1
Introduction
In the Social Security Amendments of 1972, the U.S. Congress established entitlement to Medicare benefits for people with a diagnosis of permanent kidney failure who were fully or currently insured or eligible for benefits under Social Security, and for their spouses and dependent children. The implementation of this entitlement has come to be known as the End-Stage Renal Disease (ESRD) program of Medicare. Treatments for permanent kidney failure include both dialysis and transplantation; Medicare-covered services for all medical conditions include inpatient and outpatient care from physicians as well as treatment units.
Thirty years ago, individuals who received a diagnosis of ESRD faced near-certain death. Dialysis and kidney transplantation were just then emerging as experimental procedures and were available in only a handful of medical centers. In addition, these treatments were beyond the financial reach of most Americans. The Medicare ESRD program introduced hope where there was none, saving several hundred thousand Americans from premature death by making life-saving treatment financially possible. It has been remarkably successful in fulfilling its intended objectives. Since the program began on July 1, 1973, it has grown from approximately 10,000 to over 150,000 beneficiaries.
THE CONGRESSIONAL CHARGE
The Congress, under OBRA 1987,1 asked the National Academy of Sciences Institute of Medicine (IOM) to conduct a study of the Medicare ESRD program that would examine the following issues:
-
major epidemiologic and demographic changes in the ESRD patient
-
population that may affect access to treatment, quality of care, or the resource requirements of the program;
-
access to treatment by individuals with chronic kidney failure, whether eligible for Medicare benefits or not;
-
quality of care provided to ESRD beneficiaries, as measured by clinical indicators, functional status of patients, and patient satisfaction;
-
effect of reimbursement on quality of care; and
-
adequacy of existing data systems to monitor these matters on a continuing basis.
The study was supported by the Health Care Financing Administration (HCFA) under Cooperative Agreement No. 14-C-99338/3-02.
BASIC ASSUMPTIONS
The committee believes that people with renal failure should be the primary concern of Medicare ESRD policy. An emphasis on patients and their care underlies both the congressional charge and the committee's concern with the policy issues of access, quality, reimbursement effects on quality, data, and research needs. Consequently, the early chapters of the report (as noted above) deal with patient perspectives, ethics, and the epidemiology of the ESRD patient population.
Some recommendations of this committee would entail increased funding. The recommendations (of Chapters 7 and 8) to extend the Medicare entitlement to all Americans, to remove the three-year limit on Medicare eligibility of successful transplant recipients, and to pay for immunosuppressive drugs for the period of a transplant patient's entitlement are proposed as ways to improve an already successful program by increasing equity, promoting improved patient outcomes, and encouraging the search for means to prevent ESRD. The committee recognizes, however, that Congress must weigh these recommendations against competing uses of resources and make the appropriate allocation decisions.
The IOM committee, therefore, endorses the following objectives for the Medicare ESRD program: to guarantee access to treatment for all for whom it is medically appropriate; to provide care of high quality that achieves desirable health outcomes consistent with patient health status and current professional knowledge; to develop policies that steadily improve patient wellbeing and patient outcomes; and to manage the program prudently at the lowest cost compatible with adequate care.
CONTEXT OF THIS STUDY
This study of the ESRD program has coincided with an extraordinary amount of ESRD-related activity. OBRA 1986,2 for example, reorganized
the ESRD networks, consolidating them from 32 to 17 (later increased to 18). In addition, OBRA 1986 authorized a National End-Stage Renal Disease Registry, and the U.S. Renal Data System (USRDS) was established in partial response to this legislation. (See Chapter 13.)
OBRA 1986 also called for a study by the IOM of the effects of reductions in reimbursement for physician and facilities services on access to and quality of care. The constraints of time and the narrowness of the question led the IOM to decline to submit a proposal. Subsequently, the Urban Institute conducted that study and submitted a report in December 1987 that HCFA sent to Congress in December 1988 (Held et al., 1987). (See Chapter 10.)
In 1986, Home Intensive Care, Inc. (HIC), a dialysis supplier based in Florida, began providing paid aides for home hemodialysis patients in a way not originally anticipated by Congress. In 1978, Congress had called for a study of paid home aides, but that study had failed to provide HCFA with an adequate justification for adopting such a policy (Orkand Corporation, 1982). HIC billed its carriers for supplies at a rate that allowed it to pay for a home aide without billing directly for that aide. In 1988, HCFA sought to restrict this activity of HIC through an instruction to its financial intermediaries. Litigation before the U.S. District Court of Washington, D.C., however, resulted in a temporary injunction against HCFA's enforcement of this instruction in Florida and Illinois.3 (See Chapter 9.)
Congress in OBRA 19894 limited reimbursement for home dialysis supplies purchased by patients and paid for through Method II to a level not to exceed the facility composite rate (or Method I).5 The question of how to deal with patients being treated at home with the help of a paid aide, but too ill to travel to facilities or having severe ambulatory problems, was addressed in OBRA 1990 by the authorization of a demonstration project.6
Also during the study, the first recombinant DNA-based biological for use by dialysis patients was introduced into clinical practice. In June 1989, the Food and Drug Administration (FDA) approved recombinant human erythropoietin (Amgen Inc.'s Epogen or Epoetin-alfa) for the treatment of anemia in dialysis patients (FDA, 1989). HCFA responded immediately with an affirmative coverage decision and an interim reimbursement policy (HCFA, 1989b). That interim policy is under review by HCFA as data about dosage, response rates, and administration are considered. Reports on the subject have been issued by the Office of Technology Assessment (OTA, 1990) and by the U.S. Department of Health and Human Services (DHHS) (OIG, 1990a). Congress, in OBRA 1990, authorized self-administration of erythropoietin for home patients and also modified reimbursement policy.7 (See Chapter 9.)
Congress addressed kidney transplantation as well during the study. The National Organ Transplant Act of 1984, which amended the Public Health Service Act and expanded the federal government's interest in whole-organ transplantation beyond kidneys, authorized the creation of the Organ Procurement and Transplantation Network.8 That law was amended in 1986
and 1988 and was reauthorized in 1990 as this study was nearing completion. In addition, a draft report on access to kidneys for transplantation was issued by the DHHS in August 1990 (OIG, 1990b) as this IOM report was being written. (See Chapter 8.)
Change is occurring across many dimensions, then, within the ESRD program. The patient population continues to grow and to change in composition. Treatment technology in both dialysis and transplantation changes incrementally, punctuated on occasion by major new innovations. Total program expenditures continue to grow, primarily because of growth in the patient population.
STUDY METHODS
The composition of the IOM's End-Stage Renal Disease Study Committee reflected a balance among experts in the treatment of ESRD, including a patient, nephrologists (including a pediatrician) and transplant surgeons, nurses, and a social worker; also included were a sociologist and an economist, both familiar with the ESRD program. In addition, a breadth of perspective was achieved by individuals from internal medicine, epidemiology, quality assessment, ethics, and economics who were not immediately involved with dialysis or transplantation.
The IOM committee held seven 2-day meetings and one 3-day retreat. For the most part, these meetings were open to the public, were attended by a number of observers, and involved a combination of presentations and discussions.
The committee commissioned papers on the following subjects: pediatric renal failure patients; elderly renal failure patients; diabetic renal failure patients; hypertensive renal failure patients; black and other nonwhite renal failure patients; and the effect of reimbursement on innovation. The IOM also contracted for the following activities: three patient focus groups held in Washington, D.C., Los Angeles, and St. Louis; a survey of the state certificate-of-need (CON) programs that affect the ESRD program; a survey of state expenditures for ESRD purposes by state Medicaid programs, state medical assistance programs, and state kidney disease programs; and three analyses of the effects of the 1983 composite rate on mortality, hospitalization, and dialysis unit staffing. A list of authors and titles of these commissioned papers and contractor reports appears in Appendix C.9
In addition, a methodological paper on mortality in the ESRD population was prepared by Robert A. Wolfe, University of Michigan, and appears as Appendix D to this report. Finally, a paper on ethics (Cassel and Moss, 1990) was prepared by Christine K. Cassel, a committee member, and Alvin H. Moss, a nephrologist, and used extensively in preparing Chapter 3.
The committee held two public hearings, the first in Chicago in May 1989.
Respondents were invited to comment on the five issues of the congressional charge to the IOM study, and written testimony was a prerequisite for oral testimony. Participants and their affiliations are listed in Appendix E.
The second public hearing, in Washington, D.C., in February 1990, focused on dialysis reimbursement rate-setting. Project staff prepared a draft document that was circulated for comment (Young and Rettig, 1989). Interested parties were invited to comment on this draft, which became the basis for written and oral testimony. The committee received detailed responses to this document. Participants and their affiliations are listed in Appendix F.
HCFA supported this study by a cooperative agreement, and cooperation characterized the working relations with HCFA staff throughout the study. Encouragement and support were provided by the Office of Research and Demonstrations through Carl Josephson, project officer, and Paul W. Eggers. The Bureau of Policy Development supplied audited and unaudited cost data for 1985 and unaudited cost data for 1987 and responded to a number of queries from project staff. In June 1990, the Health Standards and Quality Bureau staff briefed the IOM staff on their quality assurance initiative. The Bureau of Data Management and Strategy made data and analyses available to the study from the Program Management Medical Information System (PMMIS). The IOM project staff also established direct computer access to PMMIS that enabled epidemiologic analyses to be done that would not otherwise have been possible.
Throughout the study, the project staff and committee members interacted with HCFA, congressional staff, the renal provider community, and patients, including attendance at and participation in professional meetings. The committee benefited greatly from this interaction.
THE ESRD PATIENT POPULATION
The ESRD patient population has changed strikingly in numbers, characteristics, and treatment modalities over the history of the program. The increase in new patients (incidence) and the total patient population (prevalence) between 1974 and 1989 are shown in Table 1-1. These incidence and prevalence figures are for treated ESRD patients only and are thus somewhat lower than the true incidence and prevalence of all ESRD patients.
The proportion of new ESRD patients who are elderly grew from 24 percent in 1978 to 38 percent in 1988, while diabetic patients increased from 21 to 35 percent of new ESRD patients, as shown in Table 1-2.
Projections of Medicare ESRD patients through the year 2000 are shown in Table 1-3. These projections, developed by Eggers for this study, are described in Chapter 4. They estimate that nearly one-quarter of a million ESRD patients will be enrolled in Medicare at the end of the decade.
TABLE 1-1 Incidence and Prevalence of Patients in Medicare End-Stage Renal Disease (ESRD) Program, 1974–89
|
Year |
Incidencea |
Prevalenceb |
|
1974 |
NA |
15,993 |
|
1975 |
NA |
22,674 |
|
1976 |
NA |
28,941 |
|
1977 |
NA |
34,778 |
|
1978 |
15,174 |
44,153 |
|
1979 |
16,937 |
52,184 |
|
1980 |
18,437 |
60,053 |
|
1981 |
19,356 |
67,493 |
|
1982 |
21,927 |
76,316 |
|
1983 |
25,155 |
86,354 |
|
1984 |
26,552 |
95,522 |
|
1985 |
29,419 |
104,879 |
|
1986 |
31,561 |
114,659 |
|
1987 |
34,273 |
125,011 |
|
1988 |
36,743 |
134,786 |
|
1989 |
40,497 |
146,657 |
|
NOTE: NA = Not available. a New ESRD patients during the reference year. b Figures are for patients currently enrolled in the Medicare ESRD program as of July 1 for 1974–77 and as of December 31 for 1978–89. SOURCES: Incidence: HCFA, 1990b; prevalence: 1974–79, Eggers et al., 1984; 1980–99, HCFA, 1990c. |
||
NATIONAL EXPENDITURES FOR ESRD
Total Medicare expenditures10 for all ESRD beneficiaries were $229 million in 1974, the first full year of the program. Since then, the program has grown significantly, both in enrollments and in expenditures. By 1988, total Medicare ESRD expenditures reached $3.0 billion, a 12-fold increase from 1974 (Table 1-4). Measured in 1974 constant dollars, however, this increase is less than fivefold, from $229 million to $1.3 billion (Figure 1-1).
The primary contributor to the growth in Medicare ESRD payments has been the 10-fold increase in beneficiary enrollments from 1974 to 1989 (Table 1-1). This faster growth of patients compared to constant-dollar expenditures suggests that treatment has become more cost-effective over time (Table 1-5).
Medicare expenditures for ESRD beneficiaries, moreover, are for all medical services, not just those related to renal disease. About 36 percent of these
expenditures in 1988 went for inpatient hospital stays. Outpatient services and physician-supplier services accounted for another 40 percent and 23 percent, respectively (Table 1-6). In this context, services covered for ESRD treatment include outpatient hemodialysis and peritoneal dialysis (in-center or at home), inpatient dialysis (hemodialysis and peritoneal dialysis), kidney donor acquisition (cadaver and living donor), transplant procedures, and some medications. Medicare ESRD expenditures as a percentage of total Medicare expenditures grew to 3.4 percent in 1980, declined to 3.0 percent in 1985, and climbed again to 3.4 percent in 1988 (Table 1-7).
Medicare, however, does not cover all costs for either dialysis or transplant patients: premiums, deductibles, and copayments must be met from other sources such as state Medicaid programs, Department of Veterans Affairs (Table 1-8), private insurance companies, and patients. The U.S. Renal Data System 1990 Annual Data Report (USRDS, 1990) has estimated that the total direct medical payment from all sources for Medicare and non-Medicare ESRD patients in 1988 was $5.4 billion.
Based on 1987 cost data provided by HCFA, the annualized Medicare expenditure for a dialysis patient (for renal and nonrenal care) was $32,000. Transplant patients have a first-year per-capita expenditure of $56,000, but they cost Medicare approximately $6,000 on average in succeeding years for successful transplants (P.W. Eggers, unpublished data, 1990).
TABLE 1-2 New Elderly and Diabetic End-Stage Renal Disease (ESRD) Patients as a Percentage of New Medicare ESRD Patients
|
Year |
Over Age 65 |
Diabeticsa |
|
1978 |
24.1 |
20.6 |
|
1979 |
26.7 |
21.3 |
|
1980 |
27.8 |
24.7 |
|
1981 |
27.0 |
26.1 |
|
1982 |
29.5 |
26.8 |
|
1983 |
35.3b |
28.4 |
|
1984 |
34.6 |
30.7 |
|
1985 |
36.1 |
31.4 |
|
1986 |
37.5 |
33.5 |
|
1987 |
38.8 |
34.0 |
|
1988 |
38.4 |
34.9 |
|
a Calculated as a percentage of all patients for whom a diagnosis of diabetes was reported. b The large jump between 1982 and 1983 in the percentage over age 65 may be due to irregularities in data reporting in those years. SOURCE: HCFA, 1990a. |
||
TABLE 1-3 Projections of Medicare End-Stage Renal Disease (ESRD) Patients to the Year 2000
|
Year |
Incidence |
Prevalence |
|
1989 |
38,111 |
141,236 |
|
1990 |
40,375 |
149,868 |
|
1991 |
42,601 |
159,047 |
|
1992 |
44,766 |
168,583 |
|
1993 |
46,844 |
178,314 |
|
1994 |
48,808 |
188,099 |
|
1995 |
50,634 |
197,812 |
|
1996 |
52,298 |
207,338 |
|
1997 |
53,774 |
216,563 |
|
1998 |
55,042 |
225,388 |
|
1999 |
56,082 |
233,714 |
|
2000 |
56,877 |
241,452 |
|
SOURCE: P.W. Eggers, Health Care Financing Administration, unpublished data, 1990. |
||
TABLE 1-4 Cumulative Percentage Change in Medicare End-Stage Renal Disease (ESRD) Benefit Payments, 1974–88 Nominal and Real-Dollar Payments
|
Year |
Annual ESRD Payments (millions of dollars)a |
Cumulative Change (%) |
GNP Deflator Adj. Payments (1974 dollars) |
Cumulative Change (%) |
|
1974 |
229 |
— |
229 |
— |
|
1975 |
361 |
58 |
329 |
44 |
|
1976 |
512 |
124 |
438 |
91 |
|
1977 |
641 |
180 |
514 |
124 |
|
1978 |
800 |
249 |
598 |
161 |
|
1979 |
1,011 |
341 |
694 |
203 |
|
1980 |
1,252 |
447 |
789 |
245 |
|
1981 |
1,477 |
545 |
848 |
270 |
|
1982 |
1,662 |
626 |
897 |
292 |
|
1983 |
1,898 |
729 |
986 |
331 |
|
1984 |
2,003 |
775 |
1,003 |
338 |
|
1985 |
2,128 |
829 |
1,035 |
352 |
|
1986b |
2,423 |
958 |
1,148 |
401 |
|
1987b |
2,702 |
1,080 |
1,241 |
442 |
|
1988b |
3,011 |
1,215 |
1,343 |
466 |
|
a ESRD figures may underestimate the actual benefit payments. In 1990, HCFA introduced a new method of calculating expenditures. See note 10. b Data are incomplete because of outstanding bills. SOURCE: HCFA, 1990c. |
||||
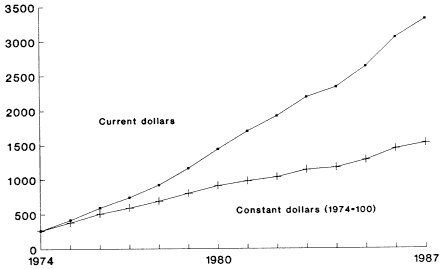
FIGURE 1-1
Medicare ESRD Benefit Payments
NOTE: Adjusted by GNP deflator. SOURCE: HCFA, 1990.
TABLE 1-5 Growth of End-Stage Renal Disease (ESRD) Program: Patient Growth Versus Real-Dollar Benefit Payment Growth
|
Year |
ESRD Annual Patient Growth (%) |
ESRD Annual Real-Dollar Expenditure Growth (%)a |
|
1974 |
— |
— |
|
1975 |
41.9 |
43.7 |
|
1976 |
27.3 |
33.1 |
|
1977 |
20.4 |
17.4 |
|
1978 |
27.0 |
16.3 |
|
1979 |
18.1 |
16.1 |
|
1980 |
15.1 |
13.7 |
|
1981 |
9.1 |
7.5 |
|
1982 |
4.5 |
5.8 |
|
1983 |
17.5 |
9.9 |
|
1984 |
9.3 |
1.7 |
|
1985 |
6.4 |
3.2 |
|
1986 |
11.6 |
10.9 |
|
1987 |
9.1 |
8.1 |
|
1988 |
9.9 |
8.2 |
|
a Adjusted by GNP deflator. SOURCES: Eggers et al., 1984; HCFA, 1990c. |
||
TABLE 1-6 End-Stage Renal Disease (ESRD) Benefit Payments by Type of Service, 1988
TABLE 1-7 Medicare End-Stage Renal Disease (ESRD) Benefit Payments, 1974–88
|
Year |
Annual ESRD Payments (millions of dollars) |
Total Medicare Payments (millions of dollars) |
ESRD as Percentage of Total Medicare |
|
1974 |
229 |
12,881 |
1.8 |
|
1975 |
361 |
15,742 |
2.3 |
|
1976 |
512 |
18,621 |
2.7 |
|
1977 |
641 |
22,175 |
2.9 |
|
1978 |
800 |
25,756 |
3.1 |
|
1979 |
1,011 |
30,221 |
3.3 |
|
1980 |
1,253 |
36,484 |
3.4 |
|
1981 |
1,477 |
43,541 |
3.4 |
|
1982 |
1,662 |
51,301 |
3.2 |
|
1983 |
1,898 |
58,579 |
3.2 |
|
1984 |
2,003 |
64,329 |
3.1 |
|
1985 |
2,128 |
69,924 |
3.0 |
|
1986a |
2,423 |
75,389 |
3.2 |
|
1987a |
2,702 |
81,511 |
3.3 |
|
1988a |
3,011 |
89,300 |
3.4 |
|
NOTE: Data may underestimate actual ESRD benefit payments. See note 10. a Data are incomplete because of outstanding bills. SOURCE: HCFA, 1990c. |
|||
TABLE 1-8 End-Stage Renal Disease (ESRD) Expenditures in Department of Veterans Affairs, 1984–89 (millions of dollars)
|
Year |
Dialysis Expendituresa |
Transplant Expendituresa |
Physician Expendituresb |
Total |
|
1980 |
— |
— |
— |
104.9 |
|
1981 |
— |
— |
— |
107.9 |
|
1982 |
— |
— |
— |
114.3 |
|
1983 |
— |
— |
— |
120.4 |
|
1984 |
124.1 |
8.5 |
— |
132.6 |
|
1985 |
141.8 |
6.6 |
— |
148.4 |
|
1986 |
144.4 |
6.4 |
— |
150.8 |
|
1987 |
137.7 |
5.8 |
— |
143.5 |
|
1988 |
123.4 |
5.7 |
— |
129.1 |
|
1989 |
115.7 |
6.0 |
9.0 |
121.7 |
|
a Before FY 1984, dialysis and transplant cost accounts were a subset of the regular cost distribution system and did not include all applicable indirect costs. b FY 1989 was the first year in which physician costs were reported as a separate component of the ESRD expenditures. SOURCE: C.R. Wichlacz, Department of Veterans Affairs, personal communication, July 3, 1990. |
||||
ORGANIZATION OF THE REPORT
Part I is an overview of the study and the ESRD program. It contains the Summary and this introductory chapter.
Part II deals with patients and providers. Chapter 2 presents the results of three patient focus groups convened to provide direct access to patient concerns. Chapter 3 addresses ethical issues related to the initiation and termination of treatment and to the problem patient. Chapter 4 includes general background information about the ESRD patient population, a projection of that population to the year 2000, and a discussion of ESRD patient mortality. Chapter 5 discusses special ESRD patient groups: pediatric, elderly, diabetic, hypertensive, and black and nonwhite renal failure patients. Chapter 6 describes the nature and structure of dialysis and transplantation providers (facilities and centers).
Part III deals with access issues. Although eligibility for Medicare coverage for ESRD treatment is estimated to cover over 90 percent of the American people, the entitlement is not universal.11Chapter 7 addresses the access problems of individuals with no Medicare eligibility for ESRD treatment,12 as well as the access problems of Medicare-eligible ESRD patients. Chapter 8 deals with kidney transplantation, focusing on the limits of
access to this treatment that stem from policy limits on eligibility and coverage of immunosuppressive drugs and from the shortage of donor organs.
Part IV addresses the relationship between reimbursement and quality. Chapter 9 discusses current reimbursement policy for facilities as well as physicians. This partial digression from the congressional charge was deemed necessary in order to deal with the issue of reimbursement effects on quality. Chapter 10 analyzes these effects with respect to mortality and hospitalization (two outcome measures), treatment unit staffing (a structural measure with substantial implications for the process of care), and innovation in treatment technology. Chapter 11 deals with reimbursement policy issues of covered services, rate-setting, and payment structure of the composite rate. Chapter 12 examines the conceptual elements of quality assessment and assurance (QA) as applied in the ESRD area, analyzes how the federal government manages the ESRD QA function, and argues for a treatment-unit QA approach.
Part V on data and research concludes the report. Chapter 13 describes the HCFA, the USRDS, and the United Network for Organ Sharing (UNOS) data systems, analyzes the implementation of the OBRA 1986 provision for the National ESRD Registry, and examines the adequacy of specific features of these data systems. Chapter 14 considers research needed to support the ESRD program, ranging from laboratory studies through clinical studies, epidemiology, and health services research.
NOTES
|
1. |
Omnibus Budget Reconciliation Act of 1987, Pub. L. No. 100–203, § 4036(1), (1987). |
|
2. |
Omnibus Budget Reconciliation Act of 1986, Pub. L. No. 99–509, § 9335, (1986). |
|
3. |
National Kidney Patients Association v. Otis R. Bowen, Civ. No. 88–3251 (D.D.C. Dec. 22, 1988). |
|
4. |
Omnibus Budget Reconciliation Act of 1989, Pub. L. No. 101–239, § 6203. |
|
5. |
Method I involves payment under the composite rate for home dialysis at the same rate as outpatient center hemodialysis (see Chapter 9). Method II involves direct billing of a fiscal carrier by the patient. |
|
6. |
Omnibus Budget Reconciliation Act of 1990, Pub. L. No. 101–508, § 4202. |
|
7. |
OBRA 1990, Pub. L. No. 101–508, § 4201(c) and (d). |
|
8. |
Pub. L. No. 98–507. |
|
9. |
Although some IOM studies publish commissioned papers, the ESRD Study Committee has treated these as inputs to its deliberations and has encouraged the authors to publish their work independently. |
|
10. |
The ESRD Quarterly Statistical Summary, prepared by the Bureau of Data Management and Strategy, HCFA, publishes the only historical ESRD expenditure data series. Expenditure data are also presented in some of the annual HCFA research reports on ESRD (HCFA 1986, 1987, 1990d). The U.S. Renal Data System publishes summary cost data only. HCFA (1990d) recently presented expenditure data for 1984–87 that are substantially higher than previously published data. These new expenditure data are shown below in relation to the data presented in Table 1-4 (which are based on the HCFA ESRD Quarterly Statistical Summary): |
|
11. |
Eligibility depends on a diagnosis of permanent kidney failure, on ''fully or currently insured'' status regarding Social Security or entitled to or receiving Social Security benefits, and on application for benefits. |
|
12. |
At any time, ESRD patients will be classified as Medicare eligible, eligibility pending, and not eligible. |
REFERENCES
Cassel CK, Moss AH. 1990. An ethical analysis of the End-Stage Renal Disease program with recommendations for physician-patient decision making and appropriate use of dialysis. Paper prepared for the Institute of Medicine ESRD study.
Eggers PW, Connerton R, McMullan M. 1984. The Medicare experience with end-stage renal disease: Trends in incidence, prevalence, and survival. Health Care Financing Rev 5:69–88.
FDA (Food and Drug Administration). 1989. Summary Basis of Approval: Drug License name: Epoetin alfa; Brand name: EPOGEN. June 1.
HCFA (Health Care Financing Administration). 1986. Health Care Financing Research Report: End Stage Renal Disease, 1984. Baltimore, Md.
HCFA. 1987. Health Care Financing Research Report: End Stage Renal Disease, 1985. Baltimore, Md.
HCFA. 1988. Health Care Financing Research Report: End Stage Renal Disease, 1986. Baltimore, Md.
HCFA. 1989a. Health Care Financing Research Report: End Stage Renal Disease, 1987. Baltimore, Md.
HCFA. 1989b. Medicare Provider Reimbursement Manual, Part 1-Chapter 27: Reimbursement for ESRD and transplant services. Medicare Provider Reimbursement Manual: Transmittal No. 11. Baltimore, Md. July.
HCFA. 1990a. ESRD Program Management and Medical Information System, March Update. Baltimore, Md.
HCFA. 1990b. ESRD Program Management and Medical Information System, August Update. Baltimore, Md.
HCFA. 1990c. End Stage Renal Disease Program Quarterly Statistical Summary. September. Baltimore, Md.
HCFA. 1990d. Health Care Financing Research Report: End Stage Renal Disease, 1988. Baltimore, Md.
Held PJ, Bovbjerg RR, Pauly MV, Garcia JR, Newmann JM. 1987. Effects of the 1983 "Composite Rate" changes on ESRD patients, providers, and spending. Washington, D.C.: The Urban Institute. December 21.
OIG (Office of Inspector General, Department of Health and Human Services). 1990a. The Effect of the interim payment rate for the drug Epogen on Medicare expenditures and dialysis facility operations. Draft Report. Washington, D.C.
OIG. 1990b. The Distribution of Organs for Transplantation: Expectations and Practices. Washington, D.C. Draft Report. August.
Orkand Corporation. 1982. Evaluation of the home dialysis aide demonstration: Executive Summary. Silver Spring, Md.
OTA (Office of Technology Assessment, U.S. Congress). 1990. Recombinant erythropoietin: Payment options for Medicare. OTA-H-451. Washington, D.C.
USRDS (U.S. Renal Data System). 1990. Annual Data Report. The National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, Md.
Young GP, Rettig RA. 1989. Issues in dialysis reimbursement rate-setting. Washington, D.C.: Institute of Medicine.














