3
Implementing Reusable Elastomeric Respirators in Health Care Settings: Routine and Surge Use
This report examines two distinct circumstances in which reusable elastomeric respirators could be considered for use in health care settings: routine use and surge use (defined in Chapter 1). Building on the evidence provided in Chapter 2, this chapter discusses the issues surrounding the implementation of elastomeric respirators in health care settings.
DATA ON THE EXTENT OF ROUTINE AND SURGE USE AND SUPPLIES OF RESPIRATORS
Few data are available on the extent of routine and potential surge use of all types of respirators in health care. A study at Fraser Health Authority in British Columbia, Canada, noted that prior to the H1N1 pandemic the health authority had used approximately 1,440 disposable filtering facepiece respirators per week (approximately 75,000 per year) and that during the peak of the pandemic more than 19,000 of the respirators were being used each week, an increase of more than 13-fold over the baseline (Ciconte et al., 2013). A 2012 survey by the Association of State and Territorial Health Officials (1,066 hospitals completed the survey) estimated that almost 60 million N95 filtering facepiece respirators and approximately 75,000 powered air-purifying respirators (PAPRs) were on hand at the 1,066 acute care hospitals that responded (ASTHO, 2014). The survey did not measure inventories of reusable elastomeric respirators. The respirator used predominantly in health care settings is the disposable filtering facepiece respirator. In a survey of occupational health professionals in health care facilities, 94 percent of the facilities reported using this type of respirator, while 78 percent reported use of PAPRs, and 31 percent re-
ported some use of elastomeric respirators (usually by fewer than 10 employees) (Wizner et al., 2016). Elastomeric respirators are not typically used by workers involved in direct patient care, but rather are used primarily by the grounds-keeping, chemical spill response, and maintenance teams in health care facilities (Brown et al., 2017; Gribogiannis, 2018).
Some focal shortages of disposable filtering facepiece respirators were reported during the H1N1 pandemic in 2009 that required a release of supplies from the Strategic National Stockpile (SNS). Because the magnitude and severity of future pandemics are unpredictable, epidemiological models are used to estimate the potential surge use rates. Emergency stockpiling models base their assumptions on moderate-to-severe pandemic scenarios. Models have been developed to estimate the likely extent of the potential rates of use of respirators during a surge situation and to compare the total cost of various types of respirators and respirator-related products (e.g., filters, batteries); these models include not only product cost but also stockpile storage and replacement costs but do not factor in use and maintenance costs (see discussion later in this chapter on total cost). A model by Baracco and colleagues (2015) projected the stockpiling need for respirators as well as the range of costs; this model estimated that during a severe pandemic there would be 6.1 million contacts between health care providers and patients for every 1 million members of the population and these contacts would require more than 6 million single-use filtering facepiece respirators or slightly more than 10,000 elastomeric respirators. The details of the analysis are provided in Table 3-1. These numbers, extrapolated to the U.S. population of approximately 320 million individuals, imply an estimated need of 1.95 billion disposable filtering facepiece respirators or 3.4 million reusable elastomeric respirators. The authors concluded that the least costly stockpiling strategy would involve “reusable elastomeric respirators and/or disposable respirators with an extended use/reuse policy” (p. 317). They noted that because few health care facilities currently use elastomeric respirators, issuing those respirators plus fit testing them and training individuals to use them would take considerable time and effort.
An analysis by Carias and colleagues (2015) developed three respirator distribution scenarios built on the assumption that in an influenza pandemic 20 to 30 percent of the U.S. population would become ill and require health care. In their base case distribution scenario, the overall demand for respirators remained proportionate to the number of patients, except during the peak of the pandemic. This scenario implied a need for 1.7 to 3.5 billion N95
TABLE 3-1 Annual RPDa Pandemic Stockpiling Costs for a Population of 1 Million, per Strategy
| N95b Single Usec | Elastomericd | PAPRe | Mixedf Single-Use | Extended Useg | ||
| N95 | Mixed | |||||
| Number of RPDs | 6,112,500 | 10,612 | 2,653 | N95: 2,791,500 | 1,222,500 | N95: 558,300 |
| Elastomeric: 5,766 | Elastomeric: 5,766 | |||||
| RPD acquisition cost, per year (thousand $) | 306–800 | 69–122 | 17,889–18,048 | 177–429 | 61–159 | 65–139 |
| Warehouseh and managementi cost, per year (thousand $) | 207 | 5 | 455 | 97 | 42 | 22 |
| Annual cost of RPD stockpile (thousand $) | 512–1,001 | 74–127 | 18,343–18,502 | 274–526 | 87–160 | 103–201 |
aRPD indicates respiratory protective device, including accessories.
bN95 indicates disposable N95 respirators ($0.25–$0.65).
cSingle use indicates one disposable respirator for every health care personnel patient contact.
dElastomeric indicates reusable half-face respirator ($25–$50).
ePAPR indicates powered air-purifying respirator ($500–$800).
fMixed indicates elastomeric for physician and registered nurse contacts and N95 for all other contacts.
gExtended use indicates 5 uses or 2.5 hours per disposable respirator.
hWarehouse indicates warehouse lease $7/square foot; utilities $3/square foot; pallets stacked 2 high; shelf life of 5 years.
iManagement indicates one full-time-equivalent employee/10,000 square feet of warehouse space; salary and benefits $80,000/year.
SOURCE: Reprinted with permission from Baracco et al., 2015.
filtering facepiece respirators, with other scenarios implying a need for up to 7.3 billion of those respirators. The researchers looked at a demand-reduction strategy that included the use of reusable elastomeric respirators; this reduced the estimated need for N95 filtering facepiece respirators (in nursing homes and other locations) to 48 to 154 million. These estimates all emphasize the critical need for preparedness planning.
STAKEHOLDERS IN IMPLEMENTATION
The perspectives of the key stakeholder groups—health care workers, health care employers and managers, manufacturers and suppliers, and governing and professional agencies and organizations—were considered by the committee. Figure 3-1 illustrates many of the respirator-related factors associated with each of the stakeholders collectively.
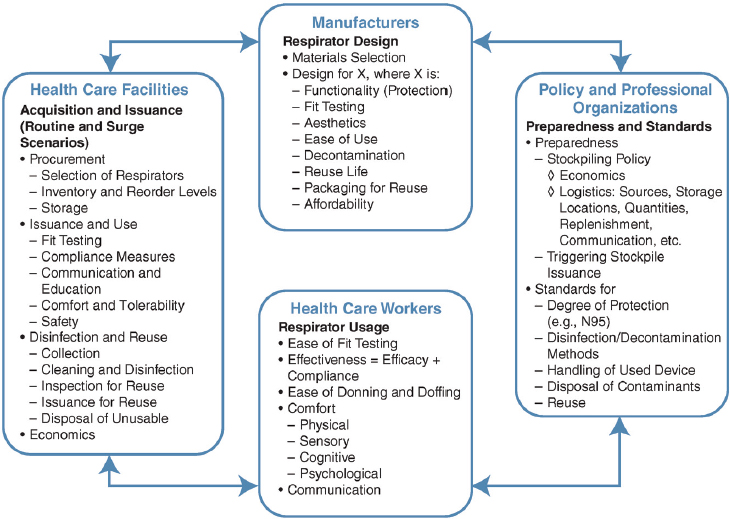
Health Care Workers: Usage and Compliance
The effectiveness of reusable elastomeric respirators in the field is determined by both the efficacy of the respirator and how correctly and consistently they are used by health care workers. Because most health care workers use respirators infrequently, their lack of familiarity makes it difficult to ensure that the procedures for correct use are followed, particularly those related to donning, doffing, user seal checks, cleaning, and maintenance of the respirator. This challenge is heightened for clinicians who move among offices and patient settings, as each facility may be using a different brand or model of respirator, each with specific requirements. Other factors influencing use are comfort, the ability to use the respirator in conjunction with other medical devices such as stethoscopes, the respirator’s interference with communication, the ease of fit testing, and the ease of use of the device as well as the overall safety culture and perceived risk (i.e., perceived need for self-protection).
Health Care Facilities, Employers, and Managers
Decisions on the selection and purchase of respirators for routine use in health care facilities often involve inputs from staff with expertise in infection prevention and control, occupational health, and industrial hygiene as well as from the purchasing and value analysis teams. Additionally, compliance with the use of respirators by health care workers is driven not only by the regulations and by the types of respirators that have been purchased and are available but also by the safety culture and climate of the health care institution, which largely emanates from the leadership and management.
Making decisions about stockpiling respirators for a potential infectious disease emergency is challenging. Although health care facilities are expected to protect their workforce during an emergency situation, there are no specific requirements about the nature and amount of supplies, including personal protective equipment (PPE), that need to be stockpiled. Furthermore, there are no clear delineations of responsibility for maintaining stockpiles of specific types of PPE among individual facilities and state and federal public health authorities. Facility leadership must decide whether to incur considerable costs in the face of uncertainty about the potential threats and a lack of clarity concerning the availability and supply of respirators from state and federal stockpiles.
Manufacturers and Suppliers
The design of respirators significantly influences their use. Even the most “protective” of devices is not effective if it is not comfortable for the user. Physical, sensory, cognitive, and psychological factors play a role, as do the respirator’s functionality (degree of protection), the ease of fit testing, requirements for decontamination or other maintenance, and affordability.
Policy and Regulatory Agencies and Organizations
Regulatory and certifying agencies (particularly the Occupational Safety and Health Administration [OSHA], the National Institute for Occupational Safety and Health [NIOSH], and the U.S. Food and Drug Administration [FDA]) and guidance and policy organizations (including the Centers for Disease Control and Prevention [CDC], the World Health Organization, and professional associations) play crucial roles in defining the standards that govern the approval of respirators and delineating the standards of use. However, in past public health crises there has been inconsistent communication on pertinent standards and inconsistent guidance on respiratory protection for health care workers for specific emerging infectious threats (Sheets and Payne, 2014).
TRAINING AND EDUCATION
Several opportunities are available for health care workers to learn about the use and care of respirators. However, the extent to which training or education is made available in public health and health care and the expectations and priority level given to respiratory protection can vary widely throughout the careers and job experiences of workers as well as among health care employers and front-line supervisors. The committee considered three types of training and education opportunities: professional education, job-based training during routine health care, and just-in-time training during emergency situations. The committee heard few examples where health care workers received training in the use of reusable elastomeric respirators; those who have been fit tested in their training programs or on their jobs are often only trained in the use of disposable
filtering facepiece respirators. The committee was asked to discuss an educational campaign for health care workers and the following section highlights ways in which a multifaceted educational effort could be conducted.
Professional Education
Studies by the American Association of Occupational Health Nurses have identified competencies and related educational information on respiratory protection which are specific for occupational health nurses for program development (Burns et al., 2014; Pompeii et al., 2016). Rogers and colleagues (2013) conducted a CDC-funded educational and practice intervention project and identified respiratory protection competencies for all health care workers (see Box 3-1). These competencies were then sent to clinicians and management in 18 hospitals in North Carolina asking them to validate the adequacy and importance of each respiratory protection competency on a 5-point Likert scale (5 = high). All competencies were validated from 4.5 to 5.0 (Rogers et al., 2018).
These competencies are practice driven and can be used as a guide to develop educational curricula and resources both in academic and in continuing education venues to ultimately help improve practice and develop respiratory protection policies. OSHA, NIOSH, and CDC have numerous resources available to support content development related to the OSHA respiratory protection standard and these competencies.
While there is some information available about continuing education focused on respiratory protection and aimed specifically at occupational health nurses (Pompeii et al., 2016), there is limited information concerning the academic content provided to occupational safety and health professionals in NIOSH-funded Education and Research Centers (IOM, 2011a). Furthermore, there is a paucity of information about academic and continuing education programs for respiratory protection for the wide array of health care workers who provide services to patients with potentially infectious respiratory diseases. The committee determined that there are knowledge gaps in training assessments for the broader community of health professionals and for settings outside of acute-care hospitals.
How do health care workers at risk of respiratory infectious agent exposure get adequate and updated information on respiratory protection, and how is this monitored in terms of effective training? Clearly there is a need to study and document the respiratory-protection-related content provided in health science schools and through continuing education.
Health professional schools (including clinical, public health, and industrial hygiene programs) and accrediting agencies should consider outlining where in their curricula they prepare students to assess potential exposure threats and to use appropriate respiratory protection. Furthermore, professional organizations should incorporate respiratory protection concepts into continuing education curricula, and professional certification bodies should include respiratory protection in certification examinations. Such content should address the array of available respiratory protection, including disposable filtering facepiece respirators, PAPRs, and reusable elastomeric respirators.
On-the-Job Training During Routine Health Care
It is essential that workers at risk of exposure to respiratory infectious agents have a fundamental understanding of the work-related exposures that can result in respiratory infections and illness and of the importance of respiratory protection and respirator use. Each health care facility that uses respirators is mandated to have a respiratory protection program (described in Chapter 1). The training associated with the OSHA requirements is often conducted in association with annual fit testing and is specific to the respirators available at that facility. This is an opportunity to educate workers about the range of respiratory protective devices, including reusable elastomeric respirators, particularly if elastomerics are an option that would be used by the health care facility in the event of an emergency. Ensuring that the trainers are knowledgeable about the demands of clinical care and the use of respirators in health care settings is essential.
On-the-job training also includes staff education to prepare for and plan for public health emergencies, and it offers another opportunity for the staff to learn about elastomeric respirators as relevant to the facility’s emergency response plan. Using “practice champions” has been shown across many work settings to be an effective mechanism to improve on-the-job training, and practice may be an effective tool for respiratory protection programs and their implementation (Rogers et al., 2009, 2018; Shaw et al., 2012).
The effects of training that are specific to elastomeric respirators have not been extensively evaluated. In the aforementioned study of elastomeric use in three hospitals in British Columbia, the researchers reported that
As [elastomeric respirators] are reusable, their use requires that cleaning, reprocessing, and equipment maintenance activities be conducted. Subsequently the education and training portion of fit testing sessions is more time-consuming, requiring an additional 10 to 15 minutes to complete. (Ciconte et al., 2013, p. 23)
Just-in-Time Training
During a surge scenario, health care facilities will often confront two issues related to respiratory protection:
- The type of respirator available from the health care facility’s backup supply or arriving from the public health stockpile—including elastomeric respirators or any brand/model of disposable respirators in the stockpile—will likely differ from the device used routinely at the facility; and
- Additional employees who were not already included in the routine respiratory protection program will need to be incorporated into the response.
The combination of these two issues means that during a surge there will be a large and acute need for just-in-time training—including training on fit testing, proper use, and proper disinfection, storage, and disposal—at a time of limited time and resources. Pre-training individuals who may participate in a potential pandemic response would familiarize health care workers with respirator use and to some extent mitigate the just-in-time needs, but such pre-training would be very resource intensive.
DECISION MAKING AND IMPLEMENTATION ISSUES
The committee explored a number of implementation issues that arise from the nature of health care work, relevant policies and practices, and the current design of reusable elastomeric respirators. These issues include
- Storage, cleaning, and disinfection;
- Medical clearance, fit testing, and respirator issuance;
- Procurement and supply logistics and emergency stockpiles;
- Safety culture and risk perception; and
- Other issues, particularly outside of hospital settings, regulatory and policy issues, and guidelines.
Storage, Cleaning, and Disinfection
The logistics of respiratory protection are complex because of the nature of health care. Health care workers spend large percentages of their work hours caring for and interacting with multiple patients who have varying health conditions and who are in a number of separate rooms or other settings. The vast majority of these interactions do not require the use of a respirator, except in specific units (such as pulmonary units) or specialized facilities (such as tuberculosis hospitals). A study by Cohen and colleagues
(2012) of seven patient care units in three hospitals, found that members of the nursing staff visited an average of 4.5 different patients per hour, while members of the medical staff visited an average of 2.8 different patients per hour. Additionally, many workers (e.g., respiratory and occupational therapists, physicians, nurse practitioners) have patients in multiple units of the facility. Furthermore, physicians and other clinicians and health care workers may be in and out of several office and clinic locations, or they may practice in one or more hospitals or nursing homes—each of which may have a different risk profile, respiratory protection program, and choice of respirators. Other health care workers deliver food or clean numerous rooms across the facility. The logistical issues of respirator use are also complicated by the fact that in most health care settings the use of respirators is rare in routine day-to-day health care. Staff may work infrequently with patients whose conditions necessitate respiratory protection use.
All of the complexities make it necessary to ensure (through infection prevention and control protocols, such as hand hygiene and contact or respiratory precautions) that the transmission of infectious agents is prevented and that cleanliness and disinfection are implemented in the most streamlined and effective manner possible. Several of the logistical issues—in particular, cleaning and disinfection—are specific to reusable respirators, and these need to be considered in the cases of both routine use and surge use.
Storage and Transport
A health care facility that decides to use elastomeric respirators would have several options for how those respirators could be distributed to the staff members who need them. An elastomeric respirator could be assigned to an individual or could be available for the health care worker to select each day from a cart or other central location. Either approach poses challenges. The Texas Center for Infectious Disease (TCID), which focuses on care of patients with tuberculosis, is one of the few hospitals in the United States that uses elastomeric respirators exclusively and routinely (see Chapter 2); each staff member there is assigned a respirator and provided with a shoulder carrying bag to transport and store the respirator (Joint Commission, 2014; Kizilbash et al., 2018). The center has found that this is a workable solution because the staff members, who are regularly coming into contact with patients with tuberculosis, must use respirators frequently. For health care facilities in which the usage of respirators is
infrequent (possibly a few times per year) and unpredictable, asking health care workers to always carry this equipment with them while on duty would be cumbersome and could result in improper storage and maintenance of the respirator. A study of the use of elastomeric respirators in three acute-care hospitals in British Columbia reported that storing on the unit
can be challenging as there is very limited counter space and storage at the nurse’s station outside the patient room. Due to similar reasons, it was challenging to identify and dedicate space for storing respirator supplies in both the clean supplies and soiled utility rooms. (Ciconte et al., 2018, p. 18)
From a warehousing perspective (i.e., storage prior to use), elastomeric respirators have both advantages and disadvantages. While the elastomeric respirators are bulkier and take up more space per unit in storage than the filtering facepiece respirators, far fewer of the elastomerics are required to meet pandemic needs. In the model developed by Baracco and colleagues (2015), a box of 10 elastomeric respirators was estimated to take up a space of 7 × 13 × 18 inches of storage pallet space (with additional space for boxes of filters), while a box of 20 disposable respirators took up 12 × 6 × 6 inches of space. Meeting the pandemic planning needs for a population of 1 million was estimated to require 6,112,500 disposable filtering facepiece respirators, with warehouse and management costs of approximately $207,000 per year (not including acquisition costs), compared with only 10,612 elastomeric respirators, with warehouse and management costs of $5,000 per year. The analysis also included PAPRs, which had larger boxes and higher warehouse and management costs. Additional information on the study is provided in a later section in this chapter on stockpile issues.
Considerations Regarding Routine Use If the decision is made to use reusable elastomeric respirators for routine health care at a facility, it will be necessary to develop a storage and transport system. The storage and transport system will need to become part of the initial staff training on respirators and also need to be incorporated into refresher training. Health care facilities that make the decision to use reusable elastomeric respirators routinely will need to have a staff whose members are fit tested, trained, and familiar with elastomerics, which will make it easier to move into a surge situation if needed, where the elastomeric respirators may be used more extensively.
Considerations Regarding Surge Use If a health care facility decides to have elastomeric respirators as part of its backup supply but not to use them routinely, then quickly implementable and frequently practiced plans need to be in place that establish, on a very practical basis, how issues of space, transportation, location, etc., will be resolved. As previously discussed, storage, transport, and administrative procedures will need to be part of the just-in-time training. For U.S. health care organizations to rapidly convert to elastomeric respirators in a just-in-time fashion during a public health emergency, the committee believes that greater clarity will be needed regarding the responsibilities for stockpiling and the specific contents of the SNS as it relates to respirators.
Cleaning and Disinfection
As discussed in Chapter 2, effective and easy-to-implement cleaning and disinfection processes and protocols are needed. Much remains to be learned about the most effective cleaning and disinfecting agents and processes for influenza viruses and other potential pathogens. Cleaning and disinfecting processes need to be standardized across manufacturers, with special attention paid to the cleaning and disinfection of the respirator between patients and at the end of the work shift. Research on the materials and protocols for disinfection and other new avenues of relevant research (discussed in Chapter 4) should address the ability of the pathogen to live on the surfaces of the respirator and potentially infect those who come in contact with the respirator. Furthermore, protocols for cleaning and disinfection are needed that account for the nature of the health care environment and that are practical to implement with limited space and time. Manufacturers’ instructions for use offer cleaning protocols that are unique to each product, are often time consuming and burdensome, and are unclear on the appropriate frequency required for disinfection as it relates to health care (after each patient, after each doffing, after each shift, etc.) (see Chapter 2).
The initial implementation of cleaning and disinfecting protocols will be challenging. If the cleaning and disinfection is to be done by individual health care workers on their units, there will be challenges in finding the space for these efforts and also in setting up and maintaining the cleaning and disinfecting stations. If the cleaning and disinfection are to be done in a centralized reprocessing facility, challenges can arise in transporting the respirators to the central location and in storing the clean respirators, as noted in the study in British Columbia (Ciconte et al., 2013). Key issues
to take into consideration when selecting a cleaning and disinfection method include material compatibility (including straps and filters), the safety and availability of the disinfecting products, the ease and time requirements of the procedure, and the space needs for the reprocessing procedure.
Considerations Regarding Routine Use Key decisions and implementation strategies need to include the identification of effective disinfection processes and the determination of where and how the cleaning and disinfection will be done and by whom. Finding dedicated space, if needed, for this effort could be a challenge in many health care facilities. At TCID, each trained staff member is responsible for cleaning his or her personal elastomeric respirator and for notifying the respiratory department if maintenance or replacement is needed (Kizilbash et al., 2018).
The cleaning and disinfection processes will need to become part of the initial staff training on respirators and also to be incorporated into refresher training. Monitoring and compliance checks will be critical. A facility that has these processes in place will be better prepared to move into a surge situation because a portion of the staff would already be trained, the disinfection system would be known, and the logistics for reusable respirators would have been addressed, contingent on the facility stockpiling the same brand of respirators or ones with a similar cleaning process.
Considerations Regarding Surge Use If reusable elastomeric respirators are a component of the emergency surge stockpile from the facility or public health authorities, but they are not used routinely at the facility, it will be necessary to have quickly implementable and frequently practiced plans (as with storage and transport issues) that establish, on a practical basis, how and where cleaning and disinfection will occur. As previously discussed, these protocols would need to be part of the just-in-time training. The familiarity and standard operating procedures that a health care facility will have when it uses reusable elastomeric respirators as part of its routine respiratory protection program should make it easier to scale up in a pandemic situation.
Medical Clearance, Fit Testing, and Respirator Issuance
Medical Clearance and Fit Testing
A critical part of respiratory protection programs for health care workers is ensuring that users are medically cleared to participate in the program and that the respirators are selected and sized to best fit the users; both of these things are the responsibilities of the respiratory protection program administrator. Resources have been developed that are specific to health care respiratory protection programs, including Hospital Respiratory Protection Toolkit: Resources for Respirator Program Administrators (OSHA and NIOSH, 2015) and Implementing Hospital Respiratory Protection Programs: Strategies from the Field (Joint Commission, 2014). The medical evaluation is focused on determining if a potential respirator user can wear a respirator or if he or she has conditions that could prevent respirator use, such as certain heart conditions, lung disease, and psychological conditions, e.g., claustrophobia (OSHA, 2018a).
Currently, fit testing is necessary for both disposable filtering facepiece and reusable elastomeric respirators. Reusable elastomeric respirators are produced in varying sizes and with varying designs, and health care facilities can choose the models and sizes that will ensure a fit for most users. Fit testing is specific to the brand and model of respirator that is being fitted and is not interchangeable. This presents a challenge when the respirator used for routine care and the one stockpiled for surge situations are different. It presents an even bigger challenge when the specific composition of the emergency stockpile is not known, as with the SNS. Fit testing is required to be conducted on an annual basis and provides an opportunity for staff training on respiratory protection and on new options that become available. Those who cannot achieve a fit or who for other reasons cannot be fit tested can use a PAPR.
Considerations Regarding Routine Use Both medical clearance and fit testing are a mandatory part of routine use of respirators. In health care facilities that stock reusable elastomeric respirators for use in emergencies, the annual fit test provides an opportunity to fit and size the elastomeric respirator as well so that the employees who are part of the respiratory protection program are ready to use either type of respirator.
Considerations Regarding Surge Use During surge use a major concern will be fitting employees for the respirators that are available; this fit testing will need to be done quickly and effectively. Several situations could make it necessary to carry out just-in-time fit testing—for example, if there are types of respirators in the health care facility’s emergency stock to which employees have not been fitted or if different makes and models or types of respirators are received from the SNS or other resources. During the 2009 H1N1 pandemic it was noted that in many cases the respirators delivered from the SNS were not the same model for which the health care workers had been fit tested, which resulted in valuable time being spent in just-in-time fit testing (HHS, 2012a). Even facilities that fit test employees for elastomeric respirators during routine fit testing would need to fit test additional staff during a large emergency.
Staffing and Respirator Issuance
Policies vary among hospitals and other health care facilities about the number and types of workers who are part of the respiratory protection program. For some institutions, it works best for a large portion of the workforce to be fit tested and trained and thus be eligible to wear a respirator. For other facilities, fewer personnel are fit tested and trained—generally those working in areas with a high potential risk for exposure, including respiratory therapy or emergency departments or other areas with the potential for exposure to tuberculosis or other airborne infectious diseases. A study on the respiratory protection programs in nine health care organizations found that the number of staff members who were part of the respiratory protection program ranged from 160 to 20,000 individuals; however, the sizes of the staff at the health care organizations were not provided (Brown et al., 2017).
The needs of the health care facility are paramount in decisions about the scope of the respiratory protection program; scope is raised here as one of many considerations regarding the use of various types of respirators. The respiratory protection program administrator, in partnership with infection prevention and control, value analysis, occupational health, and other pertinent departments, must determine the appropriate size and scope for the organization’s specific respiratory protection program during both routine and surge situations. Consideration has been given to stratifying the risks experienced by health care workers according to the types of work that they do and the locations of their work. For example, health care workers performing aerosol-generating procedures on known or suspected
pandemic patients would be at high exposure risk. This type of approach to stratifying risks (also termed the control banding approach) could be used for decision making on PPE selection and for prioritization during public health emergencies (Patel et al., 2017).
Procurement and Supply Logistics and Emergency Stockpiles
It is not possible to implement a respiratory protection program without access to a supply of respirators whose purchase and use require a complex chain of decisions involving multiple clinical and administrative teams in health care facilities working with suppliers and, in some cases, directly with manufacturers.
Manufacturing and Supply Chain
Health care is one sector of a much larger—primarily, industrial—market for respirators (see Chapter 2 for a description of the use of elastomeric respirators in other industries). It is estimated that more than 5 million workers are required to wear respirators in 1.3 million U.S. workplaces (OSHA, 2018b).
The production capacity for respirators, particularly the U.S.-based capacity, will be a major concern in a public health crisis, particularly a crisis in which there is global demand for respiratory protection. As noted by the authors of a review of lessons learned from recent public crises,
A significant proportion of the respiratory protective device supply chain is produced offshore and may not be available to the U.S. market during a public health response because of export restrictions to the United States or the nationalization of manufacturing facilities, which may favor in-country rather than foreign demands. (Patel et al., 2017, p. 245)
Thus, in a global emergency situation, respirator supplies might be quite limited and it will take time for U.S.-based manufacturing to gear up to meet the demands. Global suppliers will also be involved in supplying the raw materials necessary to manufacture respirators domestically (NASEM, 2018).
Adding to the supply concerns is the lean supply management approach used by many health care facilities, which often rely on just-in-time supply chains that deliver products, including respirators, when needed, resulting in little excess inventory to deal with an emergency situation
(Patel et al., 2017). Health care facilities often do not have the capacity to store large quantities of supplies, and the storage space they do have is needed for a wide variety of products and devices.
In 2009, the manufacturing and supply chain limitations quickly became apparent when orders for disposable filtering facepiece respirators rapidly spiked and created a 2- to 3-year backlog (Patel et al., 2017). In a study of 16 California hospitals during the H1N1 pandemic, more than 80 percent of hospital managers who reported shortages of disposable respirators said at the time they were interviewed that the orders they had placed for additional respirators could not be filled by suppliers (Beckman et al., 2013). During the response to the 2014 Ebola virus epidemic, there was an initial spike in the ordering of PPE products (estimates of the demand range from 10 to 200 times the normal amounts ordered) followed by a more strategic assessment of PPE needs once CDC’s tiered approach to triage and treatment had been implemented; in this approach hospitals were categorized as front line facilities, assessment hospitals, or Ebola treatment centers—each tier with varying PPE timelines and needs (CDC, 2016; Patel et al., 2017).
As discussed further below, the federal SNS and the connected network of state and local stockpiles has been used to help ease respirator shortages in past surge situations, along with local supplies and increased production and distribution.
Total Cost
For elastomeric respirators, the purchase cost is only one factor among many that go into purchasing and maintenance decisions. For routine use, the total cost of a specific type of respirator includes the
- Purchase price of the respirators and associated equipment (e.g., filters, transport and storage bags). Besides the initial setup cost, replacement costs are influenced by the expected lifespan of the respirators, the numbers of new staff and temporary or short-term health care workers (e.g., trainees or contractors), and the frequency of filter replacement. There is also a cost for lost or damaged equipment.
- Costs of fit testing and training (e.g., staff to conduct the medical clearance, fit tests, and training; time for clinical staff to participate; equipment and materials needed). The number of health care
-
workers included in the respiratory protection program dictates the volume of the annual fit testing required.
- Costs associated with storage (space and infrastructure on the clinical units and in warehouse storage).
- Costs associated with cleaning and disinfection (materials, staffing, and the costs—in both space and time—of processing).
- Costs associated with the disposal of respirators.
For emergency preparedness, it is expected that stockpiled respirators will spend all or most of their shelf life in the warehouse, given the infrequency of global pandemics; therefore, the bulk of the costs incurred are in the initial purchase costs, replacement costs prorated to the shelf life of the respirators, and the cost of warehouse space and inventory management. In some health care facilities, stockpiled respirators are rotated into routine use. Additional costs may be projected as part of the deployment of these respirators during an emergency, including distribution, just-in-time fit testing and training, the implementation of cleaning and disinfection processes, and potential losses from the diversion of respirators to the community.
The purchase price per unit of elastomeric respirators (estimated in one study at $25 to $50) is higher than that of disposable filtering facepiece respirators (estimated at $0.25 to $0.65 per respirator) and lower than PAPRs (estimated at $500 to $800 per respirator) (Baracco et al., 2015).1 In an estimate of the costs per worker, a draft OSHA document found elastomerics to be the most cost effective (OSHA, n.d.) (see Box 3-2). Baracco and colleagues (2015) looked at the costs of stockpiling respirators and found elastomeric respirators to have the lowest costs when considering acquisition and warehousing costs in a pandemic situation. Neither estimate took into account the implementation costs, including the cleaning and disinfection of elastomeric respirators or staff training.
___________________
1 Elastomeric respirators were estimated to need three sets of filters annually at an additional cost of $25 per set. PAPRs were estimated to have additional costs of $250 per battery (one battery needed per every 10 hours of use), additional hoods (three needed per PAPR at $30 per hood), and additional tubes (three needed per PAPR at $30 per tube) (Baracco et al., 2015).
The committee urges that more work be done to determine the total comparative costs of the various types of respirators, including elastomeric respirators, that could be used in a pandemic or other surge situation (see Chapter 5). The biggest unknown costs are data-based policy development, staff education and training time, and staff time and supply costs for cleaning, disinfection, and maintenance. Given the wide cost differences in the estimates that have been done (Baracco et al., 2015; see Box 3-2) further efforts are needed.
A value-analysis approach to health care supply decision making is one in which all relevant clinical and business issues and impacts are considered throughout the cycle of purchase, use, and product evaluation, with consideration paid to the clinical impact on patient care, quality, and safety. For respirators, the relevant clinical inputs include infection prevention and control, respiratory care, occupational health, and environmental health. In a presentation at the committee’s May workshop, Gloria Graham noted that the value-analysis perspective would incorporate patient care impact, clinical necessity and effectiveness, patient and health care worker safety, the volume of use, the uniqueness of the item or prod-
uct line, product compatibility with other systems/units, a cost–benefit determination, contract compliance, distribution compliance, storage of the units, and staff competency (Graham, 2018).
Lessons learned from the H1N1 and Ebola crises regarding the supply of respirators include the need for improvements throughout the supply chain, such as developing systems to share information on PPE use, ensuring increased transparency of PPE orders (particularly for federal purchases), providing increased opportunities for the sharing of supplies within regions, and developing more specific PPE selection guidance tools (Patel et al., 2017). Ways to ramp up domestic manufacturing surge capacity during a public health crisis are also being explored (HHS, 2015; Patel et al., 2017).
Emergency Stockpiles
When public health emergencies occur, hospitals and other health care facilities generally rely first on their own stock of supplies and then turn to local, state, and federal government resources. Of 1,066 acute-care hospitals in the United States responding to an Association of State and Territorial Health Officials survey in 2012, just under half (44 percent) indicated that they had an emergency cache of respiratory PPE (ASTHO, 2014).
Originating as a pharmaceutical stockpile, the federal SNS has expanded to include other emergency products, including respirators (CDC, 2018). The SNS is designed to supplement and resupply state and local inventories of emergency medical supplies. In 2009 the H1N1 influenza pandemic triggered
the largest deployment in SNS history when 12.5 million antiviral regimens were deployed across the country (a further 300,000 were deployed internationally), as well as 19.6 million pieces of PPE, 85.1 million N95 respirators, and 2,129 regimens of Peramivir IV (the latter were deployed in conjunction with the Biomedical Advanced Research and Development Authority [BARDA]). (NASEM, 2016, pp. 10–11)
This was a deployment of approximately 75 percent of the SNS cache of disposable filtering facepiece respirators (Patel et al., 2017). The SNS supplies were distributed to state health departments which were then responsible for distributing the supplies to facilities within each state. As noted
above, this deployment of respirators eased shortages and ordering backlogs that health care facilities were facing. Manufacturers also increased production of the products to meet the increases in demand (Patel et al., 2017).
One of the challenges in emergency planning has been the lack of clarity on the nature and extent of the responsibilities that private-sector health care organizations and federal and state government agencies each have regarding the stockpiling of respirators and other PPE products. Additionally, health care systems and facilities do not have information on the specific makes, models, and sizes of the respirators that are in the federal stockpile—information that would be helpful to better plan for transitions during surge situations. If it became possible to know the types of respirators and the specific models in the stockpiles, staff could be fit tested and trained on those specific respirators, and the transition would be expedited. Finding out this information in the midst of a pandemic or other crisis puts additional strains on what will be an already heavily burdened workforce. As noted in Box 3-3, challenges occurred during the H1N1 pandemic regarding the fit testing of a supply of respirators from the California stockpile.
Following the 2009 H1N1 pandemic, an analysis of the lessons learned noted,
In some cases, PPE that was released [from the SNS] was not the preferred or previously fit tested brand, did not fit, or required training for use. . . . Because of unique training and fit testing requirements for each brand of mask, standardizing the brand of PPE available from the SNS and soliciting input from states into decisions about purchases for the SNS contents should be considered. (HHS, 2012a)
The National Guidance for Healthcare System Preparedness had a similar recommendation: “The type of PPE that is procured for local or regional caches should be consistent with the type of PPE used locally to promote interoperability and inter-facility sharing” (HHS, 2012b, p. 49).
The committee understands the need to keep certain details of the SNS classified. However, state and federal authorities could consider disclosing to health care facilities the manufacturers, makes, and models of respirators placed in stockpiles. Such disclosure would ease the burden on health care facilities in their attempts to rapidly adopt these devices in a surge context.
Safety Culture and Risk Perception
Each organization creates a culture that exhibits its values and is evident in the workplace through the ways in which employees interact and perform their jobs. One aspect of an organizational culture is its safety culture. DeJoy (2018) defines safety culture as the attitudes, values, norms, and beliefs that people in a workplace or organization share with respect to risk and safety. The culture of safety is reflected in the organizational policies, standard operating procedures, structures, and expected norms of behavior of the health care workers. A component of the safety culture is
the “safety climate,” which is how the employees perceive the safety in their work environment. When there is a strong emphasis on a positive safety culture and climate, this has constructive impacts on the behaviors of the worker and their well-being and also has the potential for positive impacts on the outcomes of the organization’s efforts (in this case, a decreased transmission of airborne diseases to health care workers and patients).
The traits of a positive safety culture include leadership safety values and actions, problem identification and resolution, personal accountability, work processes, continuous learning, an environment for raising concern, effective safety communication, a respectful work environment, and a questioning attitude (NRC, 2011). An organization’s safety climate can be perceived differently by people in different roles. A study of 98 hospitals across 6 states in which more than 1,105 health care workers were surveyed found that front line health care workers perceived the safety climate for respiratory protection less positively than hospital and unit managers (Peterson et al., 2016). It is not clear why various workers perceive a safety climate differently. For any respiratory program to be effective, it has to be the cultural norm, a priority for leaders and managers, and the policies must be followed by every employee, every time, every day. A robust safety culture is one in which corrections or reminders are expected and are accepted as part of daily work. Additionally, it is critical that health care workers at all levels have input into safety issues and feel free to raise respiratory safety and other safety concerns (Peterson et al., 2016).
A number of industries other than health care (e.g., commercial air travel, nuclear power plants, and amusement parks) have achieved high levels of consistent safety performance. The set of characteristics that has been used to identify “high-reliability organizations” includes leadership and management’s commitment to safety; the availability of safety resources and incentives; open and candid communications; a low frequency of unsafe behavior, even under production pressures; prioritizing safety, even at the expense of productivity and efficiency; continuous safety mindfulness; openness about errors and problems; and being an organization that values learning from past experiences (Roberts, 1990; Rochlin, 1999; DeJoy, 2018).
Efforts to improve the safety culture of health care have focused largely and necessarily on patient safety. The committee urges that attention also be placed on worker safety and on the multidimensional interventions, including improvements in the area of respiratory protection, that are needed to change the safety culture to improve worker health and safety.
The committee was not tasked with exploring the behavioral and safety culture issues in depth, but it emphasizes the importance of considering these issues as part of decision and policy making regarding respiratory protection and employee safety in health care facilities. Key features of systematic, ongoing, and multidimensional safety culture change are outlined in Box 3-4.
Considerations Regarding Routine Use
Risk perception is often a challenge in implementing respiratory protection protocols in routine health care. Health care workers become accustomed to dealing with life and death situations and may not take respiratory precautions seriously, particularly since the respiratory risks for health care workers (i.e., airborne viruses or bacteria) are invisible, the onset of disease from inhaling pathogens may not be immediate, respiratory protective devices may be perceived as interfering with patient care, and even when there is a high risk of transmission, not all workers acquire the infection (Chung et al., 2015). The committee was able to identify only one study that explored safety culture and safety climate issues with attention to reusable elastomeric respirators. In a focus group of health care workers who had used elastomeric respirators, the workers noted the risk perception issues and said that they “would feel safer wearing ERs [elastomeric respirators], as they were viewed as offering more protection” (Hines et al., 2017, p. 101).
Having a strong safety culture in which compliance with respiratory protection is expected and frequently monitored can improve compliance with respiratory protection protocols. Furthermore, if a strong organizational safety culture is in place during routine health care, then when a pandemic or other crisis occurs, the staff can quickly and knowledgably respond.
Considerations Regarding Surge Use
In emergency or surge use, the driving factor will be the public health crisis. Health care staff will pay close attention to the transmission route of the disease and be attentive to safety measures including, as needed, respiratory protection. During the 2009 H1N1 pandemic, the demand at health care facilities for respirators was high and the initial supplies of the product were rapidly used (Patel et al., 2017). The Ebola crisis highlighted the need for full body protection, and precaution measures, including respiratory protection, were taken seriously (CDC, 2015; Fischer et al., 2015). The urgency and seriousness of a disease heightens the perception of risk and potentially results in greater compliance with PPE guidance. As discussed below, when different versions of PPE guidance are provided by authority organizations during a public health crisis, confusion is heightened.
Other Implementation Issues
Out-of-Hospital Settings
Much attention has been paid to emergency planning for large urban tertiary hospitals and health care institutions, but further efforts are needed that focus on home health care workers, emergency care workers, nursing home workers, health care workers in rural locations, and others (Baron et al., 2009; El Sayed et al., 2011; Rebmann et al., 2011). The Bureau of Labor Statistics estimated that in 2016 there were 2.9 million home health aides and personal care aides caring for home-based patients (BLS, 2018b), and there are also numerous health care workers in other out-of-hospital settings. In some cases these workers are employed by organizations that provide supplies such as respirators, while in other cases the workers are self-employed and may or may not purchase or use respirators. There are also more than 248,000 emergency care workers (emergency medical technicians and paramedics) with direct patient care responsibilities (BLS, 2018a). The committee noted a need to focus on respiratory protection outside of acute-care settings. In these settings, reusable elastomeric respirators may have challenges and benefits not fully explored.
Regulatory and Liability Issues
A concern has been raised about the use of reusable elastomeric respirators in a public health emergency from the legal perspective regarding manufacturers’ liability. The Public Readiness and Emergency Preparedness Act, enacted in 2005, authorizes the secretary of the U.S. Department of Health and Human Services (HHS) to issue a declaration that provides manufacturers with immunity from tort liability for claims of loss (with exceptions such as willful misconduct) due to countermeasures to help abate public health emergencies (HHS, 2017).2 The act notes that it covers medical devices that are defined, cleared, or approved under the Federal Food, Drug, and Cosmetic Act.3 This coverage would apply to the surgical N95 disposable filtering facepiece respirators approved by FDA, but it is not clear that reusable elastomeric respirators would be covered. The committee did not examine the legal issues involved but instead raises it as an issue that may need further exploration or regulatory or legislative action.
___________________
2 Public Law 109-148.
3 21 USC 9.
Guidelines for Respiratory Protection
Lessons learned from the 2009 H1N1 influenza pandemic indicate the necessity for clear and consistent guidelines and policies regarding when to use PPE and what level of protection is needed during a pandemic or surge situation. As noted in Table 3-2 and described in a 2011 Institute of Medicine report:
The lack of precise information about the modes of influenza transmission, the contagiousness, the virulence of novel H1N1 influenza A, the at-risk population, and the efficacy of different devices in preventing transmission led to a variety of recommendations at different times by federal and local government public health agencies. Delayed and/or disparate recommendations often led to confusion among health care personnel and their employers, who had to decide what to tell personnel about what type of PPE to wear and when. In addition, little research was available to guide health system officials in making decisions about the quantities of various types of PPE needed to protect their workforce. A major problem encountered was a slow response in tailoring recommendations as more knowledge about virulence and affected populations became available. (IOM, 2011b, p. 141)
The mixed messages that occurred in 2009 with the H1N1 pandemic regarding what level and type of PPE to use need to be avoided in the next pandemic influenza or another public health crisis. In a follow-up to the 2009 pandemic, HHS noted:
Implementation of PPE guidance varied across federal departments, stemming from the fact that different federal agencies released different sets of recommendations on the appropriate PPE to protect against the 2009 H1N1 influenza virus. It would be desirable in the future for the federal government to disseminate a single, consistent set of recommendations. (HHS, 2012a, p. 35)
Agreement on guidance and standards across agencies, coupled with streamlined and frequent communication through a broad array of media platforms, will increase the likelihood of adherence to recommended standards and optimal health for health care workers and their patients.
| CDC Guidance 4/29/09 Novel Pandemic Influenza | CDC Guidance 10/15/09 Novel Pandemic Influenza | CDC Guidance for Seasonal Influenza 9/20/10 | WHO Guidance | Public Health Agency of Canada Guidance for Novel H1N1 | |
|---|---|---|---|---|---|
| Recommended level of infection control precautions | Standard and contact precautions and eye protection | Standard and droplet precautions | Adhere to standard and droplet precautions | Standard and droplet precautions | Tiered approach |
| Recommended respiratory PPE | NIOSH-approved N95 respirator | NIOSH-approved N95 respirator | Medical mask except for aerosolgenerating procedures, use N95 or better | Medical mask except for aerosolgenerating procedures | Medical mask except for aerosol generating procedures |
| Did the respiratory PPE recommendation differ by work task? | Yes—direct care versus indirect patient contact | Yes—direct care versus indirect patient contact | Yes—direct care versus aerosol generating procedure | Yes | Yes |
NOTE: CDC = Centers for Disease Control and Prevention; NIOSH = National Institute for Occupational Safety and Health; PPE = personal protective equipment; WHO = World Health Organization.
SOURCES: Adapted from CDC, 2009, 2010; IOM, 2011b.
REFERENCES
ASTHO (Association of State and Territorial Health Officials). 2014. Assessment of respiratory personal protective equipment in U.S. acute care hospitals—2012. http://www.astho.org/Preparedness/Respiratory-PPE-Assessment-Report (accessed July 5, 2018).
Baracco, G., S. Eisert, A. Eagan, and L. Radonovich. 2015. Comparative cost of stockpiling various types of respiratory protective devices to protect the health care workforce during an influenza pandemic. Disaster Medicine and Public Health Preparedness 9:313–318.
Baron, S., K. McPhaul, S. Phillips, R. Gershon, and J. Lipscomb. 2009. Protecting home health care workers: A challenge to pandemic influenza preparedness planning. American Journal of Public Health 99(Suppl 2):S301–S307.
Beckman, S., B. Materna, S. Goldmacher, J. Zipprich, M. D’Alessandro, D. Novak, and R. Harrison. 2013. Evaluation of respiratory protection programs and practices in California hospitals during the 2009–2010 H1N1 influenza pandemic. American Journal of Infection Control 41(11):1024–1031.
BLS (Bureau of Labor Statistics). 2018a. EMTs and Paramedics. https://www.bls.gov/ooh/healthcare/emts-and-paramedics.htm (accessed November 13, 2018).
BLS. 2018b. Home health aides and personal care aides. https://www.bls.gov/ooh/healthcare/home-health-aides-and-personal-care-aides.htm (accessed August 16, 2018).
Brown, L. M., B. Rogers, K. Buckheit, and J. P. Curran. 2017. Evaluation of 9 health care organizations’ respiratory protection programs and respiratory protective device practices: Implications for adoption of elastomerics. American Journal of Infection Control 46(3):350–352.
Burns, C., A. M. Lachat, K. Gordon, M. G. Ryan, M. Gruden, D. P. Barker, and D. Taormina. 2014. Respiratory protection competencies for the occupational health nurse. Workplace Health and Safety 62(3):96–104.
Carias, C., G. Rainish, M. Shankar, B. B. Adhikari, D. L. Swerdlow, W. A. Bower, S. K. Pillai, M. I. Meltzer, and L. M. Koonin. 2015. Potential demand for respirators and surgical masks during a hypothetical influenza pandemic in the United States. Clinical Infectious Diseases 60(Suppl 1):S42–S51.
CDC (Centers for Disease Control and Prevention). 2009. Interim guidance for emergency medical services (EMS) systems and 9-1-1 public safety answering points (PSAPs) for management of patients with confirmed or suspected swine-origin influenza A (H1N1) infection. http://cdc.gov/h1n1flu/guidance_ems.htm (accessed August 16, 2018).
CDC. 2010. Interim guidance on infection control measures for 2009 H1N1 influenza in healthcare settings, including protection of healthcare personnel. http://cdc.gov/h1n1flu/guidelines_infection_control.htm (accessed August 16, 2018).
CDC. 2015. Guidance on personal protective equipment (PPE) to be used by healthcare workers during management of patients with confirmed Ebola or persons under investigation for Ebola who are clinically unstable or have bleeding, vomiting, or diarrhea in U.S. hospitals, including procedures for donning and doffing PPE. https://www.cdc.gov/vhf/ebola/healthcareus/ppe/guidance.html (accessed July 5, 2018).
CDC. 2016. Considerations for U.S. healthcare facilities to ensure adequate supplies of personal protective equipment (PPE) for Ebola preparedness. https://www.cdc.gov/vhf/ebola/healthcare-us/ppe/supplies.html (accessed August 20, 2018).
CDC. 2018. Strategic National Stockpile. https://www.cdc.gov/phpr/stockpile/index.htm (accessed July 5, 2018).
Chung, W. M., J. C. Smith, L. M. Weil, S. M. Hughes, S. N. Joyner, E. M. Hall, J. Ritch, D. Srinath, E. Goodman, M. S. Chevalier, L. Epstein, J. C. Hunter, A. J. Kallen, M. P. Karwowski, D. T. Kuhar, C. Smith, L. R. Petersen, B. E. Mahon, D. L. Lakey, and S. J. Schrag. 2015. Active tracing and monitoring of contacts associated with the first cluster of Ebola in the United States. Annals of Internal Medicine 163(3):164–173.
Ciconte, R., Q. Danyluk, G. Astrakianakis, P. Percy Chua, G. Ducholke, S. Daniels, T. Dickson, and I. Perry. 2013. Assessment and determination of practical considerations for wide-scale utilization of elastomeric half-facepiece respirators during a pandemic or outbreak situation. https://www.worksafebc.en/resources/about-us/research/assessment-and-determination-of-practical-considerations-for-wide-scale-utilization-of-elastometric-half-facepiece-respirators-during-a-pandemic-or-outbreak-situation?lang=en (accessed August 20, 2018).
Cohen, B., S. Hyman, L. Rosenberg, and E. Larson. 2012. Frequency of patient contact with health care personnel and visitors: Implications for infection prevention. Joint Commission Journal on Quality and Patient Safety 38(12):560–565.
DeJoy, D. 2018. Safety culture and training/education. Presentation at the May 22, 2018, public meeting of the National Academies of Sciences, Engineering, and Medicine Committee on the Use of Elastomeric Respirators in Health Care. Washington, DC. http://www.nationalacademies.org/hmd/~/media/Files/Activity%20Files/Workforce/ElastomericRespirators/Meeting%203/DeJoy.pdf (accessed August 21, 2018).
El Sayed, M., R. Kue, C. McNeil, and K. S. Dyer. 2011. A descriptive analysis of occupational health exposures in an urban emergency medical services system: 2007–2009. Prehospital Emergency Care 15(4):506–510.
Fischer, W. A., D. Weber, and D. A. Wohl. 2015. Personal protective equipment: Protecting healthcare providers in an Ebola outbreak. Clinical Therapeutics 37(11):2402–2410.
Graham, G. 2018. Value analysis and decision-making process. Presentation at the May 22, 2018, public meeting of the National Academies of Sciences,
Engineering, and Medicine Committee on the Use of Elastomeric Respirators in Health Care. Washington, DC. http://nationalacademies.org/hmd/~/media/Files/Activity%20Files/Workforce/ElastomericRespirators/Meeting%203/Graham.pdf (accessed August 15, 2018).
Gribogiannis, M. 2018. Elastomeric respirators: Infection control perspective. Presentation at the May 22, 2018, public meeting of the National Academies of Sciences, Engineering, and Medicine Committee on the Use of Elastomeric Respirators in Health Care. Washington, DC. http://nationalacademies.org/hmd/~/media/Files/Activity%20Files/Workforce/ElastomericRespirators/Meeting%203/Gribogiannis.pdf (accessed August 13, 2018).
HHS (U.S. Department of Health and Human Services). 2012a. An HHS retrospective on the 2009 H1N1 influenza pandemic to advance all hazards preparedness. https://www.phe.gov/Preparedness/mcm/h1n1-retrospective/Documents/h1n1-retrospective.pdf (accessed August 15, 2018).
HHS. 2012b. National guidance for healthcare system preparedness. Office of the Assistant Secretary for Preparedness and Response. https://www.phe.gov/preparedness/planning/hpp/reports/documents/capabilities.pdf (accessed August 20, 2018).
HHS. 2015. HHS funds development of high-speed manufacturing for N95 respirators. https://www.phe.gov/Preparedness/news/pages/n95.aspx (accessed August 20, 2018).
HHS. 2017. Public Readiness and Emergency Preparedness Act. https://www.phe.gov/Preparedness/legal/prepact/Pages/default.aspx (accessed August 14, 2018).
Hines, S. E., N. Mueller, M. Oliver, P. Gucer, and M. McDiarmid. 2017. Qualitative analysis of origins and evolution of an elastomeric respirator-based hospital respiratory protection program. Journal of the International Society for Respiratory Protection 34(2):95–110.
IOM (Institute of Medicine). 2011a. Occupational health nurses and respiratory protection: Improving education and training. Washington, DC: The National Academies Press.
IOM. 2011b. Preventing transmission of pandemic influenza and other viral respiratory diseases: Personal protective equipment for healthcare personnel: Update 2010. Washington, DC: The National Academies Press.
Joint Commission. 2014. Implementing hospital respiratory protection programs: Strategies from the field. Oakbrook Terrace, IL: The Joint Commission. https://www.jointcommission.org/assets/1/18/Implementing_Hospital_RPP_2-19-15.pdf (accessed August 14, 2018).
Kizilbash, A., C. Guenther, R. Reed, and D. Mata. 2018. Use of elastomeric respirators at a TB hospital. Presentation at the March 22, 2018, public meeting of the National Academies of Sciences, Engineering, and Medicine Committee on the Use of Elastomeric Respirators in Health Care. Washington, DC. http://nationalacademies.org/hmd/~/media/Files/Activity%20Files/Workforce/ElastomericRespirators/Meeting%202/TCID.pdf (accessed August 13, 2018).
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016. The nation's medical countermeasure stockpile: Opportunities to improve the efficiency, effectiveness, and sustainability of the CDC Strategic National Stockpile: Workshop summary. Washington, DC: The National Academies Press.
NASEM. 2018. Impact of the global medical supply chain on SNS operations and communications: Proceedings of a workshop. Washington, DC: The National Academies Press.
NIOSH (National Institute for Occupational Safety and Health). 2010. NIOSH investigation of 3M model 8000 filtering facepiece respirators as requested by the California Occupational Safety and Health Administration, Division of Occupational Safety and Health. https://www.cdc.gov/nioch/hhe/reports/pdfs/2010-0044-3109.pdf?id=10.26616/NIOSHHETA201000443109 (accessed October 3, 2018). HETA 2010-044-3109.
NRC (Nuclear Regulatory Commission). 2011. Issuance of final safety culture policy statement. Federal Register 76(114):34773–34778.
OSHA (Occupational Safety and Health Administration). 2018a. Medical evaluations for workers who use respirators. https://www.osha/gov/video/respiratory_protection/medevaluations_transcript.html (accessed July 5, 2018).
OSHA. 2018b. Respiratory protection. https://www.osha.gov/SLTC/respiratoryprotection (accessed August 15, 2018).
OSHA. n.d. Proposed guidance on workplace stockpiling of respirators and facemasks for pandemic influenza—Draft for public comment. https://www.osha.gov/dsg/guidance/proposedGuidanceStockpilingRespirator.pdf (accessed August 15, 2018).
OSHA and NIOSH. 2015. Hospital respiratory protection program toolkit: Resources for respirator program administrators. https://www.cdc.gov/niosh/docs/2015-117/pdfs/2015-117.pdf?id=10.26616/NIOSHPUB2015117 (accessed August 16, 2018).
Patel, A., M. M. D’Alessandro, K. J. Ireland, W. G. Burel, E. B. Wencil, and S. A. Rasmussen. 2017. Personal protective equipment supply chain: Lessons learned from recent public health emergency responses. Health Security 15(3):244–252.
Peterson, K., B. M. E. Rogers, L. M. Brosseau, J. Payne, J. Cooney, L. Joe, and D. Novak. 2016. Differences in hospital managers’, unit managers’, and health care workers’ perceptions of the safety climate for respiratory protection. Workplace Health and Safety 64(7):326–336.
Pompeii, L., A. Byrd, G. L. Delclos, and S. H. Conway. 2016. The American Association of Occupational Health Nurses’ respiratory protection education program and resources webkit for occupational health professionals. Workplace Health and Safety 64(12):564–572.
Rebmann, T., B. Citarella, S. Alexander, B. Russell, and J. C. Volkman. 2011. Personal protective equipment use and allocation in home health during disasters. American Journal of Infection Control 39(10):823–831.
Roberts, K. H. 1990. Some characteristics of one type of high reliability organization. Organization Science 1(2):160–176.
Rochlin, G. I. 1999. Safe operation as a social construct. Ergonomics 42(11):1549–1560.
Rogers, B., L. E. McCurdy, K. Slavin, K. Grubb, and J. R. Roberts. 2009. Children’s Environmental Health Faculty Champions Initiative: A successful model for integrating environmental health into pediatric health care. Environmental Health Perspectives 117(5):850–855.
Rogers, B., K. Buckheit, J. Ostendorf, and E. Alfano-Sobsey. 2013. Integration of respiratory protection into practice among health care workers exposed to influenza. NIOSH contract #254-2012-M-53195.
Rogers, B., K. Buckheit, and J. Ostendorf. 2018. Development of competencies for respiratory protection for health care workers. Workplace Health and Safety. https://doi.org/10.1177/2165079918798857 (accessed November 9, 2018).
Shaw, E. K., J. Howard, D. R. West, B. F. Crabtree, D. E. Nease, Jr., B. Tutt, and P. A. Nutting. 2012. The role of the champion in primary care change efforts. Journal of the American Board of Family Medicine 25(5):676–685.
Sheets, C. D., and P. L. Payne. 2014. Ebola 2014: Clarifying the confusion: A public health and laboratory containment perspective. Applied Biosafety 19(3):118–123.
Wizner, K., L. Stradtman, D. Novak, and R. Shaffer. 2016. Prevalence of respiratory protective devices in U.S. health care facilities: Implications for emergency preparedness. Workplace Health and Safety 64(8):359–368.
This page intentionally left blank.




































