60 |
120 |
140 |
160 |
180 |
|
Increase in SO2 concentration (μg/m3) |
28.6 |
12.9 |
10.2 |
8.3 |
6.8 |
|
Increase in SO4 concentration (μg/m3) |
0.66 |
0.62 |
1.32 |
1.78 |
2.1 |
|
Location of plume |
rural air |
edge of city |
metropolitan area |
||
|
Oxidation Rate % per hour |
0.5 |
5.0 |
5.0 |
5.0 |
5.0 |
Table App. 13-A3
Incremental Contributions to Ambient Levels of SO2 and Sulfate from a Power Plant in a Remote Location (104 kg/hour SO2 emissions, 240 km from plant to city).
|
Time, hours since emission |
3 |
6 |
12 |
13 |
14 |
15 |
|
Distance from plant, km |
60 |
120 |
240 |
260 |
280 |
300 |
|
Increase in SO2 concentration (μg/m3) |
28.6 |
12.9 |
5.3 |
4.5 |
3.9 |
3.3 |
|
Increase in SO2 concentration (μg/m3) |
0.66 |
0.62 |
0.53 |
.83 |
1.06 |
1.23 |
|
Location of plume |
rural air |
|
edge of city |
metropolitan area |
||
|
Oxidation Rate % per hour |
0.5 |
0.5 |
5.0 |
5.0 |
5.0 |
5.0 |
Table App. 13-A4
Incremental Contributions to Ambient Levels of SO2 and Sulfate from a Power Plant in a Remote Location (104 kg/hour SO2 emissions, 1000 km from plant to city).
|
Time, hours since emission |
6 |
12 |
18 |
24 |
50 |
51 |
52 |
53 |
|
Distance from plant, km |
120 |
240 |
360 |
480 |
1000 |
1020 |
1040 |
1060 |
|
Increase in SO2 concentration (μg/m3) |
12.9 |
5.3 |
2.87 |
1.76 |
0.35 |
0.32 |
0.29 |
0.26 |
|
Increase in SO4 concentration (μg/m3) |
0.62 |
0.53 |
0.46 |
0.40 |
0.22 |
0.24 |
0.25 |
0.26 |
|
Location of plume |
rural air |
|
|
|
edge of city |
metropolitan area |
||
|
Oxidation Rate % per hour |
0.5 |
0.5 |
0.5 |
0.5 |
5.0 |
5.0 |
5.0 |
5.0 |
APPENDIX 13-B
THE REPRESENTATIVENESS OF THE ILLUSTRATIVE CASES
Ideally, the analysis of alternative emission control strategies should be carried out for each individual power plant, but such an effort was obviously not possible for the present study. Our analysis has, instead, focused on two illustrative cases: a rural plant 300 miles upwind of a metropolitan area, and a plant in or adjacent to an urban area.
Tables App. 13-B1, 2, and 3 indicate the number and nature of the various power plants that our illustrative examples represent. The data is illustrative, not definitive: in some cases it is out of date or in need of qualification. The reader is directed to the referenced source documents for further details.
Table App. 13-B1 lists some of the larger existing coal fired plants, their locations, and some relevant technical and economic characteristics. Table App. 13-B2 lists some new coal fired plants and those units planned for construction in the near future. Table App. 13-B3 contains a tabulation of existing oil fired plants deemed convertible to coal. (The determination of which oil burning plants could be converted to burn coal was carried out by the Oil Savings Work Group, Office of Energy Conservation, Federal Energy Agency. All of those plants listed were originally designed to burn coal.)
TABLE App. 13-B1
|
Region State City |
Company |
Plant |
Capacity (MW) |
Cost Per KW of Installed Capacity ($) |
|
NEW ENGLAND |
|
|||
|
New Hampshire |
|
|||
|
Bow |
Public Service Co. of New Hampshire |
Merrimack |
459 |
132 |
|
MIDDLE ATLANTIC |
|
|||
|
New York |
|
|||
|
Dunkirk |
Niagara Mohawk Power Corporation |
Dunkirk |
628 |
139 |
|
Tonawanda |
|
C.R.Huntley |
928 |
149 |
|
Pennsylvania |
|
|||
|
Springdale |
Duquesne Light Company |
Cheswick |
565 |
209 |
|
Elrama |
" |
Elrama |
510 |
176 |
|
South Heights |
" |
F.Phillips |
411 |
161 |
|
Portland |
Metropolitan Edison Company |
Portland |
427 |
149 |
|
Shawville |
Pennsylvania Electric Company |
Shawville |
640 |
139 |
|
Homer City |
" |
Homer City |
1,319 |
144 |
|
W. Pittsburgh |
Pennsylvania Power Company |
New Castle |
426 |
128 |
|
York Haven |
Pennsylvania Power & Light Co. |
Brunner Island |
1,559 |
100 |
|
Sunbury |
" |
Sunbury |
410 |
209 |
|
Shelocta |
" |
Keystone |
1,872 |
101 |
|
Indiana Co. |
" |
Conemaugh |
1,872 |
119 |
|
Strawberry Ridge |
" |
Montour |
823 |
176 |
|
Eddystone |
Philadelphia Electric Company |
Eddystone |
707 |
226 |
|
Courtney |
" |
Mitchell |
449 |
140 |
|
Masontown |
" |
Hatfield’s Ferry |
1,728 |
139 |
|
EAST NORTH CENTRAL |
|
|||
|
Illinois |
|
|||
|
Bartonville |
Central Illinois Light Company |
E.D.Edwards |
725 |
179 |
|
Coffeen |
Central Illinois Public Service Co. |
Coffeen |
1,006 |
143 |
|
Joliet |
Commonwealth Edison Company |
Joliet |
1,787 |
118 |
|
Kincaid |
" |
Kincaid |
1,319 |
107 |
|
Pekin |
" |
Powerton |
1,213 |
182 |
|
Waukegan |
" |
Waukegan |
933 |
140 |
|
Lockport |
" |
Will County |
1,269 |
133 |
|
Joppa |
Electric Energy, Incorporated |
Joppa |
1,100 |
152 |
|
Baldwin |
Illinois Power Company |
Baldwin |
623 |
188 |
|
Venice |
Union Electric Company |
Venice #2 |
500 |
144 |
|
Indiana |
|
|||
|
Madison |
Indiana-Kentucky Electric Corp. |
Clifty Creek |
1,304 |
115 |
|
Lawrenceburg |
Indiana & Michigan Electric Company |
Tanners Creek |
1,100 |
127 |
|
Petersburg |
Indianopolis Power & Light Company |
Petersburg |
733 |
117 |
|
Dune Acres |
Northern Indiana Public Service Co. |
Bailly |
616 |
149 |
|
Gary |
" |
D.H.Mitchell |
529 |
164 |
|
Cayuga |
Public Service Co. of Indiana, Inc. |
Cayuga |
1,018 |
141 |
|
New Albany |
" |
Gallagher |
637 |
152 |
|
Terre Naute |
" |
Wabash River |
881 |
130 |
|
Michigan |
|
|||
|
West Olive |
Consumers Power Company |
J.H.Campbell |
650 |
129 |
|
Muskegon |
" |
B.C.Cobb |
511 |
134 |
|
Essexville |
" |
D.E.Korn |
530 |
158 |
|
Monroe |
Detroit Edison Company |
Monroe |
817 |
224 |
|
E. China Twp |
" |
St. Clair |
1,905 |
137 |
|
Ohio |
|
|||
|
New Richmond |
Cincinnati Gas & Electric Co. |
W.C.Beckjord #1–5 |
761 |
145 |
|
New Richmond |
" |
W.C.Beckjord #6 |
461 |
132 |
|
Ashtabula |
Cleveland Electric Illuminating Co. |
Ashtabula |
640 |
NA |
|
Avon Lake |
" |
Avon Lake |
1,275 |
143 |
|
Eastlake |
" |
Eastlake |
1,257 |
150 |
|
Cleveland |
" |
Lake Shore |
514 |
131 |
|
Conesville |
Columbus & Southern Electric Co. |
Conesville |
434 |
129 |
|
Aberdeen |
Dayton Power & Light Company |
J.M.Stuart |
1,831 |
146 |
|
Shayside |
Ohio Edison Company |
R.E.Burger |
544 |
157 |
|
Stratton |
" |
W.H.Sammis #1–7 |
3,304 |
153 |
|
Brilliant |
Ohio Power Company |
Cordinal |
1,231 |
112 |
|
Beverly |
" |
Muskingun |
1,530 |
120 |
|
Philo |
" |
Philo |
500 |
115 |
|
Cheshire |
Ohio Valley Electric Corporation |
Kyger Creek |
1,086 |
124 |
|
Oregon |
Toledo Edison Company |
Bay Shore |
640 |
165 |
|
Wisconsin |
|
|||
|
Oak Creek |
Wisconsin Electric Power Company |
N.Oak Creek |
500 |
149 |
|
Port Washington |
" |
Port Washington |
400 |
121 |
|
Oak Creek |
" |
S.Oak Creek |
1,192 |
116 |
|
WEST NORTH CENTRAL |
|
|||
|
Minnesota |
|
|||
|
Oak Park Hts. |
Northern States Power Co. (Minn.) |
A.S.King |
598 |
137 |
|
Missouri |
|
|||
|
Henry Co. |
Kansas City Power & Light Company |
Montrose |
563 |
117 |
|
Sibley |
Missouri Public Service Company |
Sibley |
519 |
151 |
|
Labadie |
Union Electric Company |
Labadie |
1,665 |
179 |
|
St. Louis |
" |
Meramac |
800 |
153 |
|
West Alton |
" |
Sioux |
978 |
113 |
|
Randolph Co. |
Associated Electric Cooperative |
Thomas Hill |
456 |
138 |
|
Annual Generation (Million KWH) |
Heat Rate (BTU/KWH) |
Operating Costs, Exclusive of Fuel (Mills/KWH) |
Coal Consumption (1000 tons) |
Avg. Heat Content (Million BTU/ton) |
Cost per Ton as Burned ($) |
Avg. Sulfur Content (%)c |
|
2,964 |
10,081 |
.69 |
1,099 |
27.18 |
13.03 |
2.33 |
|
3,266 |
9,764 |
1.02 |
1,286 |
25.39 |
12.84 |
2.54 |
|
3,450 |
10,142 |
1.36 |
1,380 |
25.30 |
14.30 |
2.20 |
|
3,242 |
9,494 |
.89 |
1,387 |
22.00 |
8.31 |
2.18 |
|
2,888 |
10,635 |
1.17 |
1,408 |
21.80 |
8.32 |
2.09 |
|
2,109 |
11,781 |
1.47 |
1,094 |
22.69 |
8.26 |
1.89 |
|
2,530 |
9,718 |
1.43 |
974 |
24.89 |
13.34 |
2.53 |
|
4,101 |
10,484 |
1.15 |
1,700 |
25.20 |
8.18 |
2.80 |
|
5,772 |
10,181 |
1.73 |
2,479 |
23.46 |
9.69 |
2.10 |
|
2,128 |
11,432 |
1.34 |
1,033 |
22.94 |
7.71 |
3.48 |
|
8,305 |
9,836 |
.80 |
3,238 |
25.04 |
12.11 |
2.84 |
|
2,518 |
12,374 |
1.60 |
1,333 |
24.17 |
8.57 |
2.48 |
|
8,341 |
9,789 |
1.48 |
3,476 |
23.33 |
8.18 |
2.23 |
|
9,957 |
9,349 |
1.28 |
3,559 |
23.22 |
9.47 |
2.59 |
|
3,464 |
9,889 |
.75 |
1,358 |
25.29 |
11.73 |
—d |
|
4,374 |
8,897 |
1.16 |
1,978 |
26.09 |
13.82 |
4.11 |
|
2,771 |
10,544 |
.74 |
1,147 |
25.50 |
7.80 |
2.59 |
|
8,857 |
9,440 |
.46 |
3,386 |
24.70 |
7.11 |
3.04 |
|
2,840 |
9,877 |
.69 |
1,339 |
20.83 |
6.74 |
2.83 |
|
1,969 |
10,369 |
1.17 |
1,110 |
18.38 |
5.77 |
4.48 |
|
8,068 |
10,444 |
1.06 |
3,819 |
20.92 |
6.95 |
2.95 |
|
5,469 |
10,503 |
1.36 |
2,982 |
19.37 |
6.29 |
4.02 |
|
1,798 |
13,176 |
1.51 |
1,114 |
21.26 |
7.71 |
3.98 |
|
4,438 |
10,261 |
1.24 |
2,131 |
20.91 |
8.12 |
2.45 |
|
4,606 |
10,273 |
1.61 |
2,561 |
18.47 |
10.79 |
2.58 |
|
6,999 |
9,986 |
1.22 |
3,102 |
22.75 |
7.98 |
2.71 |
|
3,796 |
9,969 |
.55 |
1,842 |
19.92 |
4.25 |
4.02 |
|
2,055 |
13,025 |
2.60 |
1,116 |
22.53 |
7.88 |
2.34 |
|
9,530 |
9,390 |
.59 |
4,108 |
21.60 |
6.94 |
3.56 |
|
5,974 |
9,379 |
1.01 |
2,501 |
21.76 |
7.24 |
3.18 |
|
4,369 |
9,525 |
.57 |
1,880 |
21.90 |
6.00 |
3.41 |
|
2,714 |
10,022 |
1.22 |
1,186 |
22.36 |
7.12 |
3.60 |
|
3,274 |
9,967 |
.74 |
1,396 |
21.16 |
6.23 |
3.15 |
|
4,582 |
9,744 |
.80 |
2,140 |
21.30 |
5.27 |
2.33 |
|
3,257 |
10,568 |
1.31 |
1,508 |
22.41 |
6.67 |
3.56 |
|
4,731 |
10,170 |
1.49 |
2,187 |
21.85 |
5.86 |
2.72 |
|
3,971 |
9,071 |
.80 |
1,497 |
24.00 |
9.10 |
3.39 |
|
2,874 |
10,756 |
1.27 |
1,190 |
25.89 |
12.10 |
2.85 |
|
3,490 |
9,161 |
.72 |
1,290 |
24.71 |
11.61 |
2.10 |
|
3,521 |
9,590 |
1.64 |
1,298 |
25.38 |
10.85 |
2.82 |
|
12.386 |
9,110 |
.59 |
4,508 |
23.84 |
10.39 |
3.06 |
|
3,743 |
10,042 |
1.05 |
1,767 |
21.08 |
7.81 |
NA |
|
2,247 |
9,681 |
.44 |
1,027 |
21.16 |
7.79 |
NA |
|
1,811 |
10,370 |
1.87 |
808 |
22.13 |
9.82 |
3.27 |
|
5,957 |
9,869 |
.98 |
2,448 |
23.45 |
9.69 |
2.58 |
|
5,491 |
9,853 |
.84 |
2,179 |
23.62 |
10.62 |
3.04 |
|
2,597 |
11,338 |
1.55 |
1,228 |
23.98 |
11.14 |
2.84 |
|
2,503 |
10,425 |
1.64 |
1,229 |
21.20 |
7.02 |
4.51 |
|
9,041 |
9,028 |
.44 |
3,624 |
22.46 |
8.30 |
1.54 |
|
2,642 |
10,828 |
1.47 |
1,243 |
22.99 |
9.03 |
3.23 |
|
12,735 |
9,449 |
.95 |
5,247 |
22.96 |
8.04 |
2.68 |
|
7,180 |
9,088 |
.64 |
2,861 |
22.70 |
7.46 |
2.94 |
|
8,903 |
9,404 |
.73 |
4,044 |
20.65 |
8.00 |
4.91 |
|
1,101 |
12,002 |
3.78 |
655 |
20.10 |
6.33 |
3.90 |
|
7,173 |
9,405 |
.63 |
3,051 |
22.88 |
7.07 |
3.93 |
|
4,192 |
9,301 |
.85 |
1,610 |
24.03 |
10.81 |
2.11 |
|
2,381 |
10,212 |
1.60 |
1,055 |
22.98 |
10.16 |
2.09 |
|
1,517 |
11,575 |
2.51 |
720 |
24.38 |
11.65 |
2.98 |
|
5,365 |
9,567 |
1.04 |
2,207 |
23.05 |
10.16 |
2.08 |
|
3,310 |
9,608 |
.70 |
1,476 |
21.54 |
8.68 |
3.12 |
|
3,346 |
10,584 |
.77 |
1,851 |
19.11 |
5.02 |
5.52 |
|
2,053 |
10,638 |
1.47 |
918 |
23.79 |
7.08 |
3.67 |
|
4,665 |
9,971 |
1.25 |
2,045 |
22.44 |
6.20 |
2.99 |
|
5,021 |
10,260 |
1.27 |
2,208 |
23.26 |
8.75 |
2.46 |
|
3,712 |
10,252 |
2.04 |
1,729 |
21.95 |
6.92 |
3.30 |
|
3,195 |
9,643 |
.36 |
1,539 |
20.01 |
9.75 |
4.25 |
|
Annual Generation (Million KWH) |
Heat Rate (BTU/KWH) |
Operating Costs, Exclusive of Fuel (Mills/KWH) |
Coal Consumption (1000 tons) |
Avg. Heat Content (Million BTU/ton) |
Cost per Ton as Burned ($) |
Avg. Sulfur Content (%)c |
|
1,977 |
10,581 |
.88 |
929 |
22.51 |
8.95 |
3.38 |
|
5,136 |
10,460 |
1.40 |
2,421 |
22.19 |
8.78 |
3.40 |
|
4,192 |
9,872 |
.55 |
1,778 |
23.01 |
8.41 |
3.24 |
|
3,893 |
11,126 |
.88 |
1,894 |
22.83 |
11.22 |
2.40 |
|
7,418 |
9,724 |
.65 |
2,979 |
24.23 |
10.85 |
1.17 |
|
3,403 |
9,655 |
1.35 |
1,297 |
24.94 |
13.81 |
2.12 |
|
|
||||||
|
2,119 |
9,959 |
.66 |
858 |
24.45 |
12.63 |
1.40 |
|
2,171 |
10,094 |
1.10 |
828 |
25.46 |
12.71 |
1.00 |
|
6,425 |
9,461 |
.58 |
2,438 |
24.88 |
10.25 |
1.40 |
|
2,308 |
9,585 |
.74 |
902 |
24.44 |
10.70 |
1.36 |
|
8,089 |
9,332 |
.35 |
3,230 |
23.37 |
10.69 |
1.09 |
|
2,256 |
10,814 |
1.24 |
1,020 |
24.18 |
11.14 |
.94 |
|
3,607 |
10,271 |
.45 |
1,498 |
24.74 |
9.48 |
1.11 |
|
14,227 |
8,670 |
.28 |
5,286 |
22.98 |
10.07 |
.91 |
|
3,185 |
10,605 |
.91 |
1,395 |
23.94 |
10.55 |
1.10 |
|
3,789 |
9,078 |
.45 |
1,380 |
24.63 |
11.59 |
1.50 |
|
5,485 |
8,959 |
.37 |
2,069 |
24.06 |
7.13 |
.71 |
|
2,158 |
9,744 |
.78 |
846 |
24.39 |
9.19 |
.89 |
|
2,511 |
10,143 |
1.11 |
967 |
26.11 |
15.74 |
.98 |
|
8,675 |
8,959 |
.37 |
3,115 |
24.73 |
9.61 |
1.00 |
|
3,161 |
9,186 |
.54 |
1,239 |
23.41 |
8.72 |
.89 |
|
6,343 |
9,124 |
.98 |
2,618 |
21.96 |
8.74 |
1.38 |
|
7,417 |
9,046 |
.55 |
2,763 |
24.24 |
6.94 |
3.07 |
|
3,962 |
9,865 |
.80 |
1,629 |
23.98 |
7.99 |
4.00 |
|
6,892 |
9,461 |
.69 |
2,750 |
23.37 |
7.82 |
3.34 |
|
6,261 |
10,153 |
.71 |
2,753 |
22.59 |
8.36 |
2.14 |
|
7,319 |
10,037 |
.84 |
3,081 |
23.84 |
9.77 |
2.61 |
|
4,896 |
10,681 |
.88 |
2,242 |
23.32 |
8.60 |
1.43 |
|
3,067 |
9,595 |
.80 |
1,228 |
23.96 |
7.87 |
1.88 |
|
6,845 |
9,462 |
.46 |
2,795 |
23.18 |
9.67 |
1.07 |
|
4,017 |
9,850 |
1.02 |
1,770 |
22.34 |
7.73 |
4.17 |
|
1,451 |
9,910 |
1.61 |
643 |
22.32 |
7.57 |
4.19 |
|
3,631 |
10,800 |
1.14 |
1,721 |
22.70 |
7.32 |
2.40 |
|
4,968 |
9,730 |
1.06 |
2,157 |
22.36 |
7.28 |
3.21 |
|
6,720 |
9,476 |
.69 |
2,661 |
23.29 |
8.18 |
1.04 |
|
2,796 |
10,431 |
.50 |
1,231 |
23.68 |
9.55 |
2.28 |
|
8,030 |
9,480 |
1.06 |
3,743 |
20.32 |
3.64 |
NA |
|
6,314 |
9,300 |
.80 |
2,871 |
20.38 |
3.59 |
NA |
|
9,798 |
9,870 |
.65 |
4,638 |
20.82 |
6.05 |
2.81 |
|
3,168 |
10,200 |
.50 |
1,476 |
21.51 |
5.28 |
3.85 |
|
5,082 |
9,100 |
.63 |
2,025 |
22.64 |
6.23 |
1.64 |
|
5,782 |
9,440 |
.82 |
2,514 |
21.68 |
7.49 |
3.33 |
|
6,006 |
10,410 |
.97 |
2,802 |
22.28 |
6.64 |
3.72 |
|
9,356 |
9,560 |
.67 |
4,095 |
21.82 |
8.27 |
2.05 |
|
3,797 |
9,570 |
.75 |
1,579 |
22.98 |
8.72 |
1.77 |
|
2,461 |
10,610 |
1.09 |
1,790 |
14.00 |
2.94 |
.60 |
|
5,148 |
10,417 |
.52 |
1,599 |
21.71 |
6.23 |
.50 |
|
9,846 |
9,766 |
.82 |
5,262 |
18.03 |
2.71 |
.65 |
|
3,284 |
10,577 |
.89 |
2,336 |
14.84 |
1.53 |
.62 |
|
c Sulfur content data is for the year 1971 and hence cannot be directly compared with the other data. Source: Steam-Electric Plant Air and Water Quality Control Data, 1971 [ ]. d New plant initially reported in 1972. |
||||||
TABLE APP. 13-B2
LARGE COAL FIRED POWER PLANTS-NEW PLANTS OR UNITS PLANNED OR UNDER CONSTRUCTION, 1973–1979a
|
Indiana |
|
|||
|
Indianapolis |
Indianapolis Power & Light Co. |
E.W.Stout #7 |
450 |
73 |
|
Petersburg |
|
Petersburg #3–4 |
515 |
77 |
|
Michigan City |
Northern Indiana Pub.Ser.Co. |
Michigan City #12 |
504 |
73 |
|
Unassigned |
|
Northwest Ind. #15 |
500 |
73 |
|
Wheatfield |
|
R.M.Schahfer #14 |
500 |
75 |
|
Nr. Princeton |
Public Service Co. of Indiana |
Gibson #1–4 |
650/650 |
75/76 |
|
Michigan |
|
|||
|
West Olive |
Consumers Power Co. |
Campbell #3 |
800 |
77 |
|
Monroe |
Detroit Edison Co. |
Monroe #2–4 |
1,578/786 |
73/74 |
|
Ohio |
|
|||
|
North Bend |
Cincinnati Gas & Electric Co. |
Miami Fort #7&8 |
500/500 |
75/77 |
|
Conesville |
Columbus & South Ohio Elec. Co. |
Conesville #4–6 |
842/403 |
73/77 |
|
Brilliant |
Ohio Power Co. |
Cardinal #3 |
615 |
76 |
|
Cheshire |
|
Gavin #1&2 |
1,500/1,30 |
74/75 |
|
Aberdine |
Dayton Power & Light Co. |
J.M.Stuart #4 |
580 |
74 |
|
Wisconsin |
|
|||
|
Portage |
Wisconsin Power & Light Co. |
Columbia #1 |
527 |
75 |
|
WEST NORTH CENTRAL |
||||
|
Iowa |
|
|||
|
Council Bluffs |
Iowa Power & Light Co. |
Council Bluffs #3 |
600 |
79 |
|
Sioux City |
Iowa Public Service Co. |
Neal #3 |
520 |
76 |
|
Region State City |
Company |
Plant |
Capacity MW |
In Service Date (19–) |
|
Kansas |
|
|||
|
Linn County |
Kansas Gas & Electric |
La Cygne #1&2 |
840/630 |
73/77 |
|
Topeka |
Kansas Power & Light Co. |
Unassigned |
700 |
73 |
|
Minnesota |
|
|||
|
NA |
Northern States Power Co. |
Base Load #1 |
800 |
79 |
|
Becker |
|
Sherbourne #1&2 |
680/720 |
76/77 |
|
Missouri |
|
|||
|
New Madrid |
Associated Electric Corp. |
New Madrid #2 |
600 |
77 |
|
Labadie |
Union Electric Co. |
Labadie #4 |
582 |
73 |
|
Crystal City |
|
Rush Island #1&2 |
592/592 |
75/76 |
|
Nebraska |
|
|||
|
NA |
Nebraska Public Power District |
Gentleman |
600 |
77 |
|
North Dakota |
|
|||
|
Center |
Minnkota Power Coop Inc. |
Milton Young |
400 |
77 |
|
Stanton |
Basin Electric Power Corp. |
Leland Olds #2 |
460 |
75 |
|
South Dakota |
|
|||
|
Grant |
Otter Tail Power Co. |
Big Stone #1 |
430 |
75 |
|
SOUTH ATLANTIC |
|
|||
|
Florida |
|
|||
|
Pensacola |
Gulf Power Co. |
Crest #7 |
516 |
73 |
|
Tampa |
Tampa Electric Co. |
Big Bend #2 |
425/425 |
73/75 |
|
Georgia |
|
|||
|
Centerville |
Georgia Power Co. |
Bowen #3&4 |
910/910 |
74/75 |
|
Carrollton |
|
Wansley #1&2 |
910/952 |
76/77 |
|
Newman |
|
Yates #6&7 |
725 |
74 |
|
Maryland |
|
|||
|
Morgantown |
Potomac Electric Power Co. |
Morgantown #3 |
550 |
79 |
|
North Carolina |
|
|||
|
North Winston |
Duke Power Co. |
Belews Creek #1&2 |
1,143/1,143 |
74/75 |
|
Roxboro |
Carolina Power & Light Co. |
Roxboro |
720/72 |
73/76 |
|
Virginia |
|
|||
|
Yorktown |
Virginia Electric & Power Co. |
Yorktown #3 |
845 |
74 |
|
Region State City |
Company |
Plant |
Capacity MW |
In Service Date (19–) |
|
West Virginia |
|
|||
|
Scary |
Appalachian Power Co. |
J.E.Amos #3 |
1,300 |
73 |
|
Haywood |
Monongahela Power Co. |
Harrison #2&3 |
650/650 |
73/75 |
|
Mt. Storm |
Virginia Elec. & Power Co. |
Mt. Storm |
575 |
73 |
|
EAST SOUTH CENTRAL |
||||
|
Alabama |
|
|||
|
Jefferson Co. |
Alabama Power Co. |
West Jefferson #1&2 |
880/880 |
73/79 |
|
Wilsonville |
So. Electric Generating Co. |
Gaston #5 |
910 |
74 |
|
Kentucky |
|
|||
|
Maysville |
East Kentucky Rural Elec. Coop. |
Ohio River #1 |
300 |
76 |
|
Sebree |
Henderson Municipal Utilities |
No. 2#1&2 |
334 |
73 |
|
Ghent |
Kentucky Utilities Co. |
Ghent #1&2 |
500/500 |
74/77 |
|
Louisville |
Louisville Gas & Elec. |
Mill Creek #2–4 |
336/425 |
74/77 |
|
Mississippi |
|
|||
|
Gulfport |
Mississippi Power Co. |
Watson #5 |
520 |
73 |
|
Tennessee |
|
|||
|
Cumberland City |
Tennessee Valley Authority |
Cumberland #2 |
1,275 |
73 |
|
WEST SOUTH CENTRAL |
||||
|
Louisiana |
|
|||
|
NA |
Central Louisiana Elec. Co. |
Unassigned |
550 |
78 |
|
Oklahoma |
|
|||
|
Muskogee |
Oklahoma Gas & Elec. |
Muskogee #4&5 |
550/550 |
77/73 |
|
Texas |
|
|||
|
Cason |
Southwestern Elec. Power Co. |
Cason #1&2 |
530/528 |
77/73 |
|
Tatum Rusk Co. |
Texas Utilities Co. |
Martin Lake #1&2 |
750/750 |
76/77 |
|
Mt. Pleasant |
|
Monticello #1&2 |
575/575 |
74/75 |
|
MOUNTAIN |
|
|||
|
Region State City |
Company |
Plant |
Capacity MW |
In Service Date (19–) |
|
Arizona |
|
|||
|
Page |
Salt River (AIP) District Project |
Navajo #1–3 |
750/750/750 |
74/75/76 |
|
Montana |
|
|||
|
Colstrip |
Montana Power Co. |
Colstrip #1–4 |
700/700 |
73/79 |
|
Nevada |
|
|||
|
NA |
Nevada Power Co. |
Arrow Canyon #1&2 |
500/500 |
77/78 |
|
New Mexico |
|
|||
|
Farmington |
Public Service Co. of N.M. |
San Juan #1–3 |
500 |
73 |
|
Utah |
|
|||
|
Huntington |
Utah Power & Light Co. |
Huntington Canyon #1&2 |
430/400 |
74/74 |
|
Wyoming |
|
|||
|
Rock Springs |
Idaho Power Cp |
Bridget #1–3 |
500/500/500 |
74/75/76 |
TABLE App. 13-B3
OIL FIRED PLANTS CONVERTIBLE TO COALa
|
MIDDLE ATLANTIC STATES |
|||
|
New Jersey |
|
||
|
Deepwater |
Atlantic City Electric Co. |
Deepwater |
202 |
|
Beesleys |
|
England |
299 |
|
Sayreville |
Jersey Central Power & Light Co. |
Sayreville |
248 |
|
S.Amboy |
|
Werner |
60 |
|
Holland TWP |
|
Gilbert |
126 |
|
Ridgefield |
Public Service Electric & Gas Co. |
Bergen |
650 |
|
Burlington |
|
Burlington |
481 |
|
Jersey City |
|
Hudson |
455 |
|
Kearney |
|
Kearney |
294 |
|
Sewaren |
|
Sewaren |
431 |
|
Vineland |
Vineland Electric Util. |
Down |
61 |
|
New York |
|
||
|
New York |
Consolidated Edison Co. |
Arthur Kill |
826 |
|
New York |
|
Astoria |
1,455 |
|
New York |
|
Ravenswood |
1,000 |
|
Island Park |
Long Island Lighting Co. |
Barrett |
187 |
|
Bethlehem |
Niagara Mohawk Power Co. |
Albany |
400 |
|
Tomkins Cove |
Orange & Rockland Util. |
Lovett |
495 |
|
Pennsylvania |
|
||
|
W.Norristown |
Philadelphia Electric Co. |
Barbadoes |
148 |
|
E.Pikeland |
|
Cromby |
230 |
|
Philadelphia |
|
Delaware |
272 |
|
Peach Bottom |
|
Southwark |
378 |
|
Region State City |
Company |
Plant |
Capacity Convertible MW |
|
SOUTH ATLANTIC STATES |
|||
|
Delaware |
|
||
|
Delaware City |
Delmarva Power & Light Co. |
Delaware City No. 3 |
38 |
|
Wilmington |
|
Edge Moor |
389 |
|
Georgia |
|
||
|
Brunswick |
Georgia Power Co. |
McManus |
140 |
|
Maryland |
|
||
|
Baltimore |
Baltimore Gas & Electric Co. |
Crane |
400 |
|
Baltimore |
Wagner |
268 |
|
|
Vienna |
Delmarva Power & Light |
Vienna |
67 |
|
Aquasco |
Potomac Electric Power Co. |
Chalk Point |
728 |
|
Newburg |
Morgantown |
1,114 |
|
|
North Carolina |
|
||
|
Wilmington |
Carolina Power & Light Co. |
Sutton |
671 |
|
Virginia |
|
||
|
Chester |
Virginia Electric Power Co. |
Chesterfield |
1,485 |
|
Chesapeake |
|
Portsmouth |
650 |
|
Dumfries |
|
Possum Point |
491 |
|
Yorktown |
|
Yorktown |
375 |
APPENDIX 13-C
TABLE APP. 13-C1
NATIONAL AIR SURVEILLANCE NETWORKS
URBAN SULFATE OBSERVATIONS
ANNUAL AVERAGE SULFATE CONCENTRATION, μg/m3
|
|
Year |
|
|
Station |
1969 |
1970 |
|
Bridgeport, Conn. |
11.9 |
11.6 |
|
Hartford, Conn. |
12.1 |
13.9 |
|
New Haven, Conn. |
20.0 |
20.9 |
|
Waterbury, Conn. |
13.6 |
15.1 |
|
Newark, Del. |
n.a. |
13.0 |
|
Wilmington, Del. |
19.2 |
13.7 |
|
Washington, D.C. |
13.9 |
— |
|
Baltimore, Md. |
13.9 |
19.8 |
|
Portland, Maine |
— |
17.0 |
|
Boston, Mass. |
15.3 |
— |
|
Fall River, Mass. |
12.4 |
14.9 |
|
Springfield, Mass. |
8.5 |
12.5 |
|
Worchester, Mass. |
13.0 |
15.5 |
|
Concord, N.H. |
6.5 |
9.2 |
|
Burlington Co., N.J. |
13.2 |
10.3 |
|
Camden, N.J. |
22.0 |
17.2 |
|
Elizabeth, N.J. |
11.5 |
13.2 |
|
Glassboro, N.J. |
13.4 |
12.6 |
|
Hamilton, N.J. |
9.9 |
— |
|
Jersey City, N.J. |
12.1 |
14.5 |
|
Newark, N.J. |
11.2 |
11.8 |
|
Paterson, N.J. |
12.7 |
12.7 |
|
Perth Amboy, N.J. |
13.3 |
9.9 |
|
Trenton, N.J. |
12.8 |
15.0 |
|
Albany, N.Y. |
9.4 |
— |
|
Buffalo, N.Y. |
11.4 |
16.9 |
|
New York City, N.Y. |
19.1 |
22.2 |
TABLE App. 13-C2
NASN SULFATE OBSERVATIONS—NONURBAN STATIONS
|
|
AVERAGE ANNUAL SULFATE CONCENTRATION, μg/m3 |
||
|
Station |
1968 |
1969 |
1970 |
|
Kent County, Delaware |
9.5 |
n.a. |
n.a. |
|
Monroe County, Indiana |
9.7 |
7.9 |
7.5 |
|
Park County, Indiana |
6.9 |
8.5 |
13.5 |
|
Acadia Nat. Park, Maine |
5.8 |
4.9 |
7.0 |
|
Calvert County, Maryland |
14.1 |
9.8 |
n.a. |
|
Coos County, New Hampshire |
7.3 |
3.4 |
6.5 |
|
Jefferson County, New York |
10.0 |
9.1 |
9.4 |
|
Clarion County, Pennsylvania |
9.8 |
9.1 |
12.4 |
|
Washington County, Rhode Island |
10.5 |
9.1 |
7.7 |
|
Orange County, Vermont |
7.5 |
5.8 |
8.0 |
|
Shenandoah Nat. Park, Virginia |
8.1 |
13.3 |
9.2 |
|
Source: 1968 data: U.S. EPA publication, “Air Quality Data from the National Air Surveillance Networks and Contributing State and Local Networks.” 1969–1970 data: Air Quality Data for Nonmetallic Inorganic Ions, 1969 and 1970, National Air Surveillance Networks, APTD-1466, U.S. Environmental Protection Agency, June 1973. |
|||
|
NASN URBAN SITES |
||
|
ANNUAL AVERAGE SULFATE CONCENTRATION μg/m3 |
||
|
Station |
1969 |
1970 |
|
Canton, Ohio |
11.9 |
16.7 |
|
Cincinnati, Ohio |
15.1 |
12.4 |
|
Cleveland, Ohio |
15.9 |
18.0 |
|
Columbus, Ohio |
14.4 |
— |
|
Dayton, Ohio |
15.6 |
11.9 |
|
Toledo, Ohio |
10.2 |
12.9 |
|
Youngstown, Ohio |
11.8 |
16.8 |
|
Allentown, Pa. |
16.8 |
20.4 |
|
Altoona, Pa. |
10.2 |
30.2 |
|
Bethlehem, Pa. |
11.2 |
21.0 |
|
Erie, Pa. |
15.6 |
16.5 |
|
Harrisburg, Pa. |
10.2 |
16.0 |
|
Hazelton, Pa. |
9.9 |
— |
|
Johnstown, Pa. |
15.3 |
16.9 |
|
Philadelphia, Pa. |
— |
21.9 |
|
Pittsburgh, Pa. |
— |
17.9 |
|
Reading, Pa. |
— |
18.6 |
|
Scranton, Pa. |
— |
13.9 |
|
Warminster, Pa. |
— |
10.9 |
|
Wilkes-Barre, Pa. |
— |
13.7 |
|
York, Pa. |
— |
14.3 |
|
East Providence, R.I. |
15.5 |
12.0 |
|
Providence, R.I. |
11.9 |
12.9 |
|
Burlington, Vt. |
10.9 |
12.0 |
|
Charleston, West Va. |
25.8 |
25.0 |
|
So. Charleston, West Va. |
— |
15.5 |
|
Source: Air Quality Data for Nonmetallic Inorganic Ions, 1969 & 1970, National Air Surveil lance Networks, APTD-1466, U.S. Environmental Protection Agency, June 1973. |
||
APPENDIX 13-D
COST CALCULATIONS FOR ELECTRIC POWER GENERATION WITH AND WITHOUT FLUE GAS DESULFURIZATION
Since calculations have not been included elsewhere in the report, this appendix is provided to give a detailed basis for the cost of flue gas desulfurization using the lime scrubbing process. These costs are used in Table 13–5 of this chapter, in the Executive Summary, and in Part Two in Brief. The calculations assume the use of coal of approximately 3 percent sulfur and a plant of moderate size, approximately 600 MW. Fixed charges, including amortization, of 17 percent on invested capital are assumed for new investment, and fixed charges on existing plant capacity are computed at 14 percent. Other needed assumptions are as specified in Tables 13–1 and 13–3. All numbers are rounded to the nearest 0.1 mill, so some columns do not sum exactly.
TABLE App. 13-D1
Cost Calculation for New Plant
|
Without flue gas desulfurization: |
|
|
capital cost: $500/kw×0.17÷7000 hours |
12.1 mills/kwh |
|
fuel cost: heat rate of 8,982×$1/MM BTU of fuel |
8.9 |
|
other operating costs: |
0.5 |
|
Cost of power generation without scrubber |
21.6 mills/kwh |
|
Added cost of lime scrubbing process: |
|
|
capital cost: $100/kw×0.17÷7000 hours |
2.4 mills/kwh |
|
operating cost: labor, chemicals, etc. |
0.5 |
|
sludge disposal |
0.3 |
|
Subtotal |
3.2 mills/kwh |
|
energy loss of 6% of plant output needed for scrubber operation: |
0.6 |
|
capacity derating of plant resulting from 6% loss in output capacity: |
0.7 |
|
Subtotal |
1.3 |
|
added cost of lime scrubbing process |
4.5 mills/kwh |
|
Cost of power generation with scrubber: |
26.2 mills/kwh |
TABLE App. 13-D2
New Plant: Sensitivity Calculations on Added Cost from Scrubber
|
Low case: |
|
|
$60/kw; 0.15 mills/kwh for sludge disposal |
2.1 mills/kwh |
|
5% energy loss and capacity derating |
1.1 |
|
Added cost of scrubber, low case: |
3.2 mills/kwh |
|
High case: |
|
|
$130/kw; 1.0 mills/kwh for sludge disposal |
4.7 mills/kwh |
|
7% energy loss and capacity derating |
1.5 |
|
Added cost of scrubber, high case: |
6.2 mills/kwh |
TABLE App. 13-D3
Cost Calculation for Retrofit to Existing Plant, Rural Location
|
Without flue gas desulfurization: |
|
|
capital cost $250/kw×0.14÷6000 hours |
5.8 mills/kwh |
|
fuel costs: heat rate of 10,342×$1/MM BTU of fuel |
10.3 |
|
other operating costs |
1.0 |
|
Cost of power generation without scrubber |
17.2 mills/kwh |
|
Added cost of lime scrubbing process: |
|
|
capital cost of $125/kw×0.17÷6000 hours |
3.5 mills/kwh |
|
operating cost: labor, chemicals, etc. |
0.6 |
|
sludge disposal |
0.5 |
|
Subtotal |
4.6 mills/kwh |
|
energy loss, 6% of plant output needed for scrubber operation |
0.7 |
|
capacity derating of 6% (replacement at $500/kw, 17% fixed charge) |
0.8 |
|
Subtotal |
1.5 |
|
added cost of lime scrubbing process |
6.1 mills/kwh |
|
Cost of power generation with scrubber: |
23.3 mills/kwh |
TABLE App. 13-D4
Cost Calculation for Retrofit Installation, Existing Oil Burning Plant Reconverted to Coal, Urban Location
|
Without flue gas desulfurization: |
|
|
capital cost $250/kw×0.14÷6000 hours |
5.8 mills/kwh |
|
conversion charges of $7/kw plus $4/kw for particulate removal upgrade |
0.3 |
|
fuel cost: heat rate of 10,342×$1/MM BTU of fuel |
10.3 |
|
other operating costs |
1.0 |
|
Cost of power generation without scrubber |
17.5 mills/kwh |
|
Added cost of lime scrubbing process: |
|
|
capital cost of $125/kw×0.17÷6000 hours |
3.5 mills/kwh |
|
less credit of $4/kw for particulate upgrade |
−.1 |
|
operating cost: labor, chemicals, etc. |
0.6 |
|
sludge disposal |
0.9 |
|
Subtotal |
4.9 mills/kwh |
|
energy loss, 6% of plant output needed for scrubber operation |
0.7 |
|
capacity derating of 6% (replacement at $500/kw, 17% fixed charge) |
0.8 |
|
Subtotal |
1.5 |
|
added cost of lime scrubbing process |
6.4 mills/kwh |
|
Cost of power generation with scrubber: |
23.9 mills/kwh |
APPENDIX 13-E
COMMENTS ON ESTIMATES OF MATERIAL DAMAGE
(Written by I.C.T.Nisbet, a member of the Review Committee on Air Quality and Power Plant Emissions.)
In their discussion of damage to man-made materials attributable to sulfur dioxide and sulfates, North and Merkhofer (Chapter 13) relied for numerical estimates upon the review by Waddell 1974. The following comments are offered not in criticism of the work of Waddell, North and Merkhofer, who used the best data available, but to point out that the available data are severely limited and that Waddell’s estimates of damage may be very conservative.
Waddell (1974, pp. 90–91, 128–130) estimated that the total damage caused by sulfur oxides to material property in 1970 was about $600 million (with a possible range of $400–800 million). This estimate was based primarily on studies by Gillette (1973) of damage to galvanized and painted steels, by Spence and Haynie (1972) of damage to other painted surfaces, and by Salmon (1970) of damage to non-ferrous metals and miscellaneous materials.
DAMAGE TO GALVANIZED AND PAINTED STEELS
Gillette’s (1973) estimates were based on the assumption that corrosion rates are proportional to average ambient concentrations of sulfur dioxide, and in the case of galvanized steels are also dependent on the relative humidity. Accordingly Gillette deduced that most of the damage to these materials was taking place in urbanized regions of the northeastern and northcentral states, and that damage has been decreasing rapidly as ambient sulfur dioxide levels have been reduced. His estimates of total annual damage in the United States fell from $909 million
in 1968 to $397 million in 1970 and to $75 million in 1972 (Gillette 1973, Table IV).
However, it is probable that some of the damage should be attributed to acid sulfate particulates and/or to sulfuric acid in rain. As pointed out by Gillete himself (1973, p. 4), the agent primarily responsible for damage is probably sulfuric acid, either formed by oxidation in situ after absorption of sulfur dioxide onto moist surfaces, or deposited in acid particulates or acid precipitation. If even half of the damage were attributable to sulfate particulates or acid rain, Gillette’s deduction of a drastic decrease in damage between 1968 and 1972 would be incorrect and his estimate for 1970 would be too low. Moreover if acid rain were a significant cause of corrosion, significant damage would also be predicted to occur to rural areas and in smaller towns, where Gillette’s damage function predicts negligible damage because of low ambient sulfur dioxide levels.
It should be noted also that Gillette’s estimate of about $300 million for total damage to galvanized materials in the U.S. in 1970 is much lower than estimates made independently by Salmon (1970:$778 million) and by Fink et al. (1972:$1,350 million). A study in Sweden (Bolin et al. 1971) gave rise to estimates more similar to those of Gillette—about $16 million per year, or $2 per capita—for galvanized steel. However, the Swedish study yielded estimates of damage to painted steels substantially larger than those derived by Gillette—about $25 per capita annually in Sweden, of which roughly 10 percent was attributable to sulfur oxides (Bolin et al. 1971: Tables 4.7 and 7.4), versus Gillette’s estimate of $0.88 per capita for the U.S.
DAMAGE TO OTHER PAINTED MATERIALS
Waddell (1974, p. 128) used a figure of $200 million for damage caused by air pollutants to other painted materials, based on a study by Spence and Haynie (1972). However, Spence and Haynie actually estimated total losses as $700 million, of which $540 million was attributable
to deterioration of household exterior paints (Waddell 1973, Table 14). Hence Waddell appears to have overcorrected for overlap between this and Gillette’s study, which considered only painted metal surfaces.
Moreover, Spence and Haynie’s estimates of damage appear to be based on very low figures for the total value of paintwork at risk from air pollution. Their table indicates that the total in-place value of exterior paints in the categories considered (including the labor value of painting) was only $2.5 billion, of which household exterior paints accounted for only $1.5 billion. This contrasts with a figure of $23.9 billion used by Salmon (1970) for the in-place value of exposed paints in the United States, and a figure of $2.9 billion for the total expenditure of residential property owners on maintenance painting in 1972 alone (Stat. Abstract U.S. 1973).
In addition, Waddell’s attribution of half the total damage to sulfur oxides and half to particulates appears somewhat arbitrary. Apart from soiling and staining, most particulate matter would not be expected to damage paint, except insofar as it retains materials such as acid sulfates and nitric acid and thereby helps to maintain their contact with painted surfaces.
Waddell’s final estimate of $100 million for the total amount of damage caused by sulfur oxides to painted surfaces corresponds to costs of no more than $0.80 per capita per year, even if all the damage is supposed to occur in the northeastern states. This is less than the estimates derived from the Swedish study, in which the total cost of deterioration of painted woodwork was estimated as $23 per capita in 1970, of which at least 10 percent was associated with exposure to sulfur oxides (Bolin et al. 1971: Tables 4.6 and 7.4).
DAMAGE TO NON-FERROUS METALS
Based on the study by Salmon (1970), Waddell (1974, pp. 90–91) adopted a figure of $400 million for the cost of air pollution damage to other
materials, primarily non-ferrous metals. Of this, he ascribed one-quarter to sulfur oxides, one quarter to particulates, and one-half to oxidants (Waddell 1974, pp. 128–130). This division appears questionable because others have attributed accelerated corrosion of metals primarily to sulfur oxides and acids (Salmon 1970, Bolin et al. 1971). Moreover Waddell excluded damage to copper, which is classically associated with air pollution.
DAMAGE TO AUTOMOBILES
None of the studies cited above explicitly considered damage to motor vehicles, except that Spence and Haynie (1972) included a figure of $88 million per year for the excess cost of repainting automobiles. However, it is questionable that the cost of repainting is an adequate measure of the economic damage caused by accelerated corrosion. The resale value of automobiles is substantially influenced by exterior appearance, so that accelerated deterioation of paint or metal plated trim contributes disproportionally to depreciation. The number of motor vehicles in the area most exposed to sulfur oxides and acid rain (the northeastern U.S. and southeastern Canada) is in excess of 60 million; their current value is greater than $75 billion, and rates of depreciation are of the order of 20–30 percent annually If air pollution caused even 1 percent of the depreciation in value, this would represent a substantial economic cost not included in the other surveys.
SUMMARIZING COMMENTS
The above discussion illustrates the large uncertainties involved in making estimates of economic damage caused by air pollution from the very limited data available. Waddell’s estimate of total damage, although based on a thorough and comprehensive review, is among the lowest of a range that could be obtained from the same data; an independent review suggests that a figure 4
or 5 times higher would be more consistent with the original sources. The uncertainty is further compounded by our lack of knowledge of exposure-response relationships, and of the relative importance of sulfur dioxide, suspended acid sulfates, and acid precipitation. Clearly this is another area in which the value of resolving uncertainties would exceed the cost of the research required to do so.
DAMAGE TO WORKS OF ART AND HISTORIC BUILDINGS
Sulfur oxides and acid rain also cause damage to works of art, historic buildings and monuments. The damage is almost impossible to assess in economic terms because such objects have historic or artistic value far greater than the cost of replacement; many historic objects have no market value at all. Information supplied by curators of museums and buildings suggests that the materials at greatest risk are paper (rare books, manuscripts, and prints), building stone (limestone and marble), and outdoor sculptures (bronze, steel, and stone). Although it is possible in principle to alleviate damage by protective measures—such as air-conditioning, cleaning and restoration, or protective coatings—in practice insufficient money is available for such measures and many important buildings, monuments, and collections are unprotected. Unless a method can be devised to finance the restoration and preservation of these historic and cultural objects, increased emissions of sulfur oxides are likely to speed their deterioration. Their loss should be regraded as a major intangible cost of sulfur oxide emissions, to be weighed in conjunction with the more tangible costs and benefits.
APPENDIX 13-F
INCOME REDISTRIBUTION AND EQUITY1
As noted briefly by North and Merkhofer (See Chapter 13), the assessment of alternative strategies for pollution control may need to address issues of equity and distribution. The burden imposed by pollution falls unequally on individuals within society, and the various strategies under consideration would redistribute this burden in different ways. Some—although not all—of these redistributional effects can be predicted, and should be taken into account in choosing between the alternative strategies. If the strategy which is identified as socially optimal (on the basis of a comparison between total costs and total benefits to society) involves predictable adverse effects on identifiable individuals or groups within the population, some compensatory actions may be desirable. Alternatively, it may be justified to choose a strategy which is less than socially optimal in the strict economic sense, in order to minimize social inequities. Such questions of social policy are outside the purview of this committee, but it is nevertheless important to point out what is known about redistributional effects.
The adverse effects of sulfur oxides on human health are expected to fall primarily on residents of urban areas in the northeast, where the highest ambient concentrations of both sulfur dioxide and suspended sulfates are found (Chapters 1–4, 6). Similarly, the costs imposed by damage to materials are believed to fall primarily on property owners
in northeastern cities (Appendix Chapter 13-E). Except for localized damage to vegetation close to some point sources, effects in rural areas appear to be generally smaller. However, the exact magnitude of the urban-rural difference in impact is difficult to specify, because of our lack of knowledge of exposure-response relations. For example, if the threshold level for health effects of suspended sulfates is low or zero, substantial effects might be occurring in rural areas of the northeast where ambient concentrations of sulfates are moderately high (Chapter 6). Similarly, if a substantial fraction of the damage to materials is caused by acid rain, it would be taking place in rural areas as well as in cities (Chapter 7).
It is not clear whether the costs of pollution would fall disproportionately on different income groups. To the extent that health and material damage effects fall on residents of cities, they might fall disproportionately on the poor. Also, many of the health effects fall on individuals who are old or chronically sick. However, these differentials are offset by the facts that suburban residents are also exposed to high pollution levels in some areas, and that the rural poor are relatively unaffected. More information is required to resolve this question.
In addition to the urban-rural differences in impact discussed above, there are probably major regional inequities. Chapters 6 and 7 show that sulfur oxides emitted in the east-central and midwestern states contribute significantly to airborne sulfate particulates and acid rain in the northeastern states. A special case of this regional distribution problem is the likely impact of U.S. emissions on eastern Canada. The urbanized region between Windsor and Montreal lies downwind from major emitting areas in the north-central U.S.; it is probable that the high concentrations of sulfates in rain in this region of Canada are attributable in large part to U.S. emissions, since Canadian emissions are comparatively modest (with the exception of the Sudbury smelting complex well to the northwest) (See Chapter 7). Likewise the prospective impact of acid rain on forestry would be most likely to be economically
significant in eastern Canada (Chapter 5).
A general increase in sulfur oxide emissions (as would accur if a Tall Stack-Intermittent Control strategy were widely adopted) is expected to lead to a general increase in ambient concentrations of sulfur dioxide and suspended sulfates, and an increase in impacts in rural areas. However, the models in Appendix A to Chapter 13 indicate that the highest concentrations of suspended sulfates would continue to occur in urban areas, even if emissions upwind were confined to rural sites. Accordingly the urban-rural difference in impacts would probably be maintained without major change. The regional differences would probably be maintained similarly.
Under an emission control strategy (low sulfur fuels and/or flue gas desulfurization) the increase in total exposure to sulfur oxides and sulfates would be limited, and ultimatly reversed if the policy were sufficiently widely implemented. The immediate effect would be to pass on the cost of emission control to consumers in the form of higher prices. This would help to offset the existing regional inequities, because consumers in the central states and in rural and suburban areas in the northeast would be assuming part of the social cost of generating their power that would otherwise be borne by residents in cities downwind. However, it would probably have a regressive income distributional effect, because the poor spend a higher fraction of their income on electricity than the wealthy (Ford Foundation 1974), and so would be affected disproportionately by higher prices.
The distributional effects of the adoption of marginal cost pricing for electricity also need to be considered. To the extent that it would lead immediately to higher prices, its effects are expected to be regressive, as discussed in the previous paragraph. However, this would be offset if the pricing policy involved reduction of the higher rate for the initial amount of electricity used, as might be justified on the grounds that demand for electricity in that sector of the market is inelastic. Peak load pricing (or time-of-day metering) might also offer advantages to poorer individuals, because it permits cost savings
by those with sufficient incentive to do so.
To summarize, an increase in emissions would lead to an increase in external impacts, probably without major change in the pattern of inequities. Adoption of an emission control strategy would relieve existing urban-rural, regional, and international inequities. However, it would probably have a regressive income distributional effect, unless accompanied by a pricing policy designed to reflect price elasticity of demand.
LITERATURE CITED
Amdur, M.O. (1969) Toxicological Appraisal of Particulate Matter, Oxides of Sulfur, and Sulfuric Acid. Air Pollut. Contr. Assoc. 19(9):638–644, September.
Brasser, L.J., P.E.Joosting, and O.von Zuilen (1967) Sulfur Dioxide-To What Level is it Acceptable? Research Institute for Public Health Engineering. Delft, Netherlands, Report G-300, July (Originally published in Dutch, September 1966).
Brosset, Cyril (1973) Air-Borne Acid, Ambio II (1–2), 2.
Buechley, R.W., W.B.Riggan, V.Hasselblad, and J.B.Van Bruggen (1973) Sulfur Dioxide, Levels and Perturbations in Mortality. Arch. Environ. Hlth., 27(3):134.
Bufalini, Marijon (1971) Oxidation of Sulfur Dioxide in Polluted Atmospheres: A Review, Environmental Science and Technology, 5, p. 685, August.
Burns, J.L. and J.Pemberton (1963) Air Pollution, Bronchitis, and Lung Cancer in Salford. Int. J. Air Water Pollut. 7:15.
Carnow, B.W., R.M.Senior, R.Karsh, S.Wesler, and L.V.Avioli (1970) The Role of Air Pollution in Chronic Obstructive Pulmonary Disease. Amer. Med. Assoc. 214(5):894–899, November 2.
Douglas, J.W.B. and R.E.Waller (1966) Air Pollution and Respiratory Infection in Children. Brit. J. Prev. Soc. Med. 20:1–8.
Fink, F.W., F.H.Buttner, and W.K.Boyd (1971) Battelle-Columbus Laboratories. Technical Economic Evaluation of Air Pollution Corrosion Costs in Metals in the United States. Environmental Protection Agency. Research Triangle Park, North Carolina. Final Report, 104 p., February.
Finklea, J.F., D.C.Calafiore, C.J.Nelson, W.B. Riggan, and C.B.Hayes (1974) Aggravation of Asthma by Air Pollutants: 1971 Salt Lake Basin Studies. Health Consequences of Air Pollution: A Report from the CHESS Program, 1970–1971. EPA #650/1–74–004, pp. 2–75, June.
Finklea, J.F., J.H.Farmer, G.J.Love, D.C.Calafiore, and G.W.Sovocool (1974) Aggravation of Asthma by Air Pollutants: 1970–1971 New York Studies. Health Consequences of Air Pollution: A Report from the CHESS Program, 1970–1971. EPA NO. 650/1–74–004, pp. 5–71, June.
Finklea, J.F, D.I.Hammer, D.E.House, C.R.Sharp, W.C.Nelson, and G.R.Lowrimore (1974) Frequency of Acute Lower Respiratory Disease in Children: Retrospective Survey of Five Rocky Mountain Communities. Health Consequences of Air Pollution: A Report from the CHESS Program, 1970–1971. EPA No. 650/1–74–004, pp. 3–35, June.
Freeman, A.Myrick, III, Robert H.Haveman, and Allan V.Kneese (1973) The Economics of Environmental Policy, John Wiley and Sons, Inc., New York.
French, J.G. (1974) U.S. Environmental Protection Agency, Internal Memorandum on 1971–1972 CHESS Studies of Aggravation of Asthma.
Galke, W. and D.E.House (1974a) Prevalence of Chronic Respiratory Disease Symptoms in Adults: 1971–1972 Survey of Two Southeastern United States Communities. EPA intramural report, February.
Galke, W. and D.E.House (1974b) Prevalence of Chronic Respiratory Disease Symptoms in New York Adults—1972. EPA intramural report, February.
Garland, J.A. (1974) Progress Report, 1 May 1973–1 September 1974, U.K. Proposal No. 2, Sorption of Sulfur Dioxide at Land Surfaces.
Gartrell, F.E. et al. (1963) Am. Indust. Hyg. Assoc. T., 24, 113.
Gillette, Donald G. (1973) Sulfur Dioxide Standards and Material Damage, Paper 74–170, presented at the 67th Annual Meeting of the Air Pollution Control Association, Denver, Colorado, U.S. Environmental Protection Agency.
Glasser, M. and L.Greenburg (1969) Air Pollution Mortality and Weather. New York City 1960–1964. Presented at the Epidemiology Section of the Annual Meeting
of the American Public Health Association, Philadelphia, November 11.
Goldberg, H., J.F.Finklea, C.J.Nelson, W.B.Stern, R.S.Chapman, D.H.Swanson, and A.A.Cohen (1974) Prevalence of Chronic Respiratory Symptoms in Adults: 1970 Survey of New York Communities, Health Consequences of Air Pollution: A Report from the CHESS Program, 1970–1971. EPA No. 650/1–74–004, June.
Goldberg, H.E., J.F.Finklea, J.H.Farmer, A.A.Cohen, F.B.Benson, and G.J.Love (1974) Frequency and Severity of Cardiopulmonary Symptoms in Adult Panels: 1970–1971 New York Studies, Health Consequences of Air Pollution: A Report from the CHESS Program, 1970–1971. EPA #650/1–74–004, pp. 5–85, June.
Grennard, Alf and Fraser Ross (1974) Progress Report on Sulfur Dioxide, Combustion, 4, January.
Hammer, D.I. (1974) Frequency of Acute Lower Respiratory Disease in Two Southeastern Communities, 1968–1971. EPA intramural report, March.
Hayes, C.G., D.I.Hammer, C.M.Shy, V.Hasselblad, C.R.Sharp, J.P.Creason, and K.E.McClain (1974) Prevalence of Chronic Respiratory Disease Symptoms in Adults: 1970 Survey of Rocky Mountain Communities, Health Consequences of Air Pollution: A Report from the Chess Program, 1970–1971. EPA No. 650/1–74–004, June.
Hogstrom, U. (1973a) Residence Time of Sulfurous Air Pollutants from a Local Source During Precipitation, Ambio 11.
Hogstrom, U. (1973b) Wet fallout of sulfurous pollutants emitted from a city during rain or snow. Atmospheric Environment, 8, 1291, December.
House, D.E. (1973) Preliminary Report on Prevalence of Chronic Respiratory Disease Symptoms in Adults: 1971 Survey of Four New Jersey Communities. EPA intramural report. May.
House, D.E., J.F.Finklea, C.M.Shy, D.C.Calafiore, W.B.Riggan, J.W.Southwick, and L.J.Olsen (1974) Prevalence of Chronic
Respiratory Disease Symptoms in Adults: 1970 Survey of Salt Lake Basin Communities, Health Consequences of Air Pollution: A Report from the CHESS Program, 1970–1971. EPA No. 650/1–74–004, June.
Howard, Ronald A. (1966) Decision Analysis: Applied Decision Theory, Proceedings of the Fourth International Conference on Operational Research, pp. 55–71, Wiley-Interscience.
Howard, Ronald A. (1968) The Foundation of Decision Analysis, IEEE Transactions on Systems Science and Cybernetics, Vol. SSC-4, No. 3, September.
Jacoby, Henry D., John D.Steinbruner, et al. (1973) Measuring the Value of Emissions Reductions, Chapter 8 of Clearing the Air by William R.Ahern, Jr., Ballinger, Cambridge, Massachusetts, p. 175.
Kellogg, W.W. et al. (1972) The Sulfur Cycle, Science 175, 587.
Lave, Lester B. and Eugene P.Seskin (1970) Air Pollution and Human Health, Science, 169. 723.
Lawther, P.J. (1963) Compliance with the Clean Air Act: Medical Aspects. J. Inst. Fuels (London). 36:341–344.
Lewis, T.R., M.O.Amdur, M.D.Fritzhand, and K.I.Campbell (1972) Toxicology of Atmospheric Sulfur Dioxide Decay Products. U.S. Environmental Protection Agency. Research Triangle Park, North Carolina. Publication Number AP-111, 42 p., July.
Lindeberg, W. (1968) Air Pollution in Norway. III. Correlations between Air Pollutant Concentrations and Death Rates in Oslo. Published by the Smoke Damage Council, Oslo, Norway.
Love, G.J., A.A.Cohen, J.F.Finklea, J.G.French, G.R.Lowrimore, W.C.Nelson, and P.B.Ramsey (1974) Prospective Surveys of Acute Respiratory Disease in Volunteer Families: 1970–1971 New York Studies, Health Consequences of Air Pollution: A Report from the CHESS Program, 1970–1971. EPA #650/1–74–004, pp. 5–49, June.
Lunn, J.E., J.Knowelden, and A.J.Handyside (1967) Patterns of Respiratory Illness in
Sheffield Infant School Children. Brit. J. Prev. Soc. Med. 21:7–16.
Martin, A.E. and W.Bradley (1960) Mortality, Fog and Atmospheric Pollution. Mon. Bull. Min. Health, London, 19:56–59.
McJilton, C. and R.Frank (1973) The Role of Relative Humidity in the Synergistic Effect of Sulfur Dioxide Aerosol Mixture on the Lung. Science, 182:503–504, November 2.
Miller, J.M. and R.G.DePena (1973) Contribution of Scavenged Sulfur Dioxide to the Sulfate Content of Rain Water, Journal of Geophysical Research, Vol. 77, 5905–5916.
Munn, R.E. (1973) Secular Increases in Summer Haziness in the Atlantic Provinces, Atmosphere, Vol. II, No. 4.
National Academy of Sciences (1974a) Air Quality and Automobile Emission Control on Air Quality Studies, Vol. 2, Health Effects of Air Pollutants; Prepared for the Committee on Public Works, September.
National Academy of Sciences (1974b) Air Quality and Automobile Emission Control on Air Quality Studies, Vol. 4, Costs and Benefits of Automobile Emission Control; Prepared for the Committee on Public Works, September.
National Coal Association (1974) Steam-Electric Plant Factors/1973 ed. Washington, D.C., January.
Nelson, W.C., J.F.Finklea, D.E.House, D.C.Calafiore, M.B.Hertz, and D.H.Swanson (1974) Frequency of Acute Lower Respiratory Disease in Children: Retrospective Survey of Salt Lake Basin Communities: 1967–1970, Health Consequences of Air Pollution: A Report from the CHESS Program, 1970–1971. EPA #650/1–74–004, pp. 2–55, June.
Newman, L., J.Forrest, and B.Manowitz (1975a) The Application of an Isotopic Ratio Technique to a Study of the Atmospheric Oxidation of Sulfur Dioxide in the Plume from an Oil Fired Power Plant. Submitted to Atmospheric Environment.
Newman, L., J.Forrest, and B.Manowitz (1975b) The Application of an Isotopic Ratio Technique to a Study of the Atmospheric Oxidation of Sulfur Dioxide in the Plume
from a Coal Fired Power Plant. Submitted to Atmospheric Environment.
North, D.Warner (1968) A Tutorial Introduction to Decision Theory, IEEE Transactions in Systems Science and Cybernetics, Vol. SSC-4, No. 3, September.
Nose, Yoshikatsu and Yoshimitsu Nose (1970) Air Pollution and Respiratory Diseases. Part IV. Relationship Between Properties of Air Pollution and Obstructive Pulmonary Diseases in Several Cities in Yamaguchi Prefective. J. Jap. Soc. Air Pollut. 5(1):130. Proceedings of the Japan Society of Air Pollution, 11th Annual Meeting.
OECD (1974) Environmental Directorate Co.-Operative Technical Programme to Measure the Long Range Transport of Air Pollutants Steering committee, Report on First Measurement Phase, NR/ENV/747 Paris, April 15.
Raiffa, Howard and Robert Schlaifer (1961) Applied Statistical Decision Theory, Harvard University, Boston.
Raiffa, Howard (1969) Decison Analysis: Introductory Lectures on Choices Under Uncertainty, Addison-Wesley.
Randall, A., B.C.Ives, and C.Eastman (1974) Benefits of Abating Aesthetic Environmental Damage, New Mexico State University Agricultural Experiment Station, Bulletin 618, May.
Roberts, Paul T. and Sheldon K.Friedlander (1974) Conversion of Sulfur Dioxide to Ambient Particulate Sulfates in the Los Angeles Atmosphere, paper presented at the conference: Health Consequences of Environmental Controls, Durham, North Carolina, April.
Robinson, E. (1971) Sources and Fate of Atmospheric Sulfur Compounds, Final Report, SRI Project SCC-8966, March.
Rodhe, H., C.Persson, and O.Akesson (1972) An Investigation Into Regional Transport of Soot and Sulfate Aerosols, Atmospheric Environment 6, 675–693.
Sagan, L.A. (1972) Human Costs of Nuclear Power, Science, 177, 487–493.
Sagan, L.A. (1974) Health Costs Associated with the Mining, Transport, and Combustion of Coal in the Steam-Electric Industry, Nature, 250, 107–111, July 12.
Solow, Robert M. (1971) The Economist’s Approach to Air Pollution and Its Control, Science, 173, 498.
Spence, J.W. and F.H.Haynie (1972) Paint Technology and Air Pollution: A Survey and Economic Assessment. Environmental Protection Agency. Research Triangle Park, North Carolina. Publication Number Ap-103, February.
Spetzler, Carl S. and C.A.Stael von Holstein (1972) Probability Encoding Decision Analysis, Stanford Research Institute, Menlo Park, California, to appear in Management Science.
Sugita, O., M.Shishido, E.Mino, S.Kenji, M.Kobayashi, C.Suzuki, N.Sukegawa, K.Saruta, and M.Watanabe (1970) The Correlation Between Respiratory Disease Symptoms in Children and Air Pollution. Report No. 1—a questionnaire Health Survey. Taiki Osen Kenkyu 5(1):134.
Tribus, Myron (1969) Rational Descriptions, Decisions, and Designs, Pergammon Press.
U.S. Department of Commerce (1970) Statistical Abstract of the United States; 91st Ed., Bureau of the Census, Washington, D.C.
U.S. Department of Commerce (1972) 1970 Census of Population, Characteristics of the Population, Vol. 1, Part A, Section 1, Bureau of the Census, Washington, D.C., May.
U.S. Department of Commerce (1974) Statistical Abstract of the United States; 95th Ed., Bureau of the Census, Washington, D.C.
U.S. Environmental Protection Agency (1974) 650/1–74–004, ORD, NERC/RTP, N.C., Health Consequences of Sulfur Oxides: A Report from CHESS 1970–1971, RTP, N.C., May.
U.S. Federal Power Commission (1974a) Steam-Electric Plant Construction Cost and Annual Production Expenses—1972, Washington, D.C., April.
U.S. Federal Power Commission (1974b) Steam-Electric Plant Air and Water Quality Control Data-1971, Washington, D.C., June.
U.S. Federal Power Commission (1974c) National Power Survey, Technical Advisory Committee, Task Force on Conservation and Fuel Supply, Power Generation: Conservation, Health, and Fuel Supply, Draft.
U.S. Public Health Service (1971) Acute Conditions, U.S., July 1966-June 1967, in Vital and Health Statistics, Washington, D.C.
Visher, Stephen S. (1954) Climatic Atlas of the United States, Harvard University Press, Cambridge.
Waddell, Thomas E. (1974) The Economic Damages of Air Pollution, Washington Environmental Research Center, U.S. Environmental Protection Agency, Washington, D.C. EPA-600/5–74–012, May.
Watanabe, H. and F.Kaneko (1971) Excess Death Study of Air Pollution in: Preceedings of the Second International Clean Air Congress. H.M.Englund and W.T.Beery (eds.), New York, Academic Press, pp. 199–200.
Weber, E. (1970) Paper No. CP-25F presented to 2nd International Clean Air Congress, Washington, D.C., December.
Yashizo, T. (1968) Air Pollution and Chronic Bronchitis, Osaka Univ. Med. J. 20:10–12, December.
Zeedijk, H. and C.A.Velds (1973) The Transport of Sulfur Dioxide Over a Long Distance, Atmospheric Environment, 7, 849.
PART THREE
CONTROL OF NITROGEN OXIDES FROM STATIONARY SOURCES
Part Three was prepared under the direction of the Commission on Natural Resources of the National Research Council. The discussions of nitrogen oxide sources in Chapter 14 and of tall stacks and intermittent control for nitrogen oxides in Chapter 15 are based on analyses by John Spengler, Anthony Cortese, and Douglas Dockery of the Harvard School of Public Health. The examination of control techniques in Chapter 15 is based on the work of Adel Sarofim and Richard Flagan of the Massachusetts Institute of Technology.