3
KEY FACTORS
The panel identified five key factors that influence the international leadership status of US immunology research. These factors are
-
Funding
-
Human Resources
-
Infrastructure
-
Biotechnology and Pharmaceutical Firms
-
Clinical Trials
Each is assessed in more depth below relative to how the United States compares to other countries.
3.1 Funding
Both the US and foreign members of the panel generally agreed that the structure and financial-support mechanisms of the major research institutions in the United States and the structure and mechanisms for provision of research-grant support by government and private granting agencies constitutes a major factor in the success of the US scientific enterprise in immunology and in almost every field of biomedical research.
The reasons have to do with the organization of higher education and research in contrast with the situation in the United States. Many foreign countries, US universities, medical schools, and research institutes are either privately supported or supported by individual state governments–separate administrative units, under the federal system in the United States. Thus, there is a great diversity of private institutions and a great diversity of mechanisms and approaches for funding state-supported institutions and philanthropically supported institutions throughout the United States. Most important the management, regulation, and governance of private institutions is determined by the institutions, and are therefore somewhat removed from the direct effects of federal funding decisions and federal granting agencies.
In contrast, in Europe, Japan, Australia, and several other countries, the central government supports research institutions, universities, and medical schools and allocates research-grant support for specific research projects of specific people. Government regulation of hiring, personnel practices, and many other aspects of operating research laboratories are therefore centrally controlled and do not permit the diversity that is characteristic of the US scientific enterprise. In addition to the diversity of US institutional organization and support, many research programs throughout the United States have enjoyed a greater degree of support from pharmaceutical and biotechnology firms than is true in Europe.
A second major factor in fostering innovation, creativity and rapid development of new
technologies is the National Institutes of Health (NIH) model of research-grant allocation and funding: almost all research (except small projects funded by contracts) is initiated by individual investigators, and the decision as to merit is made by a dual-review system of detailed peer review by experts in each subfield of biomedical science.
In this system, a grant is given to an individual investigator, essentially regardless of the investigator's academic rank or position, as long as he or she is given principal- investigator status by his or her institution. Almost all institutions grant principal- investigator status to scientists at the beginning of their independent careers, almost always after completion of a postdoctoral fellowship. Individual investigators in universities, medical schools, and research institutions are thus empowered to be individual entrepreneurs. They are not subject to any type of review or control of their chosen research subjects by department chairs, other faculty colleagues, or other scientific colleagues in their institutions.
This system has prevented the development of hierarchical research groups of the sort that are seen in many other countries, and it has fostered innovation and independent research initiatives to an amazing degree.
Another major source of funding of immunology (and other biomedical subjects) is the Howard Hughes Medical Institute (HHMI). HHMI selects and retains investigators (rather than projects) largely on the basis of their track record. HHMI-selected investigators are widely regarded as among the most distinguished and productive in the field at both the senior and junior investigator levels. A key to the process has been the selection of external reviewers solely on the basis of their scientific accomplishments and their standing in the field. HHMI provides superb infrastructure for its scientists, who are staff members of HHMI, but whose laboratories are integrated into major academic and research institutions, mainly in the United States. The HHMI scientists are much freer to follow their imaginations and to change the course of their projects than NIH funded investigators, in that the principal evaluation of HHMI investigators is based on productivity, whereas NIH evaluates progress mainly on prescribed projects. The funding of HHMI investigators has substantially enhanced their productivity and has relieved the pressure on NIH to fund meritorious other projects. Additional private sources of immunology funding in the United States are the American Cancer Society, the Juvenile Diabetes Foundation, the Arthritis Foundation, and the Multiple Sclerosis Society.
Funding for training grants for predoctoral fellows and postdoctoral fellows also comes from a wide variety of institutes of NIH and from private sources. Both types of funding have, over the last 40 years, influenced how science is organized in the United States. There are two major results of this entrepreneurial, individual-based system:
-
It has led to the development of multiple centers of excellence in immunology and many other fields of biomedical research at many centers around the country.
-
Many key research centers are based in or closely attached to large medical centers. This stimulates the expansion and application of immunology to many clinical problems and the study of many problems in basic immunology.
Because immunology research in the United States is based largely in medical institutions and because research, training, and clinical activities go on in parallel in these institutions, interdisciplinary research, development of clinical applications, and the application of basic immunology in solving clinical problems have all been fostered.
Further, NIH and several private funding agencies to foster basic scientific training for clinically trained people. Many medical schools have people in their departments of medicine,
pediatrics, and surgery with both a full clinical background and a basic-research background in immunology and related fields.
The current apparent eminence of US-based immunologists should not be taken as leadership in all aspects of training and immunology research, however. As shown in Table 3.1, important research in immunology rewarded by Nobel Prizes has been carried out by 16 laureates, 12 of whom were not US citizens (though some now conduct their research in the United States).
3.2 Human Resources
Its flexibility, diversity, and freedom to originate new approaches has made the United States a very attractive environment for talented researchers from other countries. This has given US research institutions a greater ability than foreign institutions to attract graduate students and postdoctoral fellows from other countries.
The flexibility of funding based primarily on peer review and the merit of applications have made the United States a more attractive country for talented researchers at higher ranks to settle and pursue their research careers. There is a much greater flow of foreign researchers into the United States than the opposite direction because of the lack of barriers (other than language) in the United States.
The US secondary-education system has numerous deficiencies. However, the flexibility allows students particularly talented students to obtain research experience in their own institutions and through summer programs, such as those at the Jackson Laboratories and the Cold Spring Harbor Laboratories. Despite those excellent opportunities at the predoctoral level for a small subset of students, the percentage of doctorate recipients with US citizenship in the combined fields of immunology, microbiology, and virology has decreased from 88.0% in 1980 to 77.9% in 1995. This drop of 11.5% in the proportion of recipients with US citizenship is not as steep as the drop of 22.1% in all the life sciences combined (82.4% in 1980 to 64.2% in 1995). The percentage of foreign doctorate recipients in immunology that were planning to obtain postdoctoral fellowships has increased from 7.0% in 1976-1985 to 13.0% in 1986-19961.
3.3 Infrastructure
The United States has been fortunate in the development of mouse genetics, inbred strains of mice, and many other variations of the basic inbred strains that have been fostered and developed at the Jackson Laboratories. A result has been that a much higher percentage of US immunology research is carried out on the laboratory mouse than was initially true in Europe and Asian countries.
The capital investment by the NIH, the National Science Foundation, and private research-granting agencies in infrastructure, equipment, and buildings for research has been a major source of growth in immunology and many other fields of biomedical research in the United States.
But European countries have proved more adept at large-scale clinical research projects in immunology than the United States, where the great diversity of institutions and institutional support has balkanized the research effort. This works to the detriment of
|
1 |
All data in this paragraph is from special analysis conducted by NRC Office of Scientific and Engineering Personnel of Survey of Earned Doctorates database for this study. |
efficient, large-scale clinical research in the United States, once basic research has led to the development of new therapeutic approaches. The European adeptness is due to many factors.
Table 3.1 Analysis of Nobel Prizes Presented for Immunology Research
|
Prize |
Laureate |
Citizenship |
Research Done In |
Currently |
|
1951 |
Max Theiler |
South Africa |
South Africa |
- |
|
1957 |
Daniel Bovet |
Switzerland |
Switzerland |
- |
|
1960 |
F. Macfarlane Burnet |
Australia |
Australia |
- |
|
Peter Medawar |
Great Britain |
Great Britain |
- |
|
|
1972 |
Rodney R. Porter |
Great Britain |
Great Britain |
- |
|
Gerald M. Edelman |
United States |
United States |
United States |
|
|
1977 |
Rosalyn S. Yalow |
United States |
United States |
United States |
|
1980 |
George D. Snell |
United States |
United States |
- |
|
Jean Dausset |
France |
France |
France |
|
|
Baruj Benecerraf |
United States |
United States |
United States |
|
|
1984 |
Cesar Milstein |
Great Britain |
Great Britain |
- |
|
Georges J.F. Kohler |
Germany |
Switzerland |
- |
|
|
Niels K. Jerne |
Denmark |
Switzerland |
- |
|
|
1987 |
Susumu Tonegawa |
Japan |
Switzerland/United States |
United States |
|
1996 |
Peter C. Doherty |
Australia |
Australia/United States |
United States |
|
Rolf M. Zinkernagel |
Switzerland |
Australia/United States/Switzerland |
Switzerland |
|
|
Source: Analysis conducted by panel members for this report. |
||||
In some cases, it is because of the centralized government control of medical schools and research institutions. In others, it is because physicians are able to maintain a single life–long comprehensive record of patients, which makes it easier to randomize individual patients or practice. Furthermore, in some countries, such as the United Kingdom, clinical-trial methodology has been a special interest of the medical research council and by a national policy that uses randomized trials as a way to introduce new treatment or diagnostic tests.
3.4 Biotechnology and Pharmaceutical Firms
Because of the nature of the venture-capital industry in the United States, the greater flexibility of this industry, and its willingness to fund small biotechnology startup firms, particularly those involved in molecular biology and recombinant-DNA technology, there has been a remarkable growth in biotechnology and a gradual shift of those firms into large pharmaceutical firms. In the last 7 years, although the number of biotechnology companies worldwide has been rather static at approximately 1,275, the amount of money spent on research and development by the industry has almost doubled from $4.9 billion to $9.9 billion (Ernst & Young, 1998a). The result of this phenomenal growth has been the creation of a new source of employment for PhD and MD trainees in immunology, which has attracted many
graduate students into immunology.
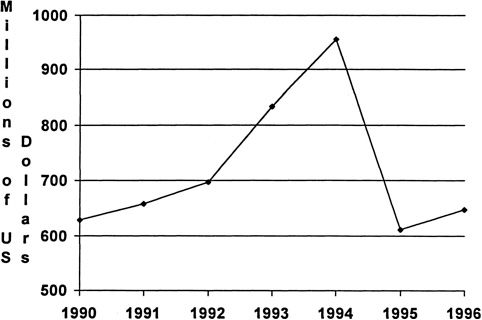
Industrywide data on the amount of money spent on immunology-specific research are not available, so the panel chose to examine trends in industry-supported research for the entire biotechnology industry. Biotechnology industry support for research is much greater in the United States than in Europe as shown in Table 3.2-3.3 (Ernst & Young, 1998b). This financing of research and the use of many academic researchers for consultation in biotech firms and large pharmaceutical firms have provided relatively direct avenues for postdoctoral immunologists to obtain employment, to move across disciplines, and to capitalize rapidly on technology developments that are fostered primarily in biotech firms. In addition, the role of many US academic researchers in founding or participating in the founding of biotechnology firms has enhanced the linkage between academic and industrial research in immunology. In some cases (decidedly a minority), the necessity for patent protection has sometimes impeded the flow of information from research developments in biotechnology and pharmaceutical firms. The ability of biotech firms and large pharmaceutical firms to take discoveries from academic research into startup companies and then large firms and into clinical application has been an overall benefit for the development of clinical immunology in the United States. This entrepreneurial approach has also translated into an economic advantage for the United States over other countries. As shown in Figure 3.1, the United States has a net positive trade balance in biotechnology-based products that was in the low $600 million range in 1990, rose to almost $1 billion in 1994 and then decreased to about $650 million in 1996. (NSF, 1998: Appendix Table 6-6)
3.5 The Clinical Trial
There is a shortage of people in the United States trained to design and administer large-scale trials of new immunology-based therapies. In addition, the impact of managed care has narrowed the patient base available for this type of clinical research, except in large, nonprofit managed-care organizations, such as the Kaiser-Permanente organization.
Table 3.2 Biotechnology Industry Comparable Metrics (Ecu in Millions)
|
European Companies |
|||||
|
Biotech Company |
Market Cap |
Turnover |
Profit/Loss |
R&D Costs |
Employees |
|
British Biotech |
1015 |
29.1 |
-42.6 |
54.2 |
454 |
|
Qiagen |
699 |
68.2 |
8.1 |
7.6 |
650 |
|
Innogenetics |
552 |
25.9 |
-0.9 |
12.6 |
380 |
|
Biocompatibles International |
544 |
16.4 |
-27.6 |
12.2 |
393 |
|
Shire Pharmaceuticals |
531 |
34.4 |
-0.2 |
16.1 |
390 |
|
Cortecs |
412 |
11.5 |
-17.5 |
17.2 |
258 |
|
Genset |
392 |
14.9 |
-17.1 |
23.1 |
355 |
|
Chiroscience |
358 |
17.2 |
-27.9 |
33.2 |
320 |
|
NeuroSearch |
357 |
2.7 |
-8.9 |
10.1 |
110 |
|
Celltech Group |
340 |
6.4 |
-17.9 |
31.2 |
220 |
|
Scotia Holding |
309 |
42.1 |
-30.8 |
34.3 |
420 |
|
Multinational Company |
Market Cap |
Turnover |
Profit/Loss |
R&D Costs |
Employees |
|
Novartis |
99554 |
19590.4 |
3274 |
2320.3 |
87239 |
|
Glaxo Welcome |
76153 |
11915.7 |
4011 |
1714.2 |
52501 |
|
Smithkline Beecham |
52447 |
11639.5 |
2464 |
1255.8 |
55400 |
|
Zeneca |
303312 |
7749 |
1120 |
975.1 |
31100 |
|
Astra |
25425 |
5074.2 |
1153 |
988.3 |
22206 |
|
Baver Group |
24741 |
27912 |
1498 |
2011.7 |
144600 |
|
Hoechst Marion Roussel |
18590 |
7091 |
838 |
1206 |
40500 |
|
US Companies |
|||||
|
Biotech Company |
Market Cap |
Turnover |
Profit/loss |
R&D Costs |
Employees |
|
Amgen |
14144 |
2115.8 |
624.7 |
485.1 |
4646 |
|
Chiron |
3588 |
1206.3 |
50.5 |
340.8 |
7434 |
|
Genentech |
2469 |
888.4 |
108.4 |
432.7 |
3071 |
|
Biogen |
2300 |
254.5 |
37.7 |
121.3 |
675 |
|
Alza |
2264 |
428.1 |
84.5 |
130.5 |
1652 |
|
Genzyme |
1938 |
488.7 |
-67.1 |
194.8 |
3516 |
|
Immunex |
1319 |
140.6 |
-49.6 |
89.1 |
808 |
|
Multinational Company |
Market Cap |
Turnover |
Profit/Loss |
R&D Costs |
Employees |
|
Merck |
114894 |
18216.9 |
3566 |
1366.1 |
49100 |
|
Johnson & Johnson |
78847 |
19531.6 |
2652 |
1750.1 |
89300 |
|
Bristol-Myers Squibb |
74174 |
13840.2 |
2618 |
1172.3 |
51200 |
|
Eli Lilly |
55517 |
6749.7 |
1400 |
1093.3 |
29200 |
|
Pfizer |
35479 |
10386.8 |
1772 |
1547.1 |
46500 |
|
Source: Ernst & Young, 1998a |
|||||
Table 3.3 Entrepreneurial Life Science Highlights (Ecu in Millions)
|
EUROPE |
Public Companies |
Industry total |
||||
|
Current Year |
Prior Year |
Percent Change |
Current Year |
Prior Year |
Percent Change |
|
|
Financial |
||||||
|
Revenues |
648 |
433 |
50% |
2725 |
1721 |
58% |
|
R&D expense |
534 |
243 |
120% |
1910 |
1508 |
27% |
|
Net loss |
347 |
73 |
375% |
2020 |
1113 |
81% |
|
Industry |
||||||
|
Number of Companies |
61 |
49 |
24% |
1036 |
716 |
45% |
|
Employees |
8418 |
5315 |
58% |
39045 |
27500 |
42% |
|
USA |
Public Companies |
Industry total |
||||
|
Current Year |
Prior Year |
Percent Change |
Current Year |
Prior Year |
Percent Change |
|
|
Financial |
||||||
|
Revenues |
12862 |
10565 |
22% |
15985 |
13413 |
19% |
|
R&D expense |
5145 |
4226 |
22% |
8268 |
7258 |
14% |
|
Net loss |
1654 |
2021 |
-18% |
3767 |
4134 |
-9% |
|
Industry |
||||||
|
Number of Companies |
317 |
294 |
8% |
1274 |
1287 |
-1% |
|
Employees |
94000 |
73000 |
29% |
140000 |
118000 |
19% |
|
Ernst & Young, 1998a |
||||||