practices that have established mechanisms for second readings of images. “There needs to be a deliberate focus on teamwork and collaboration,” he said. Cohen and Mary Zutter, vice president of integrative diagnostics at the Vanderbilt University Medical Center and professor of pathology, microbiology, and immunology at the Vanderbilt University School of Medicine, stressed that high-quality diagnosis requires a team effort: “Integration of knowledge across disciplines is essential both for the patient and for the learning health care system,” said Zutter (see also the section on Integration and Collaboration of Specialties).
ENSURING THE QUALITY AND ACCESSIBILITY OF DIAGNOSTIC EXPERTISE AND TECHNOLOGIES
A number of workshop participants examined strategies to enhance expertise in oncologic imaging and pathology, including improving education and training, expert consultations, telemedicine and telementoring, and clinical decision support.
Improving Education and Training
Several workshop participants described opportunities to improve oncologic imaging and pathology through education and training efforts, including
- Greater emphasis on improved communication and sharing of diagnostic testing results;
- Additional resources and training opportunities on molecular diagnostics and pathology informatics;
- Adoption of competency-based medical education approaches; and
- More opportunities for subspecialization in imaging and pathology and for cross-discipline education.
Improved Communication and Collaboration
Siegal noted that a significant percentage of diagnostic errors are due to inadequate communication in medical care (e.g., ensuring that the diagnostic testing results from radiologists and pathologists are received by a patient and the ordering clinician). She noted that at one hospital, radiology and emergency department residents are taught “to have open communication
between two services that often see themselves as very separate.” She added that when root cause analyses17 are performed to identify how diagnostic errors occurred, having multiple specialties present a case provides powerful lessons about how communication breakdowns contribute to diagnostic error.
To strengthen communication skills, Ritu Nayar, professor and vice chair of pathology at the Northwestern University Feinberg School of Medicine and director of cytopathology at Northwestern Memorial Hospital, reported that pathology residents and fellows participate in simulations and daily clinical practice, and then receive constructive feedback on their performance. They are also taught how to prepare their reports in a standardized fashion, communicate critical results to clinicians, and learn about factors that contribute to communication gaps. Fennessy added that appropriate communication of results is also emphasized in the Cancer Imaging Fellowship Program at Brigham and Women’s Hospital/DanaFarber Cancer Institute. Fellows are required to produce templated reports that summarize their findings, as well as suggest next steps oncologists might want to take with their patients.
Interdisciplinary team approaches to cancer diagnosis and treatment are part of the program requirements for a pathology training program accreditation, Nayar noted. Trainees actively present cases and interact with radiologists, oncologists, and other health care professionals in multidisciplinary tumor board meetings, at which patient treatment planning is discussed. Residents are also expected to attend and present at inter and intradepartmental meetings and quality assurance activities in which cancer cases are reviewed.
Nayar said Northwestern has a 24/7 call center staffed by nurses to help radiologists and pathologists reach clinicians to whom they need to report their results. “We make sure that those who need to know a result get it,” she said.
Molecular Diagnostics and Pathology Informatics
Nayar reported on a survey of “new in practice” pathologists (practicing in both academic and community hospitals) that has been conducted in concert with the Maintenance of Certification (MOC) program for the
___________________
17 A root cause analysis is a structured method used to analyze serious adverse events. See https://psnet.ahrq.gov/primers/primer/10/root-cause-analysis (accessed July 13, 2018).
past 4 years. This survey asks respondents to rate 42 areas of training to determine if residency training provided adequate preparation for their practice. In the 2016 survey, most respondents reported that residency training was about right, but a number of respondents suggested training should include a greater emphasis on molecular diagnostics and pathology informatics. In response, Nayar noted that training resources in these areas are being developed and promulgated. She added that future survey results will be used to help define the core training pathologists need to practice competently; continue to identify areas where more training is needed, as well as which training areas should be removed; and determine which competencies should be part of specialty fellowships versus residency programs.
Competency-Based Medical Education
Nayar also discussed the increased focus on competency-based medical education in medical school, as well as during residency and fellowship training (Powell and Carraccio, 2018). She said competency-based medical education recognizes that not all students or trainees master skills at the same pace, and aims to create a learner-centric education system that, in turn, can promote patient-centered care. The Accreditation Council for Graduate Medical Education/American Board of Medical Specialties specify six core competencies in medical education, including patient care, medical knowledge, practice-based learning and improvement, interpersonal and communication skills, professionalism, and systems-based practice. During their training, residents and fellows receive semiannual milestone reporting on their progress in acquiring these competencies.
She also described entrustable professional activities (EPAs), which have been defined as observable, measurable units of work that require the integration of many competencies (ten Cate, 2005, 2013). “Right now, we are judging people on individual competencies based on passing their boards after a specific amount of time served in training, and we may be better off judging them through these EPAs that require integration of several competencies and correlate better with the actual work that we do,” Nayar said. The American Board of Pathology issued a position statement supporting its participation in the further development of EPAs in pathology, including consideration of a pilot project.18 Four EPAs in anatomic and clinical
___________________
18 See http://www.abpath.org/index.php/announcements/255-abp-position-statement-on-epas (accessed April 25, 2018).
pathology (e.g., a medical autopsy, intraoperative diagnosis, workup of a non-major transfusion reaction, and a clinical pathology diagnostic study) have been suggested as potential options for a pilot project (McCloskey et al., 2017). Nayar noted that the field of pediatrics is already using this approach—from medical school through residency and fellowship—and plans to evaluate how this training compares to conventional approaches. “The American Board of Pathology is committed to exploring the EPAs and working with our organizations to see how these pilots might work out and how they might affect training and certification of individuals,” Nayar said.
Pathology and Radiology Training, Subspecialization, and Integration
Nayar said most pathology residencies are 4 years long and focus on both anatomic and clinical pathology, which includes training in oncologic pathology. A small percentage of residents pursue a 3-year residency, which focuses only on anatomic pathology or clinical pathology. Nearly all residents pursue subspecialty fellowship training after residency, and 46 percent of residents complete two or more fellowships. The American Board of Pathology certifies 11 pathology fellowships, and there are a number of additional subspecialty fellowships relevant to oncology, she said.
Nayar described current residency training in oncologic pathology, including the requirement to use standardized reporting formats with tumor staging information for resected specimens: “During residency, the utilization of templates and standardized reporting is common . . . in both academic centers as well as private practice settings that residents and trainees may rotate through,” Nayar said. Residents also perform and analyze core biopsies and body fluids obtained by minimally invasive techniques for advanced disease diagnosis, prognosis, or therapeutic decision making. She added that a key to current pathology training is correlation with ancillary studies, such as next-generation sequencing, immunohistochemistry, in situ hybridization, flow cytometry, and cytogenetics.
Brent Wagner, president-elect of the American Board of Radiology and visiting lecturer at the American Institute of Radiologic Pathology, reported on radiology residency and subspecialty fellowship requirements. He noted that radiology has four disciplines: diagnostic radiology, radiation oncology, medical physics, and interventional radiology. He said for diagnostic radiology, residents are trained in 18 content areas that include both organ systems and imaging modalities. Typically, after a 1-year internship and 4 years of residency, fellows select up to three specific clinical areas in which
they intend to specialize, and take a certifying exam in these areas and in the core elements of diagnostic radiology. This certifying exam is given after the completion of a residency and/or fellowship. “At that point, you are starting to subspecialize because you are picking areas that you think will comprise your practice moving forward based on your training and interests,” Wagner said.
Boris Brkljačić, vice president of the European Society of Radiology, said the European Society of Radiology has an online learning platform19 with more than 260 learning modules that include sections on oncology imaging. According to Brkljačić, many radiologists are using these online courses to prepare for the European radiology diploma exam. The Society also provides training in oncologic imaging at the annual European Congress of Radiology. In 2011, the European Society of Oncologic Imaging20 was established to promote research activities and other advances related to cancer imaging.
Several workshop participants discussed whether pathology and radiology residencies should have a greater focus on oncology, as well as the need for subspecialty fellowships in oncologic pathology and imaging. Hricak said there is a need for specialized expertise in oncologic imaging, and there should be educational opportunities to facilitate development of this expertise, such as offering more oncologic imaging fellowships; offering oncologic imaging training as a fourth-year mini-fellowship during radiology residency or as a rotation during a fellowship; and providing peer learning and continuing medical education (CME) courses in oncologic imaging. She said there are only 5 oncologic imaging fellowships in the United States, but more than 250 types of other imaging fellowships.
Fennessy agreed that oncologic imaging fellowships are important because cancer can affect the entire body, and traditional imaging subspecialty fellowships focus on specific body areas or systems. “It is not a disease just of the chest if the patient has lung cancer, or a disease of the abdomen and pelvis if the patient has liver cancer. We need to be able to consider the body as a whole when we give a report,” she said. Consequently, the Dana-Farber Cancer Institute has moved away from organ-based or technology-based fellowships, and instead offers more disease-centric fellowships, in which fellows receive education and training in imaging and interpretation of multiple body parts and organs using different imaging modalities (see
___________________
19 See https://cslide.ctimeetingtech.com/library/esr/home (accessed April 30, 2018).
20 See http://esoi-society.org/index.php (accessed April 30, 2018).
Box 2). Fennessy noted that these oncology fellows are expected to prepare templated reports with standardized language. Hricak and Curtis Langlotz, professor of radiology and biomedical informatics at Stanford University and medical informatics director for radiology at Stanford Health Care, said
that the uptake of structured reporting in radiology has generally been slow, but there are professional society initiatives to increase their use.21
Fennessy added that radiologists also need to understand the changing paradigm of cancer care that has been brought about by precision medicine. “When we consult with our oncology colleagues, we want to have an idea of what they are talking about,” for example, with regard to the genomic basis of a cancer, Fennessy stressed. She added that radiologists need to learn and use the technical language that their oncology and pathology colleagues use.
Several workshop participants suggested that integrating radiology and pathology disciplines in clinician training programs has the potential to improve cancer diagnosis and care. Fennessy suggested that future training programs for cancer imaging should incorporate pathology information to better evaluate whether a tumor is actively growing, or whether a response to treatment has occurred. “Digitized correlation and registration with pathology and imaging will be key to figuring out what we are missing in radiology and how we can improve upon that,” Fennessy said. William Stead, chief strategy officer at the Vanderbilt University Medical Center, suggested the creation of a new medical specialty in diagnostic oncology that could provide a collaborative track for experts who work at the intersection of those two fields (Jha and Topol, 2016). He suggested creating pilot training programs, and applying lessons learned when other disciplines were combined into the same training program, such as pediatrics and internal medicine. Becich responded that he has suggested that the Society for Imaging Informatics in Medicine create a postdoctoral or research training program that combines both radiology and pathology disciplines. “I don’t know what we’ll call them yet, but we’ve got to start the revolution through an evolution in training,” Becich said. He added that 10 years ago, Yale University explored the possibility of having a common diagnostic platform for pathology and radiology training, but it was not implemented. Becich suggested that informatics could play a critical role in merging training programs by developing an overall digital imaging and analysis architecture that encompasses pathology and radiology and would require training in both. “If you put [the fields of radiology and pathology] together around common tools, then a lot can happen,” he said. “We’re going to need [these] trainees to help fuel the advances, so this is really important.”
___________________
21 See http://www.radreport.org/specialty/oi (accessed August 17, 2018).
Training for Practice in Community Versus Academic Settings of Care
Several workshop participants discussed whether subspecialization in oncologic imaging and pathology is appropriate or feasible in all settings of care (e.g., community practice versus academic medical centers or large cancer centers). James Brink, radiologist-in-chief at the Massachusetts General Hospital and chair of the American College of Radiology (ACR) Board of Chancellors, noted that the American Board of Radiology and the American Board of Pathology devise their core competencies based on the needs of the whole country, and not for one type of setting.
Schilsky asked, “What are the core competencies with respect to cancer that a radiologist or pathologist working in a community setting needs to have in order to best meet the needs of the patient population they are serving?” He added, “It’s just not going to be possible to take the kind of radiology and pathology training and practice that exist at Dana-Farber, Memorial Sloan Kettering, and the other big freestanding cancer centers and put that out in the community. I am also not convinced that it is necessary because the case mix is very different. If you work at a big cancer center, you have 100 percent prevalence of cancer in your practice. That is not the case in most of the community hospitals around the country.”
Kojo Elenitoba-Johnson, founding director of the Center for Personalized Diagnostics and director of the Division of Precision and Computational Diagnostics at the University of Pennsylvania Perelman School of Medicine, indicated that the complexity and rapidly evolving nature of genomic diagnostics in oncologic pathology requires expertise and infrastructure that only exists in larger academic medical centers. He said that the duplication of such infrastructure in community settings may be prohibitively expensive and inefficient. He suggested it might be more feasible to develop mechanisms for community physicians to refer certain patients for testing and care available at larger academic institutions and comprehensive cancer centers if they do not have the expertise or technologies available in the community setting.
Wagner noted that “most radiology care is still provided locally—within a 50-mile radius of where you are.” He added that in some community settings, radiologists and pathologists do not see enough of certain cancers to be comfortable distinguishing normal from abnormal findings. “No matter your knowledge and training, if you are only reading 200 mammograms a year, you are not going to be good at it,” he said.
Lawrence Hofmann, co-founder of Grand Rounds and chief of inter-
ventional radiology at Stanford Hospital, added that patients also need information about the availability of subspecialists, especially for cancer care that requires a high degree of specialized expertise. “Part of the solution may be enabling the patient to seek out more specialized care when appropriate,” Hofmann said. But Sause noted that insurers may not reimburse for care that is out of network.
Ellen Baker, director of Project ECHO (Extension for Community Healthcare Outcomes) at The University of Texas MD Anderson Cancer Center, noted that “we’ll never have enough subspecialists to cover the country evenly . . . there’s got to be a way to find some middle ground so that local physicians will be able to provide more [care] than they can now.” She said this is the purpose of Project ECHO, a telementoring program (see the section on Telementoring).
Cohen added that the issue is not just about whether an individual clinician has subspecialty training, but about taking a systems approach to improving patient access to oncologic pathology and imaging. Larson suggested holding practices collectively accountable using a blended model, in which specialists supervise and provide feedback and coaching to generalists. Brink asked whether merging practices could help ensure sufficient subspecialty expertise. Even if radiologists or pathologists have subspecialty training, he noted that they may not have the scale in small practices for this expertise to be leveraged. Larson responded that there has been significant consolidation of practices, not only for purposes of aggregating expertise, but also for economic reasons. As that occurs, he suspects large private practice groups will function more like academic departments, but he cautioned that heavy coordination costs are involved.
Bruce Stewart, president of the Massachusetts Radiological Society, noted that although he is “a big fan of subspecialization, there’s a sweet spot for it,” and he cautioned against having “super specialized radiologists who are not comfortable with anything outside their realm.” For example, he pointed out that a chest CT also images the kidneys, liver, and other organs in the abdomen, so radiologists who specialize in reading chest CTs also have to recognize and evaluate abdominal lesions. “Subspecialization is critical, but at the same time, you also need to have some general radiology skills,” Stewart said.
Potential Strategies to Expand Patient Access to Subspecialty Expertise in Oncologic Imaging and Pathology
A number of workshop participants described potential strategies to expand patient access to oncology diagnostic expertise, including expert consultations that can be facilitated by telemedicine or second opinion networks, incorporating subspecialty expertise within practices, facilitating Diagnostic Management Teams (DMTs), and using telementoring.
Second Opinions
Several workshop participants discussed opportunities to leverage technology and consortia to facilitate access to second opinions in oncologic imaging and pathology. Nayar said that second opinions in pathology are often sought when a cancer diagnosis is suspected (see section on Peer Learning and Second Reviews). She added that a number of institutions are starting to use real-time digital pathology tools, such as telepathology and whole-slide imaging, within their institutions, as well as for expert consultation outside their systems (Volynskaya et al., 2017). “These tools have started making their way into pathology, but are not mainstreamed yet. . . . This is what ultimately will need to happen with personalized and precision medicine as things become more and more complex,” Nayar noted. She added that in addition to real-time approaches, access to subspecialty expertise will also require referrals and the development of consultation networks for subspecialty support.
Hricak suggested forming second opinion networks and cancer imaging consortia so that those who lack oncologic imaging expertise in their own practices have an easy mechanism to request second opinions for suspected cancer diagnoses. Fennessy noted that radiologists with Brigham and Women’s Hospital read any cancer-related images generated by its community radiology division in Foxborough. “They are not familiar or comfortable with reading these cases, so we have taken them over and our oncologists prefer that,” Fennessy said. She also reported that the Dana-Farber Cancer Institute has a consultation service in which their radiologists, as part of a multidisciplinary team, are embedded in the clinic to interpret imaging in real time. “This allows for open dialogue with the clinicians, and for quality assurance of the peer reads,” she said, while decreasing interruptions in the reading room.
Hofmann reported on Grand Rounds,22 which provides second opinions and treatment decision support services to large, self-insured companies. He said companies are increasingly seeking out these services due to the high economic costs businesses incur (e.g., disability claims, absenteeism, and health care costs) when employees are diagnosed with serious conditions such as cancer and receive poor-quality or low-value care. He said approximately 20 percent of Grand Rounds second opinions are for patients with cancer. For approximately 36 percent of oncology cases, a Grand Rounds second opinion changes the treatment plan, Hofmann reported. He added that in those cases in which an in-house Grand Rounds pathologist deems that another review of the pathology is necessary, the new review leads to a change in the cancer diagnosis approximately 40 percent of the time.
Becich asked how Grand Rounds keeps up with and curates the tremendous amount of constantly evolving knowledge about cancer biology and treatment. Hofmann responded that it relies on their experts to be up to date on the knowledge related to their specialties. “We hope over time that we will have a large enough corpus of data that we will be able to automate some of these processes or disease states that are relatively straightforward, but right now we’re relying on the experts,” Hofmann said.
Oncology Expertise Within Practices and Academic Departments
Hricak suggested instituting oncologic imaging expertise within radiology departments, and she noted that this may be more feasible in large academic settings than in small community practices. In smaller practice settings, Larson suggested that practices designate local experts in oncologic imaging or pathology who can teach and provide feedback to others in their practice. Fennessy noted that community radiologists who are part of the Brigham and Women’s Hospital/Dana-Farber Cancer Institute system can participate in educational seminars via video conferencing to stay informed and receive CME credits.
But Fennessy questioned whether community practices provide adequate learning opportunities for physicians. “Those learning opportunities may be available to them, but they often are not as aware of them. They may attend CME courses, but that does not necessarily keep them up to date, which is problematic. Potentially, you could champion a leader in those
___________________
22 See https://www.grandrounds.com (accessed April 30, 2018).
community divisions, but that person really needs to [receive] dedicated education and know how to keep up with what is new and have a formal plan. It cannot just be the assigned person in a practice because that would get lost pretty quickly,” Fennessy said. Hricak agreed, noting that an oncologic imaging fellowship is not sufficient over time, given the fast pace of cancer research. She said organized ways to ensure clinicians are keeping up with the rapidly changing knowledge base are needed.
Diagnostic Management Teams
Zutter said the complexities involved in cancer diagnosis have challenged the traditional paradigm of pathology testing, in which a clinician orders a test, a lab performs the ordered tests and returns results, and an individual clinician collates and interprets the information. She noted that there is a large, complex, and rapidly expanding menu of potential diagnostic tests for clinicians to consider and few evidence-based guidelines to help with test selection. In addition, numerous laboratories may be involved in testing, and they might provide a clinician with multiple reports, which may be difficult for clinicians to collate and interpret.
To ensure that patients receive the correct diagnostic testing and that clinicians are supported in test ordering and interpretation, Zutter said that Vanderbilt developed DMTs through a collaboration among pathologists, clinicians, and biomedical informaticians. The goals of this program are to:
- Develop the right pattern of diagnostic testing for a patient, using standard test ordering algorithms;
- Create a single, evidence-based, comprehensive report of integrated diagnostic data to guide treatment and disease monitoring; and
- Iteratively improve the algorithms as evidence-based practices evolve.
Zutter said clinicians have the option to order hemato-malignancy testing individually or via DMT, but she has found wide acceptance of the DMT approach. She added that DMT guidance is estimated to save clinicians approximately 10 minutes per patient, and ordering clinicians are reassured that the correct tests will be ordered (Seegmiller et al., 2013). “We have physician support and enthusiasm because we are not costing them any extra time,” Zutter noted.
A retrospective analysis found that DMT guidance reduced unnecessary testing, improved test concordance (consistency with standard test ordering algorithms), and resulted in lower costs (Seegmiller et al., 2013). If this approach were scaled nationally, she said it could result in an estimated savings of up to $500 million per year (Seegmiller et al., 2013). Improvements in the DMT standard test ordering algorithms continued to improve test utilization and performance, and also lowered costs (Seegmiller et al., 2016). When next-generation sequencing was incorporated into the DMT standard ordering algorithm for suspected diagnoses of myeloid malignancies, clinicians who used DMT guidance had a positive test concordance rate of approximately 81 percent, compared to approximately 18 percent for the clinicians who ordered next-generation sequencing testing a la carte. “This suggests the use of these [standard ordering algorithms] is truly remarkable in improving testing and ultimately, patient care,” Zutter said.
Vanderbilt recently started offering DMT guidance at affiliated health centers, such as Jackson Memorial Hospital. Pathologists continue morphology analyses at their own institutions, but Vanderbilt performs the more complex testing and assembles comprehensive reports. “It’s saving them money because we are doing so many fewer tests than when they were sending their lab testing out to a variety of other larger commercial laboratories,” Zutter said. Vanderbilt has also recently developed DMT standard ordering algorithms for colorectal cancer and is in the process of implementing this DMT. Plans are under way for DMTs for other gastrointestinal malignancies, as well as breast and lung cancers. “What we are doing is an example that can scale much more broadly to other disease processes,” she said.
Telementoring
Baker reported on Project ECHO,23 a teleconsulting and telementoring partnership to disseminate knowledge and build capacity in regions lacking specialty or subspecialty medical care. “We can’t duplicate what’s available at the major cancer centers in every community in America, but there is some middle ground where we can improve what is currently available and provide resources to these communities so patients get better care and physicians have more support,” Baker said. She noted that Project
___________________
23 See https://www.mdanderson.org/education-training/global-outreach/project-echo.html (accessed April 30, 2018).
ECHO was developed in 2003 by Sanjeev Arora, a gastroenterologist at the University of New Mexico, as a way for patients with hepatitis C in rural parts of the state to receive the complex specialized care they needed from primary care clinicians. Arora formed a partnership with 16 rural clinics and 5 prisons in New Mexico, for which he served as a mentor, providing case-based guidance in weekly teleconferences in order to provide the support needed to care for patients in their own communities. Baker added that an evaluation of Project ECHO found that patients treated with the telementoring support had similar outcomes compared with patients who were treated at the University of New Mexico (Arora et al., 2011).
Baker stressed that Project ECHO is not telemedicine, which involves long-distance interactions between patients and clinicians, but rather clinician-to-clinician telementoring, which enables specialists to disseminate their knowledge to partners in the community who can then treat more complex patients. “The goal is to take knowledge that usually sits at the academic center and share it with the rest of the medical community, particularly in areas that don’t have that specialty or that knowledge. By doing this, you build capacity outside of major medical centers to treat patients with complex conditions safely and effectively in the community without the need to refer them out,” Baker said. “The goal is to move knowledge and not patients,” she said, and added that telementoring can help reduce disparities in access to quality care.
The four pillars of Project ECHO are harnessing videoconference technology, sharing best practices, employing case-based learning, and monitoring outcomes (Arora et al., 2007). The University of Texas MD Anderson Cancer Center adopted Project ECHO 4 years ago to help underserved communities manage cervical dysplasia, a precursor to cervical cancer, Baker reported. Project ECHO has been expanded to other aspects of cancer care, including prevention, treatment, palliative care, and cancer survivorship care for several different types of cancer at a number of underserved regions worldwide. There are currently more than 170 Project ECHO hubs for more than 65 diseases in 23 countries, she said.
Baker noted that one of the challenges in implementing Project ECHO is the need to identify champions both in academic centers and in the community. To participate in Project ECHO, community clinicians need to take time away from direct patient care, “so you have to find somebody who is really passionately interested in moving up a notch and being able to provide services they were not previously able to provide. Once you get
that community champion, they tend to be able to bring others in with them,” Baker said.
Telehealth
Sause said that Intermountain Healthcare—which delivers health care through a network of 22 hospitals in Utah and southern Idaho, and is an insurance provider—originally began offering telehealth services to seven community hospitals serving large geographic areas with intensive care units staffed by non-intensivists. After the telehealth program to support these intensive care units was instituted, Sause said that patient outcomes improved, including a reduced risk of mortality.
Intermountain Healthcare has expanded its telehealth program to the oncology setting as well by tapping into system resources and expertise, including subspecialty expert opinions, in order to deliver cancer care in four outpatient facilities in rural settings. Patients at these facilities have participated in more than 500 telehealth visits, and report a high degree of satisfaction with the program, Sause reported. He said telehealth can augment the capacity of a generalist, and also supplement care in communities that have insufficient numbers of specialists.
Clinical Decision Support
A number of workshop participants discussed clinical decision support tools in cancer care. Ramin Khorasani, professor and vice chair of radiology at Harvard Medical School and co-founder and director of the Center for Evidence-Based Imaging at Brigham and Women’s Hospital, described clinical decision support as the iterative interaction of a clinician with a computer to improve clinical decision making by reducing unwarranted variation in care and improving adherence to what is known to benefit patients. For example, he highlighted the uneven use of CTs and MRIs nationwide among Medicare beneficiaries (Ip et al., 2015). Even within his own institution, he found wide variation on follow-up recommendations among individual cancer imaging specialists. “We need to be able to measure performance and assess whether our practitioners are adhering to evidence to reduce unwarranted variations in care,” Khorasani said, stressing that clinical decision support encourages evidence-based practice, which is an essential attribute of high-quality care, as defined by the Institute of Medicine (IOM, 2001).
Brian Shirts, assistant professor of laboratory medicine at the University of Washington, defined clinical decision support as “knowledge and person-specific information, intelligently filtered or presented at appropriate times, to enhance health and health care” (Osheroff et al., 2007, p. 141). Gregory Riely, medical oncologist at the Memorial Sloan Kettering Cancer Center, stressed that clinical practice guidelines, such as those developed by the National Comprehensive Cancer Network and ASCO, are the underpinnings for clinical decision support.
Shirts said decision support can help clinicians to appropriately interpret and act on diagnostic testing results. Jordon Laser, senior director of Cytogenetics & Molecular Pathology at Northwell Health, agreed and added that the purpose of diagnostic clinical decision support is to provide clinicians with sufficient and accurate information to order the right test at the right time for the right patient. In imaging, Khorasani said that decision support tools can be used when clinicians place orders for imaging, at the time of image analysis and interpretation through tools such as computer-aided detection or diagnosis, and at the time of results reporting, to help improve recommendations for additional imaging.
Shirts said clinical decision support tools can either be passive (requiring user effort to access or interpret) or active (pop-ups or flags that are triggered by specific situations). Examples of passive clinical decision support are drug dosing calculators, disease risk calculators, and web addresses to view additional information, but he noted that passive clinical decision support may be accessed infrequently (Shirts et al., 2009). For clinical decision support tools to be effective, Khorasani said they have to be efficiently embedded into a clinician’s workflow, reduce redundant data entry, and provide brief, actionable, and unambiguous advice (Bates et al., 2003; Khorasani et al., 2014). In addition, Khorasani said these tools should encourage or enforce clinicians to adopt evidence-based practices, and the strength and source of the evidence should be transparent to the clinician. He added that decision support tools should minimize “low-value” alerts that exacerbate alert fatigue.
Several speakers said it was essential to collaborate with patients in designing decision support tools, and to incorporate PROMs within these systems. “It’s really important to engage patients in making these recommendations applicable to the care of patients because not everybody prefers the same treatment, and patient preferences are not embedded in our guidelines and rules today,” Khorasani said. He noted that most EHRs provide only the clinician’s perspective on patient presentation. He sug-
gested collecting patient-generated information on signs and symptoms by having patients fill out an electronic form while waiting to be seen by their clinicians. “Can we partner with patients in this way so they can help us make decisions?” he asked. Riely agreed that this approach is critical to improving patient engagement, and pointed to a review concluding that PROMs appear to improve the quality of care delivered to patients with cancer (Basch et al., 2017).
Brink added that clinical decision support can be useful in explaining why a clinician may suggest a certain course of action. He gave an example of a patient who wanted an MRI to assess her lower back pain. The clinician showed the patient the computer screen with the decision support tool indicating that ordering such imaging was in the “red zone” and not recommended by clinical practice guidelines. “If you really bring it back to making an informed decision with patients and shared decision making, that can help promote cultural acceptance of decision support,” he said.
Khorasani noted that evidence on the effectiveness of clinical decision support tools is beginning to be published. Evaluations of clinical decision support tools linked to the goals of the Choosing Wisely®24 initiative to reduce unnecessary testing have found decreased use of imaging for suspected pulmonary embolisms, lower back pain, and minor traumatic brain injury. “We have had fairly significant impact in targeted interventions across many of our leading- edge institutions but these findings haven’t been replicated broadly across the country yet,” he said.
Challenges with Developing and Implementing Clinical Decision Support Tools
A number of workshop participants described challenges with clinical decision support tools, including clinician acceptance, a lack of interoperability within and among health systems, and development and maintenance costs.
Shirts said clinician acceptance is one of the largest barriers to implementing clinical decision support, noting that one review found clinicians override drug safety alerts approximately 50 to 90 percent of the time (van der Sijs et al., 2006). Brink stressed that clinicians will reject decision support if it increases their work burden and will create work-arounds if they do not believe it to be useful. Khorasani agreed and added that electronic medical records and clinical decision support are major drivers of
___________________
24 See http://www.choosingwisely.org (accessed May 1, 2018).
clinician burnout because they can cause distractions, interruptions, and inefficiencies (Noseworthy et al., 2017). “They are getting in the way of our physicians taking care of their patients,” Khorasani stressed.
Becich said most EHR systems are really billing systems. He stressed that EHR vendors should better design EHRs for patient care: “We need to compel the electronic health record companies to build decision support options directly into their systems and support them,” he said. Riely emphasized that translation of clinical practice guidelines into clinical decision support tools “is not something that can be handled by a software engineer. It requires physicians at every step, but there aren’t a lot of physicians trained to do this.” Becich suggested that physicians who are involved in developing clinical decision support tools should obtain a board specialty in informatics.
“Before we implement clinical decision support, we really should get physician buy-in,” Shirts said. He noted that if clinical decision support tools are specific, based on clinical evidence, and well designed, they are likely to foster greater clinician acceptance (Hartzler et al., 2013). Khorasani added that clinicians are also more likely to appreciate and use decision support tools if they are tailored to their specialty. “The information you’re putting in front of the person you’re trying to influence must fit their practice,” Khorasani said.
Brink added that implementation of clinical decision support will require culture shifts championed by clinical practice and hospital leadership. Khorasani agreed, adding, “Probably the most important part of [this] is leadership. If there is no leadership in the institution, it’s going to be really hard to implement decision support because these are cultural changes . . . without leadership there to want and expect change, it’s really a challenge to get some of these programs into place.”
Elenitoba-Johnson added that improved alignment among payers, regulators, and professional societies is also needed to increase uptake of clinical decision support. Because clinical decision support tools can facilitate the insurance preauthorization process for genomic testing and reimbursement, it can improve clinician acceptance and use of decision support. He added that implementation of clinical decision support tools can also foster acceptance by payers to reimburse complex genomic testing in appropriate clinical contexts.
Stewart asked whether clinicians might resist clinical decision support due to concerns about medical malpractice. For example, he wondered whether a clinician would be liable if a patient with back pain turned out to
have cancer, but the clinician did not order imaging because it was not indicated by the decision support tool. Khorasani responded that his institution has used clinical decision support for more than two decades, and there has not been a single case of medical malpractice due to following recommendations of the decision support system. Instead, he said that following clinical decision support recommendations could be used to defend against medical malpractice suits. However, this is dependent on a high-quality decision support tool and ensuring that the information entered into the system is “precise and of good integrity, and that the alert content is representative of the patient. That’s why the clinical relevance of these alerts is crucial when designing and deploying these systems,” Khorasani said.
Shirts said another challenge with clinical decision support is a lack of interoperability, or the “ability of different information technology systems and software applications to communicate, exchange data, and use information which has been exchanged” (HIMSS, 2018). Shirts noted that even within a single institution, health information systems are complex, with multiple different interfaces communicating different types of health information from different sources (see Figure 1). Sharing information
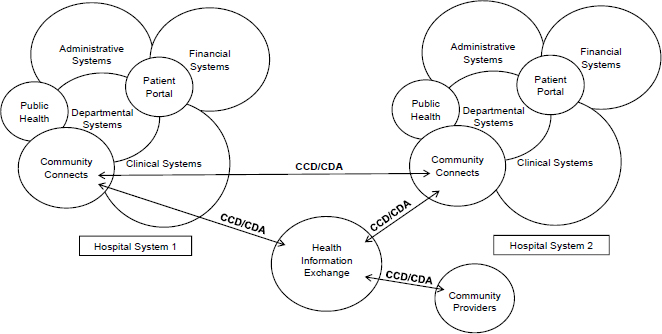
NOTE: CCD = continuity of care document; CDA = clinical document architecture.
SOURCE: Created by David Chou, Department of Laboratory Medicine, University of Washington; provided for Shirts presentation, February 12, 2018.
among health care systems can be even more challenging because there are few incentives to share data. “So you could have a clinical decision support that works very well for a patient who has been at your institution for many years and has had all [of his/her] health care at your institution, but that isn’t necessarily always the case. It’s difficult to have good clinical decision support for any type of pathology or molecular diagnostics that goes across institutions,” Shirts stressed. But he added that interoperability “is not an impossible task and many organizations are working toward it and that should be supported.” A workshop participant, Muieen Cader, suggested holding case competitions or hackathons to bring engineers and clinicians together to solve challenges with interoperability and clinical decision support.
Shirts pointed out that creating and maintaining a clinical decision support system can be extremely costly. These costs include the work of committees deciding on the clinical decision support rules, the alerts that should be included, and how the system should be designed; the time and effort to adapt the system based on pilot testing and clinician feedback; and costs to make the decision support system interoperable. “The costs of building and maintaining clinical decision support networks are often ignored or minimized; however, these costs can be substantial, especially if clinical decision support is implemented independently at each health care institution,” Shirts said.
For a genomic clinical decision support system at the University of Washington, Shirts said the estimated cost of communicating genomic information support to a clinician at the point of care was $4,600 per alert generated (Mathias et al., 2016). Shirts added that computer maintenance and system updates were expected to add 20 percent to the cost of developing the initial system per year that the system was operational, because maintenance can involve completely rebuilding the system every 3 or 4 years, or completely rebuilding decision support rule libraries to keep these interoperable with other systems. “It’s very challenging to keep systems connected with each other and to keep them up to date with current guidelines,” Shirts said. Every time a new guideline comes out, it must be translated into a computable language. “It would be wonderful if the guidelines could come out in a machine-readable format,” he added (IOM, 2015).
Shirts said that dramatic improvements in data interoperability and interinstitution collaboration will be necessary to drive decision support costs down (Mathias et al., 2016). In order to increase efficiency and reduce cost to less than $100 per alert, “we need to spread our efforts across mul-




















