THE NATIONAL ACADEMIES
Advisers to the Nation on Science, Engineering, and Medicine
| National Research Council Division on Earth and Life Studies Board on Life Sciences |
500 Fifth Street, NW Washington, DC 20001 |
September 16, 2011
Major General James K. Gilman
Commander
U.S. Army Medical Research and Materiel Command
504 Scott Street
Fort Detrick, MD 21702-5012
Dear Major General Gilman:
At the U.S. Army’s request (pursuant to Contract No. W81K04-06-D-0023 [CLIN 3005]), the National Research Council (NRC) established the Committee to Review Risk Assessment Approaches for the Medical Countermeasures Test and Evaluation (MCMT&E) facility at Fort Detrick, in Frederick, Maryland. The committee was charged with reviewing a proposed approach to preparing a risk assessment for the new biocontainment laboratory at the base. Enclosed is the committee’s second letter report on the Army contractor’s proposed work plan for conducting the risk assessment.
Sincerely,

Charles N. Haas, Ph.D.
Chair, Committee to Review Risk Assessment
Approaches for the Medical Countermeasures Test and
Evaluation Facility at Fort Detrick, Maryland
INTRODUCTION
This letter report reviews a work plan for conducting a site-specific risk assessment (SSRA) for the Army’s Medical Countermeasures Test and Evaluation (MCMT&E) facility at Fort Detrick in Frederick, Maryland. The development of the work plan was informed by findings and recommendations made by this committee (see Attachment A for roster and biographies) in a letter report issued in May 2011, who commented on proposed approaches for conducting the SSRA (NRC 2011a). The following background on the request for these reviews is excerpted from the committee’s first letter report.
The U.S. Army Medical Research and Materiel Command (USAMRMC) plans to construct and operate a new Medical Countermeasures Test and Evaluation (MCMT&E) facility at Fort Detrick in Frederick, Maryland. The proposed site of the 492,000-square-foot facility is on the north side of the fort’s National Interagency Biodefense Campus.1 The facility will be designed to handle infectious agents that are considered Category A and Category B under the Centers for Disease Control and Prevention schedules and that require safety precautions to the extent of animal biosafety level-3 (ABSL-3) and ABSL-4 and biosafety level-3 (BSL-3) and BSL-4. Researchers at the facility will develop new vaccines and drugs against such pathogens as Ebola virus and Bacillus anthracis. The laboratories will be equipped to support nonhuman primate studies and have modern aerobiology and telemetry (remote monitoring) capabilities. Research with rodents will also be conducted.
An environmental impact statement (EIS) is currently being developed by an Army contractor for the MCMT&E facility. EISs are documents required under the National Environmental Policy Act (NEPA) of 1969 to identify and characterize the probable environmental impacts from programs and actions of the federal government. Human health effects are one of the many impacts considered in EISs. Agencies with biocontainment laboratories have struggled with approaches to conducting human health risk assessments, particularly because there is no generalizable framework that can be applied to assessing the specific risks from such laboratories. Recent reviews conducted by the National Research Council (NRC) of risk assessments performed to support the construction of biocontainment facilities have identified weaknesses in both the process and technical content of the assessments by other agencies and provide guidance for improvements (NRC 2007, 2008, 2010a,b,c,d).
In 2010, an NRC committee evaluated the health and safety risks of another Fort Detrick facility with high-containment laboratories—the U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID). The evaluation included a review of a health hazard assessment for the new USAMRIID laboratories, as well as procedures and regulations for their operation. The committee found that the hazard assessment failed to provide adequate and credible technical analyses of the potential health risks to the general public. The Army was advised to improve its risk-assessment practices for infectious agents in future EIS processes and products (NRC 2010a). Thus, to support the EIS being developed for the new MCMT&E facility, the Army requested a review of its site-specific risk-assessment (SSRA) plans for the MCMT&E facility.
CHARGE TO THE COMMITTEE AND ITS APPROACH
The committee was tasked with reviewing and providing technical input to a new environmental impact statement (EIS) to be prepared for the Medical Countermeasures Test and Evaluation (MCMT&E)
________________
1 Other facilities that comprise the National Interagency Biodefense Campus include the U.S. Army Medical Research Institute of Infectious Diseases, the Department of Homeland Security’s National Biodefense Analysis and Countermeasures Center, and the National Institute of Allergy and Infectious Diseases’ Integrated Research Facility.
facility. This facility is intended to be built and operated on area A of Fort Detrick. Technical input may include, but may not be limited to, a proposed work plan for preparing risk assessments as well as information on the selection of agents, scenarios, and models to be used in the risk assessments. The committee may also be asked to review preliminary model results for the quantitative risk assessments and any qualitative assessments developed where data may be insufficient for quantitative modeling. The committee will not perform an independent evaluation of the safety of the MCMT&E facility or the EIS as a whole, but will restrict its findings to assessing the adequacy and validity of the proposed risk assessment methodology and the draft results of any assessment to be incorporated into the EIS.
The Army originally expected that the committee would review a completed SSRA as its second and final task. However, on the basis of the recommendations of the committee’s first report, the Army decided to have the committee review a formal work plan for the SSRA so that the Army could obtain further guidance before the SSRA is performed. The committee held two meetings to address this task. One meeting was held on May 18, 2011, to obtain input from members of the Containment Laboratory Community Advisory Committee2 and the general public. Another meeting was held on July 25-26, 2011, at which the Army’s contractor BSA Environmental Services, Inc., presented a work plan for conducting an SSRA for the MCMT&E facility (see Attachment B). The findings and recommendations in this letter report reflect the consensus of the committee, and the report was reviewed in accordance with standard NRC review procedures (see Attachment C). Some aspects of the work plan were vague; for example, the methods for conducting certain types of analyses were not specified. Because of this, some of our recommendations are framed broadly. In these cases, we have tried to provide citations to relevant references to case studies, guidance documents for certain analytic approaches, and other NRC reports that have evaluated similar types of risk assessments.
OVERARCHING FINDINGS
The committee first compliments BSA Environmental Services, Inc., and its team for considering elements from the first letter report (NRC 2011a) in the development of its work plan. The work plan provides a better overall description of the scope and plans for conducting the risk assessment than that of the previous plan. In particular, the work plan includes helpful illustrations of what exposure pathways will be considered in the risk assessment for specific agents and outlines steps for performing qualitative and quantitative analyses.
The purpose of any risk assessment is to help inform decision makers and stakeholders about the consequences of a particular set of decisions and scenarios. “Risk assessment should be viewed as a method for evaluating the relative merits of various options for managing risk rather than as an end in itself" (NRC 2009, p. 5). However, the objective stated in the submitted work plan is “to document the likelihood, adverse consequences, and uncertainty of reasonably foreseeable events that can affect the health of people working in and around the laboratory as well as members of the community” (BSA Environmental Services, Inc., 2011, p. 1). The committee believes that an assessment should be viewed not merely as a regulatory hurdle to be overcome but also as a tool to be used to enable the most desirable alternative or policy to be implemented. In this regard, the separation of the risk assessment from facility design reduces the overall usefulness of the assessment to decision making. In the absence of one or more actual alternatives for a facility, it should not be assumed that any SSRA would have validity under all alternatives that might be considered in an environmental impact statement.
The work plan focuses on the direct consequences of a release event from the proposed laboratory. Heavy reliance is placed on examination of retrospective reports of laboratory-acquired
________________
2The Containment Laboratory Community Advisory Committee was formed by the Frederick Board of County Commissioners and the City of Frederick to foster communication between the public and operators of containment laboratories at Fort Detrick and elsewhere in Frederick, Maryland.
infections (LAIs). There are limitations to this approach (Singh 2009). Looking retrospectively at historical events is important but cannot assess events that are of low likelihood (but possibly of higher consequence) that have not (yet) been observed. A robust risk assessment should proactively assess the likelihood and consequence of potential events. A variety of tools are available to do that, and some are discussed in detail below. It also would be prudent to consider to the extent possible the potential indirect consequences of a release.
The committee finds that the methodology of the SSRA is not sufficiently robust to assist the Army in designing a facility that will reduce the risk from potential hazards from the facility’s operations. By improving its approach, the Army can ensure the value, timeliness, and credibility of the risk assessment and develop a facility that will protect human health, the environment, and cultivate public confidence.
SCOPING AND PROBLEM FORMULATION
Agents under Consideration
The SSRA work plan proposes to review six agents or groups of agents:
- Bacillus anthracis
- Brucella spp.
- Ebola and Marburg viruses (viral hemorrhagic fevers)
- Francisella tularensis
- Venezuelan, eastern, and western equine encephalitis viruses
- Yersinia pestis
The committee was told that there are current plans for research on these agents at the proposed facility. The agents may not, by definition, be considered a representative list of all pathogens that may ultimately be investigated at the facility. Furthermore, the work plan did not address the committee’s previous recommendation that countermeasures also be included in the SSRA (e.g., vaccinia-based vaccines, antibacterial or antiviral drugs). For this reason, the first letter report questioned the decision to exclude dry-use scenarios (NRC 2011a). We continue to question this exclusion in the work plan when vaccinia virus will be used as a vector for vaccine delivery. Some vaccines are manufactured in lyophilized or freeze-dried formulations (e.g., Kastenmuller et al. 2009), and countermeasure testing would include evaluating the efficacy of such formulations. See Communication section below for discussion of the importance of clarifying who will be responsible for conducting and approving risk assessments of other agents when they are added to the research portfolio.
Range of Validity Because Facility Has Not Been Designed
In the first letter report, this committee stated that “a sufficiently detailed understanding and characterization of the design and engineering controls of the facility are needed to perform the SSRA adequately. In this case, because the facility has not yet been designed, it may be premature to scope the SSRA fully. It is conceivable for the scoping to be done in parallel with the design of the facility; however, the relative time lines for these two processes were not clear from the briefing” (NRC 2011a).
It remains the case that the facility has not yet been through the conventional infrastructure design phase, yet the SSRA is moving ahead. Therefore, the SSRA needs to be fully transparent about the assumptions being made in regard to facility design (especially in light of possible changes in size and scope [Eckstein 2011]), construction, and operation. The scope should incorporate the key elements and
assumptions with respect to the facility design and associated value engineering that may influence risk to workers and offsite populations. Substantial differences in design that have the potential to increase risk may require a supplemental risk assessment.
End Points Considered
The previous letter report recommended a clear delineation of the metrics or end points that will be used for risk characterization in the SSRA. The work plan describes all scenarios as having one or more end points, which are no adverse effect, illness, and mortality. These end points are considered direct consequences from a particular scenario (e.g., accidental exposure in a laboratory); however, other indirect consequences may occur as a result of an adverse event, which include economic, social, and healthcare impacts. For example, the 2001 anthrax letter attacks had the direct impact of killing five people and infecting 17 others. However, the indirect impact was that over 30,000 people received prophylactic treatment and many buildings were decontaminated, all at a cost of over $1 billion (CDC 2001; NRC 2011b). Other SSRAs of proposed biocontainment facilities have assessed indirect consequences of an adverse event. For example, the Department of Homeland Security estimated that the economic impact of an outbreak of disease from a laboratory release of the foot-and-mouth-disease virus from the National Bio- and Agro-Defense Facility, if it were to be located in Manhattan, Kansas, could be $9-$50 billion. The NRC committee that reviewed that SSRA estimated that the risks and costs could be even larger (NRC 2010c). These examples highlight the importance of incorporating other metrics when doing consequence analysis. Therefore, the committee recommends estimating the potential indirect and direct impacts for all scenarios to the extent possible.
Assessment of Evidence
The work plan does not adequately address the previous letter report’s recommendations for assessing risks from LAIs. Specifically, the work plan fails to consider the full range of potential occupational exposures. The Centers for Disease Control and Prevention (CDC) reports 395 cases of potential release events at national laboratories working with select agents (see Table 1). Seven LAIs were reported to CDC; four infections involved Brucella melitensis, two involved Francisella tularensis, and one involved an unspecified Coccidioides species (NRC 2011c). CDC plans to publish an analysis of these events.
TABLE 1 Activity Resulting in Potential Release of Select Agents, 2003-2009
| Activity | No. Potential Release Events |
| Animal bite or scratch | 11 |
| Needlestick or sharps injury | 46 |
| Equipment mechanical failure | 23 |
| Personal protective equipment failure | 12 |
| Loss of containment | 196 |
| Procedural issue | 30 |
| Spill | 77 |
| Total release events | 395 |
| SOURCE: CDC, unpublished material, Nov. 2010, as cited by NRC (2011c). | |
The accidental release data show the potential for exposure by accident to occur. Fort Detrick has kept records and published peer-reviewed papers on LAIs (e.g., Rusnak et al. 2004a, b, c). These and other detailed incident reports should be evaluated to determine failure modes and rates, which can inform
a formal failure analysis for assessment of exposure likelihoods and frequencies in a formal risk assessment. Failure analysis involves the systematic evaluation of engineering, procedural, and process failures that could lead to the release of a pathogen from the laboratory.
The work plan addresses infections among laboratory workers and their primary contacts. Data on LAIs in administrative, maintenance, and waste-handling staff, for example, should also be analyzed. Example references include Meyer and Eddie (1941) and Fiori et al. (2000).
Exposures have the potential to occur outside the laboratory. Therefore, failure-mode analysis should be conducted on agent transport, biohazard waste transport, treatment, storage, and disposal, recognizing the scale of work contemplated at the facility and the differences in infrastructure design, the changes made during the value-engineering phases, and the final facility “as built” and operated. The committee recognizes that failure analysis itself is imperfect; however, it is perhaps most useful in assessing the relative importance of design and operational decisions to performance and is preferable to ad hoc empirical judgments (Seife 2003).
Data Quality Standards for Literature Used
A retrospective historical analysis forms the basis of much of the proposed risk assessment approach. The systematic review required to undertake such an approach is extensive (Higgins and Green 2011). The work plan should include procedures for obtaining data and information and criteria for selecting data for inclusion. For example, the preamble to the White House Office of Management and Budget (OMB) Guidelines for Ensuring and Maximizing the Quality, Objectivity, Utility, and Integrity of Information Disseminated by Federal Agencies states that
‘agencies shall have a basic standard of quality (including objectivity, utility, and integrity) as a performance goal’…. [OMB] note[s], in the scientific context, that in 1996 the Congress, for health decisions under the Safe Drinking Water Act, has already adopted a basic standard of quality for the use of science in agency decisionmaking. Under 42 U.S.C. 300g-1(b)(3)(A), an agency is directed, ‘to the degree that an Agency action is based on science,’ to use ‘(i) the best available, peer-reviewed science and supporting studies conducted in accordance with sound and objective scientific practices; and (ii) data collected by accepted methods or best available methods (if the reliability of the method and the nature of the decision justifies use of the data).’ [OMB] also note[s] that the OMB guidelines call for an additional level of quality ‘in those situations involving influential scientific or statistical information.’ The additional level of quality concerns a standard of care for scientific or statistical analytical results, a ‘capable of being a substantially reproduced’ standard… (OMB 2001).
In this way, the risk assessment reports can be further scrutinized for data quality, bias, and assumptions. Of the 22 references cited in the work plan, eight were not peer-reviewed and three were not appropriately referenced (e.g., symposium reports, institutional reports, professional society newsletters, and Web sites). Recognizing that this document is the work plan, the committee urges the SSRA to be more scrupulously researched and documented.
Definition and Boundaries of the Problem Scope
More precision is needed in defining the terms plausible, likely, and reasonably foreseeable. A clear statement is also needed on the boundaries of the scope of the problem, which hinges on how the three terms are defined. In the text of the work plan, the terms implausible and unlikely were used synonymously; therefore, an event deemed unlikely would not be further evaluated.
The term implausibility is a descriptor based on existing knowledge of biological and physical processes as well as design features of the laboratory. The term unlikely is a metric based on a probability (unlikely adverse events have a low probability of occurring). Consistent with the work plan, it is a reasonable decision to not explore implausible routes of transmission; however, unlikely risk scenarios should not be excluded from the assessment. The work plan describes an unlikely event as one that has no historical data that it occurred; however, not all events that have not yet been experienced are unlikely. Unlikely events are not impossible, and it is the unprecedented event that often has resulted in severe outcomes.
On the basis of a more explicit definition of likelihood and plausibility, the work plan also should contain a more precise definition of a reasonably foreseeable scenario. A statement of how these scenarios will be developed would provide more clarity on the scope of the risk assessment. For example, these scenarios could be developed in a way that provides the framework for a formal failure analysis, an analysis that the committee recommended as a major component of the risk assessment (NRC 2011a). The committee’s view on the need to include failure analysis in the risk assessment is discussed further below.
Scenarios (Pathogen Maps)
The committee appreciates the convenience and creativity of the “pathogen maps,” adapted from NRC (2009), provided within the work plan (for F. tularensis) and as appendixes for the other five organisms or class of organisms. The end point of all risk assessments in this project are human infections, and the utility of these maps is that they provide an overview of all possible routes of infection, from the initial event (accidental exposures in the laboratory and accidental or intentional releases from the laboratory) to the final possible end points (no adverse effect, illness, or mortality). The approach taken in the work plan appears to be that if a pathway of exposure has been documented in the past, it is analyzed as an exposure pathway; pathways that have not been documented are excluded. The committee recommends the opposite approach of assuming that all pathways are potential routes of exposures and then assessing whether there is justification and affirmative documentation to rule that linkages to the potential routes are implausible and then to remove them. Specifically, an exposure pathway may be excluded because of a lack of information that a causal linkage is plausible or because sufficient information exists that a linkage is implausible. The decision process for construction of these maps should be fully documented with appropriate references. Producing a final map in this way will ensure that due diligence was used to analyze routes of infection or release.
The maps also should include all plausible sources and routes of exposure. For example,
- Waste streams from laboratory and animal studies.
- Accidental contamination of the ecosystem resulting in formation of a reservoir for infection (e.g., soil and water).
- Wild animals/insects entering the facility and coming into contact with agents and waste products. For example feral rodent penetration may explain outbreaks of hantavirus in animal colonies in Belgium (Desmyter et al. 1983), France (Dournon et al. 1984), Japan (Umenai et al. 1979; Kawamata et al. 1987), the United Kingdom (Lloyd et al. 1984; Lloyd and Jones 1986), and Singapore (Wong et al. 1988).
DECIDING ON ANALYSIS APPROACH
(QUALITATIVE/QUANTITATIVE DECISION MAKING)
The work plan lays out a two-tier process of conducting a risk assessment for scenarios, indicating that an initial qualitative risk assessment may be followed by a quantitative assessment. The work plan does not adequately describe the criteria that will be used to decide whether a quantitative assessment will be performed. In general, quantitative risk assessment is the preferable approach even in the presence of large data gaps:
Many factors may influence the decision to conduct a qualitative versus a quantitative risk assessment. Obviously, if no data are available to make inferences from, then a quantitative risk assessment would not be possible. Constraints in data quality, time, personnel, or resources may not permit a full quantitative risk assessment. However, data gaps are not necessarily a barrier to quantitative risk assessment. Our bias has been towards ‘Letting the data speak!’, using thorough data analysis, formal inferencing, and striving for complete documentation of variability and uncertainty” (Coleman and Marks 1999, p. 290, emphasis added)
The decision not to perform a quantitative assessment should be explicitly justified in every case. Specifically, rigorous metrics based on peer-reviewed information should be formalized when deciding the appropriate approach. Decisions with respect to deliberately induced events could be informed by risk assessment approaches developed for such incidents. Several approaches used by the Department of Homeland Security were recently reviewed by the NRC (2010e), including the Terrorism Risk Assessment and Management risk assessment (a software-based tool for performing terrorism-related relative risk analysis). Although the NRC report concluded that the model remains to be validated, it found the approach provides a structured process for conceptualizing and ranking the spectrum of risks.
CONDUCT OF THE QUALITATIVE RISK ASSESSMENT
The approach to the qualitative risk assessment in the work plan is not described in sufficient detail to be evaluated by a practitioner versed in risk assessment. The relevance of Figure 2 in the work plan to qualitative risk assessment is unclear. It does not appear that standard qualitative risk assessment approaches will be used. For example, a two-dimensional outcome matrix consisting of likelihood of hazard and magnitude of consequence is a common approach (Cox et al. 2005).
If this methodology is used, the following questions apply:
- How many categories in each dimension will be used?
- What are the criteria for assigning a likelihood value and a consequence value to a scenario?
- How will the output of this qualitative assessment be used?
The work plan should include clear statements on the precise format and techniques of a qualitative risk assessment, as well as its goals and outcomes.
CONDUCT OF THE QUANTITATIVE RISK ASSESSMENT
The goals of a quantitative risk assessment are to determine with specificity the extent and magnitude of adverse consequences associated with a policy option, scenario or decision. This
determination may be done as a point estimate or with the use of probabilistic (Burmaster and Anderson 1994), interval (Ferson and Ginzburg 1996), or other methods to determine the uncertainty of such estimates. The value of quantitative risk assessment is determined by the quality of the uncertainty analysis. Uncertainty estimation helps to ascertain which inputs are the most significant sources of uncertainty; these sources would be key targets for further data-gathering efforts.
The work plan introduces the terms “coarse grain” and “fine grain” in the discussion of quantitative risk assessment. These terms are unnecessarily confusing. There are tiered approaches of sophistication to the execution of a risk assessment, particularly with respect to uncertainty and variability analyses (see NRC 2009, 2010e), and alternative approaches have been reviewed (Paté-Cornell 1996). Figure 1 shows the spectrum of traditional risk analysis methods. The SSRA work plan should adopt more conventional risk assessment terms and should also indicate the criteria by which higher tiers of analyses might be used. For example, a lower-tier analysis (e.g, a point estimate or an exposure estimate based on lumped parameter models) that indicates the existence of a risk might justify a higher-tier analysis (e.g., one- or two-dimensional Monte Carlo analysis or spatial discretized transport models), which could identify the key variables that dominate the uncertainty in the risk estimate.
The use of quantitative risk assessment requires inputs on exposure and dose response. In the context of the MCMT&E facility, because the building is not yet designed, the most appropriate method to estimate exposure extents and likelihoods (which would be needed as inputs to quantitative risk assessment) would be failure analysis. Failure analysis is used in a number of settings in which complex facilities, such as chemical-process, nuclear-power, and space industries, are analyzed (Garrick 1988; Apostolakis and Kafka 1992; Kumamoto and Henley 1996). The committee believes that this method would be eminently appropriate for the MCMT&E facility. One advantage of using failure analysis is that information on other facilities that have common equipment or failure modes (e.g., failure of a waste backflow valve) could be used to inform the process.
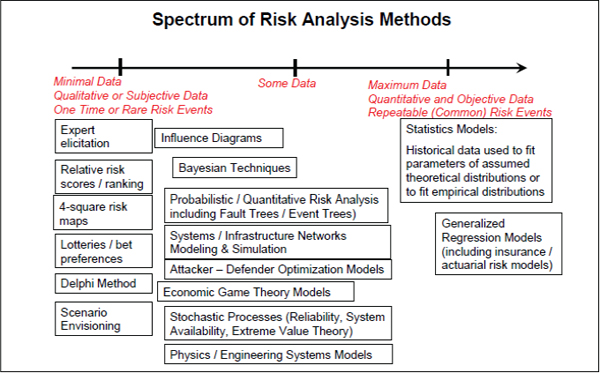
FIGURE 1 Spectrum of risk analysis methods. Source: NRC 2010e.
In the event that dose-response information for a particular pathogen of interest is not available, there are several approaches that can be used. While recognizing the limitations, one common approach is to use an organism that is considered to have similar characteristics; for example, when Escherichia coli O157:H7 rose to concern in food and water, the dose-response relationship for Shigella was used as a proxy (due to O157:H7 having common pathogenicity characteristics) (Duffy et al. 2006). Another approach is to use as an extreme the limiting upper bound of an exponential dose-response curve that has an individual organism survival probability of 1 as the upper possible limit (Teunis and Havelaar 2000).
COMMUNICATION
There is a great deal of concern among local residents that releases of biological agents (e.g., agents classified by CDC as Category A) from the research programs at Fort Detrick may lead to disease outbreaks in the surrounding community. Therefore, the committee compliments BSA Environmental Services, Inc., for its plans to devote a large amount of effort to address community concerns through risk communications. As stated in the previous letter report, the committee again acknowledges the Army’s cooperation with the Containment Laboratory Community Advisory Committee (CLCAC). Members of the committee who met with representatives of the CLCAC have seen how direct, informal interchange improves public understanding of the work of Fort Detrick’s biocontainment laboratories (information on the activities of the CLCAC is available at http://www.cityoffrederick.com/cms/page/index.php?id=547).
However, the committee urges the Army to go beyond risk communications and undertake genuine two-way community engagement, in which the general public, including the CLCAC, has the opportunity to identify, during the Army’s and BSA Environmental Services’s initial study phase, potential failure scenarios. Not only would such input improve the quality and completeness of the risk assessment, but it is also likely to strengthen community acceptance of the SSRA’s findings. Risk communication should also include discussion of risk management plans that will minimize risk of a release event, such as proper training of laboratory workers. Although LAIs will always be a risk, it has been documented that cases have been substantially reduced with improvements in biosafety practices and training (e.g., see NRC 2010a, 2011c).
Communications should go beyond the explanation of risk and risk scenarios. The Army should explain exactly how risk findings will be incorporated into the design, construction, and operation of the MCMT&E facility. For example, the Army should describe how it will meet its obligations, under the National Environmental Policy Act, to evaluate genuine alternatives. The SSRA should clearly state the assumptions with respect to planned design and operation, including security and safety systems. It will be important to convey that the goal is not risk elimination but reducing the risk to an acceptable and manageable level and developing appropriate incident response plans for the acceptable risks. Furthermore, emphasizing the distinction of likelihood versus consequences in the assessment will help to place the SSRA in context, particularly because the facility design is only beginning. Almost all facility design elements focus on reducing the likelihood of adverse accidental or deliberate scenarios, not on the consequences. A scenario that is assessed to be of high risk but is high due to the potential consequences may not be as relevant for facility design considerations but would still be an important factor for consideration in the EIS.
If additional pathogens and countermeasures—beyond those reviewed in the initial risk assessment—are proposed for testing at the MCMT&E facility, who will be responsible for conducting and approving additional risk assessments? The committee has been told that MCMT&E’s Institutional Biosafety Committee (IBC) may be responsible for reviewing and approving studies of organisms beyond those addressed in the initial risk assessment. If so, it is important that the general-public members on the IBC be identified publicly and be easily accessible to the public. The IBC should explain its charter, authority, and functions to the CLCAC.
In its previous letter report, the committee urged the Army to improve its communications with local doctors and hospitals, particularly to help community-based clinicians diagnose and treat unusual
infectious diseases. Given the possibility that some of the pathogens to be tested on MCMT&E animals might spread to household pets and farm animals in the area, it might also be advisable that local veterinarians receive training in the identification of such unusual infections among their animal patients.
In summary, the MCMT&E facility’s communications with the public should go beyond reassurances that people are unlikely to get sick because of its operations. The Army should make it clear that it will listen to the public’s concerns and use its risk findings to make the laboratory safer.
REFERENCES
Apostolakis, G., and P. Kafka. 1992. Advances in probabilistic safety assessment. Nucl. Eng. Des. 134(1):141-148.
BSA Environmental Services, Inc. 2011. Work Plan: Site-Specific Risk Assessment for the Medical Countermeasures Testing and Evaluation Facility at Fort Detrick in Frederick County, Maryland. Final Report, July 15, 2011. BSA Environmental Services, Inc., Beachwood, OH.
Burmaster, D.E., and P.D. Anderson. 1994. Principles of good practice for the use of Monte Carlo techniques in human health and ecological risk assessment. Risk Anal. 14(4):477-481.
CDC (Centers for Disease Control and Prevention). 2001. Ongoing Investigation of Anthrax—Florida, October 2001. MMWR 50(40):877.
Coleman, M.E., and H.M. Marks. 1999. Qualitative and quantitative risk assessment. Food Control 10(4-5):289-297.
Cox, L.A., Jr., D. Babayev, and W. Huber. 2005. Some limitations of qualitative risk rating systems. Risk Anal. 25(3):651-662.
Desmyter, J., K.M. Johnson, C. Deckers, J.W. LeDuc, F. Brasseur, and C. Van Ypersele de Strihou. 1983. Laboratory rat associated outbreak of haemorrhagic fever with renal syndrome due to Hantaan-like virus in Belgium. Lancet 322(8365):1445-1448.
Dournon, E., B. Moriniere, S. Matheron, P.M. Girard, J.P. Gonzales, F. Hirsch, and J.B. McCormick. 1984. HFRS after a wild rodent bite in the Haute-Savoie-and risk of exposure to Hantaan-like virus in a Paris laboratory. Lancet 323(8378):676-677.
Duffy, G., E. Cummins, P. Nally, S. O’Brien, and F. Butler. 2006. A review of quantitative microbial risk assessment in the management of Escherichia coli O157:H7 on beef. Meat Sci. 74(1):76-88.
Eckstein, M. 2011. Army May Downsize Advanced Research Testing Center. Frederick News Post.com, July 21, 2011 [online]. Available: http://www.fredericknewspost.com/sections/news/display.htm?storyid=123922 [accessed Aug. 4, 2011].
Ferson, S., and L.R. Ginzburg. 1996. Different methods are needed to propagate ignorance and variability. Reliab. Eng. Syst. Safe. 54(2-3):133-144.
Fiori, P.L., S. Mastrandrea, P. Rappelli, and P. Cappuccinelli. 2000. Brucella abortus infection acquired in microbiology laboratories. J. Clin. Microbiol. 38(5):2005-2006.
Garrick, B.J. 1988. The approach to risk analysis in three industries: Nuclear power, space systems, and chemical process. Reliab. Eng. Syst. Safe. 23(3):195-205.
Higgins, J.P.T., and S. Green, eds. 2011. Cochrane Handbook for Systematic Reviews of Interventions, Version 5.1.0 (updated March 2011). The Cochrane Collaboration [online]. Available: http://www.cochranehandbook.org./ [accessed August 4, 2011].
Kastenmuller, W., G. Gasteiger, L. Stross, D.H. Busch, and I. Drexler. 2009. Cutting edge: mucosal application of a lyophilized viral vector vaccine confers systemic and protective immunity toward intracellular pathogens. J. Immunol. 182(5):2573-2577.
Kawamata, J., T. Yamanouchi, K. Dohmae, H. Miyamoto, M. Takahaski, K. Yamanishi, T. Kurata, and H.W. Lee. 1987. Control of laboratory acquired hemorrhagic fever with renal syndrome (HFRS) in Japan. Lab. Anim. Sci. 37(4):431-436.
Kumamoto, H., and E.J. Henley. 1996. Probabilistic Risk Assessment and Management for Engineers and Scientists, 2nd Ed. New York, NY: Institute of Electrical and Electronics Engineers, Inc.
Lloyd, G., and N. Jones. 1986. Infection of laboratory workers with hantavirus acquired from immunocytomas propagated in laboratory rats. J. Infect. 12(2):117-125.
Lloyd, G., E.T. Bowen, N. Jones, and A. Pendry. 1984. HFRS outbreak associated with laboratory rats in UK. Lancet 323(8387):1175-1176.
Meyer, K.F., and B. Eddie. 1941. Laboratory infections due to Brucella. J. Infect. Dis. 68(1):24-32.
NRC (National Research Council). 2007. Technical Input on the National Institutes of Health’s Draft Supplementary Risk Assessments and Site Suitability Analyses for the National Emerging Infectious Diseases Laboratory, Boston University: A Letter Report. Washington, DC: The National Academies Press.
NRC (National Research Council). 2008. Technical Input on Any Additional Studies to Assess Risk Associated with Operation of the National Emerging Infectious Diseases Laboratory, Boston University: A Letter Report. Washington, DC: The National Academies Press.
NRC (National Research Council). 2009. Science and Decisions: Advancing Risk Assessment. Washington, DC: The National Academies Press.
NRC (National Research Council). 2010a. Evaluation of the Health and Safety Risks of the New USAMRIID High-Containment Facilities at Fort Detrick, Maryland. Washington, DC: The National Academies Press.
NRC (National Research Council). 2010b. Continuing Assistance to the National Institutes of Health on Preparation of Additional Risk Assessments for the Boston University NEIDL, Phase 1. Washington, DC: The National Academies Press.
NRC (National Research Council). 2010c. Evaluation of a Site-Specific Risk Assessment for the Department of Homeland Security’s Planned National Bio- and Agro-Defense Facility in Manhattan, Kansas. Washington, DC: The National Academies Press.
NRC (National Research Council). 2010d. Continuing Assistance to the National Institutes of Health on Preparation of Additional Risk Assessments for the Boston University NEIDL, Phase 2. Washington, DC: The National Academies Press.
NRC (National Research Council). 2010e. Review of the Department of Homeland Security’s Approach to Risk Analysis. Washington, DC: The National Academies Press.
NRC (National Research Council). 2011a. Assistance to the U.S. Army Medical Research and Materiel Command with Preparation of a Risk Assessment for the Medical Countermeasures Test and Evaluation (MCMT&E) Facility at Fort Detrick, Maryland, A Letter Report. Washington, DC: The National Academies Press.
NRC (National Research Council). 2011b. Review of the Scientific Approaches Used during the FBI’s Investigation of the 2001 Anthrax Letters. Washington, DC: The National Academies Press.
NRC (National Research Council). 2011c. Protecting the Frontline in Biodefense Research: The Special Immunizations Program. Washington, DC: The National Academies Press.
OMB (Office of Management and Budget). 2001. Guidelines for Ensuring and Maximizing the Quality, Objectivity, Utility, and Integrity of Information Disseminated by Federal Agencies. The White House, Office of Management and Budget [online]. Available: http://www.whitehouse.gov/omb/fedreg_final_information_quality_guidelines [accessed July 26, 2011].
Paté-Cornell, M.E. 1996. Uncertainties in risk analysis: Six levels of treatment. Reliab. Eng. Syst. Safe. 54(2-3):95-111.
Rusnak, J.M., M.G. Kortepeter, R.J. Hawley, A.O. Anderson, E. Boudreau, and E. Eitzen. 2004a. Risk of occupationally acquired illness from biological threat agents in unvaccinated laboratory workers. Biosecur. Bioterror. 2(4):281-293.
Rusnak, J.M.. M.G. Kortepeter, R.J. Hawley, E. Boudreau, J. Aldis, and P.R. Pittman. 2004b. Management guidelines for laboratory exposures to agents of bioterrorism. J. Occup. Environ. Med. 46(8):791-800.
Rusnak, J.M., M.G. Kortepeter, J. Aldis, and E. Boudreau. 2004c. Experience in the medical management of potential laboratory exposures to agents of bioterrorism on the basis of risk assessment at the United States Army Medical Research Institute of Infectious Diseases (USAMRIID). J. Occup. Environ. Med. 46(8):801-811.
Seife, C. 2003. Columbia disaster underscores the risky nature of risk analysis. Science 299(5609):1001-1002.
Singh, K. 2009. Laboratory-acquired infections. Clin. Infect. Dis. 49(1):142-147.
Teunis, P. F., and A.H. Havelaar. 2000. The Beta Poisson dose-response model is not a single-hit model. Risk Anal. 20(4):513-520.
Umenai, T., P.W. Lee, T. Toyoda, K. Yoshinaga, T. Horiuchi, H.W. Lee, T. Saito, M. Hongo, and N. Ishida. 1979. Korean haemorrhagic fever in staff in an animal laboratory. Lancet 313(8130):1314-1316.Wong, T.W., Y.C. Chan, E.H. Yap, Y.G. Joo, H.W. Lee, P.W. Lee, R. Yanagihara, C.J. Gibbs, Jr., and D.C. Gajdusek. 1988. Serological evidence of hantavirus infection in laboratory rats and personnel. Int. J. Epidemiol. 17(4):887-890.












