C
Federal Spending on HIV/AIDS
OVERVIEW OF FEDERAL HIV/AIDS SPENDING
Since HIV/AIDS was first recognized in the United States in 1981, annual federal spending has grown from an appropriation of several hundred thousand dollars to more than $9.7 billion in fiscal year (FY) 1999. Of this 1999 amount, $6.9 billion (71 percent) was spent on care and assistance for people with HIV/AIDS, $1.9 billion (19 percent) on research, $775.3 million (8 percent) on prevention, and $142 million (2 percent) on international efforts (Figure C.1)1 (Foster et al., 1999).
Spending for prevention efforts, in terms of both absolute spending and percentage increases, has lagged far behind investments in HIV/ AIDS-related care and assistance programs and research. From FY95 through FY99, real spending for care and assistance programs increased by 42 percent, while spending for research increased by 21 percent. In contrast, real spending on prevention remained relatively flat, with only a 9 percent increase during this same period (Figure C.2) (Foster et al., 1999).
Federal spending on HIV/AIDS is enormously complex and divided
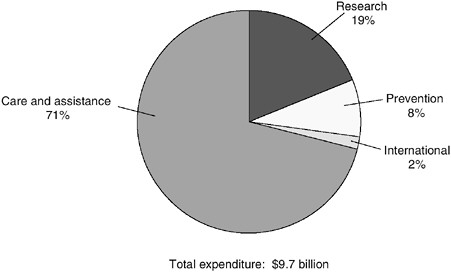
FIGURE C.1 Fiscal Year 1999 federal HIV/AIDS spending.
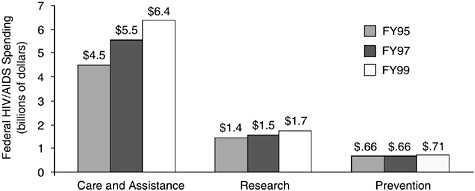
FIGURE C.2 Fiscal Year 1995–1999 domestic federal HIV/AIDS spending (in billions of 1996 dollars). SOURCE: Nominal HIV/AIDS spending data from Foster et al. (1999). Price deflators provided by the Bureau of Labor Statistics.
among many different departments and agencies within the federal government. The Department of Health and Human Services (DHHS) and its respective agencies account for approximately 75 percent of spending on prevention, research, and treatment. Other departments and agencies outside of DHHS with a role in HIV/AIDS prevention, research, and
treatment include: the Social Security Administration ($1.1 billion), the Department of Veterans Affairs ($403 million), the Department of Housing and Urban Development ($225 million), the U.S. Agency for International Development ($135 million), the Department of Defense ($107 million), the Department of Justice ($7 million), the Peace Corps ($5 million), the Department of Labor ($2 million), and the U.S. Information Agency ($0.7 million) (Foster et al., 1999).
OVERVIEW OF DEPARTMENT OF HEALTH AND HUMAN SERVICES SPENDING ON HIV/AIDS
This section describes DHHS’s operating divisions that are involved in primary and secondary HIV prevention and research. It also briefly discusses DHHS agencies whose primary function is care and assistance (e.g., the Health Care Financing Administration and the Health Resources Services Administration), as these agencies have a role in expanding prevention efforts to the clinical setting (see Chapter 4).
The spending figures reported in this section were obtained from a variety of sources, but primarily came from budgets and other documents submitted by the federal agencies. The Committee did not independently verify these figures, nor did we attempt to reconcile budgets across agencies using a common definition of prevention. As a result, there is some inconsistency across agencies with respect to the types of activities that are categorized under the heading of prevention.
Centers for Disease Control and Prevention
The Centers for Disease Control and Prevention (CDC) directs and oversees the nation’s largest and most comprehensive group of federally funded HIV prevention programs. Responsibility for these programs is divided among eight centers, institutes, and offices. HIV prevention efforts are primarily concentrated in two divisions of the National Center for HIV, STD, and TB Prevention: the Division of HIV/AIDS Prevention– Intervention Research and Support and the Division of HIV/AIDS Prevention–Surveillance and Epidemiology.
In FY99, the CDC spent $678 million on HIV-related activities (CDC, 1999b). The CDC’s FY99 HIV/AIDS funding can be divided into the following six major categories (CDC, 1999c):
-
Intervention/Program Implementation: In FY99, the CDC distributed $412 million of its HIV budget externally through cooperative agreements, grants, and contracts, to states and localities for prevention services. Approximately two-thirds of this amount ($258 million) was distributed
-
through external cooperative agreements with 65 state and select local health departments to support prevention activities such as health education and risk reduction programs and counseling and testing services (CDC, 1999c). Priorities for use of these funds are set by Community Planning Groups, comprised of representatives from groups at risk for HIV infection and providers of HIV prevention services. In addition, the CDC supports HIV prevention programs through grants and competitive funding processes for 22 national and regional minority organizations; 10 national business, labor, and faith partnerships; and 94 community-based organizations (CDC, 1999a).
-
Surveillance: The CDC supports a variety of epidemiological and behavioral surveillance activities to monitor HIV/AIDS-related trends, such as HIV and AIDS cases, AIDS deaths, risk behaviors, HIV-related knowledge, and HIV testing behaviors (CDC, 1999a). Approximately $72.3 million (11 percent) of total FY99 program funding was used for surveillance activities (CDC, 1999c).
-
Research: The CDC conducts biomedical and behavioral prevention research for a number of areas including preventing HIV transmission, mechanisms of HIV infection and disease progression, HIV and STD treatment, and microbicides and vaccines (CDC, 1999a). FY99 funding for research activities was $88.1 million (14 percent of total funding) (CDC, 1999c).
-
Technical Assistance: In FY99, approximately $51.7 million (8 percent of total funding) was used for technical assistance, training, capacity building, and information dissemination (CDC, 1999c).
-
Program Evaluation: In FY99, approximately $11.9 million (2 percent of total funding) was used for program evaluation (CDC, 1999c).
-
Policy: In FY99, approximately $1.6 million (<.01 percent of total funding) was used for policy development (CDC, 1999c).
National Institutes of Health
The National Institutes of Health (NIH) is the primary research arm of DHHS. The NIH is comprised of 25 separate institutes and centers, all of which are involved in HIV/AIDS-related research activities. The institutes and centers whose programs are most heavily concerned with HIV/ AIDS include: the National Cancer Institute, the National Institute for Drug Abuse, the National Institute of Mental Health, the National Center for Research Resources, the National Heart, Lung and Blood Institute, the National Institute of Child Health and Human Development, and the National Institute of Allergy and Infectious Diseases (OAR, 1999).
In 1988, NIH established the Office of AIDS Research (OAR) to coordinate its AIDS research and to serve as a focal point for AIDS policy and
budget development. The OAR has established and supports the efforts of six trans-NIH Coordinating Committees in each of the following areas: Natural History and Epidemiology, Etiology and Pathogenesis, Therapeutics, Vaccines, Behavioral and Social Sciences, and Information Dissemination. Prevention studies are found in each of these primary research areas. The NIH and OAR also provide support in training, infrastructure, and capacity building (OAR, 1999).
NIH’s FY99 budget for AIDS-related research was approximately $1.8 billion. The NIH reports that approximately one-third of its AIDS-related research budget supports both nonvaccine-related and vaccine-related prevention research (OAR, 2000a).
Nonvaccine Prevention Research
In FY99, $359 million (20 percent of total funding) supported studies of and interventions for primary prevention, which examined the factors, determinants, and processes of HIV risk and transmission. Approximately $181 million (6 percent of total funding) supported studies of and interventions for secondary prevention of HIV/AIDS, which examined biological and behavioral factors that influence disease progression and the negative psychosocial consequences of HIV/AIDS (OAR, 2000a).
In FY97, NIH began an HIV Prevention Science Initiative to promote comprehensive, crossdisciplinary HIV prevention science. Each year, OAR, with assistance from the Prevention Science Working Group, identifies prevention science priorities and develops a research agenda to address opportunities and gaps in nonvaccine HIV prevention science. Major priority areas of the Initiative from FY97 through FY99 have included the following (OAR, 2000b):
-
FY97 Impact of new therapies on HIV prevention; primary/acute infection; prevention of perinatal transmission; comprehensive HIV prevention strategies for injection drug users; and biobehavioral issues in the development and utilization of HIV prevention methods under female control.
-
FY98 Primary priority: Impact of early identification, counseling, and other behavioral interventions, HIV treatment on risk behaviors, the utilization of HIV prevention services, and the transmission of HIV. Secondary priorities: Comprehensive HIV prevention strategies for substance users; strategies for preventing vertical transmission of HIV; and prevention methods for women.
-
FY99 Primary priority: HIV prevention among racial and ethnic minorities. Secondary priorities: Relationship between biological and behavioral outcomes; sustainability of HIV prevention efforts; international HIV
-
prevention research; legal, ethical, and policy issues in HIV prevention; and primary prevention for men who have sex with women.
Vaccine Research
Approximately 10 percent ($182 million) of NIH’s AIDS-related budget supports basic, preclinical, and clinical research on candidate vaccine products (OAR, 2000a). The NIH also supports the Innovative Vaccine Grants Program, which provides one or two years of funding to investigators to explore new concepts in basic research related to AIDS vaccines. In addition, a cross-institute NIH Vaccine Research Center has been initiated, and which will be funded by intramural programs of the National Institute of Allergy and Infectious Diseases and the National Cancer Institute (NIH, 1999).
Substance Abuse and Mental Health Administration
The Substance Abuse and Mental Health Administration (SAMHSA) is the agency within DHHS with primary responsibility for supporting substance abuse treatment and prevention and mental health services. SAMHSA is comprised of three centers: the Center for Mental Health Services (CMHS), the Center for Substance Abuse Prevention (CSAP), and the Center for Substance Abuse Treatment (CSAT). The Agency’s current HIV/AIDS-related activities include:
-
SAPT Block Grant: The Substance Abuse Prevention and Treatment (SAPT) block grant is the largest program administered by SAMHSA, with approximately $1.6 billion in funding in FY99. Ninety-five percent of the SAPT block grant funds are distributed to states and territories, based on a formula established by Congress, to support substance abuse treatment and prevention services (SAMHSA, 2000b). While Congress provides general direction on how funds can be used, states have broad discretion in allocating these funds. Currently, however, little information exists about how the funds are used by states or about the effectiveness of programs that are funded. Efforts are under way to determine the outcomes of states’ programs (GAO, 2000).
-
SAPT Block Grant-Funded Early Intervention Services (HIV Set-Aside): As part of the ADAMHA Reorganization Act of 1992 (P.L. 102-321), Congress enacted a provision that requires states with an AIDS case rate of 10 or more per 100,000 population to use a portion (2–5 percent) of their SAPT block grant funding for early HIV-intervention services (SAMHSA, 2000b). These prevention activities may include: HIV education and risk reduction, counseling and testing, diagnostic services and assessment,
-
and medical consultation (SAMHSA, 1999). In FY99, 26 states set aside $54 million under this provision for HIV-related prevention efforts (SAMHSA, 2000a). As with the SAPT block grant, however, little information exists about how these funds are used or the effectiveness of programs sponsored by states.
-
Knowledge Development and Application (KDA) Programs: KDA programs are designed to help translate promising evidence-based prevention and treatment interventions from the controlled research environment into community settings. Examples of HIV-related KDA programs and studies include: the HIV Cost Study, the HIV/AIDS High Risk Behavior Prevention/Intervention Model for Youth Adult/Adolescent and Women Program, and the Center for Substance Abuse Prevention’s HIV Prevention Initiative for Youth and Women of Color (SAMHSA, 2000b).2
-
Targeted Capacity Expansion and HIV Outreach Grants: SAMHSA’s Targeted Capacity Expansion (TCE) program provides grants to state and local governments to address emerging and urgent substance abuse treatment and prevention needs of racial and ethnic minorities and other vulnerable populations. In addition, as part of the FY99 Congressional Black Caucus initiative, CSAT began implementing community-based substance abuse and HIV/AIDS outreach program grants, targeting minority communities with high rates of substance abuse and HIV/AIDS. Services funded under this program include outreach, HIV counseling and testing, health education and risk reduction information, access and referrals to testing for sexually transmitted diseases and tuberculosis, substance abuse treatment, primary care, and mental health and medical services. In FY99, CSAT administered $39 million in TCE and outreach grants to enhance and expand substance abuse treatment and services related to HIV/AIDS (SAMHSA, 2000c).
Health Resources and Services Administration
The Health Resources and Services Administration (HRSA) provides health care services to underserved, uninsured, and underinsured communities and individuals. The Agency has lead responsibility for administering the Ryan White CARE Act, which is the largest financial allocation specifically for HIV-related health care and support services. Programs under the CARE Act are managed by HRSA’s Bureau of HIV/ AIDS.
The Ryan White CARE Act
The Ryan White Comprehensive AIDS Resources Emergency (CARE) Act of 1990 (P.L.101-381) was passed in response to the growing AIDS epidemic. The Act was reauthorized in 1996 (P.L. 104-146) and is currently undergoing a subsequent reauthorization. The CARE Act is specifically designed to serve HIV-infected individuals who have fallen through the existing public safety net. The Act is the official “payer of last resort” and can be used only when no other funding source is available to pay for services (HRSA, 2000).
Annual funding for the CARE Act has increased substantially since its inception, from approximately $220 million in FY91 to approximately $1.4 billion in FY99. In FY99, the CARE Act accounted for 19 percent of total federal HIV/AIDS spending. In FY98, the majority of CARE Act funds were used for health care services (30 percent) and medications (34 percent). Twelve percent was used for support services (e.g., transportation, food, and emergency housing assistance), 8 percent for case management, 8 percent for planning and evaluation, and the remainder for other programs (HRSA, 2000).
The CARE Act provides assistance under four program “titles” and through Part F. Titles I–III account for 95 percent of the Act’s FY99 appropriations (HRSA, 2000).
-
Title I: Grants to Eligible Metropolitan Areas. Title I provides emergency relief grants to Eligible Metropolitan Areas (EMAs) that have been disproportionately affected by the HIV epidemic.3 An HIV Health Services Planning Council sets priorities for the allocation of funds within the EMA based on service gaps within their region. In FY99, $505.2 million was provided under Title I grants to 51 EMAs (HRSA, 2000).
-
Title II: Grants to States and Territories. Title II provides formula grants to all 50 states, the District of Columbia, Puerto Rico, Guam, and the U.S. Virgin Islands for health care and support services for people living with HIV disease. Title II also provides access to pharmaceuticals through the AIDS Drug Assistance Program (ADAP). In recent years, an increasing portion of Title II grants has been “earmarked” by Congress to fund medications through ADAP, increasing from $167 million in FY97 to $461 million in FY99. In FY99, $738 million was provided under Title II, with $277 million for grants to states and territories and $461 million for ADAPs (HRSA, 2000).
-
Title III: Early Intervention Grants. Title III provides grants to expand the service capacity of organizations providing primary care services to indigent HIV-positive individuals. Title III programs are funded to provide early intervention services that include: risk reduction counseling; partner involvement in risk reduction; education to prevent transmission; antibody testing; medical evaluation; and clinical care; antiretroviral therapies; protection against opportunistic infections; case management; and interventions to address “co-epidemics,” including tuberculosis and substance abuse. In FY99, $94.3 million was provided in early intervention grants under Title III (HRSA, 2000).
-
Title IV: Women, Infants, Children, and Youth. Title IV provides comprehensive, community-based services to children, youth, and women, who are either at-risk for or living with HIV. Title IV program services include primary and specialty medical care, psychosocial services, logistical support, outreach, and prevention. Title IV systems of care provide access to and linkage with clinical research and trials. In FY99, $46 million was provided to support 58 grants and three initiatives addressing problems in children, adolescents, and women living with HIV (HRSA, 2000).
-
Other Programs (Part F):
-
fourteen AIDS Education Training Centers that offer HIV/ AIDS training and education to clinicians across the country (FY99 funding of $20 million);
-
Special Projects of National Significance, which implement and evaluate models that can be replicated throughout the country for reaching underserved populations and delivering HIV care (FY99 funding of $25 million); and
-
Dental Reimbursement Program, which provides funds to offset the cost of uncompensated HIV care in teaching institutions, to improve access to oral health care, and to help train dental students and residents in caring for persons with HIV (FY99 funding of $7.8 million) (HRSA, 2000).
-
Food and Drug Administration
The Food and Drug Administration (FDA) has several centers and offices that are involved in HIV prevention from a regulatory perspective. The FDA is responsible for ensuring the safety and efficacy of blood and blood products, and is the principal regulatory authority regarding blood and blood products, including blood banking practices, the handling of source plasma, and the manufacture of blood products from plasma. The FDA is also responsible for the regulation and licensing of vaccines to treat or prevent HIV infection. In addition, FDA regulates topical microbicides and drugs to prevent perinatal transmission of HIV. The
FDA plays a primary role in the regulation and development of barrier products, such as condoms and surgical and examination gloves (FDA, 2000). In FY99, FDA spent an estimated $76.7 million on HIV/AIDS related efforts, with $21.8 million devoted to prevention and $54.9 million to research (Foster et al., 1999).
Health Care Financing Administration
The Health Care Financing Administration (HCFA) administers the Medicaid and Medicare programs, both of which are major sources of financing for HIV/AIDS-related medical care and assistance.
Medicaid
Medicaid is a jointly funded, federal–state health insurance program for certain low-income and medically needy individuals. Medicaid is the single largest payer of direct medical services for people living with AIDS, serving over 50 percent of all persons living with AIDS and up to 90 percent of all children with AIDS (Westmoreland, 1999). Individuals with HIV are generally not eligible for Medicaid; however, Maine was granted a demonstration waiver by HCFA in February 2000 to extend Medicaid benefits to nondisabled persons living with HIV disease (Kaiser Family Foundation, 2000). States are required to provide the full range of Medicaid services covered in the state plan to eligible persons with HIV disease, but have the option of providing services such as targeted case management, preventive services, and hospice care. In FY99, combined federal and state Medicaid expenditures were $3.9 billion. Federal Medicaid expenditures, which totaled $2.1 billion, accounted for 22 percent of total federal spending on HIV/AIDS in FY99 (Foster et al., 1999).
Medicare
Medicare is the nation’s largest health insurance program; it covers 39 million persons who are age 65 and over or who have certain disabilities. In FY99, Medicare provided $1.5 billion in care and assistance to individuals with HIV/AIDS (Foster et al., 1999).
Other DHHS Agencies with HIV/AIDS Spending:
-
Indian Health Service: The Agency spent an estimated $3.6 million on HIV/AIDS prevention programs for American Indians and Alaskan Natives in FY994 (Foster et al., 1999).
-
Agency for Health Care Quality and Research: The Agency spent an estimated $1.5 million on research programs for HIV/AIDS (Foster et al., 1999).
REFERENCES
Centers for Disease Control and Prevention (CDC). 1999a. CDC’s HIV/AIDS Prevention Activities. Atlanta: CDC.
Centers for Disease Control and Prevention (CDC). 1999b. Chart: HIV Prevention Funding FY 94–99. Atlanta: CDC.
Centers for Disease Control and Prevention (CDC). 1999c. Provisional Project List by Mission Category for FY 99 CDC HIV/AIDS Budget. Atlanta: CDC.
Food and Drug Administration. 2000. Testimony of Birnkrant D, Egan W, Klein R, and Tabor E, to the Institute of Medicine’s Committee on HIV Prevention Strategies, March 1, 2000, Washington, DC.
Foster S, Gregory A, Niederhausen P, Rapallo D, Westmoreland T. 1999. Federal HIV/AIDS Spending: A Budget Chartbook. Report Prepared for the Kaiser Family Foundation. Washington, DC: Georgetown University Law Center.
Health Resources and Services Administration (HRSA), HIV/AIDS Bureau. 2000. The AIDS Epidemic and the Ryan White CARE Act: Past Progress, Future Challenges. Washington, DC: HRSA.
Kaiser Family Foundation. 2000. Project Description: Medicaid Eligibility Expansion for People with HIV Prior to Disability. Menlo Park, CA: Kaiser Family Foundation.
National Institutes of Health (NIH). 1999. AIDS Research on HIV Prevention: FY 1999 Estimated. Bethesda, MD: NIH.
Office of AIDS Research (OAR), National Institutes of Health. 1999. National Institutes of Health Fiscal Year 2001 Plan for HIV-Related Research. Bethesda, MD: NIH.
Office of AIDS Research (OAR), National Institutes of Health. 2000a. NIH AIDS Research on HIV Prevention: FY99 Estimated. Document prepared for the Committee on HIV Prevention Strategies. (Received in e-mail communication from Judith Auerbach, March 3, 2000.)
Office of AIDS Research (OAR), National Institutes of Health. 2000b. OAR Prevention Science Initiative Awards by Priority Areas (FY97–FY99). Document prepared for the Committee on HIV Prevention Strategies. (Received in e-mail communication from Judith Auerbach, March 3, 2000.)
Substance Abuse and Mental Health Services Administration (SAMHSA). 1999. SAMHSA/ CSAT Fact Sheet on the HIV Set-Aside of the Substance Abuse Prevention and Treatment Block Grant. Washington, DC: SAMHSA.
Substance Abuse and Mental Health Services Administration (SAMHSA). 2000a. “HIV Designated States” Fiscal Year Obligations, Substance Abuse Prevention and Treatment Block Grant. Washington, DC: SAMHSA.
Substance Abuse and Mental Health Services Administration (SAMHSA). 2000b. HIV Strategic Plan. Washington, DC: SAMHSA.
Substance Abuse and Mental Health Services Administration (SAMHSA). 2000c. Targeted Capacity Expansion Program for Substance Abuse Treatment and HIV/AIDS Services. Washington, DC: SAMHSA.
United States General Accounting Office (GAO) 2000. Drug Abuse Treatment: Efforts Under Way to Determine Effectiveness of State Programs (GAO/HEHS-00-50). Washington, DC: GAO.
Westmoreland T. 1999. Medicaid and HIV/AIDS Policy: A Basic Primer. Report Prepared for the Kaiser Family Foundation. Washington, DC: Federal Legislation Clinic, Georgetown University Law Center.











