2
Why a Children's Vaccine Initiative?
PROGRESS TOWARD UNIVERSAL CHILDHOOD IMMUNIZATION
Vaccines are among the most affordable and effective health interventions available today. The development, introduction, and widespread use of vaccines in industrialized and developing countries have resulted in considerable progress against some of the most devastating infections of humankind. Indeed, the world's only complete victory over an infectious agent resulted from a vaccine. Smallpox, which many believe caused more death and sickness than any other infectious disease, was eradicated from the world in the late 1970s following a well-planned and highly effective vaccination campaign. Public health officials in the Americas are now close to achieving a similar victory over another infectious scourge: poliomyelitis.
One of the largest and most successful efforts to date to capitalize on the tremendous potential of vaccines is the Expanded Program on Immunization (EPI). The EPI, which is headquartered at the World Health Organization (WHO) and supported by numerous individual governments, nongovernmental organizations, and bilateral and multilateral agencies, was established in 1974. Its aim was to build on the success of WHO's Smallpox Eradication Program and to assist national immunization programs in the developing world. To advance the goal of universal childhood immunization, the EPI supports national governments in their efforts to implement effective vaccine delivery programs.
Among the greatest hurdles faced by the EPI during its first years of
operation was starting national immunization programs where none had existed before (Robbins, 1991). Political commitment to the goals of the EPI had to be secured from more than 90 countries. Immunization personnel had to be trained, systems had to be established to deliver and monitor immunization efforts, and adequate national and international resources had to be put in place to support the massive undertaking.
Armed with vaccines against just six diseases—diphtheria, pertussis, tetanus, tuberculosis, polio, and measles—the EPI has made remarkable strides toward achieving universal childhood immunization. By the end of 1991, an estimated 80 percent of the world's infants were reported to be vaccinated with BCG (bacillus Calmette-Guérin, the antigen used to vaccinate individuals against tuberculosis), measles vaccine, diphtheria and tetanus toxoids and pertussis vaccine (DTP), and oral polio vaccine (OPV) (Pan American Health Organization, 1993; UNICEF, 1992). Each year, EPI-sponsored immunization programs prevent some 2.9 million deaths from measles, neonatal tetanus, and pertussis as well as 440,000 cases of polio worldwide (Kim-Farley et al., 1992; Pan American Health Organization, 1993; World Health Organization, 1992).
This great achievement stands in sharp contrast to the situation in the mid-1970s, when less than 5 percent of the developing world's children were adequately immunized and when nearly 5 million children died each year from vaccine-preventable diseases (UNICEF, 1989).
Limits of the Expanded Program on Immunization
Despite tremendous progress during the 1980s toward the goal of universal immunization coverage, there is concern that the success of the six-vaccine EPI effort cannot continue indefinitely (Claquin, 1989, 1990; Poore et al., 1993; REACH Project, 1990; Robbins and Freeman, 1988; Rosenthal, 1990). Each year, a new and larger cohort of children at risk for vaccine-preventable diseases must be immunized. Some 20 percent of the world's children, many in the poorest and most remote areas of the world, have yet to be reached at all by national immunization programs (Pan American Health Organization, 1993). Indeed, more than 2 million deaths and 5 million cases of disability still occur as a result of diseases that are preventable by vaccination (Pan American Health Organization, 1993; Ransome-Kuti, 1991).
It is also worth noting that the six existing vaccines offered through EPI offset only a fraction of the burden of infectious diseases affecting children in developing countries (Figure 2-1). For example, acute diarrhea causes 3 million to 5 million deaths annually and accounts for at least one third of
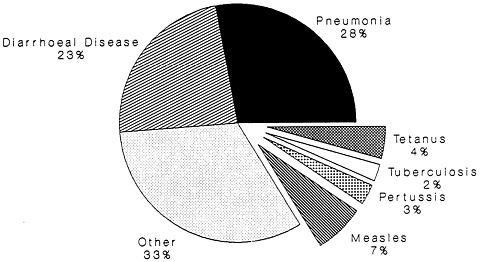
FIGURE 2-1
Under-five deaths by cause, developing countries, 1990. Cut slices: Vaccine-preventable diseases. SOURCE: State of the World's Children, 1993.
the deaths in children under age 5 years. Acute respiratory infections kill more than 2 million people every year (Ransome-Kuti, 1991), and an estimated 1 million to 2 million people, most of them children, die from malaria each year (Institute of Medicine, 1991). Vaccines for these three sets of conditions are in various stages of development but are not yet available for use.
These concerns, coupled with the recognition that genetic engineering and new vaccine technologies could permit the development of a new generation of childhood vaccines and that the translation of these scientific advances to vaccines needed by developing countries was lagging, led to the establishment of the Children's Vaccine Initiative (CVI).
THE CHILDREN'S VACCINE INITIATIVE
The CVI is both a concept and an organization. The initial focus of the CVI, launched after the World Summit for Children in New York City in September 1990, was to accelerate efforts to develop vaccines that could enhance the performance of EPI (World Health Organization/Children's Vaccine Initiative, 1991a, 1992). A number of specific, desirable features of future children's vaccines were proposed (see the box "What Is the Children's Vaccine Initiative?"). Vaccines incorporating some or all of these
characteristics offered the potential for protecting more of the world's children against a larger number of diseases at a lower cost per child or per disease prevented (Robbins, 1991). As secondary, longer-term objectives, it was hoped that the CVI could facilitate efforts to ensure an adequate supply of vaccines for children in the developing world and simplify the complex logistics of vaccine delivery.
Over time, the mission and goals of the CVI have matured. In particular, those involved in the initiative recognized that vaccine development, production, and delivery cannot be considered independently—they are intimately linked. Underlying this shift in thinking has been the realization that the manufacture of vaccines cannot be assured without taking into account the prospective development of new and improved vaccines. Vaccine development, in turn, cannot be successful without taking into account such issues as demand, intellectual property rights, production capabilities, and technology transfer.
The organization of the global CVI has also changed since the World Summit for Children. The founders of the CVI (the Rockefeller Foundation, United Nations Development Program [UNDP], United Nations Children's Fund [UNICEF], the World Bank, and the WHO) recognized at the outset that no single agency or organization has the resources and capabilities to achieve the goals of the CVI. They recognized further that the CVI needed to involve many different entities to achieve the vision of the CVI. This recognition led to the formation of the CVI consultative group, which is composed of representatives of national immunization programs, multilateral agencies, nongovernmental organizations, development-stage firms, commercial vaccine manufacturers, public-sector vaccine manufacturers, and national development assistance agencies. The consultative group, which meets annually, provides an international forum for discussion of new CVI initiatives and for marshaling broad-based support for the CVI.
The activities of the CVI are carried out primarily through product development groups and task forces. CVI task forces examine strategic, logistic, and policy issues relevant to the industrial development and introduction of CVI vaccine products. Task forces focused on the following topics have been established to date: priority setting and strategic planning, relations with vaccine development collaborators, situation analysis of the global vaccine supply, assessment of national vaccine regulatory capabilities and needs, and strengthening national epidemiological capacities to ensure the best use of vaccines. A new task force on the management of DTP combinations for the developing world has been proposed as a means to plan, coordinate, and implement a global effort to ensure the development and supply of quality DTP combination vaccines to developing countries. However, these activities are likely to be beyond the capabilities of a single task force and will need to be implemented through other means as well
|
What Is the Children's Vaccine Initiative? |
|
|
The Children's Vaccine Initiative (CVI), is an international effort to harness new technologies to advance the immunization of children. At the World Summit for Children in New York City in September 1990, world leaders called for an acceleration of the application of current science to the development of new and improved childhood vaccines. Preceding the summit, world vaccine experts proposed a number of desirable features for future children's vaccines. They are that the vaccines be: |
|
|
|
• single dose, |
|
|
• administered near birth, |
|
|
• combined in novel ways, |
|
|
• heat stable, |
|
|
• effective against diseases for which vaccines are unavailable, |
|
|
and |
|
|
• affordable. |
|
The goals of the CVI have matured. Those involved in the initiative have come to recognize that vaccine development is intimately linked to issues of vaccine production and supply. These issues deserve equal consideration. Underlying this shift in thinking is the realization that the manufacture of vaccines cannot be assured without taking into account the prospective development of new vaccines. Development, in turn, cannot be successful without taking into account such issues as local production, intellectual property rights, technology transfer, and collaboration with the private sector. |
|
|
The Children's Vaccine Initiative, which is headquartered at the World Health Organization in Geneva, Switzerland, is cosponsored by five organizations: the United Nations International Children's Fund, the United Nations Development Program, the Rockefeller Foundation, the World Bank, and the World Health Organization. |
|
(Philip K. Russell, Johns Hopkins University, personal communication, 1993).
The CVI product development groups promote, facilitate, and manage projects that lead to the development of vaccines and related products. The three current product development groups are focusing their efforts on a single-dose tetanus toxoid vaccine, a heat-stable oral polio vaccine, and
an effective measles vaccine for administration earlier in life. The heat-stable oral polio vaccine and single-dose tetanus toxoid product development groups, which were formed in late 1991, are working with a few academic and industrial partners and have identified some promising techniques. The measles product development group became operational in March 1993. Other product development groups will be established as needs and priorities are identified and objectives set.
The success of the CVI depends on the cooperation of vaccine manufacturers, governments, and multinational organizations, such as UNICEF and the Pan American Health Organization, which supply vaccines to much of the developing world. Effective cooperation will allow vaccine developers to create new and improved vaccines of use to suppliers, and it will help the suppliers make long-term plans that take into account the vaccines of the future.
Characteristics of CVI Vaccines
The long-term goal of the CVI is to develop a means of immunizing children at birth against all important disease threats with a single procedure. World vaccine experts who met before the World Summit for Children agreed upon six desirable features of future childhood vaccines. They should be single dose, administered near birth, combined in novel ways, heat stable, effective against additional diseases, and affordable. A vaccine that has some or all of these characteristics has the potential to save money, thereby allowing more money to be spent on reaching the 20 percent of children in the world who are currently unprotected (Robbins, 1991; World Health Organization/Children's Vaccine Initiative, 1991b). Some vaccines developed by the CVI will be targeted exclusively for the populations of developing countries (e.g., shigella, malaria, and dengue); others, such as combination vaccines made up of existing and improved vaccines (e.g., DTP-hepatitis B vaccine combinations), are needed by the populations of both industrialized and developing countries.
Vaccines Should Be Single Dose
Protecting a child against the six basic childhood diseases currently requires adherence to a complicated vaccination schedule (see Appendix G). The WHO immunization schedule recommends that children receive single doses each of BCG and OPV at birth and then three doses of DTP and OPV each at ages 6, 10, and 14 weeks. Measles vaccine is administered at
age 9 months. The complexity of the vaccination schedule contributes to and exacerbates two categories of problems common to many immunization programs: high dropout rates and missed opportunities for vaccination (de Quadros et al., 1992).
Whether because of the lack of information, difficulty getting to the health clinic, or inappropriate clinic hours, families may not take their children for necessary and additional booster shots, and thus drop out of the vaccination program. In other instances, health-care workers may not check whether a child requires any immunizations during a visit to a health clinic for reasons other than vaccination. In either case, children may not receive important vaccinations. Efforts to track and completely immunize every child are labor and resource intensive (de Quadros et al., 1992). Reducing the number of required vaccine doses to protect a child fully, and hence the number of contacts with the health-care system, would reduce costs and lead to enhanced coverage against disease.
Vaccines Should Be Administered Near Birth
Some currently available vaccines, for example, measles vaccine, are not immunogenic in very young children because of interference from maternal antibody. Yet by the time the vaccine is administered to an older infant, the child may already have been exposed to or contracted the disease. A vaccine that could be administered near birth would have a substantial impact on the incidence of some vaccine-preventable diseases in young children.
Vaccines Should Be Combined in Novel Ways
The discomfort of injections and the effort required to bring children to health clinics discourages many necessary visits. Combination vaccines would reduce the number of required contacts with the health-care system by protecting against more diseases in a single administration. Integrating combination vaccines into the existing vaccine schedule could be done at minimal cost—the cost of the vaccine itself—since investments in vaccine delivery systems have already been made. Major efforts are under way around the globe to develop combination vaccines by using DTP as the base to which additional antigens can be attached (Chapter 4).
There are a number of novel vaccine delivery systems in various stages of research and development that have the potential to ease vaccine administration; some such systems may even obviate the need for booster doses, needles, and syringes. Sustained-release vaccines, for example, would
release immunogenic antigens over time, thereby foregoing the for subsequent doses. Increased widespread use of oral vaccines could eliminate patient concerns about the discomfort associated with injections. Not only would clinic visits be more tolerable to patients but the costs and risks of using syringes and other equipment would also be reduced. Children could receive many vaccinations at one time, painlessly (Robbins, 1991).
Vaccines Should Be Heat Stable
Without refrigeration, vaccines have a limited usable shelf-life, and refrigeration and maintenance of the ''cold chain" have been critical limiting factors of EPI in many countries (de Quadros et al., 1992; Pan American Health Organization, 1993). An immunization program can extend only as far as the cold chain permits. By extension, an immunization program is only as effective as its cold chain.
The cold chain is expensive and difficult to operate and maintain (de Quadros et al., 1992; Pan American Health Organization, 1993), demanding refrigeration at every stop along the route from the central manufacturing facility to the point at which a child is vaccinated (Table 2–1). The public health costs when the cold chain fails are much higher, however. In such cases, children may receive ineffective vaccines. The result may be a serious erosion of public confidence in the immunization program as children become sick with the very disease against which they were vaccinated. It has been estimated that the costs associated with enhancing or extending the cold chain approach half of the total costs of immunization programs (de Quadros et al., 1992).
Increasing the heat stability of vaccine could extend the immunization efforts while at the same time reducing vaccine wastage and the cost of refrigeration. Heat-stable vaccines could be carried by health-care workers to areas previously inaccessible because of the limitations of the cold chain. The number of vaccine failures resulting from a temperature-related loss of potency could be markedly reduced (de Quadros et al., 1992).
Vaccines Should Be Effective Against Additional Diseases
The current set of vaccines offered through EPI has inherent limits. Six antigens can control only six diseases. Many other vaccine-preventable diseases are managed by less effective and often more costly methods of
TABLE 2–1 Maximum Storage Times and Temperatures for Selected Vaccines at Various Points from the Central Store to the Health Center
prevention or treatment.
There are many diseases against which vaccines may be a useful preventive tool, including malaria, which kills more than 1 million children each year, pneumococcal disease in children, and rotavirus. In 1986, in response to a request from the National Institute of Allergy and Infectious Diseases, the Institute of Medicine evaluated the costs and potential benefits of over 20 new or improved vaccines of importance to the developing world (Institute of Medicine, 1986a,b). The development costs, in 1985 dollars, were estimated to range between $10 million and $50 million per vaccine. Since that time, the UNDP/WHO Program for Vaccine Development has undertaken similar priority-setting exercises, as is the CVI itself (see Chapter 6) (World Health Organization, 1991; World Health Organization/Children's Vaccine Initiative, 1992, 1993).
In the last 10 years, WHO has sought to encourage researchers to study the health challenges facing developing countries. The UNDP/WHO Program for Vaccine Development and the UNDP/World Bank/WHO Special Program for Research and Development in Tropical Diseases, for example, were both created to bring laboratory investigators face-to-face with the problems encountered in the field. The Program for Vaccine Development is primarily a research-stimulating and research-supporting effort. The participants in the program, almost exclusively research scientists, have worked to bring vaccine research to 'proof of principle,' the point at which product development can begin. Proof of principle is the point at which the most intensive CVI efforts are needed. However, certain technologies that are important in early vaccine development, such as technologies to achieve a single dose or heat stability, will also be a focus of the CVI.
It was once hoped that if the public sector identified the needs and funded basic research, private industry would develop technically feasible vaccines (Institute of Medicine, 1992; World Health Organization/Children's Vaccine Initiative, 1991a). However, as discussed in subsequent chapters of this report, this has not yet happened; the barriers and impediments to the development of vaccines for the industrialized and the developing world are complex and variable.
Vaccines Should Be Affordable
The affordability of vaccines is of critical importance to EPI programs (Kim-Farley et al., 1992; Robbins and Freeman, 1988). Vaccine costs currently represent only about 10 percent of the overall expense of administering EPI (Figure 2-2), but a very large percentage of the foreign exchange input into national immunization programs (John Gilmartin,
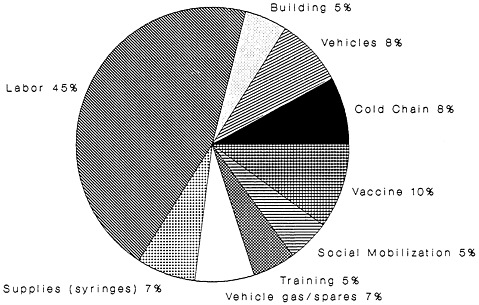
FIGURE 2-2
Estimated distribution of Expanded Program on Immunization delivery costs.
Note: This breakdown is an average global estimate. Actual costs vary considerably from country to country. Supervision costs range from 3 to 5 percent of labor costs. SOURCE: Amie Batson, World Health Organization/Expanded Program on Immunization, 1992.
UNICEF, personal communication, 1993). It now costs EPI an estimated $15 to immunize a child living in a developing country; the cost of the vaccine amounts to less than $1 (Kim-Farley et al., 1992). Not included in this calculation are investments in capital infrastructure, such as the health centers where children are vaccinated.
Studies conducted at the start of the EPI program found that it cost approximately $5.00 to immunize one child, with the cost of vaccine amounting to $0.50 (Robbins, 1991). The increased cost of immunizing a child in 1993 is not due to rising vaccine and material costs alone (supplies, transportation, cold chain equipment, and facilities), the latter of which have actually decreased (Robbins, 1991); rather, it suggests that the children who were most easily vaccinated were immunized first, increasing the per-child cost of vaccinating the remaining children (Robbins, 1991).
If past experience is any indication, the prices of new and improved vaccines on the international market fall over time. Since it was first available 10 years ago, for example, hepatitis B vaccine has dropped to less than one one-hundredth of the original price (Mahoney, 1990; Maynard, 1989). Advances in technology and competition seem likely to bring down the price of the Haemophilus influenzae type b vaccine (Hib) as well. The
introduction of Hib and hepatitis B vaccine could be the first major additions to EPI since its inception. One critical area of concern to CVI and EPI is how to ensure that new vaccines are affordable to developing countries when they first appear on the market. Adding new and improved vaccines to the EPI, whether such vaccines are purchased from international suppliers or produced locally, will inevitably increase the costs to EPI. For this reason, the committee's recommended strategy, outlined in Chapter 7, includes provisions that could change the current system for the development of affordable vaccines and the procurement of vaccines, subsidizing the prices paid by UNICEF and other agencies, and providing higher returns to private developers and manufacturers, where appropriate.
Concerns About the CVI
Some critics of the CVI approach to vaccine innovation argue that resources would be better spent improving means of delivering existing vaccines to currently underserved populations rather than developing new and more sophisticated vaccines. Others have cautioned that the prices of new vaccines may prohibit their use in developing countries (Kim-Farley et al., 1992) or lead to a reduction in existing coverage under EPI. At the same time, the sustainability of even the existing EPI is being called into question (Claquin, 1989, 1990; Poore et al., 1993; REACH Project, 1990). Many developing countries rely on outside support for their immunization programs and are unlikely to be able to sustain these efforts in the future without a continued infusion of outside resources (Claquin, 1989, 1990; REACH Project, 1990; Rosenthal, 1990); thus, expanding the program depends upon persuading donor organizations to provide more funding for global vaccine procurement (an unlikely strategy in the light of scarce resources and competing priorities) or reducing the costs of immunization. The costs of EPI can be lowered primarily in two ways: reducing the number of contacts required to protect a child and distributing vaccines that are easier to administer and that are less dependent on refrigeration. CVI is seeking to tackle both of these areas.
There is a range of complicated and practical impediments to introducing new and technologically complex vaccines into EPI. Integrating new vaccines into the EPI will require some retraining of over 100,000 healthcare workers—a Herculean task. Furthermore, countries that currently make some vaccines for their populations may not have access to, or in some cases the capability to manufacture, novel vaccines that employ complex technologies. It is feared that the capability and know-how to manufacture new vaccines will be tightly held by only a few vaccine manufacturers. Indeed, without an effort to ensure that all children have access to new and
improved-quality vaccines, children in industrialized countries will have access to the new combination vaccines, while children in developing countries will remain dependent on single-antigen vaccines and will not be protected from additional important diseases (e.g., malaria and acute respiratory infections).
RELEVANCE OF THE CVI TO U.S. IMMUNIZATION EFFORTS
Status of Immunization Efforts in the United States
The use of childhood vaccines in the United States has caused the number of cases of diphtheria, an acute bacterial infection, to fall from nearly 6,000 in 1950 to 3 or 4 a year currently (U.S. Department of Health and Human Services, 1992). Cases of pertussis (whooping cough), another illness caused by a bacterium, have dropped from over 120,000 in 1950 to 4,500 in 1990 (U.S. Department of Health and Human Services, 1992). The incidence of measles, a viral illness, has fallen off dramatically in the last 30 years, despite a major increase in 1990 (U.S. Department of Health and Human Services, 1992). Similar dramatic reductions in disease incidence have been reported for mumps, polio, rubella (German measles), and most recently, Haemophilus influenzae type b (U.S. Department of Health and Human Services, 1992).
As a counterpoint to this record of achievement against many infectious diseases is the disappointing reality that a significant proportion of children under the age of 5 years, the most vulnerable age group for vaccine-preventable diseases, are not fully vaccinated (Cutts et al., 1992a,b; National Vaccine Advisory Committee, 1991; Peter, 1992; Schlenker et al., 1992). For example, in 1985, the last year for which national data are available, only 55 percent of U.S. preschoolers received three or more doses of polio vaccine; just 65 percent were fully vaccinated with DTP (U.S. Department of Health and Human Services, 1992). Vaccine coverage rises sharply after age 5 years—to over 95 percent—since all states require proof of adequate immunization prior to enrollment in school (Cutts et al., 1992a,b; Hinman, 1991; Plotkin and Plotkin, 1988).
The Centers for Disease Control and Prevention (CDC), in conjunction with state and local health departments, recently completed retrospective assessments of vaccine coverage in 20 U.S. cities (Centers for Disease Control and Prevention, 1992a). Data from nine cities indicate that although 90 percent of children had one vaccination before their first birthday and although most children began their vaccinations on schedule, fewer than half of the children surveyed were fully immunized by age 2 years
(Centers for Disease Control and Prevention, 1992b). Another recent survey of 51 immunization projects nationwide indicated that the overall immunization levels of children under 2 years of age were low, with 16 projects reporting immunization levels below 50 percent (Centers for Disease Control and Prevention, 1992b).
Other industrialized and developing countries have been able to achieve higher rates of immunization. For example, in 1990, over 97 percent of Swedish, Danish, and Swiss children were reported to be fully immunized against polio by 1 year of age, as were over 95 percent of 1-year-olds in Pakistan, Costa Rica, and Mexico (Liu and Rosenbaum, 1992; UNICEF, 1992, 1993).
To improve immunization levels in preschool-age children, CDC embarked on an Infant Immunization Initiative with state and local health departments in 1991 (Centers for Disease Control and Prevention, 1992). The goal of the initiative is to develop novel strategies in vaccine delivery (Centers for Disease Control and Prevention, 1992a; Freeman et al., 1993). Although the U.S. Public Health Service has set a goal for the year 2000 of ensuring 90 percent immunization coverage for preschoolers (U.S. Department of Health and Human Services, 1992), given the current rates of vaccination, few believe that this goal will be attained. In fact, immunization levels among children under age 5 years for many diseases have actually declined since the late 1970s (Liu and Rosenbaum, 1992; U.S. Department of Health and Human Services, 1992).
From a public health perspective, this trend is alarming. A fundamental principle of disease control by vaccination is that enough people must be immunized to maintain so-called herd immunity. When vaccine coverage drops below a certain level, local outbreaks and, potentially, epidemics are possible. The resurgence of measles during the late 1980s and early 1990s is an example of what can happen when vaccination is carried out incompletely and vaccination rates are low (National Vaccine Advisory Committee, 1991; Schlenker et al., 1992). The number of reported cases of measles in 1990 (27,786) was the highest since 1977 and was nearly 20-fold more than was documented in 1983, the year the fewest number of cases was reported. About half of the reported cases in 1990 were among preschool-age children; among vaccine-eligible preschoolers, nearly 80 percent were unvaccinated (Cutts et al., 1992a; National Vaccine Advisory Committee, 1991).
A Role for the CVI in the United States
Despite the relatively plentiful supply of childhood vaccines in the United States, many children do not undergo the complete series of recommended immunizations on time. Although it was not the mandate of this committee
Despite the relatively plentiful supply of childhood vaccines in the United States, many children do not undergo the complete series of recommended immunizations on time. Although it was not the mandate of this committee to address this particular concern, many of the barriers that prevent children in the United States from receiving the full benefit of vaccines are similar to those in other parts of the world. These include missed vaccination opportunities, deficiencies in the health-care delivery system (most acutely in the public sector), inadequate access to health care, and lack of public awareness of required immunizations (National Vaccine Advisory Committee, 1991; Peter, 1992; Schulte et al., 1991; Szilagyi et al., 1993).
As in other parts of the world, the vaccination schedule for U.S. children, developed by the CDC's Advisory Committee on Immunization Practices, is complex (see Appendix G). Children living in the United States are required to receive more vaccines than children living in countries taking part in EPI (ten versus six). Achieving complete immunization in the United States entails a minimum of five visits to the doctor before age 2 years and additional visits at ages 4–6 years, 14–16 years, and every 10 years thereafter. The actual number of visits to a health-care provider is considerably higher, since many parents and pediatricians prefer to spread the number of immunizations out rather than give three or four shots in one sitting.
The sheer number of vaccines and contacts with the health-care system required to fully protect a child has led U.S. vaccine manufacturers to pursue the development of combination vaccines (see Chapter 4). Many of these products will be as useful to EPI as they are to the public health goals of the United States. In many respects, then, the United States and countries served by the EPI are facing a similar set of problems, and there is potential for overlap in the solutions being considered. Therefore, the vaccine development efforts of U.S. firms have relevance and are of vital interest to the international CVI.
REFERENCES
Brenzel L. Costs of EPI: 1990. A Review of Cost and Cost-Effectiveness Studies (1979–1989). REACH Project. Arlington, Virginia: John Snow, Inc.
Centers for Disease Control and Prevention. 1992. Retrospective survey results of immunization status of children at second birthday by immunization project: school year 1991/1992. letter from Walter A. Orenstein, December 15.
Centers for Disease Control and Prevention. 1992b. Retrospective assessment of vaccination coverage among school-aged children—selected U.S. cities, 1991. Morbidity and Mortality Weekly Report 41(6):103–107.
Claquin P. 1989. Sustainability of EPI: Utopia or Survival? REACH Project. June. Arlington, Virginia: John Snow, Inc.
Claquin P. 1990. Overview of Issues in the Sustainability of EPI. REACH Project. October. Arlington, Virginia: John Snow, Inc.
Cutts FT, Orenstein WA, Bernier RH. 1992a. Causes of low preschool immunization coverage in the United States. Annual Review of Public Health 13:385–398.
Cutts FT, Zell ER, Mason D, Bernier RH, Dini EF, Orenstein WA. 1992b. Monitoring progress towards U.S. preschool immunization goals. Journal of the American Medical Association 267:1952–1955.
Day LM. 1990. Toward Ensuring the Financial Sustainability of EPI. REACH Project. October. Arlington, Virginia: John Snow, Inc.
de Quadros CA, Carrasco P, Olive JM. 1992. Desired field performance characteristics of new improved vaccines for the developing world. Paper presented at the NIAID Conference on Vaccines and Public Health: Assessing Technologies and Global Policies for the Children's Vaccine Initiative, November 5–6, 1992, Bethesda, Maryland.
Freeman P, Johnson K, Babcock J. 1993. A health challenge for the states: Achieving full benefit of childhood immunization. Occasional Paper. February. The John W. McCormack Institute of Public Affairs, University of Massachusetts at Boston.
Hinman AR. 1991. What will it take to fully protect all American children with vaccines? American Journal of Disease Control 145:559–562.
Institute of Medicine. 1991. Malaria: Obstacles and Opportunities. Washington, D.C.: National Academy Press.
Institute of Medicine. 1986a. New Vaccine Development: Establishing Priorities. Volume I. Diseases of Importance in the United States. Washington, D.C.: National Academy Press.
Institute of Medicine. 1986b. New Vaccine Development: Establishing Priorities. Volume II. Diseases of Importance in the Developing World. Washington, D.C.: National Academy Press.
Institute of Medicine. 1992. Emerging Infections. Washington, D.C.: National Academy Press.
Kim-Farley R and The Expanded Programme on Immunization Team. 1992. Global immunization. Annual Review of Public Health 13:223–237.
Johnson KA. 1987. Who's Watching Our Children's Health? December. Washington, D.C.: Children's Defense Fund.
Liu JT, Rosenbaum S. 1992. Medicaid and Childhood Immunizations: A National Study. Children's Defense Fund. January. Washington, D.C.
Mahoney RT. 1990. Cost of plasma-derived hepatitis B vaccine production. Vaccine 8:397–402.
Maynard JE. 1989. Global control of hepatitis B through vaccination: Role of hepatitis B vaccine in the Expanded Programme on Immunization. Reviews of Infectious Diseases 11(Suppl. 3): S574–S578.
National Vaccine Advisory Committee. 1991. The measles epidemic. Journal of the American Medical Association 226:1547–1552.
National Vaccine Advisory Committee. 1992. Access to Childhood Immunizations: Recommendations and Strategies for Action. April. Rockville, Maryland.
Pan American Health Organization. 1992. Progress in the worldwide polio eradication effort. EPI Newsletter 14(6):1–2.
Pan American Health Organization. 1993. Global EPI progress reviewed. EPI Newsletter 15(1):6.
Peter G. 1992. Childhood immunizations. New England Journal of Medicine 327:1795–1800.
Plotkin SL Plotkin SA. 1988. A short history of vaccines. Vaccines, Plotkin SA and Mortimer EA, eds. Philadelphia: W.B. Saunders Company.
Poore P, Cutts F, Seaman J. 1993. Universal childhood immunization: Is it sustainable? Lancet 341:58.
Ramachandran S Russell PK. 1992. Draft. A new technologic synthesis: The Children's Vaccine Initiative. Paper presented at the NIAID Conference on Vaccines and Public Health: Assessing Technologies and Global Policies for the Children's Vaccine Initiative, November 5–6, 1992, Bethesda, Maryland.
Ransome-Kuti O. 1991. Introductory remarks. Meeting of the Consultative Group, Children's Vaccine Initiative. December 16–17, 1991. World Health Organization, Geneva.
REACH Project. 1990. Immunization Sustainability Study. April. Arlington, Virginia: John Snow, Inc.
Robbins T. 1988–1989. Prospectus from WHO, EPI Technical Series: The Cold Chain Product Information Sheets, No. 1. Geneva: World Health Organization.
Robbins T. 1991. The Children's Vaccine Initiative: A prospectus. September. Boston University School of Public Health. Unpublished.
Robbins T Freeman P. 1988. Obstacles to developing vaccines for the Third World. Scientific American. November. 126–133.
Rosenthal GR. 1990. The Economic Burden of Sustainable EPI Implications for Donor Policy. REACH Project. February. Arlington, Virginia: John Snow, Inc.
Schlenker TL, Bain C, Baughman AL, Hadler SC. 1992. Measles herd immunity. Journal of the American Medical Association 267:823–826.
Schulte JM, Bown GR, Zetzman MR, Schwartz B, Green G, Haley CE, Anderson RJ. 1991. Changing immunization referral patterns among pediatricians and family practice physicians, Dallas County, Texas, 1988. Pediatrics 87:204–207.
Szilagyi PG, Rodewald LE, Humiston SG, Raubertas RF, Cove LA, Doane CB, Lind PH, Tobin MS, Roghmann KJ, Hall CB. 1993. Missed opportunities for childhood vaccinations in office practices and the effect on vaccination status. Pediatrics 91(1):1–7.
UNICEF. 1989. The State of the World's Children. Oxford: Oxford University Press.
UNICEF. 1992. The State of the World's Children. Oxford: Oxford University Press.
UNICEF. 1993. The State of the World's Children. Oxford: Oxford University Press.
U.S. Congress, House. 1966. Childhood Immunizations. A report prepared by the Subcommittee on Health and the Environment, Committee on Energy and Commerce. September. Washington, D.C.
U.S. Department of Health and Human Services. 1992. Health United States 1991. May. Hyattsville, Maryland.
World Health Organization. 1991. Programme for Vaccine Development: A WHO/UNDP Partnership. December. Geneva.
World Health Organization. 1992. EPI for the 1990s. Geneva.
World Health Organization/Children's Vaccine Initiative. 1991a. Meeting of the Consultative Group. Geneva.
World Health Organization/Children's Vaccine Initiative. 1991b. Why a Consultative Group on the Children's Vaccine Initiative and Why Now? December. Geneva.
World Health Organization/Children's Vaccine Initiative. 1992. Meeting of the Consultative Group. Geneva.
World Health Organization/Children's Vaccine Initiative. 1993. Task Force on Priority Setting and Strategic Planning. Geneva.

















