7
Infant Outcomes
Because the exclusively breastfed infant is entirely dependent upon the mother for nutrition, the subcommittee examined the evidence relating maternal nutrition to infant health. In addition to nutrients in human milk, it considered constituents that have important nonnutritive functions (see Chapter 6).
As discussed in Chapters 5 and 6, the adequacy of the maternal diet may affect the formation, composition, or secretion of milk. As nutritional demands of the infant increase, milk production becomes correspondingly greater (see Chapter 5). Thus, there is a complex interrelationship between maternal nutrition, volume and composition of the milk, and the vigor of the infant.
Since infant nutrition, growth, development, and health are interrelated, the effects of breastfeeding and maternal nutrition on each of these outcomes were reviewed. The health-related outcomes include resistance to infectious diseases, allergic disorders, and chronic diseases with an immunologic basis that develop later in childhood; the passage of infectious or toxic agents in milk to the recipient; and infant mortality. Because of the specific tasks assigned to the subcommittee, this review was limited to effects on full-term infants.
In reviewing the relevant literature, the subcommittee had to contend with several confounding factors that potentially alter the interpretation of the results. For example, sick infants may be unable to breastfeed because they are separated from their mothers or because they are unable to suckle adequately. In such circumstances, if the mother does not continue lactation by pumping, breastfeeding is difficult to resume if or when the child recovers. Thus, illness may cause the cessation of breastfeeding, rather than the absence of breastfeeding causing illness. In many cultures, there is a strong relationship
between the type of infant feeding and the social status and functioning of the family. In the United States, breastfeeding rates increase with an increase in socioeconomic status. The favorable environment of these women and their infants is associated with a lower risk of many illnesses. In addition, since surveys indicate that the breastfeeding mother is less likely to smoke, her infant is at lower risk of respiratory problems from exposure to passive cigarette smoke. Further, the young infant in day care—often because the mother is working (and therefore less likely to breastfeed)—may be exposed to communicable diseases more often than the infant cared for exclusively at home. Thus, the lower risks of morbidity reported for breastfed infants may be in part due to factors other than breastfeeding. Other potential sources of bias are reviewed by Kramer (1987).
INFANT NUTRITION: VITAMINS AND MINERALS
Human milk serves as the nutritional standard for infants. Certain nutrients (vitamins A, D, K, B12, riboflavin, and folate; iron; copper; zinc; and fluoride) are reviewed in this section to illustrate the uniqueness of human milk and relationships, if any, of the infant's nutritional status to maternal nutrient stores and maternal diet. Other essential nutrients are of no less importance to the infant; information about them is presented in Chapter 6.
Three major factors contribute to the nutritional status of the exclusively breastfed infant: nutrient stores, especially those accumulated in utero; the amount and bioavailability of nutrients supplied by human milk; and environmental and genetic factors that influence the efficiency of nutrient utilization. Nutrient stores at birth are determined by the rate of placental nutrient transfer and by the duration of gestation. The stores of many nutrients increase substantially during the last trimester of pregnancy and tend to be higher in infants with higher birth weight or greater gestational age. The infant's total nutrient intake is determined by nutrient concentrations in human milk and by the volume of milk consumed. The amount of nutrient absorbed by the infant is further influenced by the bioavailability of that nutrient in human milk.
Providing the breastfed infant with supplemental foods has a complex effect on the total amount of nutrient absorbed. For example, infants consuming such foods as formula or infant cereal generally decrease their intake of human milk (see Chapter 5) and, thus, the nutrients and other specialized components it supplies. Thus, the intake of supplementary foods may add nutrients in a less bioavailable form, decrease the bioavailability of nutrients in human milk, and decrease the intake of other important factors in human milk.
Growth, infections, and differences in the efficiency of nutrient utilization affect the infant's rate of nutrient utilization, which in turn can influence the infant's nutritional status. Birth weight is inversely associated with the rate of nutrient utilization. For example, infants who are small at birth usually
experience catch-up growth during infancy. Disease or injury may adversely affect nutrient utilization either directly by increasing rates of catabolism or urinary and fecal losses or indirectly by sequestrating a nutrient in tissue compartments.
Finally, other environmental influences, such as the degree of exposure to ultraviolet light (ordinarily from sunshine) in the case of the synthesis of vitamin D, may be important determinants of the nutritional status of the infant.
Fat-Soluble Vitamins
Vitamin D
Plasma levels of 25-hydroxycholecalciferol (a vitamin D metabolite used as a measure of vitamin D status) in the mother are positively correlated with those in the neonate, providing evidence that maternal vitamin D status affects the infant's vitamin D stores (Hillman and Haddad, 1974; Hoogenboezem et al., 1989; Markestad, 1983; Markestad et al., 1983). Several investigators have found that plasma concentrations of 25-hydroxycholecalciferol in the neonate are within the normal range for adults (10 to 40 ng/ml) (Markestad, 1983; Roberts et al., 1981), whereas Ala-Houhala (1985) reported that 25-hydroxycholecalciferol plasma levels in Finnish neonates were abnormally low in winter (mean, <10 ng/ml) compared with the range of values found in summer (12 to 18 ng/ml, p < .001). These studies suggest that infants born to mothers with inadequate vitamin D status are highly dependent on a regular supply of vitamin D through diet, supplements, or exposure to ultraviolet light.
Plasma 25-hydroxycholecalciferol levels in unsupplemented breastfed infants have been compared with those of formula-fed infants or breastfed infants receiving approximately 10 µg of supplemental vitamin D per day. In six reports, plasma 25-hydroxycholecalciferol levels were substantially lower in unsupplemented breastfed infants (Ala-Houhala, 1985; Chan et al., 1982; Greer et al., 1982; Lichtenstein et al., 1986; Markestad, 1983; Roberts et al., 1981). Four studies (Ala-Houhala, 1985; Greer et al., 1982; Hoogenboezem et al., 1989; Markestad, 1983) have shown plasma levels of this compound at or below the lower limits of normal (≤10 ng/ml) (Nutrition Foundation, 1984) in the unsupplemented groups. Ala-Houhala (1985) reported that in the winter months, 10 of 18 unsupplemented breastfed infants had plasma levels of less than 5 ng/ml, which may lead to rickets (Nutrition Foundation, 1984). Despite reports of rickets in breastfed infants (Arnaud et al., 1976; O'Connor, 1977; Ozsoylu, 1977) and the low vitamin D content of human milk, breastfeeding has long been considered to be protective against rickets (Belton, 1986; Lakdawala and Widdowson, 1977).
Breastfed infants require approximately 30 minutes of exposure to sunlight per week if wearing only a diaper, or 2 hours per week if fully clothed without a hat, to maintain normal serum 25-hydroxycholecalciferol levels (Specker et
al., 1985). Darkly pigmented infants require a greater exposure to sunshine to initiate the synthesis of vitamin D in the skin (Clemens et al., 1982).
In a study by Greer and colleagues (1982), which included randomization of breastfed infants to a placebo or to a daily supplement of 10 µg of vitamin D, the bone mineral content of the placebo group was significantly lower in the first few months after birth but slightly higher than that of the supplemented group by the end of the first year.
In summary, exclusive breastfeeding results in normal infant bone mineral content when maternal vitamin D status is adequate and the infant is regularly exposed to sunlight. If the infant or mother is not exposed regularly to sunlight, or if the mother's intake of vitamin D is low, supplements for the infant may be indicated (5 to 7.5 µg/day).
Vitamin A
Although vitamin A concentrations in human milk are dependent on the mother's vitamin A status, vitamin A deficiency is rare among breastfed infants, even in parts of the world with endemic vitamin A deficiency (Sommer, 1982). Even after breastfeeding is discontinued, it appears to confer a protective effect (Sommer, 1982; West et al., 1986), presumably because some of the vitamin A provided by human milk is stored in the liver. Infants who consume human milk that provides 100 to 151 µg of retinol equivalents per day grow well and do not show signs of vitamin A deficiency. In the United States, human milk provides approximately twice this amount (FAO, 1988; NRC, 1989). These U.S. concentrations are used as the international standard for adequate vitamin A intakes in infancy (FAO, 1988; NRC, 1989). In the United States, there is no indication to routinely supplement either the infant or the mother with vitamin A.
Vitamin K
Vitamin K is essential for the formation of several proteins required for blood clotting. This vitamin has two major forms: vitamin K1 (phylloquinone), synthesized by plants, and vitamin K2 (menaquinone), synthesized by bacteria. Most of the vitamin K in human milk is phylloquinone; the extent to which infants absorb menaquinone produced by gut microflora is not known. Vitamin K stores at birth are extremely low. Therefore, newborns are immediately dependent on an external source of the vitamin, but the amount provided by human milk is low—approximately 2 µg/liter (Committee on Nutrition, 1985; NRC, 1989). Bacteria that colonize the intestinal tracts of breastfed infants produce less menaquinone than do those in formula-fed infants.
A deficiency of the vitamin produces a syndrome in infants called hemorrhagic disease of the newborn. This vitamin K-dependent disease has two different clinical forms. The classic early-onset form occurs at age 2 to 10
days in 1 of 200 to 400 unsupplemented newborns. The late-onset form occurs around 1 month of age in 1 of 1,000 to 2,000 unsupplemented newborns. Late-onset hemorrhagic disease of the newborn is a devastating, often fatal disease (Gleason and Kerr, 1989). Both forms occur more often in unsupplemented breastfed than formula-fed infants.
Although maternal supplementation with vitamin K in the last weeks of pregnancy (Owen et al., 1967) or unusually high milk intakes (>500 ml) during the first 3 days of postnatal life (Motohara et al., 1989) may reduce the risk of hemorrhagic disease of the newborn, the most dependable method of preventing this serious disorder is to inject the infant with 0.5 to 1.0 mg of vitamin K at birth or to give an oral 1.0- to 2.0-mg dose, as recommended by the American Academy of Pediatrics (Committee on Nutrition, 1985) and required by many states.
Water-Soluble Vitamins
Vitamin B12
Full-term infants of adequately nourished women are born with a total body vitamin B12 content of 30 to 40 µg (FAO, 1988). Assuming that 0.10 µg/day is required during infancy (FAO, 1988), these stores would supply an infant's needs for approximately 8 months. The 0.4 µg of vitamin B12 per day usually provided by human milk to the exclusively breastfed infant provides for ample accumulation of stores (FAO, 1988; NRC, 1989). Vitamin B12 concentrations in milk, and thus the infant's intake of this vitamin, are dependent on the mother's B12 intake and stores.
Breastfed infants born to women who eat little or no animal foods are at risk for developing vitamin B12 deficiency. In a study of six vitamin B12-deficient, exclusively breastfed infants in India, vitamin B12 concentrations in their mother's milk ranged from 0.03 to 0.07 µg/liter (Jadhav et al., 1962). Vitamin B12 deficiency has also been found in breastfed infants of complete vegetarian mothers in industrialized countries (Close, 1983; Davis et al., 1981; Gambon et al., 1986; Higgenbottom et al., 1978; Rendle-Short et al., 1979; Sklar, 1986). Urinary methylmalonic acid (UMMA) concentrations of the breastfed infants of omnivorous mothers were significantly lower (p = .05) than those of infants of complete vegetarians; maternal serum B12 concentrations were negatively associated with maternal UMMA (p = .003) and infant UMMA (p < .001) levels (Specker et al., 1988). In general, the deficiency syndrome is usually not clinically apparent until the latter half of infancy. An important finding is that breastfed infants may develop clinical signs of vitamin B12 deficiency before their mothers do (Lampkin and Saunders, 1969; McPhee et al., 1988).
For infants of mothers eating a mixed diet that includes animal foods, human milk is a generous source of vitamin B12; it provides for the infant's needs
throughout the first year of life. For mothers who are complete vegetarians, it is desirable to find an acceptable food source or supplement of vitamin B12 that will meet their needs and those of the nursing infant.
Folate
The full-term infant is born with adequate folate stores, even when maternal folate is suboptimal (Salmenperä et al., 1986b). The bioavailability of folate in human milk is high: to maintain equivalent folate status in formula-fed infants, approximately 50% more folate is required from formula than from human milk (Ek and Magnus, 1982). Serum and red cell folate levels are adequate in breastfed infants; indeed, they are several-fold greater than adult reference levels (Ek and Magnus, 1979; Salmenperä et al., 1986b; Smith et al., 1985). This is reported for infants exclusively breastfed for up to 1 year (Salmenperä et al., 1986b).
Maternal folate levels in serum and milk do not appear to be correlated; however, there are strong associations between maternal and infant serum folate levels at 6 weeks and at 3 months after birth (Smith et al., 1983) and at 4 and 9 months after birth (Salmenperä et al., 1986b). Those associations suggest that folate stores accumulated in utero are more important determinants of folate status during infancy than are levels of folate in milk.
Riboflavin
Biochemical data concerning the riboflavin status of infants are difficult to interpret. Hovi and colleagues (1979) reported a transient increase in the activation coefficient of erythrocyte glutathione reductase (EGR) in full-term healthy breastfed newborns—a finding that suggests riboflavin deficiency. The increase became even greater when the infants had received phototherapy for treatment of hyperbilirubinemia (Gromisch et al., 1977; Hovi et al., 1979; Tan et al., 1978); however, this was not accompanied by clinical signs of riboflavin deficiency. The increase in the activation coefficient did not occur with daily maternal riboflavin supplements of 0.5 mg/kg of body weight, but neither was this increase evident after 2 weeks in the infants of unsupplemented women (Nail et al., 1980). The riboflavin concentration in human milk is dependent on maternal riboflavin status (Bates et al., 1982).
High EGR activation coefficients have been reported for breastfed infants who receive only 0.13 to 0.21 mg of riboflavin per day from human milk (Bates et al., 1982). The average intake of riboflavin in exclusively breastfed infants in the United States is estimated to be 0.26 mg/day. Using several criteria, including riboflavin levels in urine and blood, Snyderman and coworkers (1949) found that riboflavin intakes of 0.3 to 0.4 mg/day provide adequate riboflavin status. Among infants undergoing phototherapy, comparable intakes of riboflavin maintain normal EGR activation coefficients (Tan et al., 1978). No
longitudinal studies of representative populations have been conducted in developed countries to determine the adequacy of riboflavin status among breastfed infants. No reports of riboflavin deficiency among exclusively breastfed infants in the United States were encountered in the review of the literature by the subcommittee.
Minerals
Iron
Iron deficiency and iron deficiency anemia remain important problems in the United States and the rest of the world. The estimated worldwide prevalence of anemia in children from birth to age 4 years is 43%. There are remarkable differences in the prevalence rates of iron deficiency anemia in economically developed regions (~12%) and developing areas (~51%) of the world (FAO, 1988). In the United States, children aged 1 to 2 years have a higher prevalence of iron deficiency (9.3%) than do people in other age groups (DHHS, 1988). Although inadequate iron intakes are not the sole cause of anemia in infants and children, diets low in iron play a major etiologic role.
A heavy demand is placed on the iron reserves of breastfed infants: the estimated daily physiologic requirement is 0.7 mg for growth and 0.2 mg to replace basal losses (Dallman, 1986). Human milk provides from 0.15 to 0.68 mg of iron per day. Approximately 50% of iron is absorbed from human milk compared with 7% from iron-fortified formula and 4% from infant cereals (Dallman, 1986). The iron concentration in milk is not influenced by the mother's iron status (Dallman, 1986; Murray et al., 1978; Siimes et al., 1984). Body stores of iron and ferritin levels increase during the first 3 months of postnatal life and then drop during the fourth to sixth months (Duncan et al., 1985; Garry et al., 1981; Saarinen et al., 1977). Despite those changes, iron deficiency is uncommon in breastfed infants during their first 6 months (Duncan et al., 1985; Garry et al., 1981; Owen et al., 1981; Picciano and Deering, 1980; Saarinen and Siimes, 1979a; Saarinen et al., 1977).
Woodruff and colleagues (1977) suggest that partially breastfed infants younger than 6 months are at risk of iron deficiency: they found a hemoglobin level lower than 11.0 g/dl in 1 of 12 breastfed infants and a transferrin saturation of less than 16% in 4 of them. Mothers in that study were instructed to feed supplementary foods to the infants at age 3 months; foods high in iron content were offered in limited amounts. Supplementary foods lead to decreased intake of human milk and possibly impair the absorption of iron from human milk (Oski and Landaw, 1980).
Two studies of a total of 43 infants indicated that there is a risk of iron deficiency by age 9 months if human milk is the infant's only food (Pastel et al., 1981; Siimes et al., 1984). Therefore, foods with bioavailable iron,
iron-fortified foods, or an iron supplement should be given beginning at age 6 months (or earlier, if supplementary foods are introduced before that time).
Copper
Full-term infants have relatively large copper stores at birth (Brückmann and Zondek, 1939; Widdowson et al., 1972). The relationship between maternal copper status and concentration in human milk is weak (Munch-Peterson, 1950; Salmenperä et al., 1986a; Vuori et al., 1980). Serum copper levels were higher among the older infants in a cross-sectional study of breastfed infants ranging from newborns through age 12 months (Ohtake, 1977). Salmenperä and coworkers (1986a) found that ceruloplasmin as well as serum copper consistently rose during 12 months of exclusive breastfeeding, despite the low copper intakes characteristic of breastfed infants. The daily intakes from months 4 to 9 ranged from 0.03 to 0.26 mg/day—less than the Food and Nutrition Board's estimated safe and adequate daily dietary intake of 0.4 to 0.6 mg/day for infants aged 0 to 6 months (NRC, 1989). Neither the subcommittee nor Mason (1979) could find case reports of copper deficiency of exclusively breastfed infants. Thus, the evidence suggests that the bioavailability of copper in human milk is high and that the copper status of breastfed infants is adequate during the first year of life.
Zinc
Human milk has been regarded as a good source of zinc (Lönnerdal et al., 1984); this form of zinc is highly bioavailable (Sandström et al., 1983). The zinc concentration in human milk does not appear to be influenced by the mother's diet (Lönnerdal et al., 1981), but the evidence is not consistent (see Chapter 6).
Plasma zinc levels in breastfed infants are similar to those of adults and of infants fed zinc-fortified formula (Hambidge et al., 1979), and they are substantially greater than those of infants fed formula not fortified with zinc (Hambidge et al., 1979; Vigi et al., 1984). Erythrocyte and hair zinc levels also are similar in breastfed infants and infants fed formula fortified with zinc (Hatano et al., 1985; MacDonald et al., 1982). The full-term breastfed infant who is born with usual liver zinc stores is at very low risk of zinc deficiency.
Fluoride
Although fluoride is not considered to be an essential nutrient, it is beneficial to humans in the prevention of dental caries (NRC, 1989). Fluoride supplementation during infancy helps prevent caries in deciduous teeth; however, caries is a multifactorial disease, and breastfeeding may affect the prevalence of caries in ways other than by the provision of fluoride. The subcommittee found
no studies that directly assessed the relationship between the mode of feeding in infancy and the incidence of caries.
From the fluoride concentrations in human milk estimated by the Committee on Nutrition (1985), one could project that an exclusively breastfed infant would consume only 0.012 mg of fluoride per day. Fluoride levels are not easily increased by maternal dietary or supplementary fluoride (see Chapter 6). The only other likely source of fluoride in the infant's diet is water. The subcommittee supports the recommendations of the American Academy of Pediatrics that infants receive 0.25-mg fluoride supplements daily if their water supply contains less than 0.3 ppm of fluoride (Committee on Nutrition, 1986).
GROWTH AND DEVELOPMENT
Historically, growth has been used as the basis to judge the adequacy of nutrient intake by the infant. A major question before the subcommittee was whether nutrition of the lactating woman influences infant growth. Because slow infant growth is sometimes used as a reason for supplementing infants with formula or solid foods or for discontinuing breastfeeding, it was essential to include a brief review of the assessment of infant growth. Interrelationships among infant growth, other indices of development, and maternal nutritional status were found to be difficult to ascertain, since few sound studies had been conducted to address them.
Pattern of Growth
Although the most commonly used indicators of infant growth have been body weight and weight gain, it is desirable to consider simultaneously length in order to assess linear growth and adiposity (the relationship of weight to length, also indicated by skinfold thickness).
Healthy, full-term infants lose an average of approximately 5 to 8% of their body weight during the first week after birth; the percentage lost is somewhat higher among breastfed infants (7.4%) than formula-fed infants (4.9%) (Podratz et al., 1986) but is unlikely to be of clinical importance. After the first week, the pattern of weight gain in infancy depends on the initial size of the infant, whether the infant is breastfed or formula fed, and other environmental and physiologic factors. In industrialized countries, the rate of weight gain of breastfed infants is similar to that of formula-fed infants and to National Center for Health Statistics (NCHS) reference data for infants up to age 2 to 3 months; however, it is less rapid over the subsequent 9 months (Chandra, 1982; Czajka-Narins and Jung, 1986; Dewey et al., 1990a; Duncan et al., 1984; Forsum and Sadurskis, 1986; Garza et al., 1987; Hitchcock et al., 1985; Saarinen and Siimes, 1979b; Salmenperä et al., 1985; Whitehead and Paul, 1984). In developing countries, breastfed infants tend to grow more rapidly than their formula-fed counterparts
throughout the first 6 months of postnatal life (Mahmood and Feachem, 1987; Seward and Serdula, 1984; Unni and Richard, 1988). This difference may reflect a greater risk of infection and malnutrition among formula-fed infants in low-income populations living in areas with poor sanitation (Brown et al., 1989).
Although most studies have linked differences in the rate of weight gain with the mode of feeding, differences in linear growth between breastfed and formula-fed infants are small if differences in size at birth are controlled (Czajka-Narins and Jung, 1986; Dewey et al., 1989; Hitchcock and Coy, 1989; Nelson et al., 1989). Weight for length tends to be somewhat lower for breastfed infants than for formula-fed infants after age 6 months (Czajka-Narins and Jung, 1986; Dewey et al., 1989; Hitchcock and Coy, 1989). Thus, it is likely that the differences in weight gain patterns represent primarily differences in adiposity.
In affluent populations, the difference in rate of growth associated with mode of feeding during infancy is consistent with energy intakes of breastfed and formula-fed infants. At 3 to 4 months of age, reported average energy intakes of breastfed infants are lower than those of formula-fed infants—74 to 91 kcal/kg of body weight compared with 92 to 104 kcal/kg, respectively (Axelsson et al., 1987; Butte et al., 1984; Dewey and Lönnerdal, 1983; Dewey et al., in press; Shepherd et al., 1988). The subcommittee found only one study of infants older than 5 months that compared intakes by feeding method. Dewey et al. (in press) reported that average gross energy intakes of breastfed infants are consistently lower than those of formula-fed infants—84 and 98 kcal/kg, respectively, at 6 months; 87 and 97 kcal/kg, respectively, at 9 months; and 92 and 95 kcal/kg respectively, at 12 months—even though both groups received solid foods beginning at 4 to 6 months. As discussed in Chapter 5, these lower intakes by breastfed infants are governed primarily by infant demand, not by insufficient milk volume.
Assessment of Growth of Breastfed Infants
Growth charts used to assess infant growth are based on data derived primarily from formula-fed infants (Hamill et al., 1977). The commonly used NCHS infant growth charts are based on information collected by the Fels Research Institute from 867 infants born between 1929 and 1975. In that study, the mode of feeding was known for 75% of the infants. Of those, only 17% were exclusively breastfed and few were breastfed for more than 3 months. Furthermore, the infant formulas used at that time were less similar to human milk (for example, they were higher in total proteins, total fats, and saturated fatty acids) and the infants were more likely to be given solid foods before age 4 months than they are today (Fomon, 1987). Therefore, a number of investigators suggest that NCHS growth charts are inappropriate for breastfed
infants (Dewey et al., 1990; Duncan et al., 1984; Hitchcock et al., 1985; Whitehead and Paul, 1984).
Similarly, it has been noted (Dewey et al., 1989) that it can be misleading to assess growth of breastfed infants by using current reference data for the increment in weight or length during specified age intervals, such as data presented by Roche and colleagues (1989) based on the Fels Research Institute population. The 5th percentile of these reference data was suggested by Roche et al. as a cutoff to indicate infants at risk of malnutrition. Caution is needed in applying this cutoff because the data are based on predicted values obtained from measurements at 3- rather than 1-month intervals, which obscures some of the short-term variation in infant growth. Thus, more than 5% of all infants, regardless of feeding mode, would be expected to be below the Fels 5th percentile in any given month.
Furthermore, Dewey and coworkers (1989) found that the percentage of breastfed infants who fell below the 5th percentile for weight gain was much larger than would be expected, averaging 31% over the six 1-month intervals up to age 6 months and 52% over the six 1-month intervals between ages 6 and 12 months. On average, infants grew less rapidly than the 5th percentile during 4.4 months out of the first 12 months. Although these percentages seem alarming, these were healthy infants who fed on demand and received solid foods beginning at 4 to 6 months. Furthermore, with regard to morbidity, activity levels, or time sleeping during the first year of life, the breastfed infants with growth rates below the 5th percentile were not different from the breastfed infants with faster growth rates (Dewey et al., in press). Therefore, it is highly likely that their growth rates were normal and not a sign of malnutrition.
Recently, data have been published on weight and length gain of 419 breastfed and 720 formula-fed infants enrolled in growth studies in Iowa between 1965 and 1987 (Nelson et al., 1989). These infants were measured up to 112 days of age. One shortcoming of the study was that the breastfed infants were allowed up to 240 ml of formula per day, and before 1979, all infants were allowed solid foods beginning at 1 month of age. Therefore, additional data are necessary to construct appropriate growth charts for infants exclusively breastfed for the first 4 to 6 months and extensively breastfed throughout the remainder of the first year.
Long-Term Growth Status
Few investigators have examined whether the differences in growth rates between breastfed and formula-fed infants during the first year of life are maintained later on in childhood. Birkbeck et al. (1985) measured children at 7 years of age who were either breastfed for at least 12 weeks (N = 283) or formula fed from birth (N = 383). Children who had been breastfed were taller, but the difference was not statistically significant when controlled for
birth weight, parental stature, and socioeconomic status. Similarly, Pomerance (1987) found no differences in growth rate from 3 to 12 years of age between formula-fed children and those who had been breastfed for at least 2 months. These studies indicate no long-term difference in growth status attributable to breastfeeding. However, more data are needed on children breastfed for 4 months or longer.
Maternal Nutrition and Infant Growth
It is often assumed that maternal nutrition during lactation will affect milk volume and composition and, therefore, infant growth. However, Chapters 5 and 6 provide evidence that some of these assumptions are not necessarily valid. For example, the influence of maternal nutrition on milk volume is difficult to demonstrate, even in populations at substantial risk of malnutrition. The links among maternal nutrition, the composition of human milk, and growth rates of breastfed infants are problematic for several reasons. First, infant growth is not a very good indicator of milk or energy intake because the amount of energy required for growth is a relatively small fraction of total energy needs. In the first 2 months of postnatal life, growth accounts for approximately 30% of energy requirements, but this decreases to 5 to 10% at 4 to 6 months and to about 3% at 10 to 12 months (Butte, 1988). Second, several factors, such as intestinal malabsorption following digestive tract infections, may affect growth independently of energy intake. Finally, the energy content of fat tissue is much higher than that of lean tissue, so some consideration of the changes in body composition is necessary in evaluating different infant growth patterns.
The results of studies in Taiwan (Adair and Pollitt, 1985), Colombia (Herrera et al., 1980), and Guatemala (Lechtig and Klein, 1980) indicate that maternal nutrition during lactation has little direct impact on infant growth. In these studies, mothers were provided food supplements during both pregnancy and lactation, and infant weight gain was a main outcome variable. In only two cases was maternal supplementation associated with increased infant growth: among a subgroup of thinner breastfeeding women in the Colombia study (Herrera et al., 1980) and in a subset of the women in the Guatemala study for whom complete information was available (Delgado et al., 1982).
Several factors limit the interpretation of results from these studies: the provision of supplements to infants as well as to the mothers (Herrera et al., 1980; Lechtig and Klein, 1980), higher mean birth weights of the infants born to the supplemented mothers (Herrera et al., 1980), lack of information of the impact of the supplement on total maternal energy intake, and self-selection bias among women consuming greater amounts of the supplement in the Guatemala study (Lechtig and Klein, 1980).
Growth of breastfed infants during the first 4 to 6 months is similar in industrialized and developing countries (Whitehead and Paul, 1984). The initial
starting point—birth weight—may differ among populations, but the shape of the weight curve among exclusively breastfed infants from 0 to 6 months is remarkably consistent.
Development
As noted below, several developmental indices have been associated with the mode of infant feeding. However, no reports have been published concerning the influence of maternal nutrition on any of these indices within a group of breastfed infants.
Gastrointestinal and Metabolic Responses
Compared with formula-fed infants, breastfed infants have slower rates of gastric filling (Lucas et al., 1981a) and faster rates of gastric emptying (Cavell, 1979, 1981). These differences may contribute to some of the differences in hormonal responses, such as the decreased insulin and gastric inhibitory polypeptide responses and the higher blood ketone levels, in breastfed as compared with formula-fed infants (Lucas et al., 1981b). These responses may have long-term effects on the fat deposition of the infant (see the section ''Obesity" below).
Studies in animals suggest that several components of human milk may contribute to gastrointestinal maturation (Koldovskỳ et al., 1988; Menard and Arsenault, 1988; Sheard and Walker, 1988). This is the subject of ongoing research but is beyond the scope of this report.
Other Physiologic Responses
Measurements of breastfed and formula-fed infants made during the first 3 postnatal days indicate a distinct physiologic state and a greater reactivity to stimuli among breastfed infants, in that their heart rates are slower and more variable and their vagal tone is elevated (DiPietro et al., 1987). Butte and coworkers (1990) reported that sleeping metabolic rate and body temperature are slightly lower in breastfed infants than in formula-fed infants at ages 1 and 4 months. Elias et al. (1986) and Wright and colleagues (1983) reported that breastfed infants are more likely to awaken at night than are formula-fed infants during the first 2 years, whereas Weissbluth et al. (1984) found no such difference among 4- to 8-month-old infants.
Cognitive Development
Several studies have compared the cognitive development of children aged 6 months to 15 years who were breastfed or formula fed (Fergusson et al., 1982; Menkes, 1977; Morley et al., 1988; Morrow-Tlucak et al., 1988;
Rodgers, 1978; Taylor and Wadsworth, 1984). Slight but statistically significant differences favoring children who had been breastfed were reported. In the most complete studies (Fergusson et al., 1982; Morrow-Tlucak et al., 1988; Taylor and Wadsworth, 1984), the influence of covariates such as maternal intelligence, educational level, socioeconomic status, child-rearing attitudes, and home environment were controlled for in the analysis. With these adjustments, the differences between groups, although smaller, remained statistically significant. The biologic importance of the findings is unclear. It is possible that the differences resulted from other confounding variables that were not identified or from enhanced maternal-infant interactions rather than from the specific biologic properties of human milk.
BREASTFEEDING AND MORBIDITY
Short-Term Effects
Infectious Diseases
Breastfeeding may protect the recipient infant against common infectious diseases in four different ways:
-
Human milk inhibits the growth of many types of microbial pathogens. By avoiding foods other than human milk through exclusive breastfeeding, the infant's exposure to environmental pathogens is limited.
-
Breastfeeding enhances the nutritional status of infants under conditions in which poor sanitary environments and the low quality of weaning foods reduce the nutritional well-being of infants.
-
Through its contraceptive effects, breastfeeding may increase the interval between births (Habicht et al., 1985; Hobcraft, 1987; Thapa et al., 1988) and may thus both reduce the number of infectious contacts within households (Abay et al., 1984; Hanson, 1986) and permit more human resources and other benefits to be directed to fewer children.
-
Human milk contains an array of antimicrobial agents (Goldman and Goldblum, 1989), anti-inflammatory agents (Goldman et al., 1986, 1990b), and immunologic stimulating agents (Goldman and Goldblum, 1989, 1990), most of which are absent from infant formula, or are present only in limited quantities, and are produced by the young infant only in limited amounts.
The frequency of gastrointestinal infections appears to be much lower in breastfed infants than in formula-fed infants in many industrialized countries (Cunningham, 1981; Cushing and Anderson, 1982; Palti et al., 1984) and developing countries (Clemens et al., 1986; Glass and Stoll, 1989; Mata et al., 1967, 1969; Plank and Milanesi, 1973; Ruiz-Palacios et al., 1986). The protection against gastrointestinal infections is more evident in populations in which sanitary standards are low and potable water is not readily available.
However, a lower incidence of gastrointestinal infections in breastfed infants in the United States (Myers et al., 1984) and lower perinatal mortality from infectious disease among breastfed, compared with formula-fed, infants in England (Carpenter et al., 1983) suggest that the protective effects of breastfeeding are important in industrialized as well as in developing countries. Moreover, since breastfed infants may remain asymptomatic even after exposure to enteropathogens (microorganisms that infect the intestinal tract) contaminating the nipples and areola of the nursing mother (Mata and Urrutia, 1971; Wyatt and Mata, 1969), the protection afforded by breastfeeding is attributable to factors in the milk in addition to an avoidance of pathogens.
For some infections, the duration is shorter and the intensity is less in breastfed infants than in formula-fed infants, but the attack rate (the incidence during a defined period of risk) is similar (Duffy et al., 1986; Glass et al., 1983; Mata et al., 1967, 1969). For example, in Bangladesh, protection against infections but not against colonization with Vibrio cholerae correlated with the amount of secretory immunoglobulin A (IgA) antibodies to that bacterium in the human milk (Glass and Stoll, 1989; Glass et al., 1983). In the United States, the protection afforded to infants against rotavirus appeared to result from factors in human milk other than specific antibodies (Duffy et al., 1986).
Protective effects of breastfeeding against certain respiratory infections have also been reported. For example, respiratory syncytial virus infections are less severe among breastfed infants than among formula-fed babies, although the attack rate is the same (Chiba et al., 1987; Downham et al., 1976; Pullan et al., 1980). In addition, serum interferon-α levels are higher in breastfed infants following respiratory syncytial virus infection (Chiba et al., 1987), even though the concentration of this antiviral agent is negligible in human milk. This suggests that the breastfed infant's immune system may be primed by human milk to respond to certain respiratory viral pathogens more rapidly and to a greater degree than is the case for formula-fed infants.
The incidence of otitis media is lower in breastfed than in formula-fed infants (Saarinen, 1982; Schaefer, 1971), but it is unclear whether this is a result of components of human milk or of other factors associated with the feeding method, such as higher enrollment of formula-fed infants in day-care centers, differences in the sucking mechanisms, or the adverse effects of foreign proteins in formula-fed infants.
The relationship between the nutritional status of the lactating woman and the susceptibility of the breastfed infant to infections requires further study.
Allergies
The effects of various infant-feeding practices on the age of onset and severity of allergies have been extensively investigated. Breastfeeding appears to be protective against food allergies (see the review by Kramer [1988]).
Evidence concerning such protective effects against other types of allergies is conflicting. When suspected food allergens were excluded from the diets of pregnant and lactating women whose families were at high risk for atopic dermatitis (Chandra, 1987; Chandra et al., 1985), the incidence of this condition was lowered among breastfed infants compared with its incidence in a group of controls. Recently, Hattevig and colleagues (1990) also found that maternal dietary restrictions for 3 months during lactation resulted in a lower rate of atopic dermatitis among the mothers' infants for 6 months. Several case reports suggest that components of foods ingested by the mother pass into the milk and then cause allergic reactions in the infant, and that these problems can be prevented by excluding the allergens from the maternal diet (Cant et al., 1985; Harmatz et al., 1987; Jakobsson and Lindberg, 1978, 1983; Kilshaw and Cant, 1984; Shannon, 1921). Nonetheless, the benefit of prophylactic use of allergen-restricted diets for the general population of pregnant and lactating women is not well established (Hattevig et al., 1990).
Early observations that atopic eczema in infants is associated with abnormal serum lipids (Hansen, 1937; Hansen et al., 1947) have recently been confirmed and extended (Bordoni et al., 1988; Manku et al., 1982; Rocklin, 1986; Wright, 1989). The findings suggest that in affected infants, the conversion of dietary linoleic acid to long-chain polyunsaturated metabolites is defective (Wright, 1985). The administration of γ-linolenic acid, a normal constituent of human milk, helps alleviate the eczema (Biagi et al., 1988; Meigel et al., 1987; Morse et al., 1989). In keeping with that finding, human milk consumed by infants with atopic dermatitis had lower concentrations of dihomo-γ-linolenic acid (a metabolite of linolenic acid) than did milk from control subjects (Morse et al., 1989).
Although the lay literature implies that maternal diet may contribute to colic (crying greatly in excess of 1 hour/day [Brazelton, 1962; Carey, 1984; Hunziker and Barr, 1986]) among breastfed infants, the subcommittee found only one study concerning this relationship. Results from that small study in Denmark (Jakobsson and Lindberg, 1978) suggest that the passage of food allergens, especially from cow's milk, through human milk accounts for 30% of the cases of colic in breastfed infants. It is difficult to generalize from that report, however, since the subjects may not have been representative of the overall population of infants with colic.
Chronic Diseases with an Immunologic Basis
Because human milk contains a host of direct-acting antimicrobial factors, anti-inflammatory agents, and substances that may hasten the maturation of the infant's immune system, there is reason to believe that development of chronic diseases having an infectious or immunologic basis may be influenced by the method of infant feeding. Recent investigations suggest that breastfeeding
exerts long-term protective effects against three such diseases: type I diabetes mellitus (Borch-Johnsen et al., 1984; Mayer et al., 1988), lymphoma (Davis et al., 1988), and Crohn's disease (Koletzko et al., 1989). Potential risk factors were identified for 114 cases of Crohn's disease and 180 controls (unaffected siblings) within 107 families. The children with this disease were less likely to have been breastfed (relative risk of 3.6 for formula-fed infants.) Even though each of these studies was retrospective and depended on long-term recall, the finding that breastfeeding may reduce the risks of developing serious disorders in later life warrants further investigation.
Effects of Infectious Agents
A discussion of infectious agents in human milk requires consideration of both exposure to the pathogen and development of overt disease. Collected human milk contains many common epidermal bacteria that are not harmful to the recipient infant. With appropriate management, it is uncommon for the infant to become infected from women with mastitis or breast abscesses (Rench and Baker, 1989; Thomsen et al., 1984). Enteropathogens such as Shigella and Salmonella species may contaminate human milk through the areola and nipples of the breast, especially if those bacteria are prevalent in the environment (Mata et al., 1967, 1969; Wyatt and Mata, 1969). However, symptomatic infections with those enteropathogens are unusual in the recipient infant, seemingly because of the activity of the host defense system in human milk.
Of more concern are the reports that human milk contains viral pathogens including cytomegalovirus, hepatitis B virus, rubella virus, human T lymphocytotropic virus type 1 (HTLV-1), and human immunodeficiency virus type 1 (HIV-1). Cytomegalovirus is common in milk produced by seropositive women (Ahlfors and Ivarsson, 1985; Dworsky et al., 1983; Stagno et al., 1980). As many as 70% of seropositive lactating women have the virus in their milk at some time (Ahlfors and Ivarsson, 1985). Although the infants become infected by ingesting the virus, the colonization does not progress to disease (Dworsky et al., 1983; Stagno et al., 1980), possibly because of specific IgG antibodies transferred via the placenta during pregnancy and immune substances provided in the mother's milk.
Both the wild and vaccine strains of rubella virus have been recovered from human milk (Buimovici-Klein et al., 1977; Klein et al., 1980; Losonsky et al., 1982). As a consequence of this transmission, mild clinical rubella has been reported, but severe disease has not.
Women who live in regions endemic for hepatitis B commonly have the surface antigens in their milk, but there is no evidence that breastfeeding is a route through which hepatitis B virus is transmitted and infection results (Boxall et al., 1974; Linnemann and Goldberg, 1974).
In parts of the West Indies, Africa, and the southwestern area of Japan, HTLV-1 (the retrovirus that causes adult T-cell leukemia) is endemic (Wong-Staal and Gallo, 1985). This virus is commonly found in milk from carrier women. Recently, epidemiologic evidence of the transmission of this virus via breastfeeding was found in an endemic population in the Nagasaki Prefecture in Japan (Hino, 1989). Since the annual incidence of adult T-cell leukemia in that population is estimated to be 0.1 to 0.2%, the demonstration of HTLV-1 in human milk is of great concern there. In a small sample investigated in Hino's study, 30% of breastfed, 10% of mixed-fed, and none of the totally formula-fed infants became positive carriers for the virus. Although it is not known whether the seropositive infants will develop the disease in later life, Hino has recommended that breastfeeding cease in that region of Japan to interrupt the spread of the disease. Follow-up studies are needed to validate or refute that recommendation. Although HTLV-1 is not a major disease in the United States, it is possible that a public health problem may develop if infected individuals immigrate to this country from endemic areas.
Of more immediate concern is the growing epidemic of HIV-1 infections in the United States and other countries and the possibility that HIV-1 may be transmitted to the infant via breastfeeding. Transplacental transmission of HIV-1 infections to the fetus occurs in about 30 to 40% of cases when women are seropositive during pregnancy. In contrast, the risk of transmitting HIV-1 via breastfeeding is not well established. Human milk contains CD4+ T cells and macrophages that are prime targets for the virus. There are two reports of the detection of HIV-1 in human milk (Thiry et al., 1985; Vogt et al., 1986). Moreover, there are several reports of the postnatal acquisition by the mother of HIV-1 by transfusion of contaminated blood and the subsequent transmission of the infection to the infant, ostensibly by breastfeeding (see the review by Davis [1989]). The risk of transmitting the infection under more natural conditions remains uncertain.
More recent studies suggest that there is little likelihood that HIV-1 is a frequent contaminant of human milk or that breastfeeding is an important route of transmission of the virus. In a preliminary report (Yolken et al., 1990), the HIV-1 virion, RNA, messenger RNA, or integrated complementary DNA could not be demonstrated in the milk of HIV-1-infected women by the polymerase chain reaction. In addition, specific antibodies to HIV-1 (Yolken et al., 1990) and other factors that inhibit the binding of HIV-1 envelope proteins (gp120) to CD4+ T cells (Newburg and Yolken, 1990) have been found in human milk.
Finally, a preliminary report of a European collaborative study on perinatal HIV-1 infections suggests that the risk of transmitting HIV-1 infections by breastfeeding women who were found to be seropositive during pregnancy is low (M.L. Newell, Institute of Child Health, London, United Kingdom, personal communication, 1990). However, the results of that study are inconclusive since only small numbers of infants were investigated; a more extensive follow-up
of the infants will be required to ascertain whether there is a discernible risk of HIV-1 attributable to breastfeeding. In addition, studies are needed to determine whether breastfeeding provides health benefits to the infant who is infected with HIV-1 during intrauterine life.
Until more definitive information is available, the reader should consult two public health pronouncements on this subject: in the United States, the Centers for Disease Control (CDC, 1985) advised mothers who are seropositive for HIV-1 not to breastfeed; the World Health Organization (WHO, 1987) took the view that the risk of transmitting the virus in human milk is uncertain and that breastfeeding should be encouraged, especially in developing countries, because of the known health benefits of human milk.
Obesity
The effects of early feeding practices—both type of feeding (formula or human milk) and timing of introduction of solid foods—on immediate and subsequent weight have been extensively studied (see reviews by Hamosh [1988], Hamosh and Hamosh [1987], Himes [1979], and Kovar et al. [1984]). However, there are limitations in many of those studies. For example, the duration and degree of breastfeeding were seldom reported, and data on infant feeding practices were often based on parental recall rather than recorded information (see reviews by Hamosh and Hamosh (1987) and Kramer [1981, 1987]).
Nevertheless, no association between breastfeeding and obesity in the adult has been established. In general, early studies indicated that breastfed infants were much leaner than formula-fed ones (Taitz, 1977; Weil, 1977), whereas later studies show a weaker association (Dubois et al., 1979; Ferris et al., 1980; Hofvander et al., 1982; Saarinen and Siimes, 1979b; Shukla et al., 1972; Sveger et al., 1975; Vobecky et al., 1983). This difference may be attributable to the introduction of new formulas in the 1980s (the later formulations are more similar to human milk) and to changes in parental attitudes that have led to less overfeeding of formula-fed infants (reviewed by Hamosh [1988], Hamosh and Hamosh [1987], and Whitehead et al. [1986]). Furthermore, Birkbeck and colleagues (1985) controlled for familial differences in socioeconomic status and stature, but their results failed to show that breastfeeding prevents obesity in children.
The genesis of obesity appears to involve many factors (Hamosh and Hamosh, in press), including critical periods of pre- or postnatal adipose tissue development, genetics (Bouchard et al., 1990; Mueller, 1983; Ravussin et al., 1988; Stunkard et al., 1986, 1990), eating style (Agras et al., 1987), energy expenditure (Berkowitz et al., 1985; Bogardus et al., 1986; Bray, 1987; Elliot et al., 1989; Ravussin et al., 1988; Roberts et al., 1988), and social factors (Agras et al., 1987; Berkowitz et al., 1985; Roberts et al., 1988). Thus, the precise role of breastfeeding in this process remains undetermined.
Atherosclerosis
For years there has been interest in whether the cholesterol content of human milk, which is higher than that of commercial infant formulas, has a beneficial or adverse effect on later development of atherosclerosis.
Studies in animals, including rats, guinea pigs, and baboons, have failed to provide clear-cut evidence that the mode of feeding affects blood cholesterol levels (see the review by Hamosh [1988]), but they suggest that feeding cholesterol may affect the mechanism of handling cholesterol in the body (Mott et al., 1990). Human studies (Fomon et al., 1984; Friedman and Goldberg, 1975; Hodgson et al., 1976; Huttunen et al., 1983; Marmot et al., 1980) show only small and inconsistent differences in serum cholesterol levels between formula-fed and breastfed children and young adults. Confounding genetic factors and dietary intake after weaning may not have been adequately considered in the interpretation of study results (Wissler and McGill, 1983). Furthermore, the extent and duration of breastfeeding have not been considered with sufficient rigor in most studies. Thus, the specific effects of the types of infant feeding on the development of atherosclerosis cannot be assessed at present.
BREASTFEEDING AND MORTALITY
Research Through 1950 in Developed Countries
Since the latter part of the last century, a great deal of attention has been given to the relationship of the mode of infant feeding with the infant's subsequent survival (see Appendix A).
Differences in postneonatal mortality rates between breastfed and bottle-fed infants in the developed countries from 1850 to 1950 are vividly summarized in Figure 7-1. The trends are striking and straightforward: there were markedly lower mortality rates among breastfed than among bottle-fed infants throughout. Although mortality rates, and thus absolute differences in mortality rates between the two groups, fell dramatically during the period, the relative advantage of breastfed infants persisted throughout, such that the postneonatal death rates of breastfed infants continued to remain at least half those of bottle-fed infants. While infant mortality rates among both breastfed and bottle-fed groups are now very low, there is no current information on whether breastfeeding confers any advantages for infant survival in the developed countries.
Research in Developing Countries
Some generalizations that can be made from the studies abstracted in Appendix B may also be relevant to policy in developed societies. First, there is a consistently increased risk of mortality among formula-fed infants under
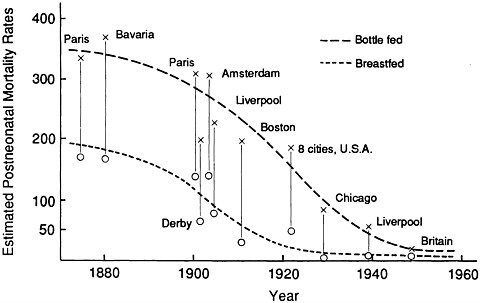
FIGURE 7-1 Estimated postneonatal mortality rates among breastfed and bottle-fed infants, various locations, 1870-1950. From Wray (1990) with permission.
1 year of age than among breastfed infants. Second, most studies suggest that infants receiving both human milk and formula have lower death rates than those of infants given only formula, but that exclusive breastfeeding is associated with the lowest mortality rates.
There are fewer data on the incidence of infectious disease than on the severity of the infection (that is, case fatality). During the neonatal period, the incidence of infectious disease appears to be lower among exclusively breastfed infants. After the neonatal period, incidence appears to be relatively less affected by mode of feeding, but severity appears to be much lessened among infants who have been breastfed. However, there are several exceptions to these generalizations: for example, Plank and Milanesi (1973) found little protection from breastfeeding among infants who were simultaneously receiving supplemental food.
The association between breastfeeding and mortality after 1 year of age has been inconsistent. For example, while Cantrelle and Leridon (1971) found no protection during the second and third years of life, Briend et al. (1988), in a careful prospective study in rural Bangladesh, observed that breastfeeding provided persistent and strong protection against mortality throughout the age span studied (up to the infants' third birthday). It does appear that even in primitive conditions excess mortality is not inevitable among those weaned
after the first birthday, but the situations in which protection is not conferred by breastfeeding are not understood.
In general, the relationship of breastfeeding to child mortality in developing countries has been found to be less strong with increased educational and social status of the mother. See, for example, the results of Palloni and Tienda (1986) from studies conducted in Peru.
Recently there has been renewed focus on the effect of child spacing among breastfed infants (see Chapter 8) on child survival (Hobcraft, 1987; Thapa et al., 1988). In countries with high infant and child mortality rates, Hobcraft (1987) estimated that a closely spaced preceding birth (a birth interval of less than 2 years) is associated with a 50% increase in the risk of mortality before age 5 for the new (index) infant. At a birth interval of less than 12 months, the risk is even higher. If another infant is born before the index child's second birthday, the index child's risk of dying before age 5 was estimated to increase by 55%. Controlling for birth weight did not influence these estimated effects, suggesting that they were in addition to the shortened interpregnancy intervals resulting from preterm delivery. In developed countries, the overall impact of child spacing on survival is probably minimal (Fedrick and Adelstein, 1973), but further research is needed to assess whether there is an impact among disadvantaged families.
Research in Developed Countries Since 1950
The subcommittee found only six published studies relating mode of infant feeding with mortality based on data collected since 1950 in the developed world (see Appendix A); three of these were concerned with sudden infant death syndrome (SIDS) (one of them focused on botulism). Naeye and colleagues (1976) found an odds ratio of 1.3 (not significant) for increased risk of SIDS among infants who were not breastfed. An odds ratio of 3.06 for formula-fed versus either breastfed or mixed-fed infants was calculated by the subcommittee from the data of Biering-Sorensen et al. (1978). Arnon and colleagues (1982) found significantly higher rates of breastfeeding among infants hospitalized with botulism than among controls but a decreased likelihood of sudden death from botulism among those who were ever breastfed.
In a study in Sheffield, England, covering the years between 1970 and 1979, Carpenter et al. (1983) attributed a decrease of 0.8 postperinatal infant deaths per 1,000 births to increased rates of breastfeeding, but no direct comparison of the rates of breastfed and bottle-fed infants was presented. A study from Canada (Department of National Health and Welfare Canada, 1963) was concerned exclusively with Canadian Indians, a relatively deprived and therefore atypical population.
Differences, even of large magnitude, may be difficult to detect in the developed countries, with their generally low infant and child death rates.
Larger and more sophisticated studies are needed to determine whether or not breastfeeding leads to lowered infant mortality in developed countries, especially among poor mothers and children.
There are many other unanswered questions about breastfeeding and infant mortality: (1) What are the relative impacts on infant mortality of improvements in nutritional status, immunologic competence, or protection from a contaminated food supply? (2) Up to what age is breastfeeding protective? (3) What are the relative merits of exclusive breastfeeding compared with partial breastfeeding supplemented by other foods? (4) What is the relative impact of breastfeeding on the incidence of infection as opposed to the severity of infection?
For further information, see the reviews by Cunningham (1981), Jason et al. (1984), Kovar et al. (1984), Kramer (1987), Winikoff (1981), and Wray (1978, 1990). Those of Cunningham (1981) and Wray (1978, 1990) are particularly informative. Since there are many sources of confounding possible when comparing death rates between breastfed and other infants, the reader is referred to the information on sources of bias at the beginning in this chapter and to the review by Kramer (1987).
MEDICATIONS, LEGAL AND ILLEGAL DRUGS, AND ENVIRONMENTAL AGENTS
The subcommittee's understanding of nutrition during lactation would be incomplete without consideration of foreign, potentially toxic substances passed from mother to infant via human milk. Such substances are discussed briefly below.
Certain drugs and environmental chemicals that enter a mother's body may appear in her milk. The hazard, if any, posed by extraneous substances in milk is dependent upon a number of factors:
-
the pharmacokinetics of the foreign substance, including its molecular size, solubility, ionization, protein binding, acidity, metabolism, storage, and excretion (Wilson, 1981);
-
the mode of entry of the agent, as well as its absorption, efficiency of metabolism, and excretion by the mother (Lawrence, 1989; Rivera-Calimlim, 1987);
-
the physiologic maturity of the infant, which influences whether or not the compound can be absorbed by the neonatal gut, detoxified, or excreted, or whether the infant is otherwise unusually susceptible to the effects of the foreign substance.
Medications
Maternal medication is usually not a reason to discourage breastfeeding.
If the mother requires a drug that poses a risk for her infant for only a short time, she can pump her breasts to maintain lactation and discard the milk. This applies to diagnostic radiopharmaceuticals, antiprotozoal compounds, and a few antibiotics that cannot be safely given to an infant (such as chloramphenicol). Compounds such as sulfadiazine may be contraindicated for the breastfeeding mother in the early postpartum period but are well tolerated when the infant is at least 1 month of age. Certain other drugs are contraindicated during breastfeeding. They include antineoplastic drugs, therapeutic radiopharmaceuticals, lithium, lactation-suppressing drugs, certain antithyroid drugs, and synthetic anticoagulants (for specific information, see Briggs et al. [1986], Committee on Drugs [1989], and Lawrence [1989]).
Caffeine or Coffee
The use of caffeine to treat immature infants with apnea has provided an opportunity to study the absorption, distribution, metabolism, and excretion of this drug in neonates. The milk-to-plasma ratio ranges between 0.5 and 0.8—an amount less than 1% of the maternal dose. However, when an infant receives frequent doses, caffeine can accumulate, causing wakefulness or irritability. Maternal consumption of one or two caffeine-containing beverages per day is not associated with unacceptable levels of caffeine in human milk (Committee on Drugs, 1989; Wilson, 1981; Wilson et al., 1985).
There is evidence from a study in Costa Rica that maternal consumption of three or more cups of coffee per day during pregnancy and lactation can affect iron concentrations in milk and infant iron status at 1 month of age (Muñoz et al., 1988). Studies in rats suggest that this effect is not due to caffeine but to some other component(s) in coffee (Muñoz et al., 1986). There is a need for further investigations in other populations and in pregnant and lactating women consuming fewer than three cups of coffee per day.
Alcohol
Small amounts of alcohol have been recommended in many cultures as a means of stimulating milk secretion. On the other hand, excessive alcohol intake has been associated with failure to initiate the let-down reflex (see Chapter 5), high alcohol levels in the milk, and lethargic nurslings (Cobo, 1973), as well as with adverse health consequences for the mother. Wilson and colleagues (1980) comment: ''The observation that acetaldehyde, the major metabolite of ethanol, is not excreted in the milk is significant since some have postulated that acetaldehyde contributes to the toxicity of alcohol" (p. 34). In a study of 1-year-old infants, Little and colleagues (1989) found a strong positive association between psychomotor development scores obtained with the Bayley Scales of Infant Development (Bayley, 1969) and a proxy measure for exposure to alcohol
through breastfeeding. Actual differences in these scores were minor for groups at the extremes of alcohol exposure. The scores of infants of breastfeeding mothers who occasionally drank (e.g., one or two drinks per week) did not differ from those of infants who were not exposed to any alcohol through breastfeeding. The amount of alcohol in the milk, the impact of confounding socioeconomic factors, and deficits in maternal interactions with the infant were not reported in this study. Therefore, it remains unclear whether alcohol in human milk presents a problem to the infant.
Cigarette Smoking
Cigarette smoking may adversely affect milk volume (see Chapter 5). Neither Luck and Nau (1985) nor the subcommittee found reports of associations between nicotine levels present in the milk of heavy smokers and symptoms in their nursing infants.
Studies to compare nicotine and cotinine levels of breastfed and formula-fed infants are most appropriate if conducted after the infants are 8 days old to eliminate the effect of transplacental exposure. The blood levels of nicotine and cotinine in infants of smokers vary widely, regardless of feeding method. It appears that absorption of nicotine in the respiratory tract, which is rapid and complete, is greater than absorption of nicotine form human milk. The infant's exposures from inspired air and from milk amount to less than 5% of the average dose inhaled by an adult smoker. Ranges of measurable nicotine in the milk vary from 1.4 to 62 ng/ml and are usually higher than the corresponding serum level. The nicotine content tends to increase with increased depth of inhalation and with an increased number of puffs per cigarette (Luck and Nau, 1985).
Although no reports have been published associating nicotine from human milk with infant health problems, it is advisable to counsel mothers to avoid smoking for 2.5 hours before feeding (Luck and Nau, 1987) and to avoid smoking in the infant's presence.
Illegal Drugs
Marijuana appears in human milk as δ-9-tetrahydrocannabinol, which is poorly absorbed in this form but may be sufficient to cause lethargy and decreased feeding frequency and duration after prolonged exposure. Heroin and cocaine also appear in human milk and place the infant at significant risk of toxicity, despite the low bioavailability from milk (Chasnoff et al., 1987).
Environmental Contaminants
In high-exposure areas, the milk of some women contains insecticides.
Organochlorinated compounds were investigated worldwide after the first report in 1951 of the presence of dichlorodiphenyltrichloroethane (DDT) in the milk of healthy U.S. women (Laug et al., 1951). Polychlorinated biphenyls (PCBs), hexachlorobenzene, dieldrin, and heptachlor epoxide have also been identified in milk from women with known heavy exposures. In a study of hundreds of subjects at delivery in the Great Lakes region of New York State in 1978, PCB was not detectable in the cord blood of the infants or in the milk of the mothers, except when the women illegally ate contaminated lake fish at least once a week (Lawrence, 1989). In general, PCB levels in milk decline over the period of lactation and with the number of children nursed (Rogan et al., 1986) if there are no new exposures during this period.
The levels of DDT and other insecticides in human milk vary with the weight of the mother, the number of children, the duration of lactation, and the residence and occupation of the mother (Rogan and Gladen, 1985; Wilson et al., 1973). Insecticide levels in human milk tend to be higher than those in cow's milk, because humans are at the top of the food chain. Levels are higher at the end of a single nursing because the fat content of the milk is increased at that time.
In a study of 858 children from birth to 5 years of age whose mother's milk contained PCBs and dichlorodiphenyl ethane (DDE, a metabolite of DDT) after a heavy exposure, there was no evidence of change in growth rate or general health, nor were there adverse effects on weight gain or differences in the number of physician visits for illness. However, DDE in the human milk was associated with a shorter duration of breastfeeding (Gladen et al., 1988; Rogan et al., 1987). These children remain under surveillance.
Agent Orange (2,3,7,8-tetrachlorodibenzo-p-dioxin), the best known of the dioxins, has been found in human milk in pooled samples from women with known exposures to high levels in Vietnam. There is no evidence that the U.S. population at large is at risk (Schecter and Gasiewicz, 1987; Schecter et al., 1986, 1987).
Heavy metals—such as lead, mercury, arsenic, and cadmium—are found in higher concentrations in certain water supplies, cow's milk, and reconstituted formula than in human milk (Dabeka et al., 1986; Jensen, 1983). Thus, breastfed infants are exposed to lower amounts than their formula-fed counterparts are. Whenever there has been an exposure or a woman has been found to have elevated mercury or lead levels, the infant's serum and the milk levels should be checked (Perkins and Oski, 1976). In the case of lead poisoning, evidence suggests that maternal serum levels under 40 mg/dl are not associated with elevated lead values in the milk (Dillon et al., 1974). Lead levels in milk are lower than would be predicted from maternal serum levels (Wolff, 1983). Based on a single case study, Ryu and colleagues (1978) state that loss of lead from the body can be expected in breastfed infants exposed to lead in utero if their lead intake is less than 5 µg/kg of body weight per day.
Filer (1968) summarized the effects of radionuclides in several parts of North America upon infants and children. Staub and Murphy (1965) found less radiostrontium in human milk than in cow's milk specimens collected in Denver and Chicago during 1959 to 1961. The deposition of strontium-90 in deciduous teeth of infants in St. Louis was also much greater in formula-fed than in breastfed infants (Rosenthal et al., 1964). Filer (1968) recommended that one of the principal methods of limiting radionuclide intake by infants would be to increase the percentage of infants who are breastfed. For more current information concerning the problem of radionuclides in foods, the reader is referred to the report of Carter (1988) of the aftermath of the Chernobyl nuclear explosion.
If there is a possibility of heavy environmental contamination, the mother should discuss the concern with the physician and, when appropriate, testing can be arranged through a state-approved laboratory.
CONCLUSIONS
-
For the biological reasons discussed in this chapter, breastfeeding is recommended for all infants in the United States under ordinary circumstances. Exclusive breastfeeding is the preferred method of feeding for normal full-term infants from birth to age 4 to 6 months. Breastfeeding complemented by the appropriate introduction of other foods is recommended for the remainder of the first year or longer, if desired. The subcommittee and advisory committee recognize that it is difficult for some women to follow the recommendations for social or occupational reasons, such as those discussed in Chapters 2 and 3.
-
During the first 2 to 3 months of lactation, exclusively breastfed infants generally stay at approximately the same weight-for-age percentile or gain weight at a slightly faster rate, although they ingest less energy than do formula-fed infants. After the third month, exclusively breastfed infants tend to follow lower weight-for-age and length-for-age percentiles. In general, those patterns are not altered by the introduction of solid foods.
-
The nutritional status of breastfed infants in the United States is generally excellent because of the uniqueness of the composition of human milk, including the high bioavailability of many of its constituents. In general, nutritional supplements are not needed for the breastfed infant. Nevertheless, certain nutritional interventions sometimes may be warranted, as described under the section "Recommendations for Clinical Practice" below.
-
In general, breastfed infants have fewer gastrointestinal or respiratory infections than do formula-fed infants in both developed and developing countries, but the extent to which breastfeeding is protective may depend upon the type of microorganism and the degree to which the infant is exposed to that pathogen.
-
Although some studies suggest that maternal nutrition may affect the immunologic components in human milk, no studies address the effect of such changes upon the susceptibility of the recipient infant to infectious diseases.
-
Breastfeeding appears to protect against food allergy and eczema, but the protective mechanisms are not understood well. The subcommittee concluded that to reduce the chance of allergy or colic in their breastfed infants, mothers should not avoid important food sources of nutrients such as cow's milk in the absence of objective evidence provided by oral elimination-challenge trials (see later section "Maternal Diet"). Although there are some reports of untoward reactions in breastfed infants linked to extrinsic food allergens in human milk, the use of food allergen-restricted diets in pregnancy, lactation, or both should be limited to those cases for which the sensitization has been proven.
-
Recent epidemiologic studies suggest that breastfeeding may lessen the risk of developing certain chronic diseases (such as lymphoma, Crohn's disease, and type I diabetes mellitus) later in life.
-
Current evidence does not warrant the conclusion that breastfeeding will prevent obesity in the offspring.
-
The effects of breastfeeding on infant and childhood mortality in the United States deserve study, especially among subgroups with higher than average mortality rates.
-
In general, human milk has not been shown to transmit infections, but more research will be required to ascertain the risk to the recipient of HTLV-1 in human milk and whether HIV-1 is a major infectious agent in human milk.
RECOMMENDATIONS FOR CLINICAL PRACTICE
Infant Nutrition
The following steps should be taken to ensure adequate nutrition of breastfed infants:
-
All newborns should receive a 0.5- to 1.0-mg injection or a 1.0-to 2.0-mg oral dose of vitamin K immediately after birth, regardless of the type of feeding that will be offered the infant.
-
The infant should be given a 5.0- to 7.5-µg supplement of vitamin D per day if his or her exposure to sunlight appears to be inadequate.
-
Fluoride supplements should be provided only to breastfed infants who live in households in which the fluoride content of the water supply is low (<0.3 ppm).
-
Human milk is a sufficient source of iron for the first 6 months of an infant's life, but foods with bioavailable iron, iron-fortified foods, or a low-dose iron supplement should be provided at 6 months, or earlier if supplementary foods are introduced before that time.
-
The milk of complete vegetarians is likely to be deficient in vitamin B12. In such cases, it is desirable to supplement the infant and either to find an acceptable food source of vitamin B12 for the mother or to provide her with a supplement of this vitamin (even if she is asymptomatic).
Infant Growth
-
The subcommittee recommends that health care providers be informed about the differences in growth between healthy breastfed and formula-fed infants. On average, breastfed infants gain weight more slowly than do those fed formula. Slower weight gain, by itself, does not justify the use of supplemental formula. When in doubt, clinicians should evaluate adequacy of growth according to the guidelines described by Lawrence (1989).
Infant Health
Breastfeeding ordinarily confers health benefits to the infant, but in certain rare cases it may pose some health risks. The following recommendations address some aspects of infant health:
-
Breastfeeding is recommended to reduce the incidence and severity of certain infectious gastrointestinal and respiratory diseases and other disorders in infancy.
-
Mothers should be encouraged to continue breastfeeding, even if they develop mastitis, with certain rare exceptions, but they should be advised to seek early treatment (see Lawrence [1989] for specific recommendations).
-
Neither cytomegalovirus nor hepatitis virus infections during lactation are contraindications to breastfeeding. In the case of hepatitis virus infections, immunizations are necessary regardless of the mode of feeding. Rubella immunization is warranted in seronegative women during the immediate postpartum period even if the woman chooses to breastfeed.
-
For mothers requiring medication and desiring to breastfeed, the clinician should select the medication least likely to pass into the milk and to the infant.
-
Medications rarely pose a problem during lactation; breastfeeding is contraindicated if certain drugs must be administered. Such drugs include antineoplastic agents, therapeutic radiopharmaceuticals, some but not all antithyroid agents, and antiprotozoan agents. Medications that can be given to infants can be taken by the lactating woman.
-
In those rare cases when there is heavy exposure to pesticides, heavy metals, or other contaminants that may pass into the milk, breastfeeding is not recommended if maternal levels are high.
Maternal Diet
Objective evidence should be obtained from an oral elimination challenge trial before advising women to make long-term diet changes to reduce the problem of allergy or colic in their breastfed infants. In such studies, the suspected food allergen is first eliminated from the mother's diet to determine whether the breastfed infant will become asymptomatic. Oral challenges should then be conducted under careful medical supervision to ascertain whether the symptoms can be reproduced. Provoked reactions should be treated with appropriate medications as rapidly as possible. If a basic food must be eliminated from the diet for weeks to months, adequate nutrient intake should be promoted through special dietary planning.
REFERENCES
Aaby, P., J. Bukh, I.M. Lisse, and A.J. Smits. 1984. Overcrowding and intensive exposure as determinants of measles mortality. Am. J. Epidemiol. 120:49-63.
Adair, L.S., and E. Pollitt. 1985. Outcome of maternal nutritional supplementation: a comprehensive review of the Bacon Chow study. Am. J. Clin. Nutr. 41:948-978.
Agras, W.S., H.C. Kraemer, R.I. Berkowitz, A.F. Korner, and L.D. Hammer. 1987. Does a vigorous feeding style influence early development of adiposity? J. Pediatr. 110:799-804.
Ahlfors, K., and S.A. Ivarsson. 1985. Cytomegalovirus in breast milk of Swedish milk donors. Scand. J. Infect. Dis. 17:11-13.
Ala-Houhala, M. 1985. 25-Hydroxyvitamin D levels during breastfeeding with or without maternal or infantile supplementation of vitamin D. J. Pediatr. Gastroenterol. Nutr. 4:220-226.
Arnaud, S.B., G.B. Stickler, and J.C. Haworth. 1976. Serum 25-hydroxyvitamin D in infantile rickets. Pediatrics 57:221-225.
Arnon, S.S., K. Damus, B. Thompson, T.F. Midura, and J. Chin. 1982. Protective role of human milk against sudden death from infant botulism. J. Pediatr. 100:568-573.
Axelsson, I., S. Borulf, L. Righard, and N. Räihä. 1987. Protein and energy intake during weaning. I. Effects on growth. Acta Paediatr. Scand. 76:321-327.
Bates, C.J., A.M. Prentice, A.A. Paul, A. Prentice, B.A. Sutcliffe, and R.G. Whitehead. 1982. Riboflavin status in infants born in rural Gambia, and the effect of a weaning food supplement. Trans. R. Soc. Trop. Med. Hyg. 76:253-258.
Bayley, N. 1969. Manual for the Bayley Scales of Infant Development. Psychological Corp, New York. 178 pp.
Belton, N. 1986. Rickets—not only the "English Disease." Acta Paediatr. Scand. Suppl. 323:68-75.
Berkowitz, R.I., W.S. Agras, A.F. Korner, H.C. Kraemer, and C.H. Zeanah. 1985. Physical activity and adiposity: a longitudinal study from birth to childhood. J. Pediatr. 106:734-738.
Biagi, P.L., A. Bordoni, M. Masi, G. Ricci, C. Fanelli, A. Patrizi, E. Ceccolino. 1988. A long-term study of the use of evening primrose oil (Efamol) in atopic children. Drugs Exp. Clin. Res. 14:285-290.
Biering-Sørensen, F., T. Jørgensen, and J. Hilden. 1978. Sudden infant death in Copenhagen 1956-1971. I. Infant feeding. Acta Paediatr. Scand. 67:129-137.
Birkbeck, J.A., P.M. Buckfield, and P.A. Silva. 1985. Lack of long-term effect of the method of infant feeding on growth. Hum. Nutr.: Clin. Nutr. 39C:39-44.
Bogardus, C., S. Lillioja, E. Ravussin, W. Abbott, J.K. Zawadzki, A. Young, W.C. Knowler, R. Jacobowitz, and P.P. Moll. 1986. Familial dependence of the resting metabolic rate. N. Engl. J. Med. 315:96-100.
Borch-Johnsen, K., G. Joner, T. Mandrup-Poulsen, M. Christy, B. Zachau-Christiansen, K. Kastrup, and J. Nerup. 1984. Relationship between breastfeeding and incidence rates of insulin-dependent diabetes mellitus. A hypothesis. Lancet 2:1083-1086.
Bordoni, A., P.L. Biagi, M. Masi, G. Ricci, C. Fanelli, A. Patrizi, and E. Ceccolini. 1988. Evening primrose oil (Efamol) in the treatment of children with atopic eczema. Drugs Exp. Clin. Res. 14:291-297.
Bouchard, C., A. Tremblay, J.P. Despres, A. Nadeau, P.J. Lupien, G. Theriault, J. Dussault, S. Moorjani, S. Pinault, and G. Fournier. 1990. The response to long-term overfeeding in identical twins. N. Engl. J. Med. 322:1477-1482.
Boxall, E.H., T.H. Flewett, D.S. Dane, C.H. Cameron, F.O. MacCallum, and T.W. Lee. 1974. Hepatitis-B surface antigen in breast milk. Lancet 2:1007-1008.
Bray, G.A. 1987. Obesity—a disease of nutrient or energy imbalance? Nutr. Rev. 45:33-43.
Brazelton, T.B. 1962. Crying in infancy. Pediatrics 29:579-588.
Briend, A., B. Wojtyniak, and M.G. Rowland. 1988. Breastfeeding, nutritional state, and child survival in rural Bangladesh. Br. Med. J. 296:879-882.
Briggs, G.G., R.K. Freeman, and S.J. Yaffe. 1986. Drugs in Pregnancy and Lactation. Williams & Wilkins, Baltimore. 495 pp.
Brown, K.H., R.E. Black, G. Lopez de Romaña, and H. Creed de Kanashiro. 1989. Infant-feeding practices and their relationship with diarrheal and other disease in Huascar (Lima), Peru . Pediatrics 83:31-40.
Brückmann, G., and S.G. Zondek. 1939. Iron, copper and manganese in human organs at various ages. Biochem. J. 33:1845-1857.
Buimovici-Klein, E., R.L. Hite, T. Byrne, and L.Z. Cooper. 1977. Isolation of rubella virus in milk after postpartum immunization. J. Pediatr. 91:939-941.
Butte, N.F. 1988. Energy requirements during infancy. Pp. 86-99 in R.C. Tsang and B.L. Nichols, eds. Nutrition During Infancy. Hanley & Belfus, Philadelphia.
Butte, N.F., C. Garza, E.O. Smith, and B.L. Nichols. 1984. Human milk intake and growth in exclusively breastfed infants. J. Pediatr. 104:187-195.
Butte, N.F., W.W. Wong, C. Garza, and P.D. Klein. 1990. Adequacy of human milk for meeting energy requirements during early infancy. Pp. 103-116 in S.A. Atkinson, L.A. Hanson, and R. Chandra, eds. Human Lactation 4: Breastfeeding, Nutrition, Infection and Infant Growth in Developed and Emerging Countries . ARTS Biomedical Publishers and Distributors. St. John's, Newfoundland, Canada.
Cant, A., R.A. Marsden, and P.J. Kilshaw. 1985. Egg and cow's milk hypersensitivity in exclusively breastfed infants with eczema, and detection of egg protein in breast milk. Br. Med. J. 291:932-935.
Cantrelle, P., and H. Leridon. 1971. Breastfeeding, mortality in childhood and fertility in a rural zone of Senegal. Popul. Stud. 25:505-533.
Carey, W.B. 1984. "Colic"—primary excessive crying as an infant-environmental interaction. Pediatr. Clin. North Am. 31:993-1005.
Carpenter, R.G., A. Gardner, M. Jepson, E.M. Taylor, A. Salvin, R. Sunderland, J.L. Emery, E. Pursall, and J. Roe. 1983. Prevention of unexpected infant death: evaluation of the first seven years of the Sheffield Intervention Programme. Lancet 1:723-727.
Carter, M.W. 1988. Radionuclides in the Food Chain. Springer-Verlag, New York, 518 pp.
Cavell, B. 1979. Gastric emptying in preterm infants. Acta Paediatr. Scand. 68:725-730.
Cavell, B. 1981. Gastric emptying in infants fed human milk or infant formula. Acta Paediatr. Scand. 70:639-641.
CDC (Centers for Disease Control). 1985. Recommendations for assisting in the prevention of perinatal transmission of human T-lymphotropic virus type III/lymphadenopathy-associated virus and acquired immunodeficiency syndrome. Morbid. Mortal. Wkly. Rep. 34:721-732.
Chan, G.M., C.C. Roberts, D. Folland, and R. Jackson. 1982. Growth and bone mineralization of normal breastfed infants and the effects of lactation on maternal bone mineral status. Am. J. Clin. Nutr. 36:438-443.
Chandra, R.K. 1982. Physical growth of exclusively breastfed-infants. Nutr. Res. 2:275-276.
Chandra, R.K. 1987. Prevention of atopic disease: environmental engineering utilizing antenatal antigen avoidance and breastfeeding. Pp. 269-274 in A.S. Goldman, S.A. Atkinson, and L.A. Hanson, eds. Human Lactation 3: The Effects of Human Milk on the Recipient Infant. Plenum Press, New York.
Chandra, R.K., S. Puri, and P.S. Cheema. 1985. Predictive value of cord blood IgE in the development of atopic disease and role of breastfeeding in its prevention. Clin. allergy 15:517-522.
Chasnoff, I.J., D.E. Lewis, and L. Squires. 1987. Cocaine intoxication in a breastfed infant. Pediatrics 80:836-838.
Chiba, Y., T. Minagawa, K. Mito, A. Nakane, K. Suga, T. Honjo, and T. Nakao. 1987. Effect of breastfeeding on responses of systemic interferon and virus-specific lymphocyte transformation in infants with respiratory syncytial virus infection. J. Med. Virol. 21:7-14.
Clemens, J.B., B. Stanton, B. Stoll, N.S. Shahid, H. Banu, and A.K.M.A. Chowdhury. 1986. Breastfeeding as a determinant of severity in shigellosis: evidence for protection throughout the first three years of life in Bangladeshi children. Am. J. Epidemiol. 123:710-720.
Clemens, T.L., J.S. Adams, S.L. Henderson, and M.F. Holick. 1982. Increased skin pigment reduces capacity of skin to synthesize vitamin D3. Lancet 1:74-76.
Close, G.Ć. 1983. Rastafarianism and the vegans syndrome. Br. Med. J. 286:473.
Cobo, E. 1973. Effect of different doses of ethanol on the milk-ejecting reflex in lactating women. Am. J. Obstet. Gynecol. 115:817-821.
Committee on Drugs. 1989. Transfer of drugs and other chemicals into human milk. Pediatrics 84:924-936.
Committee on Nutrition. 1985. Pediatric Nutrition Handbook, 2nd ed. American Academy of Pediatrics, Elk Grove Village, Ill. 421 pp.
Committee on Nutrition. 1986. Fluoride supplementation. Pediatrics 77:758-761.
Cunningham, A.S. 1981. Breastfeeding and morbidity in industrialized countries: an update. Pp. 128-168 in D.B. Jelliffe and E.F.P. Jelliffe, eds. Advances in International Maternal and Child Health, Vol. 1. Oxford University Press, Oxford.
Cushing, A.H., and L. Anderson. 1982. Diarrhea in breastfed and nonbreastfed infants. Pediatrics 70:921-925.
Czajka-Narins, D.M., and E. Jung. 1986. Physical growth of breastfed and formula-fed infants from birth to age two years. Nutr. Res. 6:753-762.
Dabeka, R.W., K.F. Karpinskl, A.D. McKenzie, and C.D. Bajdik. 1986. Survey of lead, cadmium and fluoride in human milk and correlation of levels with environmental and food factors. Food Chem. Toxicol. 24:913-921.
Dallman, P.R. 1986. Iron deficiency in the weanling: a nutritional problem on the way to resolution. Acta Paediatr. Scand. Suppl. 323:59-67.
Davis, J.R., Jr., J. Goldenring, and B.H. Lubin. 1981. Nutritional vitamin B12 deficiency in infants. Am. J. Dis. Child. 135:566-567.
Davis, M.K. 1989. The role of human milk in human immunodeficiency virus infection. Pp. 151-160 in S.A. Atkinson, L.A. Hanson, and R.K. Chandra, eds. Human Lactation 4: Breastfeeding, Nutrition, Infection and Infant Growth in Developed and Emerging Countries. ARTS Biomedical Publishers and Distributors, St. John's, Newfoundland, Canada.
Davis, M.K., D.A. Savitz, and B.I. Graubard. 1988. Infant feeding and childhood cancer . Lancet 2:365-368.
Delgado, H.L., V.E. Valverde, R. Martorell, and R.E. Klein. 1982. Relationship of maternal and infant nutrition to infant growth. Early Hum. Dev. 6:273-286.
Department of National Health and Welfare, Canada. 1963. Survey of Maternal and Child Health of Canadian Registered Indians 1962. Cited in Gerrard, J.W., and K.K.T. Tan. 1978. Hazards of formula feeding. Keeping Abreast. J. Hum. Nurturing. 3:20-25.
Dewey, K.G., and B. Lönnerdal. 1983. Milk and nutrient intake of breastfed infants from 1 to 6 months: Relation to growth and fatness. J. Pediatr. Gastroenterol. Nutr. 2:497-506.
Dewey, K.G., M.J. Heinig, L.A. Nommsen, and B. Lönnerdal. 1989. Infant growth and breastfeeding. Am. J. Clin. Nutr. 50:1116-1117.
Dewey, K.G., M.J. Heinig, L.A. Nommsen, and B. Lönnerdal. 1990a. Growth patterns of breastfed infants during the first year of life: the DARLING study. Pp. 269-282 in S.A. Atkinson, L.A. Hanson, and R. Chandra, eds. Human Lactation 4: Breastfeeding, Nutrition, Infection and Infant Growth in Developed and Emerging Countries . ARTS Biomedical Publishers and Distributors, St. John's, Newfoundland, Canada.
Dewey, K.G., M.J. Heinig, L.A. Nommsen, and B. Lönnerdal. 1990b. Low energy intakes and growth velocities of breastfed infants: are there functional consequences? In B. Schürch and N.S. Scrimshaw, eds. Proceedings of an IDECG Workshop. IDECG - Nestle Foundation, Lausanne, Switzerland.
Dewey, K.G., M.J. Heinig, L.A. Nommsen, and B. Lönnerdal. In press. Maternal vs. infant factors related to breast milk intake and residual milk volume: the DARLING study. Pediatr.
DHHS (Department of Health and Human Services). 1988. The Surgeon General's Report on Nutrition and Health. DHHS (PHS) Publ. No. 88-50210. Public Health Service, U.S. Department of Health and Human Services. U.S. Government Printing Office, Washington, D.C. 727 pp.
Dillon, H.K., D.J. Wilson, and W. Schaffner. 1974. Lead concentrations in human milk. Am. J. Dis. Child. 128:491-492.
DiPietro, J., S.K. Larson, and S.W. Porges. 1987. Behavioral and heart rate pattern differences between breastfed and bottle-fed neonates. Dev. Psychol. 23:467-474.
Downham, M.A.P.S., R. Scott, D.G. Sims, J.K.G. Webb, and P.S. Gardner. 1976. Breastfeeding protects against respiratory syncytial virus infections. Br. Med. J. 2:274-276.
Dubois, S., D.E. Hill, and G.H. Beaton. 1979. An examination of factors believed to be associated with infantile obesity. Am. J. Clin. Nutr. 31:1997-2007.
Duffy, L.C., M. Riepenhoff-Talty, T.E. Byers, L.J. La Scolea, M.A. Zielezny, D.M. Dryja, and P.L. Ogra. 1986. Modulation of rotavirus enteritis during breastfeeding. Am. J. Dis. Child. 140:1164-1168.
Duncan, B., C. Schaefer, B. Sibley, and N.M. Fonseca. 1984. Reduced growth velocity in exclusively breastfed infants. Am. J. Dis. Child. 138:309-313.
Duncan, B., R.B. Schifman, J.J. Corrigan, Jr., and C. Schaefer. 1985. Iron and the exclusively breastfed infant from birth to six months. J. Pediatr. Gastroenterol. Nutr. 4:421-425.
Dworsky, M., M. Yow, S. Stagno, R.F. Pass, and C. Alford. 1983. Cytomegalovirus infection of breast milk and transmission in infancy. Pediatrics 72:295-299.
Ek, J., and E.M. Magnus. 1979. Plasma and red blood cell folate in breastfed infants. Acta Paediatr. Scand. 68:239-243.
Ek, J., and E. Magnus. 1982. Plasma and red cell folate values and folate requirements in formula-fed term infants. J. Pediatr. 100:738-744.
Elias, M.F., N.A. Nicolson, C. Bora, and J. Johnston. 1986. Sleep/wake patterns of breastfed infants in the first 2 years of life. Pediatrics 77:322-329.
Elliot, D.L., L. Goldberg, K.S. Kuehl, and C. Hanna. 1989. Metabolic evaluation of obese and nonobese siblings. J. Pediatr. 114:957-962.
FAO (Food and Agriculture Organization). 1988. Requirements of Vitamin A, Iron, Folate, and Vitamin B12. Report of a Joint FAO/WHO Expert Consultation. FAO Food and Nutrition Series No. 23. Food and Agriculture Organization, Rome. 107 pp.
Fedrick, J., and P. Adelstein. 1973. Influence of pregnancy spacing on outcome of pregnancy. Br. Med. J. 4:753-756.
Fergusson, D.M., A.L. Beautrais, and P.A. Silva. 1982. Breastfeeding and cognitive development in the first seven years of life. Soc. Sci. Med. 16:1705-1708.
Ferris, A.G., M.J. Laus, D.W. Hosmer, and V.A. Beal. 1980. The effect of diet on weight gain in infancy. Am. J. Clin. Nutr. 33:2635-2642.
Filer, L.J., Jr. 1968. Evaluation and reduction of risks: Problems and applications of individual control measures. Pediatrics 41:308.
Fomon, S.J. 1987. Reflections on infant feeding in the 1970s and 1980s. Am. J. Clin. Nutr. Suppl. 46:171-182.
Fomon, S.J., R.R. Rogers, E.E. Ziegler, S.E. Nelson, and L.N. Thomas. 1984. Indices of fatness and serum cholesterol at age eight years in relation to feeding and growth during early infancy. Pediatr. Res. 18:1233-1238.
Forsum, E., and A. Sadurskis. 1986. Growth, body composition and breast milk intake of Swedish infants during early life. Early Hum. Dev. 14:121-129.
Friedman, G., and S.J. Goldberg. 1975. Concurrent and subsequent serum cholesterols of breast- and formula-fed infants. Am. J. Clin. Nutr. 28:42-45.
Gambon, R.C., M.J. Lentze, and E. Rossi. 1986. Megaloblastic anemia in one of monozygous twins breastfed by their vegetarian mother. Eur. J. Pediatr. 145:570-571.
Garry, P.J., G.M. Owen, E.M. Hooper, and B.A. Gilbert. 1981. Iron absorption from human milk and formula with and without iron supplementation. Pediatr. Res. 15:822-828.
Garza, C., J. Stuff, and N. Butte. 1987. Growth of the breastfed infant. Pp. 109-121 in A.S. Goldman, S.A. Atkinson, and L.A. Hanson, eds. Human Lactation 3: The Effects of Human Milk on the Recipient Infant. Plenum Press, New York.
Gladen, B.C., W.J. Rogan, P. Hardy, J. Thullen, J. Tingelstad, and M. Tully. 1988. Development after exposure to polychlorinated biphenyls and dichlorodiphenyl dichloroethene transplacentally and through human milk. J. Pediatr. 113:991-995.
Glass, R.I., and B.J. Stoll. 1989. The protective effect of human milk against diarrhea: a review of studies from Bangladesh. Acta Paediatr. Scand. Suppl. 351:131-136.
Glass, R.I., A.M. Svennerholm, B.J. Stoll, M.R. Khan, K.M.B. Hossain, M.I. Huq, and J. Holmgren. 1983. Protection against cholera in breastfed children by antibodies in breast milk. N. Engl. J. Med. 308:1389-1392.
Gleason, W.A., Jr., and G.A. Kerr. 1989. Questions about quinones in infant nutrition. J. Pediatr. Gastroenterol. Nutr. 8:285-287.
Goldman, A.S., and R.M. Goldblum. 1989. Immunologic system in human milk: characteristics and effects. Pp. 135-142 in E. Lebenthal, ed. Textbook of Gastroenterology and Nutrition in Early Infancy, 2nd ed. Raven Press, New York.
Goldman, A.S., and R.M. Goldblum, 1990. Human milk: immunologic-nutritional relationships. Ann. N.Y. Acad. Sci. 587:236-245.
Goldman, A.S., L.W. Thorpe, R.M. Goldblum, and L.A. Hanson. 1986. Anti-inflammatory properties of human milk. Acta Paediatr. Scand. 75:689-695.
Goldman, A.S., R.M. Goldblum, and L.A. Hanson. 1990a. Anti-inflammatory systems in human milk. Adv. Exp. Med. Biol. 262:69-76.
Goldman, A.S., R.M. Goldblum, and L.A. Hanson. 1990b. Anti-inflammatory systems in human milk. Pp. 69-76 in A. Bendich, M. Phillips, and R.P. Tengerdy, eds. Antioxidant Nutrients and Immune Functions. Plenum Press, New York.
Greer, F.R., J.E. Searcy, R.S. Levin, J.J. Steichen, P.S. Steichen-Asche, and R.C. Tsang. 1982. Bone mineral content and serum 25-hydroxyvitamin D concentrations in breastfed infants with and without supplemental vitamin D: one year follow-up. J. Pediatr. 100:919-922.
Gromisch, D.S., R. Lopez, H.S. Cole, and J.M. Cooperman. 1977. Light (phototherapy)-induced riboflavin deficiency in the neonate. J. Pediatr. 90:118-122.
Habicht, J.-P., J. DaVanzo, W.P. Butz, and L. Meyers. 1985. The contraceptive role of breastfeeding. Popul. Stud. 39:213-232.
Hambidge, K.M., P.A. Walravens, C.E. Casey, R.M. Brown, and C. Bender. 1979. Plasma zinc concentrations of breastfed infants. J. Pediatr. 94:607-608.
Hamill, P.V.V., T.A. Drizd, C.L. Johnson, R.B. Reed, and A.F. Roche. 1977. NCHS Growth Curves for Children from Birth to 18 Years: United States. Vital and Health Statistics, Series 11, No. 165. DHHS Publ. No. (PHS) 78-1650. National Center for Health Statistics, Public Health Service, U.S. Department of Health, Education, and Welfare, Hyattsville, Md. 74 pp.
Hamosh, M. 1988. Does infant nutrition affect adiposity and cholesterol levels in the adult? J. Pediatr. Gastroenterol. Nutr. 7:10-16.
Hamosh, M., and P. Hamosh. 1987. Does nutrition in early life have long-term metabolic effects? Can animal models be used to predict these effects in the human? Pp. 37-55 in A.S. Goldman, S.A. Atkinson, and L.A. Hanson, eds. Human Lactation 3: The Effects of Human Milk on the Recipient Infant. Plenum Press, New York.
Hamosh, M., and P. Hamosh. 1990. Obesity. Annales Nestlé 48:59-69.
Hansen, A.E. 1937. Serum lipids in eczema and in other pathologic conditions. Am. J. Dis. Child. 53:933-946.
Hansen, A.E., E.M. Knott, H.F. Wiese, E. Shaperman, and I. McQuarrie. 1947. Eczema and essential fatty acids. Am. J. Dis. Child. 73:1-18.
Hanson, L.A. 1986. The global responsibility towards child health. Aust. Paediatr. J. 22:157-159.
Harmatz, P.R., D.G. Hanson, M. Brown, R.E. Kleinman, W.A. Walker, and K.J. Bloch. 1987. Transfer of maternal food proteins in milk. Pp. 289-299 in A.S. Goldman, S.A. Atkinson, and L.A. Hanson, eds. Human Lactation 3: The Effects of Human Milk on the Recipient Infant. Plenum Press, New York.
Hatano, S., K. Aihara, Y. Nishi, and T. Usui. 1985. Trace elements (copper, zinc, manganese, and selenium) in plasma and erythrocytes in relation to dietary intake during infancy. J. Pediatr. Gastroenterol. Nutr. 4:87-92.
Hattevig, G., B. Kjellman, N. Sigurs, E. Grodzinsky, J. Hed, and B. Bjorksten. 1990. The effect of maternal avoidance of eggs, cow's milk, and fish during location on the development of IgE, IgG, and IgA antibodies in infants. J. Allergy Clin. Immunol. 85:108-115.
Herrera, M.G., J.O. Mora, B. de Paredes, and M. Wagner. 1980. Maternal weight/height and the effect of food supplementation during pregnancy and lactation. Pp. 252-263 in H. Aebi and R. Whitehead, eds. Maternal Nutrition During Pregnancy and Lactation. Hans Huber Publishers, Bern.
Higginbottom, M.C., L. Sweetman, and W.L. Nyhan. 1978. A syndrome of methylmalonic aciduria, homocystinuria, megaloblastic anemia and neurologic abnormalities in a vitamin B-12-deficient breastfed infant of a strict vegetarian . N. Engl. J. Med. 299:317-323.
Hillman, L.S., and J.G. Haddad. 1974. Human perinatal vitamin D metabolism I: 25-hydroxyvitamin D in maternal and cord blood. J. Pediatr. 84:742-749.
Himes, J.H. 1979. Infant feeding practices and obesity. J. Am. Diet. Assoc. 75:122-125.
Hino, S. 1989. Milk-borne transmission of HTLV-I as a major route in the endemic cycle. Acta Paediatr. Jpn. 31:428-435.
Hitchcock, N.E., and J.F. Coy. 1989. The growth of healthy Australian infants in relation to infant feeding and social group. Med. J. Aust. 150:306-311.
Hitchcock, N.E., M. Gracey, and A.I. Gilmour. 1985. The growth of breastfed and artificially fed infants from birth to twelve months. Acta Paediatr. Scand. 74:240-245.
Hobcraft, J. 1987. Does family planning save children's lives? Technical background paper prepared for the International Conference on Better Health for Women and Children Through Family Planning, Nairobi, Kenya, October, 1987. Population Council, New York. 77 pp.
Hodgson, P.A., R.D. Ellefson, L.R. Elveback, L.E. Harris, R.A. Nelson, and W.H. Weidman. 1976. Comparison of serum cholesterol in children fed high, moderate, or low cholesterol milk diets during neonatal period. Metabolism 25:739-746.
Hofvander, Y., U. Hagman, C. Hillervik, and S. Sjolin. 1982. The amount of milk consumed by 1-3 months old breast- or bottle-fed infants. Acta Paediatr. Scand. 71:953-958.
Hoogenboezem, T., H.J. Degenhart, S.M.P.F. de Muinck Keizer-Schrama, R. Bouillon, W.F.A. Grose, W.H.L. Hackeng, and H.K.A. Visser. 1989. Vitamin D metabolism in breastfed infants and their mothers. Pediatr. Res. 25:623-628.
Hovi, L., R. Hekali, and M.A. Siimes. 1979. Evidence of riboflavin depletion in breastfed newborns and its further acceleration during treatment of hyperbilirubinemia by phototherapy. Acta Paediatr. Scand. 68:567-570.
Hunziker, U.A., and R.G. Barr. 1986. Increased carrying reduces infant crying: a randomized controlled trial. Pediatrics 77:641-648.
Huttunen, J.K., U.M. Saarinen, E. Kostiainen, and M.A. Siimes. 1983. Fat composition of the infant diet does not influence subsequent serum lipid levels in man. Atherosclerosis 46:87-94.
Jadhav, M., J.K.G. Webb, S. Vaishnava, and S.J. Baker. 1962. Vitamin-B 12 deficiency in Indian infants: a clinical syndrome. Lancet 2:903-907.
Jakobsson, I., and T. Lindberg. 1978. Cow's milk as a cause of infantile colic in breastfed infants. Lancet 2:437-439.
Jakobsson, I., and T. Lindberg. 1983. Cow's milk proteins cause infantile colic in breastfed infants: a double-blind crossover study. Pediatrics 71:268-271.
Jason, J.M., P. Nieburg, and J.S. Marks. 1984. Mortality and infectious disease associated with infant-feeding practices in developing countries. Pediatrics 74:702-727.
Jensen, A.A. 1983. Chemical contaminants in human milk. Residue Rev. 89:1-128.
Kilshaw, P.J., and A.J. Cant. 1984. The passage of maternal dietary proteins into human breast milk. Int. Arch. Allergy Appl. Immunol. 75:8-15.
Klein, E.B., T. Byrne, and L.Z. Cooper. 1980. Neonatal rubella in a breastfed infant after postpartum maternal infection. J. Pediatr. 97:774-775.
Koldovskỳ, O., A. Bedrick, P. Pollack, R.K. Rao, and W. Thornburg. 1988. Possible physiological role of hormones and hormone-related substances present in milk. Pp. 123-139 in L.A. Hanson, ed. Biology of Human Milk. Nestle Nutrition Workshop Series, Vol. 15. Raven Press, New York.
Koletzko, S., P. Sherman, M. Corey, A. Griffiths, and C. Smith. 1989. Role of infant feeding practices in development of Crohn's disease in childhood. Br. Med. J. 298:1617-1618.
Kovar, M.G., M.K. Serdula, J.S. Marks, and D.W. Fraser. 1984. Review of the epidemiologic evidence for an association between infant feeding and infant health. Pediatrics 74:615-638.
Kramer, M.S. 1981. Do breastfeeding and delayed introduction of solid foods protect against subsequent obesity? J. Pediatr. 98:883-887.
Kramer, M.S. 1987. Breastfeeding and child health: methodologic issues in epidemiologic research. Pp. 339-360 in A.S. Goldman, S.A. Atkinson, and L.A. Hanson, eds. Human Lactation 3: The Effects of Human Milk on the Recipient Infant. Plenum Press, New York.
Kramer, M.S. 1988. Does breastfeeding help protect against atopic disease? Biology, methodology, and a golden jubilee of controversy. J. Pediatr. 112:181-190.
Lakdawala, D.R., and E.M. Widdowson. 1977. Vitamin-D in human milk. Lancet 1:167-168.
Lampkin, B.C., and E.F. Saunders. 1969. Nutritional vitamin B12 deficiency in an infant. J. Pediatr. 75:1053-1055.
Laug, E.P., F.M. Kunze, and C.S. Prickett. 1951. Occurrence of DDT in human fat and milk. Arch. Ind. Hyg. 3:245-246.
Lawrence, R.A. 1989. Breastfeeding: A Guide for the Medical Profession, 3rd ed. C.V. Mosby, St. Louis. 652 pp.
Lechtig, A., and R.E. Klein. 1980. Maternal food supplementation and infant health: results of a study in rural areas of Guatemala. Pp. 285-313 in H. Aebi and R. Whitehead, eds. Maternal Nutrition During Pregnancy and Lactation. Hans Huber, Bern.
Lewis, D.S., H.A. Bertrand, E.J. Masoro, H.C. McGill, Jr., K.D. Carey, and C.A. McMahan. 1983. Preweaning nutrition and fat development in baboons. J. Nutr. 113:2253-2259.
Lichtenstein, P., B.L. Specker, R.C. Tsang, F. Mimouni, and C. Gormley. 1986. Calcium-regulating hormones and minerals from birth to 18 months of age: a cross-sectional study. I. Effects of sex, race, age, season, and diet on vitamin D status. Pediatrics 77:883-890.
Linnemann, C.C., Jr., and S. Goldberg. 1974. HBAg in breast milk. Lancet 2:155.
Little, R.E., K.W. Anderson, C.H. Ervin, B. Worthington-Roberts, and S.K. Clarren. 1989. Maternal alcohol use during breastfeeding and infant mental and motor development at one year. N. Engl. J. Med. 321:425-430.
Lönnerdal, B., C.L. Keen, and L.S. Hurley. 1981. Iron, copper, zinc, and manganese in milk. Annu. Rev. Nutr. 1:149-174.
Lönnerdal, B., C.L. Keen, and L.S. Hurley. 1984. Zinc binding ligands and complexes in zinc metabolism. Adv. Nutr. Res. 6:139-167.
Losonsky, G.A., J.M. Fishaut, J. Strussenberg, and P.L. Ogra. 1982. Effect of immunization against rubella on lactation products. II. Maternal-neonatal interactions. J. Infect. Dis. 145:661-666.
Lucas, A., P.J. Lucas, and J.D. Baum. 1981a. Differences in the pattern of milk intake between breast and bottle fed infants. Early Hum. Dev. 5:195-199.
Lucas, A., S. Boyes, S.R. Bloom, and A. Aynsley-Green. 1981b. Metabolic and endocrine responses to a milk feed in six-day-old term infants: differences between breast and cow's milk formula feeding. Acta Paediatr. Scand. 70:195-200.
Luck, W., and H. Nau. 1985. Nicotine and cotinine concentrations in serum and urine of infants exposed via passive smoking or milk from smoking mothers. J. Pediatr. 107:816-820.
Luck, W., and H. Nau. 1987. Nicotine and cotinine concentrations in the milk of smoking mothers: influence of cigarette consumption and diurnal variation. Eur. J. Pediatr. 146:21-26.
MacDonald, L.D., R.S. Gibson, and J.E. Miles. 1982. Changes in hair zinc and copper concentrations of breastfed and bottle fed infants during the first six months. Acta Paediatr. Scand. 71:785-789.
Mahmood, D.A., and R.G. Feachem. 1987. Feeding and nutritional status among infants in Basrah City, Iraq: a cross-sectional study. Hum. Nutr.: Clin. Nutr. 41C:373-381.
Manku, M.S., D.F. Horrobin, N. Morse, V. Kyte, K. Jenkins, S. Wright, and J.L. Burton. 1982. Reduced levels of prostaglandin precursors in the blood of atopic patients: defective delta-6-desaturase function as a biochemical basis for atopy. Prostaglandins, Leukot. Med. 9:615-628.
Markestad, T. 1983. Effect of season and vitamin D supplementation on plasma concentrations of 25-hydroxyvitamin D in Norwegian infants. Acta Paediatr. Scand. 72:817-821.
Markestad, T., L. Aksnes, P.H. Finne, and D. Aarskog. 1983. vitamin D nutritional status of premature infants supplemented with 500 IU vitamin D2 per day. Acta Paediatr. Scand. 72:517-520.
Marmot, M.G., C.M. Page, E. Atkins, and J.W.B. Douglas. 1980. Effect of breastfeeding on plasma cholesterol and weight in young adults. J. Epidemiol. Commun. Health 34:164-167.
Mason, K.E. 1979. A conspectus of research on copper metabolism and requirements of man. J. Nutr. 109:1979-2066.
Mata, L.J., and J.J. Urrutia. 1971. Intestinal colonization of breastfed children in a rural area of low socioeconomic level. Ann. N.Y. Acad. Sci. 176:93-109.
Mata, L.J., J.J. Urrutia, and J.E. Gordon. 1967. Diarrhoeal disease in a cohort of Guatemalan village children observed from birth to age two years. Trop. Geogr. Med. 19:247-257.
Mata, L.J., J.J. Urrutia, B. García, R. Fernández, and M. Behar. 1969. Shigella infection in breastfed Guatemalan Indian neonates. Am. J. Dis. Child. 117:142-146.
Mayer, E.J., R.F. Hamman, E.C. Gay, D.C. Lezotte, D.A. Savitz, and G.J. Klingensmith. 1988. Reduced risk of IDDM among breastfed children: the Colorado IDDM Registry. Diabetes 37:1625-1632.
McPhee, A.J., G.P. Davidson, M. Leahy, and T. Beare. 1988. Vitamin B12 deficiency in a breastfed infant. Arch. Dis. Child. 63:921-923.
Meigel, W., T. Dettke, E.M. Meigel, and U. Lenze. 1987. Additional oral treatment of atopical dermatitis with unsaturated fatty acids. Z. Hautkr. 62 Suppl. 1:100-103.
Menard, D., and P. Arsenault. 1988. Epidermal and neural growth factors in milk: effects of epidermal growth factor on the development of the gastrointestinal tract. Pp. 105-122 in Biology of Human Milk. Nestle Nutrition Workshop Series, Vol. 15. Raven Press, New York.
Menkes, J.H. 1977. Early feeding history of children with learning disorders. Dev. Med. Child Neurol. 19:169-171.
Morley, R., T.J. Cole, R. Powell, and A. Lucas. 1988. Mother's choice to provide breast milk and developmental outcome. Arch. Dis. Child. 63:1382-1385.
Morrow-Tlucak, M., R.H. Haude, and C.B. Ernhart. 1988. Breastfeeding and cognitive development in the first 2 years of life. Soc. Sci. Med. 26:635-639.
Morse, P.F., D.F. Horrobin, M.S. Manku, J.C.M. Stewart, R. Allen, S. Littlewood, S. Wright, J. Burton, D.J. Gould, P.J. Holt, C.T. Jansen, L. Mattila, W. Meigel, T. Dettke, D. Wexler, L. Guenther, A. Bordoni, and A. Patrizi. 1989. Meta-analysis of placebo-controlled studies of the efficacy of Epogam in the treatment of atopic eczema: relationship between plasma essential fatty acid changes and clinical response. Br. J. Dermatol. 121:75-90.
Motohara, K., I. Matsukane, F. Endo, Y. Kiyota, and I. Matsuda. 1989. Relationship of milk intake and vitamin K supplementation to vitamin K status in newborns. Pediatrics 84:90-93.
Mott, G.E., E.M. Jackson, C.A. McMahan, and H.C. McGill, Jr. 1990. Cholesterol metabolism in adult baboons is influenced by infant diet. J. Nutr. 120:243-251.
Mueller, W.H. 1983. The genetics of human fatness. Yearb. Phys. Anthropol. 26:215-230.
Munch-Peterson, S. 1950. On the copper content in mother's milk before and after intravenous copper administration. Acta Paediatr. 39:378-388.
Muñoz, L., C.L. Keen, B. Lönnerdal, and K.G. Dewey. 1986. Coffee intake during pregnancy and lactation in rats: maternal and pup hematological parameters and liver iron, zinc and copper concentration. J. Nutr. 116:1326-1333.
Muñoz, L.M., B. Lönnerdal, C.L. Keen, and K.G. Dewey. 1988. Coffee consumption as a factor in iron deficiency anemia among pregnant women and their infants in Costa Rica . Am. J. Clin. Nutr. 48:645-651.
Murray, M.J., A.B. Murray, N.J. Murray, and M.B. Murray. 1978. The effect of iron status of Nigerian mothers on that of their infants at birth and 6 months, and on the concentration of Fe in breast milk. Br. J. Nutr. 39:627-630.
Myers, M.G., S.J. Fomon, F.P. Koontz, G.A. McGuinness, P.A. Lachenbruch, and R. Hollingshead. 1984. Respiratory and gastrointestinal illnesses in breast formula-fed infants. Am. J. Dis. Child. 138:629-632.
Naeye, R.L., B. Ladis, and J.S. Drage. 1976. Sudden infant death syndrome: a prospective study. Am. J. Dis. Child. 130:1207-1210.
Nail, P.A., M.R. Thomas, and R. Eakin. 1980. The effect of thiamin and riboflavin supplementation on the level of those vitamins in human breast milk and urine. Am. J. Clin. Nutr. 33:198-204.
Nelson, S.E., R.R. Rogers, E.E. Ziegler, and S.J. Fomon. 1989. Gain in weight and length during early infancy. Early Hum. Dev. 19:223-239.
Newburg, D.S., and R.H. Yolken. 1990. A human-milk fraction inhibits the binding of GP120 to CD4. Pediatr. Res. (abstract) 27:274.
NRC (National Research Council). 1989. Recommended Dietary Allowances, 10th ed. Report of the Subcommittee on the Tenth Edition of the RDAs, Food and Nutrition Board, Commission on Life Sciences. National Academy Press, Washington, D.C. 284 pp.
Nutrition Foundation. 1984. Nutrition Reviews' Present Knowledge in Nutrition, 5th ed. The Nutrition Foundation, Washington, D.C. 900 pp.
O'Connor, P. 1977. Vitamin D-deficiency rickets in two breastfed infants who were not receiving vitamin D supplementation. Clin. Pediatr. 16:361-363.
Ohtake, M. 1977. Serum zinc and copper levels in healthy Japanese infants. Tohoku J. Exp. Med. 123:265-270.
Oski, F.A., and S.A. Landaw. 1980. Inhibition of iron absorption from human milk by baby food. Am. J. Dis. Child. 134:459-460.
Owen, G.M., C.E. Nelsen, G.I. Baker, and W.E. Connor. 1967. Use of vitamin K1 in pregnancy. Effect on serum bilirubin and plasma prothrombin in the newborn. Am. J. Obstet. Gynecol. 99:368-373.
Owen, G.M., P.J. Garry, E.M. Hooper, B.A. Gilbert, and D. Pathak. 1981. Iron nutriture of infants exclusively breastfed the first five months. J. Pediatr. 99:237-240.
Ozsoylu, S. 1977. Breastfeeding and rickets. Lancet 2:560.
Palloni, A., and M. Tienda. 1986. The effects of breastfeeding and pace of childbearing on mortality at early ages. Demography 23:31-52.
Palti, H., I. Mansbach, H. Pridan, B. Adler, and Z. Palti. 1984. Episodes of illness in breastfed and bottle-fed infants in Jerusalem. Isr. J. Med. Sci. 20:395-399.
Pastel, R.A., P.J. Howanitz, and F.A. Oski. 1981. Iron sufficiency with prolonged exclusive breastfeeding in Peruvian infants. Clin. Pediatr. 20:625-626.
Perkins, K.C., and F.A. Oski. 1976. Elevated blood lead in a 6-month-old breastfed infant: the role of newsprint logs. Pediatrics 57:426-427.
Picciano, M.F., and R.H. Deering. 1980. The influence of feeding regimens on iron status during infancy. Am. J. Clin. Nutr. 33:746-753.
Plank, S.J., and M.L. Milanesi. 1973. Infant feeding and infant mortality in rural Chile. Bull. W.H.O. 48:203-210.
Podratz, R.O., D.D. Broughton, D.H. Gustafson, E.J. Bergstralh, and L.J. Melton. 1986. Weight loss and body temperature changes in breastfed and bottle-fed neonates. Clin. Pediatr. 25:73-77.
Pomerance, H.H. 1987. Growth in breastfed children. Hum. Biol. 59:687-693.
Pullan, C.R., G.L. Toms, A.J. Martin, P.S. Gardner, J.K.G. Webb, and D.R. Appleton. 1980. Breastfeeding and respiratory syncytial virus infection. Br. Med. J. 281:1034-1036.
Ravussin, E., S. Lillioja, W.C. Knowler, L. Christin, D. Freymond, W.G. Abbott, V. Boyce, B.V. Howard, and C. Bogardus. 1988. Reduced rate of energy expenditure as a risk factor for body-weight gain. N. Engl. J. Med. 318:467-472.
Rench, M.A., and C.J. Baker. 1989. Group B streptococcal breast abscess in a mother and mastitis in her infant. Obstet. Gynecol. 73:875-877.
Rendle-Short, J., J.R. Tiernan, and S. Hawgood. 1979. Vegan mothers with vitamin B12 deficiency. Med. J. Aust. 2:483.
Rivera-Calimlim, L. 1987. The significance of drugs in breast milk: pharmacokinetic considerations. Clin. Perinatol. 14:51-70.
Roberts, C.C., G.M. Chan, D. Folland, C. Rayburn, and R. Jackson. 1981. Adequate bone mineralization in breastfed infants. J. Pediatr. 99:192-196.
Roberts, S.B., J. Savage, W.A. Coward, B. Chew, and A. Lucas. 1988. Energy expenditure and intake in infants born to lean and overweight mothers. N. Engl. J. Med. 318:461-466.
Roche, A.F., S. Guo, and W.M. Moore. 1989. Weight and recumbent length from 1 to 12 mo of age: reference data for 1-mo increments. Am. J. Clin. Nutr. 49:599-607.
Rocklin, R.E., L. Thistle, L. Gallant, M.S. Manku, and D. Horrobin. 1986. Altered arachidonic acid content in polymorphonuclear and mononuclear cells from patients with allergic rhinitis and/or asthma. Lipids 21:17-20.
Rodgers, B. 1978. Feeding in infancy and later ability and attainment: a longitudinal study. Dev. Med. Child Neurol. 20:421-426.
Rogan, W.J., and B.C. Gladen. 1985. Study of human lactation for effects of environmental contaminants: the North Carolina Breast Milk and Formula Project and some other ideas. Environ. Health Perspect. 60:215-221.
Rogan, W.J., B.C. Gladen, J.D. McKinney, N. Carreras, P. Hardy, J. Thullen , J. Tinglestad, and M. Tully. 1986. Neonatal effects of transplacental exposure to PCBs and DDE. J. Pediatr. 109:335-341.
Rogan, W.J., B.C. Gladen, J.D. McKinney, N. Carreras, P. Hardy, J. Thullen, J. Tingelstad, and M. Tully. 1987. Polychlorinated biphenyls (PCBs) and dichlorodiphenyl dichloroethane (DDE) in human milk: effects on growth, morbidity, and duration of lactation. Am. J. Public Health 77:1294-1297.
Rosenthal, H.L., S. Austin, S. O'Neill, K. Takeuchi, J.T. Bird, and J.E. Gilster. 1964. Incorporation of fallout strontium-90 in deciduous incisors and foetal bone. Nature 203:615-616.
Ruiz-Palacios, G.M., Y. Lopez-Vidal, J. Calva, T.G. Cleary, and L.K. Pickering. 1986. Impact of breastfeeding on diarrhea prevention. Pediatr. Res. 20:320A (abstract).
Ryu, J., E.E. Ziegler, and S.J. Fomon. 1978. Maternal lead exposure and blood lead concentrations in infancy. J. Pediatr. 93:476-478.
Saarinen, U.M. 1982. Prolonged breastfeeding as prophylaxis for recurrent otitis media. Acta Paediatr. Scand. 71:567-571.
Saarinen, U.M., and M.A. Siimes. 1979a. Iron absorption from breast milk, cow's milk, and iron-supplemented formula: an opportunistic use of changes in total body iron determined by hemoglobin, ferritin, and body weight in 132 infants. Pediatr. Res. 13:143-147.
Saarinen, U.M., and M.A. Siimes. 1979b. Role of prolonged breastfeeding in infant growth. Acta Paediatr. Scand. 68:245-250.
Saarinen, U.M., M.A. Siimes, and P.R. Dallman. 1977. Iron absorption in infants: high bioavailability of breast milk iron as indicated by the extrinsic tag method of iron absorption and by the concentration of serum ferritin. J. Pediatr. 91:36-39.
Salmenperä, L., J. Perheentupa, and M.A. Siimes. 1985. Exclusively breastfed healthy infants grow slower than reference infants. Pediatr. Res. 19:307-312.
Salmenperä, L., J. Perheentupa, P. Pakarinen, and M.A. Siimes. 1986a. Cu nutrition in infants during prolonged exclusive breastfeeding: low intake but rising serum concentrations of Cu and ceruloplasmin. Am. J. Clin. Nutr. 43:251-257.
Salmenperä, L., J. Perheentupa, and M.A. Siimes. 1986b. Folate nutrition is optimal in exclusively breastfed infants but inadequate in some of their mothers and in formula-fed infants. J. Pediatr. Gastroenterol. Nutr. 5:283-289.
Sandström, B., A. Cederblad, and B. Lönnerdal. 1983. Zinc absorption from human milk, cow's milk, and infant formulas. Am. J. Dis. Child. 137:726-729.
Schaefer, O. 1971. Otitis media and bottle-feeding. An epidemiological study of infant feeding habits and incidence of recurrent and chronic middle ear disease in Canadian Eskimos. Can. J. Public Health 62:478-489.
Schecter, A., and T.A. Gasiewicz. 1987. Health hazard assessment of chlorinated dioxins and dibenzofurans contained in human milk. Chemosphere 16:2147-2154.
Schecter, A.J., J.J. Ryan, and J.D. Constable. 1986. Chlorinated dibenzo-p-dioxin and dibenzofuran levels in human adipose tissue and milk samples from north and south of Vietnam. Chemosphere 15:1613-1620.
Schecter, A., J.J. Ryan, and J.D. Constable. 1987. Polychlorinated dibenzo-p-dioxin and polychlorinated dibenzofuran levels in human breast milk from Vietnam compared with cow's milk and human breast milk from North American continent. Chemosphere 16:2003-2016.
Seward, J.F., and M.K. Serdula. 1984. Infant feeding and infant growth. Pediatrics 74:728-762.
Shannon, W.R. 1921. Demonstration of food proteins in human breast milk by anaphylactic experiments on guinea-pigs. Am. J. Dis. Child. 22:223-231.
Sheard, N.F., and W.A. Walker. 1988. The role of breast-milk in the development of the gastrointestinal tract. Nutr. Rev. 46:1-8.
Shepherd, R.W., D.B. Oxborough, T.L. Holt, B.J. Thomas, and Y.H. Thong. 1988. Longitudinal study of the body composition of weight gain in exclusively breastfed and intake-measured whey-based formula-fed infants to age 3 months. J. Pediatr. Gastroenterol. Nutr. 7:732-739.
Shukla, A., H.A. Forsythe, C.M. Anderson, and S.M. Marwok. 1972. Infantile over nutrition in the first year of life: a field study in Dudley, Worchestershire. Br. Med. J. 4:507-515.
Siimes, M.A., L. Salmenperä, and J. Perheentupa. 1984. Exclusive breastfeeding for 9 months: risk of iron deficiency. J. Pediatr. 104:196-199.
Sklar, R. 1986. Nutritional vitamin B-12 deficiency in a breastfed infant of a vegan-diet mother. Clin. Pediatr. 25:219-221.
Smith, A.M., M.F. Picciano, and R.H. Deering. 1983. Folate supplementation during lactation: maternal folate status, human milk folate content, and their relationship to infant folate status. J. Pediatr. Gastroenterol. Nutr. 2:622-628.
Smith, A.M., M.F. Picciano, and R.H. Deering. 1985. Folate intake and blood concentrations of term infants. Am. J. Clin. Nutr. 41:590-598.
Snyderman, S.E., K.C. Ketron, H.B. Burch, O.H. Lowry, O.A. Bessey, L.P. Guy, and L.E. Holt, Jr. 1949. The minimum riboflavin requirement of the infant. J. Nutr. 39:219-232.
Sommer, A. 1982. Nutritional Blindness: Xerophthalmia and Keratomalacia. Oxford University Press, New York. 282 pp.
Specker, B.L., B. Valanis, V. Hertzberg, N. Edwards, and R.C. Tsang. 1985. Sunshine exposure and serum 25-hydroxyvitamin D concentrations in exclusively breastfed infants. J. Pediatr. 107:372-376.
Specker, B.L., D. Miller, E.J. Norman, H. Greene, and K.C. Hayes. 1988. Increased urinary methylmalonic acid excretion in breastfed infants of vegetarian mothers and identification of an acceptable dietary source of vitamin B12. Am. J. Clin. Nutr. 47:89-92.
Stagno, S., D.W. Reynolds, R.F. Pass, and C.A. Alford. 1980. Breast milk and the risk of cytomegalovirus infection. N. Engl. J. Med. 302:1073-1076.
Staub, C.P., and G.K. Murphy. 1965. A comparison of Sr 90 component of human and cow's milk. Pediatrics 36:732.
Stunkard, A.J., T.I. Sorensen, C. Hanis, T.W. Teasdale, R. Chakraborty, W.J. Schull, and F. Schulsinger. 1986. An adoption study of human obesity. N. Engl. J. Med. 314:193-198.
Stunkard, A.J., J.R. Harris, N.L. Pedersen, and G.E. McClearn. 1990. The body-mass index of twins who have been reared apart. N. Engl. J. Med. 322:1483-1487.
Sveger, T., H. Linberg, B. Weilbull, V.L. Olsson. 1975. Nutrition, over nutrition and obesity in the first year of life in Malmo, Sweden. Acta Paediatr. Scand. 64:635-640.
Taitz, L.S. 1977. Obesity in pediatric practice: infantile obesity. Pediatr. Clin. North Am. 24:107-115.
Tan, K.L., M.T. Chow, and S.M.M. Karim. 1978. Effect of phototherapy on neonatal riboflavin status. J. Pediatr. 93:494-497.
Taylor, B., and J. Wadsworth. 1984. Breastfeeding and child development at five years. Dev. Med. Child Neurol. 26:73-80.
Thapa, S., R.V. Short, and M. Potts. 1988. Breastfeeding, birth spacing and their effects on child survival. Nature 335:679-682.
Thiry, L., S. Sprecher-Goldenberg, T. Jonckheer, J. Levy, P. Van de Perre, P. Henrivaux, J. Cogniaux-LeClerc, and N. Clumeck. 1985. Isolation of AIDS virus from cell-free breast milk of three healthy virus carriers. Lancet 2:891-892.
Thomsen, A.C., T. Espersen, and S. Maigaard. 1984. Course and treatment of milk stasis, noninfectious inflammation of the breast, and infectious mastitis in nursing women. Am. J. Obstet. Gynecol. 149:492-495.
Unni, J.C., and J. Richard. 1988. Growth and morbidity of breastfed and artificially-fed infants in urban south Indian families. J. Trop. Pediatr. 34:179-181.
Vigi, V., R. Chierici, L. Osti, F. Fagioli, and R. Rescazzi. 1984. Serum zinc concentration in exclusively breastfed infants and in infants fed an adapted formula. Eur. J. Pediatr. 142:245-247.
Vobecky, J.S., J. Vobecky, D. Shapcott, and P.P. Demers. 1983. Nutrient intake patterns and nutritional status with regard to relative weight in early infancy . Am. J. Clin. Nutr. 38:730-738.
Vogt, M.W., D.J. Witt, D.E. Craven, R. Byington, D.F. Crawford, R.T. Schooley, and M.S. Hirsch. 1986. Isolation of HTLV-III/LAV from cervical secretions of women at risk for AIDS. Lancet 1:525-527.
Vuori, E., S.M. Mäkinen, R. Kara, and P. Kuitunen. 1980. The effects of the dietary intakes of copper, iron, manganese, and zinc on the trace element content of human milk. Am. J. Clin. Nutr. 33:227-231.
Weil, W.B., Jr. 1977. Current controversies in childhood obesity. J. Pediatr. 91:175-187.
Weissbluth, M., A.T. Davis, and J. Poncher. 1984. Night waking in 4- to 8-month-old infants. J. Pediatr. 104:477-480.
West, K.P., Jr., M. Chirambo, J. Katz, A. Sommer, and the Malawi Survey Group. 1986. Breastfeeding, weaning patterns, and the risk of xerophthalmia in southern Malawi. Am. J. Clin. Nutr. 44:690-697.
Whitehead, R.G., and A.A. Paul. 1984. Growth charts and the assessment of infant feeding practices in the western world and in developing countries. Early Hum. Dev. 9:187-207.
Whitehead, R.G., M. Lawrence, and A.M. Prentice. 1986. Maternal nutrition and breastfeeding. Hum. Nutr.: Appl. Nutr. 40A (Suppl. 1):1-10.
WHO (World Health Organization). 1987. Breastfeeding/breast milk and human immunodeficiency virus (HIV). Weekly Epidemiol. Rec. 62:245-246.
Widdowson, E.M., H. Chan, G.E. Harrison, and R.D.G. Milner. 1972. Accumulation of Cu, Zn, Mn, Cr and Co in the human liver before birth. Biol. Neonate 20:360-367.
Wilson, J.T. 1981. Milk/plasma ratios and contraindicated drugs. Pp. 78-80 in Wilson, J.T., ed. Drugs in Breast Milk. ADIS Press, Balgowlah.
Wilson, D.J., D.J. Locker, C.A. Ritzen, J.T. Watson, and W. Schaffner. 1973. DDT concentrations in human milk. Am. Dis. Child. 125:814-819.
Wilson, J.T., R.D. Brown, D.R. Cherek, J.W. Dailey, B. Hilman, P.C. Jobe, B.R. Manno, J.E. Manno, H.M Redetzki, and J.J. Stewart. 1980. Drug excretion in human breast milk: principles, pharmacokinetics and projected consequences. Clin. Pharmacokinet. 5:1-68.
Wilson, J.T., R.D. Brown, J.L. Hinson, and J.W. Dailey. 1985. Pharmacokinetic pitfalls in the estimation of the breast milk/plasma ratio for drugs. Ann. Rev. Pharmacol. Toxicol. 25:667-689.
Winikoff, B. 1981. Issues in the design of breastfeeding research. Stud. Fam. Plann. 12:177-184.
Wissler, R.W., and H.C. McGill, Jr., chairmen. 1983. Conference on blood lipids in children: optimal levels for early prevention of coronary artery disease. Workshop report: experimental section. Prev. Med. 12:868-902.
Wolff, M.S. 1983. Occupationally derived chemicals in breast milk. Am. J. Ind. Med. 4:259-281.
Wong-Staal, F., and R.C. Gallo. 1985. Human T-lymphocyte retroviruses. Nature 317:395-403.
Woodruff, C.W., C. Latham, and S. McDavid. 1977. Iron nutrition in the breastfed infant. J. Pediatr. 90:36-38.
Wray, J.D. 1978. Maternal nutrition, breastfeeding and infant survival. Pp. 197-229 in W.H. Mosley, ed. Nutrition and Human Reproduction. Plenum Press, New York.
Wray, J.D. 1990. Breastfeeding: an international and historical perspective. Pp. 62-118 in Falkner, F., ed. Infant and Child Nutrition. Telford Press, Caldwell, N. J.
Wright, P., H.A. MacLeod, and M.J. Cooper. 1983. Waking at night: the effect of early feeding experience. Child Care Health Dev. 9:309-319.
Wright, S. 1985. Atopic dermatitis and essential fatty acids: a biochemical basis for atopy? Acta Dermato-Venereol. Suppl. 114:143-145.
Wright, S., and C. Bolton. 1989. Breast milk fatty acids in mothers of children with atopic eczema. Br. J. Nutr. 62:693-697.
Wyatt, R.G., and L.J. Mata. 1969. Bacteria in colostrum and milk of Guatemalan Indian women. J. Trop. Pediatr. 15:159-162.
Yolken, R., A. Ruff, P. Miotti, B. Clayman, B. Yang, N. Halsey, R. Boulos, R Viscid, and G. Dallabetta. 1990. Measurement of HIV antibodies and nucleic-acids in human milk. Pediatr. Res. (abstract) 27:279.












































