Proceedings of a Workshop
| IN BRIEF | |
|
January 2020 |
Closing the Loop on the Plastics Dilemma
Proceedings of a Workshop—in Brief
Plastics are used in various industries to produce lightweight, corrosion-resistant, durable materials, but many of the characteristics that make them valuable also make them environmentally unfriendly. Although some plastics can be recycled, the vast majority are simply discarded. The rapid proliferation of plastics in the environment has led to an urgent need to reimagine how plastics are created, used, and managed. The workshop Closing the Loop on the Plastics Dilemma, held in Washington, DC, on May 9–10, 2019, provided a venue for discussing opportunities to reduce the adverse environmental effects of plastics. Chemists, chemical engineers, and participants in related fields engaged in a rich discussion of approaches to improve plastics recycling and to promote the connection of product design with the end stages of the plastics life cycle. This brief proceedings summarizes the presentations and discussions that took place during the workshop. The workshop videos, presentations, and poster abstracts are available online.1
THE PLASTICS DILEMMA
Eric Beckman, Bevier Professor of Engineering at the University of Pittsburgh, began the workshop with an overview of the current global plastic-waste problem, recycling challenges, and potential solutions.
What Is the Global Plastic-Waste Problem?
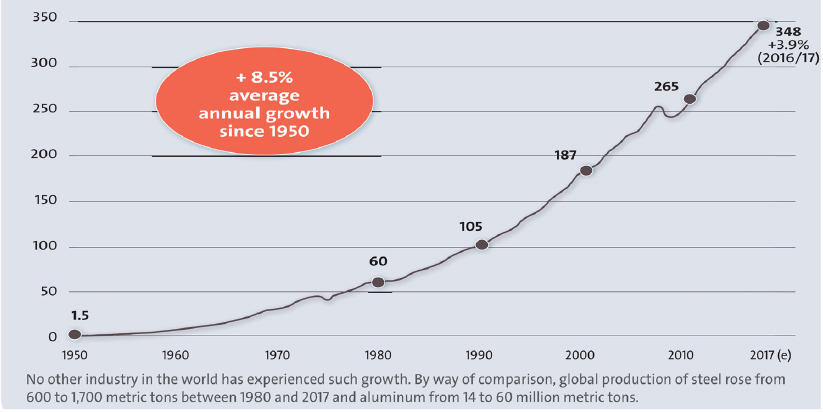
Beckman stated that synthetic polymers have fueled a remarkably successful industry thanks to their desirable physical properties and low production cost. Plastic production has risen by an average of 8.5% per year since 1950, a higher rate of sustained growth than in any other industry. About 350 million metric tons are produced globally today (Figure 1). Six resins make up more than 90% of current plastics production: polyethylene, polypropylene, polyvinyl chloride, polystyrene, polyethylene terephthalate (PET), and polyurethane. Beckman emphasized that demand for polymer-based products and packaging is likely to continue to grow as improving standards of living put these products within reach for more of the world’s population.
Although recycling technologies and adoption have improved over the last several decades, recycling rates remain low, Beckman stated. Globally, about 15% of plastic waste is recycled, a slightly greater percentage is incinerated, and the rest is discarded. However, Beckman stressed that those statistics belie the scope of the problem. Fibers, notably polyester fibers, are not typically included in the discussion. That is problematic because textile production is resource-intensive, people are buying more clothes and keeping them for shorter times, and textile-recycling rates are abysmal. Another important issue is that the production and use of polymer materials are shifting from North America and Europe to Asia, where many countries lack the infrastructure for efficient waste management and recycling.
Beckman continued that the low recycling rates have led to a dramatic buildup of plastic waste in the environment, some of which will persist for hundreds of years. About 10 million tons of synthetic polymer material enters the ocean each year; it is estimated that by 2050 the mass of polymers in the ocean will outweigh the mass of fish (WEF 2016). As plastics break down into smaller and smaller particles, polymers and associated additives find their way into ecosystems and organisms. In addition to the ecologic consequences, the proliferation of plastic in the environment raises important questions about potential effects on human health, many of which remain unanswered (Wright and Kelly 2017).
___________
1 See https://nas-sites.org/csr/closing-the-loop-on-the-plastics-dilemma/.
![]()
What Makes Recycling of Plastics So Challenging?
Beckman discussed various reasons for the recycling of only a small fraction of plastic waste. One is an emphasis on customization—the design of materials and products for particular applications or customers—which often inhibits collection, separation, and reuse of the materials. Contamination of recyclable polymers with other substances is another issue. A third is complexity: products, such as electronic products and thermosets, are often made with multiple polymers, additives, colors, and other materials, and this makes recycling challenging or impossible. Asked about rubbers or elastomers, Beckman noted that although these present a problem, one favorable aspect is that tires, a major source of rubber waste, are relatively easy to collect. He said that even when polymer recycling is feasible, materials are often converted into substances of lower value.
In addition to the technical challenges of recycling synthetic polymers, Beckman stated, the recycling industry faces economic challenges given the low cost of virgin materials used in making new polymers. Another complicating problem is that the world’s recycling systems are in a state of upheaval. Although China and India used to import millions of tons of recyclable plastic material from the United States and Europe each year, they are ending that practice and forcing former exporters to develop new ways to handle their plastic waste. That development has led to increasing efforts to prevent waste at its source through bans on some plastics.
What Might Solutions Look Like?
Previewing some of the topics that would be addressed by other workshop speakers, Beckman raised an array of potential solutions. One is to disseminate current technology for waste collection, sorting, and cleaning and to increase demand for recycled materials. Prompted by a question from the audience, Beckman acknowledged that, if technologic solutions for handling complex mixtures were available, mining landfills for recyclable polymers, as has been proposed for some metals, might eventually become practical. Chemical recycling—converting polymers to monomers—is another potential way to improve recycling capabilities and appears to be practical for some polymers, such as PET and polystyrene. Polyolefins, however, present a difficult challenge for chemical recycling.
Beckman closed by outlining new approaches for designing tomorrow’s polymers, namely, to consider designing for at least one of the following characteristics: simplicity, standardization, recoverability, upgrading, disassembly or reassembly, efficient collection and separation, or degradation at the appropriate time, place, and rate and to the appropriate end products. He noted that designing for degradability is particularly relevant for applications, such as shoes or brake pads, in which normal use makes dissipation into the environment unavoidable. He added, however, that many degradable materials will not degrade under some conditions, such as anaerobic conditions found in landfills, so it will be important to ensure that such materials find their way into industrial composters. Other approaches could focus on building a second life into a material or product from the beginning. He emphasized that life-cycle assessment should be applied to verify that any chosen approach truly constitutes an improvement.
MECHANICAL RECYCLING OF PLASTICS
Brian Riise, a project manager at the REMADE Institute, provided an overview of methods for recycling plastics mechanically. Although recycling rates are low—and dropping—mechanical recycling and chemical recycling offer promise as a way to “close the loop” by recovering materials and thereby reducing the need to produce new materials. Riise stated that mechanical recycling is particularly attractive because it uses only 10–20% of the energy required to make virgin plastics. He listed several requirements for mechanical recycling.
Purity is key for producing high-quality recycled material, Riise stated. The recycled material must behave like virgin polymers when processed, have a suitable appearance, and be free of problematic additives, such as brominated flame retardants. For that to occur, plastics must be cleaned, separated from non-plastic materials, and sorted according to polymer type. Plastic–plastic separation is critical and can require several steps in a precise sequence. Various technologies—optical, density, and electrostatic sorters—have been developed to separate plastics; their use depends on the types of materials and mixtures that need to be sorted.
Once the material stream is sorted sufficiently to achieve the target level of purity, the flakes are typically compounded and formed into pellets that can be used in place of (or mixed with) virgin material. Although some buyers of recycled materials can use plastic flakes, Riise noted that pellets are generally preferred. Depending on the intended use of the material, additional processing might be necessary to remove volatile compounds, water, and odors or to incorporate additives for desired properties, such as flexibility, stability, color, or impact strength. Riise stated, however, that using additives to adjust material properties can be problematic in that additives can be expensive, and there can be unintended consequences, for example, improving some properties might hurt other properties. Various tests to measure the composition and mechanical properties are used to ensure product quality.
Riise listed several key challenges to mechanical recycling, such as stringent separation requirements to produce materials that have sufficient purity, degradation of plastics during processing and use, and difficulties in managing complex streams and removing colorants, additives, residues, and odors. Approaches to those challenges include improvement in mechanical separations, cleaning technologies, and analytic techniques; the use of additives; dilution of virgin plastics with recycled materials; and the use of recycled materials in the most appropriate applications. Although technical limitations and economic challenges remain, Riise concluded that mechanical methods can effectively recycle plastics from complex streams and bring us closer to a circular economy.
Spectroscopic Methods for Improving Sorting of Plastics
André Bénard, an associate professor at Michigan State University, discussed opportunities for improving mechanical recycling with spectroscopy. He stated that over the last 15 years spectroscopy has emerged as a valuable tool for identifying materials in a mixed-waste stream. An array of spectroscopic technologies are available for assessing various material properties, such as near- and mid-infrared spectroscopy, Fourier-transform infrared spectroscopy, Raman spectroscopy, laser-induced breakdown spectroscopy, and hyperspectral imaging. Bénard discussed how those are increasingly used in material-recovery centers in combination with sensitive detectors, image analysis, machine learning, and robotic technologies.
Bénard stated that fast x-ray fluorescence (XRF) and coherent anti-Stokes Raman spectroscopy (CARS) are the latest spectroscopic technologies to be adopted for sorting waste streams at material-recovery centers. XRF, an older technology, is being combined with high-speed computation for sorting materials precisely; it is used primarily for metals but can also be used for plastics. CARS and single ultrafast pulse excitation for remote coherent anti-Stokes Raman spectroscopy (Bremer and Dantus 2013) are sophisticated Raman spectroscopy techniques that can rapidly detect and sort plastics moving along a conveyer belt. By enabling fast, accurate, and automated sorting, these technologies hold great potential for increasing the efficiency of plastics recycling, Bénard concluded.
Compatibilizers
Megan Robertson, an associate professor at the University of Houston, discussed an alternative approach to separation of plastics—development of methods for recycling them together by making it possible to combine different polymers into a blended product (Xu et al. 2018). Robertson stated that most polymers are immiscible and mixing them creates a phase-separated mixture that has poor properties. She outlined how some of the principles used to design surfactants for oil–water mixtures could be useful for designing compatibilizers, additives that enable blending of polymers.
Robertson noted that effective compatibilizers will stabilize the interface between immiscible polymers and thus enhance interfacial adhesion or strength, and she described several compatibilizer designs that have demonstrated success (Morkved et al. 2001; Ruegg et al. 2007). She continued that compatibilizers can be designed to have different architectures and to use diverse interactions (for example, van der Waals, dipole interactions, and hydrogen bonding). They can be synthesized and added to a mixture or formed in situ during the blending process.
Although compatibilizers are available for particular polymer mixtures, they have not been widely adopted for recycling plastics, Robertson said. One reason is cost. However, research has demonstrated that fine-tuning of the architecture and molecular-weight distribution of a compatibilizer can maximize its efficiency and reduce the necessary concentration and thus the cost of its use (Xu et al. 2018). Because most compatibilizers are optimized for specific combinations of polymers and specific conditions, other key challenges are related to handling of variable mixtures of many polymers, materials that have undergone thermal degradation, and materials that contain additives.
The “holy grail” of compatibilization is the ability to combine all types of plastic waste to create a marketable output. Robertson suggested, however, that it might be more realistic to consider compatibilizer development as complementary with sorting systems that can group plastics into mixable streams. Nonetheless, researchers are exploring promising avenues toward potential “universal compatibilizers,” including ones that use nanoparticles, reactive (in situ) compatibilization, or several types of compatibilizers simultaneously.
DISCUSSION
Tim Patten, deputy director of the Chemical, Bioengineering, Environmental, and Transport Systems Division of the National Science Foundation, moderated an open discussion that touched on key research needs and current and future challenges in the mechanical-recycling landscape. The session began with a reflection from industry.
Industry Perspective on the Plastics Dilemma
Jill Martin, a fellow with the Dow Chemical Company, stated that achieving a circular economy requires planning for a product or material to have multiple lives. One has to consider what happens at the end of the first life—Is the plastic recycled into the same market with the same uses, or is it converted to its building blocks to create a new product with different uses? Martin emphasized that life-cycle thinking is critical for finding solutions to the plastics problem. She noted that Dow uses life-cycle assessment to consider how well different solutions meet short- and long-term goals for decreasing greenhouse-gas emissions or lowering energy and water use, as well as such business considerations as economic feasibility and scale.
Martin stated that Dow invests in compatibilizer research. It is vital to consider how compatibilizers will be used—at what stage and by which party they will be added to the process and where their use will create value. She added that industry investments in compatibilizers and other advances are not intended to solve the whole problem but rather to provide “catalytic capital” that can enable long-term change in the systems and infrastructure for creating a circular economy.
Recycling offers substantial economic opportunity, Martin said. The recycling industry in North America generated $100 billion in value in 2018.2 She emphasized that building on the opportunity will require focusing on innovations and identifying the key players who can transform ideas into reality on a large scale. She concluded that it will be crucial for the plastics industry to build partnerships with converters, brand owners, and waste-management companies that can create the infrastructure necessary to solve the plastic-waste crisis.
Enabling Future Advances
Prompted by a question from Patten, panelists identified the following key advances as important for improving mechanical recycling:
- Riise identified cost-efficient methods for sorting black plastics as a critical need. Although methods exist, he noted that they are more expensive than today’s dominant technologies.
- Robertson highlighted the need to develop compatibilizers that can blend more diverse mixtures or to make waste streams more homogeneous so they are easier to compatibilize.
- Bénard pointed to the need to transfer technologies from other fields. There is an opportunity to take advantage of technologies, such as density separation methods used in mining and various spectroscopic tools, for sorting recyclables.
- Beckman and Martin focused on designing for simplicity and recyclability. Designing new materials—and revisiting the design of old ones—with a focus on simplicity and recyclability rather than function and cost could substantially reduce the complexity of recycling. A key challenge that Martin identified is to develop materials that perform well in multiple applications.
Challenges in a Shifting Landscape
A workshop participant raised a question about the composition and quality of recycled material. If recycling rates increase dramatically, a greater and greater proportion of material being recycled will have already been recycled, and this potentially would complicate the recycling process. Riise replied that the degree to which that is a problem depends on what products are created from recycled materials and their corresponding lifespans; chemical recycling methods or improvements in mechanical processing to stabilize recycled materials could also help. Robertson added that this issue also suggests a need for better analytic techniques to characterize material inputs, such as a “molecular barcode” reader, which Beckman theorized could identify the origin and properties of plastics as they enter the recycling pipeline. Beckman added that recycling practices might be affected by new business models that emphasize sharing and reuse of products rather than permanent ownership.
The development of degradable materials presents other challenges and opportunities. Riise noted that a material designed to degrade cannot be recycled; furthermore, these materials can undermine recycling systems unless waste streams are kept separate. That issue is one illustration of the broader tension between solutions that might be “best” when considered in a vacuum and solutions that are useful in the context of today’s infrastructure, he added. Beckman raised one potential solution: to be strategic about which items are made of degradable materials. For example, cigarette butts and fishing gear are notorious for being difficult to collect or recycle and, because of how they are used, often dissipate into the environment. Items that are likely to dissipate anyway, he noted, might be prime candidates for a shift to degradable materials.
___________
2 See https://www.closedlooppartners.com/3-reasons-why-recycling-is-good-business-in-america-and-a-key-driver-of-the-circular-economy/.
A workshop participant raised a question about sorting capabilities. It is useful to have a baseline to understand the efficiency of existing sorting technologies. However, Riise and Beckman pointed out that those data are often proprietary and therefore difficult to obtain. Martin, however, said that separating paper from rigid plastic, glass, and metal is highly efficient, whereas separating flexible packaging from paper is difficult. Beckman and Riise agreed that there are trade-offs between throughput, purity, and cost; 99% efficiency of the sorting processes might be possible but might require a large capital investment.
Attendees discussed the role of consumers in sorting recyclables. Beckman noted that designing for recyclability is not only about designing the polymers; consumers’ perspective also needs to be considered. It is important for consumers to understand easily what can be recycled so that they can act accordingly. Riise pointed out that many current systems use a single-stream approach, so sorting before curbside collection would not make a difference unless the processing infrastructure were also changed.
Finally, participants discussed the difficulty of weighing the many trade-offs involved from among various solutions. Although life-cycle assessment is an important tool, Beckman noted, it is difficult to compare existing materials or systems with those envisioned but not yet created. In addition, many speakers agreed that there is a lack of tools for informing decisions in the face of competing considerations—for example, human health or ecosystem effects vs carbon emissions vs functional properties of materials.
CHEMICAL AND BIOLOGIC RECYCLING OF PLASTICS
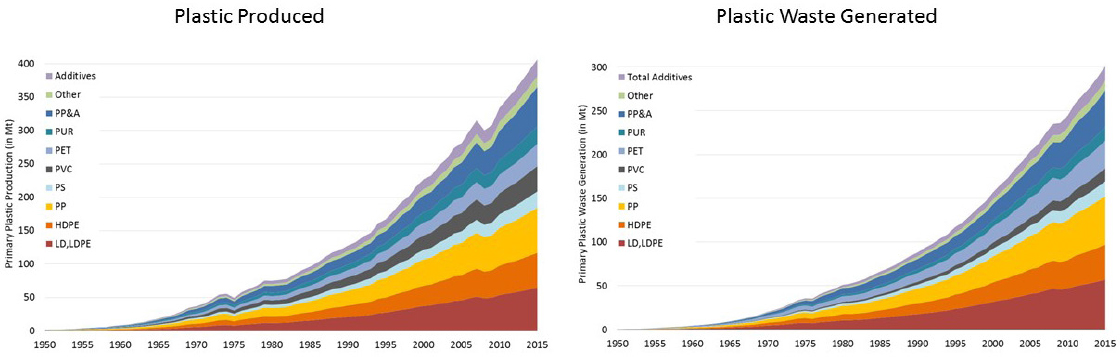
The second workshop session opened with an overview of chemical and biologic recycling by Jeannette Garcia, global lead for Quantum Applications in Quantum Chemistry and Science at IBM Research. Garcia explained that current recycling methods fall into four categories: primary recycling or reuse (recycling products for the same use), secondary or mechanical recycling (recycling products for uses other than their original use), tertiary or chemical recycling (recovering petrochemical components of plastics via a chemical process), and quaternary recycling or energy recovery (incinerating plastics to recover energy in the form of heat). The discrepancy between the large volume of plastic waste generated annually (more than 300 million tons globally) and the proportion of that waste that is recycled (about 9% in the United States) underscores both the magnitude of the challenge and the untapped opportunity to recover energy and materials (Figure 2).
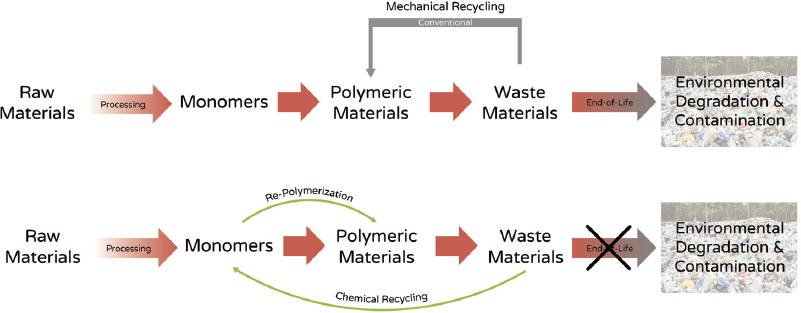
Garcia compared mechanical and chemical recycling processes (Figure 3). The goal of mechanical recycling is to convert waste materials to clean polymeric materials for reuse. A disadvantage of mechanical recycling is that it typically changes the molecular or mechanical properties of the materials, for example, by altering molecular weight or elongation properties through random scission and cross-linking. Chemical recycling, however, aims to have the polymers revert to their monomers so that there is no “end of life,” and release into the environment or into landfills is avoided.
Researchers are pursuing various approaches to recycling of plastics chemically and biologically, Garcia said. Examples are chemical strategies to “attack” the functional group or repeating unit of a polymer (Fukushima et al. 2011, 2013), enzymatic approaches to transform a polymer into useful building blocks (Yoshida et al. 2016), thermolytic techniques to convert polyolefins into liquid fuels (Wong et al. 2016), chemical methods for depolymerizing polyolefins (Williamson et al. 2018), and the use of living organisms to degrade materials (Yang et al. 2015). Garcia concluded by noting that researchers are also actively pursuing innovative strategies to design new polymers that have built-in mechanisms to facilitate chemical recycling (Garcia et al. 2014; Zhu et al. 2018; Christensen et al. 2019).
Thermal Depolymerization
Paul Dauenhauer, an associate professor at the University of Minnesota, continued the conversation by discussing the reaction-engineering fundamentals of thermal recycling of polymers. The thermolytic process of reducing polymers into smaller units that can be reassembled into new polymers involves two chemistries—melt-phase chemistry and secondary gas-phase chemistry. To develop a pyrolysis reactor that can generate the desired compounds, three length scales—reactor scale (about 1 m), particle scale (about 1 mm), and atomic scale (about 1 angstrom)—and three phases need to be considered. Dauenhauer noted that the particle scale is complicated immensely by phase change and that it is desirable to understand the chemistry that occurs when a material melts. It is also important to understand aerosol generation; aerosols are problematic because they can short-circuit the chemistry and can deposit in downstream unit operations.
Experiments with diffuse reflectance in situ spectroscopy of particles (Paulsen et al. 2014) and pulse-heated analysis of solid reactions (Krumm et al. 2016) have shed light on the foundational chemical and physical processes involved in the pyrolysis of polymers. Dauenhauer emphasized the importance of the new techniques because one cannot describe a reactor fundamentally unless one understands what is happening on the particle and molecular scales. To inform reactor design, Dauenhauer highlighted the value of a building-up approach to pyrolysis modeling by first integrating reaction models into particle models that are then integrated into reactor models. He closed, however, with a warning that the systems are extremely complex and that one needs to review methods carefully to understand what the data mean and whether they are relevant to a particular system.
Putting fundamental principles into practice, Jennifer Le Roy, research and development lead at BioCellection, Inc., described how her company is working to scale thermal oxidative depolymerization processes for converting plastic waste into new products. BioCellection recycles mixed polyethylene feedstock into short-chain dicarboxylic acids, such as adipic, succinic, and azelaic acids—virgin-grade chemical intermediates that have sizable markets. Their process can use different polyethylene sources, does not require that materials be cleaned before processing, and uses feedstocks that the company is being paid to take. Although many of the process details are confidential, Le Roy shared that the company continues to perform research and development to satisfy customer demands and reduce costs while it moves to scale up manufacturing capabilities with support from industry partners that assist with product validation.
Catalytic Depolymerization
Susannah Scott, Distinguished Professor of Chemical Engineering at the University of California, Santa Barbara, switched topics and discussed catalytic depolymerization. She focused on identifying transition-metal catalysts that could be used for polymer disassembly. Although there are many well-known transition-metal catalysts that transform small molecules or create polymers, polymer disassembly is a novel use that is based on the principle of microscopic reversibility.
Scott stated that an important barrier is that there is no universal catalyst that can be used for every polymer disassembly; a catalyst needs to be paired with a polymer on the basis of the chemistry involved. She continued that kinetics and thermodynamics present other barriers. Because plastics have been designed to be thermodynamically stable, conditions must be adjusted to make depolymerization favorable. Scott noted that if depolymerization is strongly unfavorable thermodynamically, using a catalyst will not help. Even creating thermodynamically favorable conditions can require high temperatures and a large expenditure of energy.
Scott highlighted experiments that demonstrate potential opportunities to overcome the barriers. One approach is to reverse the polymerization by using a reaction pathway different from the one used to polymerize the material (Krall et al. 2014; Rorrer et al. 2019). The key is to convert the polymer not to its building blocks but to commercially useful products, such as oligomers or different polymers—in other words, change the chemistry to improve the thermodynamics. For mixed plastics, researchers have experimented with sequential catalytic reactions by adjusting temperatures or by using multiple solvents to depolymerize particular polymers selectively (Westhues et al. 2018). Scott noted, however, that the examples
provided are not suitable for large-scale processes; there is no cost-competitive catalytic route today for chemical recycling. That is, recovered monomers are generally not competitive with fossil-derived ones. She concluded by stating that future technologies must have low energy and solvent requirements, easy separations, robust catalysts, and value-added targets.
Delving deeper into a specific catalytic depolymerization process, Robert Allen, senior manager of chemistry and materials at IBM Almaden Research Center, described the development of a glycolytic process for depolymerizing PET by using trimethylamine as a catalyst. About 150 billion pounds of PET is manufactured each year, and only a small fraction is recovered and reused. Mechanical recycling methods are available for PET, but they require high temperatures, low or no contamination, and non-colored materials. Although chemical recycling methods have potential and various approaches are being pursued, economic feasibility has presented a substantial barrier. Allen’s team at IBM created the VolCat process, which could offer a cost-effective means of recycling PET chemically.
VolCat is a simple, fast, highly selective process that uses a gentle, volatile, low-cost catalyst that is completely recovered and recycled. The process is highly tolerant of contamination and produces high-grade bis(2-hydroxyethyl) terephthalate that has good transparency and color and is well suited for reuse (Figure 4). Downstream processing is simple and effective and generates essentially no process waste. VolCat depolymerization can even be used for selective digestion of polyester in a cotton–polyester blend and thus offers a potential route to textile recycling. With further optimization and scaling, Allen said that VolCat can help PET to become a renewable resource instead of an environmental scourge.
Biologic Depolymerization
Richard Gross, professor and Constellation Chair at Rensselaer Polytechnic Institute, discussed opportunities for biocatalytic depolymerization of plastics. Biocatalysis is promising because it offers mild and selective processes that can target a specific polymer substrate in mixed-waste streams. Scientists also have the benefit of powerful tools to re-engineer enzymes to improve reaction rates and conditions. Because enzymes degrade water-insoluble polymers at their surfaces and constantly create new surfaces, enzyme degradation can be enhanced by increasing a polymer’s surface area. Furthermore, because crystalline regions are more difficult for enzymes to access, plastic substrates should be pretreated to reduce material crystallinity.
Gross stated that nature provides useful leads in the selection of candidate enzymes. One approach is to investigate the microbial communities that form around plastic waste, which appear to be evolving in ways that degrade the plastics. The enzyme activity can be improved by using rational and directed evolutionary approaches. Gross noted that although microorganisms themselves could be used to degrade plastics, expressing an enzyme and then applying it directly to a plastic is generally more efficient in producing usable products. Otherwise, the microorganism can use a substantial fraction of the degradation products for its own maintenance of metabolism and reproduction instead of converting plastics to degradation products for reuse.
Gross highlighted research on cutinase, a powerful natural hydrolase that breaks down the outer layer of leaves and has been shown to degrade PET (Ribitsch et al. 2015). He noted that the cutinase family has broad potential for breaking down various polymers and that leaf and branch compost cutinase has shown particular promise since researchers stabilized the enzyme via glycosylation (Shirke et al. 2018). His group is exploring how fungal enzymes that break down lignin might be applied to plastics that have structural features that resemble those of lignin. Gross emphasized that as further work uncovers a wider array of effective polymer-degrading biocatalysts, there will be an opportunity to identify synergies between these catalysts to accelerate enzyme-catalyzed polymer degradation. He also stressed that it will be critical to use a combination of chemical and enzymatic catalyst systems in which, for example, chemical catalysis will break large chains into smaller fragments that are then best converted to recyclable products via enzymatic processes. He added that new approaches to materials design could complement this multifaceted effort.

Eric Boyd, an associate professor at Montana State University, continued the conversation on biologic depolymerization by describing his efforts in “bioprospecting.” He focuses on identifying enzymes that have the potential to degrade PET (PETases). He noted that PET needs to be heated to above 70°C to decrease its crystallinity and thus enhance its enzymatic degradation. Given those requirements, the enzymes need to be heat-stable. Boyd said that one approach is to bioengineer an enzyme to be thermotolerant. However, his approach is to probe natural systems for a thermophilic PETase homolog or PET-degrading organism, and his preferred site for this bioprospecting is Yellowstone National Park, which has exceptionally dynamic, extensive, and chemically diverse hydrothermal features.
Using metagenomic techniques and protein clustering, Boyd can identify potential candidates to investigate from among the microorganisms that inhabit the extreme ecosystems. He stated that he has identified a catalytic triad—three residues that are conserved among all the esterases that have PETase activity—that can be used to narrow the list of candidates for investigation. Once the most promising candidates are identified, the genes can be isolated and expressed, the enzymes purified, and their activity assessed. Boyd provided data on two particularly promising candidates from his research. Next steps, he concluded, are more metagenomics analyses to identify additional PETase activity, further development of screening assays for PETase activity, and investigation of the possibilities of modifying enzyme structure to improve PETase activity or engineering a thermophile to have PETase activity.
DISCUSSION
Kathryn Beers, a group leader in the Materials Science and Engineering Division of the National Institute of Standards and Technology, moderated the panel discussion with the speakers and audience. Beers asked the speakers to name the most important advance that would propel chemical and biologic recycling forward. Boyd and Allen emphasized the need for different funding mechanisms, especially ones that would support cross-disciplinary collaboration. Several speakers discussed the need to pursue synergies among mechanical, chemical, and biologic recycling methods; to integrate life-cycle assessment and technoeconomic analyses into the process; to create academic–industry partnerships; and to encourage a greater availability of industry data that would help to inform priorities and speed efforts. Allen called for a better understanding of the scientific fundamentals, and Gross added that standard materials are needed so that results can be better compared across laboratories. Garcia noted the need for data collection over a plastic’s entire life cycle, and Dauenhauer and Le Roy highlighted the need to provide thorough data on total reaction outputs.
Several participants touched on some broader issues in the context of the overall plastics-recycling ecosystem and asked whether the focus should be on designing new plastics without being constrained by the current chemical industry. Gross emphasized the need for parallel efforts to address the current plastic-waste problem within the chemical industry and to design better materials for future collection, reuse, and recycling. Scott argued that designing all plastics to be biodegradable is clearly not a feasible approach in light of the world’s population’s simply discarding plastics into the environment. The key, she said, is to redesign products so that recovery, disassembly, and repurposing are much more efficient. Gross added that building in triggers to enhance chemical recycling at the end of a product’s life should be pursued.
A participant asked whether the focus should be on compostable plastics. Beckman noted that one problem is that composting generates greenhouse gases. Gross argued that the ideal is an enzymatic process that creates chemicals, not a microbial process that generates biomass. Another participant asked whether the focus should instead be on reuse. Garcia stated that although reuse makes sense, the material is eventually going to become waste and a combination of all the various approaches will be needed. Finally, a participant asked whether the problem is related to technology or to consumers. Scott stressed that the plastics already in the environment pose a technologic problem and that if consumers continue to demand cheap, convenient single-use plastics, redesigning plastics for the future will be difficult.
DESIGNING SUSTAINABLE PLASTICS
The third workshop session focused on redesigning today’s plastics to be more environmentally sustainable. Gregg Beckham, senior research fellow at National Renewable Energy Laboratory, moderated the session.
Biobased Polymers
David Hodge, an associate professor at Montana State University, highlighted opportunities to use renewable sources of carbon instead of fossil-based carbon as a primary feedstock for polymers. Examples of renewable sources of carbon are plant biomass, algae, and direct carbon dioxide capture. Hodge stated that food crops—such as cereal grains, sugarcane, and sugar beets—have traditionally provided the biomass raw materials. The goal, however, is to move to lignocellulose biomass, such as agricultural residues or dedicated energy crops.
Biobased feedstocks offer some key advantages, Hodge said. They have high oxygen content and can be integrated into biocatalytic approaches that use aqueous conditions at moderate temperatures. Hodge continued, saying that biobased chemicals have numerous potential applications—opportunities for direct replacement, functional replacement, and novel products that offer new properties that provide improved performance for consumers or for degradability or recyclability. Researchers and companies are actively pursuing various applications (Biddy et al. 2016; Shanks and Keeling 2017).
Hodge stated that technologies for biomass deconstruction and conversion could potentially be used to enhance chemical recycling capabilities. Plant cell walls, which are composed of various polymers, are recalcitrant to deconstruction—
lignin prevents access to the carbohydrate fraction, and cellulose embedded in crystalline microfibrils resists depolymerization (Himmel et al. 2007). For decades, researchers have pursued methods to overcome those barriers with thermal, chemical, and biologic approaches. The techniques that have been developed for extracting the components of biomass feedstocks could be used for biocatalytic or hybrid approaches to break down polymers in post-consumer plastics (Wierckx et al. 2015). Questions remain as to how cost-effective and scalable the approaches might be and whether they could be applied to more complex products, such as thermoplastics, thermosets, and composite materials.
New Building Blocks for Polymers
Geoffrey Coates, Tisch University Professor in the Department of Chemistry and Chemical Biology of Cornell University, continued the discussion on sustainable plastics by emphasizing what is required in the pursuit of an ideal plastic. It should be derived from renewable sources, facilitate decomposition or recycling, have properties equal to or better than those of current plastics, and be cost-equivalent or less expensive than current plastics. Furthermore, the synthetic methods for producing them should use less energy and raw materials.
Coates described how carbon dioxide and carbon monoxide can be used as building blocks to create new biodegradable polymers. The molecules are inexpensive, are readily available, and introduce hydrolyzable linkages into polymer backbones that facilitate decomposition. The biggest challenge, however, is that a huge thermodynamic barrier must be overcome. Researchers have been working for decades to find ways to overcome that hurdle by using catalysts and reactive molecules, such as epoxides, to drive carbon dioxide polymerization. Coates and his research team have successfully used that approach with various catalysts to make various materials (Qin et al. 2003; Cohen et al. 2005). Further experimentation and business development have yielded several applications that offer opportunities for scale-up and commercial applications, such as the use of foams in vehicles.
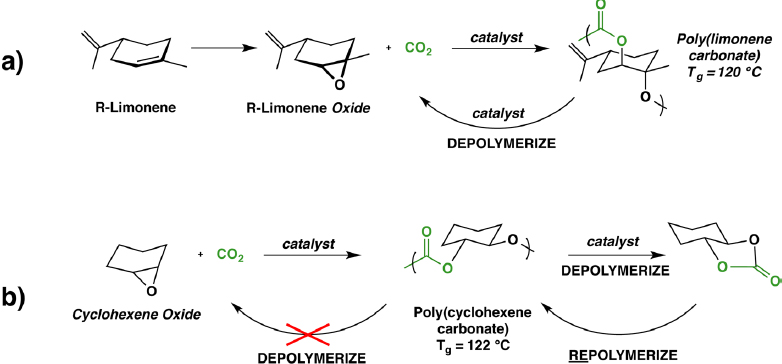
A major focus of his work is in the design of polymers for degradability or recyclability. Coates described a process for creating a polymer from carbon dioxide and limonene (a natural product of tangerines) that can be used to make containers that are easily depolymerized to the starting materials (Figure 5a). A similar process can be used to create polycyclohexene carbonate that can be depolymerized to a small molecule that can be converted to the polymer (Figure 5b). Coates stated that another promising avenue uses succinate anhydrides to create non-toxic materials that are more heat-tolerant than the materials derived by using carbon dioxide (Longo et al. 2014).
Nature has provided inspiration for using carbon monoxide as a building block for polymers, Coates said. Poly(ß-hydroxybutyrate) is a polymer made by bacteria for energy storage that has potential commercial applications, such as in milk jugs and detergent bottles that would be compostable. The problem is that fermentation processes to make the polymer are not ideal from a cost perspective or a carbon-footprint perspective. However, Coates and his research team have developed a promising approach that uses carbon monoxide, an epoxide, and a bimetallic cobalt catalyst to create poly(ß-hydroxybutyrate) (Dunn and Coates 2010). Coates concluded by saying that the bimetallic cobalt catalysts suggests new approaches for synthesizing acrylic and succinic acids, high-value commodity products.
Designing for Chemical Recyclability
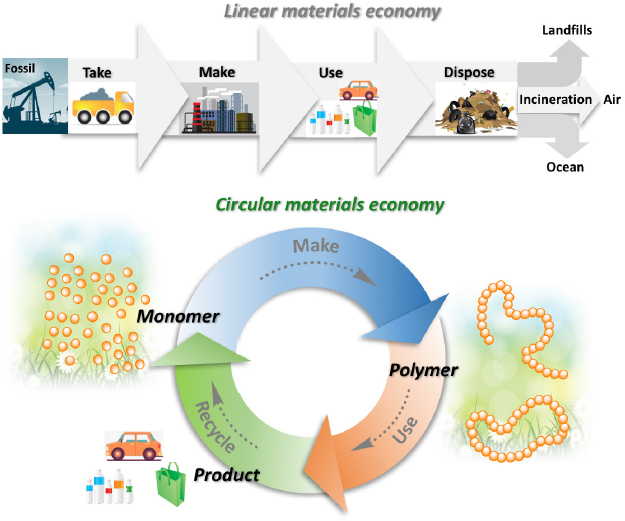
Eugene Chen, John K. Stille Endowed Chair and Millennial Professor of Polymer Science and Sustainability at Colorado State University, discussed his work to create materials that are infinitely recyclable and compared the linear and circular economies (Figure 6). He contrasted incineration and mechanical, biologic, and chemical recycling. In his view, chemical recycling methods are the most promising for creating the circular economy. Key challenges, however, include minimizing energy inputs for depolymerization, maximizing depolymerization selectivity, and balancing recyclability and performance.
As noted by other speakers, thermodynamics is a fundamental consideration: temperature, enthalpy, and entropy are key determinants of polymerization potential. Chen described experiments that elucidate thermodynamic, kinetic, and process conditions required for the synthesis of a class of polymers that has a low ceiling temperature and can be quantitatively depolymerized into pure monomers that can then be repolymerized (Hong and Chen 2016). Chen’s team ultimately created a high-molecular-weight polymer under mild conditions and room temperature that had thermal stability and high crystallinity and that could easily be depolymerized in the presence of a simple Lewis acid catalyst, such as ZnCl2. The recovered pure monomer could then be repolymerized to virgin-quality material (Zhu et al. 2018). Others have had similar success with different pathways (Yuan et al. 2019).
The ability to compete with today’s plastics will be crucial for the success of the new materials; cost and performance will be critical. With respect to research methods, Chen noted that computationally assisted design is useful for investigating infinitely recyclable plastic systems—both to develop new materials and to improve chemical recycling methods.
Modeling and Predictive Analysis
Linda Broadbelt, Sarah Rebecca Roland Professor and associate dean for research in the McCormick School of Engineering and Applied Science of Northwestern University, discussed how computational modeling and data science can be used to advance chemical recycling strategies and polymer design. She stated that those techniques are being used to study complex reaction systems, to design copolymer architectures, and to identify “bioprivileged” molecules.3
Broadbelt described three types of models that have different levels of complexity. Lumped models do not differentiate reaction products and use an overall rate coefficient. Pathway-level models are more complex; they differentiate reaction products but still lump rate coefficients. Mechanistic models are the most complex; they distinguish reaction intermediates and products and incorporate estimated or experimentally derived reaction coefficients for elementary steps. She discussed how such approaches have been applied to model the degradation of various polymers and mixtures (Kruse et al. 2005; Levine and Broadbelt 2009).
Ab initio molecular simulations, another approach, can yield insights into enzymatic behavior and even predict kinetic parameters, Broadbelt said. She and colleagues applied that approach to predict enzymes that could decompose polymers that are not their native substrates. They then complemented the simulation with machine-learning methods to rank the catalytic potential of enzymes for substrates other than their native ones.
Broadbelt described a third approach—using data science to identify bioprivileged molecules. She and colleagues started by creating a candidate list of C6HXOY molecules and developed a reactivity index as a proxy for a full reaction network. They used data science to compare reactivity indexes of candidate molecules and benchmark molecules to narrow the list from over 29,000 to about 4,600, to which the researchers applied a reaction-network generation approach to provide an overall ranking (Zhou et al. 2019). Although data science provides valuable tools for accelerating discovery, Broadbelt cautioned that it does not supplant the need for atomistic information and kinetic modeling. Rather, modeling, data science, and machine learning can be complementary; models, for example, can provide training and validation data to artificial neural networks.
___________
3 Bioprivileged molecules are ones derived from nature that can be used to create diverse chemical products (Shanks and Keeling 2017).
DISCUSSION
Participants highlighted some of the trade-offs and challenges involved in designing plastics for a circular economy. Much of the focus is on designing for production and recycling capabilities, but a participant stated that the middle piece—use—is also important. For example, plastics used for food applications typically need to withstand high-temperature sterilization processes, and a plastic designed to be susceptible to thermal depolymerization would not be suitable for such an application. Coates agreed that performance and recycling capabilities are often in conflict because materials that are designed to degrade easily might not have the robust physical properties required for many applications. Chen stated, however, that researchers have made metastable polymers that are “kinetically trapped” and have triggers—activated by such external stimuli as catalysts, thermal energy, and light—that can control and facilitate depolymerization. Chen noted that computationally assisted design can aid in the development of such materials, and Broadbelt added that modeling can also help in evaluating the effect of weathering on recyclability.
Susannah Scott asked about the challenges of designing polymers that have multiple ends of life and the difficulties that can arise when plastics intended for one process (such as mechanical recycling) end up in another process (such as chemical recycling). That question, said Chen, emphasizes the need for a systematic change in polymer coding and collection. A workshop participant added that designing plastics for recyclability is complicated given their many uses; multiple metrics for design will be required to make recyclability feasible. Regarding multiple lives, a workshop participant noted that something that was considered a waste in the past might not be one forever; the goal is that one day it will be a feedstock for another process.
Finally, Richard Gross questioned the innovative process for creating new materials. Coates, Chen, and other participants noted the need to balance fundamental discovery of new materials with research that focuses on sustainability and scalability from the outset. Chen cautioned against inhibiting innovation—something that is not commercially viable today might be viable in the future with further scientific advances. In response to a participant’s question, Broadbelt, Coates, Hodge, and Chen discussed the importance of updating training and education systems to incorporate sustainability techniques while maintaining fundamental and applied learning. Speakers and participants agreed that it is essential to equip the next generation of scientists and engineers to tackle the plastics challenge effectively.
LIFE-CYCLE ASSESSMENT OF PLASTICS
During the workshop, life-cycle assessment (LCA) in the context of plastic production and waste management was discussed. Presentations were made by Ming Xu, an associate professor at the University of Michigan, and Michael Wang, Distinguished Fellow and senior scientist at Argonne National Laboratory.
Overview
Xu began by providing an overview of LCA (Figure 7). LCA is a technique used to evaluate a product’s environmental footprint and takes into account its full life cycle, from extraction and processing of the raw materials to manufacture, distribution, and use of the product to its recycling or disposal. LCA can be used to inform comparisons among products, processes, or end-of-life pathways; to identify the key points in the life cycle that have the biggest environmental effects; to avoid “burden-shifting” from one process to another; and to guide technologic innovation, policy, and consumer decision-making. Xu noted that the publication of LCAs of plastics has increased dramatically since 2005. Three main applications are in comparison of biobased plastics with other materials, identification of “hot spots” for improvement, and life-cycle cost analysis.
Xu described the steps of an LCA. One has to define first the goal and scope of the assessment, such as determination of the environmental effects that need to be compared. One then prepares an inventory analysis in which technical inputs and outputs, resource and energy consumption, and emissions associated with all the various processes are estimated.
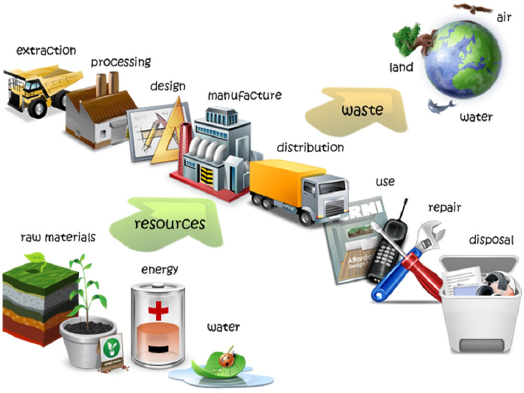
Next, one conducts the life-cycle impact assessment in which the environmental effects of all the factors in the inventory are quantified and aggregated into three categories: effects on human health, effects on ecosystem health, and effects on resource depletion. The final step is interpretation, in which the assessor communicates what was and was not included in the analysis so that people can make a fair comparison. Xu noted that the selection of factors to include in the assessment has an important influence on the results and how they should be interpreted.
Insights on Plastics
Wang offered examples of how one LCA tool, the GREET model developed by Argonne National Laboratory,4 has been used to analyze alternative means of producing plastics and managing plastic waste. The first case study compared fossil-based PET with bio-derived PET or recycled PET bottles (Benavides et al. 2018). Researchers were able to pinpoint the major sources of greenhouse-gas emissions and determine which pathways result in significant emission reductions. They concluded that bio-derived and recycled PET substantially reduce fossil-fuel consumption and greenhouse-gas emissions. The biogenic carbon credit assigned to biomass-derived components of PET bottles was a key driver of the emission reduction estimated for bio-derived PET. Furthermore, the production of terephthalic acid as a precursor in the pathway of the bio-derived PET was a major contributor of emission.
The second study that Wang discussed compared fuels derived from pyrolysis of non-recycled plastics with conventionally produced petroleum fuels. It was particularly important to account for all the co-products that are created when plastic is pyrolyzed; Wang noted that different LCA methods account for co-products in different ways, which can yield different bottom-line results and confuse the public. The study concluded that plastic-derived fuels generate somewhat less greenhouse gases than petroleum fuels but are far less water-intensive and fossil-fuel–intensive. Given that the United States produced 29 million tons of non-recyclable plastics in 2013, adoption of this technology could yield some 6 billion gallons of diesel fuel per year—roughly 10% of the US diesel-fuel market—and substantially reduce landfill inputs. In Wang’s view, the findings suggest that plastic-to-fuel technology offers an alternative waste-management strategy and a promising way to extend the value of plastics.
Xu described other LCA studies focused on plastics. One of the earliest plastic LCAs compared plastic and paper products and found that plastic products resulted in lower energy consumption and air emissions (Hocking 1991). A more recent study of baby-food packaging found that plastic pots had a lower environmental footprint than glass jars (Humbert et al. 2009). Xu stated that many LCA studies focus on greenhouse-gas emissions associated with various polymer types. A recent study, for example, estimated that upcycling carbon dioxide into polymers reduces greenhouse-gas emissions by 11–20%, and that use of bio-derived plastics reduces emissions by 20–50% compared with fossil-fuel–derived plastics (Zhu et al. 2016). Other studies examine different effects, such as ozone depletion, acidification, and ecotoxicity; by these measures, bio-derived products are not always better than products based on fossil fuels (Tabone et al. 2010).
Xu said that researchers have used LCA to identify priorities for improvements in the manufacturing process (Cheung et al. 2017) and to assess different end-of-life options. A study that compared plastic-film disposal, incineration, and recycling found that recycling of a mixed-waste stream has the greatest environmental benefit (Hou et al. 2018); the researchers also conducted a sensitivity analysis to determine which parameters were most important with respect to life-cycle effects. Xu emphasized that such analyses reveal areas with the greatest potential for improvement, which can be addressed by both technical innovation and policy incentives.
Challenges
Wang and Xu highlighted several challenges regarding the application and interpretation of LCA. Other workshop participants raised additional considerations during open discussion sessions.
Xu identified data as a key challenge. Compiling data from such disparate sources as laboratory tests, questionnaires and surveys, onsite investigation, and the scientific literature is expensive and time-consuming. Xu emphasized that data are vital for generating a life-cycle inventory and are typically captured in a matrix that can include thousands of columns and rows. Where data are unavailable, researchers typically turn to industry averages or proxies. Xu stated that researchers are actively pursuing ways to acquire data more efficiently, improve the reliability of proxies, and generate new data from existing data by using data science. However, Xu and Wang both noted that the only way to validate the assumptions included in such processes is to acquire more data from primary sources, and this takes time and money.
Noting that the traditional LCA framework assumes that a given product will have a single function, Wang pointed to a lack of both data and analytic tools to quantify all the tradeoffs involved in a truly circular economy in which a given product or material has multiple uses or multiple lifetimes. Because different uses and design goals have different sustainability implications, LCA tools will need to be expanded to account for the effects generated (or avoided) in a circular economy.
In response to a participant’s question, Xu discussed the opportunities and potential pitfalls related to using LCAs to analyze the effects of emerging designs or processes that have not yet been deployed. Researchers often make “reasonable” estimations that are based on available information, but the availability of information—even laboratory-scale information—varies case by case. Another approach is to use data-driven computational approaches, but these are themselves emerging. In either case, he said, it is important to estimate the reliability of the data and of the assumptions that are being made.
___________
REFERENCES
Benavides, P. T., J. B. Dunn, J. Han, M. Biddy, and J. Markham. 2018. Exploring comparative energy and environmental benefits of virgin, recycled, and bio-derived PET bottles. ACS Sustainable Chem. Eng. 6(8):9725–9733.
Biddy, M. J., C. Scarlata, and C. Kinchin. 2016. Chemicals from biomass: A market assessment of bioproducts with near-term potential. National Renewable Energy Laboratory Technical Report 5100-65509.
Bremer, M. T. and M. Dantus. 2013. Standoff explosives trace detection and imaging by selective stimulated Raman scattering. Appl. Phys. Lett. 103:061119.
Chalmin, P. 2019. The history of plastics: from the Capitol to the Tarpeian Rock. Field Actions Science Reports 19:6–11.
Cheung, W. M., J. T. Leong, and P. Vichare. 2017. Incorporating lean thinking and life cycle assessment to reduce environmental impacts of plastic injection moulded products. J. Clean. Prod. 167:759–775.
Christensen, P. R., A. M. Scheuermann, K. E. Loeffler, and B. A. Helms. 2019. Closed-loop recycling of plastics enabled by dynamic covalent diketoenamine bonds. Nat Chem. 11(5):442–448.
Cohen, C. T., T. Chu, and G. W. Coates. 2005. Cobalt catalysts for the alternating copolymerization of propylene oxide and carbon dioxide: combining high activity and selectivity. J. Am. Chem. Soc. 127(31):10869–10878.
Dunn, E. W. and G. W. Coates. 2010. Carbonylative polymerization of propylene oxide: a multicatalytic approach to the synthesis of poly(3-hydroxybutyrate). J. Am. Chem. Soc. 132(33):11412–11413.
Ellis, W. C., Y. Jung, M. Mulzer, R. Di Girolamo, E. B. Lobkovsky, and G. W. Coates. 2014. Copolymerization of CO2 and meso epoxides using enantioselective β-diiminate catalysts: a route to highly isotactic polycarbonates. Chem. Sci. 10(5):4004–4011.
Fukushima, K., J. M. Lecuyer, D. S. Wei, H. W. Horn, G. O. Jones, H. A. Al-Megren, A. M. Alabdulrahman, F. D. Alsewailem, M. A. McNeil, J. E. Rice, and J. L. Hedrick. 2013. Advanced chemical recycling of poly(ethylene terephthalate) through organocatalytic aminolysis. Polym. Chem. 4:1610–1616.
Fukushima, K., O. Coulembier, J. M. Lecuyer, H. A. Almegren, A. M. Alabdulrahman, F. D. Alsewailem, M. A. Mcneil, P. Dubois, R. M. Waymouth, H. W. Horn, J. E. Rice, and J. L. Hedrick. 2011. Organocatalytic depolymerization of poly(ethylene terephthalate). Journal of Polymer Science Part A: Polymer Chemistry. 49(5):1273-1281.
García, J. M. 2016. Catalyst: Design Challenges for the Future of Plastics Recycling. Chem. 1(6):813-815.
García, J. M., G. O. Jones, K. Virwani, B. D. McCloskey, D. J. Boday, G. M. ter Huurne, H. W. Horn, D. J. Coady, A. M. Bintaleb, A. M. Alabdulrahman, F. Alsewailem, H. A. Almegren, and J. L. Hedrick. 2014. Recyclable, strong thermosets and organogels via paraformaldehyde condensation with diamines. Science 344(6185):732–735.
Geyer, R., J. R. Jambeck, and K. L. Law. 2017. Production, use, and fate of all plastics ever made. Science Advances 3(7):e1700782.
Himmel, M. E., S. Y. Ding, D. K. Johnson, W. S. Adney, M. R. Nimlos, J. W. Brady, and T. D. Foust. 2007. Biomass recalcitrance: engineering plants and enzymes for biofuels production. Science 315(5813):804–807.
Hocking, M. B. 1991. Paper versus polystyrene: a complex choice. Science 251(4993):504–505.
Hong, M. and E. Y. Chen. 2016. Completely recyclable biopolymers with linear and cyclic topologies via ring-opening polymerization of γ-butyrolactone. Nat. Chem. 8(1):42–49.
Hong, M. and E. Y. Chen. 2019. Future Directions for Sustainable Polymers. Trends in Chemistry 1(2):148-151.
Hou, P., Y. Xu, M. Taiebat, C. Lastoskie, S. Miller, and M. Xu. 2018. Life Cycle Assessment of End-of-Life Treatments for Plastic Film Waste. J. Clean. Prod. 201:1052–1060.
Humbert, S., V. Rossi, M. Margni, O. Jolliet, and Y. Loerincik. 2009. Life cycle assessment of two baby food packaging alternatives: glass jars vs. plastic pots. Int. J. Life Cycle Ass. 14(2):95–106.
Krall, E. M., T. W. Klein, R. J. Andersen, A. J. Nett, R. W. Glasgow, D. S. Reader, B. C. Dauphinais, S. P. McIlrath, A. A. Fischer, M. J. Carney, D. J. Hudson, and N. J. Robertson. 2014. Controlled hydrogenative depolymerization of polyesters and polycarbonates catalyzed by ruthenium(II) PNN pincer complexes. Chem. Commun. 50(38):4884–4887.
Krumm, C., J. Pfaendtner, and P. J. Dauenhauer. 2016. Millisecond pulsed films unify the mechanisms of cellulose fragmentation. Chem. Mater. 28(9)3108–3114.
Kruse, T. M., S. E. Levine, H. W. Wong, E. Duoss, A. H. Lebovitz, J. M. Torkelson, and L. J. Broadbelt. 2005. Binary mixture pyrolysis of polypropylene and polystyrene: A modeling and experimental study. J. Anal. Appl. Pyrol. 73(2):342–354.
Levine, S. E. and L. J. Broadbelt. 2009. Detailed mechanistic modeling of high-density polyethylene pyrolysis: Low molecular weight product evolution. Polym. Degrad. Stab. 94(5):810–822.
Li, C., R. J. Sablong, R. A. T. M. van Benthem, and C. E. Koning. 2017. Unique base-initiated depolymerization of limonene-derived polycarbonates. ACS Macro Lett. 6(7):684–688.
Longo, J. M., A. M. DiCiccio, and G. W. Coates. 2014. Poly(propylene succinate): a new polymer stereocomplex. J. Am. Chem. Soc. 136(45):15897–15900.
Morkved, T. L., P. Stepanek, K. Krishnan, F. S. Bates, and T. P. Lodge. 2001. Static and dynamic scattering from ternary polymer blends: Bicontinuous microemulsions, Lifshitz lines, and amphiphilicity. J. Chem. Phys. 114(16):7247–7259.
Paulsen, A. D., B. R. Hough, C. L. Williams, A. R. Teixeira, D. T. Schwartz, J. Pfaendtner, and P. J. Dauenhauer. 2014. Fast pyrolysis of wood for biofuels: spatiotemporally resolved diffuse reflectance in situ spectroscopy of particles. Chem. Sus. Chem. 7(3):765–776.
Qin, Z., C. M. Thomas, S. Lee, and G. W. Coates. 2003. Cobalt-based complexes for the copolymerization of propylene oxide and CO2: active and selective catalysts for polycarbonate synthesis. Agnew. Chem. Int. Ed. 42(44):5484–5487.
Ribitsch, D., E. Herrero Acero, A. Przylucka, S. Zitzenbacher, A. Marold, C. Gamerith, R. Tscheließnig, A. Jungbauer, H. Rennhofer, H. Lichtenegger, H. Amenitsch, K. Bonazza, C. P. Kubicek, I. S. Druzhinina, and G. M. Guebitz. 2015. Enhanced cutinase-catalyzed hydrolysis of polyethylene terephthalate by covalent fusion to hydrophobins. Appl. Environ. Microbiol. 81(11):3586–3592.
Rorrer, N. A., S. Nicholson, A. Carpenter, M. J. Biddy, N. J. Grundl, and G. T. Beckham. 2019. Combining Reclaimed PET with Bio-based Monomers Enables Plastics Upcycling. Joule 3:1006–1027.
Ruegg, M. L., B. J. Reynolds, M. Y. Lin, D. J. Lohse, and N. P. Balsara. 2007. Minimizing the concentration of diblock copolymer needed to organize blends of weakly segregated polymers by tuning attractive and repulsive interactions. Macromolecules 40(4):1207–1217.
Shanks, B. H. and P. L. Keeling. 2017. Bioprivileged molecules: creating value from biomass. Green Chemistry 19(14):3177–3185.
Shirke, A. N., C. White, J. A. Englaender, A. Zwarycz, G. L. Butterfoss, R. J. Linhardt, and R. A. Gross. 2018. Stabilizing Leaf and Branch Compost Cutinase (LCC) with Glycosylation: Mechanism and Effect on PET Hydrolysis. Biochemistry 57(7):1190–1200.
Tabone, M. D., J. J. Cregg, E. J. Beckman, and A. E. Landis. 2010. Sustainability metrics: life cycle assessment and green design in polymers. Environ. Sci. Technol. 44(21):8264–8269.
WEF (World Economic Forum). 2016. The New Plastics Economy: Rethinking the future of plastics and catalysing action. Available: http://www3.weforum.org/docs/WEF_The_New_Plastics_Economy.pdf.
Westhues, S., J. Idel, and J. Klankermayer. 2018. Molecular catalyst systems as key enablers for tailored polyesters and polycarbonate recycling concepts. Sci. Adv. 4(8):eaat9669.
Wierckx, N., M. A. Prieto, P. Pomposiello, V. de Lorenzo, K. O’Connor, and L. M. Blank. 2015. Plastic waste as a novel substrate for industrial biotechnology. Microbial Biotechnology 8(6):900–903.
Williamson, J. B., W. L. Czaplyski, E. J. Alexanian, and F. A. Leibfarth. 2018. Regioselective C-H xanthylation as a platform for polyolefin functionalization. Angew. Chem. Int. Ed. Engl. 57(21):6261–6265.
Wong, S., N. Ngadi, T. A. T. Abdullah, and I. M. Inuwa. 2016. Catalytic cracking of LDPE dissolved in benzene using nickel-impregnated zeolites. Ind. Eng. Chem. Res. 55 (9):2543-2555.
Wright, S. L. and F. J. Kelly. 2017. Plastic and human health: a micro issue? Environ. Sci. Technol. 51(12):6634–6647.
Xu, J., J. M. Eagan, S. S. Kim, S. Pan, B. Lee, K. Klimovica, K. Jin, T. W. Lin, M. J. Howard, C. J. Ellison, A. M. Lapointe, G. W. Coates, and F. S. Bates. 2018. Compatibilization of isotactic polypropylene (iPP) and high-density polyethylene (HDPE) with iPP-PE multiblock copolymers. Macromolecules 51(21):8585–8596.
Yang, Y., J. Yang, W. M. Wu, J. Zhao, Y. Song, L. Gao, R. Yang, and L. Jiang. 2015. Biodegradation and mineralization of polystyrene by plastic-eating mealworms: Part 1. chemical and physical characterization and isotopic tests. Environ. Sci. Technol. 49(20):12080–12086.
Yoshida, S., K. Hiraga, T. Takehana, I. Taniguchi, H. Yamaji, Y. Maeda, K. Toyohara, K. Miyamoto, Y. Kimura, and K. Oda. 2016. A bacterium that degrades and assimilates poly(ethylene terephthalate). Science 351(6278):1196–1199.
Yuan, J., W. Xiong, X. Zhou, Y. Zhang, D. Shi, Z. Li, and H. Lu. 2019. 4-Hydroxyproline-Derived sustainable polythioesters: controlled ring-opening polymerization, complete recyclability, and facile functionalization. J. Am. Chem. Soc. 141(12):4928-4935.
Zhou, X., Z. J. Brentzel, G. A. Kraus, P. L. Keeling, J. A. Dumesic, B. H. Shanks, and L. J. Broadbelt. 2019. Computational framework for the identification of bioprivileged molecules. ACS Sustain. Chem. Eng. 7(2):2414–2428.
Zhu, J. B., E. M. Watson, J. Tang, and E. Y. X. Chen. 2018. A synthetic polymer system with repeatable chemical recyclability. Science 360(6387):398–403.
Zhu, Y., C. Romain, and C. K. Williams. 2016. Sustainable polymers from renewable resources. Nature 540(7633):354–362.
DISCLAIMER: This Proceedings of a Workshop—in Brief was prepared by Anne Frances Johnson and Ellen Mantus as a factual summary of what occurred at the workshop. The statements recorded here are those of the individual workshop participants and do not necessarily represent the views of all participants, the planning committee, the Chemical Sciences Roundtable, or the National Academies. To ensure that this proceedings meets institutional standards for quality and objectivity, it was reviewed in draft form by Carol Bessel, National Science Foundation, and Linda Broadbelt, Northwestern University. The review comments and draft manuscript remain confidential to protect the integrity of the process.
Planning committee members were Gregg T. Beckham, National Renewable Energy Laboratory; Kathryn Beers, National Institute of Standards and Technology; Linda Broadbelt, Northwestern University; Jeannette M. Garcia, IBM Research; Mary Kirchhoff, American Chemical Society; and Timothy Patten, National Science Foundation.
ABOUT THE CHEMICAL SCIENCES ROUNDTABLE
The Chemical Sciences Roundtable provides a neutral forum to advance the understanding of issues in the chemical sciences and technologies that affect government, industry, academic, national laboratory, and nonprofit sectors and the interactions among them and to furnish a vehicle for education, the exchange of information, and the discussion of issues and trends that affect the chemical sciences. The Roundtable accomplishes its objectives by holding annual meetings of its members and by organizing workshops on relevant important topics the published proceedings of which are made broadly available throughout the chemical-sciences community.
Chemical Sciences Roundtable members are Linda Broadbelt, Northwestern University (Co-Chair); Mark E. Jones (Co-Chair), The Dow Chemical Company; Tina Bahadori, US Environmental Protection Agency; Brian Baynes, MODO Global Technologies; Michael R. Berman, Air Force Office of Scientific Research; Carol Bessel, National Science Foundation; Martin Burke, University of Illinois at Urbana-Champaign; Michelle Chang, University of California, Berkeley; Miles Fabian, National Institutes of Health; Michael J. Fuller, Chevron Energy Technology Company; Laura Gagliardi, University of Minnesota; Bruce Garrett, US Department of Energy; Franz Geiger, Northwestern University; Carlos Gonzalez, National Institute of Standards and Technology; Malika Jeffries-El, Boston University; Jack Kaye, NASA; Mary Kirchhoff, American Chemical Society; Robert E. Maleczka, Jr., Michigan State University; David Myers, GCP Applied Technologies; Timothy Patten, National Science Foundation; Nicola Pohl, Indiana University Bloomington; Ashutosh Rao, US Food and Drug Administration; Leah Rubin Shen, Office of Senator Chris Coons; and Jake Yeston, American Association for the Advancement of Science.
This activity was supported by the US Department of Energy under Grant No. DE-FG02-07ER15872, the National Institutes of Health under Contract No. HHSN26300024, and the National Science Foundation under Grant No. CHE-1546732. Any opinions, findings, conclusions, or recommendations expressed in this publication do not necessarily reflect the views of any organization or agency that provided support for the project.
Suggested citation: National Academies of Sciences, Engineering, and Medicine. 2020. Closing the Loop on the Plastics Dilemma: Proceedings of a Workshop—in Brief. Washington, DC: The National Academies Press. doi: http://doi.org/10.17226/25647.
Division on Earth and Life Studies

Copyright 2020 by the National Academy of Sciences. All rights reserved.














