2
Jet Propellant Fuels 5 and 81
Acute Exposure Guideline Levels
PREFACE
Under the authority of the Federal Advisory Committee Act (FACA) P.L. 92-463 of 1972, the National Advisory Committee for Acute Exposure Guideline Levels for Hazardous Substances (NAC/AEGL Committee) has been established to identify, review, and interpret relevant toxicologic and other scientific data and develop AEGLs for high-priority, acutely toxic chemicals.
AEGLs represent threshold exposure limits for the general public and are applicable to emergency exposure periods ranging from 10 minutes (min) to 8 hours (h). Three levels—AEGL-1, AEGL-2, and AEGL-3—are developed for each of five exposure periods (10 and 30 min and 1, 4, and 8 h) and are distinguished by varying degrees of severity of toxic effects. The three AEGLs are defined as follows:
AEGL-1 is the airborne concentration (expressed as parts per million or milligrams per cubic meter [ppm or mg/m3]) of a substance above which it is predicted that the general population, including susceptible individuals, could experience notable discomfort, irritation, or certain asymptomatic, nonsensory
________________________
1This document was prepared by the AEGL Development Team composed of Sylvia Talmage (Summitec Corporation) and John Hinz (National Advisory Committee [NAC] on Acute Exposure Guideline Levels for Hazardous Substances. The NAC reviewed and revised the document and AEGLs as deemed necessary. Both the document and the AEGL values were then reviewed by the National Research Council (NRC) Committee on Acute Exposure Guideline Levels. The NRC committee has concluded that the AEGLs developed in this document are scientifically valid conclusions based on the data reviewed by the NRC and are consistent with the NRC guidelines reports (NRC 1993, 2001).
effects. However, the effects are not disabling and are transient and reversible upon cessation of exposure.
AEGL-2 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience irreversible or other serious, long-lasting adverse health effects or an impaired ability to escape.
AEGL-3 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience life-threatening health effects or death.
Airborne concentrations below the AEGL-1 represent exposure concentrations that could produce mild and progressively increasing but transient and nondisabling odor, taste, and sensory irritation or certain asymptomatic, nonsensory effects. With increasing airborne concentrations above each AEGL, there is a progressive increase in the likelihood of occurrence and the severity of effects described for each corresponding AEGL. Although the AEGL values represent threshold levels for the general public, including susceptible subpopulations, such as infants, children, the elderly, persons with asthma, and those with other illnesses, it is recognized that individuals, subject to idiosyncratic responses, could experience the effects described at concentrations below the corresponding AEGL.
SUMMARY
Jet propellant (JP) fuels, used in military and civilian aircraft, are complex mixtures of aliphatic and aromatic hydrocarbons made by blending various distillate stocks of petroleum. The primary military fuel for land-based military aircraft is JP-8; this fuel replaces JP-4, which is no longer in use. JP-5 was developed by the U.S. Navy for shipboard service. The composition of JP-8 and JP-5 is basically that of kerosene (with additives), and they have similar chemical and physical characteristics (ATSDR 1998). Worldwide, approximately 60 billion gallons of military JP-8 and the equivalent commercial Jet A and Jet A-1 are consumed on an annual basis. The military jet fuels contain additives that are not found in commercial jet fuels. Civilian and military personnel may be exposed to jet fuels during fuel production, aircraft fueling, aircraft maintenance, and accidental spills or pipeline leaks. The primary hazard associated with release of jet fuels is fire and explosion.
This document focuses on the toxicity of JP-8 with some attention to the chemically similar JP-5. These two fuels have a similar composition and appear to have similar toxicities (ATSDR 1998). Monitoring data indicate that exposures to JP-4, which has a higher vapor pressure than JP-8 and JP-5, were higher than those associated with JP-8 and JP-5. Data were located on acute sensory and systemic effects of JP-8 and JP-5 in mice and rats; subchronic toxicity studies have addressed systemic and pulmonary toxicity. For both fuels, eye irrita-
tion was observed at concentrations of ≥ 2,500 mg/m3. Mild skin irritation was observed after direct topical application. Several short-term and repeated exposure studies addressed the toxicity of jet fuel aerosols. Exposure to aerosolized jet fuels was associated with enhanced toxicity compared with equivalent exposure to fuel vapors, the lungs and immune system being the target organs. However, emergency exposures are expected to be in the form of vapor exposures that result from spills, whereas aerosols are relevant only to occupational exposures during aircraft-foam removal operations or aircraft cold starts. Studies that addressed the toxicity of jet fuel only in the aerosolized form were not used to derive AEGL values (Martin et al. 2010; Tremblay et al. 2010). The data collected during aerosol inhalation studies are included in this technical support document (TSD) for completeness. Animal studies also examined potential neurotoxicity, developmental and reproductive toxicity, and carcinogenicity. The JP fuels are not considered genotoxic or carcinogenic and, in a preliminary study, JP-8 failed to cause spermatotoxic effects in humans. A characteristic nephropathy and resulting renal cancer, specific to male rats exposed to jet fuels, is not relevant to humans. Concentrations of jet fuels of ≥ 2,500 mg/m3 also induce central nervous system (CNS) depression. Many of the components of jet fuels are lipophilic solvents. In general, the lipophilic solvents that induce CNS depression attain steady state in the blood within an hour.
The AEGL-1 was based on the sensory irritation study of Whitman and Hinz (2001) wherein an RD50 (the concentration that reduced the respiratory rate of Swiss-Webster mice by 50%) was measured for JP-8 vapor plus aerosol at 2,876 mg/m3. The RD50 test is a standard protocol (ASTM E981-84 [1988]) for estimating sensory irritancy of airborne chemicals. Groups of four male Swiss-Webster mice were exposed for 30 min at 681, 1,090, 1,837, or 3,565 mg/m3. Reductions in the respiratory rate within 30 min were concentration-dependent, and breathing patterns were characteristic of upper airway sensory irritation. On the basis of a correlation between the RD50 and sensory irritancy concentrations for a large number of structurally diverse chemicals, a 10-fold reduction of the RD50 results in a concentration that elicits sensory irritation in humans but that can be tolerated for hours to days (Alarie 1981; Schaper 1993). Irritation is concentration-dependent, and there is adaptation to the mild sensory irritation that characterizes the AEGL-1. Using this reasoning, the resulting concentration of 290 mg/m3 can be tolerated at each AEGL-1 exposure duration. The 290 mg/m3 value is supported by the lack of adverse health effects in subchronic toxicity animal studies with repeated or continuous exposures to JP-8 vapor at 1,000 mg/m3 (Mattie et al. 1991; Briggs 2001; Rossi et al. 2001).
The AEGL-2 is based on inhalation studies with rats and mice demonstrating that exposure to JP-8 at 1,100 mg/m3 failed to elicit signs of intoxication or CNS depression. The shorter-term studies (30 min to 4 h) with exposures to JP-8 or JP-5 in mixed vapor and aerosol forms at 3,430-5,000 mg/m3 (MacEwen and Vernot 1985; Wolfe et al. 1996; Whitman and Hinz 2001) with support from studies using repeated or continuous vapor exposures at 1,000 mg/m3 (Mattie et al. 1991; Briggs 2001; Rossi et al. 2001) were used as the basis for the AEGL-2.
No uncertainty factors were applied to the results of studies at the 1,000 mg/m3 concentration because no adverse effects were observed, and the exposures were repeated or continuous for up to 90 days. The higher concentrations of JP-8 (3,430, 3,565, and 4,440 mg/m3) and of JP-5 (5,000 mg/m3) were divided by an interspecies factor of 1 (compared with humans, systemic uptake is greater in rodents based on higher respiration rate and cardiac output) and by an intraspecies uncertainty factor of 3 to protect potentially sensitive individuals. An intraspecies uncertainty factor of 3 is considered adequate because the thresholds for both sensory irritation and CNS depression for solvents in humans and rodents do not generally differ by more than 3-fold. The lower value, 1,100 mg/mg3, in the resulting range of values, 1,100-1,700 mg/m3, is approximately the same concentration as in the no-adverse-effect repeated-exposure studies. CNS depression is a concentration-related effect. For solvents that cause CNS effects, steady state is generally approached within 1 h. In addition, because the exposure duration in the key study was 4 h, the 1,100 mg/m3 value was used for the 4-h and shorter time periods. Because the exposure of rats and mice at 1,000 mg/m3 was continuous (24 h/day) for up to 90 days (Mattie et al. 1991), the 1,100-mg/m3 value can also be used for the 8-h AEGL. The fact that the exposures in most of these studies, especially at the higher concentrations, were to mixed JP-8 vapor and aerosols supports the AEGL-2 values.
Because of their relatively low vapor pressure, the physical properties suggest JP-8 and JP-5 might not attain a sustained vapor concentration high enough to cause death. As reported by Wolfe et al. (1996), the highest vapor concentration of JP-8 that could be attained under an experimental system at 35ºC was 3,430 mg/m3, and the highest vapor and aerosol concentration that could be generated was 4,440 mg/m3. However, the highest vapor and aerosol attainable under ambient concentrations has been estimated at 700 mg/m3, and 500 mg/m3 is the upper bound for a stable JP-8 aerosol. Based on the likelihood that airborne concentrations of JP-8 or JP-5 aerosol and vapor sufficient to cause death cannot be sustained under ambient conditions, an AEGL-3 was not derived.
Although the AEGL values are based on reported mixed aerosol and vapor concentrations of jet fuels, the primary exposure is to the vapor. Exposure to aerosols will probably result in deep lung deposition. Therefore, AEGLs based on mixed aerosol and vapor exposures are more conservative than those based on gas-phase exposures. Aerosol concentrations of 10 mg/m3 result in a visible cloud. These concentrations and higher will result in liquid deposition on surfaces.
AEGL values are summarized in Table 2-1 below.
I. INTRODUCTION
Jet propellant or jet propulsion (JP) fuels are used in military aviation for turbine engine and jet aircraft. Jet fuels are complex mixtures of aliphatic and
aromatic hydrocarbons made by blending petroleum distillates, such as naphtha (the low boiling fraction of petroleum), gasoline, and kerosene to meet military or commercial specifications (U.S. Air Force 1989). Jet fuels are composed of aliphatic, monocyclane, aromatic, and alkene hydrocarbons in the C5 to C16 range. Aliphatic alkanes (paraffins) and cycloalkanes (naphthenes) are the major constituents (75-90%) of kerosene (Cavender 1994a,b,c; reviewed in ATSDR 1998). The boiling range for jet fuels is usually well above that of benzene and n-hexane. Conversely, the maximum final boiling point of middle distillate fuels tends to exclude the presence of high-boiling polycyclic aromatic hydrocarbons. The composition of jet fuels varies depending on the type of crude oil from which the fuel is derived, the refining process used, and the additives. Additives include antioxidants, metal deactivators, corrosion or icing inhibitors, and electrical conductivity agents (reviewed in ATSDR 1998). The major vapor-phase hydrocarbon components of JP-8 are listed in Appendix A.
TABLE 2-1 Summary of AEGL Values for JP-5 and JP-8a,b
| Classification | 10 min | 30 min | 1 h | 4 h | 8 h | End Point (Reference) |
| AEGL-1 (nondisabling) | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | Slight sensory irritation in humans (extrapolated from mouse RD50 test) (Whitman and Hinz 2001) |
| AEGL-2 (disabling) | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 | No clinical signs during repeated exposures at 1,000 mg/m3 to rats and mice (Mattie et al. 1991; Briggs 2001; Rossi et al.. 2001); sensory irritation at >3,430 mg/m3 in rats and mice (Wolfe et al. 1996; Whitman and Hinz 2001) |
| AEGL-3 (lethal) | Not determined | Not determined | Not determined | Not determined | Not determined | No datac |
aThe values apply to JP-8 vapor or vapor and aerosol and not to the pure aerosol.
bThe values apply to JP-8 vapor and not to JP-8+100.
cA lethal concentration was not attained in the available toxicity studies; the low vapor pressures of JP-8 and JP-5 may preclude attainment of a lethal concentration. Abbreviation: RD50, concentration that reduces the respiratory rate by 50%.
The present document focuses on the toxicity of JP-8, the jet fuel used by the U.S. military. Information on JP-5 (used by the Navy for shipboard aircraft) is included in this document because, chemically, JP-5 can be considered a subset of JP-8 (ATSDR 1998; Potter and Simmons 1998). Both JP-8 and JP-5 are middle distillates with boiling ranges of 150-275°C. JP-8 contains alkane carbon ranging from n-C8 through n-C17; whereas JP-5 contains carbons ranging from n-C7 through n-C18 (Potter and Simmons 1998). Prior to 1979, JP-4—a naphtha-based, wide-cut fuel made from straight-run, desulfurized kerosene blended with lower boiling distillates or made by blending refined shale oil distillates (ATSDR 1995)—was used by the Air Force. JP-4 was replaced by JP-8 in 1994, and JP-8 is now the standardized fuel for the U.S. military. Thus, the human monitoring and animal toxicity studies with JP-4 are not discussed in this document. Data on JP-7, a specialized high-altitude fuel restricted to reconnaissance aircraft (MacNaughton and Uddin 1984), are not included since JP-7 is no longer used.
JP-5 is a turbine engine fuel developed by the U.S. Navy for use aboard aircraft carriers because of its lower volatility and lower post-crash fire hazard compared with JP-4 (ATSDR 1998). JP-5 has a specified distillation temperature of 205°C for the 10% recovery point to 290°C for the end point (Military Specification MIL-T-5624K [1976]). The U.S. Naval Service is anticipating transition from the nearly exclusive use of JP-5 to predominant use of JP-8, consistent with the other military services and the militaries of most NATO countries.
Compared with JP-4, the less volatile JP-8 contains alkanes in the C8 to C17 range. In a survey of JP-8 fuels, the average aromatic content was 14.5%, the highest aromatic content reported being 18.8% (Martel 1989). The composition (v/v) of JP-8 consists of approximately 9% C8 to C9 aliphatic hydrocarbons, approximately 65% C10 to C14 aliphatic hydrocarbons, approximately 7% C15 to C17 aliphatic hydrocarbons, and approximately 18% aromatic hydrocarbons (NRC 1996; Carlton and Smith 2000). Typical aromatic hydrocarbons include benzene, ethylbenzene, toluene, and xylenes, but the distillation fraction of JP-8 minimizes the presence of benzene and related low-boiling aromatic hydrocarbons. Ambient air samples in aircraft fuel tank maintenance areas are dominated by C9 to C12n-alkanes; the primary n-alkanes in these samples are nonane (C9), decane (C10), and undecane (C11) (Pleil et al. 2000). The benzene content is 0.005% by volume (Carlton and Smith 2000). The typical aromatic hydrocarbons in JP-8 are the polycyclic aromatics and not the lighter aromatics, such as benzene, toluene, xylenes, and ethyl benzene found in gasoline (Appendix A).
Only the studies of Carlton and Smith (2000) discuss benzene exposures measured during maintenance operations on military aircraft fuel tanks. The exposures occur inside the fuel tanks or with personnel removing foam from the tanks. The latter operation involves the generation of aerosols as the foam is pulled out of the fuel tank. Benzene is more water soluble than other jet fuel components, and some benzene remains in the small amount of water present after many refuelings. This amount can result in measurable benzene concentra-
tions during these operations even though the levels of benzene in the bulk fuel are not detectable (see Appendix A). Benzene is not a component of concern for JP-8 AEGLs.
Except for additives included to meet military specifications, JP-8 is similar to international jet fuels A and A-1, the former used in U.S. commercial aircraft. JP-8 contains antioxidants, static inhibitors, corrosion inhibitors, fuel system icing inhibitors, lubrication improvers, biocides, and thermal stability improvers (Military Specification MIL-T-5624P [1992]). According to the Navy Environmental Health Center, additives to JP-8 typically compose <2% of the volume (NEHC 2001). Addition of antioxidants—such as 2,6-di-tert-butyl-4-methylphenol—or metal deactivators—such as N,N-disalcylidene-1,2-propanediamine—is optional. Static dissipaters—such as Stadis 450 (50-60% toluene)—organic acid corrosion inhibitors (8Q21), and icing inhibitors—such as diethylene glycol monomethyl ether—are required. JP-5 differs in that an antioxidant is required and a metal deactivator and static dissipater are not used.
To improve the thermal stability of JP-8, a proprietary package of additives including an antioxidant (butylated hydroxytoluene), a metal deactivator (N,N-disalicylal-1,2-propane diamine), and a detergent and dispersant (8Q405) are added at concentrations of 100-300 ppm. The resulting fuel is called JP-8+100 (Wolfe et al. 1996; Kornguth 1998). JP-8+100 is not widely used at present.
The chemical identification and chemical and physical properties of JP-8 and JP-5 are summarized in Table 2-2. Many of the physical properties of JP-8, such as autoignition temperature (229ºC), and flammability and explosive limits, both 0.7%-5%, are identical to those of kerosene (ATSDR 1998). The flashpoint is 38ºC, indicating that fire is the major hazard associated with jet fuels. Because of the complex and variable composition of jet fuels, the molecular weight is expressed as an average, and concentrations are expressed in terms of their total hydrocarbon content measured in mass units (mg/m3).
Worldwide, approximately 60 billion gallons of JP-8 and commercial Jet A and Jet A-1 are consumed each year (Armbrust Aviation Group 1998). Annual use of JP-8 by the U.S. military services and North Atlantic Treaty Organization forces is estimated at 4.5 billion gallons (Zeiger and Smith 1998). The U.S. military utilization of JP-8 and JP-5 exceeds 2.2 billion gallons per year (Henz 1998). In addition to fueling aircraft and tanks, the military uses JP-8 for heating tents and buildings.
Exposure to jet fuels can occur during production and refining, monitoring of storage tanks, aircraft fueling and defueling, spills during handling, and leaks at storage facilities. Under some conditions, aircraft jettison excess fuel into the upper atmosphere (ATSDR 1998; Rossi et al. 2001). Annually, several hundred thousand military personnel are involved in these operations. Thus, exposure to JP-8 represents the largest single source of chemical exposure in the U.S. military (Pleil et al. 2000); civilian exposure is restricted to the chemically similar Jet A used in commercial aircraft.
TABLE 2-2 Chemical and Physical Data for Jet Fuels 8 and 5
| Parameter | Data | Reference |
| Synonyms | ||
| JP-8 | Kerosene, aviation kerosene, fuel oil number 1, jet kerosene, turbo fuel A, straight run kerosene, distillate fuel oil-light, MIL-T-83133D, AVTUR, NATO F-34 | ATSDR 1998, NRC 1996, Chevron Phillips 2009a |
| JP-5 | Kerosene, MIL-T-5624N | Chevron Phillips 2008 |
| Molecular formula | Not applicable | |
| Structure | Not applicable | |
| Molecular weight (mean) | ||
| JP-8 | 167, 180 | MacNaughton and Uddin 1984 |
| JP-5 | 168, 170, 185 | NIOSH 2005; NRC 1996 |
| CAS Registry Number | ||
| JP-8 | 8008-20-6a/70892-10-3b | ATSDR 1998 |
| JP-5 | 8008-20-6a/70892-10-3b | ATSDR 1998 |
| Physical state | ||
| JP-8 | Clear-to-light amber liquid | ATSDR 1998; Richie et al. 2001a |
| JP-5 | Clear liquid | ATSDR 1998 |
| Solubility in water | ||
| JP-8 | 5 mg/L (kerosene) | ATSDR 1998 |
| JP-5 | 5 mg/L (kerosene) | ATSDR 1998 |
| Density (specific gravity) | ||
| JP-8 | 0.81 g/mL | Potter and Simmons 1998 |
| JP-5 | 0.82 g/mL | Potter and Simmons 1998 |
| Vapor pressure | ||
| JP-8 | 1.8 mmHg (28°C) 0.4-3.3 mmHg (20°C) |
NRC 1996 SwRI 2001 |
| JP-5 | 5.9-26.4 mmHg (kerosene) 1.8 mmHg (28°C) |
ATSDR 1998 NRC 1996 |
| Vapor density, JP-8 (air = 1) | 4.5-5 | Ritchie et al. 2001a |
| Explosive limits, JP-8 | ||
| Lower explosive limit | 0.7-0.9% | Ritchie et al. 2001a |
| Upper explosive limit | 5-6% | |
| Flash point | ||
| JP-8 | 37.8°C | Chevron Phillips 2010 |
| JP-5 | 60°C | Chevron Phillips 2009b |
| Parameter | Data | Reference |
| Liquid density (water = 1) | ||
| JP-8 | 0.788-0.845 kg/L | ATSDR 1998 |
| JP-5 | 0.788-0.845 kg/L | ATSDR 1998 |
| Melting point | ||
| JP-8 | −52ºC | ATSDR 1998 |
| JP-5 | −46ºC | ATSDR 1998 |
| Boiling range | ||
| JP-8 | 150-275ºC | Potter and Simmons 1998 |
| JP-5 | 150-275ºC | Potter and Simmons 1998 |
| Conversion factors (STP)c | ||
| JP-8 | 1 ppm ≈ 8 mg/m3 | NRC 1996 |
| 1 mg/m3 ≈ 0.12 ppm | ||
| JP-5 | 1 ppm ≈ 8.3 mg/m3 | NRC 1996 |
| 1 mg/m3 ≈ 0.12 ppm | ||
aThe CAS Reg. No. is that of kerosene.
bThe CAS Reg. No. is that of fuel oil no. 1.
cConversion factors at standard temperature and pressure (STP) are based on the average molecular weight.
2. HUMAN TOXICITY DATA
At sufficiently high exposures, liquid and vapor JP-8 may be irritating to the eyes and skin. Dermal exposure may cause defatting, drying, and irritation of the skin (U.S. Air Force 1989). Topical exposure can induce skin inflammation, which has been documented by morphologic and ultrastructural changes (ATSDR 1998; McDougal and Rogers 2004; Monteiro-Rivere et al. 2004). Workers exposed to jet fuels have complained of dizziness, headache, nausea, and fatigue (NRC 1996; ATSDR 1995, 1998). Aspiration of the liquid fuel into the lungs can give rise to chemical pneumonitis.
The toxicity data of various jet fuels have been summarized and reviewed in IARC (1989), ATSDR (1995, 1998), Bruckner and Warren (2001), Ritchie et al. (2001a, 2003), and NRC (2003). Past exposures to concentrations as high as 3,000 mg/m3 were to the more volatile JP-4 and equivalents (Knave et al. 1978; Martone 1981). Increased complaints of dizziness and fatigue have been associated with these concentrations. The low vapor pressure of JP-8 and JP-5 and the moderately high average molecular weights indicate that their relatively low volatility is such that a systemic health risk from vapor inhalation is unlikely (ACGIH 2009). In its toxicologic assessment of JP-8, the NRC (2003, pp. 4-5) noted: “No relevant adverse effects were observed for hepatotoxicity, renal toxicity, and cardiovascular toxicity, although the exposure concentrations did not
exceed 1,000 mg/m3. Adequate studies have not been conducted to assess the potential toxicity of inhaled JP-8 for reproductive toxicity, developmental toxicity, and genotoxicity”.
2.1. Acute Lethality
No reports of humans fatalities associated with JP-8 or JP-5 exposure were located in the available literature.
2.2. Nonlethal Toxicity
The odor thresholds of JP-8 and JP-5 have been reported at 1 ppm and 0.082 ppm, respectively. The odor is described as similar to that of kerosene (ATSDR 1998).
Olsen (1998) compared liver function, kidney and hematopoietic system function, serum proteins, neurocognitive function, and general physical health of 18 Air Force personnel exposed to jet fuels with 18 nonexposed subjects. The exposed subjects were evaluated while exposed to JP-4 and at 3, 6, and 18 months after JP-8 replaced JP-4. Exposure to naphthas was <3 ppm. Benzene concentrations were 0.05 ppm during exposure to JP-4 and nondetectable during exposure to JP-8. No significant differences were found between exposed and nonexposed subjects with regard to liver and kidney function, frequency of symptoms, or general physical health. Two of the subjects exposed to JP-8 developed a rash on their hands. After 18 months of exposure to JP-8, several hematopoietic parameters were affected in that exposed workers had lower mean corpuscular volume and mean corpuscular hemoglobin and higher mean corpuscular hemoglobin concentration (smaller cells with a higher concentration of hemoglobin) than the nonexposed subjects.
Norseth et al. (1998) measured circulating serum alanine aminotransferase (formerly called serum glutamic pyruvic transaminase) and serum aspartate amino transferase (formerly called serum glutamic oxaloacetic transaminase), and glutathione transferase liver enzyme activities as an indicator of liver damage in Norwegian crew chiefs exposed to JP-8. Exposures were to C5-C9 aliphatic hydrocarbons at 0.13 ppm and to C9-C13 at 3.11 ppm. Compared with controls, there were no meaningful differences between the two groups.
2.2.1. Clinical Studies
Because JP-8 is a kerosene-based fuel, the results of human exposures to kerosene offer useful comparisons. When six volunteers (age range 23-49 years) inhaled several different concentrations of deodorized kerosene, the odor threshold was 0.6 mg/m3 (0.09 ppm) (Carpenter et al. 1976). The kerosene consisted
of 55.2% paraffins, 40.9% naphthenes, and 3.9% aromatics and had a boiling range of 208-272°C. There were no complaints of irritation or discomfort when six volunteers (age range 20-63 years) were exposed to a measured concentration of 140 mg/m3 (20 ppm) for 15 min. Three of the volunteers experienced slight olfactory fatigue. The authors reported that 14,000 mg/m3 is the highest obtainable vapor concentration of deodorized kerosene at 25°C.
2.2.2. Accidental Exposures
Two individuals were exposed for 1 h to an unknown concentration of JP-5 in the cockpit of an unpressurized aircraft (Porter 1990). The odor was described as “overwhelming”, and the individuals experienced burning eyes and euphoria (one individual) during exposure and complaints of headache, nausea, coordination difficulties, and transient memory defects after exposures were made. The effects subsided within 24 h in one individual and within 4 days in the other.
2.2.3. Monitoring Data
Because of its wide use in the past, most published monitoring data involve JP-4. Because of its higher volatility than JP-8, ambient air concentrations of JP-4 at military installations were higher than the currently measured concentrations of JP-8. Measured concentrations of JP-4 jet fuel inside aircraft shelters at bases ranged from 33 to 3,090 mg/m3 and were dependent on temperature and shelter size. Concentrations of the less volatile JP-8 averaged <20 mg/m3 at three shelters. Refueling normally took 3-5 min, although in one case, aircraft refueling associated with a measured exposure concentration of JP-4 at 620 mg/m3 took 30 min (Martone 1981). At Swedish and Danish military bases where aviation fuel was equivalent to JP-4, maximum 5-min workplace concentrations ranged up to 3,226 mg/m3 (Knave et al. 1978), and 8-h time-weighted averages (TWAs) ranged up to 3,000 mg/m3 (Knave et al. 1978; Thomas and Richardson 1981; Holm et al. 1987; Døssing et al. 1985; Selden and Ahlborg 1986, 1987). Vapor concentrations often exceeded 350 mg/m3 (a specific exposure duration was not given) (Selden and Ahlborg 1986, 1987); exposure durations to unspecified concentrations ranged up to 31 years (Døssing et al. 1985).
Workplace air concentration data for JP-8 and JP-5 are summarized in Table 2-3. The highest concentrations of JP-8 were measured inside empty aircraft fuel tanks during maintenance and foam removal. Workers who enter the fuel tanks wear a supplied air respirator or a self-contained breathing apparatus, whereas the outside attendants do not. Therefore, information on potential adverse health effects (from JP-8 and JP-5 inhalation) could not be derived from these studies.
TABLE 2-3 Monitoring Data for JP-8 and JP-5
| Fuel Type | Concentration | Exposure Duration | Reference |
| JP-8 | <20 mg/m3 | Min (fueling time) | Martone 1981 |
| JP-8 | 1.83 ppm (naphthas)a | Ambient concentrations over work shift | Puhala et al. 1997 |
| JP-8 | <3 ppm (naphthas) | 18 mon | Olsen 1998 |
| JP-8 | 0.13 ppm (C5-C9) | Routine exposures | Norseth et al. 1998 |
| 3.11 ppm (C9-C13) | |||
| JP-8 | Inside fuel tanks:b | Smith and Zelnick 1998 | |
| 0.12-2,308 mg/m3 | 8-h TWA | ||
| 17-10,295 mg/m3 | 15-min samples | ||
| JP-8 | Inside fuel tanks; tanks with no foam: | Carlton and Smith 2000 | |
| 52 mg/m3 (range, 4-954 mg/m3) 14 mg/m3 Fuel tanks with foam: 431 mg/m3 (range, 7-10,295 mg/m3) 183 mg/m3 | 15-min TWA 8-h TWA 15-min TWA 8-h TWA |
||
| JP-8 | Outside fuel tank: 2.7 ppm (C8-C12) Inside fuel tank: 104 ppm (C8-C12) | Routine aircraft maintenance | Pleil et al. 2000 |
| JP-8 | 0.54 ppm (naphtha) | 8-h TWA | Smith et al. 1997 |
| JP-8 (mist) | 10->200 mg/m3 | Dissipated in <1 min | Leith et al. 1998 |
| JP-8 (aerosol) | 16-119 mg/m3 | Min to h | Robledo and Witten 1998 |
| JP-5 | <0.48-153 mg/m3 (range) 4.4 mg/m3 (mean) | TWA | NRC 1996 |
aDefined as all vapor phase hydrocarbons expected form JP-8.
bSamples were taken inside aircraft fuel tanks during maintenance operations; workers wear supplied air respirators during tank entry.
Abbreviation: time-weighted average.
Data from the U.S. Navy Occupational Safety and Health Program were reported by the National Research Council (NRC 1996). TWA personal-exposure measurements of JP-5 vapor, taken from November 1984 to February 1993, ranged from <0.48 to 153 mg/m3. The geometric mean was 4.4 mg/m3.
Smith and Zelnick (1998) reported the results of JP-8 and benzene monitoring taken during aircraft fuel tank entry during maintenance at U.S. Air Force
bases. Samples were collected on charcoal tubes and analyzed by gas chromatography with a flame ionizing detector. A total of 250 15-min short-term exposure limit (STEL) samples were taken; concentrations ranged from 17-10,295 mg/m3 for JP-8 and 0.06-41 mg/m3 for benzene. Calculated 8-h TWA concentrations ranged from 0.12-2,308 mg/m3 for JP-8 and 0.002-3.3 mg/m3 for benzene. As noted, these maintenance workers wear respirators during fuel tank entry, and inhalation exposure was precluded.
Pleil et al. (2000) reported personal monitoring data for JP-8 at various Air Force bases during routine operations. A number of marker compounds, more specific to JP-8 exposure than other fuels or solvents, were measured. These marker compounds were also monitored in exhaled breath of service personnel. Concentrations of individual fuel components in indoor air and in the vicinity of exhaust from aircraft cold-starts were all <0.02 ppm. Of the marker compounds, nonane, decane, o-xylene, undecane, and m,p-xylene were present in the highest concentrations in and around fuel tanks during maintenance operations. During fuel tank maintenance and foam removal, workers stationed outside the fuel tanks were exposed to mean nonane and decane concentrations at 1.8 and 0.6 ppm, respectively. Except for some aromatics such as o-xylene present at 0.2 ppm, most other components including benzene were present at ≈0.02 ppm. Inside the fuel tanks, mean concentrations of nonane, decane and decane were each 31-34 ppm. Summed concentrations of C8 through C12 (hexane through dodecane), outside and inside the tanks were 2.7 ppm and 104 ppm, respectively. Workers that entered the fuel tanks wore respirators, whereas the outside attendants did not. Analysis of breath samples from the latter two groups of workers showed nearly identical results. Therefore, the authors concluded that workers who entered the tanks had considerable dermal exposure (as well as inhalation exposure upon exiting the tanks).
At three U.S. Air Force bases in the United States, mean concentrations of individual components of jet fuels (JP-4, JP-5, and JP-8) ranged up to 0.009 ppm for benzene and 1.83 ppm for naphthas (Puhala et al., 1997). Maximum values at one of the bases were 4.04 ppm for naphtha and 0.03 ppm for benzene. Historical data indicated that exposures were to much higher concentrations when JP-4 was the primary fuel; at that time, the maximum TWA value for naphthas was 586 ppm, and the maximum TWA value for benzene was 13.2 ppm. Exposure concentrations were highest for aircraft maintenance workers.
Carlton and Smith (2000) measured exposures of personnel during aircraft fuel tank entry and repair at 12 U.S. Air Force bases. Different types of aircraft and fuel tank types (containing explosion suppression foam or no foam) were surveyed. The tanks were purged with air prior to worker entry. Workers who entered the tanks wore supplied air respirators during initial tank entry and foam removal; the assistant attendant and monitor who remained outside the tank but in close proximity did not wear a respirator. A total of 500 breathing zone samples involving 77 workers were taken. Nearly half of the samples were 15-min short-term samples. The partial period and short-term samples were used to calculate 8-h TWAs. The mean 15-min TWAs were 52 mg/m3 (range, 0.1 to 1,304
mg/m3) in tanks containing no foam to 430 mg/m3 (range, 4 to 10,295 mg/m3) in tanks containing foam. Respective mean 8-h TWA exposures were 14 mg/m3 and 183 mg/m3. Benzene concentrations in grab samples taken in the fuel tanks were also measured, and although in one case ranged up to 49 mg/m3, short-term and 8-h TWA values were 4.6 and 0.74 mg/m3, respectively.
Aerosols of JP-8 have been observed when ambient temperatures are low. At Eilson Air Force Base in Alaska, mist concentrations of 10 to >200 mg/m3 were present, but generally lasted less than 1 min (Leith et al. 1998). During refueling and preflight operations at Davis-Monthan Air Force Base (Tucson, Arizona) and Montana Air National Guard Base (Great Falls, Montana), time-weighted aerosol concentrations of JP-8 were measured at 16 to 119 mg/m3 (2-to 6-h exposures) (Robledo and Witten 1998). The 119-mg/m3 concentration composed a single sample during a 3.5-min refueling (Pfaff et al. 1995).
Additional JP-8 occupational exposure data taken in conjunction with complaints of symptoms are discussed in Section 2.3 (Neurotoxicity).
2.3. Neurotoxicity
The neurotoxicity of selected hydrocarbon fuels was reviewed by Ritchie et al. (2001b) who addressed exposure to low levels of certain volatile organic chemical constituents of hydrocarbon fuels.
Smith (1998) enumerated the anecdotal health complaints from ground crews handling JP-8. The complaints included headaches and dizziness, offensive odor, and local damage resulting from direct skin contact. Concentrations were not reported. Results of tests of neurocognitive function found no significant differences between personnel exposed to JP-8 compared with 18 nonexposed subjects (Olsen 1998). Details of the latter study were not reported in the available abstract.
Smith et al. (1997) measured the effect of chronic low-level JP-8 exposure on postural balance of a group of representative U.S. Air Force personnel; 27 workers employed in jet-fuel-related occupations at two bases for an average of 12 years (range, 0.8 to 30 years; average exposure to JP-8, 4.56 years) were compared with a matched control group of 25 workers. The mean age at time of evaluation was 37.5 years (range 23.6 to 57.4 years); there were 20 males and 7 females. Thirty-seven percent of the group had worked only with JP-8. Monitoring data for benzene; toluene; m-, o-, and p-xylenes; and naphthas were taken on two separate 8-h work periods for each worker. Postural sway movements while standing on a platform were measured electronically. Statistically significant associations between exposure and increased postural sway, particularly for benzene, toluene, and xylene, were found. The strongest association was between sway length and benzene concentration, and this association was strongest when tests were conducted under the most difficult condition—eyes closed and standing on 4 inches of foam. According to the authors, this association indicated a subtle influence on vestibular and proprioception function. To measure
the effect of acute exposures, the time of day of the test administration was compared with the sway results. There was no difference between exposed subjects tested early in the working day and subjects tested later in the day. The effects were not adjusted for concomitant exposure to other chemicals. Eight-hour TWA exposures to components of all fuels for all job categories were: benzene, 0.006 ppm; toluene, 0.01 ppm; xylenes, 0.008 ppm; and naphthas, 0.54 ppm. Exposures to all JP-8 components in milligrams per cubic meter could not be calculated from the published data.
An eye-blink conditioning test was used to evaluate potential neurologic changes in military personnel exposed chronically to JP-8 (McInturf et al. 2001). Workers with JP-8 exposure were matched with a control group of military personnel. Subjects learned a classically conditioned response between an auditory stimulus and a corneal air puff, the conditioned response. Subjects were tested for capacity to learn the response after a rest period from occupational exposure and for recall of the response following 4 h of occupational exposure to JP-8. Compared with matched controls, the JP-8 exposed workers were slow to learn the response and had an increase in mean time from onset of the stimulus to the eye-blink response. No further details were given in the available abstract.
2.4. Immunotoxicity
Rhodes et al. (2001) reported increased white blood cell counts in military personnel exposed to JP-8, compared with a low-exposure group, but these changes were within normal clinical ranges. Olsen (1998) found no difference in total white blood cell count and differential counts among Air Force personnel before and 18 months after the Air Force converted to JP-8.
2.5. Developmental and Reproductive Effects
No studies regarding human exposure and aspects of developmental toxicity were located in the published literature. In a study that examined sperm quality (concentration, motility, viability, morphology, morphometry, and stability of sperm chromatin) in 50 aircraft maintenance workers at an Air Force installation at 15 and 30 weeks after exposure to both JP-8 and solvents began, there was no significant association between sperm quality of maintenance workers and jet fuel exposure (primarily JP-4) (LeMasters et al. 1999). Exposures were low, as all measured fuel components—naphthas, benzene, xylenes, toluene, and so forth—and a solvent—1,1,1-trichloroethane—were below 6 ppm.
2.6. Genotoxicity
Addition of petroleum-derived JP-5 failed to interfere with sarcoma virus transformation of cultured human fibroblasts (ATSDR 1998). Incubation of cul-
tured human lymphocytes with JP-8 at dilutions of 1:75 to 1:500 resulted in increasing DNA damage with increasing dose, as measured using the Comet assay (Jackman et al. 2001).
2.7. Carcinogenicity
Following a review of available human data on occupational exposure to military fuel vapors, the NRC (2003) concluded that the available data are insufficient to draw a conclusion regarding the carcinogenicity of inhaled JP-8. Based on inadequate evidence for the carcinogenicity of jet fuels in humans and animals and the limited evidence of carcinogenicity in experimental animals of straight-run kerosene and of hydrotreated kerosene, IARC (1989) concluded that jet fuel is not classifiable as to its carcinogenic potential in human beings. According to ATSDR (1998), there are limited epidemiologic data regarding carcinogenicity in humans following chronic inhalation exposure to kerosene.
2.8. Summary
At sufficiently high exposures, JP-8 liquids and vapors may be irritating to the eyes and skin. Dermal exposure to neat JP-8 can cause skin irritation and skin damage (Olsen 1998; Smith 1998). The primary effect of acute inhalation exposure to JP-8 vapor is on the CNS where high concentrations result in dizziness, headache, nausea, and fatigue (Davies 1964; Porter 1990).
Workplace monitoring data indicate that in the past exposures to vapors of JP-4 were relatively high, whereas exposures to the currently used JP-8 are comparatively low. Measured concentrations of JP-4 ranged up to 3,090 mg/m3 at U.S. air bases (Martone 1981) and 3,226 mg/m3 at a Swedish jet motor factory (Knave et al. 1978). In the later study, the overall mean TWA was 300 mg/m3, and the highest average was 974 mg/m3. TWA exposures to JP-5 at Navy sites ranged up to 153 mg/m3 (NRC 1996). Although the Swedish study did not correlate symptoms with exposure, some of the acute exposures may have been associated with headache and dizziness. Most occupational monitoring studies reported total hydrocarbon vapor concentrations, but later reports indicated that aerosols may be present during aircraft fueling operations. More recently, aerosols of JP-8 of up to 119 mg/m3 have been measured in the vicinity of aircraft refueling operations (Pfaff et al. 1995). Particles are most commonly generated during start-up of cold jet engines. Emergency exposures are expected to be to spills resulting in vapor exposures, while aerosols are relevant only to occupational exposures during aircraft foam removal operations or aircraft cold starts. Exposures to high concentrations of JP-8 have occurred during jet aircraft fuel tank maintenance, but personnel wear respirators when entering the fuel tanks, thus minimizing inhalation exposure.
Occupational exposures may have had some influence on the hematopoietic system (Olsen 1998) and liver enzymes (Norseth et al. 1998). Increased
postural sway correlated with occupational exposure to routine concentrations of benzene, toluene, and xylene and was associated with cumulative exposure (Smith et al. 1997). On the basis of the results of the epidemiologic studies (primarily JP-4), including Selden and Ahlborg (1986, 1987, 1991), the NRC (1996, p. 5) concluded that the studies of “Swedish military personnel exposed to jet-fuel vapors at concentrations greater than 350 mg/m3 for several years did not show increased evidence of cancer.” No studies that addressed potential developmental effects in humans were located.
3. ANIMAL TOXICITY DATA
3.1. Acute Lethality
During attempts to generate a vapor concentration of JP-5 of 1,500 mg/m3 (the highest stable concentration attainable), a respirable aerosol was produced that resulted in 50% mortality of C57BL/6 mice by the end of 6 days continuous exposure (Gaworski et al. 1984; MacEwen and Vernot 1985). Oily deposits were observed on the animals and on the chamber windows. Aerosol counts indicated 3.6 × 105 respirable particles per cubic foot (ft3) (0.5-1.4 µm diameter) at this concentration. Young adult beagles were lethargic during exposures to this same concentration of vapor and aerosol.
3.2. Nonlethal Toxicity
3.2.1. Eye and Dermal Irritation
JP-8 was tested for eye and skin irritation in rats and rabbits and skin sensitization in mice and guinea pigs (MacEwen and Vernot 1984; Clark et al. 1989; Kinkead et al. 1992a,b; Kanikkannan et al. 2000). The undiluted test material (0.1 mL) was not irritating when instilled into the eyes of rabbits. The undiluted test material, placed on the intact or abraded skin and covered with an occluded patch for 24 h, produced a slight to moderate amount of reddening (MacEwen and Vernot 1984). Neat JP-8 showed a weak-to-mild sensitization reaction in guinea pigs and mice (MacEwen and Vernot 1984; Kinkead et al. 1992b; Kanikkannan et al. 2000).
JP-5 was nonirritating to the eyes and skin of New Zealand white rabbits, but it was a mild-to-moderate dermal sensitizer (Cowan and Jenkins 1981; Cowan et al. 1981; Kinkead et al. 1992a). In a more recent study, neat JP-8 applied to rabbit skin failed to elicit irritation, and repeated application to the skin of guinea pigs failed to elicit a sensitization reaction (Wolfe et al. 1996). In vitro studies with porcine or human skin showed that JP-8 penetrates the skin (Riviere et al. 1999; Kanikkannan et al. 2001), albeit not at concentrations sufficient to cause systemic toxicity (McDougal et al. 2000; McDougal and Robinson 2002).
Permeation of individual chemical components was chemical-specific. In vivo, neat JP-8 was more irritating to pig skin than an equal volume of neat toluene or neat nonane (Kanikkannan et al. 2001). When equal volumes were applied dermally, JP-8 was more irritating to rat skin than JP-4 (Baker et al. 1999). Dermal application of 25 µL of Jet A, JP-8, or JP-8+100 to pig skin in vivo under occluded or nonoccluded conditions for 5 or 24 h or 5 days continuous contact resulted in no erythema and very slight edema for all fuels only after 5 days and only under occluded conditions (Monteiro-Rivere et al. 2001). Fabric soaked with 335 µL of the fuels (to mimic worker exposure) and applied every day for 4 days under occluded conditions had the greatest effect, resulting in slight erythema and edema on day 5. Under this latter condition, epidermal thickening occurred and epidermal rete peg depth increased. The epidermal proliferative response was greatest with JP-8+100. Although irritation and dermal absorption may occur with exposure to the liquid fuel, the dermal absorption route was not considered significant for exposure to the vapor.
3.2.2. Inhalation Toxicity Studies
Several acute vapor inhalation studies with JP-8 and JP-5 and utilizing the rat and mouse were located (Table 2-4). Longer-term studies of 6 weeks to 3 months, the latter with continuous exposure of mice, rats, and dogs (JP-5) or mice and rats (JP-8) are included to compare with values derived from acute exposures. These studies addressed sensory irritation as well as systemic effects. Generation of high concentrations of vapor in laboratory chamber studies requires introduction of a mixture of vapor and aerosol (NRC 2003). Several studies with JP-8 aerosol alone, usually with repeated exposures, also used rats and mice. “The animal data show toxicity from aerosol inhalation. However, these publications do not provide adequate information to permit a judgment of aerosol size and stability nor do they speak to the extent to which the sampling systems distinguished between aerosol and vapor” (Dietzel et al. 2005; ACGIH 2009). The NRC (2003, p. 3) reviewed the methods used to generate the exposure atmospheres in the aerosol studies and “suspects that the total JP-8 concentrations in the atmosphere may have been underreported.” The data collected during aerosol inhalation studies are included in this TSD for completeness. A distinction is made in Table 2-4 between exposure to the vapor and exposure to aerosols. Where available, data on the source of the jet fuel (that is, petroleum or shale derived) is specified in the first column of Table 2-4 (although the available data indicate no substantial differences in toxicity between petroleum and shale-derived JP fuels).
Many of the reported studies were performed in the same laboratory and used the same methodology. The inhalation studies conducted at Wright-Patterson Air Force Base used 23.4 m3 Thomas domes, and all animals were exposed in groups, which were continuously monitored for toxicity during the
TABLE 2-4 Summaries of Studies on the Toxicity of Jet Fuels to Mammalian Species
| Fuel Type | Species | Exposure Concentrationa | Exposure Duration | Effects | Reference |
| JP-8 vapor and aerosol | Swiss-Webster mice (M) | 681, 1,090, 1,837, 3,565 mg/m3 | 30 min | No clinical signs RD50 of 2,876 mg/m3 | Whitman and Hinz. 2001 |
| JP-8 vapor and aerosol | F344 rat (M, F) F344 rat (M, F) | 3,430 mg/m3 4,440 mg/m3 | 4 h 4 h | No deaths; eye/upper respiratory track irritation No deaths | Wolfe et al. 1996; Feldmann et al. 1997 |
| JP-8 aerosol | C57BL/ 6 mice (M) | 5.0, 11.7, 27.8, 50.0, 112.5 mg/m3 | 1 h |
No changes in pulmonary function parameters; 27.8 mg/m3: increase in BALF parameters; 50 mg/m: increased alveolar permeability; 112.5 mg/m3: microscopic/ultrastructural lung changes |
Robledo and Witten 1998 |
| JP-8 aerosol | B6.A.D. mice | 7, 12, 26, 48, 118 mg/m3 | 7 d, 1 h/d | No changes in pulmonary function parameters, 48 and 118 mg/m3: changes in BALF components; bronchiolar edema, cellular necrosis and increased permeability | Robledo et al. 2000 , |
| JP-8 aerosol | C57BL/6 mice (M, F) | 0, 100, 250, 500, 1,000, 2,500 mg/m3 | 1 h/d, 7 d | Immediately postexposure: ≥500 mg/m3 : decreases in wet weight of spleen and thymus; decrease in viable cells of spleen; ≥100 mg/m3: decrease in viable cells of thymus; changes in numbers of immune cells of bone marrow, lymph nodes, and peripheral blood and in types of immune cells in all immune organs and tissues; alterations in immune function; many effects; and reversible at 28 days postexposure | Harris et al. 1997a,b |
| JP-8 aerosol | Swiss-Webster mice (M) | 0, 1,000, 2,500 mg/m3 | 1 h/d, 5 or 7 d | Lungs: changes in protein abundance; kidneys: changes in protein abundance | Witzmann et al. 1999; 2000b |
| JP-8 aerosol | F344 rat (M)e | 495 mg/m3 (7 d) 520 mg/m3 (28 d) | 1 h/d, 7 or 28 d |
Increased lung dynamic compliance (7 d); increased pulmonary resistance |
Pfaff et al. 1995, |
| Lower chemical mediator in BALF;f Increased alveolar permeabilityg | Chen et al. 1992, Pfaff et al. 1992, | ||||
| No pathologic lung changes | Witten et al. 1992, | ||||
| Lower body-weight gain | Parton et al. 1993 | ||||
| Increased organ weights (liver, spleen, kidney); no liver | Pfaff et al. 1993 | ||||
| lesions, ALT normal; kidney, spleen changes | |||||
| JP-8 aerosol | F344 rat (M)e | 495-520 mg/m3 813-1,094 mg/m3 | 7, 28, 56 d, 1 h/d 5 d/wk |
Changes in lung permeability: 28 d, both concentrations, 56 d, high concentration; partial recovery at 56 d |
Witten 1994 Hays et al. 1995 |
|
Lung histopathology (all groups): interstitial edema, epithelial thickening, vacuolization of type II cells |
|||||
| JP-8 vapor and aerosol | F344 Brown Norway rats (M) | 2,490 mg/m3 | 1 h/d, 5 d/wk, 4 wk | FOB: greater arousal and activity than controls; Morris swim task: no learning or memory deficits | Baldwin et al. 2001 |
| JP-8 vapor | Rat (M) | 0, 250, 500, or 1,000 mg/m3 | 6 wk, 6 h/d, 5 d/wk | No clinical signs; no effects on male fertility; no microscopic lesions in the testes; some biochemical changes in testes | Briggs 2001 |
| JP-8 vapor | Sprague-Dawley rat (M) | 1,000 mg/m3 | 6 wk, 6 h/d, 5 d/wk | No clinical signs; no effects in 9 of 10 neurobehavioral tests; significant decrease in response time in 1 of 10 tests, but no decrease in overall activity; some changes in brain neurotransmitter activities | Rossi et al. 2001 |
| JP-8 Vapor | Sprague-Dawley rat (M) | 500, 1,000 | 6 wk 6 h/d, 5 d/wk |
No clinical signs, no change in body weights;
62 days postexposure: 500 mg/m3: no effect and superior performance in operant behavior tests compared with controls; 1,000 mg/m3: threshold for operant task deficit; changes in brain neurotransmitters |
Ritchie et al. 2001c |
| JP-8 vapor | F344 rat (M, F) C57BL/ 6 mouse (M, F) | 500 mg/m3 1,000 mg/m3 | 90 d, continuous; killed at 0 wk to 21 mon | No clinical signs in either species; No or minor hematologic, clinical chemistry changes No lung lesions (rats, electron microscopy) No tumors | MacEwen and Vernot 1985; Mattie et al. 1991 |
|
Male rats: decreased body-weight gain accelerated chronic progressive nephrosis reversible kidney hyaline droplet formation liver basophilic foci, nondefinitive liver effects Mice: no treatment-related lesions |
| Fuel Type | Species | Exposure Concentrationa | Exposure Duration | Effects | Reference |
| JP-8+100 vapor and aerosol | Swiss-Webster mice (M) | 777, 1,519, 2,356 mg/m3 | 30 min | No clinical signs RD50 of 1,629 mg/m3 | Whitman and Hinz. 2001 |
| JP-5 Aerosol | F344 rat (M) C57BL/ 6 mouse (M) | 2,500, 5,000 mg/m3d | 1 h | Eye irritation (both concentrations) and CNS depression in both species (5,000 mg/m3); renal hyaline droplet formation with some urinary biochemical changes in male rats at 5,000 mg/m3; no other histopathologic effects | MacEwen and Vernot 1985 |
| JP-5 (P) | Sprague- | 1,125 mg/m3 (P) | 6 wk, | Increased water consumption; no neuropathies (no changes in SEP); no liver enzyme changes; no histopathologic effects | Bogo et al. 1983, 1984 |
| JP-5 (S) | Dawley rat (M) | 1,636 mg/m3 (S) | 6 h/d, 5 d/w | ||
| JP-5 vapor | Sprague-Dawley rat (M) | 1,200 mg/m3 | 6 wk, 6 h/d, 5 d/wk | No clinical signs; no effect on 9 of 10 neurobehavioral tests; increased forelimb grip strength; some changes in blood and brain neurotransmitter activities | Rossi et al. 2001 |
| JP-5 (P) | F344 rat (M, F) | 150 mg/m3 (P, S) | 90 d, | Necrosis of renal tubular epithelial cells and subnormal weight | Cowan and Jenkins 1981; |
| JP-5 (S) | C57BL/ 6 mouse (F) Beagle dog (M, F) | 750 mg/m3 (P, S)d | continuous; killed at 0 wk and 19-24 mon postexposure | gain in male rats exposed to JP-5 (P,S) at both concentrations; mild hepatocellular vacuolization in rats exposed to 750 mg/m3 JP-5/(S); minor hematology changes, liver glycogen accumulation in dogs exposed to JP-5 (P,S) at both concentrations; mild reversible liver cell changes and mild nasal inflammation in | Cowan et al. 1981; Gaworski et al. 1984, 1985; MacNaughton and Uddin 1984; MacEwen and Vernot 1985 |
a All exposures are to vapors except as otherwise noted; aerosol studies involve primarily vapor with some aerosol present.
b Saturated vapor (concentration estimated).
c The 5,000 mg/m3 concentration was chosen to produce a benzene concentration of 80 mg/m3 (25 ppm).
d An aerosol may have been present at this concentration.
e Nose-only exposure.
f Refers to Substance P; no differences for cell counts or the stable metabolite of prostacyclin.
g Lung epithelial permeability measured by clearance of technetium-labeled diethylenetriamine pentaacetate. Abbreviations: P, petroleum-derived jet fuel; S, shale-derived jet fuel; ALT, alanine aminotransferase; BALF, bronchoalveolar lavage fluid. FOB = functional observational battery (a series of tests designed to measure neurotoxicity); SEP = somatosensory evoked potential.
studies. Jet fuel vapors were generated by passing fuel through dual constant temperature evaporator towers operated at 50-57ºC and mixed with air to establish the desired atmospheric concentrations. Vapor concentrations were measured continuously using a Beckman model 400 hydrocarbon analyzer. The absence of aerosols was documented with a Royco aerosol particle counter. Chamber atmospheres were verified using gas chromatography and mass spectrometry (MacNaughton and Uddin 1984). Sufficient numbers of animals were used to provide statistical verification of the observations.
In a 6-week study with adult Sprague-Dawley rats (Bogo et al. 1983, 1984), petroleum- and shale-derived JP-5 was first aerosolized in an aerosol generator and then, following removal of particles >0.5 um, was vaporized and mixed with air; vapor generation was maintained with a heated liquid and air countercurrent flow system. The vapor concentration was calculated from the net loss of liquid fuel and the total volume of airflow through the system. A computer-controlled gas-sampling and chromatographic analysis system monitored the total hydrocarbons, oxygen, and carbon dioxide in the chambers. Rats were exposed in groups of six in 30-L Leach chambers.
The majority of studies with combined vapor and aerosol of JP-8 were conducted by staff at the University of Arizona: Pfaff et al. (1995), Hays et al. (1995), and Robledo and Witten (1998). Jet fuel was aerosolized by placing 3 mL of JP-8 in an Ultra-Neb 99 nebulizer (DeVILBISS). Rats were exposed nose-only in groups of 12 in a 0.5-m3 IN-TOX exposure chamber. Exposure concentrations were determined from changes in plate weights of a seven-stage cascade impactor after each exposure. Analysis was by gas chromatography. In the Pfaff et al. (1995) study, the aerosol to vapor mass ratio was 1.5. Particle size averaged 1.7 ± 2.2 µm (MMAD [mass mean aerodynamic diameter] ± GSD [geometric standard deviation]). In the Robledo and Witten (1998) study, particle MMADs for the different concentrations ranged from 2.0 ± 1.7 to 3.4 ± 2.3 µm. However, the actual aerosol and vapor concentrations in these studies are unclear (Dietzel et al. 2005). The JP-8 fuel used in many of the more recent studies was supplied by the Propulsion Directorate, Fuel Division, at Wright-Patterson Air Force Base. The fuel was prepared by blending approximately 250 fuel samples obtained from fuel manufacturers worldwide (Witzmann et al. 2000a) and is therefore considered a representative sample.
Studies addressing irritation and systemic toxicity of JP-5 and JP-8 are discussed in more detail below. Neurotoxicity, immunotoxicity, and developmental and reproductive toxicity studies are discussed in Sections 3.3, 3.4, and 3.5, respectively.
3.2.2.1. Dogs
Groups of three male and three female beagle dogs were exposed continuously to concentrations of 0, 150, or 750 mg/m3 of JP-5 petroleum or shale-oil derived vapor for 90 days (Gaworski et al. 1984; 1985). Density of aerosol par-
ticles (0.5 to 1.4 µm) was measured prior to the exposures and was found to be 1,100 and 6,200 particles/ft3, respectively, in the 150- and 750-mg/m3 exposure chambers; the particle count in laboratory air was 1,530 particles/ft3. The benzene concentrations averaged 0.1 and 0.5 ppm at the 150- and 750-mg/m3 exposures, respectively. All data for males and females were combined for analyses. These concentrations did not affect body-weight gain. Clinical chemistry parameters were within normal limits. Red-blood-cell osmotic fragility was increased in the petroleum-derived 750-mg/m3 group, and erythrocyte counts and hematocrit and hemoglobin levels were lowered in both the petroleum and shale-derived 750-mg/m3 groups, but the differences were not statistically significant compared with the control group. Dogs exposed to shale-derived JP-5 at 750 mg/m3 developed increased liver weights, and dogs exposed to petroleum-derived JP-5 (both concentrations) had glycogen accumulation in their livers. There were no other lesions.
3.2.2.2. Rats
JP-8. Wolfe et al. (1996; see also Feldman et al. [1977] for published abstract) exposed groups of five male and five female Fischer 344 (F344) rats to a target concentration of 5,000 mg/m3 of vaporized or aerosolized JP-8 for 4 h. The generation system consisted of two flasks (one for vapor and one for vapor and aerosol generation) containing a six-jet compressed air nebulizer. The flasks were kept in a 34ºC water bath. For the vapor study, an industrial HEPA filter was used to prevent aerosol from entering the exposure system. Under this system, the highest vapor concentration obtainable in the vapor-only exposure was 3,430 mg/m3. No deaths occurred, but animals exhibited signs of eye or upper respiratory irritation during exposure. Exposed animals lost weight on the first 1-2 days postexposure but gained weight during the remainder of the 14-day observation period. No control data for body weights were provided. No gross exposure-related lesions were observed. The vapor and aerosol concentrations during exposure to aerosolized JP-8 were 2,630 and 1,810 mg/m3, respectively for a combined exposure of 4,440 mg/m3. The particle MMD was 1.79 ± 1.60 µm. No deaths occurred, and exposed animals gained weight during the 14-day observation period. These tests were also performed with JP-8 containing several additive packages (JP-8+100) designed to increase thermal stability and decrease fuel fouling. Test results with the vaporized and aerosolized fuel plus additives were similar to those of the vaporized and aerosolized fuel alone. It should be noted that, under this experimental system, the vapor and vapor and aerosol atmospheres contained a higher percentage of the lower molecular weight hydrocarbons (C9-C11) than the neat fuel.
Groups of 95 male and 75 female F344 rats were exposed to JP-8 vapor at concentrations of 0, 500, or 1,000 mg/m3 continuously for 90 days (MacEwen and Vernot 1985; Mattie et al. 1991). Animals were killed immediately after cessation of exposure and up to 21 months postexposure. Clinical signs, body-
weight, and hematologic and clinical chemistry parameters were monitored during the exposures. No clinical signs of toxicity or biologically significant changes in hematologic and clinical chemistry were observed. Body weight of exposed males was significantly depressed during the exposures and postexposure (both concentrations), but there was no dose-response relationship. Twenty-one months after exposure, relative liver and kidney weights of male rats were increased and SGPT of female rats was reduced in the 1,000-mg/m3 group. Hyaline droplet formation in the kidneys of males was reversible by 2 months postexposure, and linear mineralization in the kidneys was reversible by 9 months postexposure. However, the incidence of chronic progressive nephrosis increased in males postexposure. A dose-dependent increased incidence of basophilic foci was observed in the livers of exposed males, but this effect is of uncertain biologic significance. The increased incidence of splenic hematopoiesis in female rats at 21 months was not accompanied by changes in hematologic parameters and thus was attributed to biologic variation. Scanning electron microscopy of the lungs of male rats revealed no differences between exposed and control groups.
In contrast to vapor-only studies, single and repeated exposures to JP-8 aerosol resulted in severe consequences. F344 rats were exposed nose-only to an aerosol and vapor mix of JP-8 for 1 h/day, 5 days/week for 7, 28, or 56 days to study pulmonary changes. Exposure concentrations ranged from 495 to 1094 mg/m3, and particle size for both exposures averaged 1.1 ± 2.2 µm MMAD. This study was reported in a series of articles and abstracts, the most recent being Pfaff et al. (1995) and Hays et al. (1995) (Table 2-4). Following 7 days of exposure at 495 mg/m3 or 28 days at 520 mg/m3, pulmonary resistance was significantly increased in both groups compared with their concurrent control groups, but resistance was not increased compared with baseline values (Pfaff et al. 1995). However, when corrected for body weight, resistance was significantly increased in the exposed groups compared with the control and baseline groups. Dynamic compliance was increased after the 7-day exposure but not after 28 days, indicating an adaptive response. The ratio of lung wet weights to body weights was also increased after the 7-day exposure but not after 28 days of exposure. Analysis of bronchoalveolar lavage fluid (BALF) revealed no differences in cell counts or 6-keto PGF1α, a measure of endothelial cell function; pulmonary alveolar clearance (as measured by 99mtechnetium-labeled diethylenetriamine pentaacetate) was increased as was substance P, a neuropeptide associated with airway reactivity. Light microscopic examination of the lungs found no differences between the exposed and control groups. Compared with the control groups, both exposed groups gained significantly less body weight during the exposures.
Following 56 days of exposure at 813-1,094 mg/m3, lung epithelial permeability was significantly increased; this effect was no longer present in the combined lower-dose group (average concentration reported at 500 mg/m3) (Hays et al. 1995). Electron microscopy revealed pulmonary inflammation with degeneration of Type II epithelial cells after 7 days of exposure at the high con-
centration and after 28 and 56 days of exposure at both 495 and 1094 mg/m3, the changes partially resolving in the low-dose group at 56 days. In general, the lungs had a normal appearance at 56 days. No liver pathology or liver enzyme changes were reported after 7 or 28-day exposures to aerosolized JP-8 at 5,000 or 1,000 mg/m3 (Parton et al. 1993).
Male Sprague-Dawley rats were exposed whole-body to 1,000 mg/m3 JP-8 vapor for 6 h/day, 5 days/week for 6 weeks (Witzmann et al. 2000a). During and following the exposures, there were no exposure-related deaths or visible signs of irritancy or distress. The mean body weight of the exposed group was slightly lower than that of the controls during exposure, but was similar to that of controls by 14 days postexposure (data not provided). At 82 days postexposure, liver and kidney were examined for changes in protein abundance and protein charge modification. Proteomic analysis revealed nonsignificant quantitative and qualitative alterations in the expression of lamin A in the liver and of 10-formyltetrahydrofolate dehydrogenase and glutathione-S-transferase in the kidneys of the exposed rats. Protein charge modification index analysis indicated significant alterations in the expression of lamin A and 10-formyltetrahydrofolate.
JP-5. Groups of 20 male F344 rats inhaled JP-5 vapor at 2,500 or 5,000 mg/m3 for 1 h (MacEwen and Vernot 1985). Generation of the 5,000 mg/m3 concentration resulted in development of an aerosol, which coated the fur of the animals. Ocular irritation as evidenced by mild lacrimation, eye closure, and pawing at the eyelids was observed during exposure at 5,000 mg/m3. Eye irritation (undefined) also occurred in animals exposed at 2,500 mg/m3. Lethargy and delayed righting reflex, which continued 2 h postexposure, were present at 5,000 mg/m3 but not at 2,500 mg/m3. During the postexposure period, there were some urinary biochemical changes, and at death, the animals exhibited hyaline droplet formation in the kidneys (see Sections 4.2 and 4.4.1 for a discussion of this nephropathy). There were no effects on body-weight gain or on liver or kidney weights in rats killed at 24 h or 28 days postexposure.
Groups of 75 male and 75 female F344 rats were exposed continuously to JP-5 petroleum or shale-derived vapor at 0, 150, or 750 mg/m3 for 90 days (Cowan and Jenkins 1981; Cowan et al. 1981; Gaworski et al. 1984; 1985). In addition to the vapor concentration, an aerosol may have been present in the chamber at the higher concentration. Male rats exposed to both concentrations of fuels from both sources developed renal tubular epithelial necrosis. Reduced body-weight gain, increased kidney/body-weight ratios, and slightly elevated blood urea nitrogen and creatinine levels were consistent with that effect. Those effects were absent in female rats, although exposure to the higher concentration of shale JP-5 resulted in a slight reduction in body-weight gain. Mild liver changes and mild nasal inflammation also occurred in rats exposed to shale JP-5, but these changes were not dose-related.
3.2.2.3. Mice
JP-8. The sensory irritation associated with JP-8 as well as that of JP-4 and JP-8+100 were evaluated by Whitman and Hinz (2001) using the standard RD50 test (ASTM E981-84 [1988]). Groups of four young male Swiss-Webster mice were administered the test compounds separately for a 30-min period via a head-only exposure system. The test atmospheres were administered as either vapor only (JP-4) or combined vapor and aerosols (JP-8 and JP-8+100). As noted, achieving high concentrations of JP-8 in laboratory chamber studies requires introduction of a mixture of vapor and aerosol (NRC 2003). Group mean vapor concentrations were 685, 956, 1,888, and 11,430 mg/m3 for JP-4 vapor; 681, 1,090, 1,837, and 3,565 mg/m3 for JP-8 vapor and aerosol; and 777, 1,519, and 2,356 mg/m3 for JP-8+100 vapor and aerosol. The proportion of aerosol in the JP-8 exposures ranged from 3% at 681 mg/m3 to 35% at 3,613 mg/m3. An additional group of four mice was exposed to JP-8 at 708 mg/m3 in the vapor-only phase. The test atmospheres were generated using a syringe pump to deliver the fuel to the inside surface of a heated glass countercurrent generator. The heated vapors were drawn into the exposure chamber with the air supply. Aerosol atmospheres were generated with a nebulizer. Atmospheres were collected on charcoal sorbant tubes, and exposure concentrations were determined as total hydrocarbon concentration via gas chromatography. These analyses showed that the lower molecular weight hydrocarbons were more abundant in the vapor samples than in the vapor and aerosol samples.
Exposure to JP-4, JP-8, and JP-8+100 evoked breathing patterns characteristic of upper airway sensory irritation. There was no indication of pulmonary irritation or narcosis at any of the concentrations examined. For JP-4, group mean respiratory rates decreased from baseline values by 11, 28, 51, and 59% at mean exposure concentrations of 685, 956, 1,888, and 11,430 mg/m3, respectively. The calculated RD50 for JP-4 was 4,842 mg/m3. For JP-8, group mean respiratory rates were decreased from baseline by 22, 38, 46, and 50% at mean exposure concentrations of 681, 1,090, 1,837, and 3,565 mg/m3, respectively (Table 2-5). The RD50 for JP-8 was 2,876 mg/m3. At the vapor-only exposure of 708 mg/m3, the respiratory rate was decreased by 28%, which is similar to the decrease of 22% observed with the vapor and aerosol concentration of 681 mg/m3. It should be noted that the irritant response tapered off at the highest exposure, that is, reductions in respiratory rate were similar at 1,837 mg/m3 (46%) and 3,565 mg/m3 (50%). For JP-8+100, group mean respiratory rates were reduced by 18, 53, and 63% at mean exposure concentrations of 777, 1,519, and 2,356 mg/m3, respectively. The RD50 for JP-8+100 was 1,629 mg/m3. Mice in all JP-8 groups and mice in the higher exposure groups of JP-4 and JP-8+100 exhibited a concentration-dependent delay in recovery of respiratory rate in the 10 min following the exposures. However, all mice appeared normal at clinical observations prior to, during, and immediately after the exposures. For
TABLE 2-5 RD50 Test of JP-8 with Swiss-Webster Mice
| Concentration (mg/m3) | Decrease in Respiratory Rate (%) |
| 681 | 22 |
| 1,090 | 38 |
| 1,837 | 46 |
| 3,565 | 50 |
| 2,876 | Calculated RD50 |
Source: Adapted from Whitman and Hinz 2001.
Abbreviation: RD50, concentration that reduces the respiratory rate by 50%.
these jet fuels, particularly JP-8, the slope of the respiratory rate vs concentration flattened at the highest exposure, that is, the highest concentration, 3,565 mg/m3 elicited a 50% response, which is similar to the RD50. This flattening of the response at high concentrations occurs with hydrocarbon solvents and may reflect coverage of all of the nasal tissue with the vapor and liquid.
It is interesting to note that the RD50 of n-nonane (n-C9), the primary component in the airborne volatile fraction of JP-8 in aircraft maintenance areas could not be measured at concentrations between 1,000 and 1,500 ppm (5,246-7,869 mg/m3). These concentrations failed to reduce the respiratory rate by 50% in CF-1 male mice (Kristiansen and Nielsen 1988). The same was true for decane and undecane. However, reduced respiratory rates were measured for heptane (n-C7). The RD50 for heptane was between 15,600 and 17,400 ppm, and the RD50 for octane was greater than 10,000 ppm. The authors noted that the concentrations tested for the respective chemicals were higher than 50% saturation at room temperature. In another study, an 8-h exposure of male Sprague-Dawley rats to n-nonane at 2,414 ppm failed to cause death (Nilsen et al. 1988), but 8-h exposures at ≥ 3,560 ppm resulted in lethality.
Several studies in mice that delivered JP-8 in the form of an aerosol, identified changes in protein expression in different organs. The toxicologic significance of these changes and their relationship to human health is unknown. Following a 1-h/day, 7-day, nose-only exposure of aerosolized JP-8 to male Swiss-Webster mice at 0, 1,000, or 2,500 mg/m3, molecular biomarkers in terms of protein changes in the lungs were analyzed by gel electrophoresis (Witzmann et al. 1999). Of 796 proteins resolved by electrophoresis, 42 were significantly increased or decreased by exposure to 2,500 mg/m3. The affected proteins were identified as related to protein synthetic machinery; toxic and metabolic stress and detoxification systems; ultrastructural damage; and functional responses to carbon dioxide handling, acid-base homeostasis, and fluid secretion. The study authors described the results as a significant but comparatively moderate effect of JP-8 aerosol on protein expression. In a similar study, protein expression in the cytosol fraction of kidneys was analyzed following exposure of male Swiss-Webster mice to aerosolized JP-8 at 1,000 mg/m3 for 1 h/day for 5 days (Witzmann et al. 2000b). The roles of the quantitatively altered proteins (6% of the
974 proteins resolved by electrophoresis) were identified as the same as those altered in the lung. The study authors again concluded that the changes in protein expression were moderate. (Compared with results in the control group, no change in abundance was greater than 34%.)
Groups of 25 male B6.A.D. mice (genetically identical to C57BL/6 except that they are double congenic for nonresponsiveness to aryl hydrocarbon hydroxylase induction and slow N-acetylation) were exposed nose-only to air or aerosolized JP-8 at 7, 12, 26, 48, or 118 mg/m3 for 1 h/day over 7 days (Robledo et al. 2000). Pulmonary function and respiratory permeability measurements and BALF analysis were performed followed by histologic evaluation of the lungs. At 24 to 30 h after the exposures, there were no changes in dynamic compliance or airway resistance in any of the mice. Compared with the control value, respiratory clearance of 99mtechnetium-labeled-diethylenetriaminepentaacetic acid was approximately doubled in the 7-, 48-, and 118-mg/m3 dose groups, and there was no increase in the 12- and 26-mg/m3 groups. bronchoalveolar fluid (BALF) analysis revealed increases in total protein and lactic dehydrogenase and reductions in N-acetyl-β-D-glucosaminidase and alveolar macrophages at 48 and 119 mg/m3. Light microscopic examination revealed minimal and infrequent deterioration of the alveolar-capillary barrier with sporadic areas of erythrocyte accumulation within alveolar spaces at the two higher exposures. Ultrastructural evaluation of the lungs revealed increases in lamellar bodies and vacuolation of alveolar type II epithelial cells and bronchiolar alterations characterized by perivascular edema, Clara cell vacuolization, and necrosis at the two higher doses. Ciliated epithelial cells appeared mostly unaffected except for changes to intercellular spaces. These changes were reversible following a single exposure.
Groups of 100 male and 100 female C57Bl/6 mice were exposed to vapor concentrations of JP-8 of 0, 500, or 1,000 mg/m3 continuously for 90 days (MacEwen and Vernot 1985; Mattie et al. 1991). Animals were killed immediately after exposure and up to 20 months postexposure. Clinical signs, body weights, and hematologic and clinical chemistry parameters were monitored during exposure. No clinical signs of toxicity or biologically significant changes in hematologic and clinical chemistry parameters were observed. During the 2-week to 20-month recovery period, an increase in mortality of male mice due to necrotizing dermatitis associated with fighting occurred. The incidence (47/100) was the same in both exposure groups. The incidence of fighting-induced dermatitis was also increased in exposed female mice.
JP-5. Groups of 20 male C5BL/6 mice were exposed to JP-5 vapor concentrations of 2,500 or 5,000 mg/m3 for 1 h (MacEwen and Vernot 1985). Generation of the 5,000 mg/m3 concentration resulted in production of an aerosol that coated the fur of the animals. Eye irritation was present at both exposure concentrations, but resolved at 2,500 mg/m3 after termination of exposure. There was no effect on subsequent body-weight gain. One mouse exposed at 5,000 mg/m3 exhibited hind limb paralysis upon removal from exposure. The paralysis continued with some recovery until scheduled animals were killed at 28 days postexposure. Because none of the other mice or rats in this study exhibited hind
limb paralysis, the study was repeated with 40 mice exposed at 5,000 mg/m3. None of the mice in the followup study demonstrated a loss of mobility, indicating the observation made in the first study was not exposure related.
Groups of more than 100 female C57BL/6 mice were exposed continuously to concentrations of JP-5 petroleum- or JP-5 shale-derived vapor at 0, 150, or 750 mg/m3 for 90 days (Gaworski et al. 1984, 1985). In addition to the vapor concentration, an aerosol may have been present in the chamber at the highest concentration. These exposures had no effect on body-weight gain. Non-dose-related (but statistically significantly increased) incidences of hepatocellular fatty infiltration with vacuolization were observed in the mice exposed to JP-5 from either shale or crude oil sources.
3.2.2.4. Rabbits
An increased concentration of substance P (a selective neurokinin receptor agonist) was present in the lungs of rabbits following chronic exposure to JP-5 (Witten et al. 1990). Substance P may exert a protective effect against toxicity.
3.3. Neurotoxicity
In a series of neurobehavioral studies with adult male Sprague-Dawley rats, Bogo et al. (1983, 1984) dosed groups of 6-10 rats orally (via gavage) with either petroleum- or shale-derived JP-5. Doses ranged from 1 to 24 mL/kg. The control groups received an equivalent amount of water by gavage. Rats were observed for general behavior, overnight activity, food and water consumption, and body-weight changes. Because results were similar among the separate substudies, the general results are summarized here. Food and water consumption and body weight were reduced for 2-3 days after dosing. Overnight home-cage activity increased in rats dosed with 3, 5, or 8 mL/kg, but the hyperactivity was not dose related. Activity was greater in a separate study when rats were dosed with 24 mL/kg. Also in a separate study, home-cage daytime activity increased between 2.5 and 6 h after dosing with 3 or 5 mL/kg; the increase was not observed in control rats or rats given 1 mL/kg. Although rats appeared hypersensitive to touch after dosing, two tests of motor function (the accelerod, which is a shock-motivated skilled test of motor function and a test of aggression) failed to reveal any differences between the control and the JP-5 exposed groups. The authors attributed the hyperactivity at 6 h after dosing to gastric irritation and the rat inability to regurgitate.
In the same study (Bogo et al. 1983), male Sprague-Dawley rats inhaled petroleum- or shale-oil derived JP-5 at concentrations below those resulting in anesthesia. Concentrations of 1,125 mg/m3 of petroleum-derived JP-5 (reported as decane) or 1,636 mg/m3 shale-derived JP-5 were administered 6 h/day, 5 days/week, for 35 days. Aside from an increase in drinking-water intake, there were no alterations in behavior or motor function and no changes in neurophysi-
ologic function (measured as electrical potential over the somatosensory area of the brain) or serum liver enzymes (SGPT and SGOT). There were no histopathologic effects on any organs examined, including the liver.
Groups of 32 adult male Sprague-Dawley rats inhaled either JP-5 at 1,200 mg/m3 or JP-8 at 1,000 mg/m3 for 6 h/day, 5 days/week, for 6 consecutive weeks (Rossi et al. 2001). Two groups of 16 rats each exposed to filtered, conditioned air served as controls. Atmospheres were monitored using an infrared spectrophotometer. No aerosol was detected. There were no exposure-related clinical signs, and there were no significant differences in body weight or rate of weight gain between the exposed groups and respective control groups. Sixty-five days after the last exposure, the rats were subjected to 10 neurobehavioral tests consisting of startle response (two tests), appetitive reinforcer approach sensitization, forelimb grip strength, total locomotor activity, tail flick response, social interaction with conspecifics, passive avoidance, forced swim test, and a water maze. Significant differences in response between exposed and control groups were observed in 2 of the 10 tests: the number of seconds for approach to the novel appetitive stimulus was increased over those of the respective control for both exposed groups, but the difference was significant only for the group exposed to JP-8. Times spent within different parts of the test system did not differ among exposed and control groups, indicating similar spontaneous locomotor activity. The appetitive stimulus approach sensitization test is hypothesized to quantify dopamine system sensitization. JP-5-exposed rats exhibited significantly increased forelimb grip strength compared with their control group. The forelimb grip strength test evaluated muscle strength and inhibition of motor response activity. The increase in grip strength was not observed in rats exposed to JP-8.
Following the above neurobehavioral tests (85 days postexposure), all rats were killed and the blood and five different regions of the brain were analyzed for norepinephrine, dopamine, 3,4-hydroxyphenylacetic acid, homovanillic acid, serotonin, and 5-hydroxyindoleacetic acid (Rossi et al. 2001). A single control value was used for comparison. The only significant differences in blood neurotransmitters were in the levels of 5-hydroxyindoleacetic acid, a metabolite of serotonin. Compared with the control values, 5-hydroxyindoleacetic acid was significantly increased in JP-5-exposed rats and significantly reduced in JP-8exposed rats. However, circulating concentrations of serotonin did not differ among control and exposed groups. Compared with control rats, JP-5-exposed rats exhibited increased (p <0.05) dopamine in the hippocampus, level 3,4-dihydroxyphenylacetic acid in the cerebral cortex, and significantly reduced homovanillic acid in the hippocampus. Compared with the control rats, JP-8exposed rats had decreases of 3,4-dihydroxyphenylacetic acid in both the cerebellum and brainstem. The relationship between the increased approach to a novel appetite stimulus and the changes in blood and brain neurotransmitters and their metabolites is not understood. The authors concluded that exposure to JP-5 or JP-8 did not alter basic sensory, motor, or inhibitory functions and did not modulate the capacity of rats to lean and recall tasks of minimal complexity.
In a continuation of the above study, groups of 16 adult male Sprague-Dawley rats were exposed (whole body) to JP-8 vapor at 0 (room air), 500, or 1,000 mg/m3 for 6 h/day, 5 days/week, for 6 weeks (Ritchie et al. 2001c). The 1,000 mg/m3 concentration was the highest vapor concentration that could be generated without formation of an aerosol. Chamber atmospheres were quantified by measuring hexane with infrared spectrometry. Mean body weight of the exposed and control groups did not differ during exposure or during the 7-day postexposure observation period. Clinical observations found no changes in health status or neurobehavioral activity during this time. Following exposure, the rats were rested for 65 days and then tested for ability to acquire and perform operant tasks of increasing difficulty from simple lever pressing to the learning of 3-lever, 4-response operant chains. Rats were slightly food-deprived in order to facilitate the food-reward tests. In the first three measurements (lever acquisition, fixed ratio, and lever spatial reversal), there was no significant difference between the control group and the groups previously exposed at 500 mg/m3 or 1,000 mg/m3 for any measured parameter. In the stimulus reversal and the incremental repeated acquisition task, significant differences between the 500- and 1,000-mg/m3 groups were observed on some days. Although there were more incorrect responses in the 1,000-mg/m3 group compared with the control group, these differences were not statistically significant. The attainment of significance for the high-dose group compared with the low-dose group (but not with the control) was due to the superior performance of the low-dose group on operant tasks of moderate or greater difficulty.
Following the operant training, four rats from each exposure group were killed, and five regional areas of the brain were analyzed for concentrations of norepinephrine, dopamine, 3,4-dihydroxyphenylacetic acid, serotonin, homovanillic acid, and 5-hydroxyindoleacetic acid (Ritchie et al. 2001c). Compared with the control results, there was significantly more dopamine in the cerebral cortex and 3,4-dihydroxyphenlyacetic acid (a metabolite of dopamine) in the brainstem of samples from the 500- and 1,000-mg/m3 groups. Dopamine concentrations were similar in the 500- and 1,000-mg/m3 groups, and 3,4-dihydroxyphenylacetic acid was higher in the 500-mg/m3 group than in the 1,000-mg/m3 group. There were no significant differences compared with the control results for neurotransmitters or neurotransmitter metabolites in any other regions of the brain.
Baldwin et al. (2001) reported on behavioral and memory alterations of rats exposed to aerosolized JP-8. Five F344 Brown Norway rats was exposed (nose-only) to 1,059 mg/m3 for 1 h/day for 25 days followed by exposure at 2,491 mg/m3 for 1 h/day for 3 days (overall mean exposure 1,237 mg/m3). An additional group was administered aerosolized substance P for 15 min following JP-8 exposure; a control group inhaled air only. Potential alterations in behavior were tested with the EPA functional observational battery (FOB) and two versions of the Morris swim task to address spatial and visual discrimination. Substance P was tested to determine if potential deleterious effects of JP-8 exposure on behavior and spatial learning could be ameliorated. During exposure, tran-
sient body-weight loss was observed in the two exposed groups compared with the control; the weight loss resolved by day 19. The two exposed groups showed similar behaviors during the FOB and so were compared as a group to the concurrent control. JP-8 exposed rats, tested on days 4, 9, 14, 19, and 24, showed more rearing activity (day 24), greater arousal (days 9 and 24), and less grooming behavior (day 19) than controls. Although gait and limb strength were unaffected, hind claw contracture was observed in several JP-8 exposed rats over the 28-day period compared with only a single observation in the control group. In the Morris swim test, JP-8 exposed rats swam significantly faster than controls during testing, but there were no significant differences between the exposed and control performance in either the visual discrimination or spatial version of the Morris swim tank task.
3.4. Immunotoxicity
Although emergency exposures are expected to be to spills resulting in primarily vapor exposures, many of the following studies addressed the immunotoxicity of jet fuel aerosols. Aerosols are not generated during aircraft refueling operations. Aerosols will only be developed during aircraft foam removal operations (occupational exposure) or aircraft engine cold starts (occupational exposure). Nevertheless, these studies are summarized here for complete coverage of the database. It should be noted that the lung has an innate immune system that responds to inhaled microbes and particles (University of California 2006). Lung epithelial cells rapidly release antimicrobial polypeptides in response to microbe or particle inhalation, including aerosols. The innate immune response also recruits a secondary adaptive immune response. The immunosupression response of the thymus and spleen are not fully understood.
Groups of 12 male C57BL/6 mice were exposed (nose-only) to aerosolized JP-8 for 1 h (Robledo and Witten 1998) at concentrations of 0, 5.0, 11.7, 27.8, 50.0, and 112.5 mg/m3. At 24 to 30 h after exposure, alveolar permeability, BALF markers, pulmonary function, and histopathology were evaluated. Pulmonary function (dynamic compliance, static compliance, and total resistance; measured with a pneumotachograph) in anesthetized, tracheostomized mice did not differ between control mice and those exposed to jet fuel. Alveolar permeability (as indicated by the clearance of 99mtechnetium-labeled diethylenetriamine pentaacetate) increased in a dose-dependent manner, with increases of 128% and 173% at the 50.0 and 112.5 mg/m3 concentrations, respectively. The following BALF parameters were significantly increased in a concentration-dependent manner beginning with the stated concentration: total protein, 112.5 mg/m3; lactate dehydrogenase, 27.8 mg/m3; N-acetyl-β-D-glucosaminidase, 27.8 mg/m3. Total cell counts were increased significantly and alveolar macrophages were reduced significantly at 112.5 mg/m3. Light microscopy revealed alveolar-septal thickening, monocytic infiltration, and scattered areas of interstitial edema, which became consistent at 112.5 mg/m3. At this concentration, ultra-
structural evaluation revealed an increase in the size and number of surfactant-secreting lamellar bodies of alveolar type II cells and moderate to severe vacuolization of smooth endoplasmic reticulum with normal-appearing mitochondria in nonciliated bronchiolar (Clara) cells. Sporadic edema in terminal bronchioles and areas of goblet cell hyperplasia in small bronchioles were also evident. Adjacent ciliated epithelial cells were not affected.
Male and female C57BL/6 mice exposed under the same protocol as that of Robledo and Witten (1998) above but exposed for 7 days were examined for changes in the immune system 24 h after the last exposure (Harris et al., 1997a). Exposure concentrations of JP-8 were 0, 100, 250, 500, 1,000, or 2,500 mg/m3. Exposure to ≥ 500 mg/m3 resulted in significant reductions in wet weights of the spleen and thymus. Counts of total viable cells from these organs indicated similar effects with cell counts lower in the spleen at exposure concentrations of ≥ 500 mg/m3 and in the thymus at ≥ 100 mg/m3. Changes in the numbers of immune cells in the lymph nodes, bone marrow, and peripheral blood also occurred, with a general reduction at the lowest exposure concentration (with the exception of bone marrow), increases at the next three higher concentrations, and reductions at the highest concentration. Flow cytometric analyses revealed loss of different immune cell subpopulations in different organs. In general, a concentration of 2,500 mg/m3 appeared to be immunotoxic, and mitogenesis assays with splenic cells revealed decreased immune function.
In a followup to the above study, the recovery of mice exposed to JP-8 at 1,000 or 2,500 mg/m3 was studied at 1, 7, 14, 21, and 28 days postexposure (Harris et al. 1997b). Spleen and thymus weights generally recovered by 7 days postexposure (both concentrations) with significantly increased spleen and thymus weights (over the control) for some intervals. Viable splenic immune cell numbers remained significantly lower than control values, but viable thymic immune cells recovered by 21 days postexposure. Immune cell numbers of lymph nodes, bone marrow, and peripheral blood had generally recovered by 7 days postexposure. Immunosuppression as measured by mitogenesis assays indicated recovery at 28 days postexposure for the 1,000-mg/m3 group but not for those exposed at 2,500 mg/m3. In reporting preliminary results of a study with C57BL6 mice, Harris et al. (2000) found that transient immune cell loss followed a single 1-h exposure at 1,000 mg/m3, but partial to full recovery was observed in most organs (spleen, thymus, and bone marrow) at 24 h postexposure.
Harris et al. (2001) reported preliminary results of a study in which timed-pregnant C57BL/6 mice were exposed to JP-8 to 1,000 mg/m3 for 1 h/day either early or late in gestation (days of exposure were not reported in the available abstract). Dams were allowed to deliver and numbers of offspring by sex were tallied. When the offspring were 6-8 weeks old, dams and surviving offspring were killed, and immunologic assays were performed. Fewer pups were born to JP-8-exposed dams as compared with air-exposed controls, and significantly fewer male offspring were born after the dams inhaled jet fuel. At 8 weeks post-in-utero exposure, pups of JP-8-exposed dams had lower immune organ weights,
decreased immune organ cell numbers, and suppressed immune function. This observation was particularly true for male offspring.
Male Swiss-Webster mice were exposed nose-only to aerosolized JP-8+100 at concentrations of 1,000 or 2,500 ppm for 1 h/day for 7 days to study effects on glutathione S-transferase activity in the eye and brain and effects on tissue proteins. Immunohistochemical assays showed increases of this conjugating enzyme in the radial glia of the cerebellum and retina and a reduction in the inner layers of the retina (Kornguth 1998). Electrophoresis of the proteins of the cytosolic fractions of kidney, liver, and lung tissues and serum of the exposed mice showed moderate quantitative changes in the proteins of these tissues (Witzmann et al. 1999, 2000a,b).
Keil et al. (2003) reported that repeated prenatal exposure to JP-8 at very large oral bolus doses (1,000 or 2,000 mg/kg/day on gestation days 6-15) can target the developing murine fetus and result in impaired immune function and altered T4 levels in adulthood. At weaning, these doses had no effect on body or organ weight, splenic and thymic cellularity, splenic lymphocyte subpopulations, or T-cell subpopulations.
In addition to the inhalation and oral studies cited above, dermal application of JP-8 to mice and rats has also produced a systemic immunosuppressive effect (Ullrich 1999; Ullrich and Lyons 2000; Kabbur et al. 2001). Although the steps in the immune response are not fully understood, it appears to be mediated via an increase in cytokines, such as interleukin-10.
3.5. Developmental and Reproductive Effects
A total of 150 time-mated female Crl:CD7 rats were administered JP-8 fuel on days 6-15 of pregnancy by gavage at doses of 0, 500, 1,000, 1,500, or 2,000 mg/kg/day (Cooper and Mattie 1996). Rats were killed on day 28 of pregnancy and fetuses were examined for malformations. The average body weight of dams as well as of male and female fetuses was significantly reduced at the two highest concentrations. The number of pregnant females, number of corpora lutea per female, number of fetuses per female, and post-implantation loss were all within normal limits. There was no significant difference in the number of malformations between the control and any treated group.
Mattie et al. (2000) reported on the reproductive toxicity of JP-8 in both male and female Sprague-Dawley rats treated by gavage. In the first part of the study, young male Sprague Dawley rats, weighing 180-220 g were administered 0, 750, 1,500, or 3,000 mg/kg daily for 70 days. Treatment continued during mating to untreated females (days 70-90) after which the males were killed. General toxicity, fertility (pregnancy rate and gestation duration for females) and sperm parameters of the males were evaluated. Beginning with days 26-42 of treatment, mean body weights of the groups were lower in a dose-dependent manner with males in the 3,000 mg/kg group losing weight over the treatment period. Pregnancy rates were low in all groups with percent of females pregnant
in the 0, 750, 1,500, and 3,000 mg/kg groups of 47, 39, 57, and 53%, respectively. Mean gestation lengths were similar among groups. Epididymal sperm analyses revealed no differences among groups in sperm concentration or motility parameters. Results of microscopic examinations of male rats were reported in Matttie et al. (1995). Male rat-specific α2u-microglobulin nephropathy was observed. Gastritis and a perianal dermatitis were also observed. Although several liver enzymes were increased in the treated groups (aspartate aminotransferase and alanine aminotransferase), the increases were not dose-dependent and liver weights were not increased.
Mattie et al. (2000) also administered JP-8 by gavage to groups of 35 young female Sprague-Dawley rats (180-200 g) at daily doses of 0, 750, or 1,500 mg/kg for 21 weeks. The 21-week period included 90 days of treatment followed by treatment during cohabitation, gestation, delivery, and lactation. Males were not exposed. Females were killed one day after weaning (day 22 of lactation). Litters were standardized to 4 males and 4 females. Pregnancy rate and gestation duration were recorded for the dams. Litter size, number born dead and pup weights (by sex) on various days through postnatal day 90 were recorded. On the day prior to killing, urine was collected for urinalysis; at death, blood was collected from 10 dams/group for hematology and clinical chemistry evaluation, and organs were collected, weighed, and subjected to gross and microscopic examination.
Beginning with week 7, body weights of exposed groups were slightly lower than the control group weight. By week 8 and continuing to week 20, the difference in body weight compared with the control weight was significant in the 1,500-mg/kg/day group. Mean terminal body weights (week 21) were similar among all groups. Pregnancy rates, gestation length, litter size, and percent live pups were similar among control and exposed groups. On postnatal day 1, group mean pup weights of all exposed groups were within 95% of the control pup weight. On postnatal days 4, 14, and 21, there was a trend for decreased pup weight with increasing dose; the difference was statistically significantly lower on those days that correlated with lower maternal body weights. This difference in pup groups was no longer evident by postnatal day 90.
There were no exposure-related clinical signs in dams throughout the second part of the study, and there were no effects on mortality. As noted, body weights of dams were reduced, the lowered weight attaining significance primarily in the 1,500-mg/kg/day group. At study termination, absolute liver weights and liver weights relative to both body and brain weights were significantly increased in the 750- and 1,500-mg/kg/day groups. Kidney weight relative to brain weight was also increased in the two higher dose groups. Changes in urine and hematology and clinical chemistry parameters were observed but were not dose related. Hyperplasia of the stomach and perianal dermatitis were observed microscopically. Incidences were dose-dependent, attaining statistical significance in the 750-mg/kg/day (hyperplasia of the stomach) and 1,500-mg/kg/day groups (anal dermatitis and hyperplasia of the stomach).
Briggs (2001) exposed male Sprague-Dawley rats (whole body) to JP-8 at 0, 250, 500, or 1,000 mg/m3 for 6 h/day, 5 days/week, for 6 weeks to assess the potential for JP-8 to produce reproductive toxicity. No signs of toxicity were observed during the exposure period. After an 87-day recovery period, there were no exposure-related differences in sperm concentration or motility among control and exposure groups. Microscopically, no lesions were observed in the testes. Although biochemical studies revealed some differences in protein expression in the testes (Witzmann et al. 2003), these changes did not interfere with normal sperm maturation or function.
3.6. Genotoxicity
Inhalation of JP-5 failed to increase sister chromatid exchanges or micronuclei in peripheral lymphocytes of beagle dogs. JP-5 was not mutagenic in Salmonella typhimurium standard preincubation tests, with or without metabolic activation (ATSDR 1998).
JP-8 was not mutagenic in Salmonella tester strains TA1535, TA1537, TA1538, TA98, and TA100 either with or without metabolic activation by S9; mouse lymphoma assay; or dominant lethal assays (Brusick and Matheson 1978b; ATSDR 1998). JP-8 was cytotoxic at the higher concentrations tested.
3.7. Chronic Toxicity and Carcinogenicity
No tumors were formed in rats or mice exposed at 500 or 1,000 mg/m3 continuously for 90 days and killed up to 21 months later (Mattie et al. 1991). With the exception of an increased incidence of liver adenomas in female mice exposed to shale-derived JP-5 at 150 or 750 mg/m3 for 90 days and killed 19 or 24 months later, JP-5 was negative for tumors (Gaworski et al. 1985). Tumor data in laboratory animals exposed to hydrocarbon fuels were further evaluated by Bruner (1984). No renal tumors were observed in rats following the 90-day continuous exposure to 750 mg/m3 and observed for a lifetime. Liver adenomas were not increased in female mice exposed to petroleum-derived JP-5 or in male mice or male or female rats exposed to JP-5 from either source.
JP-5 was not carcinogenic to male or female B6C3F1 mice when applied dermally for 2 years at doses of 0, 250, or 500 mg/kg in acetone (NTP 1986). There were increased incidences of chronic dermatitis, judged as mild and moderate in severity in the low- and high-dose groups, respectively, at the site of application. Skin tumors were found in C3H mice exposed dermally with neat Jet A twice a week for 2 years on an intermittent schedule (Freeman et al. 1993) and in C3H mice exposed dermally three times a week (25 mg/dose) for up to 105 weeks (Clark et al. 1988).
Following a review of skin painting studies with a variety of petroleum distillates, NRC (1996; 2003) and Nessel (1999) concluded that there is only tenuous evidence that these fuels pose a carcinogenic hazard after topical appli-
cation, and the induction of tumors at the site of (excessive) application depends on skin irritation and subsequent cell proliferation.
3.8. Summary
Jet fuels in the vapor form exhibit low acute and chronic toxicity in animal studies. Regardless of the source of the fuel or the differences in distillation temperatures, the toxicities appear to be similar in all tested species.
No deaths occurred in rats and mice exposed for 1 h to JP-5 aerosol at a concentration of 5,000 mg/m3 (MacEwen and Vernot 1985), in rats exposed to JP-8 for 4 h at either a vapor concentration of 3,430 mg/m3 or a vapor plus aerosol concentration of 4,440 mg/m3 (Wolfe et al. 1996). Furthermore, no deaths and no effects applicable to humans occurred during a 90-day continuous exposure of rats or mice to vapor of JP-8 at 1,000 mg/m3; there were no effects on the lungs (Mattie et al. 1991).
Reproductive parameters in male rats were not affected, either immediately following oral intubation (Mattie et al. 2000) or following JP-8 inhalation for 6 weeks with a recovery period (Briggs 2001).
Long-term exposures (90 days) to JP-8 and other jet fuels resulted in lower body-weight gain and produced nephropathy specific to male rats. The nephropathy, characterized by hyaline droplet formation and necrosis is exclusive to male rats (Bruner and Pitts 1983). The male rat nephropathy and resulting kidney cancer associated with exposure to jet fuels is not relevant to humans (see Section 4.2. Mechanism of Toxicity). The jet fuels discussed here (JP-5 and JP-8) are not carcinogenic or genotoxic and are not reproductive or developmental toxicants.
Aerosols present along with the vapors of the jet fuels are more toxic than vapors alone as determined by histopathologic effects on the lungs. A 1-h exposure of mice to a concentration of 112.5 mg/m3 resulted in no changes in several pulmonary function parameters, but several reversible biochemical and ultrastructural changes were observed in the lungs (Robledo and Witten 1998). The implication of these changes is unknown.
JP-8 when delivered in the aerosol phase in nose-only exposures induces more severe effects than exposures to the vapor or vapor and aerosol phase formed under most conditions. Under this exposure condition, repeated exposures of rats and mice resulted in pulmonary responses characterized by increased respiratory permeability, peribronchiolar edema, and cellular necrosis at concentrations as low as 48 mg/m3 for 1 h daily (Robledo and Witten 1998, Robledo et al. 2000). The immune system is a target of JP-8 in aerosol form (see Bruckner and Warren 2001). Daily repeated 1 h exposures at concentrations of 1,000 or 2,500 mg/m3 resulted in decreases in immune organ weights and viable immune cell numbers with a decrease in immune system function; these effects were only partially reversible after 1 month (Harris et al. 1997a,b). Aerosolized
JP-8 at a concentration of 4,440 mg/m3 for 4 h caused no gross exposure-related lesions; histopathologic examinations were not performed (Wolfe et al. 1996).
Rodent studies with repeated exposures show JP-8 can induce changes in protein expression in the liver, kidney, lungs, and testes (Witzmann et al. 1999, 2000a,b, 2003) and significant changes in pulmonary (Pfaff et al. 1995; Robledo et al. 2000) or immune system function (Harris et al. 1997a,b) (all from Rossi et al. 2001. The altered protein responses are indicative of possible repair and regeneration and detoxification mechanisms and are present long after exposure has ceased. These health effects or biomarkers of aerosol exposure have not been reported in fuel-exposed humans.
The jet fuels discussed here (JP-5 and JP-8) are not carcinogenic or genotoxic and are not reproductive or developmental toxicants.
4. SPECIAL CONSIDERATIONS
4.1. Metabolism and Disposition
No information was located that specifically addressed the absorption, distribution, or metabolism of JP fuels following oral, dermal, or inhalation exposures. Effects following human and animal exposures lend indirect evidence to the fact that absorption occurs by these routes. Because jet fuel is a mixture of chemicals, the toxicokinetics are complex. Many aliphatic, alicyclic, and aromatic hydrocarbons are hydroxylated, conjugated with sulfate or glucuronic acid, and eliminated in the urine (Cavender 1994a,b,c). See NRC (1996; 2003) for a review of the toxicokinetics of components of jet fuels: benzene, alkylbenzenes (toluene and xylenes), and C9-C13 aliphatic and aromatic hydrocarbons. Most JP-8 components are metabolized or oxidized primarily in the liver by cytochrome P-450/CYP2E1. Pharmacokinetic modeling of n-nonane, based on blood and tissue partition coefficients from a series of inhalation studies with F344 rats at 100, 500, and 1,000 ppm, predicts that human brain concentration during a 4-h exposure at 1.8 ppb would be 0.1 mg/L (Robinson 2000). n-Nonane is a lipophilic compound (log Kow of 5.65) that preferentially concentrates in brain tissue (Zahlsen et al. 1990; 1992). PBPK models of uptake and clearance of individual components, such as n-decane, have been developed, but the authors reported that the model needs refining before predictions of tissue and blood concentrations at low concentrations of decane vapor can be made (Perleberg et al. 2004).
There have been investigations of the pharmacokinetics of combinations of ≥ 3 aromatics and/or long-chain aliphatics (Pedersen et al. 1984; Zahlsen et al. 1990, 1992; Lof et al. 1999). There have also been efforts to develop physiologically based pharmacokinetic (PBPK) models for complex mixtures (Tardif et al. 1997; Haddad et al. 2001). Dennison et al. (2003) developed a chemical lumping approach to analyze the effects of gasoline on the kinetics of individual aromatic components from gas uptake studies. Gasoline contains many of the
same hydrocarbons as JP-8. The model assumes that essentially all of the components of gasoline serve as competitive inhibitors of oxidation of the other components. Campbell and Fisher (2007) developed a PBPK model to assess the effect of JP-8 vapor on individual component kinetics of the selected hydrocarbons m-xylene and ethylbenzene. At concentrations of JP-8 vapor at 390, 1,100, or 2,700 mg/m3, clearance of hydrocarbons was rapid, concentrations of monitored components decreasing in concentration from 30% to 70% by 0.5 h postexposure. The model described the kinetic binary interaction between m-xylene and ethylbenzene and adequately described the impact of the remaining aromatic fraction of the JP-8 exposure on the kinetics of m-xylene and ethylbenzene.
4.2. Mechanism of Toxicity
Aliphatic and alicyclic hydrocarbons cause CNS depression (narcosis) and asphyxia following acute exposures to high concentrations (Cavender 1994a,b, c; Bruckner and Warren 2001). Exposure to high concentrations may cause excitement, loss of equilibrium, stupor, and coma. The effectiveness of the individual components of jet fuels as CNS depressants is related to their volatilization, potency, and blood/air partition coefficients (NRC 1996). Recovery from CNS effects is rapid and complete in the majority of cases.
Because hydrocarbons are lipophilic, they partition into and accumulate in neuronal membranes and myelin. The more lipophilic the hydrocarbon is (that is, the higher its neuronal tissue:blood partition coefficient), the more potent a CNS depressant it is. The mere presence of hydrocarbons has generally been thought to disrupt the ability of the neuron to propagate an action potential and repolarize. Recent research has revealed that hydrocarbons might act by more specific mechanisms and might affect specific neurotransmitters and membrane receptors (that is, by enhancing gamma-aminobutyric acid[A]) receptor function or by activating dopaminergic systems). Hypotheses and pertinent experimental results have been published by a number of researchers, including Mihic et al. (1994), Engelke et al. (1996), Cruz et al. (1998), and Balster (1998).
In rodents, exposure to JP-8 vapor at 500 mg/m3 increases activity, whereas exposure to higher concentrations (1,000 mg/m3) may result in modulation of the capacity to learn or perform difficult tasks. These exposures have also been shown to affect the concentrations of the neurotransmitter dopamine and its metabolite 3,4-dihydroxyphenylacetic acid in different regions of the brain (Ritchie et al. 2001c). In another study, rats inhaling JP-8 at 1,000 mg/m3 exhibited a significant increase in approach to a novel appetitive stimulus and significantly decreased levels of 3,4-dihydroxyphenylacetic acid in both the cerebellum and brainstem regions (Rossi et al. 2001). The relationship of changes in levels of neurotransmitters in the brain to activity level and task performance is unknown.
Many volatile hydrocarbons are of low acute toxicity. Concentrations that cause CNS depression are generally not injurious to the lung. CNS depression in
rats and mice was observed with JP-5 at 5,000 mg/m3 (MacEwen and Vernot 1985) but not in mice inhaling JP-8 at 3,565 mg/m3 for 30 min (Whitman and Hinz 2001). The aromatic hydrocarbons are more toxic than the aliphatic and alicyclic hydrocarbons but, due to their lower boiling point, are present to a much smaller extent in jet fuels. The interactions of the individual components were not predictable with current information. These volatile hydrocarbons can be used as biomarkers of exposure in jet fuel exposed workers. For example, acute increases in benzene and toluene have been measured in the blood, urine, and exhaled breath of chronically exposed workers (Smith et al. 1997; Pleil et al. 2000). Some hydrocarbons are also primary skin irritants.
Long-term exposure to some hydrocarbons results in α2u-globulin nephropathy and associated carcinogenicity specific to male rats (Bruner and Pitts 1983; Swenberg 1993; Bruckner and Warren 2001). The nephropathy is characterized by hyaline droplet formation and necrosis of kidney cells. The toxic effect is attributed to the α2u-microglobulin protein, which is specific to the male rat. The α2u-microglobulin protein is synthesized in the liver of male rats and is readily excreted in the glomerular filtrate (Bruner et al. 1993; Swenberg 1993). The mechanism leading to nephropathy, necrosis, cell proliferation, and neoplasms involves the ability of select hydrocarbons to combine with the protein to form poorly digestible complexes and prevent efficient catabolism of the protein following resorption from the glomerular filtrate. The tubular epithelial cells become engorged with the protein, resulting in metabolic disturbances followed by cell death and exfoliation. Exfoliated necrotic cells form tubular casts that plug the nephron near the corticomedullary junction. The casts become mineralized and may be flushed into the medullary segments where they may remain. α2u-Microglobulin nephropathy is also specific to male rats. The protein is not synthesized in humans (EPA 1991). Therefore, this adverse effect is not relevant to human exposure to jet fuels. The incidence of renal tumors in male rats induced by chemicals causing α2u-globulin nephropathy is low, 0 to 26% (Swenberg 1993). Consistent with the low incidence of neoplasms are the 90-day studies of MacEwen and Vernot (1985) in which findings of nephropathy were reported in male rats exposed to either JP-5 or JP-8, but no kidney tumors were reported at the 24-month killing.
4.3. Structure-Activity Relationships
Many of the hydrocarbons in jet fuels affect the CNS. Experimental data showed that saturated hydrocarbons are absorbed to a lesser extent than unsaturated hydrocarbons in the rat (Dahl et al. 1988) and that aliphatic hydrocarbons are less efficiently absorbed by human blood than aromatic hydrocarbons (Astrand et al. 1975). Compared with aromatic hydrocarbons, aliphatic hydrocarbons are thought to have lower acute narcotic effects, lesser mucous membrane irritation, lower vapor pressure, and lower blood:air partition coefficients (Lof et al. 1999). Furthermore, in rats, uptake of several aliphatic hydrocarbons, as
measured by blood:air partition coefficients, is lower than that of the aromatic hydrocarbons (Perbellini et al. 1985).
4.4. Other Relevant Information
4.4.1. Species Differences
Except for nephropathy in the male rat, no species differences in toxicity were observed among rodents in the several studies with jet fuels. Studies with hydrocarbon vapors, including jet fuels, showed a pattern of nephropathy in several strains of male rats, including Sprague-Dawley, Wistar, and F344 (MacEwen and Vernot 1984, 1985). These lesions consist of renal tubular degeneration along with mineral deposits in the renal papillae (see Section 4.2. Mechanism of Toxicity).
Jet fuel is a complex mixture of hundreds of aliphatic and aromatic hydrocarbons. When exposed to single hydrocarbons, uptake, as measured by blood concentrations and blood:air partition coefficients, is greater in rodents than humans (Benignus et al. 1981; Gargas et al. 1989). Greater chemical uptake is also due to the more rapid respiration rate and greater cardiac output in rodents compared with humans (Witschi and Last 2001; Kale et al. 2002). For the specific chemical, n-hexane, affinity of red blood cells of rats was greater (92% uptake) than that of human blood cells (66% uptake) (Lam et al. 1990).
The extent of CNS depression caused by each chemical is dependent on the concentration of the parent chemical present in the brain (reflected in the blood concentration). Therefore, at a constant concentration, rodents may be more sensitive to the effects of specific hydrocarbons than humans. For lipophilic compounds, elimination may be faster in rodents than in humans.
4.4.2. Susceptible Populations
Although the hydrocarbons that constitute jet fuels are not primary irritants, high concentrations of JP-8 (≥ 681 mg/m3) elicit a classic respiratory depression response (Whitman and Hinz 2001). The threshold for response to irritants is expected to differ by no more than a 3-fold factor in the general population.
Robledo and Witten (1998) exposed C57BL/6 mice deficient in aryl hydrocarbon hydroxylase and N-acetyltransferase enzymes to aerosols of JP-8 ranging from 0 to 112.5 mg/m3 for 1 h. Aryl hydrocarbon hydroxylase is responsible for biotransformation of aromatic hydrocarbons, such as benzo(a)pyrene, which are present in JP-8. Aromatic amines, the substrate for N-acetyltransferases, are not major constituents of JP-8. Exposure conditions and pulmonary function tests were the same as those for congenic mice that were not deficient in these enzymes (Section 3.2.3). Pulmonary responses were similar in both strains of mice. No differences in immune function changes were found in these
two strains of mice or between males and females when exposed to aerosolized JP-8 at concentrations up to 2,500 mg/m3 (Harris et al. 1997a).
Young and Witten (2000) found that aged mice (12-14 months old) exhibited more changes in pulmonary parameters, including lung permeability, dynamic compliance, and pulmonary resistance, following exposure of aerosolized JP-8+100 to 1,000 mg/m3 than young mice (3-4 months old).
No susceptible human populations were identified from monitoring studies. No information on children was located. Children and the elderly may be more or less sensitive to the toxic effects of solvents and vapors, but age-dependent susceptibilities to acute effects usually differ by no more than 2- to 3-fold (Bruckner and Warren 2001). Although humans differ in the rate at which they metabolize chemicals, the susceptibility of the general population to CNS depressants varies by no more than 2- to 3-fold as indicated by the minimum alveolar concentration, the concentration of an anesthetic that produces immobility in 50% of patients (Kennedy and Longnecker 1996; Marshall and Longnecker 1996).
4.4.3. Concentration-Exposure Duration Relationship
For the end points of both sensory irritation and depression of the CNS by solvents, there is generally a concentration threshold. All the jet fuels administered in the phase of vapor or combined vapor and aerosol exhibited low toxicity during acute, repeated, and chronic exposures. Time to steady state for individual components depends on lipophilicity as well as chemical interactions. Once steady state is attained, the CNS effect observed with exposures to high concentrations is most likely a concentration-dependent effect with exposure duration of lesser importance. For n-nonane, inhaled by F344 rats at 100, 500, or 1,000 ppm for 4 h, steady state was approached in the blood within 2 h at the two higher concentrations (Robinson 2000). The blood:air partition coefficient was 5.13.
For many hydrocarbons administered singly, CNS depression occurs at “high” concentrations. For example, n-heptane at 15,513 ppm (63,575 mg/m3) failed to induce anesthesia in mice when administered over a 30-min period (Kristiansen and Nielsen 1988). CNS depression was observed within 20 and 8 min when exposures were at 15,668-21,746 and 24,801 ppm, respectively. The blood:air partition coefficient for n-heptane is 1.9 (Perbellini et al. 1985).
Fifteen young adult males inhaled 1,250 or 2,500 mg/m3 of white spirit (Stoddard Solvent) for 30 min during rest or exercise (Astrand et al. 1975). White spirit (boiling range 150-200°C) contains 83% aliphatic and alicyclic hydrocarbons and 17% aromatic hydrocarbons. Pulmonary absorption of the aliphatics ranged from 46% to 59% and that of the aromatics from 58% to 70%. Blood concentration of n-decane, used as a marker of aliphatic hydrocarbons, increased at the start of each exposure but tended to level off toward the end of the exposure. Aromatics continued to rise during the exposure period. In con-
trast, Stokholm and Cohr (1979) found that the aromatic fraction of white spirit reached steady state in alveolar air of 21 human subjects earlier than the aliphatic fraction. Steady state in alveolar air was reached after 20 min of exposure at rest and after 1 h during work. Exposures were at 204, 600, 1,200, and 2,400 mg/m3.
Several animal studies that addressed mixtures used exposure durations of 1 to several weeks, and blood and brain concentrations of the chemicals were not measured prior to a 1-week exposure. In rats exposed to dearomatized white spirit at 0, 400, or 800 ppm for 3 weeks, blood and brain concentrations of n-nonane, n-decane, and n-undecane did not increase after 1 week; whereas fat concentrations were still increasing (Lof et al. 1999). After 1 week of exposure to white spirit at 400 ppm, blood concentrations of n-nonane, n-decane, and n-undecane were 0.10, 0.70, and 0.16 mg/kg, respectively. Following exposure to white spirit at 800 ppm, blood concentrations were 0.26, 2.09, and 1.06, respectively.
4.4.4. Concurrent Exposure Issues
In past studies with JP-4, exposure to benzene has been an area of concern. The typical benzene content of jet fuels, such as JP-4 and JP-8, is less than 0.5% by weight or volume (Martone 1981; ATSDR 1995). In a study of Air Force personnel engaged in aircraft maintenance, the highest TWA exposure to benzene was 0.034 ppm (Smith et al. 1997). ATSDR (1998) noted that the boiling point range of kerosene and the resulting jet fuels is well above the boiling point of benzene and many polycyclic aromatic hydrocarbons (PAHs); therefore, the benzene content of jet fuels in normally below 0.02%, and the PAH content is virtually zero.
Although emergency exposures are expected to be to spills resulting in vapor exposures, exposure to respirable aerosols during aircraft fueling and maintenance is of concern because several studies have shown that aerosols are more toxic than vapors. Highly visible aerosol emissions have been observed during jet aircraft cold starts, resulting in some crew members working behind the aircraft becoming drenched in fuel (Bruckner and Warren 2001). Toxicity studies that specifically utilized aerosols rather than vapor alone have identified JP-8-induced effects on the lung and immune system (see Table 2-4; Chen et al. 1992; Pfaff et al. 1992; 1993; 1995; Witten et al. 1992; Parton et al. 1993; Witten 1994; Hays et al. 1995; Harris et al. 1997a,b). The mechanism of lung injury appears to involve reduction of substance P, which participates in the maintenance of airway epithelial cell competency in the lung (Pfaff et al. 1996). In mice, JP-8 aerosols decreased immune organ weights and cellularities, altered the number of viable immune cells of several immune tissues, and resulted in the loss of different immune cell subpopulations in immune organs. Some of these changes were present up to 1 month postexposure. Treatment with aerosolized
substance P protected against the JP-8-induced lung and immune system effects (Harris et al. 1997c). Microscopic changes in the lungs and other organs were not observed in mice and rats exposed to JP-8 vapor at 1,000 mg/m3 for 90 days (MacEwen and Vernot 1985; Mattie et al. 1991). The inhalation of JP-8 aerosol may be relevant to military personnel in close proximity to aircraft fueling, cold start, or maintenance operations, but the relevance to the civilian population, that is, community exposure, where vapor is a more likely mode of exposure is not clear.
Inhalation is the primary route of exposure for most populations. Dermal exposure is relevant for military personnel and maintenance workers. Following dermal contact, individual volatile components may evaporate (low-molecular-weight volatile components) or penetrate the skin (less volatile, longer chain [hydrophobic] hydrocarbons) and pass into the blood for distribution throughout the body (NRC 2003). The NRC concluded that the exposure via the dermal route can be substantial for military personnel.
5. DATA ANALYSIS FOR AEGL-1
The proposed AEGLs were based on jet fuels as mixtures of hydrocarbons rather than individual components because both occupational and experimental animal exposures have been to the partial to total vaporization products of the fuels. Although vapor compositions may differ in different situations and with different fuels, the large database encompassing many jet fuels and the chronic nature of the exposures allows derivation of short-term values with considerable confidence.
Although early toxicity studies with jet fuel vapors found few toxic effects, even following chronic exposures, aerosols that are more toxic may be formed under certain conditions. However, emergency exposures are expected to be in the form of vapor exposures that result from spills, whereas aerosols are relevant only to occupational exposures during aircraft-foam removal operations or aircraft cold starts. Studies that addressed the toxicity of jet fuel only in the aerosolized form were not used to derive AEGL values (Martin et al. 2010; Tremblay et al. 2010).
5.1. Summary of Human Data Relevant to AEGL-1
There are no useful acute data involving human exposures. Monitoring data compose the primary exposure data for JP-8. Specific symptoms or complaints could not be related to exposure concentrations. Furthermore, the chronic nature of occupational exposures must be taken into consideration. Highest exposures occurred during fuel tank entry during which time the workers wore respirators.
5.2. Summary of Animal Data Relevant to AEGL-1
Although jet fuels are not primary irritants, they elicit a characteristic irritant response at high concentrations. Whitman and Hinz (2001) reported results of the standard RD50 test using several jet fuels. Exposure to JP-8 vapor and aerosol at 681, 1,090 1,837, and 3,565 mg/m3 for 30 min decreased the respiratory rate of male Swiss-Webster mice by 22%, 38%, 46%, and 50%, respectively. The RD50 was 2,876 mg/m3. The respiratory rate decrease of 22% at 681 mg/m3 is considered a slight to moderate response.
Repeated exposures (6 weeks to 90 days) of rats and mice to JP-8 vapor at a concentration of 1,000 mg/m3 failed to induce clinical signs (MacEwen and Vernot 1985; Mattie et al. 1991; Briggs 2001; Rossi et al. 2001) or effects on the male reproductive system (Briggs 2001), but this concentration may be the threshold for neurobehavioral changes (Rossi et al. 2001).
5.3. Derivation of AEGL-1
The AEGL-1 is based on the sensory irritation study of Whitman and Hinz (2001), specifically the RD50 for JP-8 of 2,876 mg/m3. This is a robust study based on aerosol and vapor atmospheres in accordance with the ASTM E981-84 method. According to Alarie (1981), reducing the RD50 by 10-fold results in an exposure concentration of the JP-8 mixture that produces some sensory irritation to humans that is tolerable for hours to days. This effect is consistent with an AEGL-1 effect. The resulting value is 290 mg/m3. Because primary irritation is a concentration effect independent of time, the 290 mg/m3 value was applied to all AEGL-1 exposure durations (Table 2-6).
The primary volatile components of JP-8 present in the vapor phase, the n-alkane solvents in the C9 to C12 range, are not primary irritants. As indicated in Table 2-4, there were no other adverse clinical effects in animal studies with repeated exposure to JP-8 vapor at 1,000 mg/m3 (MacEwen and Vernot 1985; Mattie et al. 1991; Briggs 2001; Rossi et al. 2001). Rossi et al. (2001) reported some changes in brain neurotransmitter activities. Applying an interspecies uncertainty factor of 1 (chosen because the uptake of these chemicals is higher in rodents than in humans [Gargas et al. 1989]) and an intraspecies uncertainty factor of 3 (chosen to account for potential differences in human susceptibility to sensory irritation) results in a value of 330 mg/m3. This value supports the value of 290 mg/m3 determined using the RD50 study of Whitman and Hinz (2001). Appendix B contains a category plot of animal toxicity data and AEGL values.
TABLE 2-6 AEGL-1 Values for JP-5 and JP-8
| 0 min | 30 min | 1 h | 4 h1 | 8 h |
| 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 |
6. DATA ANALYSIS FOR AEGL-2
6.1. Summary of Human Data Relevant to AEGL-2
The available monitoring data indicate that exposures to jet fuels at threshold concentrations for CNS depression have occurred, but actual concentrations are unavailable. At this time, aircraft maintenance workers wear respirators when entering fuel tanks where exposures are potentially quite high. Thus, no information on adverse health effects could be derived from these studies. Past exposures to concentrations as high as 3,000 mg/m3 were to the more volatile JP-4 or an equivalent fuel (Knave et al. 1978; Martone 1981). Such symptoms as dizziness and fatigue may have been associated with these concentrations.
6.2. Summary of Animal Data Relevant to AEGL-2
A 30-min exposure to JP-8 vapor and aerosol at 3,565 mg/m3 failed to induce obvious clinical signs in mice but decreased the respiratory rate by 50% (Whitman and Hinz 2001). Four-hour exposures of rats to JP-8 vapor at 3,430 mg/m3 or aerosol and vapor at 4,440 mg/m3 induced eye and upper respiratory irritation (Wolfe et al. 1996). Conversely, as noted for the AEGL-1, repeated exposures to of JP-8 vapor at 1,000 mg/m3 generally failed to elicit adverse effects in animal studies. Finally, exposure of rats and mice to JP-5, considered a subset of JP-8, at 5,000 mg/m3 for 1 h resulted in eye irritation and signs of CNS depression (MacEwen and Vernot 1985; Mattie et al. 1991); these effects were not observed during repeated exposures to JP-5 at 1,200 mg/m3 (Rossi et al. 2001) or during repeated exposures to JP-5 at 1,636 mg/m3 (Bogo et al. 1983, 1984).
6.3. Derivation of AEGL-2
The AEGL-2 is based on several animal studies that indicate that exposure at 1,100 mg/m3 would not elicit adverse health effects or CNS depression but may be the threshold for such effects. As noted, repeated exposures of rats and mice to JP-5 or JP-8 in the vapor phase at 1,000 mg/m3 or to a single exposure of JP-5 vapor and aerosol at 5,000 mg/m3 is without adverse effects other than sensory irritation and some CNS depression at 5,000 mg/m3.
The studies using repeated exposures to 1,000 mg/m3 (MacEwen and Vernot 1985; Mattie et al. 1991; Briggs 2001; Rossi et al. 2001) and the shorter term studies (30 min to 4 h) with exposures at 3,430-5,000 mg/m3 (MacEwen and Vernot 1985; Mattie et al. 1991; Wolfe et al. 1996; Whitman and Hinz 2001) were used as the basis for the AEGL-2. No uncertainty factors were applied to the 1,000 mg/m3 concentration because there were no effects and the exposures were repeated for up to 90 days. The higher concentrations of JP-8, 3,430 and
4,440 mg/m3, and of JP-5, 5,000 mg/m3, were divided by an interspecies factor of 1 (there were no species differences in susceptibility in numerous studies) and by an intraspecies uncertainty factor of 3 to protect potentially sensitive adults. Doses of volatile organic hydrocarbons (VOCs) absorbed systemically are considerably greater in mice and rats than in humans subjected to equivalent inhalation exposures. This response is attributable to rodent’s relatively high respiratory rates, cardiac outputs, and blood:air partition coefficients. Therefore, no-effect levels for CNS depression in rodents are quite protective for humans. An intraspecies uncertainty factor of 3 is considered adequate because the thresholds for both sensory irritation and CNS depression to solvents do not generally differ by more than 3-fold (Bruckner and Warren 2001). The lower value, 1,100 mg/mg3, in the resulting range of values, 1,100-1,700 mg/m3, is approximately the same concentration as that in the no-adverse-effect repeated exposure studies.
No information was available on time to steady-state blood levels for many of the components of jet fuel. Therefore, no information was available for time-scaling. CNS depression is a concentration-related effect. Because the exposure duration in the key study was for 4 h, the 1,100-mg/m3 value was used for the 4-h and shorter time periods. Because the exposure of rats and mice at 1,000 mg/m3 was continuous (24 h/day) for up to 90 days (Mattie et al. 1991), the 1,100-mg/m3 value can also be used for the longest AEGL exposure duration of 8 h (Table 2-7). The fact that the exposures in most of these studies, especially at the higher concentrations, were to both the vapor and aerosol supports AEGL-2 values. A category graph of AEGL values in relation to toxicity data is in Appendix B.
7. DATA ANALYSIS FOR AEGL-3
7.1. Summary of Human Data Relevant to AEGL-3
No human data relevant to calculation of AEGL-3 values were located. In humans and animals, the primary effect associated with inhalation exposure to aliphatic and aromatic hydrocarbons is CNS depression. Due to the low vapor pressure of JP-8 (1.8 mmHg at 28°C), it is unlikely that atmospheric concentrations would reach lethal concentrations during community exposures. The saturated vapor concentration of JP-8 under “ambient conditions” is approximately 700 mg/m3 (F. Whitman, ExxonMobil Biomedical Science, Inc., personal commun., Oct.31, 2001). It is possible that lethal concentrations of JP-8 cannot be attained or sustained.
TABLE 2-7 AEGL-2 Values for JP-5 and JP-8
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 |
7.2. Summary of Animal Data Relevant to AEGL-3
None of the acute studies with jet fuel vapor resulted in lethality. The only reported effects of 4-h exposures to JP-8 at a vapor concentration of 3,430 mg/m3 or JP-8 at a vapor and aerosol concentration of 4,440 mg/m3 were eye and upper respiratory irritation (Wolfe et al. 1996), symptoms consistent with the definition of the AEGL-2. Those values were the highest vapor and vapor and aerosol concentrations, respectively, attained under laboratory exposure conditions.
7.3. Derivation of AEGL-3
In the study reported by Wolfe et al. (1996), the highest vapor concentration of JP-8 that could be attained was 3,430 mg/m3. The highest vapor and aerosol concentration that could be attained was 4,440 mg/m3. It is not apparent that concentrations high enough to cause death can be attained. A concentration of 500 mg/m3 is assumed to be the upper bound for a stable cloud of inhalable dust (and aerosols) (Craig and Lux 1998). On the basis of the likelihood that lethal concentrations of JP-8 cannot be attained and sustained under ambient conditions, an AEGL-3 was not determined.
8. SUMMARY OF AEGLs
8.1. AEGL Values and Toxicity End Points
The AEGL values for various exposure durations are listed in Table 2-8. Derivation summaries are in Appendix C.
TABLE 2-8 Summary of AEGL Values
| Classification | Exposure Duration | ||||
| 10 min | 30 min | 1 h | 4 h | 8 h | |
| AEGL-1 (Nondisabling) | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 |
| AEGL-2 (Disabling) | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 |
| AEGL-3 (Lethal) | Not determineda | Not determined | Not determined | Not determined | Not determined |
aThere are no data on lethal concentrations.
8.2. Comparison with Other Standards and Guidelines
Standards and guidance levels for workplace and community exposures are listed in Table 2-9. The U.S. Navy’s 8-h TWA-PEL and 15-min STEL for shipboard exposures of Navy personnel are 200 and 1,000 mg/m3. The previous PEL and STEL of 350 and 1,800 mg/m3 (NAVOSH 1992, NEHC 1993), respectively, were based on the Navy’s review of manufacturers’ technical documentation and the NIOSH recommendations for maximum exposure to refined petroleum solvents. The values are for exposure to JP-5 and JP-8. The NRC (1996; Bakshi and Henderson 1998) reviewed the U.S. Navy’s standards and recommended that the NAVOSH STEL be lowered from 1,800 mg/m3 to 1,000 mg/m3. The U.S. Air Force’s values for JP-8 for the respective exposure durations are the same as the U.S. Navy’s. The U.S. Air Force requested that the NRC review the available toxicologic, epidemiologic, exposure, and other relevant data on JP-8; independently re-evaluate the scientific basis of the PEL of 350 mg/m3; identify data gaps; and make recommendations for future research relevant to deriving the PEL (NRC 2003). The NRC Subcommittee on Jet-Propulsion Fuel 8 concluded that the interim PEL of 350 mg/m3 might be too high to be protective of human health but did not propose a specific PEL for JP-8.
The military TWAs are for healthy adults. The 8-h 200-mg/m3 value is slightly lower than the AEGL-1, which is for the general population but is for acute exposures. Most agencies have not derived guidelines specific to jet fuels, but instead use the more general category of petroleum distillates. The Swedish TWA occupational exposure limit of 350 mg/m3 for an 8-h day (and 500 mg/m3 for a TWA over 15 min) is for aviation fuels.
TABLE 2-9 Extant Standards and Guidelines for Jet Fuels, Gasoline, and Kerosene
| Exposure Durations for JP-5 and JP-8 | |||||
| Guideline | 10 min | 30 min | 1 h | 4 h | 8 h |
| AEGL-1 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 |
| AEGL-2 | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 |
| AEGL-3 | Not determined | Not determined | Not determined | Not determined | Not determined |
| PEL-TWA (NAVOSH)a | 200 mg/m3 | ||||
| PEL-TWA (AFOSH)b | 200 mg/m3 | ||||
| STEL (NAVOSH)c | 1,000 mg/m3 | ||||
| STEL (AFOSH)d | 1,000 mg/m3 | ||||
| Exposure Durations for JP-5 and JP-8 | |||||
| Guideline | 10 min | 30 min | 1 h | 4 h | 8 h |
| Exposure Durations for JP-5 and JP-8 | |||||
| Short Term | 8 h | ||||
| IDLH (NIOSH)e | 1,100 ppm (4,500 mg/m3) (petroleum distillates [naphtha]) | ||||
| TLV-TWA (ACGIH)f | 200 mg/m3 (kerosene/jet fuels) | ||||
| PEL-TWA (OSHA)g | Naphthah (coal tar) – 100 ppm (400 mg/m3) | ||||
| REL-TWA (NIOSH)i | Kerosene – 100 mg/m3 Petroleum distillates (naphtha) – 350 mg/m3 | ||||
| REL-Ceiling (NIOSH)j | Petroleum distillates (naphtha) – 1,800 mg/m3 | ||||
| TLV-STEL (ACGIH)k | None established | ||||
| PEL-STEL (OSHA)l | Petroleum distillates (naphtha) (rubber solvent) – 500 ppm (2,000 mg/m3) | ||||
aPEL-TWA (permissible exposure limit-time-weighted average, Navy Occupational Safety and Health [NAVOSH] Standard). (NAVOSH 1992).
bPEL-TWA (permissible exposure limit-time-weighted average, Air Force Occupational Safety and Health [AFOSH] Standard) (U.S. Air Force 2005).
cSTEL (short-term exposure limit, NAVOSH Standard) (NAVOSH 1992).
dSTEL (short-term exposure limit, AFOSH Standard) (U.S. Air Force 2005).
eIDLH (immediately dangerous to life or health concentrations, National Institute for Occupational Safety and Health [NIOSH]) (NIOSH 1996) represents the maximum concentration from which one could escape within 30 min without any escape-impairing symptoms or any irreversible health effects. The IDLH for petroleum distillates (naphtha) is based strictly on safety considerations, being 10% of the lower explosive limit of 1.1%.
fTLV-TWA (Threshold Limit Value-time-weighted average, American Conference of Governmental Industrial Hygienists [ACGIH]) (ACGIH 2009) is the TWA concentration for a normal 8-h workday and a 40-h workweek, to which nearly all workers may be repeatedly exposed, day after day, without adverse effect.
gPEL-TWA (permissible exposure limit–time-weighted average, Occupational Safety and Health Administration [OSHA]) (29 CFR 1910.1000 [2005]) is analogous to the ACGIH TLV-TWA, but is for exposures of no more than 10 h/day, 40 h/week.
hNaphtha is the low-boiling fraction of petroleum and is composed chiefly of pentanes and hexanes.
iREL-TWA (recommended exposure limit-time-weighted-average, NIOSH) (NIOSH 2005) is analogous to the ACGIH TLV-TWA.
jREL-Ceiling (recommended exposure limit-ceiling, NIOSH) (NIOSH 1996) is the concentration of the chemical in air that should not be exceeded.
kTLV-STEL (Threshold Limit Value-short-term exposure limit, ACGIH) (ACGIH 2009) is defined as a 15-min TWA exposure that should not be exceeded at any time during the workday even if the 8-h TWA is within the TLV-TWA. Exposures above the TLV-TWA
up to the STEL should not be longer than 15 min and should not occur more than 4 times per day. There should be at least 60 min between successive exposures in this range.
lPEL-STEL (permissible exposure limit–short-term exposure limit, OSHA) (29 CFR 1910.1000 [2005]) is analogous to the ACGIH TLV-STEL.
The NIOSH IDLH is defined comparable to an AEGL-2. The IDLH for petroleum distillates (naphtha) was originally 4,000 ppm based on a study with gasoline (Drinker et al. 1943). In the Drinker (1943) study, subjects inhaling 0.26% (9,150 mg/m3) for 1 h reported mild exhilaration and muscular incoordination; at 1.1% (38,000 mg/m3), the subjects appeared intoxicated, most within 5 min. The present IDLH value of 1,100 ppm is based strictly on safety considerations, being 10% of the lower explosive limit of 1.1% (NIOSH 1996).
Because benzene, a component of JP-8, is a known human carcinogen (EPA 2003), the benzene concentration in JP-8 at the AEGL-2 value was compared with standards and guidelines. The benzene content of neat fuel is <0.005% by volume (Carlton and Smith 2000). Ratios of benzene to total JP-8 concentrations in instantaneous samples taken during aircraft fuel tank maintenance monitoring studies ranged from 0.0004 to 0.001, that is, up to 1 mg/m3 of benzene in 1,000 mg/m3 of JP-8 vapor (Carlton and Smith 2000). Therefore, under ambient conditions of evaporation of JP-8, the atmospheric concentration of benzene in 1,100 mg/mg3 of JP-8 vapor would be 1.1 mg/m3 (0.34 ppm). This value is above the NIOSH REL of 0.3 mg/m3 for benzene, but below the OSHA PEL of 3.2 mg/m3. It is below the NRC 1-h EEGL of 160 mg/m3, the NIOSH IDLH of 1500 mg/m3, and the 8-h AEGL-2 for benzene of 200 ppm.
8.3. Data Adequacy and Research Needs
Although data on human exposures to JP-5 and JP-8 were located, most of the concentrations were too low to be of value in developing AEGLs. Exposure to higher concentration during aircraft fuel tank entry occurred only when personnel were wearing respirators, thus health effects could not be derived from these studies. Animal studies used several species (rat, mouse, and dog) and addressed sensory irritation, neurotoxicity, and male fertility, as well as other systemic effects. Studies with aerosols addressed direct lung effects and effects on the immune system. The data were sufficient for deriving AEGL-1 and AEGL-2 values. Animal studies with repeated exposures support the safety of values derived for acute exposure durations.
9. REFERENCES
ACGIH (American Conference of Governmental Industrial Hygienists). 2009. TLVs and BEIs: Based on Documentation of the Threshold Limit Values for Chemical Sub-
stances and Physical Agents and Biological Exposure Indices. Kerosene/Jet Fuels. American Conference of Governmental Industrial Hygienists, Cincinnati, OH.
AFOSH (U.S. Air Force Occupational Safety and Health). 2005. Standard 48-8. Controlling Exposures to Hazardous Substances. U.S. Air Force.
Alarie, Y. 1981. Dose-response analysis in animal studies: Prediction of human responses. Environ. Health Perspect. 42:9-13.
Armbrust Aviation Group. 1998. World Jet Fuel Almanac. Palm Beach Gardens, FL: The Armbrust Group.
Astrand, I., A. Kilbom, and P. Ovrum. 1975. Exposure to white spirit. I. Concentration in alveolar air and blood during rest and exercise. Scand. J. Work Environ. Health 1(1):15-30.
ATSDR (Agency for Toxic Substances and Disease Registry). 1995. Toxicological Profile for Jet Fuels JP-4 and JP-7. U.S. Department of Health and Human Services, Public Health Service, Agency for Toxic Substances and Disease Registry, Atlanta, GA. June 1995 [online]. Available: http://www.atsdr.cdc.gov/ToxProfiles/tp76.pdf [accessed Oct. 7, 2010].
ATSDR (Agency for Toxic Substances and Disease Registry). 1998. Toxicological Profile for Jet Fuels JP-5 and JP-8. U.S. Department of Health and Human Services, Public Health Service, Agency for Toxic Substances and Disease Registry, Atlanta, GA. August 1998 [online]. Available: http://www.atsdr.cdc.gov/ToxProfiles/tp121.pdf [accessed Oct. 7, 2010].
Baker, W., D. Dodd, J.N. McDougal, and T.E. Miller. 1999. Repeated Dose Skin Irritation Study on Jet Fuels - Definitive Study. AFRL-HE-WP-TR-1999-0022. Air Force Research Laboratory, Wright-Patterson AFB, OH.
Bakshi, K.S., and R.F. Henderson. 1998. Permissible exposure levels for selected military fuel vapors. Inhal. Toxicol. 10 (10):955-961.
Baldwin, C.M., F.P. Houston, M.N. Podgornik, R.S. Young, C.A. Barnes, and M.L. Witten. 2001. Effects of aerosol-vapor JP-8 jet fuel on the functional observational battery, and learning and memory in the rat. Arch. Environ. Health 56(3):216-226.
Balster, R.L. 1998. Neural basis of inhalant abuse. Drug Alcohol Depend. 51(1-2):207-214.
Benignus, V.A., K.E. Miller, C.N. Barton, and J.A. Bittikofer. 1981. Toluene levels in blood and brain of rats during and after respiratory exposure. Toxicol. Appl. Pharmacol. 61(3):326-334.
Bogo, V., R.W. Young, T.A. Hill, C.L. Feser, and J. Nold. 1983. The Toxicity of Petroleum and Shale JP5. AD-A142 670. Armed Forces Radiobiology Research Institute, Bethesda, MD.
Bogo, V., R.W. Young, T.A. Hill, R.M. Cartledge, J. Nold, and G.A. Parker. 1984. Neurobehavioral toxicology of petroleum-and shale-derived jet propulsion fuel No. 5 (JP5). Pp. 17-31 in Applied Toxicology of Petroleum Hydrocarbons, H.N. MacFarland, C.E. Holdsworth, J.A. MacGregor, R.W. Call and M.L. Kane, eds. Advances in Modern Environmental Toxicology Vol. 6. Princeton, NJ: Princeton Scientific Publishers.
Briggs, G.B. 2001. Evaluation of Military Fuel Potential to Produce Male Reproductive Toxicity. Presented at the International Conference on the Environmental Health and Safety of Jet Fuel, August 8-11, 2001, San Antonio, TX.
Bruckner, J.V., and D.A. Warren. 2001. Toxic effects of solvents and vapors. Pp. 869-916 in Casarett and Doull’s Toxicology: The Basic Science of Poisons, 6th Ed., C.D. Klaassen, ed. New York: McGraw-Hill.
Bruner, R.H. 1984. Pathologic findings in laboratory animals exposed to hydrocarbon fuels of military interest. Pp. 133-140 in Renal Effects of Petroleum Hydrocarbons, M.A. Mehlman, C.P. Hemstreet, J.J. Thorpe, and N.K. Weaver, eds. Advances in Modern Environmental Toxicology Vol. VII. Princeton, NJ: Princeton Scientific Publishers.
Bruner, R.H., and L.L. Pitts. 1983. Nephrotoxicity of hydrocarbon propellants to male Fischer 344 rats. Pp. 337-349 in Proceedings of the 13th Conference on Environmental Toxicology, November 16-18, 1982, Dayton, OH. AFAMRL-TR-82-101. ADA134150. Aerospace Medical Research Laboratory, Wright-Patterson AFB, OH. August 1983.
Bruner, R.H., E.R. Kinkead, T.P. O’Neill, C.D. Flemming, D.R. Mattie, C.A. Russell, and H.G. Wall. 1993. The toxicologic and oncogenic potential of JP-4 jet fuel vapors in rats and mice: 12-month intermittent inhalation exposures. Fundam. Appl. Toxicol. 20(1):97-110.
Brusick, D.J., and D.W. Matheson. 1978a. Mutagen and Oncogen Study on JP-4. ADA064952. AMRL-TR-78-24. Aerospace Medical Research Laboratory, Wright-Patterson Air Force Base, OH. September 1978.
Brusick, D.J., and D.W. Matheson. 1978b. Mutagen and Oncogen Study on JP-8. ADA064948. AMRL-TR-78-20. Aerospace Medical Research Laboratory, Wright-Patterson Air Force Base, OH. September 1978.
Campbell, J.L., and J.W. Fisher. 2007. A PBPK modeling assessment of the competitive metabolic interactions of JP-8 vapor with two constituents, m-xylene and ethylbenzene. Inhal. Toxicol. 19(13):265-273.
Carlton, G.N., and L.B. Smith. 2000. Exposures to jet fuel and benzene during aircraft fuel tank repair in the U.S. Air Force. Appl. Occup. Environ. Hyg. 15(6):485-491.
Carpenter, C.P., D.L. Geary, Jr., R.C. Myers, D.J. Nachreiner, L.J. Sullivan, and J.M. King. 1976. Petroleum hydrocarbon toxicity studies. XI. Animal and human response to vapors of deodorized kerosene. Toxicol. Appl. Pharmacol. 36(3):443-456.
Cavender, F. 1994a. Aliphatic hydrocarbons. Pp. 1221-1266 in Patty’s Industrial Hygiene and Toxicology, Vol. IIB, 4th Ed., G.D. Clayton, and F.E. Clayton, eds. New York: John Wiley & Sons.
Cavender, F. 1994b. Alicyclic hydrocarbons. Pp. 1267-1299 in Patty’s Industrial Hygiene and Toxicology, Vol. IIB, 4th Ed., G.D. Clayton, and F.E. Clayton, eds. New York: John Wiley & Sons.
Cavender, F. 1994c. Aromatic hydrocarbons. Pp. 1301-1442 in Patty’s Industrial Hygiene and Toxicology, Vol. IIB, 4th Ed., G.D. Clayton, and F.E. Clayton, eds. New York: John Wiley & Sons.
Chen, H., M.L. Witten, J.K. Pfaff, R.C. Lantz, and D. Carter. 1992. JP-8 fuel exposure increases alveolar epithelial permeability in rats [abstract]. FASEB J. 6(4):A1064.
Chevron Phillips. 2008. JP-5 Aviation Turbine Fuel. Material Safety Data Sheet. Chevron Phillips Chemical Company LP, The Woodlands, TX [online]. Available: http://www.cpchem.com/bl/specchem/en-s/Pages/JP5MILT5624.aspx [accessed Oct.7, 2010]
Chevron Phillips. 2009a. JP-8 Aviation Turbine Fuel (Mil-T-83133). Material Safety Data Sheet. Chevron Phillips Chemical Company LP, The Woodlands, TX [online]. Available: http://www.cpchem.com/bl/specchem/en-us/Pages/JP8MILT83133.aspx [accessed Oct. 7, 2010].
Chevron Phillips. 2009b. Specialty Chemicals: JP-5 (MIL-T-5624). Product Sheets. Chevron Phillips Chemical Company LP, The Woodlands, TX [online]. Available: http://www.cpchem.com/bl/specchem/en-s/Pages/JP5MILT5624.aspx [accessed Oct. 8, 2010].
Chevron Phillips. 2010. Specialty Chemicals: JP-8 (MIL-T-83133). Product Sheets, Chevron Phillips Chemical Company LP, The Woodlands, TX [online]. Available: http://www.cpchem.com/bl/specchem/en-us/Pages/JP8MILT83133.aspx [accessed Oct. 7, 2010].
Clark, C.R., M.K. Walter, P.W. Ferguson, and M.A. Katchen. 1988. Comparative dermal carcinogenesis of shale and petroleum-derived distillates. Toxicol. Ind. Health 4(1):11-22.
Clark, C.R., P.W. Ferguson, M.A. Katchen, M.W. Dennis, and D.K. Craig. 1989. Comparative acute toxicity of shale and petroleum derived distillates. Toxicol. Ind. Health 5(6):1005-1016.
Cooper, J.R., and D.R. Mattie. 1996. Developmental toxicity of JP-8 jet fuel in the rat. J. Appl. Toxicol. 16(3):197-200.
Cowan, M.J. and L.J. Jenkins, Jr. 1981. Navy toxicity study of shale and petroleum JP-5 aviation fuel and diesel fuel marine. Pp. 129-139 in Health Effects Investigation of Oil Shale Development, W.H. Griest, M.R. Guerin, and D.l. Coffin, eds. Ann Arbor, MI: Ann Arbor Science Publishers.
Cowan, M.J., L.J. Jenkins, and J. Lawrence. 1981. The toxicity of grade JP-5 aviation turbine fuel: A comparison between petroleum and shale-derived fuels. Pp. B2/1-B2/7 in Toxic Hazards in Aviation: Conference Proceedings. Neuilly sur Seine, France: Advisory Group for Aerospace Research and Development).
Craig, D.K., and C.R. Lux. 1998. Methodology for Deriving Temporary Emergency Exposure Limits (TEELs) (U). WSRC-TR-98-00080. Westinghouse Savannah River Company, Aiken, SC [online]. Available: http://orise.orau.gov/emi/scapa/files/Method_for_deriving_TEELs.pdf [accessed Oct. 8, 2010].
Cruz, S.L., T. Mirshahi, B. Thomas, R.L. Balster, and J.J. Woodward. 1998. Effects of the abused solvent toluene on recombinant N-methyl-D-aspartate and non-N-methyl-D-aspartate receptors expressed in Xenopus oocytes. J. Pharmacol. Exp. Therap. 286(1):334-340.
Dahl, A.R., E.G. Damon, J.L. Mauderly, S.J. Rothenberg, F.A. Seiler, and R.O. McClellan. 1988. Uptake of 19 hydrocarbon vapors inhaled by F344 rats. Fundam. Appl. Toxicol.10(2):262-269.
Davies, N.E. 1964. Jet fuel intoxication. Aerospace Med. 35:481-482.
Dennison, J.E., M.E. Andersen, and R.S. Yang. 2003. Characterization of the pharmacokinetics of gasoline using PBPK modeling with a complex mixtures chemical lumping approach. Inhal. Toxicol. 15(10):961-986.
Dietzel, K.D., J.L. Campbell, M.G. Bartlett, M.L. Witten, and J.W. Fisher. 2005. Validation of a gas chromatography/mass spectrometry method for the quantification of aerosolized Jet Propellant 8. J. Chromogr. A 1093(1-2):11-20.
Døssing, M., S. Loft, and E. Schroeder. 1985. Jet fuel and liver function. Scand. J. Work Environ. Health 11(6):433-437.
Drinker, P., C.P. Yaglou, and M.F. Warren. 1943. The threshold toxicity of gasoline vapor. J. Ind. Hyg. Toxicol. 25(6):225-232.
Engelke, M., H. Tahti, and L. Vaalavirta. 1996. Perturbation of artificial and biological membranes by organic compounds of aliphatic, alicyclic and aromatic structure. Toxicol. In Vitro 10(2):111-115.
EPA (U.S. Environmental Protection Agency). 1991. Alpha-2u-Globulin: Association with Chemically-Induced Renal Toxicity and Neoplasia in the Male Rat. EPA/625/3-91/019F. Risk Assessment Forum, U.S. Environmental Protection Agency, Washington, DC.
EPA (U.S. Environmental Protection Agency). 2003. Benzene (CASRN 71-43-2). Integrated Risk Information System, U.S. Environmental Protection Agency [online]. Available: http://www.epa.gov/iris/subst/0276.htm [accessed Oct. 8, 2010].
Feldmann, M.L., R.E. Wolfe, E.R. Kinkead, H.F. Leahy, W.W. Jederberg, and D.R. Mattie. 1997. Acute toxicity evaluation of JP-8 jet fuel containing additives [abstract]. Toxicologist 36:331.
Freeman, J.J., T.M. Federici, and R.H. McKee. 1993. Evaluation of the contribution of chronic skin irritation and selected compositional parameters to the tumorigenicity of petroleum middle distillates in mouse skin. Toxicology 81(2):103-112.
Gargas, M.L., R.J. Burgess, D.E. Voisard, G.H. Cason, and M.E. Andersen. 1989. Partition coefficients of low-molecular-weight volatile chemicals in various liquids and tissues. Toxicol. Appl. Pharmacol. 98(1):87-99.
Gaworski, C.L., J.D. MacEwen, E.H. Vernot, R.H. Bruner, and M.J. Cowan, Jr. 1984. Comparison of the subchronic inhalation toxicity of petroleum and oil shale JP-5 jet fuels Pp. 33-47 in Applied Toxicology of Petroleum Hydrocarbons, H.N. MacFarland, C.E. Holdsworth, J.A. MacGregor, R.W. Call and M.L. Kane, eds. Advances in Modern Environmental Toxicology Vol. 6. Princeton, NJ: Princeton Scientific Publishers.
Gaworski, C.L., J.D. MacEwen, E.H. Vernot, C.C. Haun, H.F. Leahy, R.H. Bruner, G.B. Baskin and M.J. Cowan, Jr. 1985. Evaluation of the 90-day Inhalation Toxicity of Petroleum and Oil Shale JP-5 Jet Fuels. ADA156 815. AFAMRL-TR-85-035. NMRI 85-18. Aerospace Medical Research Laboratory, Wright-Patterson Air Force Base, OH.
Haddad, S., M. Beliveau, R. Tardif, and K. Krishnan. 2001. A PBPK modeling-based approach for interactions in health risk assessment of chemical mixtures. Toxicol. Sci. 63(1):125-131.
Harris, D.T., D. Sakiestewa, R.F. Robledo, and M. Witten. 1997a. Immunotoxicological effects of JP-8 jet fuel exposure. Toxicol. Ind. Health 13(1):43-55.
Harris, D.T., D. Sakiestewa, R.F. Robledo, and M. Witten. 1997b. Short-term exposure to JP-8 jet fuel results in long-term immunotoxicity. Toxicol. Ind. Health 13(5):559-570.
Harris, D.T., D. Sakiestewa, R.F. Robledo, and M. Witten. 1997c. Protection from JP-8 jet fuel induced immunotoxicity by administration of aerosolized substance P. Toxicol. Ind. Health 13(5):571-588.
Harris, D.T., D. Sakiestewa, D. Titone, R.F. Robledo, R.S. Young, and M. Witten. 2000. Jet fuel-induced immunotoxicity. Toxicol. Ind. Health 16(7-8):261-265.
Harris, D.T., D. Sakiestewa, D. Titone, and M. Witten. 2001. JP-8 Jet Fuel: Deleterious Effects of In Utero Exposure and Potentiating Effects in a Experimental Tumor Model. Presented at the International Conference on the Environmental Health and Safety of Jet Fuel, August 8-11, 2001, San Antonio, TX.
Hays, A.M., G. Parliman, J.K. Pfaff, R.C. Lantz, J. Tinajero, B. Tollinger, J.N. Hall, and M.L. Witten. 1995. Changes in lung permeability correlate with lung histology in a chronic exposure model. Toxicol. Ind. Health 11(3):325-336.
Henz, K. 1998. Survey of Jet Fuels Procured by the Defense Energy Support Center, 1990-1996. Defense Logistics Agency, Ft. Belvior, VA.
Holm, S., D. Norback, B. Frenning, and C.J. Gothe. 1987. Hydrocarbon exposure from handling jet fuel at some Swedish aircraft units. Scand. J. Work. Environ. Health 13:438-444.
IARC (International Agency for Research on Cancer). 1989. Occupational Exposures in Petroleum Refining; Crude Oil and Major Petroleum Fuels. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans Vol. 45. Lyon: IARC
Jackman, S.M., C.J. Kolanko, D.A. Stenger, J. Noth, and G.M. Grant. 2001. In Vitro Investigations of JP-8 Genotoxicity. Presented at the International Conference on the Environmental Health and Safety of Jet Fuels, August 8-11, 2001, San Antonio, TX.
Kabbur, M.B., J.V. Rogers, P.G. Gunasekar, C.M. Garrett, K.T. Geiss, W.W. Brinkley, and J.N. McDougal. 2001. Effect of JP-8 jet fuel on molecular and histological parameters related to acute skin irritation. Toxicol. Appl. Pharmacol. 175(1):83-88.
Kale, A., S. Praster, W. Thomas, I. Amende, and T.G. Hampton. 2002. Respiratory Frequency of Mice at Rest and After Exercise. Mouse Specifics, Inc. [online]. Available: https://www.mousespecifics.com/physio/PFarticle13.html [accessed Oct. 8, 2010].
Kanikkannan, N., T. Jackson, M.S. Shaik, and M. Singh. 2000. Evaluation of skin sensitization potential of jet fuels by murine local lymph node assay. Toxicol. Lett. 116(1-2):165-170.
Kanikkannan, N., R. Patel, T. Jackson, M.S. Shaik, and M. Singh. 2001. Percutaneous absorption and skin irritation of JP-8 (jet fuel). Toxicology 161(1-2):1-11.
Keil, D.E., D.A. Warren, M.J. Jenny, J.G. EuDaly, J. Smythe, and M.M. Peden-Adams. 2003. Immunological function in mice exposed to JP-8 jet fuel in utero. Toxicol. Sci. 76(2):347-356.
Kennedy, S.K., and D.E. Longnecker. 1996. History and principles of anesthesiology. Pp. 295-306 in Goodman & Gilman’s The Pharmacological Basis of Therapeutics, 9th Ed., J.G. Hardman, L.E. Limbird, P.B. Molinoff, R.W. Ruddon, and A.G. Gilman, eds. New York: McGraw-Hill.
Kinkead, E.R., R.E. Wolfe, and S.A. Salins. 1992a. Acute irritation and sensitization potential of petroleum-derived JP-5 jet fuel. Int. J. Toxicol. 11(6):706.
Kinkead, E.R., R.E. Wolfe, and S.A. Salins. 1992b. Acute irritation and sensitization potential of JP-8 jet fuel. Int. J. Toxicol. 11(6):700.
Knave, B., B.A. Olson, S. Elofsson, F. Gamberale, A. Isaksson, P. Mindus, H.E. Persson, G. Struwe, A. Wennberg, and P. Westerholm. 1978. Long-term exposure to jet fuel. II. A cross-sectional epidemiologic investigation on occupationally exposed industrial workers with special reference to the nervous system. Scand. J. Work Environ. Health 4(1):19-45.
Kornguth, S. 1998. The Effects of Jet Fuel (JP-8) on Mouse Retinal and Cerebellar Function. Presented at the 1998 Workshop on Neurologic Assessment Protocols (Neuro Sub-IPT), August 31 -September 2, 1998, San Antonio, TX.
Kristiansen, U., and G.D. Nielsen. 1988. Activation of the sensory irritant receptor by C7-C11 n-alkanes. Arch. Toxicol. 61(6):419-425.
Lam, C.W., T.J. Galen, J.F. Boyd, and D.L. Pierson. 1990. Mechanism of transport and distribution of organic solvents in blood. Toxicol. Appl. Pharmacol. 104(1):117-129.
Leith, D., M. Boundry, R. Goodman, J. McKernan, L. Smith, and G. Carlton. 1998. Development of Aerosol Measurement Method for JP-8. Presented at the International Conference on the Environmental Health and Safety of Jet Fuel, April 1-3, 1998, San Antonio, TX.
LeMasters, G.K., D.M. Olsen, J.H. Yinn, J.E. Lockey, R. Shukla, S.G. Selevan, S.M. Schrader, G.P. Toth, D.P. Evenson, and G.B. Huszar. 1999. Male reproductive effects of solvent and fuel exposure during aircraft maintenance. Reprod. Toxicol. 13(3):155-166.
Lof, A., H.R. Lam, E. Gullstrand, G. Ostergaard, and O. Ladefoged. 1999. Distribution of dearomatised white spirit in brain, blood, and fat tissue after repeated exposure of rats. Pharmacol. Toxicol. 85(2):92-97.
MacEwen, J.D., and E.H. Vernot. 1984. Toxic Hazards Research Unit Annual Report: 1984. AFAMRL-TR-84-001. NMRI-84-42. ADA147857. Aerospace Medical Research Laboratory, Wright-Patterson Air Force Base, OH.
MacEwen, J.D., and E.H. Vernot. 1985. Investigation of the 1-h emergency exposure limit of JP-5. Pp. 137-144 in Toxic Hazards Research Unit Annual Report: 1985. AAMRL-TR-85-058. AD-A161558. Aerospace Medical Research Laboratory, Wright-Patterson Air Force Base, OH.
MacNaughton, M.G., and D.E. Uddin. 1984. Toxicology of mixed distillate and high-energy synthetic fuels. Pp. 121-132 in Renal Effects of Petroleum Hydrocarbons, M.A. Mehlman, C.P. Hemstreet, J.J. Thorpe, and N.K. Weaver, eds. Advances in Modern Environmental Toxicology Vol. VII. Princeton, NJ: Princeton Scientific Publishers.
Marshall, B.E., and D.E. Longnecker. 1996. General anesthetics. Pp. 307-330 in in Goodman & Gilman’s The Pharmacological Basis of Therapeutics, 9th Ed., J.G. Hardman, L.E. Limbird, P.B. Molinoff, R.W. Ruddon, and A.G. Gilman, eds. New York: McGraw-Hill.
Martel, C.R. 1989. Properties of F-34 (JP-8) Fuel for 1988. WRDC-TR-89-2021. ADA210188. Aero Propulsion and Power Laboratory, Wright-Patterson AFB, OH.
Martin, S.A., R.T. Tremblay, K.F. Brunson, C. Kendrick, and J.W. Fisher. 2010. Characterization of a nose-only inhalation exposure system for hydrocarbon mixtures and jet fuels. Inhal. Toxicol. 22(5):382-393.
Martone, J.A. 1981. An Industrial Hygiene Evaluation of Aircraft Refueling Inside Closed Aircraft Shelters. AD A098708. Technical Report BEES (W) 81-03. Air Force Hospital Wiesbaden APO NY.
Mattie, D.R., C.L. Alden, T.K. Newell, C.L. Gaworski, and C.D. Flemming. 1991. A 90-day continuous vapor inhalation toxicity study of JP-8 jet fuel followed by 20 or 21 months of recovery in Fischer 344 rats and C57BL/6 mice. Toxicol. Pathol. 19(2):77-87.
Mattie, D.R., G.B. Marit, C.D. Flemming, and J.R. Cooper. 1995. The effects of JP-8 jet fuel on male Sprague-Dawley rats after a 90-day exposure by oral gavage. Toxicol. Ind. Health 11(4):423-435.
Mattie, D.R., G.B. Marit, J.R. Cooper, T.R. Sterner, and C.D. Flemming. 2000. Reproductive Effects of JP-8 Jet Fuel on Male and Female Sprague-Dawley Rats after Exposure by Oral Gavage. Interim Report: June 1993-March 2000. AFRL-HEWP-TR-2000-0067. Air Force Research Laboratory, Wright-Patterson AFB, OH.
McDougal, J.N. and P.J. Robinson. 2002. Assessment of dermal absorption and penetration of components of a fuel mixture (JP-8). Sci. Total Environ. 288(1-2):23-30.
McDougal, J.N., and J.V. Rogers. 2004. Local and systemic toxicity of JP-8 from cutaneous exposures. Toxicol. Lett. 149(1-3):301-308.
McDougal, J.N., D.L. Pollard, W. Weisman, C.M. Garrett, and T.E. Miller. 2000. Assessment of skin absorption and penetration of JP-8 jet fuel and its components. Toxicol. Sci. 55(2):247-255.
McInturf, S.M., M.Y. Bekkedal, G.D. Ritchie, A.F. Nordholm, and J. Rossi. 2001. Effects of repeated JP-8 exposure on eyeblink conditioning in humans [abstract]. Toxicologist 60(1):555.
Mihic, S.J., S.J. McQuilkin, E.I. Eger, P. Ionescu, and R.A. Harris. 1994. Potentiation of γ-aminobytyric acid type A receptor-mediated chloride currents by novel halogenated compounds correlates with their abilities to induce general anesthesia. Mol. Pharmacol. 46(5):851-857.
Monteiro-Riviere, N., A. Inman, and J. Riviere. 2001. Effects of short-term high-dose and low-dose dermal exposure to Jet A, JP-8 and JP-8 + 100 jet fuels. J. Appl. Toxicol. 21(6):485-494.
Monteiro-Riviere, N., A. Inman, and J.E. Riviere. 2004. Skin toxicity of jet fuels: Ultrastructural studies and the effects of substance P. Toxicol. Appl. Pharmacol. 195(3):339-347.
NAVOSH (Navy Occupational Safety and Health). 1992. Interim Recommendations for Permissible Fuel Exposure Criteria from the Navy Environmental Health Center’s Navy Occupational Safety and Health (NAVOSH) Standards Update Board. Memorandum 6261, Ser 34/2514. Navy Environmental Health Center, Norfolk, VA. April 9, 1992 .
NEHC (U.S. Navy Environmental Health Center). 1993. Industrial Hygiene Field Operations Manual. Technical Manual NEHC-TM91-2. U.S. Navy Environmental Health Center, Norfolk, VA.
NEHC (U.S. Navy Environmental Health Center). 2001. Fuels Comparison Chart. U.S. Navy Environmental Health Center, Norfolk, VA. October 2001 [online]. Availble: http://www-nehc.med.navy.mil/downloads/ep/fuels_comp_chart.pdf [accessed Oct. 11, 2010].
Nessel, C.S. 1999. A comprehensive evaluation of the carcinogenic potential of middle distillate duels. Drug Chem. Toxicol. 22(1):165-180.
Nilsen, O.G., O.A. Haugen, K. Zahlsen, J. Halganset, A. Helseth, H. Aarset, and I. Eide. 1988. Toxicity of n-C9 to n-C13 alkanes in the rat on short term inhalation. Pharmacol. Toxicol. 62(5):259-266.
NIOSH (National Institute for Occupational Safety and Health). 1996. Documentation for Immediately Dangerous to Life or Health Concentrations (IDLH): NIOSH Chemical Listing and Documentation of Revised IDLH Values (as of 3/1/95)-Petroleum Distillates (Naphtha). U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health. August 1996 [online]. Available: http://www.cdc.gov/niosh/idlh/8002059.html [accessed Oct. 11, 2010].
NIOSH (National Institute for Occupational Safety and Health). 2005. NIOSH Pocket Guide to Chemical Hazards: Kerosene. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, Cincinnati, OH. September 2005 [online]. Available: http://www.cdc.gov/niosh/npg/npgd0366.html [accessed Oct. 11, 2010].
Norseth, T., L. Toftegaard, and O. Romerike. 1998. Measures of Liver Function in Norwegian Crewchiefs. Presented at the International Conference on the Environmental Health and Safety of Jet Fuel, April 1-3, 1998, San Antonio, TX.
NRC (National Research Council). 1993. Guidelines for Developing Community Emergency Exposure Levels for Hazardous Substances. Washington, DC: National Academy Press.
NRC (National Research Council). 1996. Permissible Exposure Levels for Selected Military Fuel Vapors. Washington DC: National Academy Press.
NRC (National Research Council). 2001. Standing Operating Procedures for Developing Acute Exposure Guideline Levels for Hazardous Chemicals. Washington, DC: National Academy Press.
NRC (National Research Council). 2003. Toxicologic Assessment of Jet-Propulsion Fuel 8. Washington, DC: The National Academies Press.
NTP (National Toxicology Program). 1986. Toxicology and Carcinogenesis Studies of Marine Diesel Fuel and JP-5 Navy Fuel (CAS No. 8008-20-6) in B6C3F1 Mice (Dermal Studies). NTP TR 310. NIH 86-2566. U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health, National Toxicology Program, Research Triangle Park, NC.
Olsen, D.M. 1998. Occupational Assessment of Air Force Personnel Exposure to Jet Fuel Before and After Conversion to JP-8. Presented at the International Conference on the Environmental Health and Safety of Jet Fuel, April 1-3, 1998, San Antonio, TX.
Parton, K.H., J. Pfaff, A.M. Hays, and M. Witten. 1993. Effects of JP-8 jet fuel inhalation on the liver of F-344 rats. Toxicologist 13:48.
Pedersen, L.M., K. Larsen, and K.H. Cohr. 1984. Kinetics of white spirit in human fat and blood during short-term experimental exposure. Act Pharacol. Toxicol. 55(4): 308-316.
Perbellini, L., F. Brugnone, D. Caretta, and G. Maranelli. 1985. Partition coefficients of some industrial aliphatic hydrocarbons (C5-C7) in blood and human tissues. Br. J. Ind. Med. 42(3):162-167.
Perleberg, U.R., D.A. Keys, and J.W. Fisher. 2004. Development of a physiologically based pharmacokinetic model for decane, a constituent of jet propellent-8. Inhal. Toxicol. 16(11-12):771-783.
Pfaff, J., K. Parton, R. Lantz, H. Chen, D. Carter, and M. Witten. 1992. Effects of JP-8 jet fuel exposure on pulmonary function [abstract]. Am. Rev. Respir. Dis. 145 (4 Part 2):A89.
Pfaff, J., G. Parliman, K. Parton, R. Lantz, H. Chen, M. Hays, M. Witten. 1993. Pathologic changes after JP-8 jet fuel inhalation in Fischer 344 rats [abstract]. FASEB J. 7:A408.
Pfaff, J., K. Parton, R.C. Lantz, H. Chen, A.M. Hays, and M.L. Witten. 1995. Inhalation exposure to JP-8 jet fuel alters pulmonary function and substance P levels in Fischer 344 rats. J. Appl. Toxicol. 15(4):249-256.
Pfaff, J.K., B.J. Tollinger, R.C. Lantz, H. Chen, A.M. Hays, and M.L. Witten. 1996. Neural endopeptidase (NEP) and its role in pathological pulmonary change with inhalation exposure to JP-8 jet fuel. Toxicol. Ind. Health 12(1):93-103.
Pleil, J.D., L.B. Smith, and S.D. Zelnick. 2000. Personal exposure to JP-8 jet fuel vapors and exhaust at Air Force bases. Environ. Health Perspect. 108(3):183-192.
Porter, H.O. 1990. Aviators intoxicated by inhalation of JP-5 fuel vapors. Aviat. Space Environ. Med. 61(7):654-656.
Potter, T.L., and K.E. Simmons. 1998. Composition of Petroleum Mixtures. Hydrocarbon Working Group Series Vol. 2. Amherst, MA: Amherst Scientific Publishers.
Puhala, E., G. LeMasters, L. Smith, G. Talaska, S. Simpson, J. Joyce, K. Trinh, and J. Lu. 1997. Jet fuel exposure in the United States Air Force. Appl. Occup. Environ. Hyg. 12(9):606-610.
Rhodes, A.G., G.K. LeMasters, J.E. Lockey, J.W. Smith, J.H. Yiin, R. Gibson, and S. Rappaport. 2001. The effects of JP8 on immune cell counts of tank entry workers. Pp. 100-120 in JP-8 Final Risk Assessment Report, R.K. Kendall, Ernest Smith, L.B. Smith, and R.L. Gibson, eds. IOH-RS-BR-SR-2005-0003. Texas Tech University,
The Institute of Environmental and Human Health, Lubbock, TX. August 2001 [online]. Available: http://airforcemedicine.afms.mil/idc/groups/public/documents/afms/ctb_044060.pdf [accessed Oct. 11, 2010].
Ritchie, G.D., M.Y. Bekkedal, A.J. Bobb, and K.R. Still. 2001a. Biological and Health Effects of JP-8 Exposure. Report No. TOXDET-01-01, Naval Health Research Center Detachment (Toxicology), Wright-Patterson AFB, OH [online]. Available: http://www.gooptroop.com/gtroop/JetFuel/JP8%20Health%20Issues.pdf [accessed Oct. 12, 2010].
Ritchie, G.D., K.R. Still, W.K. Alexander, A.F. Nordholm, C.L. Wilson, J. Rossi, III, and D.R. Mattie. 2001b. A review of the neurotoxicity risk of selected hydrocarbon fuels. J. Toxicol. Environ. Health B Crit. Rev. 4(3):223-312.
Ritchie, G.D., J. Rossi III, A.F. Nordholm, K.R. Still, R.L. Carpenter, G.R. Wenger, and D.W. Wright. 2001c. Effects of repeated exposure to JP-8 jet fuel vapor on learning of simple and difficult operant tasks by rats. J. Toxicol. Environ. Health A 64(5):385-415.
Ritchie, G.D., K.R. Still, J. Rossi III, M. Bekkedal, A.J. Bobb, and D.P. Arfsten. 2003. Biological and health effects of exposure to kerosene-based jet fuels and performance additives. J. Toxicol. Environ. Health B Crit. Rev. 6(4):357-451.
Riviere, J.E., J.D. Brooks, N.A. Monteiro-Riviere, K. Budsaba, and C.E. Smith. 1999. Dermal absorption and distribution of topically dosed jet fuels Jet-A, JP-8, and JP-8(100). Toxicol. Appl. Pharmacol. 160(1):60-75.
Robinson, P.J. 2000. Pharmacokinetic Modeling of JP-8 Jet Fuel Components. I. Nonane and C9-C12 Aliphatic Components. AFRL-HE-WP-TR-2000-0046. ADA435036. Human Effectiveness Directorate, Wright-Patterson AFB, OH.
Robledo, R.F., and M.L. Witten. 1998. Acute pulmonary response to inhaled JP-8 jet fuel aerosol in mice. Inhal. Toxicol. 10(5):531-553.
Robledo, R.F., R.S. Young, R.C. Lantz, and M.L. Witten. 2000. Short-term pulmonary response to inhaled JP-8 jet fuel aerosol in mice. Toxicol. Pathol. 28(5):656-663.
Rossi, J., III, A.F. Nordholm, R.L Carpenter, G.D. Ritchie, and W. Malcomb. 2001. Effects of repeated exposure of rats to JP-5 or JP-8 jet fuel vapor on neurobehavioral capacity and neurotransmitter levels. J. Toxicol. Environ. Health A 63(6):397-428.
Schaper, M. 1993. Development of a database for sensory irritants and its use in establishing occupational exposure limits. Am. Ind. Hyg. Assoc. J. 54(9):488-544.
Selden, A., and G. Ahlborg, Jr. 1986. Causes of Death and Cancer Morbidity at Exposure to Aviation Fuels in the Swedish Armed Forces. ASF Project 84-0308. Department of Occupational Medicine, Orebro, Sweden (as cited in NRC 1996).
Selden, A., and G. Ahlborg, Jr. 1987. Causes of Death and Cancer Morbidity at Exposure to Aviation Fuels in the Swedish Armed Forces: An Update. Department of Occupational Medicine, Orebro, Sweden(as cited in NRC 1996).
Selden, A., and G. Ahlborg, Jr. 1991. Mortality and cancer morbidity after exposure to military aircraft fuel. Aviat. Space Environ. Med. 62(8):789-794.
Smith, L. 1998. USAF Jet Fuel Program: Industrial Hygiene Perspective. Presented at the 1998 Workshop on Neurologic Assessment Protocols (Neuro Sub-IPT), August 31 to September 2, 1998, San Antonio, TX.
Smith, L.B., and S. Zelnick. 1998. Benzene Exposure Study during Aircraft Fuel Tank Entry. Presented at the International Conference on the Environmental Health and Safety of Jet Fuel, April 1-3, 1998, San Antonio, TX.
Smith, L.B., A. Bhattacharya, G. LeMasters, P. Succop, E. Puhala, II, M. Medvedovic, and J. Joyce. 1997. Effect of chronic low-level exposure to jet fuel on postural balance of US Air Force personnel. J. Occup. Environ. Med. 39(7):623-632.
Stokholm, J. and K.H. Cohr. 1979. Exposure of Humans to White Spirit. III. Concentrations in Alveolar Air and Venous Blood during Experimental Exposure [in Danish with English Summary]. Institute of Occupational Health Report No. 3/1979. Copenhagen: Danish National Labour Inspection Service.
Struwe, G., B. Knave, and P. Mindus. 1983. Neuropsychiatric symptoms in workers occupationally exposed to jet fuel - a combined epidemiological and casuistic study. Acta. Psychiatr. Scand. Suppl. 303:55-67.
Swenberg, J.A. 1993. α2u-Globulin nephropathy: Review of the cellular and molecular mechanisms involved and their implications for human risk assessment. Environ. Health Perspect. 101(Suppl. 6):39-44.
SwRI (Southwest Research Institute). 2001. JP-8 Volatility Study. IERA-RS-BR-SR-2001-0002. Southwest Research Institute, San Antonio, TX. March 2001.
Tardif, R., G. Charest-Tardif, J. Brodeur, and K. Krishnan. 1997. Physiologically based pharmacokinetic modeling of a ternary mixture of alkyl benzenes in rats and humans. Toxicol. Appl. Pharmacol. 144(1):120-134.
Thomas, T.C., and A. Richardson III. 1981. An infrared analysis method for the determination of hydrocarbons collected on charcoal tubes. Pp. 37-48 in Chemical Hazards in the Workplace, G. Choudhary, ed. ACS Symposium Series 149. Washington, DC: American Chemical Society.
Tremblay, R.T., S.A. Martin, and J.W. Fisher. 2010. Novel characterization of the aerosol and gas-phase composition for aerosolized jet fuel atmospheres. Inhal. Toxicol. 22(5):394-401.
Ullrich, S.E. 1999. Dermal application of JP-8 jet fuel induces immune suppression. Toxicol. Sci. 52(1):61-67.
Ullrich, S.E., and H.E. Lyons. 2000. Mechanisms involved in the immunotoxicity induced by dermal application of JP-8 jet fuel. Toxicol. Sci. 58(2):290-298.
University of California. 2006. Researchers Reveal Lung's Unique Innate Immune System. Science Daily, April 19, 2006 [online]. Available: http://www.sciencedaily.com/releases/2006/04/060419073650.htm [accessed Oct. 12, 2010].
U.S. Air Force. 1989. The Installation Restoration Program Toxicology Guide, Volume 4. ADA215002. Prepared by Oak Ridge National Laboratory, Oak Ridge, TN, for the Air Force Systems Command, Wright-Patterson Air Force Base, OH.
Whitman, F.T., and J.P. Hinz. 2001. Sensory Irritation Study in Mice: JP-4, JP-8, JP-8+100. Report No. IERA RS-BR-SR-2001-0005. ADA398112. Air Force Institute for Environment, Safety and Occupational Health Risk Analysis, Brooks Air Force Base TX [online]. Available: http://www.dtic.mil/cgi-bin/GetTRDoc?AD=ADA398112&Location=U2&doc=GetTRDoc.pdf [accessed Oct. 13, 2010].
Witschi, H.R., and J.A. Last. 2001. Toxic responses of the respiratory system. Pp. 515-534 in Casarett & Doull’s Toxicology: The Basic Science of Poisons, 6th Ed., C.D. Klaassen, ed. New York: McGraw-Hill.
Witten, M.L. 1994. The Chronic Effects of JP-8 Jet Fuel Exposure on the Lungs. Technical Report, AFOSR-TR-940382. ADA280982. University of Arizona, Tucson, AZ.
Witten, M.L., S.E. Leeman, R.C. Lantz, P.M. Joseph, C.H. Burke, W.K. Jung, D. Quinn, and C.A. Hales. 1990. Chronic jet fuel exposure increases lung substance P (SP) concentration in rabbits. P. 29 in Substance P and Related Peptides: Cellular and Molecular Physiology, S.E. Leeman, ed. International Symposium, University of Massachusetts and the New York Academy of Science, New York.
Witten, M.L., J.K. Pfaff, K. Parton, R.C. Lantz, D. Carter, and S.E. Leeman. 1992. JP-8 jet fuel exposure alters lung chemical mediator and substance P activity in rats [abstract]. FASEB J. 6:A1065.
Witzmann, F.A., M.D. Bauer, A.M. Fieno, R.A. Grant, T.W. Keougth, S.E. Kornguth, M.P. Lacey, F.L. Siegel, Y. Sun, L.S. Wright, R.S. Young, and M.L. Witten. 1999. Proteomic analysis of simulated occupational jet fuel exposure in the lung. Electrophoresis 20(18):3659-3669.
Witzmann, F.A., R.L. Carpenter, G.D. Ritchie, C.L. Wilson, A.F. Nordholm, and J. Rossi III. 2000a. Toxicity of chemical mixtures: Proteomic analysis of persisting liver and kidney protein alterations induced by repeated exposure of rats to JP-8 jet fuel vapor. Electrophoresis 21(11):2138-2147.
Witzmann, F.A., M.D. Bauer, A.M. Fieno, R.A. Grant, T.W. Keougth, M.P. Lacey, Y. Sun, M.L. Witten, and R.S. Young. 2000b. Proteomic analysis of the renal effects of simulated occupational jet fuel exposure. Electrophoresis 21(5):976-984.
Witzmann, F.A., A. Bobb, G.B. Briggs, H.N. Coppage, R.A. Hess, J.Y. Li, N.M. Pedrick, G.D. Ritchie, J. Rossi III, and K.R. Still. 2003. Analysis of rat testicular protein expression following 91-day exposure to JP-8 jet fuel vapor. Proteomics 3(6):1016-1027.
Wolfe, R.E., E.R. Kinkead, M.L. Feldmann, H.F. Leahy, W.W. Jederberg, K.R. Still, and D.R. Mattie. 1996. Acute Toxicity Evaluation of JP-8 Jet Fuel and JP-8 Jet Fuel Containing Additives. AL/OE-TR-1996-0136. NMRI-94-114. Prepared by ManTech Environmental Technology, Inc., Dayton, OH, for Armstrong Laboratory, Occupational and Environmental Health Directorate, Toxicology Division, Wright-Patterson AFB, OH.
Young, R.S., and M.L. Witten. 2000. Age Related Alterations in Pulmonary Function and Pulmonary Lipid Peroxidation After Exposure to JP-8 and JP-8+100 Blend Jet Fuel. Presented at the AFOSR JP-8 Jet Fuel Toxicology Workshop, January 11-12, 2000, University of Arizona, Tucson, AZ.
Zahlsen, K., A.M. Nilsen, I. Eide, and O.G. Nilsen. 1990. Accumulation and distribution of aliphatic (n-nonane), aromatic (1,2,4-trimethylbenzene) and napthenic (1,2,4-trimethylcyclohexane) hydrocarbons in the rat after repeated inhalation. Pharmacol. Toxicol. 67(5):436-440.
Zahlsen, K., I. Eide, A,M, Nielsen, and O.G. Nilsen. 1992. Inhalation kinetics of C6 to C10 aliphatic, aromatic and naphthenic hydrocarbons in rat after repeated exposures. Pharmacol. Toxicol.71(2):144-149.
Zeiger, E., and L. Smith. 1998. The first international conference on the environmental health and safety of jet fuel. Environ. Health Perspect. 106(11):763-764.
APPENDIX A
INDIVIDUAL HYDROCARBON DATA—NEAT JET FUELS,a PERCENT BY VOLUME
TABLE A-1 Hydrocarbon Data for Jet Fuels 4, 8, and 8+100
| Hydrocarbon | JP-4 | JP-8 | JP-8+100 |
| Isopentane | 0.1437 | ND | ND |
| n-Pentane | 0.3237 | ND | ND |
| 2-Methylpentane | 0.7302 | ND | ND |
| 3-Methylpentane | 0.4139 | ND | ND |
| n-Hexane | 1.5403 | 0.0011 | 0.0023 |
| Methylcyclopentane | 1.3913 | 0.0006 | 0.0014 |
| Benzene | 0.3551 | ND | ND |
| Cyclohexane | 1.7803 | 0.0033 | 0.0030 |
| 3-Methylhexane | 1.5065 | 0.0119 | 0.0078 |
| Isooctane | 2.4941 | 0.0256 | 0.0112 |
| n-Heptane | 3.3458 | 0.0481 | 0.0357 |
| Toluene | 2.0009 | 0.0721 | 0.0664 |
| 3-Methylheptane | 0.9567 | 0.0604 | 0.0524 |
| n-Octane | 3.8056 | 0.2609 | 0.2506 |
| Ethylbenzene | 0.6458 | 0.1414 | 0.1322 |
| p-, m-Xylene | 3.3541 | 0.6610 | 0.6319 |
| n-Nonane | 1.5714 | 0.9103 | 0.8886 |
| Cumene | 0.1870 | 0.1756 | 0.1672 |
| Propylbenzene | 0.1830 | 0.2846 | 0.2698 |
| p-, m-Ethyltoluene | 0.5318 | 0.6921 | 0.6801 |
| 1,3,5-Trimethylbenzene | 0.5948 | 1.0785 | 1.0677 |
| o-Ethyltoluene | 0.4586 | 0.8522 | 0.8416 |
| 1,2,4-Trimethylbenzene | 0.8171 | 1.2355 | 1.2192 |
| n-Decane | 1.2687 | 2.8907 | 2.8641 |
| n-Undecane | 1.7350 | 5.5171 | 5.5065 |
| n-Dodecane | 1.8808 | 5.3191 | 5.3032 |
| n-Tetradecane | 1.4537 | 3.0523 | 3.0658 |
| n-Hexadecane | 0.3169 | 0.7690 | 0.7602 |
| Total analytesb | 35.7868 | 24.0634 | 23.8289 |
aSource: Whitman and Hinz 2001, p. 46.
bPercent of test substance analyzed; other hydrocarbons unidentified.
APPENDIX B
CATEGORY GRAPH OF TOXICITY DATA AND AEGL VALUES
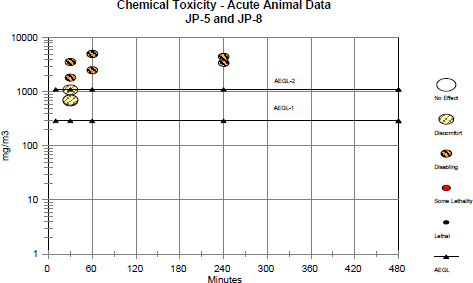
FIGURE 2-1 Category graph for JP-5 and JP-8. Note: Only acute studies are graphed.
TABLE B-2 Data Used in Category Graph
| Source | Species | mg/m3 | Minutes | Categorya |
| NAC/AEGL-1 | 290 | 10 | AEGL | |
| NAC/AEGL-1 | 290 | 30 | AEGL | |
| NAC/AEGL-1 | 290 | 60 | AEGL | |
| NAC/AEGL-1 | 290 | 240 | AEGL | |
| NAC/AEGL-1 | 290 | 480 | AEGL | |
| NAC/AEGL-2 | 1,100 | 10 | AEGL | |
| NAC/AEGL-2 | 1,100 | 30 | AEGL | |
| NAC/AEGL-2 | 1,100 | 60 | AEGL | |
| NAC/AEGL-2 | 1,100 | 240 | AEGL | |
| NAC/AEGL-2 | 1,100 | 480 | AEGL | |
| Source | Species | mg/m3 | Minutes | Categorya |
| NAC/AEGL-3 | ND | 10 | AEGL | |
| NAC/AEGL-3 | ND | 30 | AEGL | |
| NAC/AEGL-3 | ND | 60 | AEGL | |
| NAC/AEGL-3 | ND | 240 | AEGL | |
| NAC/AEGL-3 | ND | 480 | AEGL | |
| Whitman and Hinz 2001 | Mouse | 681 | 30 | 1: 22% depression, respiratory rate |
| Whitman and Hinz 2001 | Mouse | 1,090 | 30 | 1: 38% depression, respiratory rate |
| Whitman and Hinz 2001 | Mouse | 1,837 | 30 | 2: 46% depression, respiratory rate |
| Whitman and Hinz 2001 | Mouse | 3,565 | 30 | 2: 50% depression, respiratory rate |
| Whitman and Hinz 2001 | Mouse | 708 (vapor) | 30 | 1: 28% depression, respiratory rate |
| Wolfe et al. 1996 | Rat | 3,430 (vapor) | 240 | 2: Eye, upper respiratory tract irritation |
| Wolfe et al. 1996 | Rat | 4,440 (2,630 vapor + 1,810 aerosol) | 240 | 2: No deaths |
| MacEwen and Vernot 1985 | Rat | 2,500 | 60 | 2: Eye irritation |
| MacEwen and Vernot 1985 | Rat | 5,000 | 60 | 2: CNS depression |
aCategories: 0, no effect; 1, discomfort; 2, disabling; and 3, lethal.
Abbreviations: ND, not determined; CNS, central nervous system.
APPENDIX C
ACUTE EXPOSURE GUIDELINE LEVELS FOR JP-5 AND JP-8
Derivation Summary for JP-5 AND JP-8
AEGL-1 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 |
| Key reference: Whitman, F.T., and J.P. Hinz. 2001. Sensory Irritation Study in Mice: JP-4, JP-8, JP-8+100. Report No. IERA RS-BR-SR-2001-0005. ADA398112. Air Force Institute for Environment, Safety and Occupational Health Risk Analysis, Brooks Air Force Base TX. | ||||
| Supporting reference: MacEwen, J.D., and E.H. Vernot. 1985. Investigation of the 1-h emergency exposure limit of JP-5. Pp. 137-144 in Toxic Hazards Research Unit Annual Report: 1985. AAMRL-TR-85-058. AD-A161558. Aerospace Medical Research Laboratory, Wright-Patterson Air Force Base, OH. | ||||
| Test species/Strain/Number: Mouse/Swiss-Webster/4 per group | ||||
| Exposure route/Concentrations/Durations: Inhalation concentrations at 681, 1,090, 1,837, 3,565 mg/m3 (vapor + aerosol), and 708 mg/m3 (vapor only) for 30 min | ||||
| Effects: | ||||
| 681 mg/m3: 22% decrease in respiratory rate | ||||
| 1,090 mg/m3: 38% decrease in respiratory rate | ||||
| 1,837 mg/m3: 46% decrease in respiratory rate | ||||
| 3,565 mg/m3: 50% decrease in respiratory rate | ||||
| 708 mg/m3: 28% decrease in respiratory rate (vapor-only exposure) | ||||
| 2,876 mg/m3: calculated RD50 | ||||
| End point/Concentration/Rationale: 290 mg/m3 across all AEGL-1 exposure durations (0.1 times the calculated mouse RD50 of 2,876 mg/m3 [slight irritation withstood for hours,according to Alarie 1981]) | ||||
| Uncertainty factors/Rationale: | ||||
| Total uncertainty factor: Not applicable. (The mouse RD50 was reduced by a factor of 10, which reduces the sensory irritation to a concentration tolerated for hours to days by most individuals.) The factor of 10 is the same as applying interspecies and intraspecies uncertainty factors of 3 each. | ||||
| Interspecies: Not applicable | ||||
| Intraspecies: Not applicable | ||||
| Modifying factor: Not applied | ||||
| Animal-to-human dosimetric adjustment: Not applied | ||||
| Time-scaling: Not applied; the repeated nature of many of the studies ensures the safety of a single exposure. | ||||
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 |
| Data adequacy: The database of inhalation and oral studies in rodents is robust. Studies addressed irritation, neurotoxicity, immunotoxicity, developmental and reproductive effects, genotoxicity, and carcinogenicity. Human exposures to JP-8 were limited to occupational exposures; monitoring studies with other aviation fuels showed few effects. The value is supported by animal studies in which repeated and continuous exposures at 1,000 mg/m3 for up to 90 days failed to elicit clinical signs or adverse health effects. Interspecies and intraspecies uncertainty factors of 1 and 3, respectively would result in a similar value, 330 mg/m3. | ||||
AEGL-2 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 | 1,100 mg/m3 |
| Key references: | ||||
| Briggs, G.B. 2001. Evaluation of Military Fuel Potential to Produce Male Reproductive Toxicity. Presented at the International Conference on the Environmental Health and Safety of Jet Fuel, August 8-11, 2001, San Antonio, TX. | ||||
| MacEwen, J.D., and E.H. Vernot. 1985. Toxic Hazards Research Unit Annual Report: 1985. AAMRL-TR-85-058. ADA161558. Aerospace Medical Research Laboratory, Wright-Patterson Air Force Base, OH. | ||||
| Mattie, D.R., C.L. Alden, T.K. Newell, C.L. Gaworski, and C.D. Flemming. 1991. A 90-day continuous vapor inhalation toxicity study of JP-8 jet fuel followed by 20 or 21 months of recovery in Fischer 344 rats and C57BL/6 mice. Toxicol. Pathol. 19(2):77-87. | ||||
| Rossi, J., III, A.F. Nordholm, R.L Carpenter, G.D. Ritchie, and W. Malcomb. 2001. Effects of repeated exposure of rats to JP-5 or JP-8 jet fuel vapor on neurobehavioral capacity and neurotransmitter levels. J. Toxicol. Environ. Health A 63(6):397-428. | ||||
| Whitman, F.T., and J.P. Hinz. 2001. Sensory Irritation Study in Mice: JP-4, JP-8, JP-8+100. Report No. IERA RS-BR-SR-2001-0005. ADA398112. Air Force Institute for Environment, Safety and Occupational Health Risk Analysis, Brooks Air Force Base TX. | ||||
| Wolfe, R.E., E.R. Kinkead, M.L. Feldmann, H.F. Leahy, W.W. Jederberg, K.R. Still, and D.R. Mattie. 1996. Acute Toxicity Evaluation of JP-8 Jet Fuel and JP-8 Jet Fuel Containing Additives. AL/OE-TR-1996-0136. NMRI-94-114. Prepared by ManTech Environmental Technology, Inc., Dayton, OH, for Armstrong Laboratory, Occupational and Environmental Health Directorate, Toxicology Division, Wright-Patterson AFB, OH. | ||||
| Test species/Strain/Number: Rat/F344/groups of 5 to 95 | ||||
| Mouse/Swiss-Webster/4 per group | ||||
| 10 min | 30 min | 1 h | 4 h | 8 h |
| 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 | 290 mg/m3 |
| Exposure route/Concentrations/Durations: 3,430-5,000 mg/m3 for 30 min to 4 h (vapor + aerosol); 1,000 mg/m3 for up to 90 days. The exposure at 5,000 mg/m3 involved JP-5, which is similar in composition to JP-8. | ||||
| Effects: | ||||
| 3,430 mg/m3: eye and upper respiratory irritation (Wolfe et al. 1996) | ||||
| 3,565 mg/m3: no clinical signs; respiratory rate depression of 50% (Whitman et al. 2001) | ||||
| 4,440 mg/m3: eye and upper respiratory irritation (Wolfe et al. 1996) | ||||
| 5,000 mg/m3: eye irritation and signs of CNS depression (MacEwen and Vernot 1985; Mattie et al. 1991) | ||||
| 1,000 mg/m3: no severely adverse effects (Briggs 2001; Rossi et al. 2001; and others) | ||||
| End point/Concentration/Rationale: Weight of evidence from all studies; escape might be impeded at the highest concentration of 5,000 mg/m3. | ||||
| Uncertainty factors/Rationale: | ||||
| Total uncertainty factor: 3 applied to concentrations of 3,430-5,000 mg/m3; 1 applied to 1,000 mg/m3 | ||||
| Interspecies: 1 applied to all concentrations; rodent uptake is greater than human uptake based on higher respiratory rate and cardiac output and higher blood:air partition coefficients. | ||||
| Intraspecies: 3 applied to 3,430-5,000 mg/m3 (1,100 mg/m3); no susceptible populations identified; upper respiratory sensory irritation and threshold for CNS effects do not differ by more than 3-fold in the general population; 1 applied to repeated exposures of 1,000 mg/m3 because no adverse health effects identified. | ||||
| Modifying factor: Not applied | ||||
| Animal-to-human dosimetric adjustment: Not applied | ||||
| Time-scaling: Not applied; CNS depression is a concentration threshold effect. The repeated nature of many of the studies ensures the safety of a single exposure. | ||||
| Data adequacy: Robust database of inhalation and oral studies in rodents that addressed irritation, neurotoxicity, immunotoxicity, developmental and reproductive effects, genotoxicity, and carcinogenicity. Human exposures to JP-8 were limited to occupational exposures; monitoring studies with other aviation fuels showed few effects. | ||||
AEGL-3 VALUES
| 10 min | 30 min | 1 h | 4 h | 8 h |
| Not | Not | Not | Not | Not |
| determined | determined | determined | determined | determined |
| Data adequacy: There are no data on lethal concentrations. | ||||




































































