2
Test Protocol
UNDERLYING CONCEPTS
Introduction
Protecting an individual against CB agents requires a suit that not only protects against the external threat of CB agents in both liquid and vapor form, but also allows body moisture and heat transfer to the external environment from the microclimate between the body and the clothing. Thus, the materials in an effective suit must meet stringent performance demands and will be necessarily complex. The currently accepted CB protective suit, referred to as the battle dress overgarment, is constructed from a permeable, multilayered material that incorporates activated charcoal to absorb chemical agents. A permeable outer layer of fabric (a tri-blend of 58 percent cotton, 27 percent Kevlar aramid, and 15 percent nylon twill weave) is backed by a charcoal-loaded foam or nonwoven layer. The surface of the outer layer is coated with formulations containing fluoropolymers to minimize surface energy, thereby preventing wetting and wicking of aqueous and organic liquid chemical agents. Protection against vapor chemical agents is provided by the absorbing charcoal. The multilayered material is permeable to water vapor so that some body moisture (perspiration) can escape from the body microclimate; however, thermal stress to the individual is a significant limitation of this suit. To prevent liquid perspiration, which can poison the activated charcoal, from coming into contact with the charcoal-containing foam, a permeable, nonwettable inner fabric is inserted.
The committee recognizes that protection against chemicals is just one of several factors involved in the overall evaluation of a chemical protective ensemble; heat stress is another. Because the committee
was charged with reviewing the MIST program only for how well the program evaluates the chemical protection of an ensemble, other factors, such as heat stress, are not addressed directly.
Materials Evaluation
Reliable testing and evaluation procedures are essential in the constant search for better protective suits. A battery of laboratory tests have been developed and are available to evaluate the performance of candidate materials. These tests include liquid and vapor chemical agent permeation tests through fabric samples mounted on diffusion cells. These so-called ''cup tests'' are based on measurements made under controlled laboratory conditions and are valid only for homogeneous, continuous materials. They are designed to make precise, accurate evaluations of both the protective aspects and the heat and moisture transfer characteristics of materials. These tests utilize swatches of material and are designed only for continuous, uninterrupted materials that are globally homogeneous and uniform. Other tests are used to evaluate the mechanical properties of materials, which must meet standard requirements for strength, tear resistance, abrasion resistance, bending flexibility, and similar characteristics.
Suit Technology
When a protective suit is constructed from a suitable material, the final product can no longer be considered homogeneous and continuous. In other words, its performance may no longer meet the required component performance criteria despite the fact that the material from which it was constructed meets all of the established performance standards. In contrast to a homogeneous and continuous material, which is planar or two dimensional, the final product is three dimensional, discontinuous, and otherwise structurally complex. The suit is fabricated from many panels that are stitched, bonded, or otherwise held together, which creates discontinuities. In addition, the suit must be integrated with other protective gear, such as a hood, a mask, gloves, and boots, which create additional discontinuities in the overall ensemble.
Furthermore, a protective system is required to function under dynamic conditions. The individual must be able to move and perform
a variety of tasks. Physical motion creates local deformations of the suit material that can, under certain conditions, reach relatively high strains. These deformations alter the pore structure (shape and dimensions) of the material and modify both the barrier and the permeation properties. The physical motion may especially affect the discontinuities in the suit created by the stitching and bonding of component panels and by interfaces with other protective gear. In addition, CB protective suits may also be subject to wear and damage during use, potentially under extreme battlefield conditions. Perforations, punctures, and tears in the suit material will create further discontinuities.
Evaluation of Suit Systems
For the reasons outlined above, evaluating the performance of a CB protective ensemble under realistic, dynamic conditions is far more complex than evaluating component materials and does not lend itself readily to standard, controlled laboratory measurements. In recognition of this fact, the MIST program has been developed to evaluate individual CB protective ensembles under realistic, dynamic conditions. The essential elements of the MIST involve placing individuals wearing candidate suits in an enclosed environment (referred to as a defensive test chamber) containing a nonhazardous chemical compound (methyl salicylate vapor) that is intended to simulate a chemical agent. Dynamic conditions are created by individuals performing various physical tasks according to a specified time schedule. The test individuals are outfitted with sensing patches on their skin, referred to as passive sampling detectors, that absorb the chemical compound when and if it penetrates the protective suit system. The sensing patches are positioned at various places on the body and are analyzed at the conclusion of the test procedure. The raw data from the patch analyses are interpreted in terms of a model referred to as body region hazard analysis (BRHA). This model is based on the varying absorptive capacities of the skin in different regions of the body and incorporates significant assumptions to determine the overall protection factor.
Together, BRHA and the MIST attempt to provide a quantitative measure of the relative effectiveness of a protective suit system under realistic dynamic conditions. The MIST/BRHA must be viewed as a procedure for evaluating the performance of a complex system,
intended not only to characterize overall performance, but also to identify the weakest elements in the system (probably the discontinuities discussed above).
METHODOLOGY
Methods for Evaluating Protective Clothing
Protective materials traditionally have been evaluated using physical approaches in which the amount of chemical that passes through the material is measured directly. The internally absorbed dose is calculated based on estimates of skin deposition and percutaneous transport. A relatively new approach to estimating the ability of clothing to protect humans from hazardous chemicals is biological monitoring. Biological monitoring usually involves a urinalysis to determine the amount of chemical or metabolite in the urine following an exposure. If the proportion of the absorbed dose excreted in urine is known through control studies, the urine level can be used to estimate the total absorbed dose. Biological monitoring is now being widely used to assess worker exposure to pesticides and other hazardous chemicals found in the workplace and has become a benchmark for standardizing other procedures for assessing worker exposure (Wang et al., 1989).
For obvious reasons, biological monitoring cannot be used directly in human studies to evaluate protective materials against CB agents. It can be used, however, with nontoxic agent simulants, as long as the comparative penetration rates (ensemble material and discontinuities, as well as skin) and other biokinetic factors of CB agents and simulants are known.
In addition to biological monitoring, several other techniques are available to evaluate the effectiveness of protective suits in the MIST scenario. These include air sampling, passive dosimetry, and fluorescence imaging. Briefly, air monitoring involves sampling the air between the skin and the protective clothing. Passive dosimetry involves placing patches between the skin and the protective clothing to estimate dermal exposure; skin deposition is then extrapolated from patch deposition. Fluorescence imaging involves a simulant that incorporates a fluorescent dye; video imaging of the test individuals provide estimates of dermal deposition. Table 2-1 summarizes the essential features of each technique.
TABLE 2-1 Techniques for Evaluating Protective Clothing
|
Technique |
Measurement |
Advantages |
Disadvantages |
|
Air sampling |
Withdrawal and analysis of air between skin and clothing |
Simple analysis |
Restricts movement Artificial airflow Provides no skin deposition or absorption data |
|
Passive dosimetry |
Analysis of deposition on patches placed on skin or clothing |
Ease of sampling Simple analysis |
Extrapolation error Provides no skin absorption data |
|
Fluorescence imaging |
Measurement of fluorescent tracer deposited on skin |
Quantitative assessment of skin deposition |
Provides no skin absorption data |
|
Biological monitoring |
Measurement of chemical or metabolite in urine, blood, or expired air |
Integrates body exposure, skin deposition absorption, and possibly inhalation; Closest estimate of total body dose |
Requires information on distribution or metabolism |
The biggest advantage of the air sampling technique is the simplicity of direct chemical analysis. However, the removal of air from the space between skin and clothing for sampling purposes can cause local turbulence and create artificial airflow through the fabric. Air lines and connections also restrict the movement of the test individuals. Furthermore, air sampling provides no information on skin deposition and absorption, and biological effects must be extrapolated directly from air concentrations.
The biggest advantage of passive dosimetry is the ease with which samples can be obtained and analyzed. The simulant is usually analyzed, and no metabolism data are necessary. A disadvantage is that deposition on the skin may differ from deposition on the patch; thus no information on real skin deposition is obtained, and skin absorption cannot be calculated. A potential source of error in passive dosimetry is inherent in the extrapolation of deposition from relatively small patches to the entire body surface.
Video imaging of fluorescent tracers is a relatively new technique, which has the advantage of providing a visual image of affected body
areas. The technique provides a measure of dermal deposition but assumes that the transport and adsorption of the tracer and chemical simulant are similar. Again, no information on skin absorption is obtained.
The primary advantage of biological monitoring is that the actual internally absorbed dose can be determined provided that information regarding the distribution or metabolism of the compound is available. The technique integrates absorption from all body sites, eliminating the concern over positioning of patches. The integration makes it impossible to pinpoint the location of suit failures; information regarding skin deposition and absorption is inherent in the measurement. Biological monitoring can be done with compounds or their metabolites that are excreted in a variety of biological materials, including blood, urine, feces, expired air, and saliva. The technique has been used successfully by a number of investigators in human studies to determine internally absorbed doses following exposures (Franklin et al., 1981).
Combining two or more testing techniques (e.g., biological monitoring and passive dosimitry) in skin exposure studies has been useful. The first three techniques (air sampling, passive dosimetry, fluorescence imaging) are best suited to determining exposure (i.e., the amount of chemical that reaches the skin). These techniques would be most helpful for designing effective gear. In contrast, biological monitoring determines an absorbed dose and would be most useful for assessing a soldier's effectiveness.
MIST Test Procedure
The MIST and BRHA were developed to provide a system-level evaluation of CB protective suits (Fedele and Nelson, 1996). Briefly, each subject in a group of up to eight volunteers is fitted with approximately 20 passive detector patches for methyl salicylate vapor, which simulates the vapors of VX (an organophosphorus cholinesterase inhibitor) or HD (the vesicant bis(2-chloroethyl) sulfide). The detectors, which contain an absorbent powder (Tenax TA), are placed at various anatomical locations (Figure 2-1). The subjects don test suits, gas masks, and hoods and enter a test chamber containing methyl salicylate at a concentration of 100 mg/m3 at an air temperature of 21°C to 32°C, 50 to 80 percent relative humidity, and an airflow rate of 3 to 16 kph (2 to 10 mph).
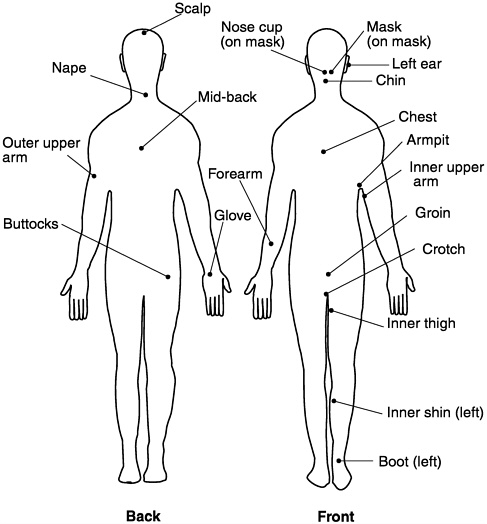
Figure 2-1 Placement of passive sampling devices for the MIST.
The test chamber is a stainless steel, environmentally-controlled unit that can accommodate eight test subjects. The chamber is maintained at a negative pressure by an air filtration system and a controlled air intake system. The chamber was designed for agent testing of large systems and has a design specification to provide controlled temperature (-32°C to 38°C) and a nominal wind speed of 2 to 10 mph, provided by two large propellers to ensure a unidirectional airflow, which is recirculated through the chamber through a false ceiling and a series of vents (see Figure 2-2).
The test chamber is supported by several modules: a control room on the west side of the chamber, which is the central location for
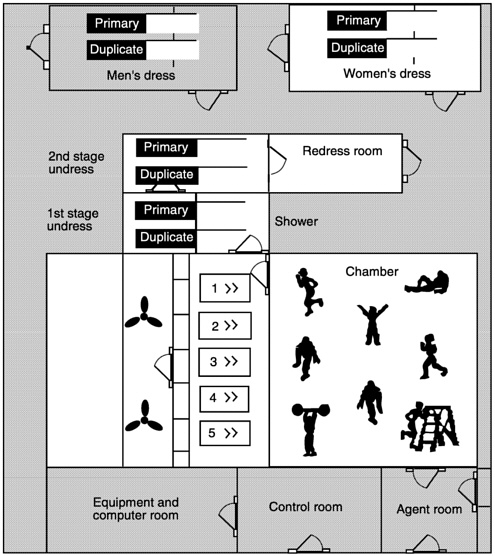
Figure 2-2 MIST system test chamber.
monitoring and controlling all physical parameters; two separate instrumentation rooms; and an airlock chamber entry/exit room, which allows test participants to enter and exit the chamber without releasing large amounts of simulant to the atmosphere. The egress area is also used as a clean undress location (Hanzalka et al., 1996).
Additional subjects wear suits virtually impermeable to vapor (i.e., control suits) with corresponding detectors on the outer surface of the suit. All subjects follow a prescribed exercise or movement routine for the duration of the exposure (120 min). The exercise protocol consists
of eight "stations." Station #1 consists of jumping jacks, Station #2 seated rest, Station #3 walking simulation (treadmill), Station #4 seated rest, Station #5 moving weights, Station #6 a "take cover" maneuver, Station #7 walking simulation (treadmill), and Station #8 climbing and reaching (Hanzelka et al., 1996). The threat or challenge is defined in terms of a concentration x time factor (Ct) of 12,000 mg/m3-min (100 mg/m3x 120 min). After the subjects exit the test chamber, the detectors are removed for gas chromatographic analysis. Because the background level of methyl salicylate in control detectors ranges from 50 to 100 nanogram (ng), detectors with values of less than 100 ng are assigned a value of 100 ng.
Body Region Hazard Analysis
When the methyl salicylate is intended to simulate mustard vapor (HD), the BRHA is "local" because HD exerts its toxicity primarily on localized regions of the skin. When exposure to VX vapor is being simulated, the BRHA is "systemic" because VX toxicity results from cumulative absorption through exposed skin.
The mass of VX required to produce systemic toxicity after exposure to various regions of the skin was derived from the work of Sim (1962) who estimated the dose of liquid VX required to cause a 70 percent depression in cholinesterase from studies in which droplets of VX were applied to various skin sites on volunteer subjects (see Table 2-2 and the Acetylcholinesterase Inhibition as a Biological Marker in Chapter 5). The symptoms exhibited by the subjects included local sweating, erythema, weakness, muscular fasciculation, dizziness, headache, abdominal cramps, repeated vomiting, and diarrhea. The wide range of the data in Table 2-2 reflects the variation of skin permeability at different anatomic sites. Factors that affect the regional permeability of the skin have not been precisely defined. However, the thickness of the stratum corneum and the density of adnexal structures (e.g., hair follicles) may be contributors. VX does not appear to undergo appreciable metabolism by the skin, in contrast to soman, which undergoes hydrolysis (van Hooidonk et al., 1983). At a given anatomic site, the absorption rate generally increases as the dose increases, whereas the efficacy of absorption (the percentage of agent absorbed) generally decreases as the dose increases. The effects of dosage on regional variations in skin permeability have not been adequately studied.
TABLE 2-2 Estimated Topical Dose of VX That Would Cause a 70 Percent Depression in Red Blood Cell Cholinesterase in a 70-kg Human
|
Area of Application |
Single-Drop Dose of VX (mg) |
|
Cheek |
0.36 |
|
Ear |
0.46 |
|
Top of head |
0.76 |
|
Forehead |
0.78 |
|
Groin |
1.22 |
|
Back of neck |
1.72 |
|
Axilla |
2.07 |
|
Popliteal space |
2.09 |
|
Abdomen |
2.23 |
|
Elbow |
2.25 |
|
Back |
2.65 |
|
Forearm (volar) |
2.80 |
|
Hand (dorsum) |
2.91 |
|
Buttocks |
4.26 |
|
Forearm (dorsum) |
6.57 |
|
Foot (dorsum) |
6.60 |
|
Foot (plantar) |
7.14 |
|
Knee |
7.14 |
|
Hand (palmar) |
9.24 |
The BRHA assumes that (1) the effective vapor exposure (Ct factor) for each region of the body would produce toxicity (e.g., nausea and vomiting); in other words, when vapor is presented to that body region only, toxicity will result; and (2) the relative differences in exposures to VX vapor required to produce an equivalent toxicity from the various skin sites are the same as the corresponding differences for the exposures to VX liquid.
For exposure to HD vapor, the Ct factor necessary to produce equivalent toxicity at various anatomic locations is based on a vapor exposure of 1,000 mg/m3-min to a human forearm causing severe burns in hot (above 27°C), humid conditions. The BRHA calculates the exposure necessary to produce equivalent toxicity at other anatomic locations based on the regional toxicity data for VX. For example, the amount of VX required to cause equivalent toxicity after
application in the chin and neck area (5.1 µg/kg, Table 2-3) is approximately one-eighth of the corresponding amount for the forearm (40 µg/kg). The associated HD vapor exposure for chin and neck is then calculated as approximately one-eighth of the forearm vapor. As indicated in Table 2-3 and by other data, the scrotum and areas of the head and neck are particularly vulnerable because of higher skin permeability. These areas are also near discontinuities in the typical chemical protective ensemble (CPE), which increases their vulnerability. These areas should receive special attention in the design of the new CPE.
Evaluation of Suits against Exposure to HD (Local Analysis)
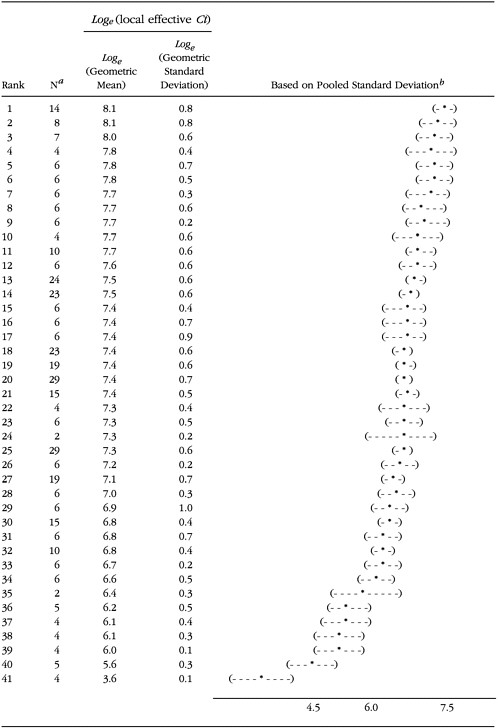
To calculate the "protection value" of test suits against a simulated exposure to HD vapor (local analysis), the "protection factor" for each anatomical site is calculated by dividing the amount of methyl salicylate found in the detector on the outside of the control suit by the amount found in the corresponding detector on the inside of the test suit. Each site protection factor is multiplied by the exposure to HD necessary to produce severe bums (column 4 in Table 2-3). The lowest value of the resulting site protection factors is assigned as the test suit's protection value, expressed as a Ct value in mg/m3-min. The results for tests of 41 different candidate ensembles are given in Table 2-4, which gives the number of test replicates (n) and the geometric mean and standard deviation of the local effect of concentration vs. time (Ct) for each protective ensemble. Ensembles are ranked from most protective (1) to least protective (41). The battle dress uniform (41) is a standard military uniform not specifically designed for chemical protection. Table 2-5 contains the loge transform of the geometric mean and standard deviation contained in Table 2-4, as well as the 95 percent confidence intervals (95 percent CIs) for the loge (local effective Ct).
The geometric: mean1 has been used to average ratios (here protection factors) when each ratio is to be given equal weight. A log transform of local or systemic Ct has the effect of further reducing the
TABLE 2-3 Parameters for Local Body Region Hazard Analysisa
variability of the data. An alternate statistical analysis of the data should be done to investigate the influence of these mathematical treatments on the ranking of CPEs. The data should also be analyzed for the influence of test subject characteristics, such as body size and weight.
TABLE 2-4 Ranking of Protective Ensembles by Local Effective Cta
|
Rank |
Ensembleb |
Nc |
Geometric Meand |
Geometric Standard Deviatione |
|
1 |
Duty uniform, non fire resistant, model 1 |
8 |
8,730 |
2.44 |
|
2 |
Duty uniform, fire resistant, model 1 (w) |
2 |
7,614 |
1.39 |
|
3 |
Overgarment, none-fire resistant, model 3 (w) |
6 |
6,933 |
1.67 |
|
4 |
Saratoga chemical protective suit (worn over personal undergarments only or over duty uniform (w) |
6 |
5,800 |
1.78 |
|
5 |
Duty uniform, none-fire resistant, model 3 |
7 |
5,689 |
2.86 |
|
6 |
Duty uniform, non-fire resistant, model 2 |
4 |
5,317 |
1.89 |
|
7 |
Overgarment, non-fire resistant, model 2 9 (r) |
6 |
5,079 |
2.31 |
|
8 |
Duty uniform, fire resistant, model 2 |
10 |
4,331 |
2.04 |
|
9 |
Overgarment, non-fire resistant, model 2 (w) |
6 |
5,079 |
3.33 |
|
10 |
Overgarment, fire resistant, model 2 (w) |
6 |
4,096 |
3.86 |
|
11 |
Duty uniform, fire resistant, model 1 |
6 |
4,052 |
2.38 |
|
12 |
Overgarment, fire resistant, model 1 (w) |
6 |
3,283 |
3.36 |
|
13 |
Overgarment, non-fire resistant, model 2 |
23 |
3,089 |
2.88 |
|
14 |
Duty uniform, non-fire resistant, model 3 (w) |
6 |
2,835 |
1.99 |
|
15 |
Overgarment, non-fire resistant, model 1 (r) |
6 |
2,788 |
1.88 |
|
16 |
Overgarment, fire resistant, model 2 |
23 |
2,700 |
2.95 |
|
17 |
Vapor protective, fire resistant, protective undergarment |
14 |
2,492 |
3.84 |
|
18 |
Overgarment, non-fire resistant, model 1 |
19 |
2,441 |
3.52 |
|
19 |
Overgarment, fire resistant, model 1 |
29 |
2,308 |
3.01 |
|
20 |
Overgarment, non-fire resistant, model 2 (r) |
6 |
2,129 |
2.12 |
|
21 |
Vapor protective, fire resistant, protective undergarment (w) |
4 |
2,101 |
2.08 |
|
22 |
Duty uniform, fire resistant, model 2 (w) |
6 |
2,002 |
2.33 |
|
23 |
Standard U.S Army chemical protective undergarment |
24 |
2,001 |
2.25 |
|
24 |
Duty uniform, non-fire resistant, model 2 (w) |
6 |
1,930 |
2.08 |
|
25 |
Overgarment, non-fire resistant, model 3 |
19 |
1,899 |
3.67 |
|
26 |
Duty uniform, non-fire resistant, model 1 (w) |
6 |
1,823 |
3.82 |
|
27 |
Saratoga chemical protective suit (worn over personal undergarments only or over duty uniform |
15 |
1,804 |
2.25 |
|
28 |
Standard Army chemical protective undergarment (w) |
4 |
1,305 |
1.97 |
|
29 |
Battle dress overgarment |
29 |
1,295 |
2.35 |
|
30 |
Overgarment, non-fire resistant, model 1 (w) |
6 |
1,221 |
1.82 |
|
31 |
Overgarment, non-fire resistant, model 2 (nr) |
4 |
1,124 |
1.72 |
|
32 |
Air crew uniforms. model 2 |
15 |
1,045 |
2.42 |
|
33 |
Air crew uniforms, model 1 |
10 |
930 |
2.07 |
|
34 |
Air crew uniforms, model 1 (w) |
6 |
846 |
1.98 |
|
35 |
Overgarment, non-fire resistant, model 1 (nr) |
4 |
680 |
2.24 |
|
36 |
Air crew uniforms, model 2 (w) |
6 |
594 |
1.28 |
|
37 |
Overgarment, non-fire resistant, model 3 (nr) |
4 |
527 |
1.84 |
|
38 |
Saratoga chemical protective suit (worn over personnal undergarments only or over duty uniform (r) |
5 |
515 |
1.57 |
|
39 |
Standard U.S. Army chemical protective undergarment |
2 |
447 |
1.14 |
|
40 |
Saratoga chemical protective suit (worn personal undergarments only or over duty uniform) (nr) |
5 |
423 |
2.33 |
|
41 |
Battle dress uniform standard military uniform) |
4 |
53 |
1.19 |
|
a Ct is agent concentration (mg/m3) x times of exposure (in minutes) b Various protective ensembles tested (w = worn, r = repaired , nr = not repaired) c Number of test replicates for each ensembled d The geometric mean, the nth root of the product of the n data or e The geometric standard deviation, Σ; (standard deviation of logarithms of values |
||||
Evaluation of Suits against Exposure to VX (Systemic Analysis)
To calculate the protection value of test suits against a simulated exposure to VX vapor (systemic analysis), the protection factor for each anatomical site is calculated in the same manner as for the local analysis. The site protection factors (PFsite) along with the site size (skin areasite) and the VX dose site (VX dosesite) that would cause a 70 percent depression of red blood cell cholinesterase (Table 2-2) are used to calculate a ''whole body effective exposure'' (WBEE) according to the following formula:2
where the total body surface area (excluding the face) is 18,950 cm2. This calculation is essentially the sum of the VX dose sites that have been weighted for the area and protection factor of each site. The WBEE for an unprotected person weighing 70-kg was calculated by setting all the site protection factors equal to unity to yield a value of 2.45 mg for the data in Table 2-3.
The protection factor for a test suit is calculated by dividing the WBEE (mg) by 2.45 mg. The protection factor for a test suit is multiplied by the Ct factor for VX (25 mg/m3-min), which is assumed to be the vapor equivalent of the WBEE for the unprotected person (Reutter and Wade, 1994). This figure is called the "systemic effective Ct, " and the geometric mean of replicate evaluations is used to rank the test suits relative to the battle dress overgarment. The results from tests involving 41 protective suits are given in Table 2-6. Geometric mean, geometric standard deviation, and CIs for these data are given in Table 2-7.
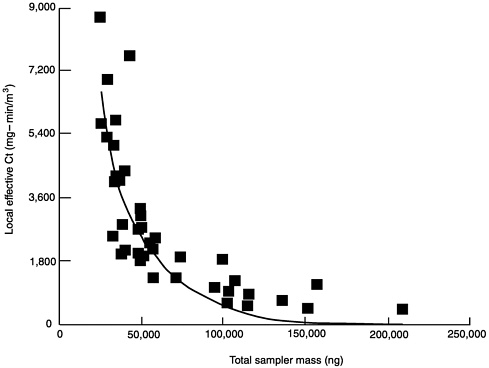
Table 2-8 shows the total mass of methyl salicylate collected in the passive samplers and the geometric mean of the local effective Ct (local analysis for HD) for the 41 protective ensembles (see also Figure 2-3). There is no linear relationship between total mass and local effective Ct for the protective ensembles. Instead, there may be an exponential relationship of the type [Y= A(-kx)] where Y is the local effect Ct and x is the total sampler mass. This equation is plotted in Figure 2-3, where the constants are A = 14,600 and k= 3.40 x 10-5. The results suggest that the total sampler mass alone may be useful for the preliminary ranking of chemical protective values.
|
2 |
See Appendix A for the derivation of this equation. |
TABLE 2-6 Ranking of Protective Ensembles by Systemic Effective Cta
|
Rank |
Ensembleb |
Nc |
Geometric Meand |
Geometric Standard Deviatione |
|
1 |
Vapor protective, fire resistant, protective undergarment |
14 |
3,225 |
2.12 |
|
2 |
Duty uniform, non-fire resistant, model 1 |
8 |
3,177 |
2.24 |
|
3 |
Duty uniform, non-fire resistant, model 3 |
7 |
3.003 |
1.89 |
|
4 |
Duty uniform, non-fire resistant, model 2 |
4 |
2,466 |
1.56 |
|
5 |
Duty uniform, fire resistant, model 1 |
6 |
2,463 |
2.05 |
|
6 |
Overgarment, non-fire resistant, model 3 (r) |
6 |
2,409 |
1.57 |
|
7 |
Overgarment, non-fire resistant, model 3 (w) |
6 |
2,278 |
1.30 |
|
8 |
Overgarment, non-fire resistant, model 2 (w) |
6 |
2,260 |
1.76 |
|
9 |
Saratoga chemical protective Suit (worn over undergarments only or over duty uniform) (w) |
6 |
2,230 |
1.24 |
|
10 |
Vapor protective, fire resistant, protective undergarment (w) |
4 |
2,189 |
1.80 |
|
11 |
Duty uniform, non-fire resistant, model 2 |
10 |
2,122 |
1.90 |
|
12 |
Overgarment, fire resistant, model 2 (w) |
6 |
2,033 |
1.92 |
|
13 |
Standard U.S. Army chemical protective undergarment |
24 |
1,835 |
1.76 |
|
14 |
Overgarment, non-fire resistant, model 2 |
23 |
1,759 |
1.80 |
|
15 |
Duty uniform, non-fire resistant, model 3 (w) |
6 |
1,725 |
1.049 |
|
16 |
Duty. uniform, fire resistant, model 2 (w) |
6 |
1.701 |
1.97 |
|
17 |
Overgarment, non-fire resistant, model 1 (w) |
6 |
1,698 |
2.40 |
|
18 |
Overgarment, fire resistant, model 2 |
23 |
1,679 |
1.83 |
|
19 |
Overgarment, non-fire resistant, model 1 |
19 |
1,642 |
1.80 |
|
20 |
Overgarment, fire resistant, model 1 |
29 |
1,552 |
2.05 |
|
21 |
Saratoga chemical protective suit (worn over persona] undergarments only or over duty uniform) |
15 |
1,552 |
1.66 |
|
22 |
Standard U.S. Army chemical protective undergarment (w) |
4 |
1,540 |
1.50 |
|
23 |
Duty uniform, fire resistant, model 2 (w) |
6 |
1,531 |
1.67 |
|
24 |
Duty uniform, fire resistant, model 1 (w) |
2 |
1,506 |
1.32 |
|
25 |
Battle dress overgarment |
29 |
1,494 |
1.91 |
|
26 |
Overgarment, non-fire resistant, model 1(r) |
6 |
1,285 |
1.27 |
|
27 |
Overgarment, fire resistant, model 3 |
19 |
1,201 |
2.08 |
|
28 |
Overgarment, non-fire resistant, model 2 (r) |
6 |
1,124 |
1.37 |
|
19 |
Duty uniform non-fire resistant, model 1 (w) |
6 |
972 |
2.65 |
|
30 |
Air crew uniforms, model 2 |
15 |
944 |
1.49 |
|
31 |
Air crew uniforms, model 1 (w) |
6 |
885 |
1.99 |
|
32 |
Air crew uniforms, model 1 |
10 |
857 |
1.55 |
|
33 |
Air crew uniforms, model 2 (w) |
6 |
838 |
1.27 |
|
34 |
Overgarment. non-fire resistant, model 1 (w) |
6 |
716 |
1.67 |
|
35 |
Standard U;S. Army chemical protective undergarment |
2 |
585 |
1.35 |
|
36 |
Saratoga chemical protective suit (worn over persona] undergarments only or over duty' uniform (r) |
5 |
487 |
1.64 |
|
37 |
Overgarment, non-fire resistant, model 3 (nr) |
4 |
451 |
1.53 |
|
38 |
Overgarment, non-fire resistant, model 1 (nr) |
4 |
429 |
1.32 |
|
39 |
Overgarment, non-fire resistant, model 2 (nr) |
4 |
411 |
1.07 |
|
40 |
Saratoga chemical protective suit (worn over personal undergarments only or over duty uniform (nr) |
5 |
270 |
1.39 |
|
41 |
Battle dress uniform (standard military uniform) |
4 |
38 |
1.07 |
|
aCt is agent concentration (mg/m3) x time of exposure (in minutes) b Various protective ensembles tested (w = worn. r = repaired, nr = not repaired) c Number of test replicates for each ensemble d The geometric mean, the nth root of the product of the n data or e The geometric standard deviation. Σ (standard deviation of logarithms of values) |
||||
TABLE 2-8 Comparison of Total Mass of Methyl Salicylate Collected in Passive Samplers versus Geometric Mean of Local Effective Ct (Local Analysis for Mustard Gas)
|
Protective Ensemblesa |
Total Mass (µg) Collected in Samplers |
Geometric Mean of Local Effective Ct (mg/m3-mm) |
|
Duty uniform, fire resistant, model 1 |
24.2 |
8,730 |
|
Duty, non-fire resistant, model 3 |
25.1 |
5,690 |
|
Overgarment, non-fire resistant, model 3 (w) |
27.5 |
6,930 |
|
Duty uniform, non-fire resistant, model 2 |
28.7 |
5,320 |
|
Overgarment, non-fire resistant, model 3 (r) |
32.1 |
5,000 |
|
Vapor protective, fire resistant, protective undergarment |
32.4 |
2,490 |
|
Duty uniform, fire resistant, model 1 |
33.6 |
4,050 |
|
Saratoga chemical protective suit (worn over personal undergarments only or over duty uniform (w) |
33.8 |
5,800 |
|
Overgarment, non-fire resistant, model 2 (w) |
33.9 |
4,210 |
|
Overgarment. fire resistant, model 2 (w) |
36.5 |
4,100 |
|
Duty uniform, non-fire resistant, model 3 (w) |
37.4 |
2,840 |
|
Duty uniform, fire resistant, model 2 (w) |
38.0 |
2,000 |
|
Duty uniform fire resistant, model 2 |
39.6 |
4,330 |
|
Vapor protective, fire resistant, protective undergarment (w) |
40.0 |
2,100 |
|
Duty uniform, fire resistant, model 1 (w) |
42.0 |
7,610 |
|
Standard U.S. Army chemical protective undergarment |
47.7 |
2,000 |
|
Overgarment, fire resistant, model 2 |
48.0 |
2,700 |
|
Saratoga chemical protective suit (worn over personal undergarments only or over duty uniform.) |
49.1 |
1,800 |
|
Overgarment, non-rite resistant, model 2 |
49.3 |
3,090 |
|
Overgarment. fire resistant, model 1 (w) |
49.3 |
3,280 |
|
Overgarment. non-fire resistant, model 1 (r) |
49.9 |
2,780 |
|
Duty uniform, non-fire resistant, model 2 (w) |
51.3 |
1,930 |
|
Overgarment fire resistant, model 1 |
55.6 |
2,310 |
|
Overgarment. non-fire resistant, model 2 (r) |
56.6 |
2,130 |
|
Standard U.S. Army chemical protective undergarment (w) |
57.4 |
1,300 |
|
Overgarment. non-fire resistant, model 1 |
58.0 |
2,440 |
|
Battle dress overgarment |
71.2 |
1,300 |
|
Overgarment, non-fire resistant, model 3 |
73.8 |
1,900 |
|
Air crew uniforms, model 2 |
94.8 |
1,040 |
|
Duty uniform, non-fire resistant, model 1 (w) |
99.6 |
1,820 |
|
Air crew uniforms, model 2 (w) |
102.4 |
590 |
|
Air crew uniforms, model 1 |
103.8 |
930 |
|
Overgarment, non-fire resistant, model 1 (w) |
107.2 |
1,220 |
|
Overgarment, non-fire resistant, model 3 (nr) |
114.9 |
530 |
|
Air crew uniforms, model 1 (w) |
115.3 |
850 |
|
Saratoga chemical protective suit (worn over personal undergarments only or over duty uniform (r) |
115.5 |
510 |
|
Overgarment, non-fire resistant 1 (nr) |
135.5 |
680 |
|
Standard U.S. Army chemical protective undergarment |
151.3 |
450 |
|
Overgarment, non-fire resistant, model 2 (nr) |
156.6 |
1,120 |
|
Saratoga chemical protective suit worn over personal undergarments only or over duty uniform (nr) |
209.0 |
420 |
|
Battle dress uniform, (standard military uniform) |
1,788.8 |
50 |
|
a w = worn, r = repaired, nr = not repaired |
||