4
Test Methods and Sampler Selection
PURPOSES OF MONITORING
The MIST (man-in-simulant test) program is intended to establish protective factors for a personal protective equipment ensemble. Estimates of possible battlefield concentrations of chemical agent, the BRHA (body region hazard analysis), estimates of the cutaneous toxicity of agents, and personal factors, such as heat loading, may ultimately provide a rational basis for determining the relative acceptability of chemical protective ensembles. A protective factor is defined as the ratio of the concentration of the contaminant in air outside the suit to the concentration of the contaminant in air inside the suit. In the MIST program, the concentrations inside and outside the suit are measured at multiple sites by passive samplers. These concentrations can be determined by a variety of methods, which are discussed below. The precision and accuracy of any system is limited by its least precise or least accurate component. Measurements inside the suit will obviously be more difficult to take than measurements in the chamber and will be a more likely source of error.
The current monitoring system includes two layers of sophistication beyond determining a simple protective ratio, an estimate of the protection afforded to each region of the body and, ultimately, an estimate of the absorbed dose of simulant. Measuring protection by body region requires taking multiple samples at the same time from different places inside the suit. Although this creates a few special problems, it is still easier than estimating the absorbed dose of simulant.
Absorbed dose refers to the amount of simulant that penetrates the skin and enters the regional tissue or blood. The effective dose is the amount that actually reaches and injures living cells. Because the outer layer of skin consists of dead cells, simple skin exposure cannot be
equated with poisoning. But there are many other routes to a living cell. For example, the toxicant may be ingested with food or water, it may be injected through the skin (carried on a shell fragment, injected by rupture of a pressure line, or deliberately injected), or it may be inhaled and absorbed through the large surface area of the lungs. Injection and ingestion are routes of exposure that are beyond the scope of the MIST program and are not considered here.
The absorbed dose differs from the effective dose because some of the agent that enters viable tissue or the blood stream may be destroyed before it reaches cells in the target organs. In addition, rates of detoxification may vary greatly among individuals. These differences can be genetically determined or modified by the environment (e.g., exposure to other drugs or chemicals).
Direct measurements of absorbed dose would require biological monitoring (i.e., drawing blood specimens, collecting urine, saliva, or breath samples, or taking tissue samples for analysis). Indirect estimates of absorbed dose require either simulating resistance to penetration by the skin or knowledge of how skin penetration varies with body region. The skin acts as a barrier in several ways. First, the keratinized epithelium (the layer of dry dead cells on the surface) acts as a passive barrier. The effectiveness of the passive barrier depends partly on the lipid solubility of the simulant but also on the thickness of the keratinized layer and the degree to which the skin is populated with sweat and sebaceous glands, which allow easier access to the blood. The thickness of the living layers of tissue below the keratinized layer also varies widely. Differences in thickness have a slight effect on penetration by simply increasing the opportunity for passive diffusion but may have a greater affect by increasing the hydrolysis and metabolism of the simulant during passage.
RATIONALE FOR THE SELECTION OF MONITORING METHODS
The monitoring methods for both chamber and in-suit concentrations should be precise and accurate. Ideally, monitoring methods should report concentrations to within a few percentage points of the true concentration of the simulant. In fact, the concentrations in the test chamber may vary more than a few percentage points; and the variations among suit trials and different wearers may greatly exceed the error introduced by monitoring. Nevertheless, precise and accurate
measurements should be attainable within the design constraints of the test program and should be one of the most important criteria for choosing monitoring systems.
A monitoring method should not introduce errors. The chamber monitors should not alter the airflow within the chamber in such a way as to cause the concentrations to vary in different parts of the chamber. The in-suit monitor must not compromise the integrity of the CPE. Sampling systems that penetrate the suits alter the barrier properties of the suits or alter the form, fit, or function of the suit in other ways and could influence test results. Tubes that penetrate the suit to draw air from sampling points are the most obvious example of a monitoring system that alters the suit integrity. The barrier properties of a suit might be altered by adhesive tape used to affix samplers to the outer shell or by belts that compressed the sorbent layers or held them against the body allowing them to absorb perspiration. Alterations in the form, fit, or function most often apply to masks but could also affect suit performance. For example, bulky objects under a protective garment could increase the ''bellows effect'' whereby contaminated air enters the suit during some motions and leaves during others. Relatively small exchanges, if repeated often enough, can rapidly equilibrate concentrations inside and outside a garment. Bulky samplers under the garment could stretch neck, wrist, or ankle openings and degrade suit performance.
The weight and bulk of sampling equipment that a subject carries under the suit must not change activities or the level of effort required to accomplish tasks. Thus, the subject could not carry sampling pumps next to the skin, even though they would probably give the most precise measurements.
Many studies have shown dramatic differences in the absorption of chemicals through the skin of various body regions. In other words, absorption through the skin, unlike absorption through the lungs, is not simply proportional to the vapor concentration. Therefore, it may be desirable for the MIST program to simulate at least some skin penetration and absorption properties with the sampler rather than attempting to estimate the absorption from air concentrations.
CHAMBER MONITORING
The concentration in the chamber (outside the suit) is determined by the airflow rate into the chamber and the rate of simulant release
into the air. According to the MIST test plan (Hanzelka et al., 1996), surfaces of the test chamber are stainless steel. Air flows through the chamber at 3.2 to 16.1 km/hr (2 to 10 mph or 0.89 to 4.5 m/s). The concentration is monitored by fixed samplers, called miniature infrared analyzers (MIRANS), located on the upwind end of the room and on the right and left sides of the inflow wall. Additional miniature automated chemical air monitoring systems are located in antechambers but are not used to monitor concentrations in the chamber. The detailed test plan does not indicate how the simulant is introduced into the air stream, what mechanisms are used to assure uniformity of simulant concentration within the air stream, or how much of the front wall is occupied by the inflow ducts. The MIST plan provides no information about the precision or accuracy of the MIRANS.
Supplying air with a fairly constant concentration of simulant over an extended period of time, in the absence of a mechanism for removing or absorbing significant amounts of the simulant, would tend to create a uniform concentration in all parts of the chamber. Maintaining uniform concentrations in animal exposure chambers, however, has proved to be a problem in toxicology experiments (Smith and Fowler, 1985).
The simulant is removed from the chamber air by respiration through the mask filters at a rate equal to the concentration in the air times the respiratory minute volume. The breathing rate will vary with activity. The respiratory rate for moderate activity in a workplace setting is assumed to be 10 m3/8-hr shift, or 20.83 1/min. Exact chamber dimensions are not given in the test plan, but if one assumes a flow path of 5 m, there would be more than 600 air changes per hour even at the lowest flow rates. Therefore, removal is unlikely to appreciably diminish even the local concentration at any point in the chamber.
The simulant is also removed from the air by adsorption in or on the chamber surfaces and especially on protective garments. That rate will clearly vary with the efficacy of the suit and the type and intensity of activity and the position of the subject within the chamber. Army officials indicated that the chamber is allowed to reach equilibrium before tests are begun (Malabarba and Fidele, 1996). This should adequately control for the deposition of simulant on chamber surfaces but not necessarily on the suits. The large number of air changes per hour, even at low flow rates, should preclude an appreciable effect of simulant adsorption to chamber surfaces or protective garments on the simulant concentration. Actual chamber measurements are the only way to confirm this.
As an alternative to chamber monitoring at various locations, the MIST program has adopted the ingenious approach of placing passive monitors, identical to those used inside candidate ensembles, at identical positions on the outside of an impermeable garment. The impermeable suit is then worn by another subject who mimics the motions of the subject testing the candidate protective ensemble. This allows for comparisons of simulant concentrations at specific locations around the suit (e.g., elbow, knee, etc.)
OPTIONS FOR IN-SUIT MONITORING
This section outlines three major approaches that might be used to determine the amount of simulant inside a protective ensemble. Advantages and disadvantages of each sampling system are considered.
Active Sampling Systems
Overview
Active sampling systems, whether internal or external to the suit, draw air through a sorbent at a measured rate. The sorbent removes most (ideally all) of the chemical of concern. The chemical is then removed from the sorbent by solvent extraction or by heating the sorbent until the chemical is released. A variety of chemical analysis systems can then be used to measure the total amount of chemical desorbed. The total is then divided by the volume of air drawn through the sorbent to approximate the concentration. Because some chemical is usually retained on the sorbent and cannot be measured, chemists usually refine the estimate by calculating the fraction of chemical recovered from sampling tubes that have been spiked with a known amount of the analyte and dividing the quantity observed in samples by that fraction. This mechanism adjusts for proportional errors (the slope of the recovery curve). Other measurements can be used to adjust the zero-point (the interception of the recovery curve).
Active systems can introduce errors through uncertainties or variations in the flow rate. The pump is one potential source of variation. If tubing connects the pump to the sorbent tube or canister, bends or kinks in the tubing may also introduce error. The pump rate of
battery-powered pumps may diminish as the battery discharges, but that problem has been well studied, and industrial hygienists usually calibrate the pumps at the beginning and end of each sampling period. For small changes, an average flow may be used. Nickel-cadmium batteries usually fail along a gradual, nearly linear curve, with rapid decay in voltage and current near the end of the cycle life. By avoiding the late phase, an average flow rate will give a satisfactory estimate provided that the concentration does not vary greatly during the test period. With some analytes, especially particulates or aerosols, a mechanical barrier may be created by clogging of the sorbent bed, which may increase resistance and decrease flow rates during the sampling period. It is not anticipated that this problem will arise with MeS as the primary simulant.
With either active or passive systems, the recovery of chemical decreases as the sorbent becomes saturated. For that reason, the size of the sorbent bed, the sampling rate, and the sampling time must be chosen so that the sorbent remains well below its saturation point. The problem is simpler with active systems, in which retained quantities may be large as long as the sorbent binds the analyte tightly enough to prevent breakthrough (passage of analyte completely through the system).
Other sources of error with either passive or active systems include influence of temperature, humidity, and the presence of other vapor constituents. These factors can be assessed by controlled trials of the system prior to a study. Data for the passive samplers show small, predictable variations with temperature and little variation with humidity effect. The effect of humidity on active samplers would probably be greater than on passive samplers with nonporous membranes.
External Pumps and Tubes
A system with pumps, connecting tubes, and samplers on the outside of a suit, with tubes passing through openings into the suit, has the advantage of permitting the use of bulkier equipment with better control over flow rates, sorbent bed volume, and operating temperature of the sorbent bed. Instruments that take direct readings might even be adaptable for intermittent monitoring. Measurements of airborne chemical concentration, the sorbent-to-analysis phase of the measurements, should not be affected, providing that the simulant does not adhere strongly to the tubing. Because of its low volatility,
however, adherence to tubing is a severe problem with VX analysis systems, making direct collection of VX on a sorbent essentially impossible. With VX, the monitoring problem has been solved by installing a conversion pad, which substitutes a fluorine atom for the N,N-diisopropylamino-2-ethyl-thiol group of VX (see Figure 3-1). Similar solutions might be found for testing low volatility simulants.
The major disadvantages of external monitors are: (1) they compromise the integrity of the chemically protective ensemble being tested; (2) they limit motion if a fixed or bench-top system is connected to the subject; and (3) they add weight and bulk if the subject carries portable samplers.
The MIST study is designed to monitor in-suit concentrations at a large number of body sites. Tubes running either through the shell of the suit or parallel to the skin through neck, wrist, waist, or ankle openings could easily introduce gaps that would allow contaminated air to penetrate in a way that would not occur with an intact suit. Holes in the shell could be more readily sealed than gaps at openings, but this would require either multiple openings or passing tubes through the waistline or other sealing points, which creates the risk of abnormal simulant migration between body regions. Tubes running through the suit to external samplers would need to be collected into a bundle to prevent tangling during activities. To prevent distortion in the fit, the bundle would need to be attached to the suit with adhesives or to the soldier with a belt or harness. Adhesives can alter the barrier properties of the suit either by increasing or reducing permeability and can alter the fit by pulling on and distorting the suit, potentially aggravating the bellows effect that moves air in and out of the suit with motion. Straps may alter the results in a number of ways: they could decrease the normal flow of air between body regions by constricting the space between suit and skin; they could alter permeation qualities by compressing the suit material; they could decrease the area exposed directly to simulant concentrations and thereby decrease the in-suit concentration; and they could increase sweat absorption by holding the suit next to the skin, which could alter suit permeability. Finally, an active sampling system draws make-up air from outside the suit into the suit, which would necessarily increase the amount of simulant beneath the shell. This might be partially corrected by introducing an amount of clean air that precisely matches the amount of air that was removed. The process, however, would alter the motion of simulant within the suit leading to artificially high or, more likely, abnormally low readings.
Although many of these problems could be overcome with sufficient ingenuity and effort, collectively they indicate a significant potential for altering the form, fit, and function of the suit in ways that are difficult to predict. Using a single vacuum line or pump with a manifold system to multiple sampling points, for example, would solve some of the problems but would introduce uncertainty in the volume of air pulled through each sampler. Balancing a manifold system is difficult even with rigid tubing and would be even more difficult with flexible tubing. If a fixed or bench-top system were connected to the subject, the connecting tubes would serve as a tether, limiting the activities the subject could perform. Several pumps could be carried within the weight limits of a field pack, but then exercise protocols that do not require use of the field pack would be severely limited. The bulk of external portable samplers would be less problematic than the weight but might create problems if exercise protocols required crawling. The design of active samplers would make it difficult to simulate the barrier properties of skin. An absorbed dose could not be approximated with this system.
The problems described here do not necessarily eliminate sampling with pumps external to the suit. That strategy might still be the best option because of the physical constraints of active samplers and the inaccuracy of passive samplers. The precision and accuracy of the analytical results of active samplers would probably not compensate for their disadvantages.
Pumps and Sampling Systems inside the Protective Garment
As an alternative to penetrating the shell of a protective ensemble with sampling tubes, pumps could be worn between the garment and the skin. An advantage of this scheme is that the pump would exhaust air into the space inside the garment, so no make-up air would have to be drawn from the outside. A disadvantage is that a new error would be introduced by placing a clearing mechanism within the suit. If the amount of simulant removed on the sampling medium were small in comparison to the flux through the suit, however, the error would be negligible. The precision and accuracy of internal pumps could be comparable to external samplers.
Although the structural integrity of the chemical protective ensemble being tested would be maintained by sampling pumps hung inside the garment, the garment's form, fit, and function would all be altered significantly. Form would be altered by pulling the garment away from
the body over the pumps, which could open gaps at the waist or neck. It would also pull the garment closer to the skin on the opposite side, increasing sweat loading. The fit could be altered in many ways at various places, especially at friction points (waist, groin, underarm, neck, etc.) Protective garments that are designed to be "form fitting" would make the introduction of air sampling equipment particularly difficult. Function could be altered by increasing the heat loading within the garment as the result of the heat generated by the pump. If the number of pumps required does not fit practically under the suit, compromises in the number of sample points or the number of tests might be necessary.
In general, the design of active samplers does not lend them to simulation of skin barrier properties. An absorbed dose could not be approximated by a physical system. Sampling with pumps under the suit might still be the best option if the inaccuracies of passive samplers or other constraints preclude their use. Reducing the number of sampling points per test with active sampling would be troubling, but the requisite data could be gathered using more tests. The effect of heat loading could be calculated from information about the rate of heat generation by the pumps. If only a few pumps are used, the added stress might be negligible.
Passive Samplers
Passive dosimetry has become very popular for personal monitoring in recent years (Soule, 1991). The dosimeters or monitors use Brownian motion to control the sampling process, enabling lightweight, low-cost personal monitors that do not require a power source. They rely on a concentration gradient across a static or placid layer of air or other medium to induce a mass transfer. The following equation, based on Fick's law, gives the steady-state relationship for the rate of mass transfer:
W = D(A/L) (C1–C0)
where W is the mass transfer rate, D is the diffusion coefficient, A is the frontal area of the static layer, L is the length or depth of the static layer, C1 is the ambient concentration, and C0 is the concentration at the collection surface.
If an effective collection surface is chosen, C0 can be essentially zero, so the mass transfer or collection rate is proportional to the vapor
concentration C1. Note that the units of D(A/L) are volume per unit time, the same as for volumetric flow in a pump monitoring system. The rate of sampling of the contaminant is then the product of the D(A/L) term and the average ambient concentration.
The precision and accuracy of the overall system depend on the sampling process and the analytical steps. Precision and accuracy of the sampling process depend on the measured exposure time, velocity, and temperature, whereas the precision and accuracy of the analytical steps depend on the calibration standards, properties of the collection media, and the analytical method. The potential effects of velocity and temperature distinguish this type of monitoring device from the conventional dynamic or flow monitor.
When L (dependent on the resistance of the film barrier in this type of sampler) is large compared with the average boundary-layer thickness, sampling is barely affected by velocity. For temperatures between 10°C and 31°C, the variance should be no more than 1.8 percent. At higher or lower temperatures, corrections may be required.
An internal Army memo from 1994 discusses the effects of several factors on accumulated mass. The temperature effect is about 0.1 percent/degree F. In a cavity-type passive collector, the ratio of the sampler length to its diameter must be greater than three so as to minimize the effect of convection within the chamber. At low tangential face velocity, V, the uptake rate decreases because of external resistance to mass transfer in the boundary layer. Diffusion resistance through the boundary layer is proportional to V0.5 for laminar flow and V0.8 for turbulent flow. A commercial passive sampler (DuPont PRO-TEK™) exhibits a marked decrease in sampling rate below a laminar flow velocity of 1,000 cm/min (0.17 m/s). At a face velocity of 12 cm/min (0.2 m/s), the sampler collects 20 percent less vapor than the predicted amount, regardless of boundary-layer effects.
Theoretical models, laboratory evaluations, and field studies agree that diffusion samplers provide a reliable measure of mean vapor concentration if the fluctuation frequency is at least five 5 seconds. Specifically, steady-state mass uptake is approached closely if the period of the concentration change is less than 1.4 times the mean residence time of vapor in the diffusion zone tR, where tR = L2/2D. The DuPont PRO-TEK™, which has a diffusion path length of 0.95 cm, will attain steady-state uptake if the fluctuation period is less than nine seconds, assuming that the diffusion coefficient for MeS at 80°F (27°C) in air is about 0.05 cm2/s.
The key selection criteria for the MIST passive sampler include: (1) projection into the under-the-garment air space is sufficiently small so that it accurately samples the unaltered vapor stream in the vicinity of the sampling device; and (2) the absorption velocity Wv for MeS under MIST conditions is between 1 and 4 cm/min, the range observed for skin uptake of agents. Orifice-based samplers for which Wv = D/L would require a diffusion path length of 0.75 to 3 cm to achieve the desired Wv range with MeS. Although this exceeds the dimensional requirement, interposing a barrier membrane in proximity to the adsorbent with a minimal diffusion path would solve the problem.
Natick Sampler
The Natick sampler is a type of passive sampler developed at the Natick RDEC specifically to detect MeS vapor for the MIST. The Natick system clearly protects the integrity of the protective ensemble. The samplers are no thicker than a common adhesive bandage and are less than 1 inch square (see Figure 4-1). Solubility of MeS in high-density polyethylene (HDPE) was reasonably linear over concentrations from 0 to 135 mg/m 3 with a coefficient of (1-mg MeS/gm HDPE)/ (125-mg MeS/m3) = 0.0082 m 3 /gm HDPE. The weight of a 1-mil HDPE cover on a Natick sampler is about 0.018 gm, so at a chamber concentration of 150 mg/m3, the quantity of adsorbed MeS would be:
(0.0082-m3/gm HDPE)(0.018-gm HDPE/sampler)(150 mg/m3)
= 0.022 mg per sampler
After treating the polyethylene films by heating to 95°C to 100°C for 16 hours, permeability to carbon tetrachloride increased substantially. At 18°C, permeability increased from about 55 gm/m2-day before treatment to about 80 gm/m2-day after treatment. At 40°C, the corresponding values were 120 and 160. Army scientists recommend pretreating the films in all cases to assure uniformity.
Water vapor often competes with other chemicals for binding sites on the sorbent, decreasing the sorbent's capacity to absorb the test chemical. Ambersorb and Tenax show less variation with humidity than carbon, but Army officials assert that the humidity effect has been insignificant in the Natick RDEC sampler. In contrast, humidity increased the skin absorption of organic compounds with partition
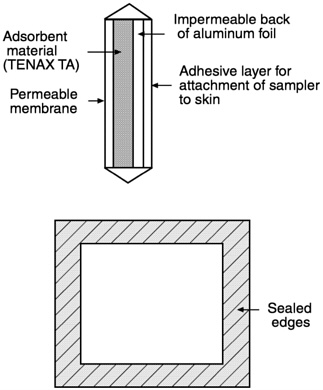
Figure 4-1 Diagram of Natick sampler.
coefficients in the range of 0 to 3 (Hawkins and Reifenrath, 1984). The partition coefficients of many chemical warfare agents fall within this range. These findings lead to the obvious conclusion that the permeability properties of skin and polyethylene are different, especially for a nonporous lipophilic membrane like polyethylene, which does not readily transmit water to the sorbent layer.
The committee asked whether the same basic design would work with a different simulant. Army officials stated that the membrane has been tested successfully with a large number of chemicals. Confirming the effectiveness of a complete sampler would involve repeating some tests, but it seems likely that a suitable sorbent could be attached to the membrane and the design used to test other simulants.












