6
Interactions Among Stressors and Challenges to Understanding Their Cumulative Effects
INTRODUCTION
The assessment of aggregate and cumulative effects from stressors (anthropogenic or natural) on any particular species or stock of marine mammal involves two fundamental elements: conceptualizing the process by which the potential stressors might influence the mammal population, and designing and implementing approaches to test specific hypotheses for relationships among stressors and demographic responses. Both of these needs present particular challenges in the case of marine mammals. Chapter 6 explores these challenges in further detail.
CONCEPTUALIZING PROCESS
Understanding the impacts of a potential stressor on any species in nature is always best served by first establishing a conceptual model that defines the pathways and processes by which that impact might occur. This general approach further involves defining the relationship between dosage of the stressor and response of the individual marine mammal, the population, or the associated ecosystem. Multiple potential stressors add to the challenge of understanding impacts. One commonly used approach to this difficulty that has been used in biomedical research involves estimating whether the impacts of two or more stressors occur via common pathways. Sharing common modes of action is thought to increase the likelihood of interaction (see Table 4.1). However, demonstrating or even predicting how the diverse set of stressors considered in this report may interact to influence marine mammals will be no mean feat. In this chapter the problem is treated in a manner that is broadly conceptual. The discussion begins by introducing the “interaction web” as a way of envisioning how the distribution and abundance of marine mammals will be influenced by stressors of any sort. Next is a discussion of functional relationships between stressor level and marine mammal response. In the third short section of this chapter, “ecological surprises” are introduced and discussed as the likely manifestation of what science does not yet understand about the way interaction webs are assembled and how they function. The section on ecological surprises is followed by an exploration of how the understanding of stressor–response relationships for marine mammals might be improved through a discussion of the principles of experimental design and scientific inference. The chapter concludes with a section on adaptive management: how best to use the insights derived from the various studies of marine mammals, stressors, and responses for their conservation and management.
THE INTERACTION WEB
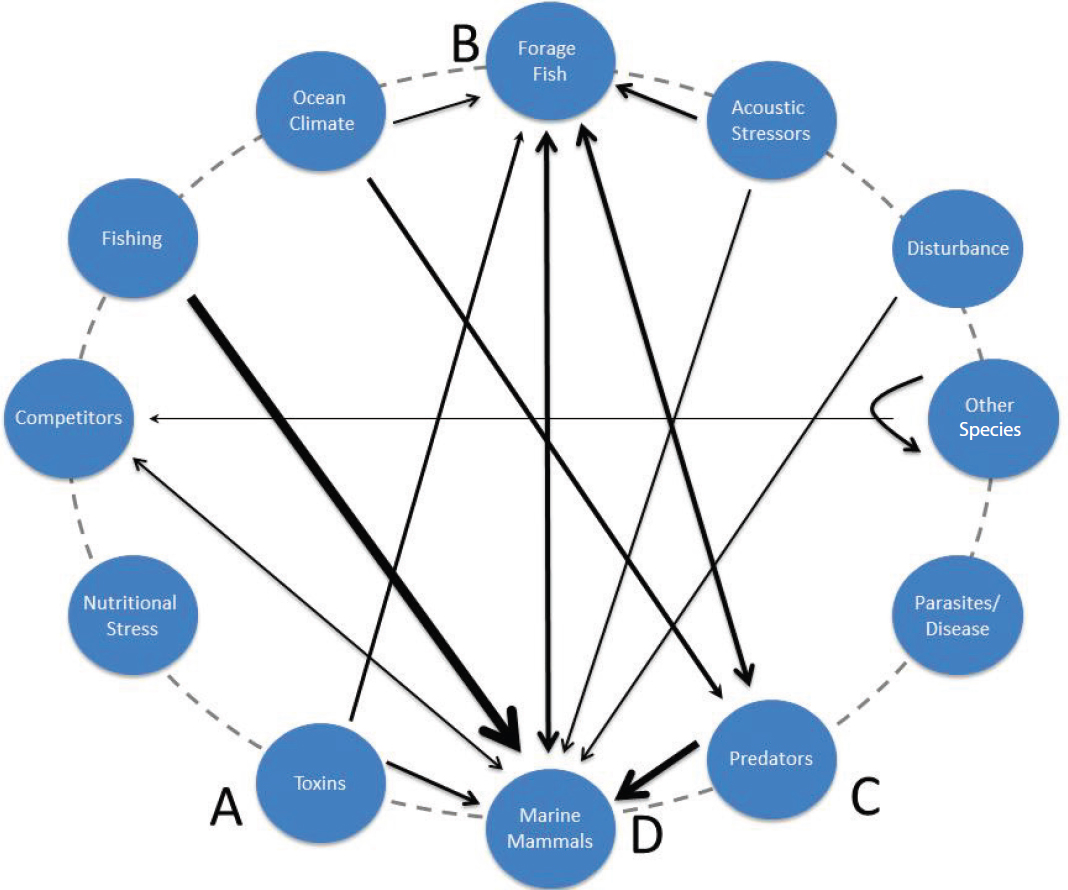
Although various approaches have been taken to define the network of interactions among species and between species and their abiotic environments, in this report the idea of an interaction web, as defined by Dunne et al. (2002) is used. The older, more well-known, and more widely used notion of a food web (the network of trophic interactions among species [Pimm, 1979]) is embedded in the interaction web concept. The conception of the interaction web is based on a single broad premise—that the distribution and abundance of species in any ecosystem is dictated by interactions among species and between these species and their abiotic environment. In the case of food webs, abiotic factors are not considered, and species interactions are restricted to those involving consumers and their prey. The interaction web broadens the concept of interactions to include abiotic and biotic ecological drivers that have effects on populations that are broadly similar to the effects of stressors on individuals. Stated in the specific context of this report, a stressor stimulates the
physiological response in an individual, and an ecological driver is a species or abiotic element of the environment that has an influence on a population. The key feature of ecological drivers is that they are biotic or abiotic features of the environment that affect individual animals indirectly by changing exposure to a whole suite of extrinsic stressors.
Interaction webs can be characterized in various ways. In this report it is done visually—as an oval with species and abiotic environmental elements arrayed around the perimeter (referred to subsequently as nodes) and direct interactions among species and/or elements of the abiotic environment (referred to subsequently as linkages) as the interconnecting lines (see Figure 6.1). The distribution and abundance of species in nature are largely dictated by these linkages, which are further defined by three properties: directionality, sign, and strength. For any two nodes A and B, A may influence B while B has little or no influence on A (in which case A is said to be the driver and B is said to be the recipient); or two nodes B and D may influence one another (in which case both B and D are drivers and recipients). Interactive effects might be positive (e.g., the influence of a prey species on its consumer) or negative (e.g., the influence of consumer on its prey). Anthropogenic stressors may be negative drivers, in the sense that at the levels occurring in nature they exert a
negative influence on the distribution and/or abundance of a marine mammal species, population, or stock. In this context it is important to recognize that stressors at the individual level may have little or no influence, or in some cases even a positive influence, on the species or stock of interest. Interaction strength, defined as the magnitude of the direct effect of one node on another node, is visually characterized by line weight (see Figure 6.1).
Interaction web nodes can also affect one another via one or more intervening nodes, in which case their interplay is defined as an indirect effect. For example, node A might affect node D both directly and even more strongly through an indirect effect on node D via node B. Indirect effects are often imagined to be weaker than direct effects because the likelihood of a weak link occurring in the interaction chain increases with chain length, and the strength of any indirect effects will be limited by the weakest link in the chain. However, indirect effects can be as strong as or stronger than direct effects, and, in all but the simplest ecosystems, the number of potential indirect effects greatly exceeds the number of potential direct effects (Estes et al., 2013a). The net effects of anthropogenic drivers on marine mammal populations might thus be composed of either direct or indirect effects, or, most likely, both types of effects.
Interaction webs, by their fundamental nature, are exceedingly complex. Endeavors to quantify or otherwise analyze interaction web behavior have employed two broadly similar approaches, use of the community matrix (May, 1972; Yodzis, 1988) and network analysis (Proulx et al., 2005). Although these general methods of analysis will not be discussed further in this report, they may be used for further understanding the influence of anthropogenic stressors on marine mammals and their associated ecosystems.
Finding 6.1: Interaction webs characterize the numerous pathways in which all species within an ecosystem interact with one another and the various elements of their physical environment. This approach can be used to conceptualize the myriad ways extrinsic stressors may influence marine mammals.
Finding 6.2: Any two species may link together in the interaction web via direct or indirect interactions. Direct interactions are those in which there are no intervening species, whereas indirect interactions are those in which there are one or more intervening species. Indirect effects can link species with stressors via long interaction chains that may involve both bottom-up and top-down forcing processes.
RELATIONSHIP BETWEEN STRESSOR LEVEL AND INTERACTION WEB RESPONSE
The effects of a stressor on a population or ecosystem depend on the functional relationship between stressor level and an individual’s response through changes in vital rates, the proportion of the population that is exposed to the stress- or, and, for those exposed individuals, the level of exposure that each individual experiences.
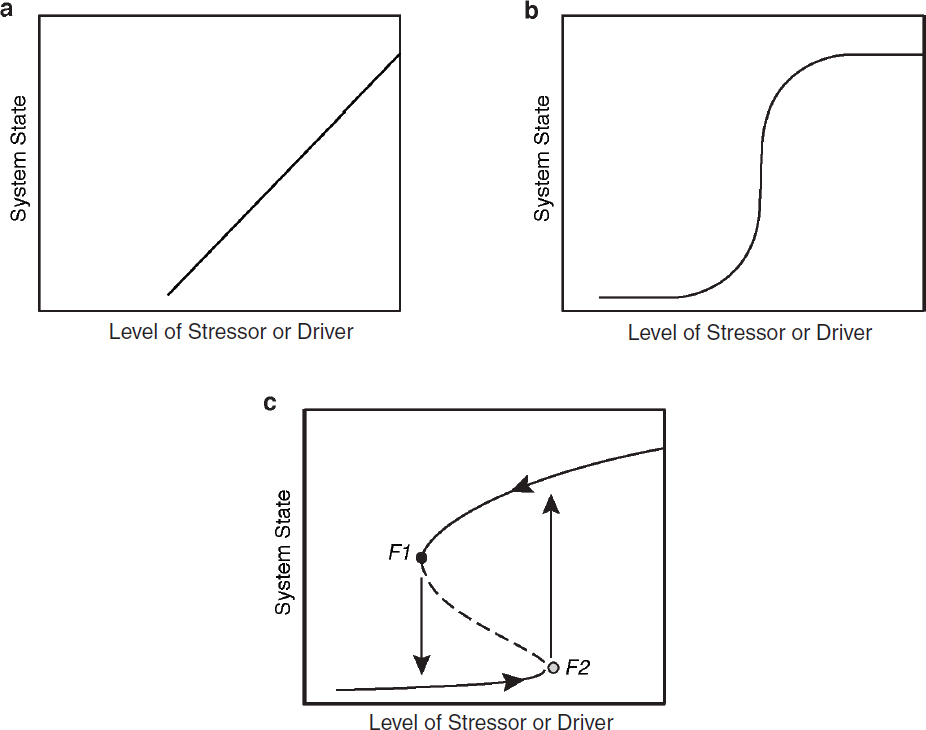
A critical question here is: How sensitive are the predictions of population- and ecosystem-level effects from stressors to the form of the mathematical function that describes these relationships? If for example this function is linear (see Figure 6.2a), then some change in stressor level is predicted to lead to a constant proportional change in the system in which it acts, whatever the specific value of the stressor. Using this simple function, the magnitude of stressor impact can be estimated from the slope of the stressor–response function and the magnitude of change in the stressor, and even very low doses will have some effect. If the stressor has a point source, large numbers of individuals may be exposed to these very low stressor levels (see Box 2.2), and this could have important population-level effects. If, however, a sigmoidal function of the form shown in Figure 6.2b is assumed, very low doses are predicted to have little or no effect, and the population-level effects associated with the linear function would be ignored. In contrast, if the true function is in fact sigmoidal but linearity is assumed, unanticipated strong effects from small increases in stressor level may occur.
There are many reasons why a nonlinear function is more likely to be appropriate. Some of the more obvious reasons at both the individual and population levels are summarized below:
- For toxicants whose effect depends on binding with a receptor, the well-developed theoretical understanding of receptor-ligand kinetics predicts a nonlinear function.
- The physiological mechanisms that animals use to maintain homeostasis in the face of stressors often mean that adverse effects may not be visible until these systems break down, after which an adverse effect can suddenly appear. This nonlinear pattern can lead to sharp thresholds for effects.
- Any pattern of threshold variation (i.e., any particular density function) among individuals in response to a stressor within a population is likely to lead to a nonlinear cumulative distribution function.
- For a noise effects example, animals are not expected to respond to sounds at levels below their hearing threshold, and responsiveness may not increase above a certain high intensity of sound.
The preceding discussion is not meant to imply that these functional relationships must be understood before stressor effects can be documented. Such functional relationships will likely remain unknown in many cases. Even under this more limiting circumstance, stressor impacts might still be detected.
As explained further in Chapter 5, the Population Consequences of Acoustic Disturbance model (NRC, 2005) aimed
to break the causal chain from exposure to the stressor of noise to population effects into a series of sequential functional relationships. Chapter 5 describes recent applications of this model that use measures of body condition to integrate effects of stressors, from which the influences on reproduction and survival are predicted. There is evidence for nonlinear relationships between body condition, which integrates effects of many stressors, and reproduction, and this in turn varies among marine mammal species. Analysis of data from several species of pinnipeds showed that maternal state variables explained twice the variation in natality rates in capital breeders compared with income breeders (55% compared to 25%) and that the relationships between maternal state variables and pregnancy were distinctly nonlinear in capital breeders (Boyd, 2000). Thus, even if disturbance of feeding had a linear effect on body condition, the combined effect of disturbance on condition and then condition on pregnancy would be nonlinear, and the form of this function would likely vary between capital and income breeders.
Hunsicker et al. (2016) reviewed 736 relationships between driver levels and ecosystem responses in marine pelagic ecosystems. They report that nonlinear responses are more common than linear ones. Strongly nonlinear relationships were particularly common among climate and trophodynamic variables but also were associated with anthropogenic drivers, such as overfishing and pollution. The results of their meta-analysis of ecological studies led Hunsicker et al. (2016) to suggest that “in the absence of evidence for a linear relationship, it is safer to assume a relationship is non-linear.”
The shape of the functional relationship between a stressor or driver and its effect on an individual, population, or ecosystem has significant implications for management. If managers can assume that gradual changes in intensity of the stressor or driver lead to roughly linear changes in recipients, as in Figure 6.2a, then they can aim to monitor the effects over time to make sure these effects are not becoming adverse. If the slope of this linear relationship is known at low driver levels, this relationship can be extrapolated to predict effects at higher driver levels. By contrast, if the functional relationship is as in Figures 6.2b and 6.2c, then no
effect may be seen over a considerable range of driver levels, but beyond this range effects may escalate rapidly with only a small increase in the driver. Functional relationships of this nature lead to what are called phase shifts or regime shifts (Conversi et al., 2015), defined as abrupt and sometimes catastrophic responses by a system to small changes in driver intensity. The net effects of anthropogenic stressors on marine mammal populations and their associated ecosystems might thus be small and imperceptible until some critical level is reached, at which point the effect is strong. Selkoe et al. (2015) argue that this situation is common enough that resource managers should, “[i]n the absence of evidence to the contrary, assume nonlinearity.”
In some situations, the functional relationship between the level of a stressor or driver and the state of a system may vary depending on the directionality of change in stressor or driver level (see Figure 6.2c). This phenomenon is called hysteresis. For example, an individual marine mammal that has been exposed to a sound may habituate or become sensitized, changing its responsiveness to later exposures. Similarly, the initial response of an individual to increasing numbers of a pathogen following infection will differ from the response as the body reduces the number of pathogens. In this case, the state of the organism has changed from when the infection starts to when its immune system is causing the infection to decrease. At the population level, if abundance is reduced to a very low level by a driver, the population may not recover following driver relaxation because of such factors as demographic stochasticity or inverse density dependence (Allee effect; Stephens et al., 1999). For populations governed by the generalized logistic growth equation, the rate of decline following overshoot beyond carrying capacity will be more rapid than the rate of recovery from a similarly sized reduction in abundance below carrying capacity (Gotelli, 2008, p. 30). In multispecies systems (i.e., biological communities), a driver-induced reduction in one species might alter species interactions such that the driver relaxation is not followed by a similar pattern of recovery. A critical point about hysteresis for this report is that managers should not assume the response of a system will follow the same path when the level of a stressor is reduced as it did on increase of the stressor.
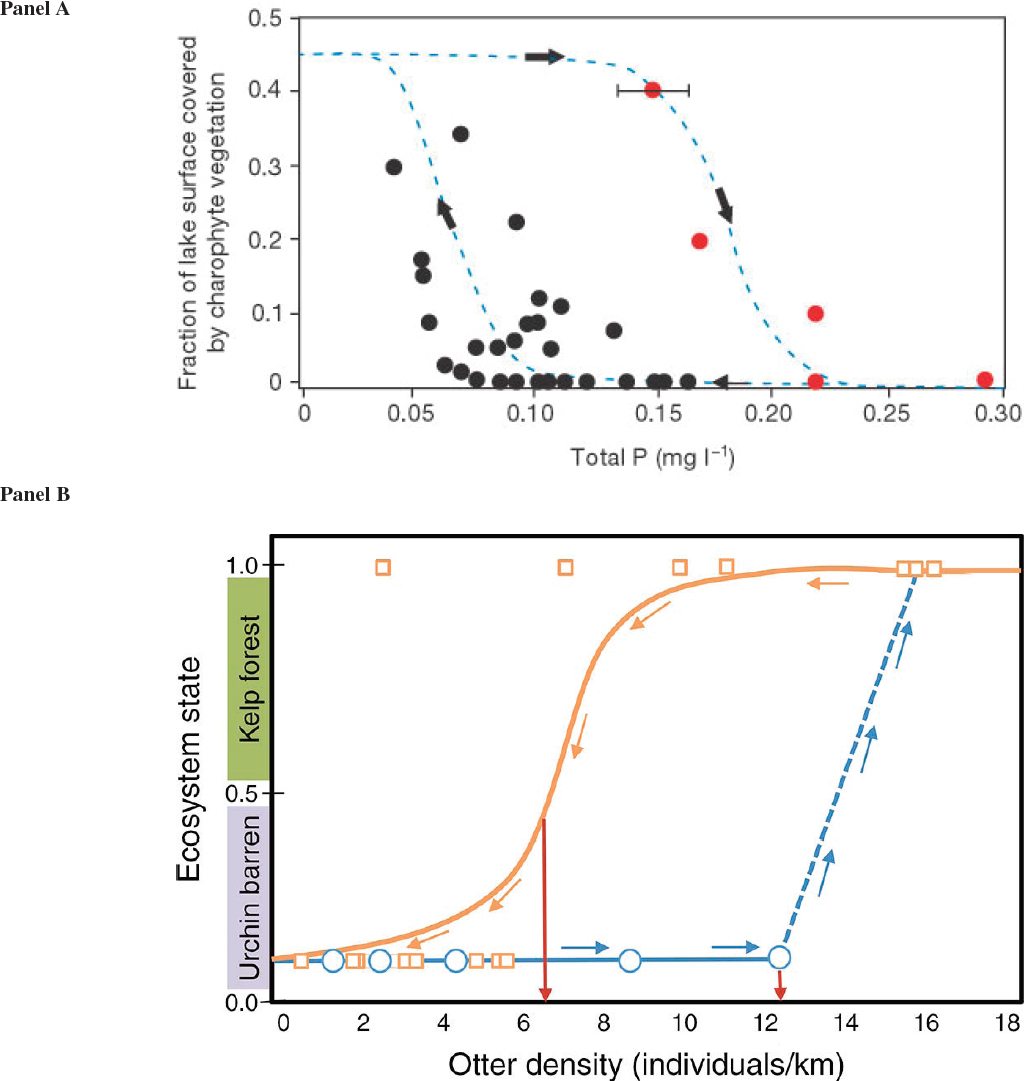
Ecosystems can shift among different basins of attraction (Scheffer et al., 2001)—different configurations to the distribution and abundance of species, in which movement from one basin to another requires a strong perturbation. This situation can be likened to the behavior of a ball over a three-dimensional surface of ridges and valleys, in which the valleys are basins of attraction and the ridges are tipping points (also known as breakpoints). Perturbations (changes in driver level) that are sufficient to push the ball over a ridge and into another valley result in regime shifts. The consequences of this process for the functional relationship between driver level and system state is illustrated graphically by Figure 6.2c. When driver level changes from just below F2 to just above F2 (a tipping point), the system jumps from one state to another (a regime shift). Once a regime shift has occurred, driver level must be reduced to below F1 for the system to return to the initial state. These breakpoints or tipping points can be thought of as unstable equilibria between alternative stable states (May, 1976). The first explorations of ecological tipping points and regime shifts were based on theoretical analyses (e.g., Lewontin, 1969; May, 1976). A large and growing body of empirical study confirms the existence of these state shifts and regime shifts in nature (Sutherland, 1974; Scheffer, 2009), including the shift from coral-dominated systems to macroalgae-dominated systems in the Caribbean (Hughes, 1994; Knowlton, 2004), changes in fishery yield (Steele, 2004; Vert-pre et al., 2013), shifts between kelp forests and sea urchin barrens (Steneck et al., 2002); and changes at larger system-wide scales (Beaugrand, 2004; Hare and Mantua, 2000; Möllman et al., 2009). Empirical evidence for hysteresis, although more limited, does exist (see Figure 6.3).
The general situation in which the state or condition of an individual, population, or ecosystem is largely unresponsive over one range of stressor or driver levels but responds strongly at other levels presents a substantial challenge to management. Under this circumstance, managers must know the range of stressor levels over which the desired state is maintained, thereby allowing them to set a threshold below which the risk of transition to the adverse state is suitably low. The actual forms of the functional relationship between stressor levels and their effects on marine mammal physiological systems, individual condition and life-history metrics, or the distribution and abundance of populations are largely undocumented. To the extent possible, the choice of such functional relationships should be based on data and/or theory, not on scientific preconceptions.
ECOLOGICAL SURPRISES
The preceding sections of this chapter establish two key points: (1) that interaction webs are highly complex structural entities, given the great diversity of species and the even greater diversity of ways these species can interact with one another and their physical environment, and (2) that functional relationships among species and between species and their physical environments are commonly nonlinear. Given these two key points, the responses of natural systems to stressors are expected to be difficult to predict and thus often characterized by what have been referred to as ecological surprises. In a paper based on analyses of various case studies and a survey of established field ecologists, Doak et al. (2008) concluded that major surprises (defined as “a substantial change in the abundance of one or more species resulting from a previously unknown or unanticipated process of any kind,” p. 593) should be expected in any effort to understand and predict ecological dynamics (Peetchey et
al., 2015). Key attributes of ecological surprises (Doak et al., 2008) include the following:
- Surprises are both dramatic and widespread in scientific studies of all kinds.
- Ecological surprises are especially common and underreported.
- Ninety percent of well-established field ecologists who responded to a questionnaire in which they were asked if they had ever been surprised (as defined above) answered in the affirmative.
- Eighty-eight percent of those who responded in the affirmative believed that they understood the reasons for having been surprised after the
-
-
fact, thus suggesting that the causes were easy to understand but previously unanticipated.
- Many of these examples remained unpublished because the individual investigators thought they were either uninteresting (scientifically) or unpublishable.
-
- Efforts to improve predictability and quantify uncertainty in ecological models are unlikely to reduce the frequency of ecological surprises because these modeling efforts necessarily are built around things that are known as opposed to things that are unknown.
- Sooner or later, most natural resource management strategies will not work as planned, thus reinforcing the need for management plans that are precautionary.
In keeping with this general view of nature, studies of marine mammals have resulted in numerous surprises. For example, while most populations and species of great whales recovered following protection from exploitation during the whaling era, some (like southern blue whales) have not recovered for reasons that remain unknown (Branch et al., 2007). In Chapter 4, several case studies of population decline were explored where it has been difficult to infer causes, including beluga whales in Cook Inlet, Alaska, pinnipeds and sea otters in the Northern Pacific and Southern Bering Sea, and harbor seals in the United Kingdom. Other examples of surprises involving marine mammals could be described and cited. However, the committee is not aware of any cases where these surprises were subsequently attributed to cumulative impacts or the interaction among multiple stressors. This does not imply that such cumulative or interactive effects are unimportant in causing ecological surprises, but rather that they are not well understood.
To reiterate, the basic reasons for these various surprises are (a) insufficient understanding of interaction web structure, especially with regard to the various important pathways that lead from potentially diverse drivers to marine mammals; (b) complex functional relationships in the interactions among species and between species and the abiotic environment; and (c) overly simplistic views of interaction web structure and process.
Finding 6.3: The functional relationships between interacting species are often nonlinear and characterized by hysteresis. These complex functional relationships, coupled with immensely complex interaction web topologies, often result in unanticipated outcomes, sometimes referred to as ecological surprises.
DESIGNING APPROACHES TO UNDERSTANDING STRESSOR IMPACTS AND THE PRINCIPLES OF SCIENTIFIC INFERENCE
Empirically based scientific inquiry in ecology involves two main elements: a search for pattern (which is commonly based on one’s view of interaction web structure and dynamics, as discussed above), and distinguishing between causation and correlation. Empirically based patterns nearly always derive from observation of variation in space or time. These two elements of scientific inquiry are in turn often challenged by two essential inadequacies: (1) inherent difficulties in observing patterns associated with purported or hypothesized causal agents (in the context of this report, stressors and drivers) and (2) the inability to distinguish between causation and correlation with a high level of confidence. These shortcomings are best overcome through the experimental method, wherein the influence of some purported causal agent or agents (e.g., anthropogenic stressors or drivers) is assessed by observing differences between experimental units (e.g., behavior or physiological parameters in the case of stressors; individuals or populations in the case of driver effects on the distribution and abundance of species) that have been treated with the purported causal agent (i.e., by adding or removing the imagined stressor or driver) and those that have not (controls).
The three basic principles of experimental design are randomization, replication, and local control, which exist because experimental units always contain some level of intrinsic variation, independent of that which might be caused by their experimental treatments. For example, no two individuals are exactly the same. One needs to be able to detect and measure experimental treatment effects through this intrinsic variation in experimental units. Randomization (the random matching of experimental treatments to experimental units) is done in order to ensure that intrinsic variation among the experimental units is as likely as possible to be spread evenly between treatments. Replication provides a measure of experimental error, defined as the difference among identically treated experimental units, and causes the average value of the intrinsic variation among identically treated experimental units to converge on zero with increased replicate number. Local control is accomplished by choosing and arranging the experimental units and then assigning treatments to these experimental units so as to reduce experimental error.
Scientific experiments that are conducted in accordance with these design principles have three important properties. First, they minimize the likelihood of mistaking correlation for causation. Second, they provide an inferential template for the assessment of multiple agents of causality and the interactions among these agents. Third, they often permit increased inferential efficiency through the processes of blocking, stratification, and the analysis of covariance, all
of which help reduce experimental error. These broad principles are discussed and explained in greater detail in any introductory text on experimental design (e.g., Fisher, 1937; Montgomery, 1997).
As observed in Chapter 4, the predominant approach to studying interactions between stressors uses experiments with a simple factorial design. Although this approach is both powerful and broadly applicable, it has drawbacks and limitations for answering the many questions about nature that scientists have been unable to address experimentally. This is the current state of affairs for the committee’s charge in this report, which is to evaluate the cumulative influences of anthropogenic stressors on marine mammals. As noted in Chapter 3, the lack of strong evidence for an influence of fisheries on marine mammals through competition for prey or other indirect interaction web effects is due to the failure to be able to assess these effects experimentally. Instead, the conclusions are more often based on observations of individuals and populations of marine mammals between otherwise similar areas with and without fisheries effects. Other approaches have been used in an effort to make these assessments (most commonly correlative analyses or inferences based on modeling approaches), but in many cases the signal is weak, and in most cases the distinction between causation and correlation is equivocal. For example, despite the great biomass of fish removed from the North Pacific Ocean/southern Bering Sea ground fisheries, it has proven both difficult and contentious to establish whether or not these potential prey removals have contributed to the declines of fur seals, harbor seals, Steller sea lions, and sea otters in southwest Alaska (NRC, 2003b). Moreover, pinniped populations in the northwestern Atlantic Ocean have generally increased, despite the collapsed ground fisheries (Estes et al., 2013b). Similar obstacles apply in the assessment of noise on marine mammals, although in this latter case experimental or quasi-experimental approaches are less problematic because noise is more manageably controlled than fisheries in space and time. However, the assessment of noise effects in combination with other potential stressors on marine mammals is exceedingly challenging because not only is it difficult or impossible to experimentally assess most singular (main) effects, doing so in sufficiently orthogonal combinations to be able to sort out the interactive effects is vastly more challenging. This is the fundamental nature of the problem at hand.
Understanding the influence of anthropogenic or natural stressors on marine mammals can only be rigorously assessed through observations of the manner in which individuals and populations respond to changed intensities of these stressors in their surrounding environments. Such information can be obtained in two general ways—through purposeful experimentation and through correlative studies from regions in which data from marine mammals are available in areas where the purported or hypothesized stressor has also varied. The strength of the experimental method is that, when properly done, the likelihood of misinterpreting results because of potentially confounding factors is eliminated or greatly diminished. As explained previously, the difficulty with experimental approaches for marine mammals is that they are difficult or even impossible to implement at appropriate scales of space and time for a host of fairly obvious reasons, including logistical limitations and legal, social, and economic constraints. Many of the experimental approaches that have been implemented lack sufficient samples to have the necessary statistical power or precision to detect effects. With proper planning, correlative studies are easier to conduct, but these are also usually plagued with uncertainties over whether the purported or hypothesized stressor is the cause of any marine mammal response in the face of other potential confounding variables. This fundamental limitation to correlative analyses will be greatly magnified in efforts to assess the potential influences of multiple stressors or the aggregate influences of single stressors on marine mammals.
The strength of inferences from nonexperimental information can often be improved through various analytical approaches. One of these is a weight-of-evidence analysis in which the array of relevant information is contrasted against the expectations of alternative competing hypotheses. Using this approach, it is sometimes possible to determine the most likely of two or more alternative hypotheses, or to exclude one or more of these hypotheses based on internal inconsistencies with available data. More recently, Sugihara et al. (2012) proposed a general method for distinguishing causality from correlation based on nonlinear state-space reconstruction of time-series data.
Finding 6.4: Controlled experiments are the most rigorous way of testing for the influences of potential stressors on any species. For marine mammals, such experimental approaches are often not possible, in which case inferences must be based on quasi-experiments. Although quasi-experimental data are subject to confounding and thus multiple interpretations, reasonably strong inferences are often possible from time-series analyses and weight-of-evidence approaches.
ADAPTIVE MANAGEMENT
As described above, classical factorial experiments are impractical as a vehicle for evaluating potential cumulative influences of stressors on marine mammal populations, while observational (correlative) studies are more practical to undertake but are likely to result in ambiguous inferences. Despite this, regulators must make decisions on whether and where to allow potentially harmful anthropogenic activities to take place. The concept of adaptive (resource) management offers a framework for making such decisions in the situation where there is some scientific understanding of the link between management action and outcome, and where repeated decisions must be made over time (such as issuing annual permits for activities, or setting harvest limits). Key
texts describing the concept include Walters (1986) and Williams (2011a, 2011b). A brief overview is provided here.
Adaptive management involves first setting a conservation objective and then formulating multiple hypotheses about the population response to the different management options, together with an assessment of the probability of each hypothesis being correct. The optimal decision is determined (see later for how “optimal” is defined), and this action taken. The population response is monitored, and the new information gained is used to update the probabilities for each hypothesis, whereupon the process is repeated. A key concept is that “we learn more about the system as we go along” and hence can adapt management decisions in the light of the improved information. There are broadly two approaches of adaptive management, depending on how “optimal” is defined: in passive adaptive management, the optimal decision is the one most likely to bring scientists closest to the conservation objective given the current state of knowledge; in active adaptive management, determining the optimal decision also involves accounting for the learning that is anticipated to occur as a result of each possible decision. (See Williams [2011b] for a more nuanced discussion of the various closely related definitions that have been used.) Hence, in active adaptive management, it is sometimes considered optimal to take management decisions that result in moving away from the conservation objective in the short term if this means one learns more about the biological system and so can make better conservation decisions in the future. Classical experiments may be contemplated, where different management actions are assigned at random to spatially replicated regions (if possible). Active adaptive management is therefore riskier, in that it relies more on having an accurate assessment of the consequences of selected actions (in terms of how much each possible action will help us distinguish between the multiple alternative hypotheses).
Although adaptive management ideas are much discussed, they are relatively little used in practice. A recent literature review by Westgate et al. (2013) identified 1,336 articles published between 1978 and 2011 using the term “adaptive management.” Of these only 61 (<5%) explicitly claimed to enact the methods, and only 13 projects were found that the review authors felt met the criteria for actually using adaptive management. There are multiple possible reasons for this lack of usage. First, the method requires the formulation of multiple competing hypotheses, typically expressed as alternative quantitative conceptual models of the system, and it may be that there is simply not enough knowledge about most systems to do this adequately. Second, the realistic rate of learning may be too slow to be useful. This may be because there is strong natural variability (e.g., from ecological drivers such as El Niño in the Pacific or the North Atlantic Oscillation) that nearly masks any signal coming from alternative management actions; because possible management options do not generate a strong signal (e.g., if they can only be applied to a small component of the population); because any signal may take a long time to be manifest (as will be the case for long-lived, slow-reproducing animals like most marine mammals); because standard experimental practices like replication and blocking are not possible; or because the monitoring of outcomes that are feasible is too imprecise to be useful. Third, although adaptive management is designed to cope with uncertainty about which hypothesis is correct, and with observation error in the outcome measurements (both “known unknowns” [Logan, 2009]), it is not robust to the kinds of ecological surprises that were discussed earlier in this chapter (the “unknown unknowns”); hence, focusing only on measuring the best metrics for distinguishing between alternative hypotheses risks missing other important conservation issues. The topic of monitoring is explored in the next chapter. Finally, implementing adaptive management is complex, typically requiring a team with skills in theoretical ecology, applied conservation, statistics and modeling, and, potentially, social sciences if the human aspect of management decisions is to be considered. Resources and commitment over the long term are required, and these are rarely available.
Despite these issues, there does not appear to be a superior alternative to adaptive management as a rational and structured system for making optimal conservation decisions. Trial and error, or “reactive management” (Sutherland, 2006), is clearly inferior. For this reason, the application of adaptive management principles to the management of cumulative effects is encouraged wherever this is possible.
Recommendation 6.1: Adaptive management should be used to identify which combinations of stressors pose risks to marine mammal populations, and to select which stressors to reduce once a risk is identified. In this approach, hypotheses are developed which guide management actions and data collection to assess the strength and impact of individual stressors and their cumulative effects.
CONCLUSIONS
In addition to direct mortality from entanglement in fishing gear, ship strikes, and purposeful killing, marine mammals are exposed to a broad range of potential anthropogenic stressors, including but not necessarily limited to noise, prey depletion by fisheries, disease, pollutants and toxins, and a broad (but still largely unknown) array of indirect effects of these various stressors on the associated ecosystems. In particular cases, each of these direct effects is known or suspected to have negative impacts on marine mammal individuals and populations. A separate literature from experimental studies (see Chapter 4) has demonstrated the cumulative or synergistic influences of stressors on a wide range of aquatic plant and animal species. Therefore, cumulative influences of anthropogenic stressors on marine mammals are nearly a certainty.
The challenge is in conceiving of and especially then
demonstrating these effects on marine mammals. The important outstanding questions are these: For which particular stressors under what specific conditions and for which marine mammal species will cumulative effects occur, and what are the functions that relate stressor dosage to the linked effect? Answering these questions in a scientifically rigorous manner is beset by three significant challenges. The first challenge is to properly characterize a topology of influence by stressors on marine mammals. Simple direct effects of singular stressors on marine mammals are relatively easy to imagine, but the potential influences of multiple stressors, acting through both direct and indirect interaction web pathways, will be substantially more difficult. The second key challenge will be in designing studies in which the interactive influences of multiple stressors on marine mammals can be evaluated. Experimental designs that are capable of demonstrating interactive effects while controlling for confounding influences are nearly impossible to carry out without purposely manipulating the purported drivers in an orthogonal manner. A final challenge is in the detection of any real impact from stressors on a marine mammal stock at the individual and especially the population level. Rigorous demonstration of population change has proven to be exceedingly difficult for most marine mammal species. Thus, even when the process by which multiple stressors might influence a marine mammal is well conceived and a study can be properly designed to put the resulting hypothesis to a test, the ability to document an effect on the marine mammal species, population, or stock of interest will often be limiting.










