7
Coordination of Research Priorities
Many organizations within the United States—public, private, governmental, nongovernmental, academic, industrial, and advocacy—are involved in conducting or funding medical research. Over the years the number of such organizations has grown as health conditions have captured the public’s attention or new health threats have emerged. However, the amount of money going to such research is finite, necessitating the judicious use of the available funds to support health research.
Coordinating research priorities and funding among federal and nongovernmental agencies, including international organizations, can serve several purposes in addressing this issue. First, it can help ensure that research dollars are allocated where they have the greatest potential to advance science, thus reducing research redundancies and overlaps, encouraging high-quality research, funding a broad range of researchers, and promoting responsible fiscal stewardship. Second, coordination can leverage dollars so that organizations with the most experience, expertise, or innovative approaches to a problem are most likely to receive funding. Third, it can help organizations and institutions develop much needed expertise by assisting them in focusing their efforts, rather than requiring them to be generalists, and training the next generation of researchers. And, finally, coordination among organizations can help them identify and implement best practices in all areas of medical research so that others might benefit from the lessons learned.
In this chapter, the committee responds to its congressionally man-
dated task to evaluate how the Department of Defense’s (DoD’s) Congressionally Directed Medical Research Programs (CDMRP) coordinates its research priorities with two federal organizations, the National Institutes of Health (NIH) and the Department of Veterans Affairs (VA), which are also leaders in the field of medical research for the health of service members, veterans, their families, and the general public. Medical research funding in the United States is briefly discussed in order to provide background to the committee’s subsequent consideration of how the major funding organizations establish their research priorities. The chapter then discusses how these and other organizations attempt to avoid funding duplicative or redundant research. The committee considers establishing research priorities and avoiding duplication to be critical aspects of research coordination.
MEDICAL RESEARCH FUNDING IN THE UNITED STATES
As briefly discussed in Chapter 1, medical research in the United States is funded by both the federal government, primarily NIH and DoD, and nongovernmental organizations. Within the federal government, funding allocations for medical research are divided among many departments and agencies. Federal agencies receive “core” funding for research as part of their annual budgets set by Congress and the president (known as the “President’s budget”). Additionally, some research funds are also allocated by congressional appropriations committees separately from core funding.
In 2016, the NIH budget was $31.3 billion (NIH, 2016a), the 2016 budget for the DoD Defense Health Agency was more than $980 million (DoD, 2016), and the 2016 VA budget for medical and prosthetic research was $525 million (VA, 2016). In 2016, CDMRP received almost $1.3 billion1 for 29 research programs (CDMRP, 2015f).
Among the industrial and philanthropic organizations that fund medical research, the majority of monies in 2012 came from the pharmaceutical, biotechnology, and medical devices industries (Moses et al., 2015). The largest U.S. philanthropic funders of medical research are the Howard Hughes Medical Institute and the Bill & Melinda Gates Foundation (Viergever and Hendriks, 2016). Other major funders of medical research include other foundations, academic institutions, venture capital firms, and state and local governments (IOM, 2004). There are many other philanthropic and nongovernmental organizations that fund research on the same health conditions that CDMRP funds, such as cancer, amyotrophic lateral sclerosis, neurofibromatosis, muscular dys-
___________________
1 This number does not include $207 million for Restore Core Research Funding Reduction.
trophy, and tuberous sclerosis. In 2012, foundations, charities, and other private organizations spent more than $4 billion on medical research (Moses et al., 2015); many of those organizations have representatives on the CDMRP programmatic panels. Each of these organizations has a mission statement and review mechanisms for grant applications that help it fund research it believes will help cure or treat the health condition of concern to its constituency.
COMMITTEE’S APPROACH TO COORDINATION
While the focus of this report is on the CDMRP and its coordination of research priorities with NIH and VA, there are other federal and nongovernmental organizations that are involved in advancing research on one or more of the same health conditions that are addressed by these three organizations. For example, breast cancer research is funded not just by NIH, CDMRP, and VA, but also by numerous advocacy organizations as well (e.g., Susan G. Komen and the American Cancer Society [ACS]). This means that the potential for overlapping or redundant research is strong, and in an era of tight research dollars and competing priorities, it is critical to use research funds judiciously in a way that will do the most good. It is equally true that the multitude of organizations funding research on a particular health condition provides an opportunity for coordinating research dollars among them to not only maximize the dollars spent but also to move more effectively from basic to translational research and thus to improve treatment and care services for consumers.
In order to fulfill its task of evaluating CDMRP’s coordination of research priorities with NIH and VA, the committee performed a qualitative comparison of the three entities. The comparison was conducted using information available on the NIH and VA websites and the expertise and familiarity of the committee members with the organizations. The sections below summarize how NIH, VA, CDMRP, and selected other research funding organizations establish research priorities and their attempts to avoid funding duplicative research for health conditions in the CDMRP portfolio. The committee notes, however, that comparisons with these organizations must be made with caution as these organizations have different missions, stakeholder populations, funding mechanisms, and research program goals. For example, one of the first concerns when examining whether and how NIH, VA, and CDMRP coordinate research priorities is the noticeably different missions of the three organizations (see Box 7-1).
ESTABLISHING RESEARCH PRIORITIES
The ways in which research priorities are established for NIH, VA, and CDMRP are discussed below. In addition, the committee briefly considers how one nongovernmental organization, the American Cancer Society, determines its research priorities.
National Institutes of Health
NIH consists of 27 individual institutes and centers that conduct and/or fund medical research on a wide variety of human health conditions; the research ranges from basic biologic research to preventive medicine, applied medical advances, and public health. Although some research is conducted in-house by NIH researchers and clinicians, NIH’s focus is providing extramural funding to support researchers at academic institutions, health care industries, and biotechnology groups that are exploring scientific frontiers. Much of this extramural research is investigator-initiated.
The individual institutes and centers have individual research agendas and priorities, which may be aligned with legislative mandates, and they often have resources and budgets that may be related to specific diseases or body systems such as cancer, Alzheimer’s disease, H1N1 influenza, the immune system, or complementary and integrative medicine as directed by Congress (NIH, 2015c). Each institute also has its own
policies and procedures for establishing research priorities which may include strategic planning, annual planning, and the periodic review and assessment of research portfolios. Although the individual institutes and centers make their own decisions about which research projects to fund, they must factor in the needs and priorities of numerous stakeholders. Those stakeholders may include NIH in general, the scientific research community, those with the health conditions and their caregivers and families, and Congress (Kington, 2009). Research priorities are typically posted on the websites of the various institutes and centers.
The institutes and centers’ research priorities need to align with scientific opportunities, research gaps, the burden of disease in a population, public health needs, the expertise and experience of applicants, and emerging challenges (Kington, 2009). Each institute or center has an advisory council that makes recommendations on priority areas of research, pending policy, and funding of particular applications. The advisory council members are from outside NIH—scientific members, primarily from academia, and public representatives—chosen for their expertise, interest, or activity in matters related to health and disease (NIH, 2014a). Patients and patient representatives and other important stakeholders such as insurers, policy makers, and industry professionals may be involved in the priority setting at NIH, but their participation varies among the institutes and centers.
Stakeholders that may help the institutes and centers establish research priorities include the scientific community, both individual researchers and professional societies; patient organizations and voluntary health associations; Congress and the administration; the Advisory Committee to the NIH Director; NIH staff; and the general public. Together these scientists and stakeholders strengthen the research priority setting process for each institute or center. For example, the National Heart, Lung, and Blood Institute developed four strategic goals with input from its four advisory committees and also had two opportunities for public comment. According to the institute, these goals “will guide future Institute-solicited research, ultimately leading to new funding opportunity announcements, scientific workshops, and other activities” (NHLBI, 2015).
The NIH Division of Program Coordination, Planning and Strategic Initiatives (DPCPSI) in the Office of the Director is charged with providing leadership and coordinating priority-setting activities across NIH institutes and centers, especially for those activities that involve multiple centers. The division is tasked to “identify important areas of emerging scientific opportunity, rising public health challenges, and gaps in knowledge that deserve special emphasis” (Kington, 2009). However, other institutes within NIH also engage in coordinated and collaborative activities with internal and external organizations outside of DPCPSI, such as
the BRAIN initiative and the Interagency Pain Research Coordinating Committee. The director of NIH meets with all institute and center directors weekly to discuss research priorities, investments, and concerns that may affect an institute or center or NIH in general (GAO, 2014).
Thus, although the research needs of each NIH institute and center may differ considerably, the process by which the various institutes and centers establish their research priorities is similar across NIH. The presence of an oversight group such as the DPCPSI also ensures that there is a greater ability to address specific scientific needs and gaps and to help eliminate redundant research efforts. Research coordination is highlighted by the NIH Common Fund in the Office of Strategic Coordination in DPCPSI, which supports “cross-cutting, trans-NIH programs that require participation by at least two NIH institutes and centers or would otherwise benefit from strategic planning and coordination” (NIH, 2016b). The Common Fund encourages collaboration across institutes and centers but also maintains the necessary flexibility to address short-term, high-impact programs. The programs must include milestones, timeframes, and goals for the research to be conducted by them (NIH, 2016b).
Department of Veterans Affairs
VA is the largest health care system in the United States. It not only treats millions of veterans each year, it also conducts research on health conditions of particular interest to veterans and the health care providers who treat them. The VA strategic plan for 2014–2020 includes a focus on veterans’ health care research, particularly in coordination with DoD, with an emphasis on the long-term consequences of traumatic brain injury and posttraumatic stress disorder (PTSD) and their effects on underlying health, injury, and disorder mechanisms (VA, 2014). To develop this plan, VA received input from stakeholders across VA, external partners such as NIH and DoD, veterans and eligible beneficiaries, and the general public. Stakeholder involvement continued throughout the plan development process via workshops and notices in the Federal Register.
VA research is conducted by the VA Office of Research and Development (ORD), which oversees four service areas: biomedical, health services, clinical science, and rehabilitation. In addition to taking guidance from the strategic plan, ORD continuously receives input from Congress and veteran service organizations that may advocate for more research on specific health conditions, such as Gulf War illness, spinal cord injury, and prosthetics. ORD leadership also interacts with Congress, veteran service organizations, other stakeholders, other funders (such as pharmaceutical companies), and universities to identify the interests and needs of these communities. Based on this input, ORD establishes its research priorities, which are communicated to the VA research community (see Figure 7-1).
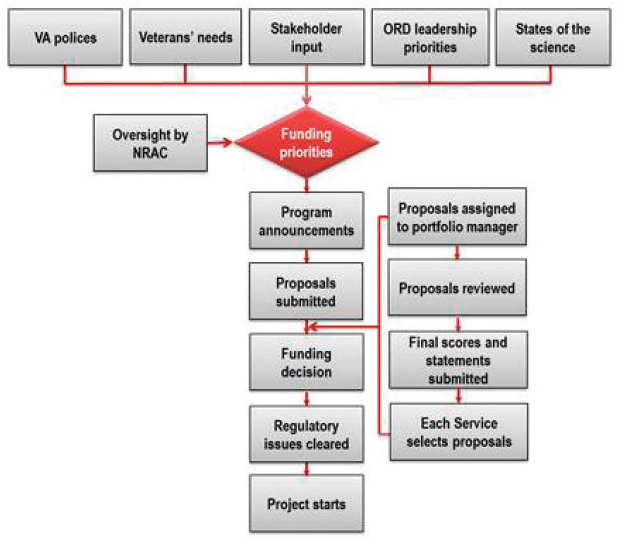
NOTE: NRAC = National Research Advisory Council; ORD = Office of Research and Development.
SOURCE: Katz, 2012.
VA also coordinates its research agenda with NIH. ORD staff members may be ex officio members of several NIH advisory councils such as the Advisory Council for the National Institute of General Medical Sciences. ORD staff frequently meets with program managers at the National Institute of Mental Health, the National Institute on Aging, and the National Institute of Neurological Disorders and Stroke. VA and NIH (along with DoD) co-sponsor conferences on various research topics such as the Second Annual Trauma Spectrum Disorders Conference: A Scientific Conference on the Impact of Military Service on Families and Caregivers, and may also issue joint requests for applications for health conditions of mutual interest, including PTSD, traumatic brain injury, and substance abuse. In preparation for the Million Veterans Program initiative, VA partnered with the National Human Genome Research Institute at NIH
to understand veterans’ knowledge of and attitude toward genomic medicine. VA recently started using the eRA (for electronic research administration) Commons, an NIH-sponsored electronic grants management system used to post and exchange grant-related information between the agency and the research community. Most, but not all, VA awards can be found in the NIH RePORTER (Research Portfolio Online Reporting Tools Expenditures and Results) database, although some awards, such as those in the VA Cooperative Studies Program, which funds large multi-site trials, have not yet been added to the NIH database. However, these awards are accessible via the VA website or at clinicaltrials.gov (Teresa Gleason, VA ORD, personal communication, July 7, 2016).
VA and DoD collaborate in areas of mutual interest such as mental health, traumatic brain injury, vision, and other health care areas. A special body, the Health Executive Council, was created to coordinate health care initiatives and programs under the two departments’ Joint Executive Council. There are 500 projects in the combined VA–DoD research portfolios (Katz, 2012), and the most recent Joint Executive Council report (VA and DoD, 2014) indicated numerous projects where DoD and VA are collaborating to actively improve service member and veteran health. As noted by a 2014 Institute of Medicine report on PTSD, “DoD and VA are funding broad PTSD research portfolios and are working collaboratively with the National Institutes of Health and other organizations to fill research gaps, for example, developing the joint National Research Action Plan for Improving Access to Mental Health Services for Veterans, Service Members, and Military Families for improving access to mental health services” (IOM, 2014, p. 228).
VA researchers may also collaborate with pharmaceutical and biotechnology companies on clinical trials. Many VA researchers have university appointments which permit them to coordinate and collaborate on research with their academic colleagues at their affiliated university and at other universities. Unlike other federal agencies such as NIH and DoD, VA does not have the statutory authority to make research grants to non-VA entities. Non-VA researchers may apply for funding, but a VA employee must be the principal or co-principal investigator on the application to receive VA funding, and, therefore, all VA research is essentially intramural. VA research funds are relatively limited, and intramural funding for specific research may be seen by VA leadership as seed money, which should be leveraged with other federal and extramural funding. VA researchers are allowed to apply for extramural funding from NIH and other organizations, such as CDMRP, through nonprofit research corporations or foundations. Thus, the research priorities of other potential funders, particularly NIH, are considered by VA research leadership when making funding decisions.
Congressionally Directed Medical Research Programs
As discussed in Chapter 2, CDMRP must fund research projects for the health conditions determined by Congress. For a few research programs, Congress may be relatively specific with regard to the particular areas of research that are to be funded. For example, in the House of Representatives report accompanying the 2012 Department of Defense Appropriations Bill, the House recommended that the Secretary of Defense (that is, CDMRP) “fund research for the advancement of prostate imaging technologies.”
Once Congress passes the DoD appropriations legislation that funds each research program, CDMRP, through its vision setting process, has discretion as to the specific research areas to be funded, the award mechanisms to be used, and the amount of funding to be allocated for each award mechanism. The vision setting process and how research priorities are established during that process are discussed in detail in Chapter 4.
CDMRP has no legislative mandate to coordinate its research priorities or funding with other governmental or nongovernmental organizations, although it does so informally to a varying extent in the individual programs. During the vision setting and programmatic review process, the programmatic panel assesses the state of the science, including a review of what the program and other agencies—both public and private—are currently funding and the potential for duplicative research by CDMRP and other organizations. This assessment is possible, in part, by having representatives of other funding agencies or organizations (e.g., NIH, VA, ACS, or other advocacy groups) participate on a programmatic panel. The committee notes that some programmatic panels had no representation from organizations that the committee believes would have valuable input for that program. For example, it appears that there has never been a VA representative on the Prostate Cancer Research Program’s programmatic panel, although the committee finds that aging veterans are a likely population for prostate cancer. Furthermore, there has been no representation in recent years from the National Cancer Institute on the Breast Cancer Research Program’s programmatic panel, although they are a large funder of breast cancer research.
Other major funders for a specific health condition may be engaged and asked to give briefings at the vision setting meeting. These participants may or may not be part of the programmatic panel. For example, the Gulf War Illness Research Program invites the senior program manager of the Gulf War illnesses portfolio at VA to give a briefing at its vision setting meeting; however, CDMRP reports that this participant does not sit on the programmatic panel (Salzer, 2016b). The committee notes, however, that there has been a VA representative on each programmatic panel for the
Gulf War Illness Research Program since 2010. This program has recently begun to list all of its funded research, including the principal investigator, project title, funding level, and lay and technical abstracts “in an effort to facilitate collaborations and speed GWI research” (CDMRP, 2016i).
During its open-session meetings with some of the CDMRP program managers, the committee learned that program managers often participate in interagency groups that look at research efforts across the members’ portfolios. For example, the Breast Cancer Research Program’s program manager sits on several federal interagency groups—such as the Advisory Committee on Breast Cancer in Young Women, the Trans-Agency Early-Life Exposures and Cancer Working Group, and the Interagency Breast Cancer and Environmental Research Coordinating Committee—that include not only representatives from a variety of federal agencies (NIH, the Food and Drug Administration [FDA], VA), but nongovernmental members as well (Vaday, 2016).
The committee notes that although each CDMRP research program engages in vision setting (see Chapter 4), the programs do not develop long-term (e.g., 5-year) strategic plans that might include more formal mechanisms for determining who should be included on the programmatic panels (that is, what other organizations should be represented in the near- and long-term); what research should be tracked in other organizations and how this information might be tracked; and what areas of promising research, if not currently funded, might be considered for funding by CDMRP in the future (e.g., epigenetics or new treatments in the pipeline). One exception to this is the Breast Cancer Research Program, which in 2013 developed a strategy and landscape document to spell out what the programmatic panel believed to be the overarching challenges in breast cancer and the funding opportunities that the panel hoped would move the science forward. The Ovarian Cancer Research Program has also recently developed a landscape document that identifies long-term goals, areas of research encouragement, and funding mechanisms as well as providing an overview of the ovarian cancer research work being undertaken or funded by numerous organizations in the United States and around the world (OCRP, 2015). However, these landscape documents, while informative for vision setting and to a lesser extent, programmatic review, do not provide a framework for achieving each program’s mission and goals beyond the current year’s funding. The committee finds that although CDMRP funding may be for 1 year only, this does not preclude the development of a long-term strategic plan by the CDMRP research programs. Furthermore, while the landscape documents developed by two programs are helpful, the committee does not consider them to be equivalent to a strategic plan. Lack of such a plan makes it more difficult to ensure that
CDMRP research priorities are innovative, not redundant, and that they are coordinated with the research being performed or funded by other organizations.
Nongovernmental Organizations
Although the committee was tasked with evaluating how CDMRP coordinates research priorities with NIH and VA specifically, the committee believes that given the amount of research that is conducted by industry and private organizations and foundations in areas that are also part of the CDMRP portfolio, it was important to consider how these organizations set their research priorities and interact with CDMRP. The committee used ACS as a representative organization when considering how nongovernmental organizations set research priorities as it is a large funder of cancer research (in 2016, more than $400 million for almost 750 grants). ACS has an informative website (www.cancer.org) from which the following information was obtained.
ACS prioritizes research that, in its judgment, will have the biggest impact in the fight against cancer and save the most lives, but there is no strict set of research priorities. Research priorities are not established by a committee. In general, ACS does not prescribe specific research topics as it prefers research to be investigator initiated. However, ACS does list the types of awards to which investigators might apply in areas such as immunotherapy, training, and fellowships (ACS, 2016). Researchers may submit proposals for any type of cancer research project, including quality of life, cancer control, and patient support and survivorship (ACS, 2015).
Representatives of ACS have served on several CDMRP programmatic panels, but it is unclear to the committee whether CDMRP staff, particularly the CDMRP program managers for the cancer programs, is involved with ACS. Representatives of many other nongovernmental organizations have participated on both peer and programmatic review panels at CDMRP. These organizations typically include the major advocacy and funding groups for a health condition, such as Susan G. Komen for breast cancer, the Multiple Sclerosis Society, Autism Speaks, and the Muscular Dystrophy Association as well as smaller foundations such as Children’s Tumor Foundation and the Craig H. Neilsen Foundation. The committee finds that the participation of CDMRP program managers in research reviews at nongovernmental organizations, if appropriate, might strengthen the CDMRP knowledge base in terms of what organizations are funding research for the health conditions of concern as well as specifics on current or anticipated research activities.
AVOIDING RESEARCH DUPLICATION
One of the goals of coordinating research funding among government agencies and nongovernmental organizations, including international organizations, is to minimize the likelihood of one group funding the same research as another group. Although some duplication in research is desirable in order to verify the results of prior studies, in general most agencies attempt to fund new research that moves the health and science fields forward. The goal of reducing research redundancy is laudable, but in actuality it is often difficult to achieve. In this section, the committee examines the mechanisms that CDMRP, NIH, and VA have in place to avoid research duplication among the three agencies.
The committee finds that many federal agencies already have formal mechanisms in place to reduce redundancies in the research they fund. For example, the Interagency Pain Research Coordinating Committee, which is composed of representatives from the Agency for Healthcare Research and Quality, the Centers for Disease Control and Prevention, DoD, FDA, NIH, and VA, began its work with an analysis of each participant’s research portfolio in order to determine potential redundancies and collaborative opportunities across the agencies. Although it is recognized that each agency has a unique mission and specialization with regard to pain research, assessing the portfolios revealed many areas of overlapping interests across the agencies. Further analysis showed that the research within these overlapping topic areas was not redundant but for the most part was complementary and that some agencies had developed ongoing collaborative efforts to facilitate overlapping areas of research interests and identify future partnerships (NIH, 2014b). A further example of CDMRP’s efforts to coordinate research efforts and avoid duplication is its membership in the International Cancer Research Partnership. This multinational organization, established in 2000, seeks to improve global collaboration and strategic coordination of cancer research being conducted worldwide. It maintains a database of information on 77,351 grants in a common searchable format from 110 member organizations, including CDMRP, NIH, and other U.S. organizations that fund cancer research (ICRP, 2016). The committee was not informed of who from CDMRP participates in this organization’s activities.
On the other hand, although there are numerous federal interagency groups, with members from the NIH, VA, and DoD (although not necessarily CDMRP), that work to establish research priorities and reduce redundancies in research, their efforts are not always adequate. In 2012 the Government Accountability Office (GAO) issued a report, 2012 Annual Report: Opportunities to Reduce Duplication, Overlap and Fragmentation, Achieve Savings, and Enhance Revenue, that looked at a number of federal government agencies with the goal of streamlining activities within and
between agencies in order to make them more efficient and effective. GAO performed searches on funded applications for breast cancer and PTSD research using the databases that the agencies used primarily to identify applications and projects that might be duplicative. Most of the applications GAO looked at did not appear to be duplicative, but it did identify two applications—one funded by VA and the other by DoD—that a VA program manager confirmed were duplicative. It turned out, however, that modifications to the applications’ research aims had since been made at VA, but these changes had not been updated in the databases, nor shared with the other agencies.
Of relevance to this committee’s task, GAO assessed the interactions between NIH, DoD, and VA with regard to reducing redundancies in funding health research among the three organizations. GAO (2012) concluded the following:
NIH, DOD, and VA each lack comprehensive information on health research funded by the other agencies, which limits their ability to identify potential areas of duplication in the health research they fund. NIH, DOD, and VA program managers—officials who typically manage agency research portfolios and may provide input to senior agency officials responsible for making funding decisions—told GAO that, when reviewing health research applications, they typically search publicly available databases for potentially duplicative research projects funded by other federal agencies. . . . According to NIH, DOD, and VA officials, the information provided in the research databases they use to identify any potential duplication when making funding decisions is generally not sufficient. In such cases, officials said they typically obtain information not contained in the databases by contacting colleagues at other federal agencies to obtain details on specific applications. . . . Officials at NIH, DOD, and VA acknowledged that duplication may sometimes go undetected. . . . Because multiple federal agencies fund research on topic of common interest, there is potential for unnecessary duplication. As long as research on similar topics continues to be funded by separate agencies, it is incumbent on the agencies to coordinate effectively with each other.
The GAO report then offered specific ways in which NIH, DoD, and VA could collaborate to permit program staff to more efficiently and effectively determine the potential overlap in applications that each organization reviews. A major recommendation was to enhance the respective databases that are used by the organizations to catalog applications by including more information about the applications, including if and when the applications are modified, and providing a contact person in each organization to answer questions about the applications.
In response to the GAO report, in April 2015, DoD delivered a short report to Congress that summarized the progress that DoD had made toward improving access to comprehensive electronic information for
funded health research among the organizations, specifically a pilot project to upload DoD information to the NIH RePORTER database.
National Institutes of Health
Although NIH institutes and centers may consult with outside organizations when developing their strategic plans and annual research priorities, there is no specific requirement that they do so. The DPCPSI identifies and reports on research that represents important areas of emerging scientific opportunities, highlights new public health challenges, and assists with program evaluations (NIH, 2016c).
NIH has standing policies to avoid funding duplicative research applications within the institutes. Specifically, the NIH policy is that an investigator may not submit essentially the same research application to more than one institute or funding announcement at the same time. If that happens, NIH administrators will return one of the applications without review. In addition, when applications are being considered for funding after peer review, investigators must provide up-to-date information that includes all other funding they have or are currently receiving, to insure that duplication in funding to any individual investigator is avoided. There is no NIH policy related to how program officers and directors should check for duplicative research when the research teams are different, although this is likely to happen on an individual basis.
Department of Veterans Affairs
VA recently started using eRA Commons, an NIH-sponsored electronic grants management system used to post and exchange grant-related information between the agency and the research community. In addition, VA ORD-funded projects are included in the public NIH RePORTER database (Katz, 2012). However, GAO (2012) found that only about one-quarter of VA-funded research was included in the NIH RePORTER as of 2012 making it difficult to determine whether there was overlap or duplication of funded research. As of 2016, most, but not all, VA awards can be found in the NIH RePORTER database (Teresa Gleason, VA ORD, personal communication, July 7, 2016).
VA and DoD have developed a collaboration guidebook that recommends that efforts be made to identify any related research prior to beginning any research collaborations. These efforts might begin with literature searches for published work, but the guidebook acknowledges the challenge to finding ongoing research projects in either organization. The guidebook also recommends that grants.gov, the official website for
information on federal grants, including those awarded by VA and DoD, be searched as well (Resnick et al., 2013).
Congressionally Directed Medical Research Programs
The Procedures to Avoid Research Duplication document on the CDMRP website informs applicants, awardees, other funding agencies, and the public of how CDMRP identifies and avoids funding duplication. Investigators may apply for funding for the same research from different funding agencies, but applicants may not accept funding for the same research from more than one source.
There are several points throughout the CDMRP program cycle where duplication of research is checked and, if necessary, mitigated, including at application submission, peer review, programmatic review, and the negotiation and monitoring of funded awards. Principal investigators and all key personnel are required to submit a comprehensive list of previous, current, and pending funding support. CDMRP reports that applicants must identify where they perceive there to be proposed research overlaps with other previous, existing, and pending research projects, or they must state that there is no such overlap (CDMRP, 2016h). The committee notes, however, that it was unable to find this requirement in the program announcements or the general application instructions, although applicants are prohibited from submitting “applications that are essentially identical or propose essentially the same research project to different funding opportunities within the same program and fiscal year” (CDMRP, 2015b).
CDMRP states that duplicative research may also be identified during peer review. Peer reviewers, particularly the scientist reviewers who have extensive knowledge of the subject they are reviewing and may serve on other agencies’ review panels, might be able to provide feedback as part of their evaluations on whether the proposed research has already been published or is the subject of an application to another funding agency. The CDMRP science officer may review these reviewer comments if the application moves to award negotiations (CDMRP, 2016h). The committee cautions, however, that peer reviewers need to respect the confidentiality of applications submitted to other agencies for which they may also serve as reviewers. Thus, peer reviewers may need to limit their discussion of potential research overlaps to their knowledge of the published literature.
Programmatic review is another opportunity for assessing the duplication of research. Many of the programmatic panels include at least one representative from other relevant federal agencies, such as NIH and VA, who are familiar with the research program area and can provide
information about similar research that is being funded by their own organizations (CDMRP, 2016h). CDMRP program managers may also be aware of similar funding opportunities through direct contact with their counterparts at other organizations and by attending other agencies’ and organizations’ meetings, working groups, and national conferences (Vaday, 2016). The committee finds that although these contacts are helpful, they are ad hoc and not systematic or necessarily comprehensive; thus, the program manager’s knowledge of other funded research efforts may be incomplete.
After an application has been recommended for funding, but before the award negotiations begin, the applicant must supply an updated, comprehensive list of support. On the basis of this information, combined with the peer and programmatic review notes, CDMRP’s internal grants management database (eBRAP), NIH’s eRA Commons system, NIH RePORTER, International Cancer Research Partnership, the Federal RePORTER, and other program-specific sites, CDMRP science officers try to determine whether the application has potential research overlaps with other federal or nonfederal agencies (CDMRP, 2016h). The committee recognizes that even using a multitude of approaches such as these may not capture all redundancies. All CDMRP awards are entered into the Federal RePORTER database and made available for public scrutiny.
















