CHALLENGE SUMMARY
This challenge has several related parts: (1) Maintaining a reliable supply of Technetium-99m (Tc-99m) for single-photon imaging; (2) Availability and further development of PET radiopharmaceuticals; and (3) Expanding the supply of PET radionuclides beyond C-11, N-13, O-15, and F-18.
Technetium-99m is the workhorse of single-photon imaging. Its precursor is reactor-produced molybdenum-99 (Mo-99). Production of Mo-99 has a history of several problems: (1) Most has been produced by reactor irradiation of HEU, which has required the sole export of such materials from the United States. Alternative reactor production is available using LEU and there are now efforts to make this the only method of reactor production. (2) There have been a number of interruptions in the supply of Mo-99 as aging reactors have been removed from service. (3) U.S. supplies of Mo-99 have had to rely on foreign sources (Canada, The Netherlands, South Africa, etc.) and there has been considerable effort to develop a sustainable supply in this country (see American Medical Isotope Production Act of 2012). Also, there has been recent interest in alternative methods for the production of Tc-99m using accelerators, but issues of expense, specific activity, and distribution need to be addressed.
(Another radionuclide with desirable properties for single-photon imaging is I-123. This iodine isotope brings with it the advantages of halide chemistry and avoids the bulky coordination cage properties of technetium. In the past, its accelerator production had its own problems including cost, purity, and availability, but these seem to have been mostly solved. It has
never enjoyed the anticipated uses predicted for it outside of iodide for thyroid imaging. Newly developed radiopharmaceuticals such as ioflupane [DaTscan™] might expand its utility.)
Radiopharmaceuticals labeled with positron-emitting radionuclides are now used to map a number of cellular functions including glucose metabolism, oxygen utilization, cell proliferation, amino acid uptake, and neurotransmitter status, to name a few. Produced, on-site, by cyclotrons capable of making Carbon-11, Nitrogen-13, Oxygen-15 and Fluorine-18 the most available compounds are primarily labeled with Fluorine-18 (F-18). This is partly due to its longer half-life (110 minutes) than the others and partly because of relatively facile fluorine chemistry. Carbon-11 labeled agents, despite the centrality of carbon compounds as biological substances, have been less used, in part because the short half-life (20 minutes) requires very rapid syntheses to produce pure products with high specific activity.
At present, there is a robust commercial supply of F-18 labeled FDG and NaF. Commercial supplies of other F-18 labeled compounds are severely limited. As a greater repertoire of tracers is developed for more specific diagnoses and monitoring response to therapy, the management of patients outside of academic medical centers is certain to be compromised unless an economic pathway for these can be developed.
In addition to the light elements (C, N, O, F), longer-lived positron-emitting radionuclides are likely to be desired for medical application. Some already showing promise are Cu-64, Zr-89, and I-124. These radionuclides are produced by cyclotrons or linear accelerators. Except for I-124, which can be used as iodide for thyroid studies, all need to be incorporated into complex organic compounds for imaging purposes.
For all medical imaging procedures there is a need to minimize the radiation dose received by patients without sacrificing diagnostic accuracy. The realization that medical imaging from CT and nuclear studies now represents the major source of public radiation exposure has mobilized the profession of Radiology into campaigns for minimizing exposure (viz. Imaging Gently and Imaging Wisely).
In addition to imaging, certain radionuclides are used for therapy. These include I-131 for thyroid disease, Sm-153 and Sr-89 for bone pain, Y-90 for liver metastases, In-111 and Lu-177 for neuroendocrine tumors. Newer ones, such as the alpha particle emitter Ra-223, have been used in research. Some are produced in reactors others in accelerators. For their full potential to be realized, continued availability of the radionuclides will need to be assured and specific delivery systems will need to be devised.
Key Questions
• Is the American Medical Isotope Production Act of 2012 sufficient to secure the continued need for Tc-99m? Should alternative methods of production continue to be pursued? Given the superior resolution of F-18 labeled agents, what advances in SPECT or CT technology will be required to justify the continued use of Tc-99m?
• As newer more specific PET agents are created, how will the manufacture and distribution of these be accomplished for use by other than major academic medical centers? The time for new agents to go from bench-to-bedside/clinic is quite a bit shorter in Germany and Japan than in the United States. What are the impediments to such transfer in this country, regulatory and otherwise, and how might they be eliminated?
• Is the use of radiopharmaceuticals labeled with longer-lived positron-emitting radionuclides likely to be employed other than in research? If so, how are they to be produced and distributed?
• As optimization of radiation dose in medicine becomes dictum, efforts will be made to use diagnostic nuclear medical studies appropriate to the clinical questions asked. In addition, technologies are, and will be, developed to reduce amounts of administered radioactivity without loss of diagnostic accuracy. What will these be, how much will they add to cost, and what are their limits?
• For a number of reasons, the use of radionuclides in therapy has been relatively restricted. What factors have limited their use (even when their efficacy has been demonstrated) and what might be done to overcome these hurdles?
Suggested Reading
Atcher RW. Medical radioisotopes—what steps can we take to ensure a secure supply? Journal of Nuclear Medicine 2009; 50(4):17-18N.
Congress passes American Medical Isotope Production Act. Journal of Nuclear Medicine 2013; 54(2):11N.
Fowler JS, Wolf AP. The synthesis of carbon-11, fluorine-18 and nitrogen 13 labeled radiotracers for biomedical applications. Nuclear Science series: NAS-NS 3201. National Technical Information Service: Springfield, VA, 1982.
Goske MJ, Applegate KE, Bulas D, et al. Image Gently 5 years later: what goals remain to be accomplished in radiation protection for children? American Journal of Roentgenology 2012; 199(3):477-479.
Hricak H, Brenner DJ, Adelstein SJ et al. Managing radiation use in medical imaging: a multifaceted challenge. Radiology 2011; 258(3):889-905.
Institute of Medicine. Isotopes for medicine and the life sciences. Report of Committee on Life Sciences, Division of Health Science Policy; National Academy Press: Washington, DC, 1995.
James ML, Gambhir SS. A molecular imaging primer: modalities, imaging agents, and applications. Physiological Reviews 2012; 92(2):897-965.
Kircher MF, Hricak H, Larson SM. Molecular imaging for personalized cancer care. Medical Oncology 2012; 6(2):182-195.
Mettler FA Jr, Bhargavan M, Faulkner K et al. Radiologic and nuclear medicine studies in the United States and worldwide: frequency, radiation dose, and comparison with other radiation sources—1950-2007. Radiology 2009; 253(2):520-531.
National Research Council. Medical isotope production without HEU. The National Academies Press: Washington, DC, 2008.
Peplow M. Technetium: nuclear medicine’s crisis. Proto Summer 2013.
Sheehy N, Tetrault T, Zurakowski D, et al. Pediatric 99mTc-DMSA SPECT using iterative reconstruction with isotropic resolution recovery: improved image quality and reduction in radiopharmaceutical administered activity. Radiology 2009; 251:511-516.
SNM Position Statement on dose optimization for nuclear medicine and molecular imaging procedures. Society for Nuclear Medicine: June 2012.
Tu Z, Mach RH. C-11 radiochemistry in cancer imaging applications. Current Topics in Medicinal Chemistry 2010; 10(11):1060-1095.
Because of the popularity of this topic, three groups explored this subject. Please be sure to review the other write-ups, which immediately follow this one.
• Narasimhan Danthi, National Institutes of Health
• Patrick Hahn, Johns Hopkins University
• Efstathios Karathanasis, Case Western Reserve University
• Jason S. Lewis, Memorial Sloan-Kettering Cancer Center
• Michael A. McDonald, Johns Hopkins University
• Todd E. Peterson, Vanderbilt University
• Jonathan K. Pokorski, Case Western Reserve University
• Satish Viswanath, Case Western Reserve University
IDR TEAM SUMMARY—GROUP 1A
Patrick Hahn, NAKFI Science Writing Scholar, Johns Hopkins University
“The poison is in the dose.”—Philippus Aureolus Theophrastus Bombastus von Hohenheim, the father of toxicology.
IDR Team 1A was asked to identify improvements in technology and other approaches, such as educational initiatives and manufacturing and distribution plans that will ensure the future development and supply of radionuclides and radiopharmaceuticals for diagnostic imaging and therapy in the United States.
The team focused on four areas: the supply of stable isotopes, the availability of radioisotopes and tracers, translation of research from bench to clinic, and education.
Supply of Stable Isotopes
Stable isotopes are the starting materials needed to create the radioisotopes used for diagnostic nuclear imaging (SPECT and PET) and targeted radiotherapy, which are common in contemporary medicine. The major advantages of using enriched stable isotopes for radioisotope production are that purer, higher specific activity radioisotopes can be produced. The United States has relied on the isolation of stable isotopes using calutrons, but these currently are not being operated and have been in a standby mode since 2008. As a consequence, there is no domestic supply of these stable isotopes. A supply from international sources cannot be counted on indefinitely.
The team agreed that innovative technologies, including miniaturization of the cumbersome calutron technologies currently in use, are imperative to make the domestic manufacture of stable isotopes cost-effective once more. Public–private partnerships could help to make the adoption of these new technologies economically feasible, with public funding defraying startup costs, after which the day-to-day operations could be spun off to the private sector.
Availability of Radioisotopes and Tracers
The supply of radioisotopes and tracers available to clinicians is limited and inconsistent. The current centralized system is highly vulnerable to the failure of one of its components. Also, multiple tracers for the same target often make it to first-in-human use, with no clear first choice among them.
The vast majority of commercial cyclotrons are used to manufacture FDG, a radiolabeled glucose analog. This tracer is indeed a powerful tool in nuclear medicine, but focusing on this one tracer limits the availability of new tracers for clinical trials, making the approval of potentially beneficial new tracers a problem.
The United States needs a decentralized and coordinated infrastructure of cyclotrons to manufacture a variety of radiotracers for clinical use. The country needs a competitive but unbiased method to pick a winner among different radiotracers for the same target, making the commercial development and widespread distribution of that tracer economically feasible. The ultimate goal is the development of innovative single-stream technologies combining accelerators/reactors, separation, and radiopharmaceutical preparation for point-of-care application.
Translation to Clinic
The process of translation from lab bench to clinic needs to be streamlined. Before a New Drug Application can be approved, the applicants must submit data from all the relevant pathology and toxicology studies to the Food and Drug Administration (FDA). This information is called the path-tox package, and the burden of generating path-tox packages represents one potential barrier to translation. Another is poorly designed human trials without clear medical relevance or a well-defined endpoint. Finally, radiotracers may be approved by the FDA but not reimbursed by Medicare or Medicaid.
Radiotracers typically are administered in picomolar quantities, yet are held to the same safety standards as therapeutic agents administered in quantities many orders of magnitude greater. The process of generating toxicology studies could be streamlined by the increased use of existing data, and by the increased use of in vitro studies and/or computer modeling to reduce or eliminate the need for time-consuming and expensive animal studies. A nationwide database should be established to enable investigators to share data from path-tox studies, with the short-term goal of eliminating
redundant trials and the long-term goal of eliminating the requirement for such studies for radiotracers altogether.
The cost of human trials could be reduced through smart trial design, with a well-defined clinical endpoint specified from the beginning. As therapies become increasingly specific for specific types of cancers, clinicians need to employ image-guided biopsies from the earliest stages of drug development in order to ascertain whether a given patient is expected to respond to a given drug before recruiting that patient to a trial. This in turn will make it increasingly difficult to recruit sufficient numbers of patients for a trial. In the future, multicenter trials will become the norm, in turn necessitating the development of new software to collate data from imaging studies, genomics, and proteomics from multiple centers, to help clinicians identify appropriate patients for different kinds of therapy.
A promising area of research is the new field of theranostics, or the combination of diagnostic and therapeutic entities into one drug delivery vehicle. All this should ultimately lead to greater quantitative precision in medicine as opposed to the current system of dosing according to body weight.
Finally, government agencies such as the FDA, National Institutes of Health, Nuclear Regulatory Commission, and the Center for Medicare and Medicaid Services need to work together at all stages of development of new radiotracers to facilitate reimbursement for approved radiotracers.
Education of New Workers
The average worker in the nuclear medicine field is 55-58 years old and the majority of the workforce is expected to retire within 10 years. The country is on the verge of losing an incredible resource, and little is being done to train new scientists specializing in this field. Few universities have programs in radiochemistry and nuclear medicine, and most have only a few students, scattered among different departments. Communication among different subspecialties relating to nuclear medicine is often poor.
The country needs programs to ignite young people’s interest in nuclear science, such as the Department of Energy’s recently closed Nuclear Summer School. Universities, professional societies, and government agencies need to work together to develop a curriculum enabling practitioners of all subspecialties relating to nuclear medicine to work together effectively. Studies are needed on the economics of radiopharmaceuticals. Finally, ef-
fective education/lobbying is needed to educate lawmakers of the benefits of nuclear medicine.
Conclusion
By means of all stakeholders working together, the full promise of nuclear medicine can be realized, resulting in improved patient outcomes at reduced costs.
• Sujata K. Bhatia, Harvard University
• Georges El Fakhri, Massachusetts General Hospital; Harvard Medical School
• Jacob M. Hooker, Massachusetts General Hospital; Harvard Medical School
• Julie L. Sutcliffe, University of California, Davis
• Yuan-Chuan Tai, Washington University in St. Louis
• Izabela Tworowska, RadioMedix Inc.; RITA Foundation Houston
• Frank J. Wessel, University of California, Irvine
• Julianne Wyrick, University of Georgia
IDR TEAM SUMMARY—GROUP 1B
Julianne Wyrick, NAKFI Science Writing Scholar University of Georgia
IDR Team 1B was asked to identify improvements in technology and other approaches that will ensure the future development and supply of radionuclides and radiopharmaceuticals for diagnostic imaging and therapy. The challenge included several specific questions, such as how to manufacture and distribute new radionuclides for PET imaging so that they are available for widespread clinical use. Team 1B agreed that while these questions are very important, another fundamental issue must be tackled first—the need to develop radioactive imaging assays that are likely to be useful to the academic, clinical, and industrial communities.
The team identified two different approaches that could be used by imaging scientists to generate imaging assays with promise of value. The two
approaches are the “bottom up” approach and the “top down” approach, as shown in Figure 1. The bottom-up approach refers to searching for new imaging agents by screening many potentially useful radionuclides. The top-down approach refers to first identifying clinical needs and searching for imaging agents to meet these needs. The team members emphasized that both approaches should be used.
They also noted that integrated training between academia and industry as well as between academia and the clinic must be used along with both approaches. For example, graduate students in the imaging sciences could benefit from spending a year in industry or a nonacademic clinical setting. Better partnerships among these three entities—academic institutions, clinics, and companies—are also needed to ensure the development of useful radionuclide imaging assays. The IDR Team noted that these partnerships should involve the commitment of money by all parties. However, the team chose to focus its discussion on the top-down and bottom-up methods of
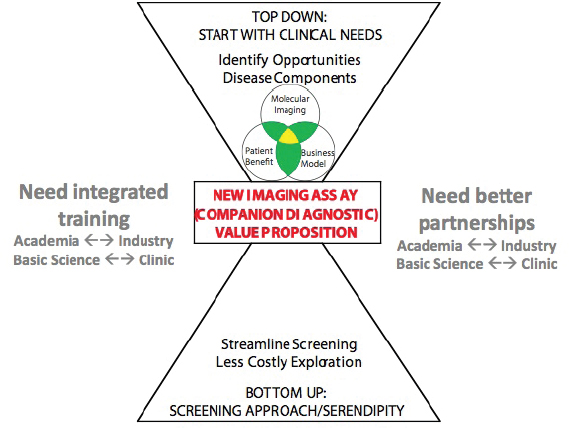
FIGURE 1 The two approaches that IDR Team 1B identified for developing imaging assays that would have promise of clinical, academic, and industrial value are shown. IDR Team’s strategies for improving these approaches are also shown.
discovery because another IDR Team was concentrating on the issues of training and partnerships.
Bottom-Up Approach
The first approach the IDR Team identified for generating imaging assays was the bottom-up approach, which is the approach traditionally used in scientific exploration. This approach could also be referred to as a screening approach. Specifically, it involves investigating many potential imaging agents and disease targets with the goal of identifying an agent-target combination that will be a high-value imaging assay for humans. The major challenge is the high cost of screening. The more radionuclides a researcher tests, the higher the cost. IDR Team 1B identified resources that could be shared to reduce costs, including labs, molecular libraries, synthesis methods, imaging protocols, personnel, and toxicology and pharmacokinetics data.
The IDR Team chose to focus in particular on developing a way to share toxicology and pharmacokinetics data. Currently, researchers must do toxicology and pharmacokinetics studies on potential radionuclide imaging agents when they submit an investigational new drug (IND) application before beginning clinical trials. Costs of studying imaging agents could be reduced if researchers did not have to repeat these studies for compounds that have already been studied by other researchers.
The IDR Team’s strategy for sharing toxicology and pharmacokinetics data is the creation of a database called Free RIDES, an acronym for “Radiopharmaceutical + Imaging Database to Enable Sharing.” The idea is based on the way protein sequences are already shared through public databases, such as the RCSB Protein Data Bank (PDB). When publishing a paper with data about a new protein sequence in a journal, researchers must submit the new protein sequence to a community-endorsed, public database. The team suggests a similar process when publishing studies that test a new radionuclide in humans for the first time. IDR Team 1B listed several key steps to achieving this goal. First, funding would be obtained for computing resources to create the database. Then, a prototype database would be built with volunteer contributions of toxicology and pharmacokinetics data for some number of radionuclides. For this process to succeed, NIH and FDA would be asked to require submission of these data to the Free RIDES database. Then, it would also be necessary to work with peer-reviewed journals to implement use of the database prior to publishing.
The IDR Team noted that there would be both short-term and long-term benefits of such a system. The short-term benefits would be for researchers applying for INDs for imaging agents. They could use the toxicology and pharmacokinetics database rather than having to conduct these studies again—a free ride. The system could reduce the time and money associated with studying imaging agents by approximately 2 to 4 months and drop the cost by approximately $200,000 per compound that received a “free ride.” The long-term benefit would be the ability to build a scientific case to eliminate the need for toxicology and pharmacokinetics studies prior to clinical studies that involve microdoses of radionuclides. A microdose refers to a dose that is so low that it is unlikely to produce whole-body effects, as a therapeutic agents does. In other words, if toxicology and pharmacokinetics data consistently show no detrimental effects with microdoses of radionuclides, the requirement for these data to be obtained prior to applying for an IND could be removed.
Top-Down Approach
The second approach the IDR Team identified for developing useful imaging assays is the top-down approach, as shown in Figure 1. This involves starting with the clinical needs for imaging and then identifying radionuclide imaging agents to meet these needs. The challenge for this approach is identifying the principal components of many diseases, such as inflammation, for which imaging agents are needed. These disease components would identify areas of opportunity for developing new radionuclides that would have broad applicability. Then, research funding could be provided for studies seeking relevant imaging agents. The IDR Team developed a strategy to determine these components using an expert consensus panel.
First, the IDR Team suggested that a search for potential disease components be conducted using a mathematical approach. The components identified would later be used to begin discussion among a panel of experts. To conduct the initial search, publications and other disease data would be analyzed using a statistical procedure, such as principal component analysis, to identify commonalities among diseases. Conducting this analysis would first involve obtaining a grant to pay for the analysis. The grant would be publicized as a challenge grant, meaning people would submit algorithms for the analysis. The person with the best algorithm would receive the grant to conduct the analysis. The IDR Team suggested that once the principal component analysis was conducted, there would be an initial workshop and
“request for comments” period in which stakeholders in the nuclear medicine community could give their opinion on whether the analysis identified the proper disease components.
Next, an expert panel would be assembled to further debate the disease components identified. Ultimately, the panel would identify several major disease components on which imaging research could be focused. The panel would develop a report on these high-impact areas so that funding for imaging assay research could be prioritized. Team 1B suggested that the panel include physicians, basic scientists, applied imaging scientists, business leaders, patient advocates, and regulatory body representatives. After the expert panel identifies the highest priority disease components, the IDR Team envisions that a funding body, such as the NIH, would develop grants for research of imaging assays addressing these components. Team 1B suggested a time frame of 2 years to complete the process of identifying disease components and allotting funding. Using this strategy, the chance of developing an imaging assay with both clinical relevance and marketability would increase, as research groups worked toward several main target areas.
IDR Team 1B’s overall goal was to find ways to develop imaging assays with clinical, industrial, and academic value. Within this goal, they identified approaches that could be used to develop these assays: the bottom-up approach and the top-down approach. They also wanted to provide examples of innovative ways to improve these approaches. Specifically, these strategies included a toxicology database to reduce research time and cost, as well as an expert consensus panel to identify major imaging needs. Using these strategies to develop high-value imaging assays is an important way to ensure the future development and supply of radionuclides.
• Nsikan Akpan, University of California, Santa Cruz
• Richard E. Carson, Yale University
• Anthony J. Di Pasqua, University of North Texas System College of Pharmacy
• Michael T. Fasullo, College of Nansoscale Sciences and Engineering
• Sundaresan Gobalakrishnan, Virginia Commonwealth University
• Daniel A. Pryma, University of Pennsylvania
• Michael van Dam, University of California, Los Angeles
• Alan Waltar, Pacific Northwest National Laboratory
• Weian Zhao, University of California, Irvine
IDR TEAM SUMMARY—GROUP 1C
Nsikan Akpan, NAKFI Science Writing Scholar University of California, Santa Cruz
IDR Team 1C was asked to identify ways that will ensure the future development and supply of radionuclides and radiopharmaceuticals for diagnostic imaging and therapy in the United States.
One challenge presented to the IDR Team is securing the domestic supply of technetium-99m (Tc-99m) and its precursor molybdenum-99 (Mo-99) in order to achieve nuclear medicine independence. The United States imports its entire supply of Mo-99 from a small number of overseas facilities.
The IDR Team disagreed with this directive, concluding that the time is ripe to alleviate nuclear medicine’s Tc-99m reliance by focusing on the development of radiopharmaceuticals for positron emission tomography (PET) that do not require Tc-99m. They stated that if new PET radiopharmaceuticals are approved to replace Tc-99m radiopharmaceuticals, technetium would not be a major player in 10 years, with the SPECT industry outdated and not growing. This paradigm shift would obviate some consequences related to Tc-99m shortages and help safeguard the future success of the field as a whole.
Adopting a policy of domestic production of other isotopes could also answer the logistical dilemmas associated with purchasing Tc-99m abroad. Though President Obama ratified the American Medical Isotope Production Act in 2013 to protect the interests of nuclear medicine, more emphasis has been placed on phasing out enriched uranium rather than securing and stabilizing the medical isotope market.
The IDR Team agreed on four specific challenges that must be addressed to advance next-generation PET radiopharmaceuticals and nuclear science.
First, the desires of scientists, academic institutions, and the general public require better harmony. Current regulatory procedures unnecessarily stymie progress in nuclear medicine. A lack of coherent, consistent regulatory standards across government agencies, including the Food and Drug Administration (FDA), Centers for Medicare & Medicaid Services (CMS), and the National Institutes of Health, threatens innovation. The IDR Team proposed resolutions to expedite the bench-to-bedside journey
for the most promising pharmaceuticals and for diagnostic tools solely for basic scientific discovery.
Second, a new dawn for radiopharmaceuticals will require fresh technology, with the IDR Team offering a list of the most pressing needs.
Third, recruiting the next generation of nuclear medicine professionals is critical, which was a concept that permeated throughout the NAKFI Conference. The IDR Team proposed a specific plan to encourage more young scientists to enter nuclear medicine.
Finally, education initiatives should be implemented to correct public myths connected to the safety of nuclear medicine.
The IDR Team ultimately developed a series of directives to tackle each hurdle, which they posited as the framework for a future policy statement to be issued by a major nuclear organization, such as the Society of Nuclear Medicine and Molecular Imaging.
Trial Harmony
Research institutions, scientists, the general public, and regulatory agencies aspire for radiopharmaceuticals to satisfy different objectives.
Research institutions have altruistic ambitions, but also must develop unique ideas that will bolster their reputations and attract young investigators. Scientists want freedom to make their developments accessible to as many patients as possible. Patients want safe, effective drugs as quickly as possible at the lowest costs. Regulatory bodies are charged with brokering those goals, while providing safety for patients.
Yet the development cycle for radiopharmaceuticals is long, expensive, and unnecessarily risky due mainly to shifting targets in the regulatory approval of nascent radiopharmaceuticals, according to the IDR Team.
They recommended holding a stakeholder summit where investigators, regulators, and institutional officials could discuss “trial harmony”—new guidelines to unify trial endpoints.
Regulatory cohesion
One major impediment raised during the meeting was shifting targets in the regulatory approval of radiopharmaceuticals. The two regulatory organizations in question are FDA and CMS.
Recent tribulations with Amyvid (18F-florbetapir), the first FDA-approved PET radiotracer for Alzheimer’s disease, was offered as a prime
example. Spawned from academic research, the tracer received FDA approval in January 2011, but patient access was ultimately denied by CMS last autumn. The decision represented a scary prospect for nuclear medicine, suggesting that heavy investments in the research and development of prospective tools may not be reimbursed at the end of the day. The average cost of inventing and obtaining FDA approval of a new drug is $1.3 billion, according to Eli Lilly, which now owns Amyvid and stands to lose up to $650 million across international markets. Moreover, patents and licenses are now vital revenue streams for many universities.
The IDR Team asked for more transparency with regulatory standards from the FDA and CMS.
The FDA’s current regulations for investigational new drugs (INDs) and diagnostic agents were viewed by the team as another barrier. These rules require an imaging radiotracer to meet many of same standards as a therapeutic drug. This reasoning is unnecessarily stifling, in the team’s opinion, because exposure to the radiotracer will likely be limited, relative to the use of an average drug. For instance, a person with heart disease might take a beta blocker every day for 30 years, whereas a PET imaging agent for cancer may only be used a handful of times for an individual patient. Furthermore, radiotracers are given in miniscule amounts, below the expected limits for any physiologic effects.
The team proposed expanding the FDA’s Radioactive Drug Research Committee (RDRC) Program. This program permits human use of fledgling radiotracers, but without the heavy restrictions and costs of maintaining IND status.
Precision medicine is an arena that stands to benefit from these policy changes, according to the IDR Team, with companion diagnostics and theranostics given as examples. Companion diagnostics are assays that screen genes, mutations, or proteins and provide direct treatment strategies for a patient, while a theranostic is a diagnostic agent that can also be repurposed into a drug.
Pairing diagnostics with therapeutics tackles disease heterogeneity, while cutting costs. This is especially true with theranostics because the diagnostic agent often provides the molecular backbone for the drug, limiting the amount of time and effort invested in medicinal chemistry. Expanding the RDRC program could give scientists more freedom for basic research on companion diagnostic and theranostic radiotracers, while also unclogging the pipeline for drugs specifically destined for the clinical track.
Academic cohesion
A further ambition for the stakeholder summit is promoting greater academic cooperation within nuclear medicine. One problem within diagnostic imaging research is that individual scientists set their own standards for determining the sensitivity and specificity of new radioagents. This trend makes it harder to compare results among researchers studying radiotracers for identical biological targets.
The IDR Team recommended harmonizing trial endpoints to permit more prospective meta-analysis among laboratories working on similar classes of radiopharmaceuticals. The IDR Team cited the success observed with the Alzheimer’s Disease Neuroimaging Initiative (ADNI), in which investigators across the world have partnered to collect findings on predetermined groups of imaging biomarkers for the neurological disorder.
The goal would be to unify the way scientists design their projects, produce gold standards for radiopharmaceutical validation, and gauge the best endpoints for clinical design.
Marquee example: prostate cancer PET imaging agents
Prostate cancer was posited as a promising realm for attempting trial harmony.
The principal test for the disease—serum screening for prostate-specific antigen—is prone to false positives with localized low-grade prostate tumors, leading to unnecessary biopsies. The assay also tends to miss small high-grade tumors, creating the opportunity for missed diagnosis. Finally, this testing gives little to no information on the location of the disease in the body (which can have significant implications on prognosis and optimal treatment strategy). More sensitive and accurate diagnostics are needed to guide therapy and measure clinical responses to cancer drugs.
PET radiotracers are gaining momentum as clinical tools for evaluating prostate tumor biology. 11C-choline and 18F-fluorocholine have garnered much attention in Europe and Japan over the past few years as lipogenesis markers. The former was approved by the FDA to detect recurrent prostate cancer in 2012, but the approval is currently limited to a single academic institution. The androgen-receptor tracer 18F-fluoro-5α-dihydrotestosterone and 18F-fluorodeoxyglucose (FDG) are in clinical trials as early-stage assessors of treatment outcomes, while other tracers are proving useful for staging or tracking bone metastasis.
Trial harmony and prospective meta-analysis could bolster this emerging field by unifying perspectives on which novel PET tracers should be pursued in earnest.
Future Technology for Nuclear Medicine
The IDR Team listed advances in technology that could help PET agents drive the modern era of radiopharmaceuticals.
The installation of small medical reactors and cyclotrons at more hospitals across the nation would facilitate the generation and testing of radionuclides for novel pharmaceuticals as well as PET mainstays such as 18F-FDG. This would accelerate the synthesis, dispensing, and quality control of radioactive raw materials and radiopharmaceuticals for investigators. Furthermore, neutron-activation of radionuclides could be performed at hospitals, increasing the impact of this type of pharmaceutical, a specific example of which is TheraSphere. Evaluating new or rarely used isotopes for PET would also be aided by having more small reactors.
The emergence of microfluidics has made it possible to create accelerators on a microchip, and the IDR Team felt further investments should be made into microreactor technology. The broader incorporation of nanotechnology lends itself to developing fully automated pathways that reduce personnel costs and synthesis time for producing new delivery vehicles for radiopharmaceuticals.
Most of these concepts represent multidisciplinary challenges, according to the IDR Team, that do not fit under current funding opportunities from the National Institutes of Health and that were once, but no longer, funded by the Department of Energy. New avenues for support would be discussed at the summit.
Teaching Millennials About Nuclear Medicine
Two groups were spotlighted as important targets for education initiatives: young scientists and the general public.
The next generation of nuclear medicine scientists
No amount of innovation can replace bright minds, and a concern that loomed over the entire conference is the nuclear industry’s aging
population. Nuclear medicine is no exception, and the IDR Team reviewed recruitment strategies.
One fear is that students who could make contributions to this field are missing the chance because they are simply unaware of its existence. More programs that introduce undergraduates and recent graduates to concepts in radiation research were posited. Striking early could plant the seeds of expertise that bloom into basic research careers in nuclear medicine.
On the clinical side, the IDR Team said medical training programs need more synergy in their curricula. They described the typical medical university as having many nuclear disciplines or courses—for example, radiation oncology, diagnostic radiology, and nuclear medicine—but surprisingly little interplay between the programs of study. They would push for more interaction to streamline the education process.
The final suggestion involved the creation of a summer program for interested students. Dubbed the “theranostic boot camp,” this program would provide hands-on experience and be geared toward reviewing the multiple disciplines involved with precision radiopharmaceuticals. North Carolina was suggested as a prime location for the camp, because NC State University runs a research reactor, while the University of North Carolina Chapel Hill has a renowned nuclear medicine program.
A new nuclear dictionary for the public
Public relations must improve for nuclear medicine, according to the IDR Team. Limiting radiation exposure is paramount feature of nuclear medicine, while many radiopharmaceuticals, especially diagnostic tracers, are safe and pose few health risks. Yet trepidation still filters into general attitudes toward radiation therapy and diagnostics.
A new vocabulary is needed so patients can comprehend radiation safety on their own. A visual lexicon, such as a color-coding system for risk levels, could provide a simple, but engaging solution.


















