1
Introduction
Lead is a ubiquitous toxicant. It is especially toxic to young children and the fetus, and it was estimated that in 1984 about 6 million children and 400,000 fetuses in the United States were exposed to lead at concentrations to an extent that placed them at risk of adverse health effects (i.e., blood lead concentrations of at least 10 micrograms per deciliter (µg/dL) (CDC, 1991). Equally important, past screening programs were based on the blood lead concentrations of the U.S. Centers for Disease Control and Prevention (CDC) guideline of 25 µg/dL. Screening programs identify about 12,000 children with evidence of lead toxicity each year (ATSDR, 1988), but results of screening programs might seriously underestimate the magnitude of childhood lead exposure, primarily because few children are screened and because the false-negative rate of screening is high when screening is done with erythrocyte protoporphyrin.
In 1985, the CDC screening guideline for childhood blood lead associated with toxicity changed from a minimum of 30 µg/dL to a minimum of 25 µg/dL (CDC, 1985). The CDC guideline was set on the basis of health implications; of sensitivity, specificity, and cost-effectiveness of a screening program; and of the feasibility of effective intervention and followup. Evidence gathered since 1985 has shown that lead at less than half the previous CDC guideline (EPA, 1986a; Grant and Davis, 1989) can cause impairment of cognitive and physical development in children and increases in blood pressure in adults. The U.S. Environmental Protection Agency (EPA) Science Advisory Board has therefore proposed setting 10 µg/dL as a maximal safe blood lead con-
(EPA, 1990a). In response to the same evidence of effects at 10 µg/dL and even below, CDC has recently revised its 1985 statement (CDC, 1991). The 1991 statement's major points include (1) an acknowledgment that the current evidence on adverse effects associated with low-dose lead exposure requires a response from the federal medical and public-health community, (2) a reduction in the 1985 intervention or action concentration from 25 µg/dL to 10 µg/dL, and (3) the implementation of a multitiered, graded response that depends on measured blood lead concentrations. Responses will range from community-level actions to reduce lead exposure to emergency medical responses.
Persons exposed to lead and with blood lead above 10 µg/dL are likely to number in the millions, so appropriate methods for measuring concentrations of lead in blood at the revised guidelines will need to be developed and refined. In addition, new reliable and reproducible techniques for measuring lead in blood and other tissues, such as bone, will also need to be developed. Methods for detecting and measuring low-dose lead biologic markers are also needed, because the erythrocyte protoporphyrin (EP) test lacks sensitivity at blood lead concentrations below 25 µg/dL. For those reasons, the U.S. Agency for Toxic Substances and Disease Registry (ATSDR) requested that the National Research Council (NRC) provide information on techniques for measuring environmental exposure of sensitive populations to lead. The Board on Environmental Studies and Toxicology in the NRC Commission on Life Sciences formed the Committee on Measuring Lead Exposure in Critical Populations to meet the need.
PERSPECTIVE ON ISSUES
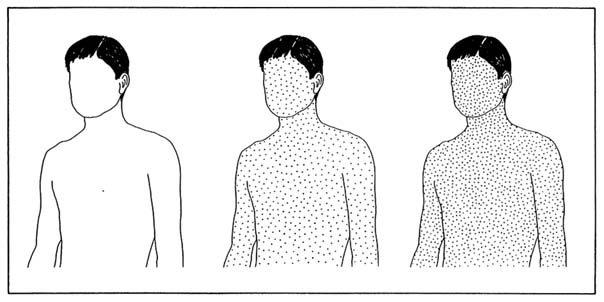
The finding of health effects of lead at blood concentrations previously considered low (i.e., about 10 µg/dL) is not surprising. First, the typical body burdens of lead in modern North Americans are at least 300 times greater than (reconstructed) burdens in North American Indians before European settlement (Ericson et al., 1991; Patterson et al., 1991). Thus, 10 µg/dL is not low, compared with the concentrations that used to prevail until relatively recently in humans. The increasing body burdens of lead with time are illustrated in Figure 1-1. Second, lead interferes with normal cellular calcium metabolism, causing intracellular
buildup of calcium. It binds normally to most calcium-activated proteins with 100,000 times the affinity of calcium; once bound, it interferes with the normal actions of these proteins.
Calcium and calcium-binding proteins serve as the messengers for many basic cellular processes. Some lead-caused disturbances, such as activation of protein kinease, show a dose-response relationship with no evidence of a threshold—hence the apparent absence of a threshold for some of the adverse health effects of lead. Third, death from encephalopathy or massive brain damage is common in children with untreated blood lead concentrations of 150 µg/dL and higher (NRC, 1972), and approximately 10% of the concentration that can cause death from brain damage might cause cognitive disturbances (as shown in epidemiologic studies).
Regulation of most toxic substances is based on safety factors: the presumed "safe" concentration for exposed people is set to be lower than the lowest-observed-effect concentration in humans by a factor of 10 to 100 (NRC, 1986). In contrast, the mean blood lead concentration of urban black children in 1978 was about one-sixth of the potentially fatal concentration. For all children, the mean was one-tenth of the potentially fatal concentration and was above the concentration at which decrements in IQ and other cognitive entities have since been established. The concentrations of lead in blood at which lead-abatement actions have been recommended over the past several years is shown in Figure 1-2. Cognitive effects of lead have been found in infants and children with blood lead concentrations of 10 µg/dL (ATSDR, 1988; Grant and Davis, 1989; Baghurst et al., 1992; Bellinger et al., 1992; Dietrich, 1992). Other studies have reported effects of lead at 10–15 µg/dL on growth rates, attained stature, birthweight, gestational age, auditory functioning, attention span, blood pressure, and some of the general metabolic pathways (Schwartz et al., 1986; Dietrich et al., 1987a,b, 1989; Schwartz and Otto, 1987, 1991; Shukla et al., 1987, 1991; Bellinger et al., 1988, 1991a; Bornschein et al., 1989; Thomson et al., 1989).
Prevention of disease is preferable to treatment, particularly if treatment is not certain to reverse damage. A wide variety of public agencies and offices are trying to reduce exposures to lead. The greatest successes have been in reducing lead in gasoline and food, and the introduction of new lead into paint and plumbing systems has been substantially reduced. But relatively little has been done to reduce exposure to

FIGURE 1-2 Children's blood lead concentrations at which lead-abatement action has been recommended. Cross-hatch columns, blood lead concentration that if untreated is potentially fatal to young child (150 µg/dL). Black columns, percentage of potentially fatal blood lead concentration at which intervention has been recommended.
existing lead in paint and plumbing systems and the reservoir of lead-contaminated soil and dust. For example, 42 million families live in housing that contains an estimated 3 million tons of lead in paints in the immediate environment (ATSDR, 1988)—equivalent to about 140 lb per household. Ingestion of as little as 2.4 x 10−7% of that household burden each day (150 µg/day) would result in a steady-state aggregate lead concentration now considered toxic. Similarly, over 90% of U.S. housing units have lead-soldered plumbing (Levin, 1986). Occupational exposure at concentrations above those associated with reduced nerve conduction velocity, increased blood pressure, reduced reserve capacity for blood formation, and adverse reproductive effects is still common and legally permissible.
Figures 1-3 and 1-4 show the decline in gasoline lead use and the decline in food lead in the typical infant diet in the United States. The reductions have been accompanied by a substantial reduction in the average blood lead concentration of the population, as shown in Figure 1-3. However, children with high lead exposures to such sources as paint have not benefited proportionately, and the lack of substantial progress in reducing the source of their exposure constitutes a great failure in exposure prevention. Reducing lead in drinking water should be more easily accomplished by controlling corrosion. Apart from the benefits derived from reduced lead exposure, the economic saving in pipe and water-heater replacement would exceed the costs (Levin, 1986, 1987; Levin and Schock, 1991).
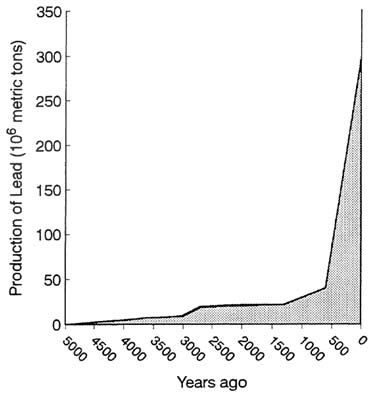
Once lead is mined and introduced into the environment, it persists. Over time, lead in various forms becomes available to the body as small particles. Most of the 300 million metric tons of lead ever produced (Figure 1-5) remains in the environment, largely in soil and dust. That explains, in part, why background concentrations of lead in modern North Americans are higher by a factor of 102–103 than they were in pre-Columbian Americans. Today's production evolves into tomorrow's background exposure, and despite reductions in the use of lead for gasoline, overall lead production continues to grow and federal agencies have not addressed the impact of future increases of lead in the environment.
Until very recently, lead poisoning had been perceived as a potentially fatal illness associated with acutely high exposure to lead and manifested as encephalopathy, acute abdominal colic, and acute kidney damage
(Fanconi's syndrome). The association between chronic large exposure and peripheral neuropathy and gout, mainly in lead workers, has also been recognized. In the United States until about 1970, upper limits of acceptable or normal blood lead concentrations were set just below the concentrations associated with overt clinical illness (60 µg/dL in children, 80 µg/dL in adults), and ''prevention'' referred only to the prevention of clinical symptomatic episodes of lead poisoning. In the late 1960s and early 1970s, however, the concept of preventing lead toxicity became prevalent. In 1972, NRC published Lead: Airborne Lead in Perspective, the first comprehensive document on the subject published in the United States in almost 30 years. That report recommended a "further search for possible subtle effects of prolonged, low-level exposure to lead," inasmuch as the information available at the time did not "afford an adequate basis for the evaluation of this critical concern."
There has since been a great increase in both experimental and clinical investigations, and they have led to the conclusion that chronic low-level lead exposure, insufficient to produce recognizable clinical symptoms of toxicity, has adverse and probably long-lasting effects, particularly on neurodevelopment. An 11-year followup study has indicated that the neurodevelopmental effects persist and can have profound consequences for school achievement (Needleman et al., 1990). The neurodevelopmental studies have led to the identification of infants, children, and pregnant women (as surrogates for fetuses) at greatest risk of lead toxicity from low-level exposure. In this report, these are described as the most sensitive populations. The EPA Science Advisory Board and the Centers for Disease Control and Prevention concluded that published data clearly indicate that the upper limit of acceptable blood lead concentration is 10 µg/dL. This reduced acceptable blood concentration necessitates substantial improvement in analytic methods for measuring lead, as well as development of newer methods for measuring very low blood lead concentrations. Those measurement issues are the focus of this report.
HISTORICAL BACKGROUND
Lead was known and widely used by ancient civilizations. A lead statue displayed in the British Museum, discovered in Turkey, dates
from 6500 BC (Hunter, 1978). Lead has been mined, smelted, and used in cosmetics, internal and topical medicinal preparations, paint pigments, and glazes since early in recorded history (Nriagu, 1983a). In ancient times, the uses of lead medicinally and as a food additive, sweetening agent, and wine stabilizer were probably the principal means by which humans became poisoned by lead. Food and beverage containers crafted from lead compounds, such as pewter, were also likely exposure routes. Since the Middle Ages, a particularly common means of exposure has been the adulteration of wine by lead (Stevenson, 1949; Wedeen, 1984).
Beginning of Public-Health Interest in Lead
Although it is probable that workers involved in the mining, smelting, and working of lead in ancient times were poisoned, technical details to support the assumption are not available. With the advent of the Industrial Revolution, however, interest in hazardous occupational lead exposures began to develop.
The clinical manifestations of lead poisoning were first described by Nikander, a Greek poet and physician, in the second century BC, who wrote:
The harmful cerussa [white lead], that most noxious thing
Which foams like the milk in the earliest spring.
With rough force it falls and the pail beneath fills
This fluid astringes and causes grave ills. …
His feeble limbs droop and all motion is still.
His strength is now spent and unless one soon aids.
The sick man descends to the Stygian shades.
(Nikander, cited by Major, 1945, p. 312)
Physicians who came after Nikander also described the clinical manifestations of lead poisoning, but many failed to make a connection between the symptoms and the causative agent. Even today, the clinical diagnosis of lead poisoning from low doses is elusive (Harris and Elsea, 1967; Crosby, 1977; Wallace et al., 1985).
Today's interest in lead's impact on the health and occupational fields can be said to date from the 1839 publication of Tanquerel des Planches,
in Paris, in which he described the clinical course of 1,207 persons with lead colic and the types of work that exposed them to lead. More than 800 of the cases were in painters or workers involved in the manufacture of white or red lead pigments.
Somewhat later, particularly during the industrialization of Europe, reproductive failures and congenital lead poisoning were described by Paul (1860). Workers recognized sterility, abortion, stillbirth, and premature delivery as common, not only among female lead workers, but also among the wives of men who worked in the lead trades (Oliver, 1911; Hamilton and Hardy, 1949; Lane, 1949). Indeed, those observations led a British Royal Commission in 1910 to recommend that women be excluded from the lead trades, a recommendation that was enforced in some countries by law (Lane, 1949). In the United States, occupational-hygiene actions toward protection of women from lead in the workplace have included the exclusion of women from lead exposure in an early stage of pregnancy (29 CFR 1910.1025) and even an effort to exclude all women of childbearing age. The U.S. Supreme Court has recently ruled that women of childbearing age cannot be excluded from workplaces with lead present.
Lead poisoning in children was first described by Gibson et al. in 1892 in Brisbane, Queensland, Australia. By 1904, Gibson, an ophthalmologist, had identified the source of lead and its probable route of entry into children, using both observation and experiment. He wrote:
I … advance a very strong plea for painted walls and railings as the source of the lead, and for the biting of finger nails and sucking of fingers, as in a majority of cases, the means of conveyance of lead to the patient.
In Europe, at about the same time, the general health hazards of lead in paint might have already been recognized. Awareness of the hazards was reflected in advertising of the period: Figure 1-61 depicts an advertisement

FIGURE 1-6 Advertisement for Aspinall's enamel, which appeared in the Diamond Jubilee issue of the Illustrated London News, 1897. Note that it is not made with lead, is not toxic, and represents 60 years of progress. The latter probably refers to the work of Tanquerel des Planches in Paris in the 1830s. He published his famous treatise, "Les Maladies de Plumbe" in 1839. Note that Aspinall's enamel could be purchased in 1897 in Paris, London, and New York.
appearing in England in 1897 emphasizing the nonpoisonous nature of the product, in contrast with toxic leaded paint.
Thomas and Blackfan in 1914 described the first American case of lead-paint poisoning (in a 5-year-old boy). They also offered the opinion that children must in some way be peculiarly susceptible to lead. On the occasion of the centennial of the Royal Children's Hospital in Brisbane in 1978, Fison (1978) cited Gibson's work and noted that at around the turn of the century physicians in southern Australia had been skeptical, because the condition seemed to stop abruptly at the Queensland border, and regarded the condition as a "delusion held by their despised colleagues in the primitive northern state." However, in the warm humid climate, paints weather quickly, and children would soon have their hands coated with powdery leaded material, which was inevitably carried to their mouths and digestive tracts.
After the report of Thomas and Blackfan in 1914, sporadic case reports appeared, and McKhann, in 1926, published a series of 17 cases of lead poisoning in children. That was followed by a classic report of lead poisoning due to the burning of battery casings in Baltimore homes in 1931 and 1932 (Williams et al., 1933). More case studies began to appear in the 1950s, as the condition became more widely known in larger cities in the United States, including New York, Chicago, Philadelphia, Boston, Cincinnati, St. Louis, and Cleveland. By 1970, the epidemiology of childhood lead poisoning was well established. Lin-Fu (1982) has summarized the history of childhood lead poisoning in the United States.
History of U.S. Childhood Lead-Screening Programs
Although several cases of childhood lead poisoning in the United States were reported in the first half of this century, little effort was made to understand the extent of poisoning in children until the 1950s, when caseworkers in a few large cities attempted to identify poisoned children as part of their family nutrition work. Limited results were obtained. In 1966, Chicago was the site of the first mass screening program where many poisoned children were identified; it was followed shortly by New York City and other large cities (Lin-Fu, 1980), where
similar results were obtained. In 1971, the Surgeon General issued a statement that emphasized the need to shift the focus from identifying poisoned children to prevention.
The 1971 Lead-Based Paint Poisoning Prevention Act led to the Categorical Grants Program to help communities carry out screening and treatment programs. The act initiated a national effort to identify children with high blood lead concentrations and to attempt abatement of their environmental sources of lead. Funds appropriated under the act were first administered by the Bureau of Community Environmental Management of the Department of Health, Education, and Welfare and later by the Centers for Disease Control and Prevention. Annual expenditures under the act rose from $6.5 million in fiscal year 1972 to $11.25 million in fiscal year 1980. The money supported up to 62 screening programs in 25 states (NCEMCH, 1989).
While CDC conducted the categorical program, criteria for identifying lead toxicity underwent a number of changes. A confirmed blood lead concentration of 40 µg/dL had been used to "define" lead toxicity. The 1975 and the 1978 revisions of the earlier CDC guidelines (CDC, 1975, 1978) used 30 µg/dL or above and different definitions of high erythrocyte protoporphyrin (EP) concentrations to produce several risk categories. Over 2.7 million children were screened from July 1, 1972, to June 30, 1979; 183,452, or 7%, tested positive by the prevailing criteria. In 1981, over 500,000 children were screened; in 18,000, the results were "defined" as lead poisoning (CDC, 1982).
In 1981, the Maternal and Child Health Services Block Grant Act and the Omnibus Budget Reconciliation Act transferred the national administrative responsibility for childhood lead-poisoning prevention programs to the Division of Maternal and Child Health of the Bureau of Health Care Delivery and Assistance. Under the provisions of the block grant act, each state decides whether to use federal funds to support childhood lead-poisoning prevention efforts (NCEMCH, 1989). In the transition from the categorical to the block grant programs, the screening-data reporting requirement was eliminated. Furthermore, federal policy from the U.S. Office of Management and Budget discouraged continued reports of screening from existing programs.
In early 1985, CDC reduced its criterion of childhood lead poisoning (CDC, 1985). A blood lead concentration at or above 25 µg/dL in tandem with an EP concentration 35 µg/dL or above was now considered
evidence of potential lead toxicity. The 1985 statement made clear that adverse effects were recognized as occurring blood lead concentrations below 25 µg/dL (so the chosen criteria represented the best compromise between health protection and practical matters related to limitations in screening methods at that time). The 1985 recommendations by CDC have now been re-evaluated, and an updated statement on lead-poisoning prevention was recently issued (CDC, 1991), which defines a lead concentration in whole blood at 10 µg/dL or greater as the action level, i.e., level of intervention.
The Lead Contamination Control Act of 1988 authorized, over 3 years, $20, $22, and $24 million per year for CDC to administer a childhood lead-poisoning prevention program. The money was to be given to state and local agencies to perform childhood lead screening for medical and environmental followup and education about lead poisoning. The act specifically stated that the money was not to supplant other funding for childhood lead-poisoning prevention. Although no money was appropriated for fiscal 1989, $4 million was appropriated for fiscal 1990, about $8 million for fiscal 1991, and $21.3 million for fiscal 1992. At present, national systematic collection of screening results does not exist.
The most recent national projection data on lead on children are available in the 1988 ATSDR report. The authors estimated that 250,000 children under the age of 6 had blood lead concentrations above 25 µg/dL in 1984. That estimate was based on data from NHANES II, census data, and projected environmental quantities of lead (ATSDR, 1988).
Sensitive Populations
People are not equally vulnerable to the effects of environmental exposure to lead. That fact yields the idea of sensitive populations that should be protected by monitoring programs from the adverse health effects of lead exposure. The focus of this report is infants, children, and pregnant women (as surrogates for fetuses). These populations are the most sensitive to lead exposure and are defined by the committee as the sensitive populations for this report. Some vulnerability is intrinsic, such as the age-dependent vulnerability in developing organ systems, and
some is extrinsic, such as that related to type or location of childhood exposure.
The magnitude of external exposure to lead varies among populations and influences both the severity of effects and the incidence of effects within a defined population. Besides the sensitive populations described in this report, other populations such as lead-industry workers may be at increased risk of adverse health effects because of large exposures. Nonetheless, the consideration of occupational exposures except where it may affect these sensitive populations is beyond the scope of this report. The time and magnitude of exposure may also influence the nature of lead intoxication. In addition, the constellation of organ systems affected can vary with age, with persistence of lead exposure, and with nutritional status, all of which are summarized in this report.
SCOPE AND ORGANIZATION OF THE COMMITTEE REPORT
The text and organization of the committee's report constitute a direct response to its charge, i.e., an assessment of appropriate, accurate, and precise methods for determining lead exposure in sensitive populations. The magnitude of human lead exposure deemed to be safe or without health effects continues to be reduced, to reflect improved identification of subtle toxic effects in sensitive populations. The committee considered measurement methods that could be useful in a world in which there might be no apparent threshold for particular neurobehavioral and other adverse health effects in vulnerable segments of the human population. The committee has evaluated exposure-measurement methods in the light of the existing broad range of population lead exposures.
This report has two main portions. One provides a summary of toxicity, public-health concerns and scientific context that helps to define the need for the committee's report. The second addresses the specifics of the committee's charge and some closely linked issues.
The background portion of this report consists of three chapters: this chapter has provided a perspective on key issues in the lead problem and a brief history of lead exposure assessment in the United States. Chapter 2 provides a summary of the toxicity of lead in sensitive populations. Chapter 3 deals with the nature and scope of source-specific environmental
lead exposure in sensitive populations and the relative contributions of specific sources to adverse health effects.
The second portion consists of Chapters 4–6. Chapter 4 deals with the biologic basis of the markers of early effects of lead exposure. It summarizes the markers of exposure, effect, and susceptibility and establishes the biologic basis for the methods evaluated in Chapter 5. Chapter 5 describes the current and developing methods that are most suitable for lead exposure monitoring at low concentration, i.e., current CDC guidelines—including their population monitoring advantages and disadvantages. The methods are primarily those for use in internal (i.e., biologic) monitoring of lead exposure. They are described in relation to sampling and transporting, instrumentation, quality assurance and quality control, and statistical methods. The chapter describes the public-health implications monitoring sensitive populations for lead exposure at low concentrations as recommended by CDC. Chapter 6 sets forth a comprehensive set of recommendations dealing with both the specifics and the generalities of the committee's charge.