To understand the potential role of biomarkers in disability determination, it is first necessary to define biomarkers and explore their potential use in the diagnosis and prognosis of health and function in general. In the first panel of the workshop, Joseph Menetski, associate vice president of Research Partnerships at the Foundation for the National Institutes of Health (FNIH), discussed the history and definition of biomarkers broadly. Amy Wagner, professor of neuroscience and Endowed Chair for Translational Research at the University of Pittsburgh, followed by presenting how biomarkers relate to health and function. The panel concluded with a discussion, led by Sara Rosenbaum, Harold and Jane Hirsh professor of Health, Law, and Policy at The George Washington University, on challenges in the development and advancement of biomarkers, as well as the importance of the context of use (COU). This chapter reviews the history and current uses of biomarkers, how they relate to health and function, and their future development and challenges in COU.
HISTORY AND DEFINITION OF BIOMARKERS
Menetski started with an overview of FNIH’s work with biomarkers. The goal of FNIH, he stated, is “to attract and share resources and create consensus.” This is done through the various public–private partnerships it facilitates, such as the Biomarkers Consortium. The Biomarkers Consortium has raised more than $95 million over the past 12 years to fund partnerships that involve a wide array of public and private stakeholders such as the Food and Drug Administration (FDA), the National Institutes of Health (NIH), the Centers for Medicare & Medicaid Services, the Pharmaceutical Research and Manufacturers of America, other pharmaceutical and nutritional companies, and not-for-profit organizations (Menetski et al., 2019). The consortium was created because of the novelty of the field of biomarkers and the need for regulation, he explained. FNIH efforts, he said, have created and led cross-sector efforts that validate and qualify biomarkers and other drug development tools to accelerate decision making for the development of new therapeutics and health technologies. So far, the Biomarkers Consortium has contributed to the advancement of 14 therapeutics and generated tools being used in drug development (FNIH, 2020; Menetski et al., 2019); these efforts have greatly improved the field of biomarkers.
Biomarkers Overview
Menetski continued by discussing the definition and classification of biomarkers. He found that the definitions of key terms in biomarker research provided by SSA are similar to the definitions included in the Biomarkers, EndpointS and other Tools (BEST) glossary, which was created by FDA and NIH (see Box 2-1).
Biomarker Classification
Biomarkers can be classified into seven categories (FDA-NIH Biomarker Working Group, 2016), said Menetski. The first three categories—susceptibility and risk biomarkers, diagnostic biomarkers, and prognostic biomarkers—are used to measure disease presence and status. The additional four categories—monitoring biomarkers, predictive biomarkers, pharmacodynamic and response biomarkers, and safety biomarkers—are used to measure aspects of response to treatment. He offered various examples of biomarkers and their classifications (see Table 2-1).
TABLE 2-1 Examples of Biomarkers and Their Key Uses
| Biomarkers | Examples | Key Uses |
|---|---|---|
| Susceptibility and risk biomarkers |
|
|
| Diagnostic biomarkers |
|
|
| Prognostic biomarkers |
|
|
| Monitoring biomarkers |
|
|
| Predictive biomarkers |
|
|
| Pharmacodynamic and response biomarkers |
|
|
| Safety biomarkers |
|
|
NOTE: KIM-1 = kidney injury molecule 1.
SOURCES: Joseph Menetski presentation, July 21, 2020. Adapted from Stein, 2020. Information from FDA-NIH Biomarker Working Group, 2016.
Evidentiary Criteria Framework
Building on the examples, Menetski described the evidentiary criteria framework, which is used to determine biomarker qualification (if enough data are present to use a biomarker) and provide a consistent set of characteristics to describe and define the biomarker program with a regulatory
agency, such as FDA (Leptak et al., 2017). The goal of the framework is to enhance submission quality and predictability of the qualification process, and clarify the type and amount of evidentiary criteria needed. Menetski described the framework as a five-component process:
- Identifying the need statement (i.e., what is the knowledge gap?);
- Defining the COU (i.e., what question is the biomarker addressing, what information will it provide, and/or what is the specific fit-for-purpose use?);
- Determining the benefit (e.g., improved sensitivity);
- Determining the risk (e.g., consequences of false positives or false negatives); and
- Defining the evidentiary criteria, using analytical and clinical validation:
- Analytical validation is used to determine whether the performance characteristics of the biomarker test are acceptable for the proposed COU.
- Clinical validation is used to determine whether the correlation between the biomarker and the outcome of interest are acceptable for the proposed COU (CDER, 2018; Leptak et al., 2017).
Biomarker Development in the Drug Development Process
To conclude, Menetski acknowledged that biomarker development can be as difficult and as resource intensive as drug development. He noted that the correlation of a biomarker to a clinical observation is only as good as the precision that the observation can be quantified, and he emphasized that the type of biomarker being developed will dictate the type of data that are needed to ensure confident decision making. He stated that a biomarker is not the tool used to measure it. For example, “Blood pressure is a biomarker. It does not matter how you measure blood pressure,” Menetski said. “The biomarker is the actual analyte, or the actual measurement. It is not how you make the measurement,” he concluded, so there may be various methods to detect a given biomarker.
RELATING BIOMARKERS TO HEALTH AND FUNCTION
Wagner began by defining health and function as concepts and then discussed the World Health Organization’s (WHO’s) International Classification of Functioning, Disability and Health (ICF) framework to demonstrate how biomarkers can be used as tools in research (see Figure 2-1). She then highlighted ways to move forward in biomarker research and initiatives and programs that use biomarkers for functional assessment.
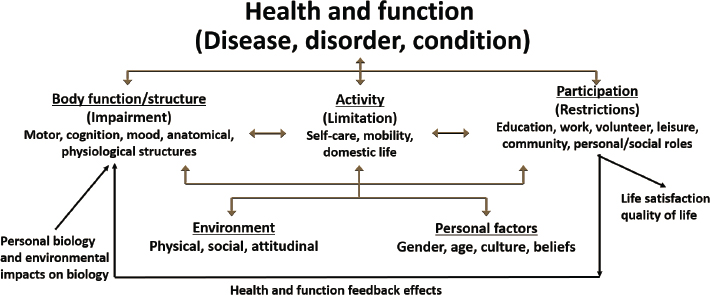
SOURCES: Amy Wagner presentation, July 21, 2020. Adapted from WHO, 2002.
Health and Function Conceptualized
Wagner offered three definitions of health from Sartorius (2006), beginning with the absence of disease, which she stated “is most relevant to the medical model and is based on the state of science, signs, and symptoms.” She acknowledged that this is likely the most widely accepted definition of health in the health care field. The second definition describes health as “a state that allows individuals to adequately cope with all demands of life.” She added, “This definition is important because it begins to make assumptions about health that are based on level of function.”
The third definition of health is “a state of balance or equilibrium between individuals and their social and physical environments.” She emphasized the importance of this third definition because “here, health reflects an internal equilibrium to get the most from life despite disease, and inherent to this definition then is the incorporation of values that individuals have about their cultures and communities.” Based on these definitions, Wagner concluded that health and function are interrelated, and multiple elements should be considered in terms of functioning. Biomarker research occurs within this complex landscape.
The WHO International Classification of Functioning, Disability and Health Framework
Wagner continued by elaborating on WHO’s ICF, which provides a standard language and framework for measuring and categorizing functioning and disability, in the context of health (WHO, 2001). It is focused on impairments, activities, and participation. Impairments are defined
as problems with body functions and structures that are associated with health conditions. Types of impairments include emotional, motor, cognitive, or other sensory functions. Wagner pointed out that these impairments can “lead to symptoms such as cognitive dysfunction, balance deficits, paralysis, and changes in mood that can be related to common diseases associated with disability assessments, such as dementia, post-traumatic stress disorder, stroke, schizophrenia, spinal cord injury, traumatic brain injury, and degenerative joint disease.” From this first element of the WHO ICF, Wagner said, we can “link symptoms to disease.”
The second element of the WHO ICF is characterized by activities, which are “things individuals do to be independent members of the community,” she said. She said that activities are
focused on execution of a daily task or action and are things we need to do to care for ourselves and carry out our goals in life. This includes various aspects such as functional mobility, household management, recreation, work, personal care, and learning new skills. These activities help us fulfill our wants and needs.
The third defining element of the WHO ICF is participation. This element “is distinguished from activity through the ideas of intent and impact,” said Wagner, adding that “participation focuses more on involvement in a life situation and is related to the various roles we assume in our daily lives.” Participation encompasses meaningful engagement, personal and societal responsibility, making an impact on others, taking advantage of access and opportunities, and social connectedness and inclusion. These factors highlight “the idea of respect and dignity resulting from participating fully in all areas of our lives,” she said. She offered the everyday scenario of getting dressed as an example to differentiate activities and participation. One might define “getting dressed as an activity, but getting dressed to go to the prom or a job interview is participation because you are engaging with others in social connectedness and social roles,” she explained.
Wagner said the ICF framework also incorporates the idea that both personal factors and environmental factors can moderate daily functions across multiple dimensions. She further emphasized that functioning at the fullest level affects life satisfaction and quality of life.
Tools for Measuring Health and Function
Wagner explained how the WHO ICF framework can be used to link biomarkers to function by using it to help understand how biomarkers relate to rehabilitation processes. She explained further:
The framework has allowed novel opportunities to work toward precision care for populations with disabilities in terms of diagnostic and prognostic markers, biomarker-guided clinical decision algorithms, and biosusceptibility tools for screening prevention, as well as biological stratification for clinical trials.
Milleville et al. (2019) found that inflammation in a population with traumatic brain injury contributed substantially to cognitive dysfunction. Downstream effects from the inflammatory burden were also noted and helped researchers understand more about its impact on daily functions that require cognitive capacity, as well as its indirect impact on quality of life. This work was provided as an example of the role biomarkers can play in “bridging the gap between health and function,” said Wagner.
Envisioning a Future Path and Leveraging Existing Resources
Wagner discussed the long-term goal for biomarker use of helping people monitor and manage their health and function. One way this may happen is through the use of point-of-care technologies and telehealth tools where information can be entered into a large data infrastructure that would provide personalized readouts for the individuals needing care. She acknowledged the immense progress needed to reach this goal, but she also highlighted existing resources that will help bridge the gap, such as the Precision Medicine Initiative, All of Us Research Program, and the Million Veteran Program (MVP).
Wagner reviewed the NIH Precision Medicine Initiative that was launched in 2015 by President Obama;1 she explained that it is a long-time research endeavor whose goal is to improve understanding of individualized approaches to disease and treatment. The initiative’s short-term focus is cancer, and its long-term focus is intended to encompass all areas of health and disease (Collins and Varmus, 2015). Wagner believes potential opportunities exist to “use this type of resource to learn more about how disability and functioning can be prioritized as a focus of biomarkers research.”
Wagner elaborated on the All of Us Research Program,2 a program within the Precision Medicine Initiative. The goal of this program is to improve and personalize the ways health conditions are prevented, diagnosed, and treated (All of Us Research Program Investigators, 2019). The program intends to build a database of 1 million individuals with
___________________
1 See https://ghr.nlm.nih.gov/primer/precisionmedicine/initiative (accessed September 8, 2020).
2 See https://allofus.nih.gov (accessed September 8, 2020).
genomic biomarker and health care information to study various areas where personal biology can inform clinical care. “The program focuses on genomic heterogeneity and personal biology to better understand disease mechanisms,” said Wagner. The National Center for Medical Rehabilitation Research has also been involved with the All of Us Research Program “by incorporating principles of health and function within enrollment processes and research efforts,” she said. Further opportunities exist for “large-scale studies to address biomarkers as functional assessment tools,” but she pointed out that it will require ongoing engagement with disability researchers and consumers on barriers to participation, as well as continued engagement with disability advocates and populations to prioritize focus within the research.
The final resource Wagner highlighted is the Department of Veterans Affairs’ (VA’s) MVP,3 which launched in 2011. The goal of the MVP is to “learn more about how genes, lifestyle, and military exposures particularly affect health and function” (VA, 2020). The program has enrolled 825,000 individuals (VA, 2020) and partnered with other entities such as the Department of Energy for its Big Data Science Initiative. The MVP has “created a large biobank for future use in various areas of research [available to VA researchers],” said Wagner, and it has “a variety of ongoing research programs that center on the idea of individualized approaches to disease diagnosis and management.” She noted that significant opportunities exist for consumer engagement in defining and assessing the genomics that might be affecting function.
DISCUSSION
Rosenbaum moderated a discussion on challenges in the development of biomarkers and important future considerations. Workshop participants and planning committee members posed questions related to progress in biomarkers research and discussed various factors that should be considered when implementing biomarkers in practice.
Challenges in Development and Advancement of Biomarkers
Rosenbaum asked the panelists about the timeline of implementing biomarker research into standard practice. “There is still a lot to be done in terms of characterizing biomarker relationships to disease and drug development,” Wagner replied. She emphasized the need to strengthen these characterizations to be able to map them onto disease-specific func-
___________________
3 See https://www.mvp.va.gov/webapp/mvp-web-participant/#/public (accessed September 8, 2020).
tions. Menetski supported Wagner and said, “Many existing definitions of disease are not precise enough to address,” and there is a “need to look at the process of redefining diseases so a strong comparative baseline exists from which to make quantitative measures.”
Srinivasa Murthy, a retired psychiatry professor, posed a question on the development of pain as a biomarker. Wagner explained that the challenge of using pain as a biomarker, versus factors such as oxygen or cholesterol, is that pain is a self-reported, subjective phenomenon. She noted
There is still much to be learned about the substrates of pain and the complexities that go into translating a biomarker to a self-reported subjective measure, but areas such as neuroimaging have been useful in bridging the gap.
Menetski added that “pain as a biomarker relates to precision in measuring.” For example, he explained, people with arthritis often categorize various pains (e.g., back pain, knee pain) in the same way, even though pain can vary.
Robert Wallace, emeritus professor of epidemiology and internal medicine at the University of Iowa, enquired about the regulatory pathway to deep learning-based biomarker discovery. In response, Menetski acknowledged the investments that have been made in artificial intelligence by looking at multianalytes, such as omic technology. FDA requires a more comprehensive understanding of the process of using omic technology to produce a reliable biomarker score. Multiple variables are involved in calculating each biomarker score, which can complicate regulation, Menetski said.
Importance of Context of Use
Questions were posed by workshop participants regarding parameters that exist between people who exhibit biomarkers and those who do not, as well as biomarkers that are not readily tied to pathophysiology but have a strong association with a clinical outcome measure. Menetski responded by emphasizing the importance of COU in both scenarios. He explained that biomarkers can require varying levels of precision and that accuracy also factors into each scenario, as explained in the evidentiary criteria framework. Using cholesterol as an example, he explained that a biomarker could have long-term effects and require less precision in measuring versus oxygen levels that could have more immediate effects.
Continuing the topic of COU, workshop participants asked about the role of demographic factors such as age, environment, race, and ethnicity in biomarker use and validation. Wagner noted that the interplay
between age and environment is complex because “as our bodies age, we have more environmental exposures.” Because of this, “there is not going to be a one-size-fits-all approach in how biomarkers are applied based on age, gender, or similar factors,” and that these factors are taken into consideration in biomarker development, she said. Wagner and Menetski both agreed that race and ethnicity play a role as well. Wagner said, “There are some biological differences that come in part as a result of our personal biology that we inherit over the generations, and race and ethnic background can factor into that.” Other factors, such as access to health care and health care maintenance and prevention, also play a role in understanding and applying biomarkers, said Wagner. Together, these all create a complex environment that we will need to navigate when using these biomarkers to try and understand disease and function more clearly, she added.
This page intentionally left blank.












