5
Heavy Industry Decarbonization
The fourth session of the workshop focused on the challenges and opportunities of deploying decarbonization technologies within heavy industrial sectors, including the manufacturing of steel, cement, commodity chemicals, and petrochemicals. Heavy industry is often considered a hard-to-decarbonize sector, featuring large amounts of carbon dioxide process emissions that are inherent to the manufacturing process. Decarbonizing these sectors will require a combination of low carbon electricity, increases in efficiency, fuel switching, carbon capture technologies, and likely large-scale, disruptive changes to the manufacturing process, such as electrified steel production.
José Santiesteban (ExxonMobil Engineering, moderator) introduced the three speakers: Tahmid Mizan (ExxonMobil), Adam Rauwerdink (Boston Metal), and Cathy Tway (Johnson Matthey).
EXXONMOBIL PERSPECTIVE
Tahmid Mizan, ExxonMobil
Tahmid Mizan spoke of ExxonMobil’s dual challenge: to provide reliable, affordable energy products while managing environmental impacts, including risks associated with climate change. Mizan noted that quality of life improvements in developing countries require access to affordable energy, and the refining industry is motivated to deliver this product and meet the world’s increasing energy demands. Within global efforts to satisfy rising demand for energy, the refining industry contributes
energy-dense liquid fuels, produced by transforming crude oil into fuel products used mainly for transportation.
Mizan described a typical refinery (illustrated in Figure 5.1) as a set of chemical process units, consisting of separations, purifications, and chemical reactions that require a large energy input. These energy inputs include a combination of heat (either through steam or direct firing) to drive chemical processes, electricity to power mechanical components, and hydrogen gas, each representing a unique challenge for decarbonization. Many common refinery components have large carbon footprints, including the steam methane reforming units that produce hydrogen gas for refining, as well as catalytic cracking units that produce gasoline.
Mizan described a few potential methods for decarbonizing refineries. He suggested that one way refineries could partially decarbonize their thermal requirements is by substituting heat inputs with low carbon electricity-produced heat or nuclear heat. Alternatively, refineries can deploy carbon capture technology on their direct firing furnace units. Mizan went on to describe “blue hydrogen” production (adding CCS to steam methane reforming units) and “green hydrogen” production (using renewable electricity and electrolysis). Another alternative is to focus on process intensification—redesigning refinery processes to be more energy and heat efficient. Mizan noted that refinery equipment typically has frequent turnover cycles, providing opportunities to iteratively implement new technologies or methods.
Mizan noted that historically, energy efficiency has provided more potential for lowering emissions than has decarbonization technologies across all sectors. He suggested that meeting our emissions targets will require both efficiency gains as well as lower carbon energy sources, both of which require innovation and deployment of new technologies. Moreover, Mizan believes that improving energy efficiency represents “low-hanging fruits” with significant future potential. In addition, he listed several decarbonization options available to refineries, including utilizing co-generation (producing power and heat from the same unit), low energy separations, process intensification, lower carbon energy and fuel sources, CCS, low carbon production of hydrogen, and biomass fuels. Mizan believes that CCS will be an important element of all deep decarbonization solutions, including for refineries.
He noted that decarbonization, if pursued, will require supportive policies, such as market-based systems that assign a cost to carbon dioxide emissions, as well as regulations that are technology neutral. Mizan concluded with the idea that all decarbonization solutions need to be cost-effective and meet consumers’ needs, in addition to being protective of the environment.
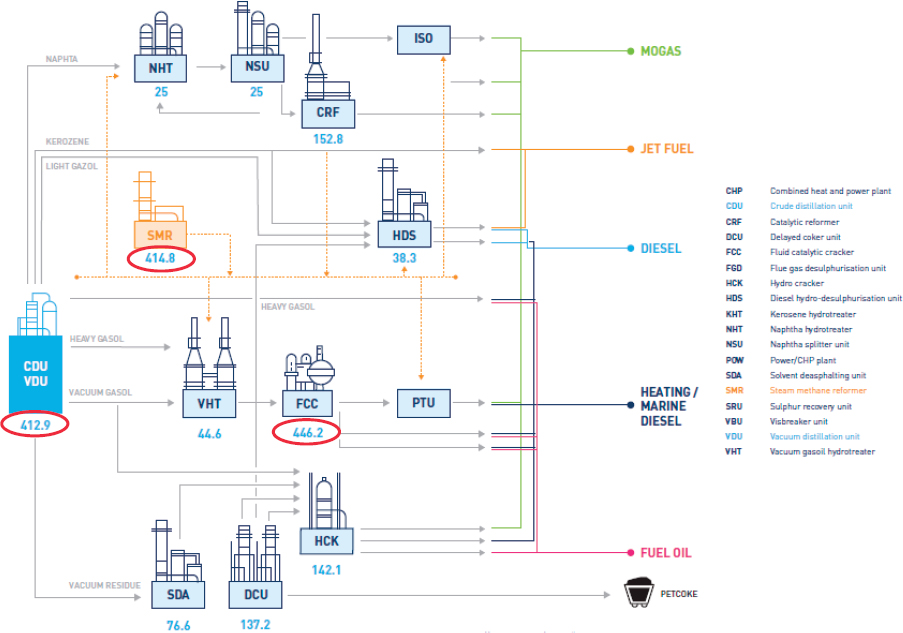
SOURCE: SINTEF Energy Research, “Understanding the cost of retrofitting CO2 capture to an integrated oil refinery,” June 2017, https://www.sintef.no/recap.
METALS MADE BETTER
Adam Rauwerdink, Boston Metal
Adam Rauwerdink described how Boston Metal is striving to decarbonize the production of steel. Rauwerdink showed the chemical reaction used for traditional steel production, in which iron ore and a source of carbon (either coal or coke) are heated in a furnace to produce iron and a large amount of carbon dioxide (greater than 2 gigatons per year globally). Rauwerdink stated that while the electricity sector is beginning to decarbonize and many solutions exist for further efforts, the steel manufacturing sector is predicted to contribute an increasing percentage of global emissions. While emissions per ton of steel produced should continue to decrease as the industry gains further incremental improvements to efficiency, other sectors will likely decarbonize more quickly. Boston Metal seeks to disrupt the steel manufacturing industry by implementing an alternative production method called molten oxide electrolysis (MOE), which requires inputs of iron ore and electricity, producing pure iron and oxygen gas. Rauwerdink noted that the process requires less energy per ton than the incumbent process but that electrifying primary steel production would require tremendous amounts of electricity (it requires a tremendous amount of coal today), and thus the level of achievable decarbonization will depend on access to low carbon electricity.
Rauwerdink suggested that Boston Metal steel can be used in the same products as steel produced by the traditional methods, and that it will be cost competitive even without a carbon tax due to the ability to use lower grade iron ore to produce high quality steel. He noted Boston Metal’s desire to create a more modular process for steelmaking, in which mining companies can operate small MOE units with incremental capacity near their iron ore mines to lower operational costs. The manufacturing processes for traditional steel production and electrified production are compared in Figure 5.2. Rauwerdink stated that steel production currently accounts for over 7 percent of global carbon dioxide emissions, representing a huge opportunity for decarbonization using Boston Metal’s MOE technology.
DECARBONIZATION OF THE CHEMICAL INDUSTRY
Cathy Tway, Johnson Matthey
Cathy Tway began by scoping out the size of the chemical industry, including its contribution to global greenhouse gas emissions. She suggested that the chemicals industry is involved in nearly all goods-producing sectors. With the total gross value of all chemicals listed at

SOURCE: Adam Rauwerdink, Boston Metal.
$5.7 trillion (7 percent of global GDP), the sector supports 120 million workers and invests $51 billion in research and development annually.1 While the chemicals industry is the largest industrial energy consumer, it is largely because the feedstocks, which are the building blocks for their products, are also considered fuels. The energy in those feedstocks stays locked into the products, so the CO2 emissions are lower than would be expected from the energy demand.
Tway noted that the chemicals sector has already optimized their manufacturing processes, with only 15 percent of carbon dioxide emissions currently coming from process losses. This means that there is less room for improving process efficiency in chemicals than in other sectors. As volumes in the chemical industry are expected to grow two to three times by 2050, emissions in the sector will need to decline by 75 percent per unit of production to have a 50 percent chance of limiting global warming to 2°C by 2050,2 said Tway.
With highly optimized manufacturing processes within the sector, Tway suggested that many of the opportunities to lower the carbon footprint lie upstream and downstream on the value chain. Upstream on the
___________________
1 International Council of Chemical Associations, “The Global Chemistry Industry: Catalyzing Growth and Addressing Our World’s Sustainability Challenges,” March 2019.
2 P.G. Levi and J.M Cullen, 2018, Mapping global flows of chemicals: From fossil fuel feedstocks to chemical products, Environmental Science and Technology 52:1725-1734.
value chain, Tway highlighted the difficulty of decarbonizing the chemical inputs to the sector (e.g., olefins, BTX, ammonia, methanol), as they are globally traded commodities with extreme price competition. She suggested a few solutions, including novel processes and energy optimization, alternative feedstocks and fuels (e.g., bioethanol dehydration to produce ethylene, coke oven gas to generate methanol), electrification, and CCS. Tway presented a map of potential chemical pathways that can be used to generate chemical products from alternative feedstocks, shown in Figure 5.3. Tway remarked on the expense of CCS technologies. She said, according to recent research, the cost of implement carbon capture and utilization for use as feedstock in the production of the 20 large volume chemicals identified in the 2013 Technology Roadmap: “Energy and GHG Reductions in the Chemical Industry via Catalytic Processes” assessment,3 which could add up to $1.5 billion/year in additional manufacturing costs depending upon the cost of oil and electricity. These costs could be up to 164 percent more than the 2017 market value for those chemicals.4
Tway suggested that a major obstacle to innovation in the sector is a paid off (or non-fully-depreciated) plant, because the long life of installed capital and its integration make consideration of new technologies more difficult. In addition, the cost of deploying low carbon technologies by retrofitting is high. A recent study5 suggested that building a new plant with electrified ethylene crackers, which produce about 10 percent of the carbon dioxide emissions from the sector, would require a $25/MWh renewable electricity price to be cost competitive with CCS, while retrofitting an existing unit would require a $15/MWh price. Tway stated that while deep decarbonization of the sector is possible, even with current technologies, we need more technological development to improve the economics to support such a transition.
DISCUSSION
A participant wondered how the electricity sector should prepare for the increase in electricity consumption caused by heavy industry electrification. Rauwerdink gave an estimate of the electricity requirements if all primary (non-recycled) steel was produced through MOE: a staggering
___________________
3 International Energy Agency, International Council of Chemical Associations, and DECHEMA, “Technology Roadmap: Energy and GHG Reductions in the Chemical Industry via Catalytic Processes,” 2013.
4 A. Katelhon, R. Meys, S. Deutz, S. Suh, and A. Bardow, 2019, Climate change mitigation potential of carbon capture and utilization in the chemical industry, Proceedings of the National Academy of Sciences U.S.A. 116(23):11187-11194.
5 McKinsey & Company, “Decarbonization of Industrial Sectors: The Next Frontier,” June 2018.
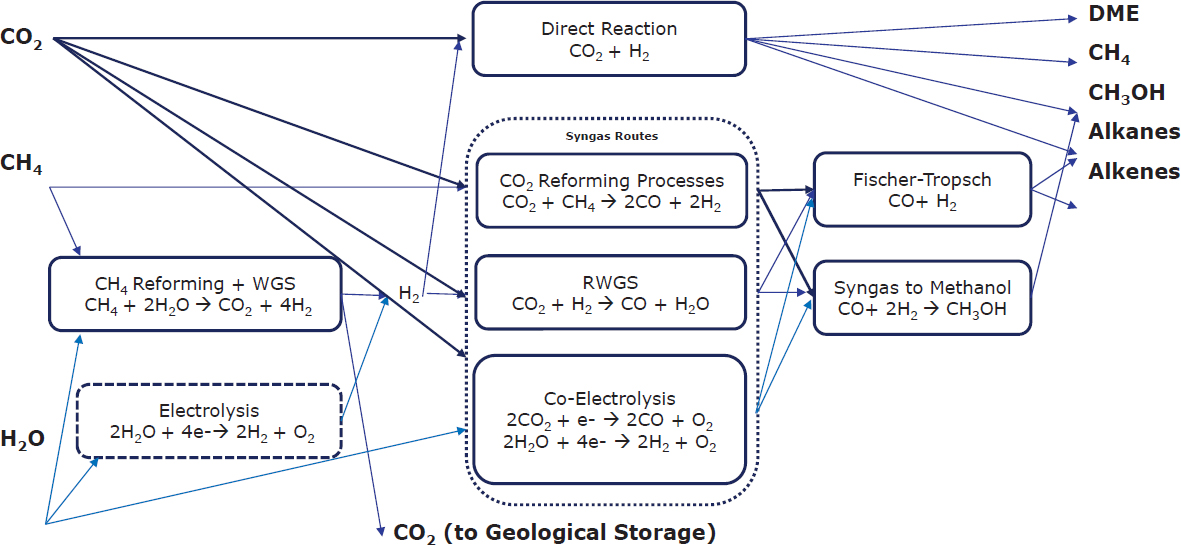
SOURCE: Cathy Tway, Johnson Matthey, presentation to the workshop; adapted from Carbon Dioxide Utilization (CO2U) ICEF Roadmap 2.0, Nov. 2017. Conducted by Lawrence Livermore National Laboratory, document LLNL-TR-739322, reprinted under copyright license CC BY-NC-SA 4.0.
4 billion megawatt hours would be required to produce 1 billion tons of steel (about half of the global annual demand). Mizan suggested that forecasts for energy consumption are highly uncertain, and that the risk of overinvesting or underinvesting in particular decarbonization technologies is high without clear policy and market signals that are unbiased and competitive.
A participant asked what can be done to accelerate energy efficiency improvements—and whether setting corporate goals might be helpful, as energy efficiency was deemed an important pillar in efforts to decarbonize heavy industry. Mizan pointed to the success of the corporate average fuel economy (CAFE) standards to encourage higher fuel economy for vehicles, suggesting that standards and goals can be helpful. He added that public education and labeling of energy efficiency metrics of appliances has been used to incentivize environmentally-conscious consumer purchasing behavior.
A participant asked what percentage of emissions from the refining industry come from end use rather than manufacturing, and if there will still be a need for oil refineries if society is fully decarbonized by mid-century. Another participant wondered if current petroleum products will simply be used for other purposes, rather than primarily for transportation. Mizan stated that about 80 percent of the emissions from refined products occur during end use as tailpipe emission, while less than 10 percent occur during refining; the remainder, he noted, comes from transportation and lifting crude oil from the ground. Tway added that, in the future, refineries will probably primarily produce inputs for the chemical sector. Mizan further noted that refineries are simply chemical process facilities, and that the products generated by those assets are flexible and can evolve with changing societal needs.
The panel was asked for their thoughts on the costs and operational challenges of bringing new decarbonized processes, like green hydrogen production from thermochemical cycles, into large, established plants. Santiesteban said that the top two costs in green hydrogen production are electricity, mostly, and to a lesser extent the electrolyzer itself, and that the challenge of building larger electrolyzers remains. A participant informed the workshop of a Department of Energy program called H2@Scale, which seeks to partner with industry to bring the cost down of deploying hydrogen fuel and process technologies in their operations.
A participant asked Rauwerdink how many megawatt hours per kilogram of steel produced is projected as Boston Metal scales up their electrified steel production. They inquired also about their ramp up time, and their ability to operate with intermittent electricity sources? Rauwerdink stated that the energy requirement for the electrified steel production is around 4 megawatt hours per ton of steel produced, and
that the process relies on reliable and stable electricity to maintain high temperatures. Rauwerdink added that their furnaces are designed to be run continuously for years of operation, like the units used for aluminum production.









