D
Dietary Iron: Trends In The Iron Content Of Foods, Use Of Supplemental Iron, And The Framework For Regulation Of Iron In The Diet
Trends in the Iron Content of Foods and Consumption From Diet and the Use of Supplements
Dietary Consumption Patterns
The foods that contribute most to dietary iron are grain products and meat, poultry, and fish (LSRO, 1989). As shown in Figure D-l, the contribution from grain products has increased since estimates of specific food contributions to the food supply were first made in 1909. The reasons for the increases are enrichment of white flour with iron and other nutrients (since World War II) and the increased use of iron-fortified cereal products.
Data from several sources indicate that the levels of iron in the diets of infants and toddlers increased during the 1970s and 1980s. Although data from the Total Diet Study, which provided reporting on the chemical analyses of typical diets, showed no change from 1974 to 1982 (Table D-l) (Pennington et al., 1984), the average iron intakes from four national surveys conducted from 1971 to 1986 indicate increases for children 1-2 and 3-5 years of age (Table D-2) (LSRO, 1989).
For adult women, no change in the iron contents of typical diets (Table D-1) or intakes (Table D-2) during the 1970s and 1980s was observed.
Infant Cereals
Iron preparations that are soluble in water or in dilute acid (such as in the stomach) are generally of high bioavailability, whereas forms of iron that are insoluble in water or dilute acid solutions are of low bioavailability (Hurrell, 1992). Examples of iron salts that are freely soluble both in water and in dilute acid are ferrous sulfate, ferrous ascorbate, ferrous gluconate, and ferric ammonium citrate. Examples of salts that are poorly soluble in water but soluble in
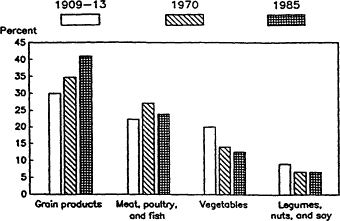
Figure D-1 Major food sources of iron ha fine food supply. Source: LSRO (1989).
dilute acids are ferrous fumarate, ferrous succinate, and ferric saccharate. Many of the iron preparations commonly used for food fortification are poorly soluble in water and poorly soluble in dilute acid (e.g., ferric pyrophosphate and ferric orthophosphate, which were used in the past, and elemental iron powders of intermediate particle size).
In the presence of oxygen, water-soluble forms of iron react with various components of food to produce oxidative rancidity. Such forms of iron can be used to fortify foods that can be packaged to limit prolonged contact with oxygen (e.g., infant formulas). Dry cereals packaged in cardboard boxes (including cereals specifically marketed for infants) permit exchange of air between package contents and the environment, and it is therefore not feasible to fortify them with ferrous sulfate or with most other iron salts known to have good bioavailability (Coccorilli et al., 1976; Hurrell, 1984). In the United States, most dry infant cereals are fortified with a metallic iron powder, specifically, electrolytic iron.
The bioavailabilities of metallic iron powders are closely related to particle size and solubility (Björn-Rasmussen et al., 1977). Metallic iron powders of extremely small particle size are readily soluble in dilute acid (Björn-Rasmussen et al., 1977) and have good bioavailability (Björn-Rasmussen et al., 1977; Rios et al., 1975). Unfortunately, the greater the solubility (and, presumably, the greater the bioavailability) of a metallic iron powder, the greater its chemical reactivity and the less suitable it is for food fortification. Circumstantial evidence has led several authors to conclude that electrolytic iron powder of the
TABLE D-1 Iron Levels in Diets of Adult Males, Infant, and Toddlers
TABLE D-2 Mean Iron Intake by Sex and Age in 1 day from NHANES I and II, NFCS, and CSFII
|
|
Iron Intake (mg)a |
|||||||
|
|
NHANES I 1971-1974 |
NFCS 1977-1978 |
NHANES II 1976-1980 |
CSFII 1985-1986 |
||||
|
Sex and Age (yr) |
Mean |
SEM |
Mean |
SEM |
Mean |
SEM |
Mean |
SEM |
|
Both sexes |
|
|
|
|
|
|
|
|
|
1-2 |
7.35 |
0.16 |
8.1 |
0.16 |
8.57 |
0.13 |
10.2 |
0.51 |
|
3-5 |
8.58 |
0.11 |
9.5 |
0.12 |
10.02 |
0.09 |
11.0 |
0.34 |
|
6-11 |
10.81 |
0.17 |
12.2 |
0.12 |
12.34 |
0.31 |
— |
— |
|
Male |
|
|
|
|
|
|
|
|
|
12-15 |
14.13 |
0.42 |
15.6 |
0.20 |
16.01 |
0.45 |
— |
— |
|
16-19 |
16.70 |
0.51 |
16.9 |
0.26 |
18.15 |
0.60 |
— |
— |
|
20-29b |
16.55 |
0.40 |
16.2 |
0.23 |
17.59 |
0.45 |
16.7 |
0.79 |
|
30-39b |
16.54 |
0.48 |
15.9 |
0.21 |
16.48 |
0.49 |
15.6 |
0.70 |
|
40-49b |
15.26 |
0.42 |
16.1 |
0.24 |
16.58 |
0.53 |
15.6 |
0.81 |
|
50-59 |
13.85 |
0.38 |
15.9 |
0.23 |
15.21 |
0.43 |
— |
— |
|
60-69 |
13.08 |
0.18 |
14.9 |
0.25 |
14.73 |
0.20 |
— |
— |
|
70+c |
11.68 |
0.16 |
14.2 |
0.28 |
13.24 |
0.29 |
— |
— |
|
Female |
|
|
|
|
|
|
|
|
|
12-15 |
10.44 |
0.28 |
11.9 |
0.21 |
10.71 |
0.32 |
— |
— |
|
16-19b |
54 |
0.30 |
11.2 |
0.20 |
10.04 |
0.34 |
— |
— |
|
20-29b |
10.06 |
0.13 |
10.7 |
0.17 |
10.67 |
0.23 |
11.1 |
0.27 |
|
30-39b |
10.36 |
0.14 |
11.1 |
0.15 |
11.08 |
0.31 |
11.1 |
0.26 |
|
40-49 |
10.40 |
0.18 |
11.0 |
0.14 |
11.10 |
0.34 |
10.6 |
0.22 |
|
50-59 |
10. 15 |
0.28 |
11.5 |
0.17 |
10.30 |
0.30 |
— |
— |
|
60-69 |
9.53 |
0.14 |
11.0 |
0.15 |
10.53 |
0.13 |
— |
— |
|
70+c |
8.63 |
0.13 |
10.4 |
0.16 |
10.18 |
0.22 |
— |
— |
|
a SEM is standard error of the mean, NHANES I and II are the first and second national Health and Nutrition Examination Surveys, NFCS is the Nationwide Food Consumption Survey, and CSFII is the Continuing Survey of Food Intakes of Individuals. b Data from CSFII are for 1985 only. c Ages 70-74 years only for NHANES I and NHANES II. SOURCE: LSRO (1989). |
||||||||
particle size currently used to fortify infant cereals in the United States is of low bioavailability (Fomon, 1987; Hurrell, 1984; Hurrell et al., 1989). A similar conclusion was reached by Hallberg et al. (1986) for another metallic iron powder with similar solubility.
Infant cereals fortified with electrolytic iron powder are among the first breakfast items introduced into the infant's diet. A large study carried out in Chile (Walter et al., 1993) demonstrated that feeding of infant cereal fortified with electrolytic iron powder can exert a favorable effect on iron nutritional status; at an intake level of 26-30 g of iron per day, fortified cereal was slightly but not significantly less effective in preventing iron deficiency anemia than iron-fortified infant formula. Whether the much lower intakes of cereal likely to be fed to infants in the United States (mean consumption for 73 percent of 6- to 12-month-old infants was 19 g/day in one infant nutrition survey [Gerber Products Company, 1989]) would contribute substantially to meeting infants' needs for absorbed iron is unknown. Findings from a recent, as yet unpublished Canadian study indicate that fortified cereal is effective in meeting the iron needs of infants; intake of cereal was about 33 g/day for 6- to 12-month-old infants (Beaton et al., in press).
Potential Role Of Meat In Meeting Infants' Need For Absorbed Iron
The potential role of meat in the diets of older infants and preschool-age children is based on its contribution of heme iron and on the enhancing effect of meat on absorption of non-heme iron. Studies of iron absorption from meals indicate that adults with moderate iron stores (500 mg) generally absorb less than 5 percent of dietary non-heme iron and about 25 percent of dietary heme iron (Monsen et al., 1978). Although heme iron provides only 5 to 10 percent of the iron in the adult Western diet, it accounts for more than one-third of the absorbed iron (Cook, 1983).
The iron content of cooked beef generally ranges from 2.0 to 3.0 mg/100 g (Pennington, 1989). Assuming that heme iron makes up about 70 percent of the iron present in cooked beef (the heme content of raw beef has been reported by Hazell [1982] to be 78 percent of total iron and by Schricker and colleagues [1982] to be 62 percent of total iron; little heme iron is likely to be destroyed by cooking), 100 g of cooked beef provides about 1.75 mg of heme iron. Thus, with 25 percent absorption, consumption of 30 g of cooked beef per day by an infant or preschool-age child might result in absorption of 0.13 mg of heme iron per day (1.75 mg/100 g × 30 g × 0.25). In addition, the presence of meat in the meal would enhance absorption of non-heme iron from meat and other foods in the meal. The effect of animal tissue protein on absorption of non-heme iron is dose related, with 1 g of meat exerting about the same enhancing effect as 1 mg of ascorbic acid (Cook, 1983). Therefore, an intake of 30 g of meat would be expected to exert a major effect on total iron absorption.
Use of Supplemental Iron Products
Supplement use has been assessed by three national surveys conducted between 1971 and 1986. The first National Health and Nutrition Examination Survey (NHANES I), conducted from 1971 to 1974, found that 23 percent of U.S. adults took supplements daily (Block et al., 1988). A special survey conducted by the U.S. Food and Drug Administration (FDA) in 1980 found that about 40 percent of adults reported using a vitamin or mineral product regularly (Stewart et al., 1985). In the 1986 National Health Interview Survey, investigators conducted detailed interviews and found that 36 percent of adults took a vitamin-mineral supplement (Moss et al., 1989). The survey found that 24.5 percent of women 18-44 years of age reported using a supplement containing iron, and the median average daily intake of iron was 100 percent of the Recommended Dietary Allowance (RDA). The estimated 90th percentile of intake from supplements was 247 percent of the RDA, and the 95th percentile of intake was 345 percent of the RDA. In further analysis of this survey, Looker and colleagues (1990) reported that iron supplement use among women 18 to 44 years of age was highest for whites (26 percent) and lower for blacks (15 percent) and Hispanics (16 percent).
Regulatory Framework For Iron Enrichment And Fortification
Iron in various forms is used for both enrichment and fortification of food products. FDA currently allows added iron compounds to be used as a nutrient or flavoring in food products. The FDA currently certifies 16 types of iron for use as a nutrient or flavor (Table D-3).
Current food industry practice for iron fortification and enrichment of food varies. By regulation (21 CFR §107.100), iron-fortified infant formula must contain between 0.15 and 3.0 mg of iron per 100 kcal of formula. Infant cereals and foods generally use electrolytic iron (dry cereals) and ferrous surf ate (jarred, ready-to-eat [RTE] infant cereals) and provide approximately 45 percent of the U.S. Recommended Daily Allowance (U.S. RDA) for infants per serving.
RTE breakfast cereals are generally fortified with reduced iron, the particle size of which is controlled. Many products also contain supplemental ascorbic acid (vitamin C). RTE breakfast cereals generally supply 10 to 100 percent of the U.S. RDA for iron per serving, but most, on the average, contain between 25 and 45 percent of the U.S. RDA for iron per serving. Forty-five percent of the U.S. RDA is the minimum amount of iron per serving required for an RTE breakfast cereal to be approved for use with the Special Supplemental Food Program for Women, Infants, and Children (WIC).
TABLE D-3 Iron Compounds Certified for Use in Foods
|
Compound |
21 CFR §a |
Useb |
|
Ferric: |
|
|
|
Ammonium citrate |
184.1296 |
N |
|
Chloride |
184.1297 |
F, other |
|
Citrate |
184.1298 |
N |
|
Phosphate |
182.5301/184.1301 |
N |
|
Pyrophosphate |
182.5304/184.1304 |
N |
|
Sulfate |
184.1307 |
F |
|
Sodium pyrophosphate |
182.5306 |
DS |
|
Ferrous: |
|
|
|
Ascorbate |
184.1307a |
N |
|
Carbonate |
184.1307b |
N |
|
Citrate |
184.1307c |
N |
|
Fumarate |
184.1307d |
N |
|
Gluconate |
182.5308/184.1308 |
N |
|
Lactate |
182.5311/184.1311 |
N |
|
Sulfate |
182.5315/184.1315 |
N |
|
Reduced iron (elemental iron; electrolytic or carbonyl) |
182.5375/184.1375 |
N |
|
Iron-choline citrate complex |
172.370 |
N, DS |
|
a Citations from Section 182 are generally recognized as safe (GRAS) for use in dietary supplements (pills); citations from Section 184 are GRAS and are affirmed for use in foods. b DS, dietary supplement; F, flavor; N, nutrient. SOURCE: 21 CFR (1992). (Additional information was obtained from personal communication with George Pauli, Center for Food Safety and Applied Nutrition, FDA, May 1993.) |
||
Food companies began to enrich milled grain products in the late 1940s. Enrichment of flour and other grain products with iron (and thiamin, riboflavin, and niacin) began as a mechanism to reduce disease and conditions of nutrient deficiency. Over the years, this public intervention has been remarkably effective and efficient for enhancing the nutrient quality of the food supply. The most common types of iron used to fortify flour and other grain products are hydrogen-reduced elemental iron (cereals, rice, flours) and ferrous sulfate (pasta).
At present, a significant amount of research is being conducted to evaluate ways to increase iron intake. Many new iron compounds for use in fortification and enrichment are being evaluated. The problem is that the bioavailability of iron compounds is generally inversely related to their reactivities in foods. Thus, the most readily available forms tend to discolor food and catalyze fat oxidation. Other approaches involve the addition of substances to foods that increase iron absorption from the gut (i.e., ascorbic acid or meat). More research is being conducted on the precise component of meat that influences iron absorption. In projects in developing countries, iron-EDTA is being evalu-
ated as a vehicle for delivering iron. In Sweden, ferrous ammonium phosphate is being studied for use in foods.
New food labeling regulations required by the Nutrition Labeling and Education Act of 1990 (P.L. 101-535) have implications for iron-enriched and fortified food products. The law and implementing regulations change both the legal status and the expression of iron on the food label. Past regulations required a listing of iron content only if it was added to a food or if a claim was made about its content. New serving sizes for RTE breakfast cereals may require additional consumer education to inform consumers about the amount of iron in a serving and may force some product reformulation for the cereals to continue to be certified for use with the WIC program.
References
Beaton, G.H, P. Tanaka, S. Zlotkin, G.H. Anderson, I.A. Menon, and D.L. Young. In press. Efficacy of Iron Fortification of Infant Cereals. Final report to the National Health Research Development Program, Canada, Toronto, Ontario, Canada.
Björn-Rasmussen, E., L. Hallberg, and L. Rossander. 1977. Absorption of ''fortification'' iron: Bioavailability in man of different samples of reduced Fe, and prediction of the effects of Fe fortification. Br. J. Nutr. 37:175-188.
Block, G., C. Cox, J. Madans, G.B. Schreiber, L. Licitra, and N. Melia. 1988. Vitamin supplement use by demographic characteristics. Am. J. Epidemiol. 127:297-309.
Coccodrilli, G.D., Jr., G.H. Reussner, and R. Thiessen, Jr. 1976. Relative biological value of iron supplements in processed food products. J. Agric. Food Chem. 24:351-353.
Cook, J.D. 1983. Determinants of non-heme iron absorption in man. Food Technol. (October):124-126.
Fomon, S.J. 1987. Bioavailability of supplemental iron in commercially prepared dry infant cereals. J. Pediatr. 110:660-661.
Gerber Products Company. 1989. Infant Nutrition Survey. Fremont, Mich.: Gerber Products Company.
Hallberg, L., M. Brune, and L. Rossander. 1986. Low bioavailability of carbonyl iron in man: Studies on iron fortification of wheat flour. Am J. Clin. Nutr. 43:59-67.
Hazell, T. 1982. Iron and zinc compounds in the muscle meats of beef, lamb, pork and chicken. J. Sci. Food Agric. 33:1049-1056.
Hurrell, R.F. 1984. Bioavailability of different iron compounds used to fortify formulas and cereals: Technological problems. Pp. 147-148 in Iron Nutrition in Infancy and Childhood, A. Stekel, ed. New York: Raven Press.
Hurrell, R.F. 1992. Prospects for improving the iron fortification of foods. Pp. 193-208 in Nutritional Anemias, S.J. Fomon and S.H. Zlotkin, eds. New York: Raven Press.
Hurrell, R.F., D.E. Furniss, J. Burr, P. Whittaker, S.R. Lynch, and J.D. Cook. 1989. Iron fortification in infant cereals: A proposal for the use of ferrous fumarate or ferrous succinate. Am. J. Clin. Nutr. 49:1274-1282.
Looker, A.C., C.M. Loria, M.A. McDowell, and C.L. Johnson. 1990. Dietary habits of blacks and other ethnic minorities in the U.S. with special references to iron status. Pp. 15-23 in Functional Significance of Iron Deficiency, C.O. Enwonwu, ed. Nashville: Meharry Medical College.
LSRO (Life Sciences Research Office). 1989. Nutrition Monitoring in the United States—An Update Report on Nutrition Monitoring. DHHS Publication No. (PHS) 89-1255. Public Health Service. Washington, D.C.: U.S. Government Printing Office.
Monsen, E.R., L. Hallberg, M. Layrisse, D.M. Hegsted, J.D. Cook, W. Mertz, and C.A. Finch. 1978. Estimation of available dietary iron. Am. J. Clin. Nutr. 31:134-141.
Moss, A.J., A.S. Levy, I. Kim, and Y.K. Park. 1989. Use of vitamin and mineral supplements in the United States: current users, types of products, and nutrients. Advance Data from Vital and Health Statistics. 174.
Pennington, J.A.T. 1989. Bowes and Church's Food Values of Portions Commonly Used, 15th ed. New York: Harper & Row.
Pennington, J.A.T., D.C. Wilson, R.F. Newell, et al. 1984. Selected minerals in foods surveys, 1974 to 1981/82. J. Am. Diet. Assoc. 84:771-782.
Rios, E., R.E. Hunter, J.D. Cook, N.J. Smith, and C.A. Finch. 1975. The absorption of iron as supplements in infant cereal and infant formula. Pediatrics 55:686-693.
Schricker, B.R., D.D. Miller, and J.R. Stouffer. 1982. Measurement and content of non-heme and total iron in muscle. J. Food Sci. 47:740-743.
Stewart, M.L., J.T. McDonald, A.S. Levy, R.E. Schucker, and D.P. Henderson. 1985. Vitamin and mineral supplement use: A telephone survey of adults in the United States. J. Am. Diet. Assoc. 85:1585-1590.
Walter, T., P.R. Dallman, F. Pizarro, L. Veloso, G. Peña, S. Bartholmey, E. Hertrampf, M. Olivares, A. Letelier, and M. Arredondo. 1993. Effectiveness of iron-fortified infant cereal in prevention of iron deficiency anemia. Pediatrics 91:976-982.
| This page in the original is blank. |










