7
Collaborative Models and New Paradigms for Supporting Regulatory Science Research and Practice
Key Messages
• Collaboration involving all stakeholders is a key element to creating a new eco-system that is more efficient at turning discovery into therapeutics that benefit human health.
• Real-world pilot demonstration projects that apply regulatory science to the development of therapeutics can generate important insights while keeping collaborators engaged and committed to their partnerships.
• An initiative to create a self-sustaining Virtual Development Institute comprising a network of university-based organizations could be initiated using funds from existing translational research programs.
• A “champion” for regulatory science could help define and promote the discipline.
Given budgetary constraints that are unlikely to ease in the near future, efforts to develop the discipline of regulatory science and a regulatory science workforce may increasingly seek to rely on indirect support more than direct funding. To explore this issue, the workshop discussed funding opportunities and collaborative models that would need to be available to strengthen and support regulatory science research and practice in therapeutics development. Workshop presentations and discussions also examined whether there are institutions, public or private, that
could offer funding to create an infrastructure and ecosystem to support innovative regulatory science.
Gigi Hirsch, Executive Director, Center for Biomedical Innovation, Massachusetts Institute of Technology (MIT), described a new initiative designed to transform the drug development ecosystem. William Greenlee, President and Chief Executive Officer, the Hamner Institutes for Health Sciences at Research Triangle Park, summarized the Hamner Institutes’ approach to collaborative efforts among government, industry, and academia. Theodore Reiss, Research Professor of Medicine, Vanderbilt University School of Medicine, proposed a new model for research and education that would reflect the needs of today’s world. A discussion followed that explored the resources and stakeholder engagement needed to build the discipline of regulatory science and establish regulatory science career paths.
CREATING A COLLABORATIVE ENVIRONMENT IN AN ACADEMIC SETTING1
In an attempt to repair dysfunction in the drug development ecosystem, MIT created the New Drug Development Paradigms (NEWDIGS) initiative. NEWDIGS is a collaborative environment for innovation and learning that takes a systems approach to transforming processes, technologies, and policy elements of innovation. The stakeholder community, Hirsch said, includes major pharmaceutical companies, the global regulatory community, academia, payers, and patient advocacy groups. A group of MIT faculty serves as strategic advisors to the initiative. NEWDIGS emphasizes tight coordination between real-world pilot projects and academic research in engineering, science, management, and clinical medicine. Lessons learned by NEWDIGS might offer useful ideas and insights for approaches to strengthening a workforce for innovative regulatory science in drug development, said Hirsch.
After identifying a high-impact area of need or opportunity within the innovation space, NEWDIGS convenes a subteam that is interested in this topic and proceeds in a modular fashion (Figure 7-1). The first module takes a regulatory science approach to focus on “adaptive licensing” (including staggered approval, progressive licensing). The second module focuses on oncology, specifically codevelopment of two or more investigational compounds in combination therapies and helping FDA develop a guidance document on the subject. A proposed payer-centric third module would examine value-driven innovation. Within each mod-
________________
1 This section is based on the presentation by Gigi Hirsch, Executive Director, Center for Biomedical Innovation, MIT.
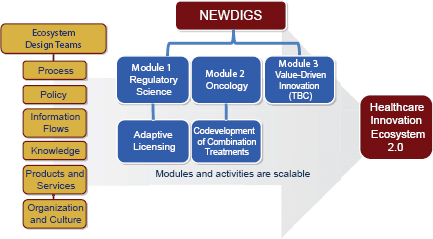
SOURCE: Hirsch, 2011. Presentation at IOM workshop on Strengthening a Workforce for Innovative Regulatory Science in Therapeutics Development.
ule, NEWDIGS researchers build test beds that are designed to combine stakeholders, real-world demonstration projects, and academic research in a way that enables rapid-cycle learning.
NEWDIGS also looks across the modules it has established to find improvements in the architecture of the overall innovation system. The cross-module approach relies on methodologies that focus on preserving value. In this way, everything in the drug development environment enables and contributes to innovation through understanding of how to improve processes, policies, and information flows.
Adaptive Licensing
NEWDIGS has adopted the approach of taking advantage of an opportunity for proactive, strategic design of policy with broad stakeholder input, followed by the empiric evaluation of these designs to inform discussions about change. Regulatory agencies, major pharmaceutical companies, two payers, and researchers from MIT and Harvard are participating in this effort and are designing initial demonstration projects involving compounds in development that will be implemented
in Singapore in collaboration with the Singapore Health Sciences Authority. The group has been using simulations to try to understand what adaptive licensing might look like across different drugs and different drug classes.
Collaboration among the participants is well integrated. Data sharing about drugs currently in the pipeline is undertaken pursuant to formal confidentiality agreements. The work includes an active academic and educational component, including monthly research seminars. Hirsch noted that some 30 students are participating and learning about regulatory science through the project.
Developing a Collaborative Workforce
Hirsch described a regulatory science workforce as a virtually collaborative workforce of individuals from different disciplines and market sectors. She offered the following observations for supporting such a workforce:
• Establish a “safe haven” environment through specific ground rules for workshops as a critical element for enabling learning and innovative thinking. These ground rules include items such as “no decisions are made in workshops” and “individuals do not speak on behalf of their organizations.”
• Systems approaches to design and problem solving are critical but challenging given the enormously complex interdependencies across disciplines and the silos that inhibit communication and sharing. NEWDIGS has been experimenting with a variety of systems engineering methodologies, finding that they are falling somewhat short when they are applied across an entire industry. In addition, a diversity of expertise and perspectives is critical. Thus, it is particularly important to involve individuals from outside of the health care industry.
• Having a safe–haven “test bed” for ideas is important not only for making progress but to fight “consortium fatigue” and to keep participants involved in working on solutions to problems involving regulatory science. Ability to demonstrate that the learning and innovation activities that fuel continuous improvement in execution have been central to the sustainability of NEWDIGS.
A COLLABORATIVE MODEL FOR RESEARCH, TRAINING, AND BUSINESS DEVELOPMENT2
The fundamental philosophical paradigm of the Hamner Institutes is that publication of findings is not the end game of research. Rather, the goal is to capture research knowledge in a way that will improve global public health by driving the development of safer medicines, informing public health policy, and realizing economic development.
The Hamner Institutes’ Institute for Drug Safety Sciences was launched 3 years ago in partnership with the University of North Carolina at Chapel Hill (UNC), and involving faculty from Duke University and North Carolina State University. This new entity operates on the principle that modernizing toxicology is not about hazard identification. Rather, it is about understanding environmental perturbation on biological systems and being able to understand the health outcomes of that perturbation. The institute is developing approaches that link postmarketing surveillance, clinical studies, and advanced animal model systems. The institute is also collaborating with investigators in China to develop virtual models of the Caucasian and Asian liver to understand subtle differences in drug response between these two populations.
The Hamner Institutes is now developing additional partnering relationships that aim to bring academia, industry, and government to the same table as a means of accelerating drug development. These new partnerships will also involve the Hamner Biosciences Accelerator, which has dedicated staff, education and training support through postdoctoral fellowships, and a business development mindset to move discoveries into technology development programs and out into the market. One partnership that has developed out of this effort involves an effort to accelerate drug development in China under U.S. regulatory standards with the goal of bringing products to market worldwide.
REGULATORY SCIENCE: SOLVING FOR A LARGER CONTEXT3
Regulatory science must be addressed within a broader context to address important public health needs and accelerate drug development. This broader context starts with the drug development environment; the pharmaceutical industry is experiencing transformational trends, including very difficult scientific challenges and rapidly increasing expenses and price pressures, while the public sector and academia are developing new skills
________________
2 This section is based on the presentation by William Greenlee, President and Chief Executive Officer, the Hamner Institutes for Health Sciences at Research Triangle Park.
3 This section is based on the presentation by Theodore Reiss, Research Professor of Medicine, Vanderbilt University School of Medicine.
and capabilities for drug discovery and development. Taken together, these trends suggest collaborative solutions for a new and broader educational, scientific, and funding environment. Reiss laid out a set of foundational principles for collaboration around development and regulatory science:
• Addressing issues piecemeal will have limited impact.
• Industry, government, and academia must collaborate to create a vision for a more integrated biomedical science environment.
• Academia must value and promote translational, development, and regulatory thinking, and it must bring industry scientists to the table to help with this transformation.
• The new environment must be efficient and robust and leverage structures already in place, such as the CTSA institutions.
A model organization would be university centered, according to Reiss, and would involve NIH, with a department of “bench-to-bedside science.” The faculty would have expertise in development, regulatory issues, and translation and would serve as a center for collaborative translational projects. These projects would include scientists from other academic departments as well as from industry, regulatory agencies, and foundations. The department would serve as a center to teach translational, development, and regulatory science, instilling a team-oriented, collaborative mindset in students and postdoctoral fellows. It also would be linked across institutions through the CTSAs into what Reiss termed a “Virtual Institute of Drug Development.”
Reiss acknowledged that there are real barriers to creating such an organization. Universities lack exposure to and comfort with this type of broad versus deep thinking. The cognitive framework of academia will need to change from one focused on absolute knowledge to one that considers confidence in benefits and risks based on significant, consistent evidence. The polarization among academia, industry, and the regulatory sector will need to be reduced. The lack of reward structure for translational, development, and regulatory projects is a significant barrier, as is the lack of coordinated teaching, training, and research programs within today’s academic structure.
Funding such an initiative will require a concerted effort from all stakeholders. Reiss suggested that initial funding should come from the proposed NCATS, with universities following by providing matching infrastructure funds, adding that NIH should ensure that there is a balanced portfolio of funding initiatives between discovery and development projects.
Reiss noted that such an initiative would provide a critical signal to investigators regarding potential careers in development, translational, or regulatory science.
Other sources of funds also need to be available, said Reiss. The pharmaceutical industry—both large and small companies—and foundations should contribute through collaborative projects and by participating operationally in the Virtual Development Institute. Though a tax may currently have trouble finding support, Reiss argued that an “approval tax” on drugs, or a revenue tax on sales exceeding $1 billion, should be considered at some point to provide a self-sustaining source of funding.
In a closing panel session, several workshop speakers engaged in a panel discussion with the workshop co-chairs and the workshop participants to discuss resources, stakeholder engagement, and next steps needed to build a discipline of regulatory science and establish career paths in innovative regulatory science. This section lists ideas presented by the panelists and workshop participants. Statements, recommendations, and opinions expressed are those of individual presenters and participants and are not necessarily endorsed or verified by the Forum or the National Academies, and they should not be construed as reflecting any group consensus.
The following are observations by the panelists:
• There is increasing participation by the nonprofit sector to advance the discipline of regulatory science.
• There continues to be a lack of respect within academia for the field of regulatory science.
• There are roles for every sector in the drug discovery ecosystem to participate in the development of a regulatory science workforce.
• Existing programs in regulatory science do not include or emphasize the social sciences.
• Without a separate discipline of regulatory science, it will be difficult to train a workforce with the necessary mindset to understand the needs of regulatory versus discovery science.
________________
4 Participants in the summary panel were Barry Coller, Vice President for Medical Affairs, Physician-in-Chief, and David Rockefeller Professor, The Rockefeller University; Elaine Gallin, Principal, QE Philanthropic Advisors; Steven Galson, Vice President for Global Regulatory Affairs, Amgen Inc.; William Greenlee, President and Chief Executive Officer, the Hamner Institutes for Health Sciences at Research Triangle Park; Gigi Hirsch, Executive Director, Center for Biomedical Innovation, MIT; Carl Peck, Professor of Pharmacology and Medicine, UCSF; Theodore Reiss, Research Professor of Medicine, Vanderbilt University School of Medicine; and Alastair Wood, Partner and Managing Director, Symphony Capital LLC.
• There are more career opportunities and paths for regulatory scientists than might be commonly understood, including international, national, and state regulatory agencies, the Federal Trade Commission, Congress, payers, investors, and journals.
• The collaborative, multidisciplinary nature of innovative regulatory science may undermine the ability to identify a champion or lead stakeholder charged with advancing the science.
• Considering that one goal of the investment in basic health sciences research is to improve health outcomes, investing in regulatory science could support the case for such investment.
The following are suggestions for a way forward from the panelists:
• Create a standing panel sponsored by FDA and/or NIH that would focus on ways to strengthen the regulatory science workforce.
• Ensure that social science research and evidence is built into regulatory science research and practice.
• Identify a champion that can take responsibility for advocacy for support of regulatory science and the workforce to support regulatory science. It was suggested that proposed NCATS could serve as a key champion for the discipline.
• Design regulatory science training programs to reflect the different training backgrounds of the individuals who come to regulatory science. The “menu” approach applied in the European PharmaTrain program could be a good model.
• Convene a series of conferences that would be charged with defining the big needs or “big questions” in regulatory science. Link this effort to RFAs that would support pursuit of these big questions.
• Opportunities to increase interest in the discipline among students and investigators include reinvigorating and expanding research fellowships in clinical pharmacology and embedding regulatory science research fellowships in translational medicine and therapeutics through such programs as the CTSA institutions and the proposed NCATS.
• Researchers who have lost jobs as the pharmaceutical industry has downsized could offer a pool of scientists that could be recruited to do regulatory science in the federal agencies and in academia through innovative and creative pilot projects.
• Make it clear that “if innovation is the goal, regulatory science is essential.”








