Advancing Knowledge and Shaping a Research Agenda
Situated outside of government and with a broad view of the research enterprise, the Institute of Medicine (IOM) is ideally placed to help guide medical and scientific research and identify priorities for the nation. Government agencies and public organizations alike depend on the findings and recommendations in IOM reports to determine how to structure their ongoing research activities or to embark on new directions. The IOM also looks farther forward to identify opportunities and challenges that lie on—or just over—the scientific horizon.
Examining today’s research
Two of today’s pressing health concerns illustrate the diversity of challenges in health research and health policy. One centers on the need to find better ways to reduce, and ultimately reverse, the pandemic caused by the human immunodeficiency virus (HIV). The other stems from the accelerating push to electronic medical recordkeeping and its attendant privacy concerns.
Protecting patient privacy while enhancing research
The modern electronic world offers many benefits for health care and health research. Using electronic methods for gathering, sharing, and analyzing patient health information can drive medical advances, including the development of new therapies, improved diagnostics, and more effective ways to prevent illness and deliver care. But the free flow of information
also creates the need for privacy protections to ensure that health care and research are carried out in ways that preserve patients’ dignity and protect them from harms such as discrimination or identity theft.
In 1996, Congress enacted the Health Insurance Portability and Accountability Act (HIPAA), which called for a set of federal standards, now known as the HIPAA Privacy Rule, for protecting the privacy of personally identifiable health information. Since then, some privacy advocates, researchers, and other observers have argued that the HIPAA Privacy Rule is not sufficient for the task and may, in fact, have the unintended consequence of undermining public health research. At the request of the U.S. Department of Health and Human Services (HHS) and a number of private groups, the IOM reviewed the rule’s track record.
Beyond the HIPAA Privacy Rule: Enhancing Privacy, Improving Health Through Research (2009) finds that the rule does not protect privacy as well as it should, and that it impedes important health research. The report recommends that the HHS work with other federal agencies to develop an entirely new approach to protecting personal health information in research. The new framework should apply privacy, data security, and accountability standards uniformly to information used in all health-related research in the United States, regardless of who funds or conducts the research.

To aid in advancing research while protecting privacy, the new framework should facilitate the use of health data in which personally identifiable information is removed and provide legal sanctions against unauthorized reidentification of individuals. In addition, the framework should provide ethical oversight of research in which use of personally identifiable information without individual consent is necessary, enlisting local ethical review boards to assess proposed projects on a case-by-case basis. Alternatively, institutions could be certified at the federal level to carry out this kind of research, having proved they have policies and practices in place to protect data privacy.
In the event that policy makers continue to rely on the current HIPAA Privacy Rule rather than adopt the new approach, the report also provides a series of recommended changes to improve both the rule as it currently stands as well as the guidance that the HHS provides for complying with it.
Designing better HIV intervention trials
In the more than 25 years since HIV was discovered, researchers have made significant strides in identifying effective prevention interventions. Yet the epidemic continues to expand. With an estimated 2.5 million new HIV infections occurring globally each year, efforts are needed to make better use of existing HIV prevention strategies and to identify new ones. In particular, women need additional methods for preventing HIV, given the many women who become infected under circumstances not under their control.
With an estimated 2.5 million new HIV infections occurring globally each year, efforts are needed to make better use of existing HIV prevention strategies and to identify new ones.
A near-perfect biomedical intervention for preventing HIV infection is unlikely to be available in the near future. New methods currently in late-stage clinical trials are likely to offer modest levels of protection, and perhaps only in specific circumstances. For this and other reasons, researchers have found it difficult to determine whether or not methods or products being tested actually provide useful levels of protection against HIV infection, and precisely how much protection they offer. At the request of the Bill & Melinda Gates Foundation, the IOM examined how best to meet this challenge.

Methodological Challenges in Biomedical HIV Prevention Trials (2008) presents a coordinated set of steps for improving the design, monitoring, and analysis of late-stage clinical trials of various types of interventions, excluding vaccines. In sum, the report concludes that alternative trial designs, more extensive site preparation, and careful monitoring and analysis of trial results are critical in evaluating prevention interventions and determining which of them can exert the greatest possible long-term impact on the HIV epidemic.
|
Biomedical Approaches to HIV Prevention Tested in Late-Stage Efficacy Trials Male circumcision, or removal of the penile foreskin, has been shown to reduce the risk of HIV infection in men. Microbicides are topical substances applied to the vagina or rectum that can potentially prevent HIV. Pre-exposure prophylaxis, employing antiretroviral drugs used for HIV treatment, may help prevent HIV infection. Cervical barriers were hypothesized to protect women from HIV by covering the cervix and blocking the upper genital tract, which is more vulnerable to HIV infection. Suppression of HSV-2, the primary cause of genital herpes, may help reduce sexual acquisition and transmission of HIV. Vaccines may enhance the body’s immune defenses to prevent HIV infection. SOURCE: Methodological Challenges in Biomedical HIV Prevention Trials, p. 3. |
Assessing government research programs
The National Institute for Occupational Safety and Health (NIOSH) is one of the key agencies charged with protecting the well-being of millions of workers nationwide who face potential dangers on their jobs. As part of its effort to establish and evaluate performance measures for each of its research programs, NIOSH requested that the National Research Council and the IOM review the performance of a number of its research programs and suggest emerging issues that should be studied in order to achieve future improvements in worker protection. The first report was issued in 2006, on hearing loss prevention, and eight additional reports in the series Reviews of Research Programs of the National Institute for Occupational Safety and Health have since been released. For each program, a key challenge is determining how to obtain maximum scientific and social pay-offs.
Traumatic Injury Research at NIOSH (2008) reviews the agency’s research conducted between 1996 and 2005 on this type of injury, defined
as “any damage inflicted to the body by energy transfer during work with a short duration between exposure and health event.” In sum, the report finds that the research program focused on the correct priority areas and yielded important gains in protecting workers’ safety. In addition, the program’s stated strategic goals for future research correctly target major contributors to occupational injuries and deaths and adequately consider populations and groups of workers who are at disproportionate risk.

The report offers nine recommendations to help NIOSH build on its success. Among them, the agency should work with other federal agencies that support injury prevention and control research to outline fruitful areas of collaboration; develop a strategic plan for evaluating its research-to-practice efforts and for building the capacity to carry out these efforts; and expand research on the safety impacts of changes in the nature of work and on interventions intended to improve organization policies and practices.
|
Goals and Subgoals of the NIOSH Traumatic Injury Research Programa
|
|||||||||||||||||||||
The Personal Protective Technology Program at NIOSH (2008) reviews the agency’s research conducted between 2001 and 2007 on the safety gear that its regulations require workers to wear. Such equipment includes respirators worn by construction workers and miners to protect against exposure to silica, dust, and hazardous gases; protective clothing, respirators, and gloves worn by firefighters and mine rescue workers to avoid burns and smoke inhalation; and respirators and protective clothing worn by health care workers to prevent acquiring an infectious disease. The report finds that overall, the research program made meaningful contributions to improving worker health and safety, correctly focused on areas of highest priority, and incorporated efforts to translate research results into products and processes that have proved useful in the workplace.

|
Personal Protective Technology Program Strategic Goals and Objectives Strategic Goal 1: Reduce Exposure to Inhalation Hazards
|
Strategic Goal 2: Reduce Exposure to Dermal Hazards
Strategic Goal 3: Reduce Exposure to Injury Hazards*
NOTE: CBRN = chemical, biological, radiological, and nuclear.
SOURCE: The Personal Protective Technology Program at NIOSH, p. 4. |
In advancing its mission, NIOSH should push to implement a 2001 congressional mandate that calls for developing a comprehensive state-of-the-art federal program focused on personal protective technology. Among other steps, the agency should establish extramural research centers of excellence; expedite the revision of respirator certification regulations; increase research on human factors, including individual behaviors and organizational behaviors, that influence workers’ use of protective equipment; and emphasize pre- and postmarket testing of protective products.
Beyond providing guidance to help protect workers on the ground, IOM committees also have taken on the task of safeguarding the health of workers in space. As part of its quest to expand humanity’s reach into the cosmos, the National Aeronautics and Space Administration (NASA) is preparing to send astronauts on various sorts of long-duration flights beyond low Earth orbit. These voyagers will face a complex set of safety and health hazards that result from the often-harsh space environment and the limits of available in-mission medical care. In determining its plans, NASA has asked the IOM for advice on a number of matters. Based on this advice, NASA has developed a new process to ensure that it gives proper attention to the full range of potential human health risks. From this effort, the agency has compiled a series of 25 “evidence books” that encapsulate the health-risk evidence gathered from both scientific studies and spaceflight experiences. Again, NASA turned to the IOM to evaluate the content of the evidence books and the process that the agency used in selecting which risks to explore.
Review of NASA’s Human Research Program Evidence Books: A Letter Report (2008) concludes that the agency has developed a thorough and well-conceived framework for documenting the evidence base, establishing research priorities, and integrating research findings into occupational health and safety measures for the space crew. The report also offers a number of recommendations to help NASA strengthen the content, composition, and dissemination of the evidence books. One key step will be to update the books continuously to reflect the most up-to-date information available. In this way, the cumulative knowledge base will best serve the interests of mission planners, researchers, and ultimately the individuals who will face the risks in their role as space travelers.
Looking toward tomorrow
In addition to facing issues with immediate import, the nation’s research enterprise and the policy makers who shape it must pay sharp attention to new and emerging challenges—and opportunities. The IOM helps to identify and illuminate these emerging challenges.
Understanding the brain
Neuroscience has made phenomenal advances over the past 50 years, and the pace of discovery continues to accelerate. Such prospects led the IOM’s Forum on Neuroscience and Nervous Systems Disorders to convene a workshop in which participants from diverse fields would examine emerging insights and, as an ultimate goal, excite both the scientific community and the public about the richness of knowledge that neuroscience is providing.
From Molecules to Minds: Challenges for the 21st Century: Workshop Summary (2008) explores the latest achievements, and remaining challenges, in deciphering the inner workings of that most complicated and exquisite organ—the brain. Researchers have crafted gains by combining past knowledge with information obtained using a host of new tools, including neuronal “light switches” and computer learning technologies. In light of such collective advances, numerous participants argued that neuroscience is on the cusp of even greater transformational progress in plotting how the brain operates and how its operations result in mental activity.

Looking ahead, participants discussed three “Grand Challenges” that neuroscientists, working in tandem with researchers in a number of related fields, might profitably consider. The first challenge centers on determining how the brain’s internal activity gives rise to thought, emotion, and behavior. The second focuses on determining how biology and experience interact to shape a person’s brain and make that person a unique individual. The third will involve identifying ways for people to keep their brains healthy, and especially how they can protect, restore, or enhance the functioning of their brains as they age. Such questions, having once simply been
assigned to future study, now are becoming approachable in a scientifically rigorous manner.
Advancing the genomic revolution
Until recently, the sequencing of the human genome—the body’s fundamental blueprint—also was thought to be purely science fiction. Yet in recent years, such sequencing has generated excitement about the potential of genomic innovations to improve medical care, preventive and community health services, and public health. Until fairly recently, physicians have been able to use a patient’s genetic information in diagnosing only a few relatively rare genetic diseases, such as cystic fibrosis and Huntington’s disease. But a transformation in the field is under way, and the IOM’s Roundtable on Translating Genomic-Based Research for Health has held workshops to foster discussions about how genomics is changing.

Diffusion and Use of Genomic Innovations in Health and Medicine: Workshop Summary (2008) reports on a workshop aimed at speeding the transfer of research findings to health care, public health, and health policy. The discussions centered around several key areas, including the processes or pathways by which new scientific findings in genomics move from the research setting into health care; the lessons that can be learned from the translation of other new technologies into practical application; and the practical incentives and barriers that now promote or hinder the translation of genomic advances into health care both within the United States and globally.
Innovations in Service Delivery in the Age of Genomics: Workshop Summary (2009) discussed how new discoveries in genomics are changing the way in which diseases are diagnosed and treated. Whereas previously, genetic testing could only screen for rare genetic disorders, increasingly, patients and their physicians now are able to use genetic information to predict the risk of common diseases such as diabetes and breast cancer and to help determine prevention and treatment options. Genetic specialists have long been the main providers of genetic services, offering intensive counseling for rare genetic disorders. However, as the trend shifts
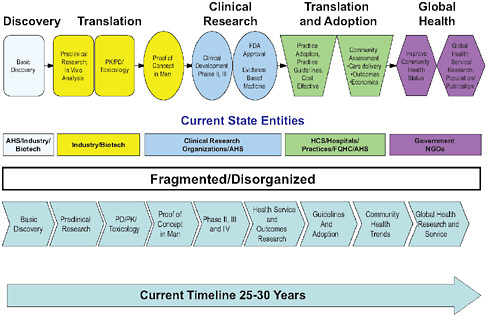
Translation of innovations.
SOURCE: Diffusion and Use of Genomic Innovations in Health and Medicine: Workshop Summary, p. 4.
from genetic testing largely being undertaken for rare genetic disorders to, increasingly, individuals being screened for common diseases, providers need to be knowledgeable about and comfortable using genetic information to improve their patients’ health.












