7
Biotechnology and Germplasm Conservation
Biotechnology requires germplasm, as both raw material and a source of natural variation. As a way of shaping and using genetic information, biotechnology has implication for germplasm conservation and use. This chapter discusses these opportunities and the allocation of resources.
Although biotechnology is commonly thought of as recombinant DNA (deoxyribonucleic acid) technology, it is used here in a broader sense to include tissue culture, cryopreservation, plant micropropagation, and animal regeneration from early embryos. Biotechnology influences germplasm conservation in several ways. First, it provides alternatives in some cases to conserving whole organisms. Second, it can assist with the exchange of germplasm. Third, the techniques of molecular biology can be applied to the problems of managing and using germplasm. The fourth influence results from the increased demand for germplasm and conservation services by the biotechnologists themselves.
Molecular biology provides a scientific framework that describes the elements of the genetic system as sequences of four nucleotide bases that make up DNA. Knowledge of how these DNA sequences are expressed and how expression is regulated and coordinated during development is growing rapidly. It is now commonplace to introduce foreign gene constructs into an organism, and the ability to add regulatory sequences that determine when and how strongly the introduced genes will be expressed to alter the phenotype of the recipient is often possible. The numbers of genes that have been isolated,
cloned, and sequenced increase daily, and the information thus obtained already represents a genetic resource of considerable and growing scientific and commercial value.
ALTERNATIVES TO STORING SEEDS AND WHOLE ORGANISMS
Any living cell appears to have the genetic information needed to regenerate the complete organism, but among more complex and highly organized multicellular forms, relatively few have developed this capacity as a means of reproduction. In plants, clonal or asexual propagation occurs naturally in the form of propagules such as bulbs, tubers, runners, and stolons. Horticultural propagation techniques as well as in vitro propagation methods have been developed for many plants and have extensive commercial use. In vitro cell and tissues cultures are providing new approaches to multiply germplasm resources and open opportunities for long-term cryopreservation.
In Vitro Conservation of Plants
The germplasm of vegetatively propagated crops is normally stored and shipped as tubers, corns, rhizomes, roots, or in the case of woody perennials, as cuttings. Although some of these crops have seeds that could be stored, they are frequently highly heterozygous and thus do not breed true to type from seed. Perennials are usually maintained ex situ either in plantations (for example, temperate and tropical tree fruits, sugarcane, and strawberries) or are stored during the winter and planted the following spring (for example, potatoes, sweet potatoes). For these and other crops whose seeds are shortlived, there are a number of potential advantages in storing them as in vitro cultures. These include economies in space and labor and, provided that appropriate conservation methods are chosen, greater genetic stability. Disadvantages, however, include the need for special facilities and trained technicians and the small amount of experience in the use of in vitro methods for germplasm conservation on a large scale. The following summary is drawn from recent reviews of the subject (Withers, 1985, 1986, 1991a,b)
In vitro storage of germplasm was first suggested in the mid-1970s (Henshaw, 1975; Morel, 1975). Although whole plants can be regenerated from the cells of many plants (totipotent), the preservation of unorganized cultures, such as cell suspensions or callus, carries some risk of generating spontaneous somaclonal mutants. In contrast, cultures of organized meristems frequently are not only more stable but also propagate more rapidly, since meristematic areas do
not have to differentiate after recovery from storage. Effective storage systems are often inexpensive and easy to maintain, and should reduce the overall work load in germplasm banks. Frequent monitoring of culture viability and for microbial contamination should not be necessary.
The full exploitation of in vitro genetic conservation is impeded if a species cannot be propagated from cultured tissue or cells. For example, a satisfactory mass propagation technique for coconut palm does not yet exist (unlike oil palm, which can be mass-propagated via callus culture). Coconut callus can be grown in vitro, but it cannot be reproducibly induced to form new plantlets. Plantlets can only be produced in vitro from germinating zygotic embryos. Each coconut embryo produces only one plant so there is no multiplication of the original material. Similarly, the impact of in vitro culture on conserving woody species has been less than for other species because of difficulties in the culture and regeneration of woody species. In these cases, insufficient research has been conducted to develop appropriate in vitro culture methods. A thorough investigation of

Advances in biotechnology provide tools for conserving and managing plant genetic resources. Here a small clump of pine tree plantlets, regenerated by individual plant cells in tissue culture, grow in a test tube. Credit: U.S. Agency for International Development.
the technical problems encountered in developing in vitro methods for woody species is urgently needed (National Research Council, 1991b).
In Vitro Storage
Two approaches to in vitro storage have proved successful, namely slow growth and cryopreservation. The slow growth approach involves applying retardant chemicals or reducing the culture temperature. Subculture intervals can be extended up to 1 or 2 years, thereby greatly reducing the time, labor, and materials required to maintain the cultures. Slower growth reduces the frequency of cell division and consequently the number of times a random mutation is multiplied in the culture. Such genetic changes that occur in tissue cultures are called somaclonal variations. Stress is an intrinsic factor in slow growth, and little is known about its effect on somaclonal variation. What began as a clonal culture may change into a population of cells consisting of the original genotype plus variant genotypes. Also, stress factors may act differently on such a population of genotypes, favoring some somaclonal variants. This could result in a changed population of cells and the failure to conserve the genetic integrity of the original clonal material. Undifferentiated callus cultures are more susceptible to somaclonal variation than organized tissue systems, such as shoot cultures. Only organized cultures are recommended for slow-growth storage. This technique of long-term root, tuber, or shoot tissue culture storage is well developed for some crops such as banana (Musa).
Cryopreservation involves suspending growth by keeping cultures at an ultralow temperature, typically that of liquid nitrogen (-196°C). It offers the prospect of storage for indefinite periods with minimal risk. However, certain cultures can suffer damage during freezing and thawing. Until recently, routine cryopreservation methods were available or under development only for cell suspension cultures. Larger, organized structures frequently suffered serious structural injury and loss of viability.
However, two new approaches to cryopreservation may lead to more widespread applications for genetic conservation. They focus on reducing cell damage from ice crystal formation. One approach is through vitrification of cellular water by a cryoprotectant mixture and the other involves encapsulation of specimens within an alginate gel that is then dehydrated. For vitrification the specimen is infused with a cryoprotectant mixture that promotes the conversion of much of the cellular water into a noncrystalline, vitreous solid when rapidly cooled (Sakai et al., 1990). For encapsulation the specimen, such

Seed sample stored in liquid nitrogen are inspected at the National Seed Storage Laboratory at Fort Collins, Colorado. Cryopreservation cuts storage costs because seed life is extended. Fewer grow-outs are needed to replenish seed when it begins to lose its ability to sprout. Credit: U.S. Department of Agriculture, Agriculture Research Service.
as a shoot tip or somatic embryo, is encased in an alginate gel to form an artificial seed. This artificial seed is then dehydrated in her air before cooling (Dereuddre et al., 1990). The enveloping gel appears to minimize deleterious effect from dehydration and also protects the specimen from physical damage, being larger and more robust than an isolated shoot tip or embryo.
Despite these optimistic developments using plants tissues, much research is needed to bring the level of development of cryopreservation techniques for plant materials to that available for microbial systems and animal semen and embryos. Many technical barriers remain that prevent the routine use of cryopreservation for plant meristems, pollen, and plant cell cultures. No conservation collection or germplasm banks are yet using cryopreservation for non-seed germplasm storage, although several are involved in cryopreservation research.
Collecting Germplasm In Vitro
The laboratory facilities required for in vitro culture normally include a sterilizer, a laminar flow hood (to provide a clean, sterile
work surface),incubators, growth chamber and greenhouse in addition to a pure water source, chemicals, glass and plastic labware, and other standard items of equipment. Some success has been claimed for collecting material in the field directly in vitro. The level of sophistication ranges from using a fully equipped local laboratory, for transfer of collected material as soon as possible, to working in the field in a portable glove box or on a clean table with a simple box-like cover to exclude contamination. Following surface sterilization, explants are removed and either inoculated to sterile culture media in the field or held in sterile buffer for later inoculation in the laboratory (Sossou et al., 1987; Withers, 1987). A cruder alternative is to sterilize tissue explants with nontoxic agents and inoculate them to media containing antibiotics and fungicides.
Although there is some potential for using in vitro collection for vegetatively propagated crops and those with short-lived seeds, there are serious implications for plant quarantine since the collected explants may carry pests or pathogens that might well be detected or excluded in material cultured by more rigorous methods.
In Vitro Exchange of Germplasm
During the past 20 years, advances in tissue culture technology have led to the development of commercial micropropagation, a relatively new industry that supplies young plants for a variety of horticultural, agronomic, and plantation crops (Constantine, 1986). One consequence has been the rapid development of in vitro exchange as a means of transferring germplasm between different laboratories. An International Board for Plant Genetic Resources data base assembled by a survey (Withers and Wheelans, 1986) indicated that some 135 plant genera were exchange from 1980 to 1985. Some 94 percent of more than 480 attempts were successful. The same data show that 49 countries attempted 110 international exchanges.
Several of the international agriculture research centers now distribute in vitro cultures. For example, the distribution of potato germplasm in this form is now a routine procedure. Shoot cultures, which are inoculated into small tubes of semisolid medium, are cultured for 2 to 3 weeks to induce rooting and reveal any microbial contamination. Transfer to fresh culture medium for a passage before potting enhanced survival. In 1984 the Centro Internacional de la Papa (International Potato Center) exported 2,500 culture tubes to more than 30 countries. In that year more than 88 percent of certified-pathogen free material was distributed as cultures rather than as field-grown tubes (Huaman, 1986). Cultures are now being replaced by the distribution
of small tubers produced in vitro. These are more robust than plantlets. They can be stored in the dark for more than 4 months, and the recipient can plant them directly into pots or nursery beds without a further culture step. In modern potato breeding programs, in vitro cultures provide a disease-free reference collection during the years of field testing needed to select the most desirable clones.
In 1984 the Centro Internacional de Agricultura Tropical (CIAT, International Center for Tropical Agriculture) distributed more than 2,000 accessions of 238 cassava clones as cultures to 21 countries and received 240 clones as in vitro cultures from Costa Rica, Guatemala, Panama, and the Philippines. By 1985 the International Institute for Tropical Agriculture (IITA) had distributed in vitro cultures of sweet potato germplasm to 47 countries. IITA also imports sweet potato, cassava, yam, and cocoyam as in vitro cultures and is testing in vitro tubers of yam as a more convenient material for germplasm exchange (Ng,1986).
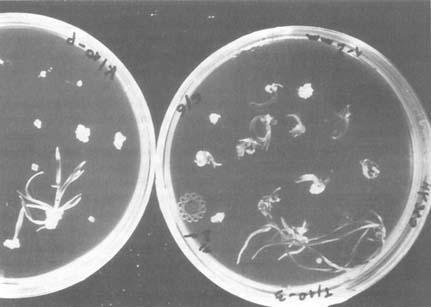
Pictured are haploid barley plants recovered from anther culture. Used to create diploids, they greatly accelerate the production of true breeding lines. Credit: Food and Agriculture Organization of the United Nations.
In Vitro Conservation of Animals
The in vitro conservation of livestock is discussed in greater detail in a separate report of the committee (National Research Council, 1993). The two principal methods for conserving animals germplasm in vitro are as frozen semen and embryos. To recover the germplasm, both methods require the maintenance of female animals for either insemination or implantation. However, embryos have an advantage over semen because they provide the complete genotype.
In some domestic species embryos for transfer or cryopreservation are generally collected from animals that have received hormonal treatment to induce an excess of eggs for insemination (superovulation). The resulting embryos are evaluated for their potential to produce a pregnancy or to withstand freezing. Embryos from cattle, sheep, goats, and horses have all been frozen in liquid nitrogen, thawed, and successfully implanted. Similar success has not been achieved for swine embryos.
The collection of embryos can be carried out surgically or non-surgically, by flushing. Implantation may also be a surgical or non-surgical procedure, depending on the species. Generally one embryo is transferred per recipient in cattle and horses, two in sheep and 20 in pigs. Related technologies allow in vitro fertilization, embryo sex determination, splitting of embryos into parts to produce identical clones, and the creation of chimeras by associating cells derived from embryos of different genotypes. Mapletoft (1987) recently reviewed embryo transfer technology.
Singh (1988) summarizes the potentials for disease transmission by embryo transfer. None of the disease agents tested replicated in the embryonic cells of embryos in which the zona pellucida was intact. However, because some agents can replicate in the cells of zona pellucida-free embryos, the integrity of this structure is important. Proper washing in the presence of antibiotics was shown to be effective in rendering embryos free of many disease agents (Singh, 1988). However, two or three bovine viruses and five of the porcine viruses tested adhered to the zona pellucida after in vitro viral exposure and washing. These agents could thus be transmitted by embryos if they are excreted in the reproductive tract. Only one disease agent (pseudorabies virus) was found to be transmitted when pig embryos were transferred from infected donors.
Two approaches are used to ensure that embryos are free of pathogenic organisms. The tradition method is based on testing donor animals over extended periods of time to establish that they are healthy. The other is based on the research on the transmissibility of agents
via embryo transfer and requires that embryos be processed in accordance with international standards. Although the protocols have been shown to be reliable for certain diseases, the health of the embryo depends entirely on the care taken during embryo collection and processing. The second method is therefore likely to require additional safeguards, such as a period of quarantine for the recipients, particularly when testing for the more serious diseases.
MOLECULAR CONSERVATION TECHNOLOGIES
Within the past decade rapid progress has been made in developing and applying methods to identify, isolate, and characterize individual genes at the molecular level. At the larger scale of the genome, the DNA sequence can be cut up into fragments and reassembled in the form of a linkage map. While the task of characterizing germplasm at the molecular level has just begun, these molecular technologies can offer new approaches for both preserving and evaluating germplasm resources.
DNA as a Genetic Resource
The DNA sequences in the genomes of germplasm accessions are the sources of the genes required in breeding programs. When identified and isolated by cloning, these genes may be used to prepare transgenic organisms that express them. Transformation may avoid much of the genetic disruption that accompanies sexual hybridization and, since it is not limited by sexual compatibility, can make use of genes from other life forms. The genes used to breed crop plants that are resistant to various herbicides or insect larvae or to increase the growth rate of transgenic fish, are most conveniently conserved as cloned DNA. The extent of this kind of conservation is limited by the technical problems of identifying and cloning the commercially important genes that breeders would like to have and obtaining high rates of stable transformation in a broad range of recipient genera. Genes of this kind that have been cloned so far are also protected by patents and so are not freely available to anyone who wishes to exploit them commercially (see Chapter 12).
For economic and technical reasons it is unlikely, in the foreseeable future, that gene synthesis will make physical storage of germplasm in the form of seeds, whole plants, or tissue cultures obsolete. The conservation of DNA and the assembly of sequence data bases are not alternatives to conventional germplasm conservation because they are not coordinated in a genome (Peacock, 1984). Recent
progress in the synthesis of artificial chromosomes in yeast raises the possibility of conserving coordinated assemblies of genes that would allow more complex phenotypic changes to be engineered. To be of practical use, however, DNA and artificial chromosomes require the conservation of recipient organism. Their genetic information is incomplete. Unlike a seed or an embryo in a surrogate mother, a DNA sequence cannot yet be used to regenerate a whole organism. Nevertheless, cloned genes, genomic libraries, and sequence data bases have significant potential uses in germplasm conservation and management. They may be more compatible with genetic stock collections, rather than national and international germplasm collections because of the specialty nature of the material and data.
In the broader context of conserving global genetic diversity, some (Adams et al., 1992; Adams, 1993) have proposed the collection of small samples of plant material (leaf tissue, seeds, and so on) for conservation as sources of DNA. Tissue samples from all taxa threatened with extinction would be collected and preserved in liquid nitrogen. These samples would, in theory, be available indefinitely. DNA extraction would be deferred until needed. At that time, the DNA could be isolated, immobilized on membranes, used as a source of specific genes or DNA sequences (see polymerase chain reaction technique below), and then returned to liquid nitrogen storage. Several conservation groups are now establishing a network of such DNA banks with duplicate samples as a safeguard against loss through extinction (Adams et al., 1992; Adams, 1993). This technique may be useful for the many underscribed or unstudied plant species with seeds that cannot be stored, but it is unlikely to be of direct value for those of recognized importance that are already conserved in germplasm banks.
DNA Sequence Data Banks
There is now much DNA sequencing carried out in various laboratories worldwide that the ability to compare new sequences with those that have already been described and well characterized is of considerable importance. Comparisons may suggest unsuspected functions or may reveal useful homologies between unrelated organisms. Several organizations support the central storage and coordination of DNA sequence information. The two major data banks are the European Molecular Biology Laboratory and, in the United States, GenBank, which is operated by IntelliGenetics and the U.S. Department of Energy's Los Alamos National Laboratory.
The rate at which data on DNA sequences is accumulating is likely to continue to increase exponentially in the near term as a
result of such major initiatives as sequencing the entire human genome and the genomes of rice and Arabidopsis. This rapidly growing body of data raises important problems of storage and access to facilitate rapid comparisons with newly obtained information.
Restriction Fragment length Polymorphisms
When DNA is extracted from an organism and digested with one or more restriction enzymes and the products (called restriction fragments) are separated by electrophoresis on a gel, the result is a smear of many DNA fragments with different lengths. The longest fragments move slowly in the gel, staying near the origin; the smallest move faster and are farthest from the origin. If the smear is transferred to a membrane by blotting, it may be hybridized with a radiolabelled DNA probe. After autoradiography, the regions of hybridization are revealed as one or more distinct bands at constant and characteristic distances from the origin of the gel. Each band identifies a restriction fragment that contains a stretch of DNA complementary to the sequence of nucleotide bases in the DNA probe. The same digest tested with different DNA probes shows different band patterns that are also constant and characteristic for each probe.
When the DNA digests from separate organisms are compared, the differences in banding patterns are called restriction fragment length polymorphisms (RFLPs) and correspond to points, or very small regions, of physical difference on the chromosomes. The number of polymorphisms (RFLPs) provides some indication of the number of differences that exist between the genomes of the organisms being compared. The RFLPs segregate as allelic differences and may be tested for linkage with each other and with other characters of agronomic importance. RFLPs, in theory, provide an almost inexhaustible number of markers and should make possible the rapid construction of linkage maps even in species in which this has been hampered because of the lack of morphological markers or long generation times.
There are several practical limitations, however. The DNA extracts, digests, gel separation, blots, hybridizations, auto radiography, and pattern interpretation are expensive and time-consuming and require trained personnel. It may be necessary to test several hundred, or even thousand, different DNA probes to find RFLPs suitably spaced throughout the genome. The ideal is to locate one at about every 10 map units. Some of these difficulties may be overcome by automation of the procedures. The analysis of the resulting data can be complex. In some cases the results obtained for one
segregating F2 population cannot be directly applied to an F2 generation from another, different cross because of major duplication at the DNA level that may even obscure the chromosomal assignments obtained with the first set of probes. This difficulty is proving to be an obstacle to the use of RFLPs as an aid to selection in maize breeding, because maize contains an excessive number of polymorphisms in the noncoding regions of its DNA that complicate the banding patterns and often make them difficult to decipher. In contrast, among some inbreeders, such as the tomato, there are not enough RFLPs among commercial cultivars, evidently reflecting their genetic similarity.
Recent research indicates that RFLPs can be used to map quantitative trait loci (Paterson et al., 1988). Linked RFLPs that identify relevant portions of chromosome arms may potentially be used to select characters that are inherently difficult to assay, such as resistance to insects that cannot be reared, drought tolerance, and processing quality of harvested product. In germplasm enhancement programs, linked RFLPs could be used to mark genes for resistance to pests or pathogens that do not occur when or where the work is being done and that cannot be introduced because of quarantine regulations. An example might be breeding U.S. maize for resistance to African streak virus.
A barrier to using RFLPs is the investment of effort needed to find useful probes. Probes could be used to screen germplasm accessions for alternative alleles for important disease resistance loci.
Polymerase Chain Reaction
The polymerase chain reaction (PCR) is an extremely sensitive and accurate method for recovering microgram amounts of single specific DNA sequences present in biological samples at very low concentrations. The reaction requires pairs of single-stranded primers (DNA template molecules) of 20 to 30 bases in length found on opposite strands at either end of the double-stranded DNA sequence of interest. These primers are added to a solution of DNA extracted from the sample, together with the four nucleotide bases that make up DNA and a DNA-polymerizing enzyme from the bacterium Thermophilus aquaticus (Taq), which is stable at high temperatures. The reaction mixture is heated to 92° C for 30 seconds to denature the DNA into single strands, cooled to 50° C for 1 minute to allow annealing to occur, and then heated to 72° C for 2 minutes to allow DNA synthesis to fill in the missing bases from one primer toward the other, using the single strands of sample DNA as templates for the
complementary strand copies. The preparation is then heated again to denature the newly formed double-stranded DNA molecules, and another cycle of cooling-incubation-heating is begun. Since the primers are present in excess at each cycle, the amount of double-stranded DNA selected by the primers steadily builds up until after 40 cycles the preparation is sufficiently pure for that segment of DNA that it can be readily sequenced.
The ability to use PCR to select and amplify specific DNA sequences from desiccated dead seeds and inviable frozen semen and embryos raises the question of whether these "dead" materials are useful genetic resources. Depending on their scientific interest, commercial value, or rarity, there may be a case for keeping them as a source of particular DNA sequences. Already collected, stored, inventoried, and evaluated, there would be little more to do with them until they needed for DNA isolation. However, the fact that they had become inviable could mean that the original storage conditions were unsatisfactory and that, if left in place, further changes to the DNA might eventually reduce their usefulness. The DNA banks discussed above avoid these difficulties.
The use of PCR to recover sequences from herbarium specimens (Rogers and Bendich, 1985) may mean that specimens should be regarded as a form of germplasm. PCR analyses will be useful for comparative genetic studies, which, until now, have depended on morphological or chemical comparisons rather than functional genetic analyses using the tools of molecular biology.
As with isolated DNA, cloned DNA, and DNA sequence information stored in data banks, the DNA of inviable specimens and cryogenically stored tissues cannot be used directly to reconstitute an organism. These forms of DNA are presently only valuable as a germplasm resource to the extent that they can be incorporated into other living organisms or for research. For the long-term preservation of genetic variability, they are only useful to the extent that viable hosts in which they are readily expressed can also be maintained.
PCR is a useful technique, but the requirement for primers means that it can only be used to find sequences that are already known in some detail. PCR is useful, however, for recovering allelic forms of well-characterized genes from any source, whether or not it is viable.
Randomly Amplified Polymorphic DNA Markers
PCR can also be used with single, arbitrary, 9- or 10-base primers to generate probes for detecting RFLPs. These polymorphisms are
called randomly amplified polymorphic DNA or RAPD markers (Welsh and McClelland, 1990; Williams et al., 1990). It is expected that any arbitrary 9- or 10-base sequence will occur at a sufficient frequency at points on opposite strands of DNA so that synthesis of the intervening segments will occur in PCR from points where the primers has hybridized. The optimum size range of these fragments can be controlled by prior digestion of the extracted DNA with one or more restriction enzymes. The several kinds of DNA fragments amplified in the PCR step can then be used as probes to detect RFLPs. The advantage of the method is that the original primers are relatively inexpensive and easy to make and that many probes are available that should cover all parts of the genome (Anderson and Fairbanks, 1990).
Other Uses for Probes
DNA probes of particular genes can be used to detect their chromosomal location by in situ hybridization and to detect the presence of those genes in segregating populations or new alleles in germplasm accessions. Although these methods are now used in plant breeding in material with known genetic backgrounds, they will likely also be of use as a research tool in germplasm conservation when simpler, cheaper methods are available. However, the same technology can detect the presence of pest and pathogen nucleic acids and could simplify quarantine procedures by greatly reducing the time needed to test imported plant materials for diseases or organism, including viruses, of quarantine significance.
RECOMMENDATIONS
Recent advances in biotechnology provide powerful tools conserving, evaluating, and using genetic resources. Cloned DNA fragments synthetic DNA are unlikely to replace conventionally stored seeds other germplasm in the foreseeable future. In any case whole organisms must be conserved as recipients for the expression of introduced DNA.
Transformation works for a number of plants and animals, and is now being applied to commercial cultivars or breeds. Many field tests of various transformed crop plants have been carried out since 1987. However, for the present, the impact of transformation on germplasm enhancement will likely be small because of the effort and resources now required to characterize, isolate, and clone useful genes.
PCR for recovering and amplifying selected DNA sequences makes possible comparative studies of DNA from herbarium and museum specimens and dead seeds and sperm. As a consequence, these materials may become much more useful for ecogeographic and evolutionary genetic studies of germplasm. They may eventually become a source of alleles at well-characterized loci.
Recombinant DNA biotechnology is rapidly evolving. Its techniques are expensive, time-consuming, and complicated and call for highly trained staff. Until it becomes more user friendly, it is best regarded as a potentially useful adjunct to germplasm conservation rather than a means of revolutionizing this important activity. This limitation applies in particular to the larger collections of plant germplasm, where current resources seriously limit activities.
Research is needed to apply in vitro culture and cryogenic storage methods to a broad range of plants and animals germplasm.
In vitro conservation of plant germplasm as growing tissue cultures has potential as a means of conserving forms that cannot easily be kept as seeds, and for some species, the maintenance of cultured plantlets in slow growth is proving more efficient and less expensive than maintaining whole plants. In vitro techniques are a useful means of exchanging and distributing some clonally propagated germplasm. Cryogenic preservation has potential for long term storage but the method is not as well developed for plants as it is semen and embryos of some livestock species.
Biotechnology research efforts should focus on developing enhanced methods for characterizing, managing, and using germplasm resources. These efforts should include the urgent need for more effective data handling systems for storage, retrieval, and sequence comparisons, some of which may be by-products of the considerable investment in sequencing the human genome.
Biotechnological innovations, including computerized information handling techniques, heighten the utility of germplasm collections rather than obviating their need. The rapid development of DNA sequence data banks, plasmid libraries, and cloned DNA fragments has created a genetic resource of growing size and importance. However, with this expansion comes a vast amount of data and information. Without sufficient attention to the management of these data, the resources they describe will be of little value.
Biotechnology could improve access to collections by providing tools for characterizing their accessions. Information on the genetic similarity between accessions could aid in developing priorities for acquisition. By linking molecular markers to specifically desired genetic
traits, biotechnology may provide a mechanism for rapid searching of large collections without the need for lengthy field trials. Molecular techniques for characterizing genetic material, such as restriction fragment length polymorphism analysis, appear likely to provide the breeder with greater efficiency in selecting and developing new breeding lines and varieties.
DNA sequences used as probes can detect viruses in germplasm bank materials and also reveal RFLPs that can be used to construct linkage maps and, as linked markers, to select desirable traits. Linked RFLP markers might be used to detect genes for resistance to pests and pathogens that themselves cannot be used because of quarantine restrictions. Information from RFLPs could potentially assist in the selection of core subsets of larger germplasm collections by providing another measure of genetic diversity, but only if the technology becomes much less expensive.
















