3 Pharmacokinetics of Permethrin
ABSORPTION OF PERMETHRIN
The two routes of exposure most relevant to evaluating permethrin toxicity from military uniforms are the oral and dermal routes. Consideration of oral absorption of permethrin in animals is necessary to estimate the absorbed dose in carcinogenicity studies, which were conducted by oral gavage. Dermal exposure, in contrast, is the most likely route of exposure to military personnel wearing uniforms impregnated with permethrin.
Oral Absorption
Oral absorption of permethrin was investigated in rats given permethrin in dimethyl sulfoxide at 1.6-4.8 mg/kg of body weight and was estimated to be about 70% for the cis/trans (35:65) isomer mix (CEPA, 1992). Only 3-6% of the dose was detected in feces as unmetabolized and, presumably, unabsorbed permethrin, suggesting that actual absorption might be higher than 70%. Likewise, Anadon et al. (1991) estimated bioavailability of permethrin to be 60% by comparing the “area under the curve” (AUC) for permethrin in blood after gavage with the AUC after intravenous injection. That low estimate could be due to first-pass biotransformation of permethrin by the liver following absorption.
Thus, although the precise gastrointestinal absorption of permethrin is not known, those two studies suggest that 70% is a conservative estimate of absorption by rats. That information is needed to estimate the actual absorbed dose in carcinogenicity studies in which permethrin is administered by gavage.
Dermal Absorption
The dermal absorption of permethrin is more relevant to human exposure and has been investigated in a number of laboratory animals, including rats (Shah et al., 1987), monkeys (Sidon et al., 1988), and mice (Shah et al., 1981). Limited studies on the dermal absorption of permethrin in humans have also been conducted. The percutaneous absorption of permethrin is generally lower in humans than in other mammalian species (CEPA, 1992). The results of several relevant studies are summarized below.
Absorption in Humans
The absorption of permethrin was determined in humans when the permethrin was applied to the scalp in a cream-rinse shampoo or applied to the entire body in a dermal cream (CEPA, 1992). The absorption rates in three studies were reported to be less than 1%. However, it was suggested that the hair rinse and cream vehicles might have hindered absorption. A fourth study was conducted in which isopropanol was used as the vehicle and 14C-labeled permethrin (labeled in the carbonyl moiety) was applied to the shaved backs of volunteers (CEPA, 1992, ref. 16). (Studies with animals showed that when permethrin is labeled in this position, there is no loss of label through production of 14CO2.) In the fourth study, from 0.3% to 2% of the applied dose of 14C-label was excreted in urine in 5 days. This is not a precise estimate of dermal absorption because of the possibility of binding of permethrin to macromolecules or storage of permethrin in body tissues such as fat. Based on these studies, a dermal absorption rate of 2% was assumed for humans by the Medical Toxicology and Worker Health and Safety Branches of the Department of Pesticide Regulation of CEPA in their
risk characterization of permethrin (CEPA, 1992). Corresponding parenteral studies were not conducted; therefore, that absorption value can only be considered an estimate.
In another study, the transdermal absorption of permethrin, formulated in a cream at a 5% concentration, was reported (van der Rhee et al., 1989). Patients with scabies were treated with one application of that cream to the skin of the whole body, excluding the head and neck, for 8 hr, after which the patients were instructed to wash their skin. The degree of permethrin absorption was assessed indirectly by determination of conjugated and unconjugated cis/trans CVA (3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylic acid, a metabolite of permethrin) excreted in urine. The results of these studies showed that permethrin could be absorbed through the skin, although they did not determine precisely how much was absorbed. They also demonstrated that excretion of CVA, which began within the first 7 hr, was at a maximum over the first 48 hr but continued at a low rate for at least 14 days, suggesting continued elimination of absorbed permethrin.
Estimates of permethrin absorption derived from metabolite excretion studies in healthy individuals suggested that the maximum absorbed by any one individual over the first 48 hr after application of 16 g of cream (800 mg of permethrin) was 18 mg and that the overall mean absorption by the group was 10 mg (unpublished data, cited by van der Rhee et al., 1989). Those studies with healthy volunteers were similar to the studies with scabies patients in that the amounts of permethrin absorption, as measured by the mass of urinary metabolites, are of the same order of magnitude in both studies.
In summary, although several dermal absorption studies were conducted in humans, these studies lacked corresponding parenteral controls and must be interpreted cautiously. In addition, no studies have been conducted in humans that focus on permethrin absorption under conditions of intended use by the Army, namely, dermal absorption of permethrin when applied to cloth.
Absorption in Animals
An acetone solution of permethrin was applied to the shaved backs of
mice to investigate dermal penetration (Shah et al., 1981). Of the 14 insecticides studied, permethrin penetrated the skin of mice most rapidly, the T1/2 being 5.9 min. Within 5 min, 40% of the permethrin had moved from the site of application. Absorption of the insecticides did not appear to correlate with physicochemical factors, such as molecular weight, solubility, charge distribution, or partition coefficients (Shah et al., 1981). Those investigators also examined age dependence in dermal penetration of insecticides in young (33-day) and adult (82-day) female F344 rats. There was no age-related difference in the penetration of permethrin in young and adult rats. There was, however, a decreased absorption of permethrin with increasing dose (Shah et al., 1987).
The percutaneous absorption of cis/trans permethrin was determined in rhesus monkeys and rats (Sidon et al., 1988). Permethrin labeled at the alcohol or cyclopropyl portion of the molecule was applied to either the forehead or the forearm of rhesus monkeys or to the mid-lobosacral (back) region of the rat. Urine was collected for 7-14 days, and the recovered radioactivity in the urine was compared with that obtained after an intermuscular injection to determine the percent absorption. In monkeys, the forehead was more permeable than the forearm (14-28%, forehead; 5-12%, forearm). According to Siddon et al., the forehead is more absorbent than the forearms because the forehead is more glabrous (smooth and without hairs) and more highly vascularized than the forearms.
The investigators also noted that absorption of permethrin applied to the backs of rats was significantly greater than the forehead or the forearm in the monkey (rats, 43-46%). The interspecies difference supports the finding of higher permeability of rat skin demonstrated previously with other pharmaceutical compounds and underscores the importance of using caution in extrapolating the results of pesticide dermal absorption studies in nonprimate species to humans. Migration of permethrin from clothing to the skin surface is important for assessing the toxicity of permethrin when impregnated into uniforms. There are no human data on the absorption of permethrin from impregnated cloth rather than from direct application to skin. However, studies on the transfer of permethrin from treated cloth to skin surface have been conducted in rabbits (Snodgrass, 1992). In those studies, permethrin migration from cloth to skin was determined by measuring the fate of the 14C label when swatch-
es of fabric impregnated with 14C-labeled permethrin were applied to the backs of rabbits for 1 week. The results showed that fabric treated with permethrin at a rate of 0.125 mg/cm2 lost the substance to the skin surface at an average rate of 0.49% per day. At the end of 7 days, about 3.2% of the available permethrin had reached the skin, 2% having been recovered from the excreta (absorbed) and 1.2% remaining on the skin surface. Other variables, such as temperature, fabric type, sweat, and prelaundering, had little effect on migration rate (Snodgrass, 1992). Although the rabbit studies do not address the important issue of absorption of permethrin through the skin of humans from permethrin-impregnated cloth, they can be used to estimate the rate of transfer of permethrin from cloth to the skin. Although absorption of permethrin through the skin of humans and rabbits is likely to be much different, physical transfer of permethrin from cloth to skin should be similar.
INTERACTIONS
A combination insecticide and repellent product (Hartz's Blockade) composed of 9% N,N-diethyl-m-toluamide (DEET) and 0.09% fenvalerate (a Type II pyrethroid insecticide) has been widely used for the control of fleas and other ectoparasites on dogs and cats. During 1987, the Illinois Animal Poison Information Center observed 200 times more poisonings in dogs or cats exposed to Blockade than to all other DEET-containing products. This report indirectly suggests that the use of DEET in combination with a pyrethroid insecticide increases the toxic response possibly by facilitating fenvalerate absorption (Dorman, 1990).
The possibility for enhancement of permethrin absorption by DEET is supported by a study on the use of DEET to enhance transdermal delivery of drugs. In in vitro diffusion studies using the hairless mouse skin as a barrier membrane, the absorption of a number of drugs (hydrocortisone, benzocaine, ibupropen, erythromycin, etc.) was studied (Windheuser et al., 1982). Although the diffusion cell results are not necessarily applicable to skin in vivo, they can demonstrate relative penetration enhancement. Enhancement of drug delivery is dramatic when DEET is present in the formulation. Drug diffusion, as expressed as a percent, increased from 1.6% to 35% for hydrocortisone and from not
detectable to 83% for erythromycin. These results suggest that DEET used in combination with permethrin might serve to facilitate the absorption of permethrin through the skin. The transdermal penetration of trisodium phosphonoformate (PFA) in the presence of sorption promoters, including DEET, was studied in excised rat skin. Ten-percent DEET doubled the amount of PFA in the skin, increased the amount permeated across the skin fourfold, and increased the flux fivefold (Hussain and Ritschel, 1988).
Other studies have shown that DEET enhances the dermal penetration of fenitrothion, an organophosphorus pesticide in rats and to a lesser extent in monkeys, when measured by recovery of absorbed fenitrothion in urine of exposed animals (Moody et al., 1987).
The U.S. Army conducted a study to determine the synergistic effect of permethrin and DEET combinations as measured by acute toxicity (Nelson, 1989). The results indicated a 25-40% potentiation of toxicity from the mixtures compared with single chemicals. In that study, the permethrin-DEET mixture was given orally, so the effect of DEET on transdermal absorption of permethrin could not be evaluated. However, the observed potentiation of permethrin toxicity by DEET is consistent with a facilitated absorption of permethrin by DEET across the gastrointestinal surfaces. Additional studies are recommended to assess the potential interactive effect on absorption (see “Recommendations” in this chapter for more details).
METABOLISM
Because permethrin is neurotoxic and carcinogenic in laboratory animals at high doses, an understanding of its metabolic fate after absorption, regardless of the route, is useful. As with other xenobiotics, it is most likely that the liver is quantitatively the most important site for permethrin biotransformation. Given what is known about the similarities in biotransformation enzymes in animals and humans, it is also likely that the metabolic pathway operant in animals will be present in humans.
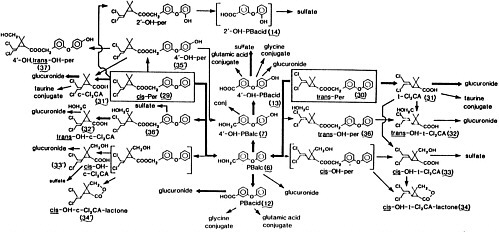
The two major pathways for metabolism of permethrin are oxidation and hydrolysis (Hutson, 1979). Figure 3-1 and Figure 3-2 show the metabolic pathways and sites of metabolic attack. The relative contributions of the
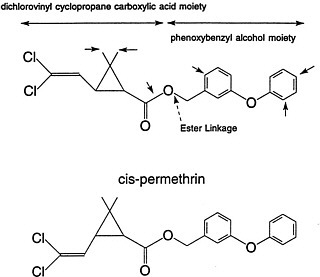
FIGURE 3-2 Chemical structures of cis- and trans-permethrin (3-phenoxybenzyl 3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylase) indicating the position of the ester linkage and the alcohol and acid portions of the molecule relative to the ester linkage. The small arrowheads designate known sites of metabolic attack.
esterase and oxidase in the in vitro hepatic metabolism of permethrin have been estimated for the R, S, cis, and trans isomers. As with other pyrethroids, trans isomer metabolism is dominated by hydrolysis and cis isomer metabolism is dominated by oxidation (Hutson, 1979).
HYDROLYSIS
The major pyrethroid hydrolyzing esterase is located in mammalian liver microsomes and is probably a carboxyl esterase (Hutson, 1979).
The cis and trans pyrethroid isomers show dramatic substrate specificity —the trans form being hydrolyzed up to 50 times faster than the cis form (Casida et al., 1976). Pyrethroid hydrolysis is inhibited by dialkylphosphorylating agents, such as organophosphorus pesticides, in vitro and in vivo (Hutson, 1979; Casida et al., 1983). That raises the question of hazard to pesticide users who might be exposed simultaneously or sequentially to the two types of pesticides. That situation is not likely under the field conditions proposed for use by the Army. However, nonfield military personnel who are wearing permethrin-treated uniforms might have occasion to use organophosphorus pesticides as part of their work duties. Therefore, the possibility of combined exposure should be taken into account when assessing potential risk of acute toxicity. The presence of a hydrolysis inhibitor should prolong the tissue distribution and retention of permethrin.
OXIDATION
Oxidation is also an important route of metabolism for pyrethroids and might be of paramount importance for the cis isomers, since they are less likely to be metabolized by hydrolysis (Glickman et al., 1981; Glickman and Lech, 1981). Oxidative reactions occur at the cyclopropane carboxylic acid moiety, at the alcohol moiety, and also probably in the proximity of the ester bond so that its cleavage is catalyzed. This later process might be very important for cis isomers, which are more resistant to hydrolysis. Additionally, oxidation at peripheral sites, while leaving the ester bond intact, affords points at which conjugation reactions occur, leading to biliary and fecal elimination of the esters (Hutson, 1979). The various c-hydroxylations are probably catalyzed by cytochrome P-450.
RELATIONSHIP TO TOXICITY
The rate of metabolism of a pyrethroid is profoundly important in its acute toxicity (Hutson, 1979). The trans isomers (rapidly hydrolyzed) are more rapidly eliminated and much less neurotoxic than their cis analogs. Inhibition of hydrolytic reactions enhances neurotoxicity. The inhibition of oxidative metabolism also increases neurotoxicity. Piperonyl
butoxide, the most important pyrethroid synergist and a classical mixed-function-oxidase (MFO) inhibitor, increases the toxicity of all pyrethroids to insects (Casida et al., 1976). It also increases the neurotoxicity of pyrethrins to mice. Induction of hepatic microsomal monooxygenases lowers the acute neurotoxicity of permethrin. These facts suggest that hydrolysis and oxidation are important in limiting acute toxicity, although they are not the only factors controlling acute toxicity (Hutson, 1979).
In summary, all of what is known about the metabolism of pyrethroids comes from studies conducted in laboratory animals and from in vitro studies using hepatic tissues. Information on the biotransformation of permethrin at the site of absorption (skin or gastrointestinal tract) or in the target tissue (nervous system) is lacking. Likewise, specific studies of the biotransformation by human tissues have not been conducted. However, the classes of enzymes responsible for permethrin metabolism are found in all mammalian systems, including humans, so it is likely that these pathways are operant in humans also. It is likely that biotransformation will serve to reduce the neurotoxic effects of permethrin in humans as it does in animals.
Several classes of biotransformation reactions might result in the formation of reactive intermediates. Epoxidation is one such biotransformation. Epoxidation can occur at the unsaturated side chains of the natural pyrethrins or at the dihalovinyl side chain of permethrin (Ruzo and Casida, 1977). Epoxides have been implicated in the carcinogenic action of such chemicals as vinyl chloride, styrene, ethylene oxide, and butadiene. However, in the metabolism studies conducted with permethrin reported so far, there has been no evidence of oxygenation at the dihalovinyl group (Hutson, 1979).
In summary, all of what is known about the metabolism of pyrethroids comes from studies conducted in laboratory animals and from in vitro studies using hepatic tissues. Information on the biotransformation of permethrin at the site of absorption (skin or gastrointestinal tract) or in the target tissue (nervous system) is lacking. Likewise, specific studies of the biotransformation by human tissues have not been conducted. However, the classes of enzymes responsible for permethrin metabolism are found in all mammalian systems, including humans, so it is likely that these pathways are operant in humans also. It is likely that biotrans-
formation will serve to reduce the neurotoxic effects of permethrin in humans as it does in animals.
ELIMINATION
The pyrethrin metabolites are generally excreted as alcohols, phenols, or carboxylic acids and their glycine, sulfate, or glucuronide conjugates. At least 80 metabolites have been identified from cis and trans permethrin in various species and systems (IUPAC, 1981).
In a study by Gaughan et al. (1977), the 1R trans, 1RS trans, 1R cis, and 1RS cis isomers separately labeled in the acid and alcohol moieties were given orally to rats, and metabolites in urine and feces were identified. The results showed that there was no significant metabolic difference between 1R and 1RS isomers, although, as noted previously, cis permethrin isomers were more likely to undergo oxidative metabolism than the trans counterparts. Twelve days after administration, 97-100% of the radioactivity was recovered in urine and feces. Unchanged permethrin was detected only in the feces. Virtually no 14CO2 was expired. Radioactivity from the cis isomers tended to be retained longer than that from the trans isomers and that from the alcohol label longer than that from the acid label. The most striking difference was that only 45-54% of the radiocarbon from the cis isomer appeared in the urine, whereas 81-90% of that from the trans isomer appeared in the urine. The more hydrolytically stable cis isomer resulted in metabolites that retained the ester bond, and these metabolites were excreted in the feces, presumably via the bile. The large molecular weight of permethrin (370) suggests that it would be an excellent candidate for biliary excretion. The major metabolites from both isomers were the sulfate and glucuronide conjugates of the phenoxybenzoic acid portion of the molecule and the glucuronide conjugate of the cyclopropane carboxylic acid portion.
Similar results were found in studies using rhesus monkeys (Sidon et al., 1988). As in the previous study, the position of the 14C radiolabel on the permethrin (alcohol or cyclopropyl group) affected the radioactivity recovered in urine. In addition, a lower recovery of radiocarbon in urine was observed following an intramuscular injection of cis permethrin as compared with the recovery after injection of trans permethrin.
This observation is consistent with findings in other studies for intravenous or oral administration. Again, these results are most likely attributed to the more metabolically labile ester group of the trans isomer as compared with the cis isomer. However, they probably also reflect greater excretion of the cis residues via the fecal route or greater retention of the cis isomer in body tissues.
DISTRIBUTION
Tissue Concentrations
Pyrethroids are lipophilic molecules that generally undergo rapid absorption and distribution following ingestion by mammals (Hutson, 1979). Unless isolated in lipid depots, they are quickly metabolized and eliminated from the body. Permethrin persists longer in fat than in other tissues when measured in chickens, rats, goats, and cows (Casida et al., 1983). Cis permethrin is retained longer than its more metabolically labile trans isomer (Marei et al., 1982).
Although many factors combine to determine the toxicity of the chemical in a target organ, one of the major determinants is the tissue concentration of the chemical. In general, 14C-labeled permethrin and its hydrolysis products are excreted from the body in a relatively short time. For example, the toxicokinetics of permethrin taken orally at 460 mg/kg or taken intravenously at 46 mg/kg was studied in male Sprague-Dawley rats (Anadon et al., 1991). The elimination half-time of permethrin was slower for the hippocampus, medulla oblongata, front cortex, and sciatic nerve (16-24 hr) than for plasma (12 hr). Higher amounts of permethrin were also found in those tissues than in plasma, indicating the accumulation of permethrin by nervous tissue. The metabolites of permethrin, m-phenoxy benzoalcohol and m-phenoxy benzoic acid, were detected in plasma and in all selected tissues for 48 hr after dosing.
Studies in Sprague-Dawley rats, in which a variety of pyrethroid insecticides were administered orally, demonstrated that the residues of permethrin in fat and brain were much higher and more persistent with cis permethrin than with trans permethrin (Marei et al., 1982). Fat and brain concentrations of the trans isomer but not the cis isomer were
greatly elevated on pretreatment with either esterase or oxidase inhibitors. Phenobarbital was moderately effective in lowering pyrethroid residues in fat most likely because of enhanced metabolism of permethrin.
Thus, although there is some information on the distribution of permethrin in tissues of mammals, the majority of this information has been obtained in rodents. There have been no studies conducted on the distribution of permethrin in the tissues of primates, including humans.
Protein Binding
The amount of the permethrin in the tissues is of some importance even at concentrations that are lower than the concentrations necessary to produce neurotoxicity. Studies using human skin fibroblast androgen receptors have demonstrated that nonsteroidal compounds, including permethrin, can interact competitively with human androgen receptors and the sex hormone binding globulin (Eil and Nisula, 1990). Those studies provide a mechanism by which chronic exposure of humans to pesticides containing nonsteroidal compounds might result in endocrine disturbances relating to androgen action. The competitive binding studies demonstrate that permethrin is a weak binder compared with other agents, such as R1888, a nonmetabolizable synthetic androgen. There is insufficient evidence, in vivo, to indicate whether these insecticides act as weak androgens, inhibitors of androgen activity, or a combination of both mechanisms.
SUMMARY AND CONCLUSIONS
In studies conducted in rodents, it has been shown that permethrin is extensively and rapidly metabolized following absorption. The two major pathways for metabolism are hydrolysis, essentially splitting the molecule in two, or oxidation at a number of carbon atoms throughout the molecule. Both of these metabolic processes result in a metabolite that is more water soluble than the parent compound and thus more likely to be excreted in the urine. Thus, one could view metabolism as an important detoxification pathway for permethrin, because it is the parent chemical
that exerts its neurotoxic effects. Given the similarities in xenobiotic metabolism in rodents and primates, metabolism can be assumed to be an important detoxification pathway for humans.
From studies conducted in rodents, permethrin, upon absorption, is found to be distributed throughout the body but appears to concentrate most predominantly in the fat. This observation might explain its high concentrations in brain and nervous tissue. One study has suggested that permethrin in vitro can weakly bind human androgen receptors. The toxicological implications of this binding are uncertain. There are no data on distribution of permethrin in human tissues.
The enhancement of dermal penetration of a variety of chemicals by DEET suggests that use of DEET in combination with permethrin might actually facilitate the dermal absorption of permethrin. Research specifically on the interaction of DEET and permethrin has not been conducted and represents an area of uncertainty regarding the dermal absorption of permethrin. Facilitated absorption of permethrin by DEET is a possible concern for the military personnel assigned to combat areas in which DEET applied to the skin is used in combination with permethrin-treated military uniforms. Dermal absorption might be enhanced on the surfaces of the skin that are not only covered with DEET cream but are also in contact with the military uniforms. Although the potential area of skin at risk is small, the magnitude of the effect is not known with certainty but is assumed to be of minor importance.
RECOMMENDATIONS
The subcommittee recommends that the Army conduct a human pharmacokinetic study with permethrin and DEET. Specifically, this study would involve three groups of volunteers: a group wearing untreated uniforms (control); a group wearing uniforms treated with permethrin (nonfield conditions); and a group wearing uniforms treated with permethrin and using DEET for skin protection (combat conditions). Urine samples would be collected from these individuals over a specified time, and the appearance of the permethrin metabolite CVA could be quantitated by mass spectrometry following the methods of van der Rhee et al. (1989). An increase in CVA in the urine of the DEET-permethrin
group would suggest a potent interactive effect. Similar values of CVA in the urine of the DEET-permethrin and permethrin-only groups would suggest no interactive effect under the Army's intended conditions of use. Military personnel should consider minimizing areas of skin that are covered by both DEET and the permethrin-treated uniforms to reduce potential interactive effects of DEET on permethrin absorption. In other words, the interactive effects could probably be minimized if the areas of the body covered by uniforms were not covered by DEET. DEET should be applied only to areas of skin not covered by uniforms.