Proceedings of a Workshop
| IN BRIEF | |
|
January 2017 |
|
International Perspectives on Integrating Ethical, Legal, and Social Considerations into the Development of Non-Invasive Neuromodulation Devices
Proceedings of a Workshop—in Brief
Emerging neurotechnologies—devices and techniques designed to collect information about the brain or affect its function—are becoming increasingly important due to scientific and technological advances and a persistent need to develop effective therapies to address the large global burden of neurological and psychiatric disease.1 While offering the potential to significantly improve the health outcomes of patients and the quality of life of individuals, the rapid advances in development of neurotechnologies and their growing availability raise many important ethical, legal, and social implications (ELSI) and considerations related to the conduct of responsible research and innovation (RRI). Although these topics often have been discussed separately from the established research and development process, there is a growing interest in developing better mechanisms for thoughtfully anticipating and integrating ELSI throughout the course of innovation in neurotechnology.
On September 15–16, 2016, the Organisation for Economic Co-operation and Development (OECD)—in collaboration with Arizona State University and the National Academies of Sciences, Engineering, and Medicine—organized a workshop in Washington, DC, on Neurotechnology and Society: Strengthening Responsible Innovation in Brain Science. The workshop brought together individuals—representing public and private sectors, research organizations and universities, public and private funders, and civil institutions from around the world—to explore approaches to better integrating societal values, scientific advancement, and economic considerations during the development and use of novel neurotechnologies.
As part of this overall workshop, the Forum on Neuroscience and Nervous System Disorders of the National Academies organized a session that used non-invasive neuromodulation as a case study to prompt further, concrete discussion about practical concerns and opportunities related to integrating ELSI and RRI throughout research, development, regulation, and use of new neurotechnologies. This Proceedings of a Workshop—in Brief summarizes the presentations and discussions from that session.2,3
___________________
1 Bloom, D. E., E. T. Cafiero, E. Jané-Llopis, S. Abrahams-Gessel, L. R. Bloom, S. Fathima, A. B. Feigl, T. Gaziano, M. Mowafi, A. Pandya, K. Prettner, L. Rosenberg, B. Seligman, A. Z. Stein, and C. Weinstein. 2011. The global economic burden of noncommunicable diseases. Geneva, Switzerland: World Economic Forum.
2 For a broad summary of the workshop and additional background information, including a discussion of ELSI, RRI, and anticipatory governance frameworks, see Garden, H., D. M. Bowman, S. Haesler, and D. E. Winickoff. 2016. Neurotechnology and society: Strengthening responsible innovation in brain science. Neuron 92(3):642-646. doi: http://dx.doi.org/10.1016/j.neuron.2016.10.053.
3 The OECD is preparing a comprehensive report on this topic. For additional information, visit: https://www.innovationpolicyplatform.org/project-emerging-technologies-and-brain-oecd-bnct (accessed December 27, 2016).
![]()
A SNAPSHOT OF DEVELOPING ISSUES IN NEUROTECHNOLOGY
Non-invasive neuromodulation devices, which apply electrical or magnetic fields through the scalp to change human brain function without the need for invasive procedures, are already being used and studied to treat symptoms across a variety of nervous system disorders, including, for example, depression and epilepsy, and to augment cognitive performance in non-therapeutic settings.4 The application of non-invasive neuromodulation technology to impact human performance and health raises important ethical and societal issues concerning equity, performance enhancement, access to treatment, clinical use in vulnerable populations and involuntary treatment, and unsupervised device use. Non-invasive neuromodulation therefore provides a snapshot of developing issues in the broader nascent field of neurotechnology, said Henry T. Greely, the Deane F. and Kate Edelman Johnson professor of law at Stanford Law School, and moderator of the session.
There is no clear structure for dealing with the ethical, legal, and social implications of neuromodulation technologies, Greely said, and the systems currently in place are works in progress involving many different stakeholders around the world. In the United States, for example, both the Food and Drug Administration (FDA) and the National Institutes of Health (NIH) have important roles in considering ELSI. At NIH, one such recent effort includes the establishment of a Neuroethics Workgroup for the Multi-Council Working Group (MCWG) that oversees the Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative.5 The workgroup charge includes reviewing research applications and providing guidance on proposed funding areas. Greely added that other groups outside the government, such as nonprofit foundations and professional societies, also encourage more work on ELSI topics.
The ability to effectively treat mental and neurological disorders is still wanting, and a huge burden now exists, noted Greely. Electrical, magnetic, and other forms of neuromodulation present a real opportunity and a different path forward, and they require stakeholders to address barriers in ethical, medical, regulatory, scientific, and industry issues at the same time, he added.
NON-INVASIVE NEUROMODULATION: A SURVEY OF SCIENCE AND USE
Given ever-developing scientific advances, clinical practices, and access to technology, meaningful integration of ethical and societal considerations in the research and development process starts with understanding the current landscape of neurostimulation device knowledge and use.
Sarah H. Lisanby, director of the Division of Translational Research at the National Institute of Mental Health (NIMH), described the tools and scientific principles of non-invasive neuromodulation and the state of the art in their clinical application. Although a broader array of tools is now available, non-invasive neuromodulation has been used for more than seven decades in psychiatry in the form of electroconvulsive therapy (ECT), which she said remains one of the most effective and rapidly acting treatments available for severe depression.
Current neurostimulation tools used in psychiatry (see Figure 1) include surgically implantable devices for vagus nerve stimulation, which are FDA approved for treatment-resistant depression; and deep brain stimulation, which has a humanitarian use device exemption from the FDA for the treatment of obsessive-compulsive disorder and is being investigated by several research groups for use in other disorders, said Lisanby. Non-invasive devices that use magnetic fields include transcranial magnetic stimulation (TMS), which induces small electric currents in specific target areas of the brain and is FDA cleared for the treatment of depression, and magnetic seizure therapy, an experimental tool designed to combine the non-invasive and focal targeting aspects of TMS with the high efficacy of ECT. The relatively new transcranial direct current stimulation or transcranial alternating current stimulation devices use electrical fields to change brain function and are currently under investigational use.
New neuromodulation tools are needed because neuropsychiatric conditions are now the leading source of disability and economic burden worldwide, said Lisanby. In addition, existing therapies, such as pharmacological intervention for the treatment of depression, are not as effective as they need to be, she noted. From a scientific standpoint, non-invasive neuromodulation is changing the way we investigate brain–behavior relationships and probe the pathophysiology of diseases. Having the ability to experimentally modulate brain activity non-invasively in humans, she explained, allows going beyond observations and conducting true hypothesis testing to understand what is driving these disorders.
___________________
4 The Forum on Neuroscience and Nervous System Disorders held a workshop on Non-Invasive Neuromodulation of the Central Nervous System on March 2–3, 2015, in Washington, DC. For a summary of the workshop, including additional information on noninvasive neuromodulation, visit: https://www.nap.edu/catalog/21767 (accessed December 27, 2016).
5 The MCWG Neuroethics Workgroup includes clinicians, researchers, and experts on law and ethics. For additional information, visit: https://www.braininitiative.nih.gov/about/newg.htm (accessed December 27, 2016).
Non-invasive neuromodulation is a current-day clinical reality in the practice of psychiatry, Lisanby said. At the same time, off-label use also exists today, with unapproved devices being marketed in some instances as a safe treatment for a broad range of clinical conditions. There are also claims for recreational effects from neuromodulation devices, such as enhanced sports performance, improved attention, and lucid dreaming for which there is no evidence basis in the mainstream medical literature. Overall, Lisanby noted, research needs to catch up with the pace of clinical adoption.
Lisanby concluded by noting that as the field moves forward, there is a need to learn more about the dosing of non-invasive neuromodulation, which can involve complex interactions among physical stimulation parameters, the environment, and other factors not well controlled. She also expects to see increases in the supply of clinics and clinicians who are offering non-invasive neuromodulation treatments to patients and increases in the number of companies and industries built around offering these products, including direct-to-consumer devices for the “do-it-yourself” (DIY) community. Lisanby noted that this highlights the need for professional guidelines to inform medical practice and for guidelines to inform over-the-counter and direct-to-consumer uses.
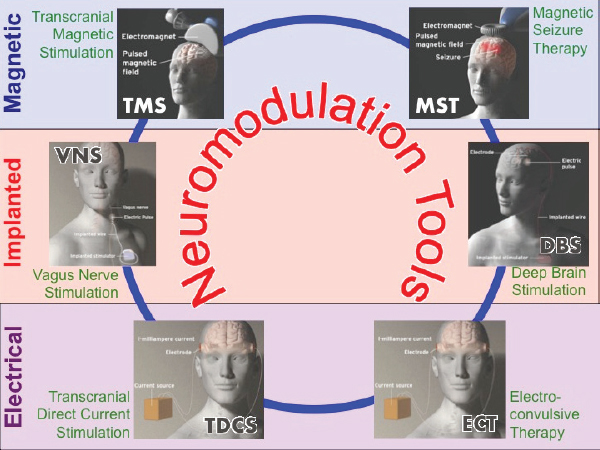
SOURCE: Presentation by Lisanby, September 15, 2016.
NON-INVASIVE NEUROMODULATION: PERSPECTIVE OF A LARGE PHARMACEUTICAL COMPANY
Companies in the private sector undertake a large portion of the research and development of new therapeutics and therefore are stakeholders in efforts to integrate ELSI and RRI considerations during the life cycle of future product development.
Jeffrey S. Nye, vice president of Neuroscience Innovation and Scientific Partnership Strategy at Janssen Research and Development, LLC, spoke about the perspective of large pharmaceutical companies on non-invasive neuromodulation technologies. He discussed how the different approaches from the regulatory, payer, and industry sectors to these devices can influence patients’ access to treatments, including potentially creating barriers to the use of treatments that may benefit patients more than those readily available.
From the perspective of industry, explained Nye, there should be no bias or preference between a treatment in the form of a pill or a device; therapeutics that are valuable to patients, caregivers, and society should be remunerated at a level corresponding to the benefit created by those interventions. When Nye examined non-invasive neuromodulation devices, however, adoption levels were much lower than would be expected given the very high response rates to treatment in patients with treatment-resistant major depressive disorder. Nye said that in fact, the adoption rate for TMS, which has been around for about 10 years, is approximately 50 times lower than that of a new antidepressant medicine that has been on the market for only 1–2 years. Nye noted that while this type of measure may not be important to everyone, it certainly does drive investment decisions in industry.
Several factors may be contributing to the low rate of adoption and usage of non-invasive neuromodulation devices, according to Nye. These include regulatory issues—such as different requirements across countries—and what
may be a lower bar for clinical trials involving devices considered widely safe in the United States. If very small trials are sufficient for regulatory decisions, Nye explained, the resulting data can be limiting and not sufficiently influential to drive adoption.
The second issue affecting adoption involves reimbursement. As an example, Nye described the TMS market, which took many years to develop the reimbursement system currently in place. This long period of time is a barrier compounded by having to obtain reimbursement approvals region by region and with private insurers individually, he said. The relatively small size of device trials also affects how much evidence can be provided to payers, which may result in a lower tier of coverage that requires prior authorization or high co-pays, or both. Nye noted that it would be helpful if professional organizations provided guidelines on how to situate devices in the context of practice guidelines. He added that this may not be currently feasible when the data available from small trials do not include comparison against other devices, such as ECT, or pharmaceutical treatments.
A third factor adversely affecting the adoption of neurostimulation technologies involves patient preferences. Some people would prefer to take a pill rather than show up repeatedly at a clinic for a non-invasive neuromodulation procedure, said Nye. Even if the procedure itself is brief, the overall time commitment required for each visit may be burdensome, especially for patients who work or have other demands on their time. He added that two more considerations influencing patient demand include travel distance, which can be a problem in areas where few treatment centers are available, and sensitivity to co-pays.
Nye also examined why psychiatrists have not widely adopted non-invasive neuromodulation. Although psychiatrists are relatively well aware of these technologies and willing to refer patients to treatment, many are unwilling to adopt and implement these treatments in their own practices. However, psychiatrists who completed residency more recently and were exposed to these technologies during training are beginning to adopt it to a greater extent, Nye said. He also noted that many practices do not have procedure rooms or technicians, and that adopting these devices requires a different business model than a traditional psychiatry practice; practices have to buy or lease the instrument, which can become a financial barrier. Finally, the price of treatments is going to affect how widely one can expect them to be adopted. In the United States, generic antidepressant medicines can cost as little as $20 USD per month, compared to TMS therapies, which may cost around $4,000 USD per month, Nye explained, adding that this big difference plays a role in payers’ decision making.
Reflecting on potential future opportunities, Nye noted that if device companies invested more heavily in conducting larger studies and performing more analyses of how a new technology fits into the existing therapeutic modality, there would be greater understanding by professional societies and a greater ability to adopt the treatment. The device sector may benefit from conducting more economic impact studies on devices; this is a common practice for pharmaceutical companies developing new medicines. Nye also suggested aligning the requirements for what is considered appropriate data for regulatory determinations, and what is appropriate for reimbursement decisions by payers. While initially this might not be in the interest of either party, he said, it would be in the interest of patients in the end. As it stands, Nye noted, the disparity between regulatory and reimbursement requirements in the United States may result in a device that has solid clinical data and potential for benefiting patients, but is not supported by the reimbursement authorities.
NON-INVASIVE NEUROMODULATION: PERSPECTIVES FROM A DEVICE START-UP COMPANY
Although some regulatory, technical, and economic opportunities and barriers are shared across the private sector, understanding the particular circumstances, environment, and interests of varied stakeholders can be critical for establishing relevant and lasting frameworks that integrate ELSI and RRI in research and development of devices. Many companies specializing in neurostimulation technologies are smaller start-up ventures with unique needs and interests. Ana Maiques, chief executive officer of Neuroelectrics, based in Barcelona, Spain, and Cambridge, Massachusetts, described the devices currently being developed and marketed by her company and shared some of the experiences and concerns that are important to smaller businesses in the field.
Responsiveness to patient preferences and improving user experience when interfacing with the product are important priorities, explained Maiques. To illustrate the advances in mobility and comfort of recent non-invasive neuromodulation devices, Maiques demonstrated one of her company’s products, a cap embedded with small electrodes that can record and display her brain’s electrical activity in real time using 32-channel electroencephalography (EEG). In addition to recording EEG, the device can also deliver electrical currents to the brain using transcranial current stimulation (tCS).
Importantly, this type of technology holds promise for future use in home diagnostics, said Maiques, adding that with advances in technology and data analysis tools, such as machine learning, EEG may become a powerful biomarker for some disorders in coming years. Currently, the cap is used by many hospitals to record EEG for diagnosing epilepsy and sleep disorders. The company is also engaged in research funded by The Michael J. Fox Foundation for Parkinson’s Research to analyze EEG data from patients with sleep disorders. Using data from 118 adult patients, it was able to predict later development of Parkinson’s disease up to 8 years before onset, Maiques said, and it is now in the second phase of the project involving follow-up in multiple sites across seven countries.
One barrier faced by entrepreneurs, both inside and outside of the United States, involves their initial apprehension of the FDA. Maiques emphasized that while the regulatory process is very complex, the FDA has been extremely helpful as her company has gone through the regulatory process. She added that the vast differences between regulators in the European Union and the United States can pose another barrier and suggested that more harmonization and more “fast lanes” for innovations would be helpful.
Other areas could also benefit from additional attention, according to Maiques. For example, she would like to see more preparation by government agencies, regulators, and medical professionals to support prescribing treatments using non-invasive neuromodulation devices for home use, as well as additional education on such technology for mental health providers. Based on her experience, there is still fear of neuromodulation or brain science in general among some providers both in the European Union and the United States. Maiques emphasized that professional societies, governments, and other organizations should ideally share in the responsibility to provide more education because companies are not able to do it alone.
Lastly, Maiques noted that several ethical issues exist for companies developing non-invasive neuromodulation technology. For example, if this technology can make people smarter, who is going to have access? Who will regulate access to these technologies? These are important issues to address now, she said, because developments in the technology may happen much faster than predicted.
ETHICAL AND REGULATORY ISSUES ASSOCIATED WITH CONSUMER USE OF NON-INVASIVE NEUROMODULATION
The introduction of non-invasive neuromodulation technology to clinical settings has been accompanied with a proliferation of non-therapeutic and DIY use of such devices among individuals around the world. Though many people may regard non-invasive methods as inherently safe, invasive medical procedures performed by trained health care providers in a clinical setting may involve less risk than non-invasive stimulation technology used at home in an unsupervised manner. Furthermore, while some consumers seek therapeutic benefits, others may be interested in pursuing non-clinical or recreational effects through neurostimulation. Hannah Maslen, postdoctoral research fellow at The Oxford Centre for Neuroethics, focused on consumer use of non-invasive neuromodulation devices, specifically consumers using them at home, whether they have made them themselves or bought them from a company. Maslen described the current regulatory landscape in which manufacturers can circumvent approval or the certification process and sell devices marketed for gaming, athletics, or cognitive enhancement by using claims that they are not for therapeutic purposes. At the same time, counterpart devices sold for therapeutic purposes or for research are regulated by medical devices legislation in the European Union, the United States, and other jurisdictions.
Maslen outlined the European Union Medical Devices Directive’s definition of a medical device, which includes products intended by their manufacturer to be used specifically for diagnostic and/or therapeutic purposes.6 A similar situation exists in the United States, with medical devices defined as products intended to be used for diagnosis or therapy. Nevertheless, new device regulations and associated legislations have just undergone major review and have gained support in the European Union. These new regulations include a list with groups of products without an intended medical purpose that will be brought within the remit of the medical devices regulatory oversight, explained Maslen, adding that one of the major challenges will be working out how to approach risk–benefit assessment.
Many people are accustomed to thinking about medical device benefits such as reduced pain or reduced symptoms of depression. But Maslen noted that while there is an understanding of the significance of those benefits for individuals’ lives, when it comes to weighing the benefits of improved gaming, improved working memory, or enhanced athletic performance, it becomes much harder to say how substantial these benefits are for individuals. Indeed, consumers are buying these products because they think the benefits are going to be meaningful for them. If the FDA
___________________
6 Council Directive 93/42/EEC, Article 1. For additional information, visit: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CONSLEG:1993L0042:20071011:en:PDF (accessed December 27, 2016).
were to follow suit, things would get even more complicated, said Maslen. This is because the FDA has to be convinced that the probable benefits of the device outweigh its probable risks, that is, not only are the risks acceptable, but that the risks are outweighed by the benefits.
Maslen is currently working with the European Commission’s Working Group on New and Emerging Technologies in Medical Devices to look at potential approaches to this sort of risk–benefit assessment for new neurostimulation devices. She said that at this important juncture, one question to ask is what level of risk presented by these devices should be considered acceptable for the direct-to-consumer market. Another question is how safety could be designed into direct-to-consumer devices and whether such design characteristics should be required as a condition for approval and placement on the market.
Lastly, Maslen talked about the DIY community in which individuals build their own neurostimulation devices using readily available electrical components. She said that while there are individuals in that community who sincerely try to use due diligence, read research papers, and emulate as much as possible procedures used by researchers, there are still those who are going to be less careful and who might be putting themselves at a lot of risk. Maslen noted that another ethical question that should be asked is what obligation, if any, do scientists have when individuals are taking their research and trying to understand and apply it themselves?
FINAL THOUGHTS
As the development of non-invasive neuromodulation devices accelerates and their clinical and non-therapeutic application grows, several workshop participants noted that ethical and societal considerations could inform regulators, clinicians, academia, and private-sector stakeholders to promote effective and safe use of neurotechnology. Individual workshop participants suggested potential future efforts to facilitate this integration may include additional education and increased awareness about non-invasive neuromodulation technology among health care providers, policy makers, payers, and society at large. Other workshop participants noted that current directions include anticipating and planning the potential frameworks, infrastructure, and workforce that would be required to support technological, regulatory, and economic challenges associated with the introduction of new technologies and their use in innovative ways. Lastly, several workshop attendees said it may be helpful to consider the interactions among the various sectors involved in the life cycle and application of non-invasive neuromodulation devices and how differences in their alignment may influence ELSI considerations, such as availability and access to effective treatments for patients across the world or the evaluation of benefits and risks to individuals outside the clinical environment.♦♦♦
DISCLAIMER: This Proceedings of a Workshop—in Brief was prepared by Noam I. Keren and Clare Stroud as a factual summary of what occurred at the meeting. The statements made are those of the rapporteurs or individual meeting participants and do not necessarily represent the views of all meeting participants; the planning committee; the National Academies of Sciences, Engineering, and Medicine; or any of the other organizations involved in the planning of the meeting.
REVIEWERS: To ensure that it meets institutional standards for quality and objectivity, this Proceedings of a Workshop—in Brief was reviewed by Diana M. Bowman, Arizona State University, and Sebastian Haesler, Neuro-Electronics Research Flanders. Lauren Shern, Health and Medicine Division, served as the review coordinator.
SPONSORS: This Proceedings of a Workshop—in Brief was supported by contracts between the National Academy of Sciences and the Alzheimer’s Association; Brain Canada Foundation; Cohen Veterans Bioscience; the Department of Health and Human Services’ Food and Drug Administration (5R13FD005362-02) and National Institutes of Health (NIH) (HHSN26300089 [Under Master Base #DHHS-10002880]) through the National Center for Complementary and Integrative Health, National Eye Institute, National Institute of Mental Health, National Institute of Neurological Disorders and Stroke, National Institute on Aging, National Institute on Alcohol Abuse and Alcoholism, National Institute on Drug Abuse, and NIH Blueprint for Neuroscience Research; Department of Veterans Affairs (VA240-14-C-0057); Eli Lilly and Company; Foundation for the National Institutes of Health; Gatsby Charitable Foundation; Janssen Research & Development, LLC; Lundbeck Research USA; Merck Research Laboratories; The Michael J. Fox Foundation for Parkinson’s Research; National Multiple Sclerosis Society; National Science Foundation (BCS-1064270); One Mind; Pfizer Inc.; Pharmaceutical Product Development, LLC; Sanofi; Society for Neuroscience; and Takeda Development Center Americas, Inc.
For additional information regarding the meeting, visit http://www.nationalacademies.org/hmd/Activities/Research/NeuroForum/2016-SEP-15.aspx.
Suggested citation: National Academies of Sciences, Engineering, and Medicine. 2017. International perspectives on integrating ethical, legal, and social considerations into the development of non-invasive neuromodulation devices: Proceedings of a workshop—in brief. Washington, DC: The National Academies Press. doi: 10.17226/24643.
Health and Medicine Division

Copyright 2017 by the National Academy of Sciences. All rights reserved.







