1
Introduction
In 2008, the Institute of Medicine (IOM) published the report Preparing for an Influenza Pandemic: Personal Protective Equipment for Healthcare Workers (IOM, 2008). When the report was released, the major influenza-related concern was avian influenza (H5N1). As novel H1N1 influenza A became a reality in 2009, the many unknowns about the virulence, spread, and nature of the virus raised to the forefront issues regarding personal protective equipment (PPE) for healthcare personnel. One of the major issues was the nature of respiratory protection required because much remains to be learned about the mechanisms of influenza transmission. This current report comes at a time when controversies continue on issues related to PPE for healthcare personnel while new horizons in PPE research and attention to PPE innovations offer promise for improvements in healthcare worker safety. Keeping the research momentum moving forward is critical, because between pandemics the focus of research efforts often moves to other issues and the nation remains underprepared.
SCOPE OF THE REPORT
The 2009–2010 H1N1 experience and its accompanying unanswered research questions provided the impetus for the National Personal Protective Technology Laboratory (NPPTL) at the National Institute for Occupational Safety and Health (NIOSH) to ask the IOM to conduct a study that would update the progress on research and identify future directions regarding PPE use for healthcare personnel.
This report is the result of a 12-month study conducted by an ad hoc IOM committee composed of experts in the fields of infectious disease, infection control, public health, occupational safety and health, pulmonary medicine, health promotion, microbiology, emergency preparedness and response, epidemiology, nursing, community health, industrial hygiene, and materials engineering. The IOM committee was charged with identifying new research directions, certification1 and standards-setting issues, and risk assessment issues specific to PPE for healthcare personnel to prevent transmission of pandemic influenza and other viral respiratory infections. The committee was asked to focus specifically on the following areas:
-
research needed to understand and improve the efficacy and effectiveness of personal protective equipment, particularly face masks and respirators for preventing transmission of pandemic influenza or other viral respiratory infections. Specific attention was sought on issues related to the research needed to determine the type of respiratory protection needed for the given exposure, to determine the requirements for protective ensembles to provide an appropriate level of protection based on work tasks, and to improve functionality and address human factor issues, such as wearability, compliance, and communications;
-
necessary certification, testing, and standards development issues; and
-
priorities and resources for research and certification efforts.
To accomplish its charge, the committee held three meetings and gathered information through a scientific workshop (Appendix A) that included a public comment session, discussions with numerous individuals in the infection control and occupational safety and health fields, and a review of the relevant literature. As mentioned above, this report builds on the work of the IOM committee that released the 2008 report. Throughout this report, the prior work is summarized. In large part, this committee’s task was to examine research conducted since the 2008 report in order to assess where research stands on issues that are key to improving PPE for healthcare personnel exposed to infectious respiratory diseases and to make recommendations to address current research gaps.
Many PPE issues relevant to healthcare personnel are also directly relevant to the PPE needs of workers in other occupations as well as the general public.
In 2009, an Institute of Medicine committee addressed the question of what type of PPE was needed for the 2009 H1N1 pandemic. In addition to recommending the use of respirators because respiratory protection was deemed appropriate, the report recommended increased research on “the next generation of personal respiratory protection technologies for healthcare workers to enhance safety, comfort, and ability to perform work-related tasks” (IOM, 2009).
BACKGROUND AND CONTEXT
Readers of this report may be familiar with the 2008 report or knowledgeable about the extensive background issues regarding PPE and its use in healthcare settings. In lieu of including extensive background materials, the committee provides an overview of the context for this report by addressing the following series of questions.
What Are PPE and Personal Protective Technologies?
The term “personal protective equipment” encompasses the specialized clothing or equipment worn by workers for protection against health and safety hazards. Specific types of PPE are selected based on the occupational hazard faced in specific work tasks or sites. “Personal protective technologies” is a broader term that includes the protective equipment as well as the technical methods, processes, techniques, tools, and materials that support their development, evaluation, and use (NIOSH, 2007; OSHA, 2002). This report most often discusses the equipment used by healthcare personnel and uses the term “PPE,” although in some cases the broader term is needed.
For healthcare personnel, PPE may include respirators, face masks, gloves,2 eye protection, face shields, gowns, and head and shoe coverings. Respirators provide respiratory protection; the other products are designed primarily to provide a barrier against microbes contacting the skin or mucous membrane surfaces. Integrating the various types of pro-
tective equipment to ensure that they work together as ensembles (e.g., eye protection with a respirator) is an ongoing concern. Infection prevention and control in healthcare workplaces involve, among many other measures, the use of PPE.
What Are the Roles of Face Masks and Respirators?
One of the challenges for the healthcare field is to clearly understand the differences between respirators and face masks as well as their appropriate uses. In this report, the terminology used by the investigators or authors of the cited journal article or report is used, but in some cases, determining whether the authors’ use of the term “masks” refers to face masks, respirators, or both was impossible.
Face masks, including surgical and procedure masks, are loose-fitting coverings of the nose and mouth that are designed to protect the patient from secretions from the nose or mouth of the physician, nurse, or other healthcare professional. Face masks are not designed or certified to protect the wearer from exposure to respiratory hazards. Some studies have looked at the variation in filtration capabilities of face masks (Chapter 3), but the role of face masks as PPE requires further research.
Respirators are specifically designed as respiratory protection and are certified by NIOSH (42 Code of Federal Regulations [CFR] Part 84). They work either by purifying the air inhaled by the wearer through filtering materials or by independently supplying breathable air. For air-purifying respirators (often the type used by healthcare personnel), the major issues are the filtration and the fit—the effectiveness of the filter and the extent to which the respirator has a tight seal with the wearer’s face to restrict inward leakage. A type of respirators, termed surgical N95 respirators, is cleared by the Food and Drug Administration (FDA) as medical devices. To be effective, respirators must fit tightly to the face. Annual fit testing for respirators is required by the Occupational Safety and Health Administration (OSHA), and user seal checks are required with each use (29 CFR 1910.134).
How Does PPE Fit into the Range of Other Workplace Safety and Preventive Measures?
Efforts to promote worker safety and health traditionally follow a hierarchy of controls. Engineering and environmental measures, such as air exchanges (including non-recirculated exchanges) or negative-pressure rooms that can isolate the hazard or reduce exposure, are ubiquitous measures that affect a large number of workers and patients and do not depend on individual compliance (IOM, 2008). Administrative controls are next in the hierarchy and include the policies, standards, procedures, and practices established within an organization to limit hazardous exposures and improve worker safety (e.g., vaccination policies, cohorting or isolating patients, hand hygiene measures, provision of appropriate and effective PPE, organizational commitment to creating and sustaining a culture of worker safety) (IOM, 2009). At the level of work practice controls, including individual practices, the healthcare employer and individual personnel are responsible for appropriate use of PPE as well as being vaccinated and adhering to work safety practices.
Measures to prevent transmission of influenza or other viral respiratory diseases to healthcare personnel include all levels of hazard controls. PPE, along with vaccination and antiviral medications, are components of an overall infection prevention and control program that uses engineering, administrative, and work practice controls. Vaccination against influenza has been found to be effective in preventing the illness in the recipient with overall efficacy rates over 70 percent (Fiore et al., 2009; Treanor et al., 1999).3 Although all levels of this hierarchy are important, this report focuses on opportunities to improve PPE and the correct use of PPE in healthcare settings.
What Federal Agencies Are Involved in Healthcare Personnel PPE?
The testing, regulation, and use of PPE by healthcare personnel involve several federal departments and agencies. Responsibilities for occupational health and safety are within the purview of both the Department of Health and Human Services (HHS) and the Department of
Labor (DOL). The Occupational Safety and Health Act of 1970 (Public Law 91-596) created two federal agencies that focus on worker safety and health: NIOSH (in HHS) is designated with responsibilities for relevant research, training, and education; and OSHA (in DOL) is designated with responsibilities for developing and enforcing workplace safety and health regulations.
NPPTL, a NIOSH laboratory, conducts and funds research on improvements in PPE and ensembles used in a variety of occupations. NPPTL also plays an integral role in standards-setting efforts relevant to PPE. Respirators used by personnel in OSHA-regulated workplaces, including healthcare workplaces, must be NIOSH certified. OSHA respirator regulations detail employer responsibilities for establishing and maintaining a comprehensive respiratory protection program, including selection, training, and fit testing requirements. For other types of PPE, OSHA specifies the voluntary consensus standards that the equipment must meet as well as relevant selection and training requirements. NIOSH, through NPPTL, conducts an extensive array of tests to assess respirator performance characteristics and to determine if the respirator meets the certification requirements.
To be marketed in the United States as medical devices, manufacturers of respirators, face masks, and some other types of healthcare PPE are required to obtain FDA approval or clearance by demonstrating equivalence with a similar product on the market. A number of other departments and agencies are involved in various aspects of healthcare PPE. In addition to NIOSH within the Centers for Disease Control and Prevention (CDC), the National Center for Emerging and Zoonotic Infectious Diseases, in conjunction with the external Healthcare Infection Control Practices Advisory Committee (HICPAC), develops infection control guidance for healthcare settings (CDC, 2010c). Public health agencies at the local, state, and federal levels play an instrumental role in developing guidelines, providing training, and assisting healthcare facilities with PPE-related issues. The Department of Defense and the Department of Veterans Affairs are actively involved in testing and developing PPE for military and veterans’ care applications. The Department of Homeland Security focuses on emergency response PPE and works to coordinate and improve standards and equipment-related issues. The Environmental Protection Agency addresses PPE issues relevant to emergency response readiness. The Consumer Product Safety Commission has oversight responsibilities for products sold in the commercial marketplace, including
PPE.4 Professional associations, standards development organizations, and other entities also play important roles in ensuring that PPE products meet performance criteria.
How Does This Report Define the Scope of the Term “Healthcare Personnel”?
More than 14 million U.S. workers are employed in health care (BLS, 2010). The committee broadly defines “healthcare personnel” to encompass all workers in direct patient care and support services who are employed by private and public healthcare offices and facilities as well as those working in home healthcare and emergency medical services, including those who are self-employed. This broad definition of healthcare personnel encompasses those working in administration, patient care, and facilities upkeep, and it includes health professional students who are receiving instruction or who are working in healthcare facilities as well as volunteers trained to provide systematic, regulated, and licensed healthcare services (including emergency medical responders) (IOM, 2008, 2009). All relevant work situations with the potential for infection risk (e.g., cleaning patient rooms, delivering food) are considered part of the healthcare workforce. This definition is expanded from the definition used in the 2008 report. The committee acknowledges that, in the midst of an influenza pandemic, many people outside the traditional healthcare workforce will become caregivers, including many family members, and they may need access to PPE.
HEALTHCARE PERSONNEL AND PPE
Many work environments offer challenges for protecting personnel—the heat and smoke of firefighting, the heights of construction work on rooftops and high-rise buildings, and worksite noise or hazardous chemicals in industrial settings—to name a few. For healthcare personnel, several aspects of the job provide challenges for designing and wearing
appropriate PPE. These issues include interactions with patients and family members that make communication critically important, split-second actions in some healthcare situations that can have major consequences, and challenges in exposure monitoring. Because most types of PPE work by acting as a barrier to hazardous agents, healthcare personnel face challenges posed by barrier materials, including having difficulties in verbal communications and interactions with patients and family members, maintaining tactile sensitivity through gloves, and addressing physiological burdens.
Some types of PPE are used routinely by clinicians as part of standard infection control precautions designed to protect both the healthcare professional and the patient from disease acquisition. CDC has developed a tiered approach to infection control precautions that is detailed and reviewed by HICPAC. Standard precautions5 (first tier) are applied to the care of all patients and include the use of gloves and hand hygiene. The second tier of precautions is used in cases where patients have documented or presumed infections or conditions that could be transmitted to healthcare personnel. The details of these transmission-based precautions are specific to situations with the potential for contact, airborne, or droplet transmission of infectious agents as defined by HICPAC (Siegel et al., 2007). Care of patients with suspected respiratory infections with viral agents—such as respiratory syncytial virus, adenovirus, parainfluenza, influenza, or human metapneumovirus—requires the use of contact and droplet precautions to protect the healthcare worker from exposure to droplet spray and contact. The guideline further notes that once adenovirus and influenza have been ruled out, droplet precautions can be discontinued (Siegel et al., 2007, p. 121).
As noted in the 2008 report, opportunities abound to provide innovative approaches that could improve PPE design to better fit healthcare needs, incorporate an emphasis on worker safety, and integrate worker and patient safety efforts. In discussing the issues relevant to the use of PPE by healthcare personnel, the committee identified a set of criteria as a starting point for decisions on selecting and using PPE. PPE for healthcare personnel should
-
effectively reduce risks of disease or injury to healthcare personnel;
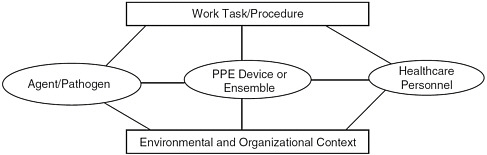
FIGURE 1-1 The major components and factors involved in the use of personal protective equipment in health care.
-
minimize negative interactions with or effects on patients and their families and caregivers;
-
be acceptable and usable by healthcare personnel in their day-to-day tasks, including ease of communication and comfort;
-
be practical regarding issues of cost, time, and training; and
-
be appropriate to the occupational risk being encountered.
In developing the report, the committee considered the issues relevant to the pathogen (virus), the device, and the worker while realizing it needed to keep in mind the various work tasks of healthcare personnel, the safety culture, and other organizational issues regarding where and how they work, and the broader policy and regulatory issues. Figure 1-1 is a depiction of the major components or factors involved in PPE use in health care.
OVERVIEW OF INFLUENZA AND OTHER VIRAL RESPIRATORY DISEASES
This report draws on the 2009–2010 experience with H1N1 influenza A. However, a brief background section is included here to set the context for the report and to broaden the discussion to include other viral respiratory diseases. Although not the focus of this report, bacterial pathogens may also be transmitted via respiratory aerosols.
Influenza is a serious respiratory illness caused by infection with influenza type A or type B virus. Each year, more than 200,000 U.S.
hospitalizations result from seasonal influenza and its complications (Thompson et al., 2004). Estimates of the number of deaths in the United States associated annually with seasonal influenza (from 1976 to 2007) ranged from a low of about 3,300 to a high of about 49,000, with most of the excess mortality in persons 65 years and older (Thompson et al., 2010). Cases of influenza peak during the winter months in each hemisphere. The influenza A virus is categorized by the subtypes of its major surface glycoproteins: hemagglutinin and neuraminidase.6 The influenza virus undergoes frequent changes in antigenicity. Vaccines and antiviral medications have been developed to prevent or mitigate the disease, although major challenges remain, particularly in determining the appropriate viral strain to be included in the annual vaccine.
In contrast to seasonal occurrences of influenza, global outbreaks or pandemics occur more rarely. In the twentieth century, influenza pandemics occurred in 1918, 1957, and 1968. The highest mortality was in the 1918 pandemic, which is estimated to have resulted in 675,000 deaths in the United States and 50 million or more deaths worldwide (Johnson and Mueller, 2002; Morens and Fauci, 2007). The 2009 H1N1 virus was first detected in Mexico in April 2009 and quickly appeared in the United States. Although the incidence was high and difficult to measure (as only a small percentage are laboratory-confirmed cases), it was milder than expected and more likely to affect people under age 65 than seasonal influenza (CDC, 2010e). In the United States, CDC estimates there were between 43 and 89 million cases of H1N1 between April 2009 and April 10, 2010, with an estimate of 8,870 to 18,300 deaths related to the 2009 H1N1 (CDC, 2010e).
In late April 2009, CDC began to release 25 percent of the supplies in the Strategic National Stockpile that could be needed to protect against the influenza virus and treat influenza patients. This equated to approximately 11 million regimens of antiviral drugs and 39 million pieces of PPE, including face masks, respirators, gowns, gloves, and face shields (allocations were based on each state’s population) (CDC, 2010a).
Other viral respiratory diseases are also a concern for the health and safety of healthcare personnel, ranging from emerging diseases, such as severe acute respiratory syndrome (SARS), to the highly prevalent and seasonal respiratory syncytial virus (RSV). In 2003, worldwide attention focused on an outbreak of infections caused by a previously unrecognized coronavirus that resulted in SARS. Healthcare personnel, partic-
ularly those not using PPE and other infection control practices, were among the vulnerable groups (CDC, 2003). RSV infections are especially a concern in infants and older adults. The symptoms of RSV infections are similar to other respiratory diseases, including influenza, and transmission routes of RSV include direct and indirect contact, droplet spray, and aerosol routes. Each year in the United States, 75,000 to 125,000 infants are hospitalized with RSV (CDC, 2010d), and their care may involve many types of healthcare personnel.
Having recently been through the 2009–2010 experience with H1N1 influenza, the committee is well aware of the ongoing challenges and controversies surrounding PPE for healthcare personnel. Lessons continue to be learned regarding strategies to address why some healthcare personnel do not use preventive measures that are available and effective, particularly because preventing disease has implications for both improving the health of the workers and their patients. At this time, it is particularly important to build on the recent H1N1 experience and take the actions needed to address the research and policy questions that will allow the healthcare community to be better prepared for the next epidemic or pandemic. While the optimist can seek comfort in the fact that progress has been made in the past several years, the pragmatist must wonder about the rate of progress (or lack thereof) on such a critical issue that potentially threatens the security and economic well-being of the nation and the world. Experience has shown that relevant research on these issues wanes between pandemics. Avoiding that scenario this time is crucial to resolving the research questions and setting evidence-based policies in place.
DEFINING AN INTEGRATED APPROACH TO PPE RESEARCH
Providing care to ill or injured patients involves a range of potentially hazardous exposures for healthcare personnel. Current infection control precautions address this challenge by providing guidance on PPE and other precautions that varies depending on the mode of transmission of the pathogen. The ultimate goal would be to have definitive information that would match the appropriate type of PPE with the pathogen, its mode of transmission, the infectious dose, and its risk to healthcare personnel. In many cases in industrial settings, this level of specificity is available for chemical exposures, although other industrial settings with
unknown or mixed exposures continue to pose challenges. Reaching that point for protecting healthcare personnel will require concerted research efforts.
This report explores an integrated approach that addresses the full spectrum of research (from basic research to policy research) and translates research findings into improvements in the standards of healthcare practice (Figure 1-2). This approach ensures that basic science initiatives are fully explored, while also addressing clinical needs and testing the results in real-world settings, with the expectation that adaptations will be made and tested along the way. Feedback loops to prior stages are also critically important. Such an integrated approach calls for active collaboration and discourse among scientists and clinicians who may not have had previous interactions.
Chapters 2 to 5 each begin with a short summary of the relevant discussion of the 2008 report, followed by an update of recent research efforts outlining the state of progress in that area, and concluding with the committee’s recommendations on research needs and directions. The committee examined four major areas of research:
-
transmission of influenza and other viral respiratory diseases and the use of PPE in preventing transmission (Chapter 2);
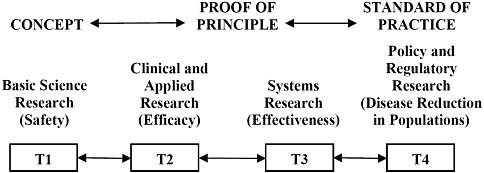
FIGURE 1-2 An integrated system moving research into practice, depicting the translation of research from basic science research (T1) through policy and regulatory research (T4).
SOURCE: Adapted from Henderson and Palmore (2010) with permission from University of Chicago Press.
-
design and engineering of PPE to be effective and wearable (Chapter 3);
-
use of PPE by healthcare personnel (Chapter 4); and
-
policy, standards setting, and certification (Chapter 5).
The report covers PPE issues that are relevant to preventing the transmission of a number of viral respiratory diseases; however, much of the recent research and discussion is focused on influenza and, as a result, influenza is the primary disease discussed in the report. The priorities noted throughout this report are to determine the type of PPE needed for given exposures and to move effective, wearable, and innovative PPE products through the basic research and initial testing phases and on to active use and evaluation by the worker community.
REFERENCES
BLS (Bureau of Labor Statistics). 2010. Career guide to industries, 2010-2011 edition: Healthcare. http://www.bls.gov/oco/cg/cgs035.htm (accessed November 19, 2010).
CDC (Centers for Disease Control and Prevention). 2003. Cluster of severe acute respiratory syndrome cases among protected health-care workers—Toronto, Canada, April 2003. Morbidity and Mortality Weekly Report 52(19):433-436.
———. 2010a. The 2009 H1N1 pandemic: Summary highlights, April 2009-April 2010. http://cdc.gov/h1n1flu/cdcresponse.htm (accessed November 19, 2010).
———. 2010b. 2010-11 influenza prevention and control recommendations: Additional information about vaccination of specific populations. http://www.cdc.gov/flu/professionals/acip/specificpopulations.htm (accessed November 30, 2010).
———. 2010c. Prevention strategies for seasonal influenza in healthcare settings. http://www.cdc.gov/flu/professionals/infectioncontrol/healthcaresettings.htm (accessed December 20, 2010).
———. 2010d. Respiratory syncytial virus activity—United States, July 2008-December 2009. Morbidity and Mortality Weekly Report 59(8):230-233.
———. 2010e. Updated CDC estimates of 2009 H1N1 influenza cases, hospitalizations and deaths in the United States, April 2009-April 10, 2010. http://www.cdc.gov/h1n1flu/estimates_2009_h1n1.htm (accessed November 19, 2010).
Fiore, A. E., C. B. Bridges, and N. J. Cox. 2009. Seasonal influenza vaccines. Current Topics in Microbiology and Immunology 333:43-82.
Henderson, D. K., and T. N. Palmore. 2010. Critical gaps in knowledge of the epidemiology and pathophysiology of healthcare-associated infections. Infection Control and Hospital Epidemiology 31(Suppl. 1):S4-S6.
IOM (Institute of Medicine). 2008. Preparing for an influenza pandemic: Personal protective equipment for healthcare workers. Washington, DC: The National Academies Press.
———. 2009. Respiratory protection for healthcare workers in the workplace against novel H1N1 Influenza A: A letter report. Washington, DC: The National Academies Press.
Johnson, N. P., and J. Mueller. 2002. Updating the accounts: Global mortality of the 1918-1920 “Spanish” influenza pandemic. Bulletin of the History of Medicine 76(1):105-115.
Morens, D. M., and A. S. Fauci. 2007. The 1918 influenza pandemic: Insights for the 21st century. Journal of Infectious Diseases 195(7):1018-1028.
NIOSH (National Institute for Occupational Safety and Health). 2007. Evidence for the National Academies’ review of the NIOSH personal protective technology program. http://www.cdc.gov/niosh/nas/ppt/pdfs/PPT_EvPkg_090707_FinalR.pdf (accessed November 30, 2010).
OSHA (Occupational Safety and Health Administration). 2002. OSHA fact sheet: Personal protective equipment. http://www.osha.gov/OshDoc/data_General_Facts/ppe-factsheet.pdf (accessed November 30, 2010).
Siegel, J. D., E. Rhinehart, M. Jackson, L. Chiarello, and the Healthcare Infection Control Practices Advisory Committee. 2007. 2007 guideline for isolation precautions: Preventing transmission of infectious agents in healthcare settings. http://www.cdc.gov/hicpac/pdf/isolation/Isolation2007.pdf (accessed December 20, 2010).
Thompson, M., D. Shay, H. Zhou, C. Bridges, P. Cheng, E. Burns, J. Bresee, and N. Cox. 2010. Estimates of deaths associated with seasonal influenza—United States, 1976–2007. Morbidity and Mortality Weekly Report 59(33):1057-1062.
Thompson, W. W., D. K. Shay, E. Weintraub, L. Brammer, C. B. Bridges, N. J. Cox, and K. Fukuda. 2004. Influenza-associated hospitalizations in the United States. Journal of the American Medical Association 292(11):1333-1340.
Treanor, J. J., K. Kotloff, R. F. Betts, R. Belshe, F. Newman, D. Iacuzio, J. Wittes, and M. Bryant. 1999. Evaluation of trivalent, live, cold-adapted (CAIV-T) and inactivated (TIV) influenza vaccines in prevention of virus infection and illness following challenge of adults with wild-type influenza A (H1N1), A (H3N2), and B viruses. Vaccine 18(9-10):899-906.














