1
Introduction
Historically, 20 percent of all injured combatants die in battle. Although there has been tremendous variation in this number since World War II (depending on the campaign), this has been the mean mortality rate for all U.S. conflicts combined. A more important statistic, however, is that of every 10 combatants who die from battle injuries, 9 die on the battlefield (i.e., are killed in action) and never make it to the field hospital (Bellamy, 1998). It is also important to understand that, unlike in the civilian setting, there is no ''golden hour'' in combat trauma (Bellamy, 1984, 1987a,b, 1995); that is, whether the casualty receives care within the first hour after the injury is incurred is not a predictor of survival. Of those combatants who die on the battlefield, 40 percent die immediately and are not salvageable, and 25 percent die within 5 minutes and are probably not salvageable. As a role, Bellamy estimates that approximately 3 to 5 percent of those who make it to a higher-echelon point of care will die at a later time (i.e., will die of their wounds) (Bellamy, 1984, 1987a,b, 1995). In addition, those who die at the hospital will die of injuries to the central nervous system, whereas of those who die in the field, the majority will die of exsanguination (see Figure 1-1).
As noted above, of the battlefield casualties who die, 65 percent do so within 5 minutes of their injury (see Figure 1-2) and are not salvageable. The remaining 35 percent are probably at least potentially salvageable, in that 15 percent die up to 30 minutes after their injuries and the remaining 20 percent die after 30 minutes (Bellamy, 1984, 1987a,b, 1995). On the basis of data from the Vietnam conflict (Maughon, 1970), analyzed by Bellamy, almost 50 percent of the battlefield casualties died of exsanguination. Twenty-five percent died of massive mutilating torso injuries, 10 percent died of torso injuries that with timely surgical intervention could have been salvageable, and another 9 percent had peripheral exsanguinating injuries that also could have been salvaged with
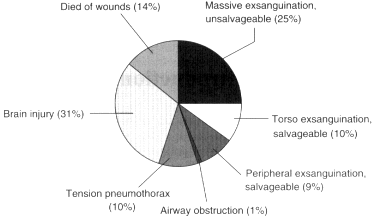
Figure 1-1
Distribution of fatal battlefield injuries in Vietnam.
Source: Adapted from Bellamy (1984, 1987a,b, 1995).
timely intervention. In addition, another 1 percent died of airway obstruction, which was potentially reversible, whereas 10 percent died of tension pneumothorax, which also was potentially reversible. Thirty-one percent died of severe brain injuries and 12 percent died later of their wounds.
In all, Bellamy estimates that just under 20 percent of those who die on the battlefield are potentially salvageable (Bellamy, 1984, 1987a,b, 1995). Bruttig (1998) estimates that approximately 10 to 15 percent of battlefield casualties have potentially surgically correctable injuries. However, because of the delays to definitive surgery, these patients do not survive. Unfortunately, no good comprehensive wound data for the latter group are available, so it is impossible to identify clearly what percentage of these patients are truly potentially salvageable (Bowen and Bellamy, 1998; Koehler et al., 1994). It is clear, however, that the single major cause of death in the potentially salvageable battlefield casualty is hemorrhage.
It may thus be reasonable to expect that with immediate and appropriate care a significant number of these patients could be salvaged. This impression is bolstered by the experience of the Israeli Defense Forces, which have an aggressive system of field treatment by physicians who stabilize the wounded in the battlefield and then rapidly evacuate them to field hospitals. In addition, every other soldier in the Israeli Defense Forces is trained as a medic. It should be pointed out, however, that, in conflicts in which Israel has been involved, evacuation to definitive care required very short flight times (Krausz, 1998).
Still, the battlefield of the 21st century will be very different from that of the past. The new battlefield will be "asymmetric and non-linear" (Bruttig, 1998); the large-scale wars of the past are less likely. Wars will most likely be
fought in settings such as cities and towns, with vaguely identified combatants who will involve large numbers of civilians in their campaigns. The fighting of the future is also likely to involve terrorists and guerrilla interdictions as opposed to highly visible armies, and will be fought by small groups of combatants over shorter time periods with smaller numbers of casualties at any point in time. Because of the likely locations of these conflicts, evacuation by air may be difficult or impossible. As a result, immediate and even ongoing treatment of casualties may be significantly extended. As a consequence, lifesaving medical treatment may well come from a fellow combatant instead of a medic—both of whom are included in the term first responders as used in this report. Thus, the therapies used in the field may vary depending on the time frame from the injury to medical evacuation, the skills and resources of the first responder, and the field site of combatant injury. The availability of fluids of the appropriate volume and physiologic effect in the field may play a part in the provision of lifesaving treatments given the time frame from the injury to medical evacuation and the availability of care after initial resuscitation.
Thus, it is reasonable to conclude that there is a definite subset of battlefield combatants who now die of hemorrhagic shock but who are potentially salvageable with timely battlefield interventions (e.g., fluid resuscitation). As defined by the committee, fluid resuscitation is a treatment regimen involving fluid replacement that is intended to minimize the effects of hemorrhagic shock and to stabilize the hemodynamic response to trauma and hypovolemia. The report focuses on fluid resuscitation of the combat casualty, where a casualty is defined as a combatant who has been physically injured. The committee defines shock as a condition of inadequate tissue perfusion and inadequate removal of cellular waste products, leading to subsequent failure of oxidative metabolism. Shock may result from defects in (1) delivery, (2) transport, or (3) utilization of oxygen, or combinations of all three. Shock is described in greater detail in Chapter 2. To understand the issues involved in saving the lives of combat casualties it is useful to examine the historical developments in fluid resuscitation.
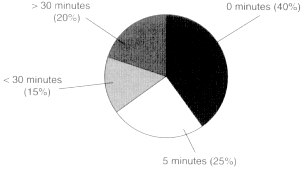
Figure 1-2
Time from injury to death of battlefield casualties.
Source: Adapted from Bellamy (1984, 1987a,b, 1995).
History of Fluid Resuscitation
In recent conflicts, the majority of potentially salvageable fatalities in the battlefield setting involved exsanguinating hemorrhage (Bellamy, 1984, 1987b). Yet therapeutic approaches to the acute care of hemorrhagic shock have not substantially been altered in over 30 years. The committee found that there are at least theoretical disadvantages to existing resuscitation fluids (although these fluids are rarely questioned in clinical practice) and, as with therapeutic approaches, many have not been modified for several decades. In contrast, there have been significant advances in organ preservation resulting from new fluid formulations. Although lactated Ringer's solution has been the most widely used resuscitation fluid for the past 50 years, there remains substantial disagreement among respected researchers and practitioners in the field regarding the relative effectiveness of this and other resuscitation fluids. Part of the difficulty in achieving consensus is that trauma and hemorrhage do not produce standard clinical situations; and the variation in injuries, the duration of periods of hypotension, hypoperfusion, and hypothermia, and the time lapse before definitive care is rendered make it particularly difficult to evaluate treatment protocols. Furthermore, much of the past research has been done with animals whose responses do not wholly parallel those of humans and whose experimental injuries are dissimilar from those incurred in battle; that is, induced hemorrhage in healthy laboratory animals only partially resembles the hemorrhage with additional tissue damage typically observed in human trauma.
Scientists who try to understand the effects of blood loss and fluid replacement have investigated and documented the relative benefits of whole-blood transfusion, plasma, oxygen-carrying fluids, and colloid, crystalloid, and saline solutions at various volumes since the 17th century. To date, there remains considerable debate over the relative effectiveness of colloid versus crystalloid resuscitation fluids as well as hypertonic saline solutions. Even though studies of each approach have been conducted, the history of investigation has been primarily cumulative rather than comparative.
One of the first significant discoveries occurred in 1883, when Sidney Ringer found that hearts perfused with solutions made with tap water functioned longer than those perfused with distilled water solutions. He determined that the calcium in tap water was responsible. Over 40 years later, Krebs created a fluid that mimicked the composition of plasma, and in 1934, Hartmann, working in St. Louis, Missouri, found that children with infantile diarrhea were dying of hyperchloremic acidosis following a significant loss of sodium from the gastrointestinal tract that depleted the serum of sodium. The addition of sodium lactate to the intravenous solution allowed the sodium to bind with the excess chloride after the lactate was metabolized, and with additional modification, it became the lactated Ringer's solution now in widespread use. Nonetheless, occasional concerns have been voiced about the use of lactated Ringer's solution, and recent laboratory studies have suggested potential clinical complications from the administration of high volumes (this is discussed in more depth in Chapter 3).
For obvious reasons, military medicine has focused on hemorrhagic shock and fluid resuscitation for some time. During World War I, clinical observations of soldiers with mild to moderate blood loss were most often made because those suffering from severe hemorrhage usually died before they reached facilities capable of rendering definitive treatment. This experience seemed to support the prevailing concept of secondary shock, or multiple-organ failure following apparently successful resuscitation, observed after an initial period of stabilization. In his 1923 landmark work, Walter Cannon summarized the World War I experience and concluded that shock most often resulted from intravascular volume deficits and must be treated with restoration of blood volume to achieve homeostasis (Cannon, 1923).
Prior to and during World War I, work was also being done with whole blood and blood components, such as plasma, for fluid replacement. Karl Landsteiner described red blood cell isoagglutinins in 1900, thereby facilitating blood typing and cross matching. It was not until World War I that whole blood became available with the addition of citrate as an anticoagulant. Shortly after Landsteiner's discovery British researchers introduced gum acacia and gutta-percha for colloid replacement. In 1916, Bayliss reported the use of 6 percent gum acacia colloid for combat casualties during World War I, thus marking an important moment in what has been an ongoing debate on the use of colloids versus crystalloids. Subsequently, plasma replaced the 6 percent gum acacia as the colloid of choice, but plasma was itself not without drawbacks (see Chapter 3). The use of plasma products was restricted by limited supply, the availability of an adequate preservative solution, and access to refrigerated areas for proper storage (Imm and Carlson, 1993).
Alfred Blalock, Professor of Surgery at the Johns Hopkins University, extensively documented the importance of fluid volume and its effect on survival and was one of the first to note the importance of plasma in resuscitation. In more than 30 papers on shock published prior to World War II, he provided extensive documentation that hypovolemia (reduced blood volume in the body) was the most frequent cause of death and that relative hypovolemia from vasodilation (neurogenic shock) and mediator-caused vasodilation (vasogenic shock) were less frequent causes of death. In 1930, he demonstrated that tissue trauma resulted in the loss of extracellular fluid, which became unavailable to the intravascular compartment, and if the loss was sufficiently severe, the effectiveness of the systemic circulation was impaired (Blalock, 1940).
Blalock was an original member of the National Research Council Committee on Transfusions, which was chaired by Walter Cannon and which advised the armed forces. At its July 24, 1940, meeting the committee recommended the use of plasma, not blood, for resuscitation for four reasons:
|
1. |
Most instances of shock are associated with hemoconcentration (a decrease in the volume of plasma in relation to the number of red blood cells), and a given quantity of plasma is more effective than an equal quantity of whole blood in treatment. |
|
2. |
Blood plasma is approximately as effective as whole blood in the treatment of hemorrhage. |
|
3. |
The difficulties of preservability and transportability of plasma are considerably less than those of whole blood. |
|
4. |
Matching and typing are not necessary when plasma is pooled (National Research Council, Minutes of Transfusion Committee). |
Edward Churchill, in his memoir Surgeon to Soldiers (1972), disagreed with the first two reasons. The first statement about shock associated with hemoconcentration may be traced to a World War I observation by Cannon, who reported higher red blood cell concentrations in capillary blood than in venous blood. This became the centerpiece of the erroneous concept that shock was different from hemorrhage. Churchill further challenged the second reason, that plasma is as effective in treating hemorrhage as blood, because it ignored the advantage of the oxygen-carrying capability of whole blood. The last two reasons for recommending plasma are logistical and are valid to a large degree. Military physicians regarded desiccated plasma favorably because of its small volume, ease of transport, and oncotic effect, and they supported the decision.
Churchill, while Consulting Surgeon to the Eighth Army in Africa, was impressed with the results achieved by British surgeons who used blood for resuscitation. This was accomplished despite rudimentary collecting and storage systems (washed beer bottles). He applied unsuccessfully to the Pentagon for permission to use whole blood, and when he persisted was told to confine his communications to normal channels. Dissatisfied with this response, he contacted a reporter with the New York Times, telling him to report that plasma is not adequate for the treatment of wounded soldiers. The article was printed on August 23, 1943, and Churchill believed that it was critical to receiving authorization and equipment for the administration of whole blood in the Italian campaign.
Meanwhile, others also pursued methods to solve the problems related to the collection and transportation of whole blood. Paul Hawley, the European theater surgeon, pooled type-specific blood in gallon containers in England in 1943, and used this blood for transfusion in France in 1944. In 1944, the U.S. Navy successfully shipped blood by air from the United States to the South Pacific with stops at Pearl Harbor and Guam to change the ice in the containers. Several months later the U.S. Army sent its own shipments of blood from the United States to Europe.
In 1945, Coller and Moyer, of the University of Michigan, described fluid translocations produced by the administration of saline to postoperative patients (Jenkins et al., 1950), and later, G. Tom Shires in Dallas, Texas, documented the necessity of adding crystalloid to whole blood and plasma for successful resuscitation (Roberts et al., 1985; Shires et al., 1960a). This work has remained valid and central to present concepts of volume replacement. For the past 50 years, fluid balance in the treatment of shock and bums and understanding of the physiologic parameters of shock have been at the center of much scientific debate in the treatment of trauma.
On the basis of the work described above and related supporting work, the addition of crystalloids, either as saline or as a balanced salt solution, became the standard of care in the Vietnam conflict and resulted in a significant reduction in the rate of renal failure. Extremely high volumes were used in the care of severe casualties in a naval facility near Danang and highlighted the pulmonary problems in nonthoracic trauma which required prolonged respirator therapy and intensive care stays. The syndrome was popularly referred to as "Danang lung," "shock lung," or "traumatic wet lung" and was later labeled "acute respiratory distress syndrome.'' It had occasionally been noted in World War II and was described under the name of "congestive atelectasis'' by Jenkins and colleagues (1950). Overhydration was the most frequently cited etiology. Although high-volume crystalloid infusion lowered the rate of posttraumatic renal failure, spared the use of blood or blood products from being the more exclusive resuscitation tools, and was successful in resuscitation, some adverse consequences were identified, pertaining to the complexity of distinguishing the process of resuscitation from the delayed effects of the shock situation. For example, high-volume fluid resuscitation in patients with shock can result in cell injury and fluid retention within the cell. Moreover, the capillary leak syndrome has been shown to have consequences from other types of resuscitation (e.g., crystalloid), including albumin, which will cross the cell membrane, and be retained within the cell, increasing wet-lung consequences.
Concern over the frequency and severity of this problem led to a conference conducted by the Committee on Trauma, Division of Medical Sciences, of the National Research Council on February 29, 1968. Numerous theories of the etiology of this problem were discussed, including fat embolism, oxygen toxicity, changes in surfactant, infection, overhydration, and vasoactive agents. A description of a final common pathway emerged: damage to the pulmonary capillaries permitted loss of proteinaceous material into the pulmonary parenchyma. Since then, observations on the etiology and treatment of acute respiratory distress syndrome have been helpful in reducing the rates of morbidity and mortality, but fluid volume remains a key consideration in the development of an effective resuscitation approach.
Although many researchers have compared casualties in the inner city to those that occur on the battlefield, those that occur on the battlefield are very different. The primary difference is that the vast majority of combat injuries are penetrating, whereas those in the civilian sector are blunt. Furthermore, in combat, penetrating wounds are caused not by bullets but by shrapnel from explosive munitions. This is compounded by the fact that evacuation in a combat setting, as opposed to the civilian sector, is not rapid and rarely transports a casualty to a setting where true definitive care can be administered. (It should also be recognized that the injuries in naval operations are still different again, in that the majority of these come from blasts, bums, and inhalation [Bellamy, 1984, 1987a,b, 1995; Ordog et al., 1984].)
Origin, Scope and Organization of the Report
In 1998 the Office of Naval Research requested that the Institute of Medicine appoint a committee that included individuals with expertise in cellular metabolism and biochemistry, emergency medicine, surgery, hematology, anesthesiology, and transfusion medicine to:
- review the state of the art of fluid resuscitation,
- identify targets for therapy, and
- make recommendations for future research directed at acute treatment of massive blood loss on the battlefield.
This report provides an independent assessment of the current status of resuscitation fluid design and resuscitation protocols for combat casualties and a series of findings and recommendations for future research directed at the acute treatment of massive blood loss on the battlefield. Additional attention is given to resuscitation strategies needed in the civilian sector.
The remainder of this report is organized into chapters that address the pathophysiology of acute hemorrhagic shock (Chapter 2), experience with and complications of fluid resuscitation (Chapter 3), novel approaches to the treatment of shock (Chapter 4), protocols of care at the site of injury (Chapter 5), and future directions (Chapter 6). Several appendixes (A to D) are included: Appendix A, acknowledgments; Appendix B, acronyms; Appendix C, the agenda from the committee's conference; and Appendix D, committee and staff biographies.
| This page in the original is blank. |











