ACRONYMS AND ABBREVIATIONS
AAVLD
American Association of Veterinary Laboratory Diagnosticians
AFMIC
DoD-Armed Forces Medical Intelligence Center
AIQ
Agricultural inspection and quarantine
APHIS
USDA Animal and Plant Health Inspection Service
AEOC
APHIS Emergency Operations Center
ARS
Agricultural Research Service
AVIC
Area veterinarian in charge
BSE
Bovine spongiform encephalopathy, also mad cow disease
BSL
Biosafety level
BTS
Border and Transportation Security (DHS)
CBP
Bureau of Customs and Border Protection (DHS-BTS)
CADIA
Center for Animal Disease Information and Analysis (CEAH)
CDC
Centers for Disease Control and Prevention
CEAH
Center for Epidemiology and Animal Health (USDA-APHIS-VS)
CFSAN
Center for Food Safety and Applied Nutrition (FDA)
CIA
Central Intelligence Agency
CSREES
USDA- Cooperative State Research, Education, and Extension Service
CVB
Center for Veterinary Biologics (APHIS-VS)
CVM
Center for Veterinary Medicine (FDA)
DHS
U.S. Department of Homeland Security
DHHS
U.S. Department of Health and Human Services
DOC
U.S. Department of Commerce
DOE
U.S. Department of Energy
DoD
U.S. Department of Defense
DOI
U.S. Department of Interior
DOJ
U.S. Department of Justice
DOS
U.S. Department of States
DOT
U.S. Department of Transportation
EPA
U.S. Environmental Protection Agency
EMS
Emergency Management Response
FAD
Foreign animal diseases
FADD
Foreign animal disease diagnostician
FADDL
Foreign Animal Disease Diagnostic Laboratory
FAS
Foreign Agricultural Service (USDA)
FBI
Federal Bureau of Investigation
FDA
Food and Drug Administration
FEMA
Federal Emergency Management Agency
FMD
Foot-and-mouth disease
FSIS
Food Safety and Inspection Service
FWS
U.S. Fish and Wildlife Service
HHS
U.S. Department of Health and Human Services
HSPD
Homeland Security Presidential Directive
HS-Centers
University-Based Homeland Security Centers of Excellence
IAHI
International animal health information
INS
Immigration and Naturalization Service (DOJ)
IS
International Services (USDA-APHIS)
JSA
Joint Subcommittee on Aquaculture
LRN
Laboratory Response Network
MOU
Memorandum of understanding
NAAHC
North American Animal Health Committee
NADC
National Animal Disease Center (USDA-ARS)
NAHEMS
The National Animal Health Emergency Management System
NAHLN
National Animal Health Laboratory Network
NAHMS
National Animal Health Monitoring System
NAHRS
National Animal Health Reporting System
NBII
National Biological Information Infrastructure (USGS)
NBACC
National Biodefense Analysis Countermeasure Center (DHS-S&T)
NCIE
National Center for Import and Export (USDA-APHIS-VS)
NCID
National Center for Infectious Diseases (CDC)
NMFS
National Marine Fisheries Service (DOC-NOAA)
NIAA
National Institute of Animal Agriculture
NIC
National Incidence Coordinator (USDA)
NIMS
National Incident Management System
NOAA
National Oceanic and Atmospheric Administration
NRMT
USDA-APHIS-National Response Management Team
NRP
National Response Plan
NSS
National Surveillance System (USDA-APHIS-VS)
NSU
National Surveillance Unit (USDA-APHIS-VS)
NVSL
National Veterinary Services Laboratories (USDA-APHIS-VS)
NWHC
National Wildlife Health Center (USGS)
NWRC
National Wildlife Research Center (APHIS-VS)
OIE
Office International des Epizooties
OIG
Office of Inspector General
PIADC
Plum Island Animal Disease Center (DHS)
POE
Port(s) of entry
PPQ
Plant Protection and Quarantine (USDA-APHIS)
READEO
Regional Emergency Animal Disease Eradication Organization (APHIS-VS)
S&T
Science and Technology Directorate (DHS)
SITC
Smuggling Interdiction and Trade Compliance
SCO
State Coordinating Officer
TECS
Treasury Enforcement Communications System (U.S. Treasury)
USAHA
U.S. Animal Health Association
USAIP
U.S. Animal Identification Plan
USARMRIID
U.S. Medical Research Institute for Infectious Disease
USDA
U.S. Department of Agriculture
USERPS
U.S. Emergency Response Plan System
USFWS
U.S. Fish and Wildlife Service
USGS
U.S. Geological Survey
USTR
U.S. Trade Representative
VMO
Veterinary medical officer
VS
Veterinary Services (USDA-APHIS)
WDIN
Wildlife Disease Information Node (USGS)
WS
Wildlife Services (USDA-APHIS)
EXISTING FEDERAL SYSTEM FOR ADDRESSING ANIMAL DISEASES
During the past 10 years, emerging and reemerging pathogens have become a major human and animal health concern. The globalization of trade, increased international travel, changing weather patterns, rapid population growth in cities, intensive agriculture, limited genetic diversity in farm animals, and changes in farm practices are creating new opportunities for the reemergence and spread of infectious diseases in both humans and livestock.1 Reservoirs of infection in the wild also pose constant and increasing threats to domestic livestock population.2 Cost of losses from disease in livestock and poultry in the United States is currently estimated at over $17.5 billion dollars per year.3 Further, nearly 200 zoonotic diseases can be naturally transmitted from animals to man.4
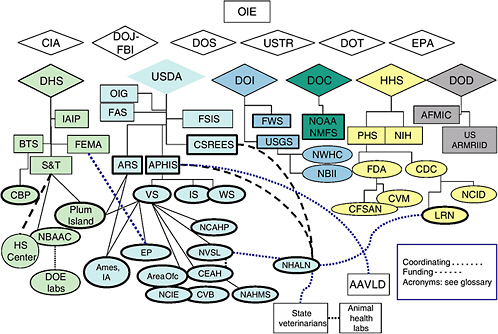
Rapid diagnostic tests, novel genetic vaccines, vigilance in monitoring and surveillance, and increased biosecurity measures will be needed to effectively detect and control emerging diseases and to prevent future animal disease outbreaks. Expanded research will also be needed to accelerate the development of information and technologies for the protection of U.S. livestock, poultry, wildlife and human health against zoonotic diseases. A complex infrastructure for protecting animal health has arisen at the federal level from a number of statutory mandates and regulatory authorities. This infrastructure is based on a large number of programmatic components of several federal agencies. A flow diagram of the existing organizational web of the major agencies involved in efforts to ensure animal health in the United States is presented in Figure C-1.
This paper outlines the existing legal authorities that establish the existing federal infrastructure for addressing animal diseases. Program-
matic activities to carry out the following functions: deterrence and prevention, monitoring and surveillance, detection and diagnosis, research and education, and emergency response and communication, are profiled in details for key federal agencies that are directly involved in the management of farm animal health, disease prevention and monitoring, and response to disease outbreaks. Readily available budget and capacity information is also summarized for several key USDA agencies and DHS. Because of the potential for wildlife to impact farm animals, the programmatic functions performed by federal agencies involved in the management of wildlife diseases are also profiled. Since fisheries are part of the U.S. agricultural system, federal agencies responsible for their health management are also described, albeit in a limited form, in this report. Finally, diseases in farm animals could have direct impact food safety; thus, federal agencies managing food safety are briefly summarized.
1 FARM ANIMALS
1.1 LEGAL FRAMEWORK
The USDA Animal and Plant Health Inspection Service (APHIS) has the lead role in protecting animal health. The Animal Health Protection Act passed into law on May 13, 2002, (Public Law 107-171; Farm Security and Rural Investment Act of 2002, 116 Stat. 134) repealing previous Animal Health and Quarantine laws and providing the Secretary of USDA broad authority and discretion to prevent, detect, control, and eradicate diseases and pests of animals.5 Animal disease means any infectious or noninfectious disease or condition affecting the health of livestock or any condition detrimental to production or marketing of livestock.6 The Secretary of USDA has delegated the responsibility to APHIS.
Within APHIS, the majority of the responsibility to protect animal health resides in Veterinary Services (VS). VS also derives its authorities from the Virus-Serum-Toxin Act of 1913. Recently, the Agricultural Bioterrorism Protection Act added responsibilities for overseeing agents or toxins deemed a severe threat to animal health.7,8 Other USDA agencies, including the USDA Agricultural Research Services (ARS) and the USDA Cooperative State Research, Education, and Extension Service (CSREES), also play critical roles in protecting animal health. The legal and regulatory foundation for APHIS and other USDA agencies that engage in activities to address agricultural animal diseases are summarized in Table C-1.
Interstate and Foreign Quarantine regulations (42 CFR70 and 71) authorize the Secretary of the Department of Health and Human Services (DHHS), through the Centers for Disease Control and Prevention (CDC)
to make and enforce regulations to prevent transmission of infectious disease from foreign countries into the United States. Under these authorities, CDC can set policy to embargo certain animals from entering the United States.9 Title III of the Bioterrorism Act provides the Secretary of the Department of Health and Human Services with new authorities to protect the nation’s food supply. DHHS legal authorities in addressing animal diseases are summarized in Table C-1.
The Public Health Security and Bioterrorism Preparedness Response Act requires notification and controls on the movement of agents or toxins deemed to be a threat to animal or plant health and to animal and plant products. To prevent the incursions of adverse animal health events, USDA-APHIS units are working with DHHS to implement the provisions of this act.23
The Homeland Security Act of 2002 establishes the Department of Homeland Security (DHS) and its directorates. More than 22 federal agencies were consolidated into the new department, including components of the USDA-APHIS that conduct inspection and animal quarantine activities at U.S. ports and Plum Island Animal Disease Center (PIADC).24Table C-2 provides a description of statutory authorities for the various DHS directorates with Homeland Security programmatic responsibilities that are either directly or indirectly addressing animal diseases.
1.2 FUNCTIONS
As authorized by various laws and regulations, the USDA agencies’ programs that address agricultural animal health cover a wide range of functions. In this paper, these functions are grouped as follows: deterrence and prevention, monitoring and surveillance, detection and diagnosis, emergency response, research, education and training, and communication. The programmatic activities that address these functions by various USDA agencies are summarized in Table C-3. Other federal agencies carry out a number of these functions to protect agricultural animal health. In the following sections, existing programs at USDA and other federal agencies performing these functions are described in more detail.
1.2.1 Deterrence and Prevention
Deterrence and prevention are considered the first lines of defense against the introduction of animal and plant pests and pathogens from foreign or domestic sources.35 Several strategies are involved including global and regional strategies that are directed at reducing a potential threat before it reaches the U.S. borders, and border strategy that focuses on interdicting a threat agent at U.S. ports of entry.36
TABLE C-1 Legal Authorities
|
Agency |
Legal and Regulatory Foundation |
Authorities |
|
USDA, Animal and Plant Health Inspection Service (APHIS) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
USDA, Agricultural Research Service (ARS) |
|
|
|
|
|
|
||
|
|
|
|
USDA, Cooperative State Research, Education and Extension Service (CSREES) |
|
|
|
USDA, Food Safety and Inspection Service (FSIS) |
|
|
|
Agency |
Legal and Regulatory Foundation |
Authorities |
|
DHHS-FDA |
|
|
|
DHHS-CDC |
|
|
|
|
|
DHS-FEMA |
|
|
|
DOD |
|
|
|
DHS |
|
|
TABLE C-2 DHS Statutory Authorities and Responsibilities
|
Agency/Directorate |
Statutory Authority25 |
Responsibilities |
|
Bureau of Customs and Border Protection |
Subtitle A, Title IV, of the Homeland Security Act of 2002 (6U.S.C 201 et seq.) establishment of Border and Transportation Security (CBP resides w/in this directorate) |
|
|
Emergency Preparedness and Response Directorate (FEMA) |
Section 502 of the Homeland Security Act of 2002 (6 U.S.C. 312) Manages and coordinates the Federal response to major domestic disasters and emergencies of all types in accordance with the Robert T. Stafford Disaster Relief and Emergency Assistance Act. |
|
|
Science and Technology Directorate |
Title III of the Homeland Security Act of 2002 (6 U.S.C. 181 et seq) |
|
|
radiological nuclear and related weapons and materials and to protect against and respond to terrorist threats.
|
||
|
Information Analysis and Infrastructure Protection Directorate |
Title II of the Homeland Security Act of 2002 (6 U.S.C. 121 et seq) |
|
Table C-3 USDA Agencies and Programmatic Functions Addressing Animal Disease
|
|
Functions: 1) Deterrence & Prevention 2) Monitoring & Surveillance 3) Detection & Diagnosis 4) Education & Research 5) Communication |
||||||
|
Agency |
Description of Programmatic Activities |
1 |
2 |
3 |
4 |
5 |
|
|
APHIS |
Veterinary Service (VS) |
|
X |
X |
X |
X |
X |
|
staff also enlists the help of more than 40,000 federally accredited veterinarians from the private sector who assist with disease exclusion and control.
|
|||||||
|
Wildlife Service (WS) |
|
X |
X |
X |
X |
X |
|
|
International Service (IS) |
|
X |
X |
||||
|
Agricultural Research Service (ARS) |
|
X |
X |
|
Functions: 1) Deterrence & Prevention 2) Monitoring & Surveillance 3) Detection & Diagnosis 4) Education & Research 5) Communication |
|||||||
|
Agency |
Description of Programmatic Activities |
1 |
2 |
3 |
4 |
5 |
|
|
Cooperative State Research, Education, and Extension Service (CSREES) |
|
X |
X |
X |
|||
|
|||||||
|
Food Safety and Inspection Service (FSIS) |
|
X |
X |
X |
X |
X |
1.2.1.1 Border Strategy
On November 21, 2002, President Bush signed legislation creating the Department of Homeland Security (DHS) to unify federal forces and protect the nation from a new host of terrorist threats. Approximately 2,600 employees from APHIS’ Agriculture Quarantine and Inspection (AQI) force became part of DHS’ Border and Transportation Security’s Bureau of Customs and Border Protection (CBP) on March 1, 2003.37 This network of veterinary inspectors and animal health inspectors at all U.S. ports of entry is the first line of defense in identifying materials entering the United States that may be introducing foreign animal diseases. A summary of the programmatic elements and functions of the DHS’ CBP is provided in Table C-4.
Although DHS is now responsible for protecting the nation’s border, APHIS through risk assessment, pathway analysis, and rule making, continues to set agricultural policy, including specific quarantine, testing, and other conditions under which animals, animal products and veterinary biologics can be imported, which is then carried out by DHS.38 At ports of entry, there are also USDA-APHIS-VS port veterinarians who inspect live animals at border ports and animals in quarantine until testing is completed. They are located at 43 VS office areas and report to the veterinarian in charge of the VS-Area Office.39 With agricultural border inspectors now being a part of the DHS, VS has identified the need for developing new protocol for training and interacting with these inspectors and the need to work with DHS to implement improvements recommended in the Animal Health Safeguarding Review regarding exclusion activities in its strategic plan.40
The Foreign Quarantine regulation (42 CFR Part 71.54, Etiologic Agents, Hosts, and Vectors) governs the importation of hazardous materials (etiologic agents, vectors and materials containing etiologic agents.51 CDC has established regulations that govern the importation of dogs, cats, turtles, monkeys, other animals, and animal products capable of causing human disease. Under these regulatory authorities, CDC has established embargo on monkeys and other animals that could carry the monkeypox virus and on birds from specified Southeast Asian countries.52 At present, the CDC-National Center for Infectious Diseases (NCID), Division of Global Migration and Quarantine has quarantine stations in Atlanta, New York, Miami, Chicago, Los Angeles, San Francisco, Seattle, and Honolulu. The quarantine operations involve coordination of numerous agencies, including:53
-
Epidemic Intelligence Service (EIS) and other parts of CDC
-
State and local health departments
-
Customs and Border Protection (DHS)
TABLE C-4 DHS, Border and Transportation Security, Bureau of Custom and Border Protection, and Components Addressing Animal Diseases41–50
|
Agencies |
Function |
|
Border and Transportation Security (BTS) |
|
|
Bureau of Custom and Border Protection (CBP)—(Commissioner Bonner) |
|
|
Agencies |
Function |
|
Office Field Operations (OFO) |
|
|
OFO—Associate Commissioner of Agricultural Inspection Policy & Programs |
|
|
CBP Port Director |
|
|
CBP Ag. Specialist |
|
|
|
|
CBP & FDA |
|
|
National Targeting Center (NTC) |
|
|
CBP Laboratories and Scientific Sciences Division (LLS) |
|
-
U.S. Department of Agriculture
-
U.S. Fish and Wild Life Service
-
The aircraft and maritime industry
The APHIS National Center for Import and Export (NCIE) also works to facilitate international trade, monitors health of animals presented at border, regulates import and export of animals, animal products and biologics, diagnoses foreign and domestic animal diseases. This APHIS center works in partnership with DOI’s Fish and Wildlife Service, APHIS Plant and Protection Quarantine and DHS’s CBP54
1.2.1.2 Offshore Strategy
Offshore activities are designed to mitigate pest and pathogen threats to the United States at points of origin. APHIS’ International Service (IS), through international contacts, gathers and exchanges information on plant and animal health. APHIS-IS cooperates in major surveillance, eradication, and control programs in foreign countries, focusing on nations where economically significant pests or diseases are found. It has implemented the Offshore Pest Information System to monitor and document changes in distribution and outbreak status of specific, designated high risk exotic pest plants and animal diseases, including pathways, in their countries of origin. APHIS-IS currently has 64 foreign service officials stationed in 27 countries on six continents.55 These APHIS personnel are engaged in surveillance and barrier programs, import and export trade facilitation, and commodity preclearance programs.56
USDA-ARS also operates six overseas locations for research on biological control of pests and pathogens. The research contributes to accurate identification of foreign pests and pathogen species; knowledge of basic biology; habitat characterization; assessment of ecological requirements; knowledge of limiting environmental conditions and patterns of occurrence; climate matching; and identification of potential control agents for foreign species. These programs target primarily unintentional threats.57
USDA-APHIS-VS’ Center for Epidemiology and Animal Health (CEAH) is a collaborating center of the Office International des Epizooties (OIE) for Animal Disease Surveillance and Risk Analysis. The OIE serves as the world animal health organization. The VS’ Center for Veterinary Biologics (CVB) and National Veterinary Services Laboratories (NVSL) participate as collaborating centers for the Diagnosis of Animal Diseases and Vaccine Evaluation in the Americas through their involvement in the Institute for International Cooperation in Animal Biologics. The NVSL also serves as an OIE reference laboratory for numerous diseases and pro-
vides training, consultation, and assistance to both domestic and international laboratories. As collaborating centers and reference laboratories, VS provides training, consultation, and other services to OIE members.58,59
1.2.1.3 Early Detection and Intelligence
In recent years, a few USDA staff have been detailed to intelligence and law-enforcement organizations. However, from the review of the these activities, the National Academy of Science has indicated in a 2003 report that it is unclear what information or approaches have been gleaned from these details, or whether findings have been incorporated or used by USDA.60 Recent inquiries also showed that APHIS coordinates with DoD-Armed Forces Medical Intelligence Center (AFMIC) on intelligence information through detailing its intelligence analysts.61 AFMIC is a field production activity of the Defense Intelligence Agency and the sole DoD producer of medical intelligence. The Center provides all source intelligence on worldwide infectious disease and environmental health risks. AFMIC maintains extensive databases; monitors foreign research, development, production, and transnational flow of medical materiel for military interest; and provides intelligence liaison services. APHIS intelligence analysts detailed at AFMIC review AFMIC data and coordinate monthly meetings on domestic and international threats for APHIS.62
The DHS Information Analysis and Infrastructure Protection (IAIP) Directorate gathers and assesses intelligence and information about threats and vulnerabilities from other agencies and takes preventive and protective action. The Department of Commerce’s Critical Infrastructure Assurance Office (CIAO) and the FBI’s National Infrastructure Protection Center were folded into this directorate. Agriculture and food are 2 of 14 critical infrastructure and key assets identified in the President’s National Strategy for Homeland Security and thus fall into the domain of the IAIP Directorate.
Recently issued, the Homeland Security Presidential Directive/HSPD-9 establishes policy to defend the agriculture and food system against terrorist attacks, major disasters, and other emergencies.63 The directive makes the DHS responsible for coordinating federal programs aimed at protecting U.S. agriculture and food from diseases, pests, and toxins. In coordination with the Secretaries of USDA, HHS, and the Administrator of EPA, the Attorney General, the Secretary of DHS, and the Director of CIA are to develop and enhance intelligence operations and analysis capabilities focusing on the agriculture, food, and water sectors (section 9, HSPD-9). The heads of Interior, Agriculture, Health and Human Services, the Administrator of EPA, and other agency heads are responsible for
expanding the current monitoring and surveillance programs (section 8, HSPD-9) to develop:
-
Robust, comprehensive, and fully coordinated surveillance and monitoring systems, including international information, for animal disease, plant disease, wildlife disease, food, public health, and water quality that provides early detection and awareness of disease, pest, or poisonous agents.
-
Tracking system for specific animal, plants, commodities of food.
-
Nationwide laboratory networks for food, veterinary, plant health, and water quality that integrate existing federal and state laboratory resources.
Additionally, the Secretary of DHS is directed to provide a report on options of creating a new biological threat awareness capacity that is based on this upgraded surveillance system to enhance detection and characterization of an attack. This report is not yet available at the preparation of this paper.
1.2.2 Monitoring and Surveillance
APHIS defines monitoring as the routine collection of information for a disease condition, characteristic, or state in an animal population; and surveillance as the analyses of the collected data.64 A surveillance system that provides adequate early information about diseases and other animal health situations is crucial for rapid response. APHIS considers surveillance as the foundation for its Veterinary Services (VS) program activities, which include: domestic disease control and eradication programs, emergency preparedness, response, and trade. The APHIS Animal Health Monitoring Surveillance program is mandated by the Animal Health Protection Act and is conducted through partnerships with states, industry, and other federal agencies.
1.2.2.1 Current Animal Health Surveillance Program
The APHIS-VS Centers for Epidemiology and Animal Health (CEAH) in Fort Collins, Colorado, administers the National Animal Health Monitoring System (NAHMS) and other surveillance efforts. NAHMS collects, analyzes, and disseminates data on animal health, management, and productivity across the United States. APHIS officials collaborate with state and other federal agencies to conduct animal health surveillance activities, including: pre- and post-entry testing of imported animals, sample collection at slaughter, and routine testing of animals for export and interstate movement. APHIS also conducts surveillance for domestic animal
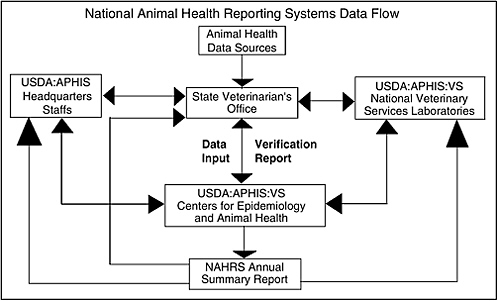
FIGURE C-268
disease eradication programs, such as for brucellosis, tuberculosis, chronic wasting disease, and others.65,66
The National Animal Health Reporting System (NAHRS) is a joint effort of the U.S. Animal Health Association (USAHA), the American Association of Veterinary Laboratory Diagnosticians (AAVLD), and the USDA-APHIS. It is a reporting system designed to collect data on the presence of confirmed Office International des Epizooties (OIE) List A and B clinical diseases in commercial livestock, poultry, and aquaculture species in the United States. Using well defined reporting criteria, the chief animal health official of the state reports OIE List A and B clinical diseases for his or her state. USDA APHIS VS utilizes the data to complete monthly/annual animal disease status reports to OIE and to support trade negotiations. The report utilize multiple sources in reporting to OIE (the United States has been reporting for 25 years).67Figure C-2 describes the NAHRS data flow.
APHIS also conducts surveillance for early identification of foreign animal diseases (FAD). Surveillance is based on clinical symptoms (syndromic). Throughout the United States, APHIS has foreign animal disease diagnosticians (FADD) who are available within a 4-hour drive of any continental U.S. location. This is a network of 3-4,000 accredited veterinarians nationwide and about 400 state veterinarians.69 The FADDs are especially trained to recognize, test, and diagnose FADs at Plum Is-
land. Samples are collected and confirmatory tests are carried out. Tissue samples are sent to the DHS Plum Island Animal Disease Center or to the NVSL in Ames, Iowa, to rule out the presence of a FAD. The total number of FAD investigations in the United States has increased over the last few years. In FY 2002, VS FADDs conducted 837 investigations, an increase from 801 in FY 2001.70 The scenario of a FAD involving live animals entering the United States is not likely, given the safeguards at ports of entry. More likely scenarios are passengers bringing in the virus inadvertently or intentionally.71
1.2.2.2 Animal Health Surveillance Enhancement at USDA
The demands of detecting foreign and emerging animal diseases, monitoring disease trends and threats in the United States and abroad, and providing adequate animal health information to various audiences require a flexible and dynamic surveillance system. Toward this goal, in October 2003, APHIS created the national surveillance unit (NSU) as the operational unit for the development of the National Surveillance System (NSS). It is a unit within the Center for National Surveillance at the VS-CEAH. Also created are several new positions to improve coordination of surveillance activities, including a National Surveillance Coordinator and a FSIS liaison. The main task of the appointed NSS Coordinator is to enhance and integrate national animal health surveillance to implement the enhancements recommended in the National Association of State Departments of Agriculture Animal Health Safeguarding Review and to facilitate effective interaction between APHIS and other federal agencies and stakeholders with surveillance activities.72,73 Additional positions will be created at the NVSL and at the CEAH. In addition, several working groups, including a field implementation team, are being developed or are already in place.74
1.2.2.3 National Animal Identification and Tracking System
In collaboration with industry and stakeholders, VS is developing a National Animal Identification System to meet current and future animal health needs of American agriculture. The National Identification Development Team, a group of over 70 individuals representing over 30 organizations has developed the U.S. Animal Identification Plan (USAIP, see box). During the next 5 years, VS will also implement a new electronic permit system. This system will draw data from numerous sources, such as the ePermits system and Import/Export databases, by identifying key electronic linkages between these sources. This system should improve customer service and allow better tracking of products imported into the United States.75,76
|
The United States Animal Identification Plan (January 2004)77 has evolved from the initial report, the National Identification Work Plan, that was presented at the 2002 USAHA meeting, with the recommendation that the USDA APHIS establish a joint state, federal, and industry group to further advance the work plan. Throughout 2003, approximately 100 animal and livestock industry professionals representing more than 70 associations, organizations, and government agencies, have formed the National Identification Development Team (NIDT) and worked to advance the work plan. This national identification and tracking plan will enhance disease preparedness by rapidly identifying animals exposed to disease, thus allowing quick detection, containment, and elimination of disease threats. When operational, the plan will be capable of tracing an animal or group of animals back to the herd or premises that is the most logical source of a disease concern. The plan’s long term is to establish a system that can complete the traces (back and forward) within 48 hours of discovery of a disease. The USAIP intends to define the standards and framework for implementing and maintaining a national animal identification system for the US. The immediate priority is to have these standards recognized in the Code of Federal Regulations. The cattle, sheep, and swine industries have already developed preliminary implementation plans. All other livestock, including goats, cervids, equine, aquaculture, poultry, llamas, and bison, are becoming engaged in the plan. |
1.2.3 Detection and Diagnosis
Early detection and reporting systems are key elements in a quick response in the event of an animal disease outbreak.78 Much of the nation’s expertise and the laboratories designed to make critical diagnoses of agriculturally important pests and pathogens are in the universities and USDA-Agricultural Research Services (ARS).79
USDA-APHIS-VS’ National Veterinary Services Laboratories (NVSL) are the only federal laboratories dedicated to the testing of diagnostic specimens for domestic and foreign animal diseases. NVSL operates facilities in Ames, Iowa, and has diagnostic capabilities at Plum Island, New York (high security biocontainment FAD Diagnostic Lab, FADDL). On June 1, 2003, the property and facility of Plum Island Animal Disease Center, jointly run by USDA’s APHIS and ARS, were transferred to DHS. NVSL analyzes blood, tissues, and environmental samples to promote disease tracking and identification.80 VS continues to work with manufacturers, the ARS and other research agencies, and animal industry groups to facilitate the licensing of diagnostic tests used in the detection of foreign and emerging animal diseases.81
Although the authority for a federal response to animal diseases in the United States resides with APHIS, as delegated by the Secretary of Agriculture, the initial detection of new pathogen on a farm or ranch, preliminary diagnosis, and development of a program for its control relies heavily on collaborations with other groups, agencies, and individuals. Groups working together to ensure early detection and response to animal disease outbreaks are: USDA- Cooperative State Research, Education, and Extension Service (CSREES), Regional Emergency Animal Disease Eradication Organization (READEO), USDA-VS-NVSL (Ames, Iowa), DHS’s Plum Island (FADDL), academics, professional societies, industry groups, other USDA agencies, other federal agencies, state departments of agriculture, state officials, and international organizations.82,83
In the past, VS has been able to rely on its close programmatic interface and share regulatory responsibilities with the state veterinarians to create consistency and standards in approaches taken to address animal health concerns. VS, however, has a different relationship with state fish and game agencies and with other federal agencies such as the DHS’ Federal Emergency Management Agency (FEMA) and organizations such as the American Association of Veterinary Laboratory Diagnosticians (AAVLD). Additional models of cooperation will need to be developed and refined with these organizations to ensure consistency and define VS’s leadership role.84
1.2.3.1 Laboratory Networks
1.2.3.1.1 National Animal Health Laboratory Network (NAHLN) In June 2002, the Public Health Security and Bioterrorism Preparedness and Response Act of 2002 was signed into law. Section 335 authorizes the Secretary of Agriculture to develop an agricultural early warning surveillance system enhancing the capacity and coordination among state veterinary diagnostic laboratories and federal and state facilities and public health agencies and provides authorization for Congress to appropriate funding to the NAHLN.85 NAHLN addresses diagnostic needs for routine animal disease surveillance as well as diagnostic capacity for investigations and control and eradication programs.86 The overall goal of the NAHLN is to contribute to the improvement of national disease surveillance capabilities. The concept was developed in discussion with NVSL that resulted in an MOU with AAVLD. The initial support was provided by CSREES and APHIS.87
The philosophy behind the design and implementation of NAHLN is that animal disease surveillance functions are most effectively accomplished as a shared responsibility among all animal health agencies. Under the NAHLN concept, state laboratories could provide significant surge capacity during a disease outbreak. The state labs could assist in
defining herds for depopulation, delimiting the extent of the outbreak, and conducting follow-up surveillance to determine “free status.”88 The key goals of the NAHLN are to expand detection and response measures for pathogens that threaten animal agriculture and bolster laboratory capability for select agents with support for personnel, equipment, testing, and training. Among the elements planned for the NAHLN systems are to support the development and deployment of standard diagnostic approaches for identification of select agents, rapid diagnostic techniques, modern equipment, and experienced personnel trained in the detection of emergent, foreign, and bioteror agents; national training; proficiency testing, and quality assurance; and upgraded facilities meeting biocontainment requirements.89 NAHLN is also to bolster data sharing among animal health agencies through the creation of a secure, two-way communications network and the creation of a national repository for animal health data; bolster cooperation and communication among animal health officials through maintenance of confidentiality of source data and providing alerts at appropriate response level.90
The network is currently in a pilot phase and modeled after the comprehensive response network in place for public health threats. The pilot NAHLN involves 12 state/university diagnostic laboratories to develop capacity and surveillance programs for eight high priority foreign animal diseases considered to be a bioterrorist threat (agent for foot-and-mouth disease, hog cholera, African swine fever, rinderpest, contagious bovine pleuropneumonia, lumpy skin disease, highly pathogenic influenza, exotic Newcastle disease). Other agents of interest for potential future inclusion include agents of zoonotic importance like West Nile encephalitis virus, Rift Valley fever, Nipah encephalitis virus, Hendra encephalitis virus, other encephalitides, and bovine spongiform encephalopathy.91 Specifically, VS provided NAHLN laboratories with training in the standard nomenclature to be used in the pilot lab results reporting tool. VS anticipates that training in additional techniques will be offered and the number of NAHLN laboratories will increase significantly by FY 2009, attaining a broader pool of expertise to tap for surge testing capacity in an outbreak.92
1.2.3.1.2 The Laboratory Response Network (LRN) VS and the AAVLD are also partnering with the CDC to enlist state veterinary diagnostic laboratories into the CDC Laboratory Response Network (LRN). VS will serve as the gatekeeper for the veterinary diagnostic laboratory connection to this wider network. Figure C-3 outlines NAHLN structure with linkage to CDC-LRN. NVSL’s Diagnostic Bacteriology Laboratory has received approval from the LRN to conduct diagnostic testing for Clostridium botulinum, Francisella tularensis, and Yersinia pestis. NVSL had previously been
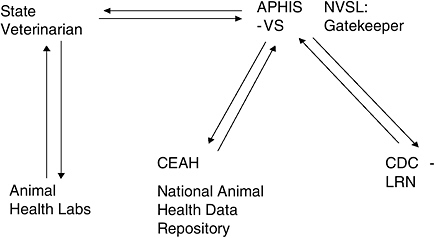
FIGURE C-3 NAHLN Structure.
approved for Bacillus anthracis and Burkholderia spp. The LRN laboratories function as confirmatory laboratories for other diagnostic laboratories and will process overflow samples in the event that a bioterrorist event were to occur.93
LRN was instituted in 1999 in preparation for the U.S. response to bioterrorism. The mission of LRN and its partners is to maintain an integrated national and international network of laboratories that is fully equipped to respond quickly to acts of chemical or biological terrorism, emerging infectious diseases, and public health threats and emergencies.94 CDC runs the LRN program with direction and recommendations provided by the following agencies and organizations:95
-
The Association of Public Health Laboratories
-
The Federal Bureau of Investigation (Department of Justice)
-
The American Association of Veterinary Laboratory Diagnosticians
-
The American Society for Microbiology
-
The EPA
-
USDA
-
DoD
-
FDA
-
DHS
LRN is a consortium of about 120 laboratories, which provide immediate and sustained laboratory testing and communication in the event of public health emergencies, particularly bioterrorism-related events. The network includes the following types of labs:96
-
Federal—labs at CDC, the USDA, the FDA, and other facilities run by the federal agencies
-
State and local public health—these are labs run by state and local departments of health
-
Military—labs operated by the Department of Defense, including the U.S. Army Medical Research Institute for Infectious Diseases (USAMRIID) at Fort Detrick, Maryland
-
Food testing—the LRN includes FDA labs and others that are responsible for ensuring the safety of the food supply
-
Environmental—includes labs that are capable of testing water and other environmental samples
-
Veterinary—some LRN labs, such as those run by USDA, are responsible for animal testing. Some diseases can be shared by humans and animals, and animals often provide the first sign of disease outbreak.
-
International—the LRN has labs located in Canada, the United Kingdom, and Australia
The LRN labs are designated as either national, reference, or sentinel. The structure is as follows:97
-
National labs include CDC and USARMRIID labs with unique resources to handle highly infectious agents and the ability to identify specific agent strains.98
-
Reference labs, sometimes referred to as confirmatory reference, can perform tests to detect and confirm the presence of a threat agent. These labs ensure a timely local response, rather than having to rely on confirmation from labs at CDC, allowing quick local response.
-
Sentinel labs represent the thousands of hospital-based labs that are in the front lines. Their responsibility is to refer a suspicious sample to the right reference lab.
For example, in the case of severe acute respiratory syndrome (SARS), the CDC labs identified the unique DNA sequence of the virus that causes the disease. LRN developed tests and materials needed to support these tests and gave LRN members access to the tests and materials.99
1.2.4 Research, Education and Training
Research and education programs that are needed for managing and recovering from a new pest and pathogen typically involve a land-grant university and ARS programs, state departments of agriculture, state officials, and APHIS.100
1.2.4.1USDA-Cooperative State Research, Education, and Extension Service (CSREES)101
CSREES distributes money to geographically disperse state laboratories to fund facility and equipment upgrades; participates in a nationwide system of agricultural research and education program planning and coordination between state institutions and the USDA; assists in maintaining cooperation among the state institutions, between the state institutions and their federal research partners; administers grants and payments to state institutions to supplement State and local funding for agricultural research and higher educations; funds animal health and disease research by formula to support livestock and poultry disease research in 67 colleges of veterinary medicine and in eligible agricultural experiment stations.
Under the National Initiative competitive grants, CSREES supports research in plants and animals; natural resources and the environment; nutrition; food safety; health; markets, trade, and rural development; and processing for adding value or developing new products.
CSREES’s objective is to support animal health and disease research at eligible schools and colleges of veterinary medicine and state agricultural experiment stations whose purpose is to improve the health and productivity of food animals and horses through effective prevention, control, or treatment of disease, reduction of losses from transportation and other hazards, and protect human health through control of animal diseases transmissible to people.102
Through cooperative efforts with APHIS, CSREES established a unified network of public agricultural institutions to identify and respond to high-risk biological pathogens in the food and agricultural system. This program develops and promotes curricula for higher education programs to support the protection of animals, plants, and public health; support interdisciplinary degree programs that combine training in food and agricultural sciences, medicine, veterinary medicine, epidemiology, microbiology, chemistry, engineering, and mathematics (statistical modeling).103
Under the National Research Competitive Grants Program (NRI), CSREES funds research on key problems relevant to agriculture, food, and the environment on a peer-reviewed, competitive basis. It was established in 1991 in response to recommendations outlined in Investing in Research: A Proposal to Strengthen the Agricultural, Food and Environmental System, a 1989 NRC report.104 Partnering with the university system, CSREES programs support expertise in plant and animal sciences along with extension and outreach capability that can be mobilized to provide an immediate response to critical issues. Program efforts will focus on early intervention strategies to prevent, manage, or eradicate new and emerging plant and animal diseases. Funding also will facilitate improved
diagnostic tests for rapid response to emerging disease agents by expanding the knowledge base of microbial genomics for both animal and plant diseases.105
CSREES provides resources necessary to foster regional and national joint planning, encourage multistate planning and program execution, and minimize duplication of effort. In order to assure responsiveness to emerging issues such as foot-and-mouth disease, E. coli, Salmonella, Listeria, sorghum ergot, potato late blight, etc. CSREES provides funds along with matching funds from the states. It has provided $43 million to states, university, and tribal lands to increase homeland security prevention, detection, and response efforts.106
1.2.4.2 USDA-Agricultural Research Service (ARS)
Over 1,200 USDA-ARS research projects are organized into 22 national programs and three major areas of focus: Animal Production, Product Value and Safety; Natural Resources and Sustainable Agricultural Systems; Crop Production, Product Value, and Safety. Under the Animal focus is the Animal Health Program.107 The mission of ARS-National Animal Health Program is to conduct basic and applied research on selected diseases of economic importance to the U.S. livestock and poultry industries.108 The research is intended to provide scientific information for the control or elimination of animal diseases, optimize animal production systems, and help to ease problems relating to sanitary regulations and food safety disputes.
The ARS-National Animal Health Program, Pathogen Detection and Diagnostics Component is intended to produce a new generation of diagnostic tools that will facilitate detection and identification of known pathogens and diseases, new variants of infectious and noninfectious agents, and emerging organisms and diseases.109 USDA-ARS, National Animal Disease Center (NADC) in Ames, Iowa, is the major federal center for domestic and emerging animal disease research. The NADC opened in 1961 and consists of more than 80 buildings on 318 acres, with an annual budget of $23M, and is staffed with 300 people, including 56 scientists.110 ARS’s other research locations include:111
-
Animal Disease Research Unit, Pullman, Wash.
-
Arthropod-Born Animal Diseases Research Laboratory, Laramie, Wyo.
-
Avian Diseases and Oncology Laboratory, East Lansing, Mich.
-
Beltsville Agricultural Research Center, Beltsville, Md.
-
Plum Island Animal Disease Center, Orient Point, N.Y.
-
Poisonous Plant Research Laboratory, North Logan, Utah
-
Poultry Research Unite, Mississippi State, Miss.
-
Roman L. Hruska U.S. Meat Animal Research Center, Clay Center, Neb.
-
Southeast Poultry Research Laboratory, Athens, Ga.
Most ARS animal health programs are oriented toward understanding the role of individual agents in disease or animal specific metabolic problems. However, many current and future disease problems have a multifactorial etiology, exist in a subclinical or chronic state, and production losses are not always obvious. Research on these complex problems requires multidisciplinary, multivariate dynamic analysis of real life field situations, such as epidemiology. However, there is little epidemiological expertise within ARS research programs. Although epidemiology expertise in APHIS is a valuable resource, it is mostly devoted to disease monitoring and other APHIS programs, not hypothesis driven for research purposes. ARS has indicated in its action plan that its goal is to incorporate hypothesis driven epidemiological analysis into selected current and future ARS research programs and to establish cooperative agreements with the APHIS-CEAH to promote ARS conducting experimental epidemiology research.112
This directorate coordinates DHS efforts in research and development, including preparing and responding to the full range of terrorist threats involving weapons of mass destruction. It conducts research on models, simulations, and tabletop exercises designed to:
-
Explore epidemiological and economic consequences
-
Analyze research and development requirements for foreign animal disease and food security scenarios
-
Develop key technologies and tools to prevent, detect, respond, and recover from intentional and unintentional introduction of biological agents to the national agricultural and food systems
The S&T plans to conduct systems studies to explore the potential utility of technology such as BioWatch for agricultural scenarios. Its strategy is to overlay protection form agricultural terrorism on the existing research and regulatory programs at USDA and FDA. Two of the four high-consequence biological scenarios that constitute the research programs for S&T Biological and Chemical Countermeasures Portfolio address major concerns for agriculture and food, mainly, the deliberate in-
troduction of foot-and-mouth disease into the United States and a classified food security event.
Plum Island Animal Disease Center (PIADC) June 1, 2003, the S&T Directorate assumed responsibility for the operation of PIADC as mandated by the Homeland Security Act of 2002 (PL 107-296 Section 310). PIADC conducts research to protect the health of livestock across America from high-consequence foreign animal diseases such as foot-and-mouth disease. PIADC continues its veterinary sciences and animal health research under the transition:
-
Research on foreign animal diseases that are not present in the United States (current focus on FMD and African swine fever)
-
Developing a joint DHS/USDA comprehensive national strategy for foreign animal disease with emphasis on FMD
-
International collaboration and commercialization of vaccines and diagnostic tools
-
Work on zoonotic agents
-
Biocontainment facilities operate at a biosafety level of 3
DHS and USDA have joint management over PIADC. USDA-ARS and APHIS continues their research and development of diagnostic programs, respectively. USDA research staff remains with USDA while contributing to PIADC’s expanded agroterrorism mission. DHS assumed administration and management responsibilities for the facility.
Office of Research and Development (ORD)-National Biodefense Analysis Countermeasure Center (NBACC) NBACC is dedicated to protect health and agriculture by advancing the scientific community’s knowledge of bioterrorism threats and vulnerabilities. NBACC integrates facilities and technical expertise in biodefense through a hub and spoke structure. The NBACC hub is based on the National Biodefense Campus at Fort Detrick in Maryland. The PIADC is one of the spokes. Additional operations are at the DOE National Laboratories. The Center serves three main functions:
-
Provides scientific assessment of emerging biological threats both to people and to agriculture
-
In partnership with the FBI, establishes new national capability for performing forensic analyses on bioterror events
-
Works with the PIADC to conduct research to help defend against agroterrorism
University-Based Homeland Security Centers of Excellence (HS-Centers)115 In December 2003, the S&T Division released a broad agency announcement calling for proposals that will focus on research effort to combat agroterrorism. DHS intends to establish two HS-Centers by April 2004; one will focus on animal related agroterrorism and the other on postharvest food security.
1.2.4.4 DHHS-NIH
Within HHS, a nontrivial amount of food safety research is funded by the National Institutes of Health (NIH). It is unclear, however, whether this research is coordinated with, or even complements, the research conducted by USDA, the FDA, and the EPA.116
1.2.4.5 DoD-U.S. Army Medical Research, Institute of Infectious Diseases (USAMRIID)
USAMRIID conducts research to develop strategies, products, information, procedures, and training programs for medical defense against biological warfare threats and infectious diseases. USARMRIID, an organization of the U.S. Army Medical Research and Materiel Command (USAMRMC), is the lead medical research laboratory for the U.S. Biological Defense Research Program. The institute plays a key role in national defense and in infectious disease research as the largest biological containment laboratory in the DoD for the study of hazardous diseases. USAMRIID has over 10,000 square feet of Biosafety Level 4 (BSL-4) and 50,000 square fee of BSL-3 lab space. Its 450 civilian staffs include veterinarians. Veterinary medicine is one of the major divisions of the Institute. Its current studies include work on improving vaccines for anthrax, Venezuelan equine encephalitis, plague, and botulism, and on new vaccines for toxins such as staphylococcal enterotoxins and ricin. Research on medical countermeasures to viral hemorrhagic fevers and arboviral illnesses also is conducted. A significant effort is devoted to developing both laboratory and field diagnostic assays for agents considered to be biological warfare or endemic disease threats.117
It has been previously suggested that research related to agricultural bioterrorism be expanded in non-USDA agencies with relevant capabilities or responsibilities, such as the FDA, CDC, USAMRIID, and Defense Advanced Research Projects Agency (DARPA). DARPA does not presently have the legislative authority to support agricultural research.118
1.2.5 Emergency Response and Communication
The Homeland Security Presidential Directive #5 (HSPD-5) enables the Department of Homeland Security to assume responsibility for coordinating federal response operations under certain circumstances. In particular, the DHS secretary will coordinate the federal government’s resources in response to or recovery from terrorist attacks, major disasters, or other emergencies, when any one of the following conditions applies:119
-
The federal department or agency acting under its own authority has requested assistance.
-
The resources of state and local authorities are overwhelmed and federal assistance has been formally requested by states/local authorities.
-
More than one federal agency has become substantially involved in responding to the incident.
-
The secretary has been directed to assume responsibility for managing the domestic incident by the president.
HSPD-5 describes the National Incident Management System (NIMS), which is to cover all incidents, natural or unnatural, for which the federal government deploys emergency response assets. Under this directive, the Secretary of DHS is responsible for leading the development and implementation of NIMS.120
As directed by HSPD-5, APHIS has structured its Emergency Management Response Systems (EMS) systems according to NIMS. APHIS’ EMS is a joint federal-state-industry effort to improve the ability to deal successfully with animal health emergencies, ranging from natural disasters to introductions of foreign animal diseases. The EMS program identifies national infrastructure needs for anticipating, preventing, mitigating, responding to, and recovering from such emergencies. In FY 2003 APHIS established five incident command posts in three states in response to the exotic Newcastle disease (END) outbreak in California, Arizona, Nevada, and Texas.121 Recent APHIS’ efforts to build emergency management capacity have included:122
-
Develop and implement plans for a secure operation in Riverdale, Md.
-
Continually update two Regional Emergency Animal Disease Eradication Organization (READEO) units capable of addressing an animal health emergency and interacting with the APHIS operations center in Riverdale, Md.
-
APHIS, with the NAHEMS Steering Committee, developed strategy for funding the construction of new emergency management
-
biocontainment and laboratory facilities at Plum Island and in Ames, Iowa.
1.2.5.1 Existing Federal Emergency Response Plan
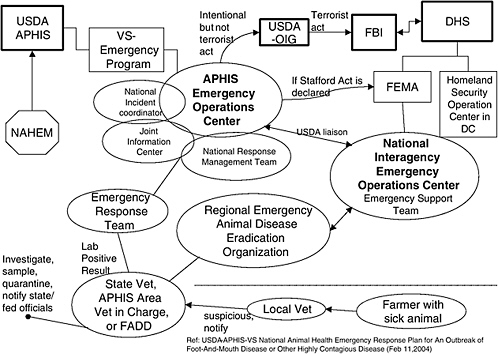
USDA-APHIS-VS—National Animal Health Emergency Response Plan For An Outbreak of Foot-And-Mouth Disease or Other Highly Contagious Animal Diseases123 outlines the national organization and concepts of operation for responding to a widespread highly contagious disease of animals. Much of the information provided in this section came from this plan. Specifically, this plan provides a unified response to all aspects of an FMD outbreak and primarily addresses the coordination and resources that would be required in a multiple-state outbreak. While the plan was written to address a widespread outbreak of FMD, its operating principals would also apply to large-scale outbreaks of other diseases, including, but not limited to, highly pathogenic avian influenza, Newcastle disease, classical swine fever, and African swine fever. Response for an outbreak of highly contagious zoonotic disease (transmitted to people from animals) may be addressed under this plan or may be addressed under contingency plans of the national human health system but will be coordinated under the National Response Plan.124
Depending on the scope of the situation, USDA will ask the Department of Homeland Security’s Federal Emergency Management Agency (FEMA) to coordinate logistical response functions with other federal agencies and state(s) where FMD exists. FEMA would use the framework of the National Response Plan (NRP) in accordance with operational requirements and priorities established by agricultural authorities. (A memorandum of agreement [MOA] will be established between USDA and FEMA to outline respective roles and responsibilities in the event of a major FMD outbreak). The operational structure of the Federal Response Plan (FRP) will be utilized, with or without a presidential emergency or major disaster declaration, to provide an established federal/state coordination mechanism. (Currently an agricultural emergency is not covered under a Stafford Act Declaration.)
When the FMD outbreak or threat is intentional, USDA’s Office of the Inspector General (OIG) would be notified. As warranted by the situation, OIG will then notify and coordinate with the appropriate law enforcement agencies at the local, state, and federal levels. If there is a suspicion that the outbreak was caused by criminal activity, the OIG would work closely with the responding veterinary staff for the proper handling, packaging, and shipment of any samples to the appropriate research laboratory for testing and forensic analysis. OIG would conduct any subsequent criminal investigation. If an FMD outbreak is determined to be a
criminal but not terrorist act, OIG will assume federal lead responsibility for a law enforcement response. If an FMD outbreak is the result of a terrorist act, OIG would notify the Federal Bureau of Investigation (FBI), Weapons of Mass Destruction Unit. OIG and the FBI would jointly conduct a criminal investigation. Well-coordinated interagency mechanisms must be established among the FBI, USDA, and DoD for collaborative forensic investigations.125
1.2.5.2 Federal Response to a FMD outbreak or similarly infectious diseases126
If an FAD passes the first line of defense, responses are conducted through six APHIS-VS national incident management teams (comprising of federal employees) and one incident management team in each VS area office (comprising both federal and state employees). Additional support for the national response would be from the FEMA National Management System, and would be coordinated out of DHS. There is a USDA liaison (at present, the APHIS-VS Associate Deputy Administrator for Emergency Management) to DHS Federal Emergency Management Agency.127
USDA is the lead agency for all agricultural emergencies at the federal level. Within USDA, APHIS is the lead agency for managing an animal disease outbreak such as FMD and within APHIS, the VS-EP (Emergency Program) monitors foreign animal health and maintains surveillance, detection, and diagnosis of outbreaks of exotic diseases in the United States. In order to effectively deal with animal disease emergencies, coordinated response among a number of local producers, veterinarians in private clinical practice, and state-level veterinarians and animal health organizations are required. The APHIS-VS-EP also enlists the help of more than 40,000 federally accredited veterinarians from the private sector who assist with disease exclusion and control.
The initial response to an animal disease outbreak involves the existing USDA expertise, resources, and authorities. The authority for establishing the national strategic response policy for containment and eradication of an FMD outbreak reside in the National Incident Coordinator (NIC) (usually the APHIS-VS- Associate Deputy Administrator for Emergency Management). The USDA-NIC coordinates activities through the APHIS Emergency Operations Center (AEOC) in Riverdale, Maryland. USDA-APHIS Senior Public Affairs Officer establishes a Joint Information Center (JIC), collocated with the AEOC, to serve as the principal source of information and coordinate with other federal agencies, industry communications officials, and state-level JICs.128
APHIS opened the AEOC in March 2003. The center serves as the national command and coordination center for APHIS emergency programs
disaster management. Teams working in the AEOC (the National Response Management Team, NRMT) have enhanced ability to collect, analyze and disseminate information. The NRMT can direct necessary resources and communicate with appropriate stakeholders by coordinating with other federal, state, and international organizations, including the Department of Homeland Security. Communication capabilities include video-teleconferencing, advanced computer interfaces, geographical information system mapping, and a strong multimedia component.129,130,131
At the regional level, in each of the two APHIS-VS regions (Eastern and Western), the APHIS Regional Director manages regional VS resources and communications. APHIS-VS has a disease eradication team made up of APHIS employees. These team members, as an additional duty, train for a specific function of disease response and will deploy individually or as a group in support of the affected state or states based on their needs. The state veterinarian can request that the APHIS area veterinarian in charge (AVIC) deploy this resource.132
In each infected state, the assigned AVIC is the authorized representative of USDA and is the senior federal official who interacts with the state coordinating officer (SCO). The SCO is the designated lead state official and may be the state veterinarian or some other official from either the agricultural or emergency management community. The AVIC leads the overall federal component of the integrated response within the state and establishes operational requirements. The AVIC and the SCO coordinate all activities at the state level.
In the field, the assigned incident commander (IC) will manage all response operations at an infected site. The IC may be a state or federal animal health official as designated by the SCO/AVIC team.
The Stafford Act, DHS-FEMA133 Under the Stafford Act, FEMA serves as the coordinating agency for disaster response and recovery activities. Without a Stafford Act declaration, USDA can request FEMA and other federal agencies to provide support in performing traditional emergency management functions using the framework of the Federal Response Plan. In the event the President declares an emergency or major disaster under the Stafford Act, FEMA assigns a primary federal official (PFO) and the PFO. The PFO, in coordination with the SCO and AVIC, oversees federal response support operations authorized under the presidential emergency or major disaster declaration, and the AVIC continues to represent APHIS for containment and eradication operations. At FEMA headquarters, the FEMA Assistant Director for Readiness, Response, and Recovery provides general direction and oversight for FEMA support of USDA. FEMA activates the interagency emergency support team (EST) located at the National Interagency Emergency Operations Center (NIEOC) and coordi-
nates with the Homeland Security Operations Center in Washington, D.C. To facilitate coordination of animal health and emergency management response functions, a USDA liaison reports to the NIEOC at FEMA headquarters and a FEMA liaison reports to the AEOC.
In the event of an agroterror attack, DHS leads the team of first responders to contain and manage the threat while APHIS provides crucial scientific and diagnostic expertise. APHIS’ expertise is used in managing a potential disease outbreaks as well as in assisting DHS in its investigative and intelligence-gathering efforts to find those responsible for the terrorist attack.134Figure C-4 outlines the infrastructure for a federal response to an FMD outbreak.
Other Federal Agencies Other possible federal agency resources available for response to an FMD outbreak in the United States include:
-
DOT for logistical, quarantine, decontamination, and animal carcass disposal; prevention of vessels with suspected FMD cargoes from entering U.S. waters; provision ships, planes, helicopters, and communication systems.
-
National Communications System (NCS).
-
U.S. Army Corps of Engineers for FMD debris and disposal, decontamination procedures, technical assistance in environmental site assessments, ground water monitoring, soil sampling, contract preparation, and GIS mapping.
-
USDA/U.S. Forest Service can ensure that carcasses are disposed of in accordance with state law or local ordinance; enforcement of federal or state quarantine regulations.
-
American Red Cross can provide hotline for affected farmers to request assistance and for concerned citizens to get information on activities, current conditions, and referrals to other relief agencies; community education to prevent disease spread.
-
General Services Administration (GSA) can provide a number of assists, including:
-
provide contract support for risk assessment; public education; monitoring; surveillance; detection; testing/diagnosis services; epidemiology; biosecurity; appraisal; vaccination; depopulation/disposal; cleaning and disinfections (site/premises); decontamination of equipment; public information and rumor control.
-
GIS mapping services; environmental monitoring and plume projections; feeding and lodging (mass care) of support response personnel; computer equipment, support, and operations in response to federal operations; transportation services for team personnel and
-
-
hazardous waste; public health and safety; mental health counseling; and temporary personnel and services.
-
There exists an MOA between GSA and USDA-APHIS detailing the mechanism for requesting and receiving support in the event that the presence of animal/plant disease and/or pests constitutes an actual or potential emergency situation.
-
DHHS:
-
The FDA can provide food safety assistance, technical assistance, vaccination assistance, veterinary surveillance, carcass disposal assistance, direct animal care and assist in analyzing potential control issues; regulate domestic and imported animal feed products to prevent or curtail FMD; assist in environmental health, disease surveillance, and vector control regarding food and feed.
-
The CDC can assist in disease surveillance, epidemiological and pest management, environmental risk assessment, worker safety issues and the provision of veterinary/medical personnel.
-
The NIH can assist in the assessment of environmental impacts, disease surveillance, vector control issues and the provision of veterinary/medical personnel and has a grant program that can assist laboratories for addressing FMD.
-
-
EPA can provide oversight in the decontamination effort by ensuring chemicals are contained and disposed to prevent environmental contamination; can review decontamination, chemical, and disposal plans; environmental air and water sampling.
-
USDA, Food and Nutrition Service (FNS) educates the public about the safety of the food supply.
-
DOE, through its atmospheric release advisory capability (ARAC), can model the spread of the FMD virus as well as the dispersion of smoke from burning debris.
-
DOC-NOAA can provide weather forecast and observational data upon request; provide on-site meteorologists to support incident command operations; provide dispersion wind forecasts in coordination with other elements of NOAA; broadcast time-critical FMD information on NOAA Weather Radio.
1.2.5.3 Communication
Communication is an important part of an emergency management system. While communication flow through APHIS National Emergency Operation Center at Riverdale for all national coordination during a disease outbreak is well detailed, a written plan for coordination and communication of information that flows from the coordinating bodies at DHS does not appear to exist.135
1.3 BUDGET AND CAPACITY
Budget information and existing capacity at key USDA agencies with programmatic activities that address animal health are summarized in Table C-5. Budget information for the components of DHS with functions that may include functions such as early detection, prevention, and management of incidents involving animal diseases is summarized in Table C-6. APHIS FY 2005 budget for various programs addressing animal diseases reflects request for increase in capacity in the following areas:136
|
Program Areas |
Requested Budget Increase FY 2005$ |
|
|
• |
Animal Health Monitoring and Surveillance increase: to support and enhance BSE surveillance |
8.641M |
|
• |
National Animal Identification program |
33.197M |
|
• |
State Cooperators |
6.1M |
|
• |
Biosurveillance program: to enhance data collection system, improve surveillance capabilities and establish connectivity with the integration and analysis function at DHS. Increase also allow increase in the number of FAD tests at the NVSL and approved state labs |
5M |
|
• |
Low pathogenic avian influenza program: to conduct surveillance and control program |
11.783M |
|
• |
FMD/FAD program: to reduce domestic threats through increase offshore threat assessment, including more officers overseas |
4.229M |
|
• |
Emergency Management Systems program |
10.625M |
|
• |
Veterinary biologics |
1.861M |
|
• |
Veterinary diagnostics program: to enhance the NAHLN and continues its diagnostic work at the FADDL on Plum Island to help protect U.S. herd against potential acts of bioterrorism |
4.347M |
|
• |
Import/Export program: to develop and implement an automated system to track animal and animal product movements |
1.355M |
1.3.1 Laboratory Capacity
Adequate space at the appropriate biosafety level is essential to conducting diagnostic activities. With DoD appropriations, VS began construction in December 2002 on a facility to relocate laboratories from leased space to the main APHIS site at Ames, Iowa. With other appropriate funds, construction began in the fall 2003 for the High Containment (BSL-3AG) Large Animal Housing Facility, associated with infrastructure, and miscellaneous support structures, which are targeted for completion in 2006. In addition, planning/design are well underway for the remainder of the National Cen-
TABLE C-5 Capacity and Level of Funding for USDA Agencies Addressing Animal Diseases
|
Agency |
Overall Capacity and Funding |
Diagnostic, Laboratory Network, Biocontainment Level |
Surveillance and Monitoring |
Deterrence and Prevention |
Risk Analysis |
|
APHIS |
Overall APHIS: • Jan 2002 Defense Appropriation Act: $105M for pest and disease exclusion; $80M for upgrading USDA facilities for operational security • APHIS spent well over $100 million on disease diagnostic and epidemiology and pest detection infrastructure.137 Veterinary Service (VS): • Approximately 1,600 permanent employees • Nationally distributed with field offices in each of the 50 states & major ports of entry. • Operating funds approx $210M (FY’03) VS-CVB operates on a $11M annual budget and has 101 employees, including 48 scientists. The CVB and NVSL shared some personnel.138 |
• The NVSL has a $15 M annual operating budget and 210 employees, of whom 60 are scientists.139 • $20.6 M for national animal and plant diagnostic laboratory networks (2002 Homeland Security Supp.Fund)140 • $14 M for increased security at NVSL in Ames, Iowa, and $23M for Plum Island Animal Disease Center (Jan 2002 Defense Appropriations Act) • Few facilities crucial for accurate diagnoses exist nationwide. Those in existence have limited resources to receive, analyze, and identify many potential agroterrorist agents. The NAHLN remains in pilot stage with laboratories in only 12 states. Further, these labs lack the capacity to test for more than 8 of the 37 FAD agents.141 |
• $138M (Est. FY’04, total APHIS plant & animal health monitoring)144 • $351M (Actual FY’03, total APHIS plant & animal health monitoring)143 • $219M (Est. FY’05, total APHIS plant & animal health monitoring; approx $36M is for BSE)145 |
• $134M (Actual FY’03, pest and disease exclusion)146 • $285M (Est. FY’04, pest and disease exclusion)147 • $315M (Est. FY’05, pest and disease exclusion)148 |
APHIS has a statistically based risk-assessment system to evaluate effectiveness of ongoing inspection operations. |
|
|
|
• $381M (Requested FY2005); of this ○ $178M is for NCAH (Ames, IA). ○ Only $30 M is requested for both plant and animal laboratory upgrades elsewhere. |
|
|
|
|
Agency |
Overall Capacity and Funding |
Diagnostic, Laboratory network, Biocontainment Level |
Surveillance and Monitoring |
Deterrence and Prevention |
Risk analysis |
|
|
|
○ The American Association of Veterinary Laboratory Diagnosticians estimated that at least an additional $85 M above the current funding is required to expand the network142 |
|
|
|
|
APHIS-WS |
• $64M (Estimate FY’03) • $66M (Budget FY’04) |
|
|||
|
Agricultural Research Service (ARS) |
• 100 research locations, a few locations overseas • 2,100 scientists and 6,000 other employees • Protection of livestock from diseases/ pathogens:149 ○ $59M(Actual FY’03) ○ $65M (Est. FY’04) ○ $61M (Est. FY’05) • Defending against catastrophic threat, homeland security:150 ○ $23M (Actual. FY’03) ○ $21M (Est. FY’04) ○ $49M (Est. FY’05) |
|
|||
|
Food Safety and Inspection Service (FSIS) |
• $15M for security upgrades and bioterrorism protection (Jan 2002 Defense Appropriation Act)151 • $754M (Actual FY’03)152 • $775M (Est. FY’04)153 • $839M (Est. FY’04)154 |
FY 2003, FSIS laboratories expanded capability: • To test for nontraditional microbial, chemical, and radiological threat agents. • Increased in surge capacity. • Has more than 7,600 inspectors and |
|
||
|
|
|
veterinarians in more than 6000 Federal meat, poultry, and egg product plants, and at ports of entry, to prevent, detect, and respond to food-related emergencies. • Constructed a Bio-security Level-3 lab • Participated with HHS, EPA, DOE, and states to integrate the nation’s laboratory infrastructure and surge capacity. • Over 60 labs in 27 states and 5 federal agencies have agreed to participate in Food Emergency Response Network (FERN)155 |
|
|
|
|
Cooperative State Research, Education, and Extension Service (CSREES) |
Extension/education/integrated activities—enacted for Homeland Security Mission:156 • $31.6M (FY’03) • $39.2M (FY’04) • $66.3M (FY’05) |
|
Critical plant/ animal issues (funds to develop early intervention strategy to prevent, manage or eradicate new and emerging diseases, both plant and anaimal: • $1M (Actual, FY’03) • $1M (Es. FY’04) • $2M (Est. FY’05) |
||
|
Food safety157 • $15M (Actual FY’03) • $13 (Est. FY’04) • $15M (Est. FY’05) |
|||||
|
Homeland security—Unified network of public agricultural institutions to identify and respond to high risk biological pathogens in the food and agricultural system:158 • $8M (Est. FY’04) • $30M (Est. FY’05) |
|||||
|
The Agricultural Research, Extension, and Education reform Act of 1998 authorized the annual appropriation of $120 M for high priority research159 |
|||||
TABLE C-6 DHS Funding
|
Agency-Focus Area |
Funding160 |
FTEs |
||||||
|
FY 2003 |
FY 2004 |
FY 2005 |
|
|||||
|
DHS |
$31.2B |
|
$36.5B |
|
$40.2B |
|
|
|
|
SECURITY ENFORCEMENT & INVESTIGATIONS |
$21.6B |
|
$22.6B |
|
$24.7B |
|
|
|
|
Bureau of Custom & Border Protection |
|
$5.9 B |
|
$5.9B |
|
$6.2B |
|
41,001 |
|
|
(proposed for Agriculture Quarantine Program) |
|
$407M |
|
||||
|
PREPAREDNESS & RECOVERY |
$5.2B |
|
$5.5B |
|
$7.4B |
|
|
|
|
Emergency Preparedness and Directorate Response (FEMA) |
|
|
|
|
$5,493 M |
|
$7,374M |
2,511 FTEs (full time staff) |
|
|
2,265 Disaster Staff |
|||||||
|
NIMS |
|
$7M |
|
|||||
|
IMT |
$8M |
(20 FTEs) |
||||||
|
Biodefense |
$2,528 M |
|
||||||
|
RESEARCH, DEVELOPMENT, TRAINING, ASSESSMENTS |
$2.3B |
|
$3.6B |
|
$3.8B |
|
|
|
|
Science & Technology Directorate |
|
|
$553M |
|
$913M |
|
$1,039M |
|
|
Plum Island |
|
|
|
$20M |
|
$32.9M |
|
|
|
Biosurveillance |
|
|||||||
|
Initiative |
|
|
|
$53M |
|
$118M |
|
|
|
|
|
$185M |
|
$834M |
|
$864M |
|
|
|
Information Analysis & Infrastructure Protection Directorate |
Vulnerability Risk Assessment |
|
|
|
$710 M |
|
$702M |
|
|
HSOC |
|
|
|
$25M |
|
$35M |
|
|
|
Biosurveillance Initiative |
|
|
|
|
|
$11M |
|
|
ter for Animal Health Modernization Project. This project will bring VS’ NVSL and CVB together in one facility with the ARS’ National Animal Disease Center, which will enhance collaboration. Subject to appropriations, construction will begin on the Low Containment (BSL-2) Large Animal Facility, the Consolidated Laboratory Building, Phase 2, and the balance of the infrastructure in FY2005, with completion of the entire project targeted for 2007.161 When completed the National Centers for Animal Health would include nearly 1 million square feet of space with state-of-the-art capabilities for research and diagnosis.162
A pilot NAHLN involving 12 state/university diagnostic laboratories was funded through USDA in May 2002 for a 2-year period to develop capacity and surveillance programs.163 A two-tiered funding structure is in place for the first 2 years, with tier 1 funded at $2M (laboratories in Calif., Colo., Ga., Tex., Wis.) and tier 2 at $750K (laboratories in Wash., Fla., N.Y., Iowa, Ariz., N.C., La.).164 Though these funds provide critical pilot project start-up costs, they fall far short of developing a true national network that will effectively provide surveillance for zoonotic and foreign animal diseases, bioterrorist agents, and newly emergent diseases like West Nile virus. Federal funding to continue the pilot program and address these deficiencies is critically needed. Both additional start-up costs (estimated at $85M) and continuing funding (est. at $22M annually) have been requested in FY’05 to expand an enhanced, coordinated, and modernized NAHLN. Grants would be awarded directly to and coordinated by an accredited animal disease diagnostic laboratory when such a laboratory exists in the state, or in lieu of an accredited laboratory, to the primary animal disease diagnostic laboratory within the state.165
The NAHLN is in an early development stage so any money received for the state laboratories in FY 2004 will be concentrated on getting the 25 laboratories trained and provided with lab equipment, not adding an additional state. However, future targets reflect an increase of one new state each FY in 2005, FY 2006, and FY 2007.166
Currently, USDA has no BSL-4 facilities. BSL-4 facilities are required for research on pathogens that confer highly contagious, hot diseases, including the animal diseases bovine spongiform encephalopathy, as well as Hendrah and Nipah viruses.167
CDC-National Center for Infectious Diseases (NCID) whose mission is to prevent illness, disability, and death caused by infectious disease in the United States and around the world, conducts surveillance, epidemic investigations, epidemiological and laboratory research, training, and public education programs to develop, evaluate, and promote prevention and control strategies for infectious diseases. In October 2003, the center announced grants to build 11 new biodefense laboratories around the country, including 2 that will be authorized to study the most dangerous
pathogens. Boston University Medical Center and the University of Texas Medical Branch at Galveston each will receive about $120M to build BSL-4 labs. BSL-4 labs can handle pathogens that pose a high risk of causing a life threatening diseases for which there is no vaccine or treatment, such as Ebola virus. In addition, the agency announced grants of $7M and $21M each for nine “Regional Biocontainment Laboratories,” which include BSL-3 and BSL-2 space. Those facilities will be in Colorado State University, Fort Collins; Duke University, Durham, N.C.; Tulane University, New Orleans; the University of Alabama at Birmingham; the University of Chicago; the University of Medicine and Dentistry of New Jersey; the University of Missouri; the University of Pittsburgh; and the University of Tennessee.168
1.3.2 Veterinarian Capacity169
APHIS currently employs 2,053 veterinarians. Over half, 1,059, work for the Food Safety and Inspection Service (FSIS) and 531 work for APHIS. Of the 531 APHIS’ veterinarians, 322 work in the field, a 20 percent decrease from the 404 field veterinarians in 1994. More significant is the comparison between today’s resources and what was available in 1984. In 1984, when there was a large avian influenza outbreak in Pennsylvania, APHIS-VS had nearly 3,000 employees, in contrast to today’s approximately 1,400. In contrast to shrinking resources, demand for investigation of suspected foreign animal diseases has risen from an average of 300 per year during the 1990s to 384 in 2000, 792 in 2001, and 837 in 2002.170 This imbalance between demand and available human resources was characterized in the report on Declining Infrastructure of Governmental Animal Health Professionals Puts American Agriculture at Risk by Dr. Ron DeHaven, as follows:
The current APHIS cadre of veterinarians and animal health professionals is clearly insufficient to handle the increased workload associated with trade obligations, emergencies, and already-apparent future demands…. APHIS faced two extensive outbreaks—END in California and last summer’s avian influenza program in Virginia…. VS has detailed over half of its workforce to California, jeopardizing ongoing programs and leaving the United States vulnerable to any additional disease incursions. It is accurate to say, though very disturbing, that APHIS could not successfully respond to a significant foot and mouth disease (FMD) outbreak and continue to operate the END program in California.
To prepare for shortage of veterinarians, APHIS has developed the National Animal Health Reserve Corps to mobilize close to 300 private veterinarians from around the United States to assist locally during an emergency.171
2 DISEASES IN WILDLIFE
Managing animal health in wildlife is a daunting task. Reservoirs of infection in wild animals offer a constant threat to domestic livestock population and human health. Reducing the risk of transmission and spread may become an even larger factor in the future, with the potential for wildlife-based transmissible encephalopathies.172
2.1 THE DEPARTMENT OF THE INTERIOR, U.S. FISH AND WILDLIFE SERVICE (FWS)
FWS is responsible for the protection of wildlife from environmental hazards, safeguarding habitat for endangered species, and the inspection of wildlife shipments to ensure compliance with laws and treaties and detect illegal trade.173 Generally, all wildlife imported into or exported from the United States for any purposes must be declared to the U.S. Fish and Wildlife Service and cleared prior to release by U.S. Customs and Border Protection. The U.S. Fish and Wildlife Service has a system of ports to allow for the import and export of wildlife, including parts and products.174 Some wildlife inspection requires coordination with USDA APHIS, DOC’s NMFS, INS, FDA and CDC.175
2.2 THE DEPARTMENT OF THE INTERIOR, BUREAU OF U.S. GEOLOGICAL SURVEY (USGS), BIOLOGICAL RESOURCES DIVISION (BRD), THE NATIONAL WILDLIFE HEALTH CENTER (NWHC)
NWHC is one of the 18 science and technology centers in the BRD of the USGS, a bureau of the DOI, located in Madison, Wis. The NWHC was
established in 1975 as a biomedical laboratory dedicated to assessing the impact of disease on wildlife and to identifying the role of various pathogens contributing to wildlife losses.176 The center provides a multi-disciplinary, integrated program of disease diagnosis, field investigation and disease management, research, and training, and maintains extensive databases on disease findings in animals and on wildlife mortality events.
NWHC has over 70 scientists (specialists in such fields as wildlife ecology, epidemiology, veterinary medicine, pathology, virology, bacteriology, parasitology, chemistry, biometry, and population ecology) and support personnel. Center field personnel respond to catastrophic events, such as major die-offs, that threaten the health of wildlife populations. The NWHC has specialized biological containment facilities that allow investigation of infectious diseases affecting a broad spectrum of wildlife, such as amphibians, eagles, sea turtles, sea otters, migratory birds, wolves, large mammals, and other species. National wildlife refuge personnel, law enforcement agents, state conservation agency biologists, university-affiliated scientists, and others send wildlife carcasses and tissue samples to the NWHC for diagnostic examination.177,178,179
The NWHC is an international focal point for research, information, and exchange of information on the study of wildlife health and disease. The center’s researches on zoonotic diseases concentrate on better understanding the ecological relationships among free-ranging wildlife, domestic animals, and public health concerns. This understanding is fundamental for developing effective disease prevention and control strategies. Other center research is directed toward developing enhanced technology for disease detection and diagnosis and toward developing biologics to protect animals against infection.178,179
NWHC is currently monitoring the outbreak in Western Europe and other parts of the world and gathering information from numerous sources. Specialists are interacting with the USDA, and working with the U.S. DOI land management and conservation agencies to provide information on disease status and risks and assist in developing FMD prevention and contingency plans.180 Congress requested that USDA and DOI work together to create a national plan to assist the states and tribes in addressing CWD in both farmed and wild animals. Budget and implementation plan has been developed. Implementation of this plan is proceeding as budgets allow.181
With available specialized containment facilities, NWCH is also providing diagnostic support and research results to federal, state, and local wildlife agencies, as well as public health departments that are utilizing dead wild birds as sentinel, for detecting the West Nile virus (WNV). USGS is working with CDC to learn the current geographic extent of WNV. Scientist at USGS Geographic Science Branch are providing CDC
and public health agencies with real-time geographic information on land-use and land-cover data, roads, and hydrography in areas where the virus is active. These data are used to identity bird and mosquito habitat for placement of mosquito traps.182
2.3 THE DEPARTMENT OF INTERIOR, USGS, NATIONAL BIOLOGICAL INFORMATION INFRASTRUCTURE (NBII) PROGRAMS
The NBII is a broad collaborative program to provide increased access to data and information on the national’s biological resources. This web-based, biological information system draws together vast amounts of scientific data and provides information via the Web. NBII partners include fish and wildlife agencies and including NOAA, EPA, National Science Foundation, Oak Ridge National Laboratory (ONL), USDA-ARS, USDA Forest Service, U.S. Fish and Wildlife Service.183,184
NBII was created in 1993, based on the recommendation of a special panel convened by the National Research Council to examine critical national biological resource issues. In 1998, the need for the NBII was reaf-firmed by a team of internationally renowned scientists who also recommend the creation of NBII “nodes” as focal points for various biological and regional issues. The NBII Program initiated 10 nodes in FY 2001 and began new prototype in FY 20002. The Fisheries and Aquatic Resource node provides access to fisheries information resources from across the world.185 The Wildlife Disease Information Node is to develop collaborative national database of wildlife mortality events to facilitate tracking and study of emerging wildlife diseases such as WNV and CWD.186
2.3.1 THE NBII WILDLIFE DISEASE INFORMATION NODE (WDIN)187
The WDIN provides access to near real-time data on wildlife mortality events and other critical related information. The major objectives of the Wildlife Disease Information Node include:
-
Documenting the prevalence and spread of wildlife diseases at the most discrete spatial and temporal levels possible via a nationwide web-based reporting system.
-
Maintaining current databases on wildlife mortality events and other critical information.
-
Providing Web access to wildlife and zoonotic disease information for management, research, epidemiological, and educational purposes.
-
Providing Web access to the general public for educational purposes and to disseminate information on the importance of wildlife and zoonotic diseases and related ecosystem and community effects.
-
Developing partnerships to share wildlife mortality and other critical information in a distributed fashion and in a secure, partner-based data system.
In FY 2004, the major WDIN undertaking in partnership with NWHC was the development of a robust Web-based CWD national clearinghouse that can accommodate contributed testing results as well as research, monitoring, and surveillance data from state, federal, and tribal agencies as well as other organizations doing CWD work into a common database scheme. Mechanisms by which data can be queried, analyzed, and visualized to make CWD data and information accessible to all parties dealing with CWD issues are being established. Wild and captive cervid data would be included.188
A prototype effort is also being funded by the National Science Foundation and the Intelligence Technology Innovation Center, which is aimed at developing scalable technologies and related standards and protocols needed for a full implementation of a national infectious disease information infrastructure for human, plant, and animal (domestic and wild) diseases. The interdisciplinary team consists of the following groups for this prototype effort:
-
Artificial Intelligence Laboratory at the University of Arizona.
-
The New York State Department of Health and its partner Health Research Inc.
-
The California State Department of Health Services and its partner PHFE Management Solutions.
-
The U.S. Geological Survey’s National Biological Information Infrastructure and the National Wildlife Health Center.
The two diseases selected for this prototype are West Nile virus and botulism because of their significant public health and homeland security implications. After an extensive 4-month research and system development efforts, the University of Arizona completed a research prototype called the WNV-BOT Portal System, which provides integrated, Web-enabled access to a variety of distributed data sources related to WNV and botulism. It also provides advanced information visualization capabilities as well as predictive modeling support.188
2.4 USDA-APHIS-VS WILDLIFE SERVICE (WS)189
WS provides expertise to resolve wildlife conflicts that threaten livestock and protect livestock from wildlife-borne diseases such as CWD. WS works with state counterparts to monitor wildlife diseases like rabies and has cooperative agreements with state wildlife agencies to fund surveillance and management.
The National Wildlife Research Center (NWRC) in Fort Collins, Colo., functions as WS’ research arm. It is the only federal research facility devoted exclusively to resolving conflicts between people and wildlife. WS conducts research projects to study the interaction of deer and cattle, coyotes as reservoirs of tuberculosis, and CWD in elk.
The WS Aquatic Animal Health (AAH) program provides diagnostic assistance to aquaculture producers experiencing health problems with their products. VS works with U.S. Department of the Interior’s U.S. Fish and Wildlife Service (FWS), the U.S. Department of Commerce’s National Marine Fisheries Service, and members of the Joint Subcommittee on Aquaculture to disseminate information and outline agency roles to best meet the aquaculture industry’s animal health needs and develop aquatic animal health monitoring and surveillance program.
3 FISHERIES
3.1 DEPARTMENT OF COMMERCE, NATIONAL OCEANIC AND ATMOSPHERIC ADMINISTRATION, NATIONAL MARINE FISHERIES SERVICE (NMFS)
NOAA oversees fisheries management in the United States and, through the 1946 Agriculture Marketing Act, provides a voluntary inspection service to the industry. The NOAA Seafood Inspection Program offers a variety of professional inspection services, which assure compliance with all applicable food regulations. These services are available nationwide, at all types of establishments such as vessels, processing plants, and retail facilities. All edible product forms ranging from whole fish to formulated products and fishmeal products used for animal foods are eligible for inspection and certification.190
Aquaculture: Global aquaculture now produces more than 31 million metric tons of farm products (fish, crustaceans, and mollusks) annually, which have a value of some $38 billion. The United States is eighth among leading aquaculture producers worldwide, with annual market share value approaching $1 billion.191
Statutory authority: The National Aquaculture Act of 1980, as amended (16 U.S.C. 22801 et seq.) allows the development of a U.S. aquaculture industry. The Act established the Joint Sub-Committee on Aquaculture as coordination group for the federal government activities relating to aquaculture, and charged JSA with development of a National Aquaculture Development Plan. Amendments to the Act in 1985 designated the Secretary of Agriculture as the permanent chair of the JSA. The secretaries of Agriculture, Commerce, and Interior make up the Executive
Committee.192 These agencies are among those listed as resources to aquaculture programs and services within the federal government by the JSA:193
-
U.S. Department of Agriculture: ARS, CSREES, Regional Aquaculture Centers, Farm Service Agencies, AMS, NASS, FAS, APHIS, Federal Crop Insurance Information, National Agriculture Library, Alternative Farming Systems Information Center, Current Research Information System
-
U.S. Department of Commerce: NMFS, National Sea Grant College Program, Economics and Statistic Administration, National Weather Service, National Environmental Satellite Data and Information Service
-
U.S. Department of Interior: U.S. Fish and Wildlife Service, U.S. Geological Survey
-
U.S. DHHS: FDA-CVM, FDA-CFSAN
-
National Science Foundation
-
EPA
-
Tennessee Valley Authority (TVA)
-
U.S. Agency for International Development (USAID)
4 FOOD SAFETY
Four federal agencies share primary responsibility for federal food safety. The largest of these, the USDA-FSIS, regulates meat and poultry through continuous inspection of processing operations and review and approval of product labels. The EPA Office of Pesticide Program (OPP) register pesticides and sets tolerances that are enforced by FDA and FSIS. Finally, the CDC is the federal government’s primary clearinghouse for disease morbidity and mortality surveillance data and its chief resource for epidemiological investigations.194 See Table C-7 for food safety responsibilities for selected food products.
4.1 USDA-FSIS
FSIS inspects most meat, poultry, and processed eggs sold for human consumption for safety, wholesomeness, and proper labeling. The Federal Meat Inspection Act of 1906, as amended [21USC 601 et seq.] requires USDA to inspect all cattle, sheep, swine, goats, and horses brought into any plant to be slaughtered and processed into products for human consumptions. The Egg Products Inspection Act, as amended [21USC 1031 et seq.] is the authority under which FSIS ensure the safety of egg products. For bioterrorism preparedness, FSIS’ Food Biosecurity Action Team (F-BAT) has placed the agency’s 7,600 inspectors on high alert to look for antemortem and postmortem irregularities in meat animals and poultry, and has conducted mock exercises to respond in emergency situations. The Food Threat Preparedness Network (PrepNet) is a joint FSIS/FDA group that works on threat prevention and emergency.195
TABLE C-7 Food Safety Responsibilities for Selected Food Products204
|
Food |
Regulators |
Comments |
|
Eggs |
FDA, AMS, FSIS, APHIS |
FDA has lead jurisdiction over shell eggs; FSIS continuously inspects egg products. AMS operates a voluntary grading program. APHIS monitors animal health. |
|
Meat and poultry |
FSIS, FDA |
FSIS inspects meat during processing. FDA holds regulatory authority once meat leaves the slaughtering or manufacturing plant. |
|
Processed foods |
FDA |
FDA is responsible for most nonmeat products. |
|
Seafood |
FDA, NMFS |
FDA oversees seafood safety generally. NMFS run a voluntary inspection service. |
4.2 FDA
The FDA regulates 80 percent of the nation’s food supply, except for meat, poultry and certain egg products. Through its Center for Food Safety and Applied Nutrition (CFSAN), FDA monitors the safety and labeling of most nonmeat and processed foods and licenses food-use chemicals other than pesticides. CFSAN has the authority to regulate food producers and distributors involved in interstate commerce and to issue recommendations on food safety issues, including foods and cosmetics using bovine ingredients.196 FDA also operates an oversight compliance program for fishery products under which responsibility for the product’s safety, wholesomeness, identity, and economic integrity rests with the processor or importer, who must comply with regulations promulgated under the Federal Food, Drug and cosmetic (FD&C) Act, as amended, and the Fair Packaging and Labeling Act (FPLA). In addition, FDA operates the Low-Acid Canned Food (LACF) Program, which is based on the hazard analysis critical control point (HACCP) concept and is focused on thermally processed, commercially sterile foods, including seafood such as canned tuna and salmon.197
Title III of the Bioterrorism Act provide the HHS Secretary with new authorities to protect the nation’s food supply against the threat of intentional contamination and other food-related emergencies. The interim final rule promulgated under this act requires domestic and foreign facilities that manufacture or process, pack, or hold food for human or animal
consumption in the United State to register with FDA. FDA expects up to 420,000 facilities to register under this requirement.198
The FDA-Center for Veterinary Medicine (FDA-CVM) ensures the safety, efficacy and quality of drugs used in animals, including animal feed and companion animals, animal food and feed, and medical devices used on animals. CVM regulates all animal drugs and feed and works to increase the availability of products to sustain the health, relieve the suffering, and increase the productivity of all farm animals. CVM’s current top three priorities are to prevent BSE, counter the risk of food associated with antibiotic resistance in humans, and ensure safe food derived from genetically modified animals.199
CVM monitors and establishes standards for feed contaminants, approves safe food additives, and manages the FDA’s medicated feed and pet food programs. Office of Surveillance and Compliance monitors marketed animal drugs, food additives, and veterinary devices. Also involved in these activities are the USDA, EPA, and other state and other federal agencies.200
CVM’s specifics activities in a BSE emergency response include201:
-
Collaborating with public health agencies (CDC, HHS, and USDA) and with states, regarding feed contaminant, tissue residue programs, and other monitoring programs for meat and poultry involving a BSE emergency
-
Providing information regarding manufacturer’s GMP compliance and other relevant animal drug quality issues
-
Providing advice in the assessment of animal drugs or feed products involving a BSE emergency
4.3 CDC
In the last decade, the CDC established more than 10 surveillance systems to identify and track the source of outbreaks of foodborne illnesses and to assist regulatory agencies in their food safety activities. Consequently, the agency now has separate surveillance systems to track botulism, Creutzfeldt-Jacob disease (human form of mad cow disease), E. coli O157:H7, Giardia, Salmonella, and Salmonella enteritidis, viral hepatitis, trichinellosis, typhoid fever, and Vibrio infections in foods.202 CDC’s surveillance systems for the most part depend on reporting capabilities of local- and state-level health and agriculture officials. Since 9/11/2001, the agency has been training these officials and laboratory technicians to recognize hazards in foods. It has also begun to refurbish public health laboratories in most states to increase the capacity of these facilities to quickly identify the act of terrorism.203
In addition to the four major organizations, there are a number of other federal agencies with ancillary or supporting roles in the government’s regulatory programs to ensure food safety and counterterrorism efforts. These partners include: the USDA’s Agricultural Marketing Service; the USDA’s Grain Inspection, Packers, and Stockyards Administration (GIPSA); the USDA’s Office of Risk Assessment and Cost-Benefit Analysis; the USDA’s Agricultural Research Service (ARS); the USDA’s Animal and Plant Inspection Service (APHIS); the USDA’s Cooperative State Research, Education, and Extension Service (CSREES); the USDA’s Economic Research Service (ERS); USDA-Foreign Agricultural Service, USDA-ARS, USDA-Food and Nutrition Service, Department of Commerce’s (DOC) National Oceanic and Atmospheric Administration (NOAA), the Treasury Department’s Bureau of Alcohol, Tobacco and Firearms (ATF); Department of State, the Federal Trade Commission (FTC); Custom and Border Protection (CBP) in DHS; Department of Army Veterinary Services Activity, Department of Treasury’s Alcohol and Tobacco Tax and Trade Bureau (TTB), the FBI and CIA.205,206
There is an increased participation by veterinary diagnostic labs in public health programs, as evident in these programs:207 ELEXNET—an integrated web-based data exchange system for food testing information that allows multiple agencies engaged in food activities to compare and communicate and coordinate findings of laboratory analyses;208 FERN (food emergency response network)—to increase surge capacity. FDA and USDA-FSIS are working with CDC to expand the LRN to include a substantial number of counterterrorism laboratories capable of analyzing foods for agents of concern. As of November 2003, there are 63 labs representing 27 states expressing interest in participation in FERN), CELDAR (CA DHS and CAHFS).
5 Endnotes
|
1 |
USDA-ARS Program Summary: Program Rationale. (http://www.ars.usda.gov/research/programs/programs.htm?np_code=103?docid=820; accessed 3/1/04) |
|
2 |
DeHaven, Ron. A Report on “Declining Infrastructure of Governmental Animal Health Professionals Puts American Agriculture at Risk.” August 2003. |
|
3 |
USDA-ARS, Program Summary: Program Rationale. (http://www.ars.usda.gov/research/programs/programs.htm?np_code=103&docid=820; accessed 3/1/04) |
|
4 |
USDA-ARS Program Summary: Program Rationale. (http://www.ars.usda.gov/research/programs/programs.htm?np_code=103?docid=820; accessed 3/1/04) |
|
5 |
USDA-APHIS-VS: ESF 11b. Animal production. National Animal Health Emergency Response Plan for an Outbreak of Foot-And-Mouth Disease or Other Highly Contagious Animal Diseases (February 11, 2004) |
|
6 |
US Congress, 2001, as cited in NAS-NRC Countering Agricultural Bioterrorism, 2003 |
|
7 |
USDA-Veterinary Services Strategic Plan, FY 2004 to FY 2008, Updated February 2004 |
|
8 |
http://www.aphis.usda/gov/lpa/about/strategic_plan/archive/strategic_plan_00-05/strategic_mission.htm; accessed 2/27/04 |
|
9 |
CDC Fact Sheet, NCID, Division of Global Migration and Quarantine, Importation of Pets, Other Animals, and Animal Products into the United States. (www.cdc.gov/ncidod/q/animal.htm; accessed 3/18/04) |
|
10 |
Creekmore,L., USDA, APHIS, VS, NCAHP, Eradication and Surveillance Team, Power Point Slide Presentation. |
|
11 |
Grannis, J., Center for Emerging Issues, USDA-VS, CEAH, Power Point Slide Presentation. |
|
12 |
OMB Budget Documents, Department of Agriculture Part Assessment. (http://www.whitehouse.gov/omb/budget/fy2005/pma/agriculture.pdf; accessed 4/12/04) |
|
13 |
Grannis, J., Center for Emerging Issues, USDA-VS, CEAH, Power Point Slide Presentation. |
|
14 |
USDA-APHIS-VS: ESF 11b Animal Production. National Animal Health Emergency Response Plan for an Outbreak of Foot-And-Mouth Diseases or Other Highly Contagious Animal Diseases (2/11/2004) |
|
15 |
OMB Budget Documents, Department of Agriculture Part Assessment. (http://www.whitehouse.gov/omb/budget/fy2005/pma/agriculture.pdf; accessed 4/12/04) |
|
16 |
ARS, FY2000 and 2001 Annual Performance Plan |
|
17 |
CFDA, 10.207 Animal Health and Disease Research. (http:www.cfda.gov/public/viewprog.asp?progid=38; accessed 4/8/04) |
|
18 |
CRS, Issue brief for Congress, Food Safety and Protection Issues in the 107th Congress, Updated August 28, 2002. No.1B0099. |
|
19 |
Crawford, Lester, Statement before the Committee on Government Affairs, U.S. Senate, November 19, 2003. |
|
20 |
CDC Fact Sheet, NCID-Division of Global Migration and Quarantine (http://wwwcdc.gov/ncidod/dq/mission.htm; accessed 4/6/04) |
|
21 |
CDC Fact Sheet, Office of Health and Safety, Etiologic Agent Import Permit Program. (http://www.cdc.gov/od/ohs/biosfty/imprtper.htm; accessed 4/6/04) |
|
22 |
CDC Fact Sheet, NCID, Division of Global Migration and Quarantine, Importation of Pets, Other Animals, and Animal Products into the United States (http://www.cdc.gov/ncidod/dq/animal.htm; accessed 3/18/04) |
|
23 |
USDA-VS Strategic Plan FY 2004 to FY 2008, Updated February 2004. |
|
24 |
APHIS Fact Sheet, The Animal and Plant Health Inspection Service and Department of Homeland Security: Working Together to Protect Agriculture. May 2003. |
|
25 |
OMB FY2005 Budgets. (http://www.whitehouse.gov/omb/budget/fy2005/pdf/ap_cd_rom/homeland.pdf) |
|
26 |
President George W. Bush, Securing the Homeland, Strengthening the Nation |
|
27 |
Creekmore, L., USDA, APHIS, VS, NCAHP, Eradication and Surveillance Team, Power Point Slide Presentation. |
|
28 |
ARS, FY 2000 and 2001 Annual Performance Plans. |
|
29 |
USDA-Agricultural Research Service, About ARS. (http://www.ars.usda.gov/aboutus/ accessed 2/27/04) |
|
30 |
USDA-ARS, National Programs. (http://www.ars.usda.gov/research/programs.html; accessed 2/27/04) |
|
31 |
USDA, ARS National Program, Program Summary: Program Component Definitions. (http://www.ars.usda.gov/research/programs/program.htm?np_code=303&docid=795; accessed 3/1/04) |
|
32 |
USDA-National Animal Disease Center. (http://www.nadc.ars.usda.gov/about/Mission/bmission.asp; accessed 3/1/04) |
|
33 |
CFDA, 10.207 Animal Health and Disease Research. (http:www.cfda.gov/public/viewprog.asp?progid=38; accessed 4/8/04) |
|
34 |
CSREES, NRI. (http://www.reeusda.gov/nri/nriinfo/about.htm; accessed 4/13/04) |
|
35 |
Dr. J. Annelli, APHIS-VS-EP, Personal Communication, April 2004. |
|
36 |
NAS-NRC, Countering Agricultural Bioterrorism, The National Academies Press, 2003. |
|
37 |
APHIS Fact Sheet, The Animal and Plant Health Inspection Service and Department of Homeland Security: Working Together to Protect Agriculture. May 2003 |
|
38 |
APHIS Fact Sheet, The Animal and Plant Health Inspection Service and Department of Homeland Security: Working Together to Protect Agriculture. May 2003. |
|
39 |
Dr. Annelli, APHIS-VS-EP, Personal Communication, April 2004. |
|
40 |
USDA-VS Strategic Plan FY 2004 to FY 2008 Updated February 2004. |
|
41 |
DHS Organization - Dept Components. (http://www.dhs.gov/dhspublic/display?theme=9&content=2973; accessed 3/1/04) |
|
42 |
Testimony of Commissioner Robert C. Bonner, US CBP Before the National Commission on Terrorist Attacks Upon the US, January 26, 2004. |
|
43 |
FCBF News Flash - CBP Agriculture Specialist Fact Sheet - 9/1/01. (http://www.fcbf.com/NewsFlashDetail.asp?NewsId=47; accessed 4/6/04) |
|
44 |
FCBF News Flash - CBP Agriculture Specialist Fact Sheet - 9/1/01. (http://www.fcbf.com/NewsFlashDetail.asp?NewsId=47; accessed 4/6/04) |
|
45 |
USAHA 2003 Resolution No. 21 from the 2003 USAHA Annual Meeting Oct. 15, 2003. (http://www.usaha.org/resolutions/reso03/res-2103.html; Accessed 4/6/2004) |
|
46 |
CBP today, March 2003. (March/ag.xml” http://www.cbp.gov/xp/CustomsToday/ 2003 March/ag.xml; accessed 3/31/2004) |
|
47 |
US CBP website press release Jan. 14, 2004. (http://www.customs.gov/xp/cgov/newsroom/press_releases/0012004/01142004_4.xml; accessed 4/2/2004 |
|
48 |
No retraining for Agricultural Inspectors in Border Agency Plan. (10/30/2003) (http://www.fass.org/fasstrack/news_item.asp?news_id=1646; accessed 4/6/2004) |
|
49 |
Dr. Ahmad, Veterinarian in Charge at JFK (718-553-1727). Telephone communication, (4/6/05). |
|
50 |
DHS Protecting Against Agricultural Terrorism, (http://www.dhs.gov/dhspublic/display?theme=43&content=3117; accessed 4/2/04) |
|
51 |
CDC Fact Sheet, Office of Health and Safety, Etiologic Agent Import Permit Program. http://www.cdc.gov/od/ohs/biosfty/imprtper.htm; accessed 4/6/04) |
|
52 |
CDC Fact Sheet, NCID, Division of Global Migration and Quarantine, Importation of Pets, Other Animals, and Animal Products into the United States. (http://www.cdc.gov/ncidod/dq/animal.htm; accessed 3/18/04) |
|
53 |
CDC Fact Sheet, NCID, Division of Global Migration and Quarantine, History of Quarantine. (http://www.cdc.gov/ncidod/dq/history.htm) |
|
54 |
NCIE Fact Sheet. (www.aphis.usda.gov/vs/ncie/; accessed 3/1/04) |
|
55 |
Statement of Dr. Charles Lambert, Senate Committee on Government Affairs. “Agroterrorism: The Threat to America’s Breakfast.” November 19, 2003. (http:/govt-aff.senate.gov/index.cfm?Fuseaction=Hearings.Testimony&HearingID=127&WitnessID=482; accessed 4/2/2004) |
|
56 |
NAS-NRC, Countering Agricultural Bioterrorism, National Academy Press, 2003. |
|
57 |
NAS-NRC, Countering Agricultural Bioterrorism, National Academy Press, 2003 |
|
58 |
USDA VS Strategic Plan FY 2004 to FY 2008, updated February 2004 |
|
59 |
Accord, Bobby, Statement before the House Subcommittee on Agriculture, Rural Development, Food and Drug Administration, and Related Agencies. |
|
60 |
NAS-NRC, Countering Agricultural Bioterrorism, The National Academies Press, 2003. |
|
61 |
Dr. J. Annelli, APHIS-VS-EP, Personal Communication, April 2004. |
|
62 |
Dr. J. Annelli, APHIS-VS-EP, Personal Communication, April 2004. |
|
63 |
HSPD9, January 30, 2004. (http://www.whitehouse.gove/news/releases/2004/02/20040203-2.html; accessed 3/32/04) |
|
64 |
APHIS Fact Sheet, Veterinary Services, July 2003. |
|
65 |
APHIS Fact Sheet, Veterinary Services, July 2003. |
|
66 |
Accord, Bobby, Statement before the House Subcommittee on Agriculture, Rural Development, Food and Drug Administration, and Related Agencies. |
|
67 |
Bruntz, Stan, APHIS-VS-CEAH, Presentation on the National Animal Health Reporting System |
|
68 |
Bruntz, Stan, APHIS-VS-CEAH, Presentation on the National Animal Health Reporting System |
|
69 |
Dr. J. Annelli, APHIS-VS-EP, Personal Communication, April 2004. |
|
70 |
APHIS Fact Sheet, Veterinary Services, July 2003. |
|
71 |
Dr. J. Annelli, APHIS-VS-EP, Personal Communication, April 2004. |
|
72 |
USAHA, Report of the Joint USAHA/AAVLD Committee on Animal Health Informa- |
|
|
tion Systems, 2003 Reports. (Http://www.usaha.org/reports/reports03/r03ahis.html; accessed 4/12/04) |
|
73 |
APHIS Fact Sheet, Veterinary Services, July 2003. |
|
74 |
APHIS-VS Strategic Plan FY 2004 to FY 2008, Updated February 2004. |
|
75 |
USDA-VS Strategic Plan FY 2004 to FY 2008, Updated February 2004. |
|
76 |
USDA-VS Strategic Plan FY 2004 to FY 2008, Updated February 2004. |
|
77 |
USAIP, the United States Animal Identification Plan, January 2004. |
|
78 |
NAHEMS Steering Committee. Standard for Animal Health Emergency Management Systems. January 2000. |
|
79 |
NAS-NRC, Countering Agricultural Bioterrorism, The National Academies Press, 2003. |
|
80 |
APHIS Fact Sheet, Veterinary Service, July 2003. |
|
81 |
APHIS-VS Strategic Plan FY 2004 to FY 2008, updated February 2004. |
|
82 |
Roy, Alma, Louisiana Vet Med Diag Lab, School of Vet Med, LSU, Presentation on Animal Diagnostic Capabilities. |
|
83 |
NAS-NRC, Countering Agricultural Bioterrorism, The National Academies Press, 2003. |
|
84 |
USDA-VS Strategic Plan FY 2004 to FY 2008. Updated February 2004. |
|
85 |
USDA Fact Sheet, National Animal Health Network, May 30, 2003. |
|
86 |
APHIS Fact Sheet, Veterinary Service, July 2003. |
|
87 |
USAHA, Report of the Joint USAHA/AAVLD Committee on Animal Health Information Systems. 2002 Committee Report. (http://www.usaha.org/reports/report02/r02ahis.html; accessed 4/12/04) |
|
88 |
APHIS Fact Sheet, Veterinary Service, July 2003. |
|
89 |
APHIS Fact Sheet, Veterinary Service, July 2003. |
|
90 |
USAHA, Report of the Joint USAHA/AAVLD Committee on Animal Health Information Systems. 2002 Committee Reports. (http://www.usaha.org/reports/reports02/r02ahis.html) |
|
91 |
USAHA, Report of the Joint USAHA/AAVLD Committee on Animal Health Information Systems. 2002 Committee Reports. (http://www.usaha.org/reports/report02/r02ahis.html; accessed 4/12/04) |
|
92 |
USDA VS Strategic Plan FY2004 to FY 2008, Updated February 2004. |
|
93 |
USDA VS Strategic Plan FY2004 to FY 2008, Updated February 2004. |
|
94 |
Gilchrist, Mary, “The progress, priorities and concerns of public health laboratories.” Forum on Emerging Infections, Biological Threats and Terrorism, Institute of Medicine, November 28, 2001. |
|
95 |
CDC Fact Sheet, Laboratory Preparedness for Emergencies. February 4, 2004. |
|
96 |
CDC Fact Sheet, Laboratory Preparedness for Emergencies. February 4, 2004. |
|
97 |
CDC Fact Sheet, Laboratory Preparedness for Emergencies, February 4, 2004. |
|
98 |
CDC, National Center for Infectious Diseases. (http://www.cdc.gov/ncidod/about.htm; accessed 3/18/04) |
|
99 |
CDC Fact Sheet, Laboratory Preparedness for Emergencies. February 4, 2004. |
|
100 |
NAS-NRC, Countering Agricultural Bioterrorism, The National Academies Press, 2003. |
|
101 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
102 |
CFDA, 10.207 Animal Health and Disease Research. (http:www.cfda.gov/public/viewprog.asp?progid=38; accessed 4/8/04 |
|
103 |
CSREES, FY2005 President’s FY2005 Budget Proposal—Advancing Knowledge for the Food and Agricultural System, Overview. Feb 2004. |
|
104 |
CSREES, NRI. (http://www.reeusda.gov/nri/nriinfo/about.htm; accessed 4/13/04) |
|
105 |
CSREES, FY2005 President’s FY2005 Budget Proposal—Advancing Knowledge for the Food and Agricultural System, Overview. Feb 2004. |
|
106 |
USDA Fact sheet, Homeland Security Efforts, June 2003. |
|
107 |
USDA-ARS National Programs. (http://www.ars.usda.gov/research/programs.htm; accessed 2/27/04) |
|
108 |
USDA-ARS Action Plan. (http://www.ars.usda.gov/research/programs/programs.htm?np_code=103?docid=820; accessed 3/1/04) |
|
109 |
USDA-ARS Action Plan. (http://www.ars.usda.gov/research/programs/programs.htm?np_code=103?docid=820; accessed 3/1/04) |
|
110 |
Iowa State University, USDA and International Resources. (http://www.vetmed.iastate.edu/academics/international/resources.html; accessed 4/12/04) |
|
111 |
USDA-ARS Action Plan. (http://www.ars.usda.gov/research/programs/programs.htm?np_code=103?docid=820; accessed 3/1/04) |
|
112 |
USDA-ARS Action Plan, Epidemiology of Diseases. (http://www.ars.usda.gov/research/programs/programs.htm?np_code=103?docid=820; accessed 3/1/04) |
|
113 |
DHS Research and Technology. (http://www.dhs.gov/dhspublic/display?theme=27&content=937; accessed 3/1/04) |
|
114 |
Albright, Penrose, Assistant Secretary for Science and Technology, Department of Homeland Security, Testimony before the U.S. Senate Committee on Governmental Affairs, November 19, 2003. |
|
115 |
DHS-Homeland Security Centers Program. (http://www.cals.wisc.edu/research/hlsecurity.html; accessed 4/5/04) |
|
116 |
Merril, R.A., and J.K. Francer, Organizing Federal Food Safety Regulation. Seton Hall Law Review, No.1,Vol.31, 2000. |
|
117 |
USAMRIID Fact Sheet. (http://www.usarmiid.army.mil/programs/index.html; accessed 3/8/04) |
|
118 |
McNair Paper 65. Chapter 4, Recommendations and Conclusions (http//www.ndu.edu/inss/McNair/mcnair65/08_ch04.htm; accessed 4/12/04) |
|
119 |
CDC Continuation Guidance for Cooperative Agreement on Public Health Preparedness and Response for Bioterrorism—Budget Year Four, Program Announcement 99051. May 2, 2003. |
|
120 |
Homeland Security Presidential Directive/HSPD-5. |
|
121 |
Accord, Bobby, Statement before the House Subcommittee on Agriculture, Rural Development, Food and Drug Administration, and Related Agencies. |
|
122 |
NAHEMS Strategic Plan for years 2000 through 2005. (http://www.usaha.org/NAHEMS/Strategy.html; accessed 3/1/04) |
|
123 |
USDA-APHIS-VS: ESF 11b. Animal Production. National Animal Health Emergency Response Plan for An Outbreak of Foot-and-Mouth Disease or Other Highly Contagious Animal Diseases (February 11, 2004). |
|
124 |
USDA-APHIS-VS: ESF 11b. Animal Production. National Animal Health Emergency Response Plan for An Outbreak of Foot-and-Mouth Disease or Other Highly Contagious Animal Diseases (February 11, 2004) |
|
125 |
McNair Paper 65. Chapter 4, Recommendations and Conclusions (http//www.ndu.edu/inss/McNair/mcnair65/08_ch04.htm; accessed 4/12/04) |
|
126 |
USDA-APHIS-VS: ESF 11bAnimal production. National Animal Health Emergency Response Plan For An Outbreak of Foot-And-Mouth Disease or Other Highly Contagious Animal Diseases (February 11, 2004). |
|
127 |
Dr. J. Annelli, APHIS-VS-EP, Personal Communication, April 2004. |
|
128 |
USDA-APHIS-VS: ESF 11b Animal production. National Animal Health Emergency Response Plan For an Outbreak of Foot-and-Mouth Disease or Other Highly Contagious Animal Diseases (February 11, 2004). |
|
129 |
Accord, Bobby, Administrator, APHIS, Statement before the House Subcommittee on Agriculture, Rural Development, Food and Drug Administration, and Related Agencies. |
|
130 |
APHIS Veterinary Services Fact Sheet, March 2003. |
|
131 |
APHIS Veterinary Services Fact Sheet, The New APHIS Emergency Operations Center, March 2003. |
|
132 |
USDA-APHIS-VS: ESF 11b Animal Production. National Animal Health Emergency Response Plan For an Outbreak of Foot-and-Mouth Disease or Other Highly Contagious Animal Diseases (February 11, 2004). |
|
133 |
USDA-APHIS-VS: ESF 11b Animal Production. National Animal Health Emergency Response Plan for An Outbreak of Foot-and-Mouth Disease or Other Highly Contagious Animal Diseases (February 11, 2004). |
|
134 |
APHIS Fact Sheet, Veterinary Services, July 2003. |
|
135 |
Dr. J. Annelli, APHIS-VS-EP, Personal Communication, April 2004. |
|
136 |
Accord, Bobby, Administrator, APHIS, Statement before the House Subcommittee on Agriculture, Rural Development, Food and Drug Administration, and Related Agencies. |
|
137 |
Accord, Bobby, Administrator, APHIS, Statement before the House Subcommittee on Agriculture, Rural Development, Food and Drug Administration, and Related Agencies. |
|
138 |
Iowa State University, USDA and International Resources. (http://www.vetmed.iastate.edu/academics/international/resources.html; accessed 4/12/04) |
|
139 |
Iowa State University, USDA and International Resources. (http://www.vetmed.iastate.edu/academics/international/resources.html; accessed 4/12/04) |
|
140 |
Statement of Dr. Charles Lambert, Senate Committee on Government Affairs. “Agroterrorism: The Threat to America’s Breakfast.” November 19, 2003. (http:/govt-aff.senate.gov/index.cfm?Fuseaction=Hearings.Testimony&HearingID=127&WitnessID=482; accessed 4/2/2004) |
|
141 |
David Kinker, National Animal Health Laboratory Network, Presented to the Secretary’s Committee on Foreign Animal and Poultry Disease, Riverdale, MD, February 4, 2004; Committee on Foreign Animal Diseases of the United States Animal Health Association, Foreign Animal Diseases, 1998 Edition, Richmond, VA: USAHA, 1998. (Cited in House Democrats statement 2004) |
|
142 |
House Democrats, 2004 House Democrats Statement: Protecting the Food Supply. (www.houselgov/hsc/democrats/pdf/press/protectingthefoodsupply.pdf, Accessed 4/5/04) |
|
143 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
144 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
145 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
146 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
147 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
148 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
149 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
150 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
151 |
Statement of Dr. Charles Lambert, Senate Committee on Government Affairs. “Agroterrorism: The Threat to America’s Breakfast.” November 19, 2003. (http:/govt-aff.senate.gov/index.cfm?Fuseaction=Hearings.Testimony&HearingID=127&WitnessID=482; accessed 4/2/2004) |
|
152 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
153 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
154 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
155 |
Statement of Dr. Charles Lambert, Senate Committee on Government Affairs. “Agroterrorism: The Threat to America’s Breakfast.” November 19, 2003. (http:/govt-aff.senate.gov/index.cfm?Fuseaction=Hearings.Testimony&HearingID=127&WitnessID=482; accessed 4/2/2004) |
|
156 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/index.html) |
|
157 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
158 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
159 |
OMB Budget Documents. (http://www.whitehouse.gov/omb/budget/fy2005/appendix.html) |
|
160 |
Homeland Security, Budget in Brief, Fiscal Year 2005. |
|
161 |
USDA VS Strategic Plan FY2004 to FY 2008, Updated February 2004. |
|
162 |
AVMA, Javma News. March 15, 2004. (http://www.avma.org/onlnews/javma/mar04/040315n.asp) |
|
163 |
USDA Fact Sheet, National Animal Health Network, May 30, 2003. |
|
164 |
USAHA Report of the Joint USAHA/AAVLD Committee on Animal Health Information Systems. 2002 Committee Reports. (http://www.usaha.org/reports/reports02/r02ahis.html) |
|
165 |
USDA Fact Sheet, National Animal Health Network, May 30, 2003. |
|
166 |
OMB’s Department of Agriculture Part Assessments (Performance Measurements) February 2004. (www.whitehouse.gov/omb/budget/FY2005/pma/agriculture.pdf; accessed 4/12/04) |
|
167 |
McNair Paper 65, Chapter 4, Recommendations and Conclusions. (http//www.ndu.edu/inss/McNair/mcnair65/08_ch04.htm; accessed 4/12/04) |
|
168 |
CIDRAP. (http:www.cidrap.umn.edu/cidrap/content/bt/bioprep/news/oct0103labs.html; accessed 4/2/04) |
|
169 |
DeHaven, Ron. A Report on “Declining Infrastructure of Governmental Animal Health Professionals Puts American Agriculture at Risk.” August 2003. |
|
170 |
OMB’s Department of Agriculture Part Assessments (Performance Measurements) February 2004. (www.whitehouse.gov/omb/budget/FY2005/pma/agriculture.pdf; accessed 4/12/04) |
|
171 |
USDA Fact Sheet, Homeland Security Efforts, June 2003. |
|
172 |
DeHaven, Ron. A Report on “Declining Infrastructure of Governmental Animal Health Professionals Puts American Agriculture at Risk.” August 2003. |
|
173 |
US Fish and Wildlife Service, Division of Law Enforcement, Annual Report FY 2001. |
|
174 |
US Fish and Wildlife, Office of Law Enforcement. www.le.fws.gov/Contact_Info_Ports.htm |
|
175 |
US Fish and Wildlife Service, Division of Law Enforcement, Annual Report FY 2001. |
|
176 |
USGS, National Wildlife Health Center. (http://www.nwhc.usgs.gov; accessed 3/8/04) |
|
177 |
USGS, National Wildlife Health Center, About the NWHC. (http://www.nwhc.usgs.gov/about_nwhc/index.html; accessed 3/8/04) |
|
178 |
USGS, NWHC, Diagnosing and Controlling Wildlife Disease. (http://www.nwhc.usgs.gov/about_nwhc/diagnosing_controlling.html; accessed 3/8/04) |
|
179 |
USGS-NWHC Information Sheet, June 2001. |
|
180 |
USGS-NWHC, Foot and Mouth Disease. (http://www.nwhc.usgs.gov/research/fmd/fmd.html; accessed 3/8/04) |
|
181 |
APHIS-VS Strategic Plan FY 2004 to FY 2008, Updated February 2004. |
|
182 |
USGS-NWHC, West Nile Virus Project. (http://www.nwhc.usgs.gov/research/west_nile/west_nile.html; accessed 3/8/04) |
|
183 |
USGS, The National Biological Information Infrastructure, an Overview, May 2001. (http://www.nbii.gov) |
|
184 |
NBII, Federal NBII Partners. (http://www.nbii.gov/about/partner/feds.html; accessed 3/18/04) |
|
185 |
NBII, Fisheries and Aquatic Resource Node. (http://www.nbii.gov) |
|
186 |
National Biological Information Infrastructure, A National Biological Information Infrastructure Overview. January 2003. (http://www.nbii.gov) |
|
187 |
NBII Fact Sheet, Wildlife Disease Information Note. November 2002. (http://www.nbii.gov) |
|
188 |
Vivian Pardo Nolan, Program Coordinator, Wildlife Disease Information Node, National Biological Information Infrastructure (NBII) Program, U.S. Geological Survey, Biological Informatics Office (Personal Communication, 4/13/04). |
|
189 |
USDA Fact Sheet, USDA Wildlife Services Protects Livestock. |
|
190 |
USDOC Seafood Inspection Program Fact Sheet. (http://www.seafood.nmfs.noaa.gov); accessed 3/17/04) |
|
191 |
US DOC. The Rationale For A New Initiative in Marine Aquaculture. September 2002. |
|
192 |
US DOC. The Rationale For A New Initiative in Marine Aquaculture. September 2002. |
|
193 |
Guide to Federal Aquaculture Programs and Services. (http://ag.ansc.purdue.edu/aquanic/jsa/federal_guide/index.htm; accessed 3/17/04) |
|
194 |
Merril, R.A., and J.K. Francer, Organizing Federal Food Safety Regulation. Seton Hall Law Review, No.1,Vol.31, 2000. |
|
195 |
CRS Issue Brief for Congress, Meat and Poultry Inspection Issues, Updated June 6, 2003. IB10082 |
|
196 |
FDA Bovine Spongiform Encephalopathy, Emergency Response Plan, December 24, 2003. (http://www.fda.gov/oc/opacom/hottopics/bseplan/bseplan.html; accessed 4/2/ 04) |
|
197 |
FDA Fact Sheet, The Food and Drug Administration’s Seafood Regulatory Program. (http://www.cfsan.fda.gov/~lrd/sea-ovr.html; Accessed 3/17/04) |
|
198 |
Crawford, Lester, Statement before the Committee on Government Affairs, US Senate, November 19, 2003. |
|
199 |
FDA Fact Sheet, Safeguarding Animal Health to Protect Consumers. Publication No FS01-6. Revised Feb 2002. |
|
200 |
FDA Fact Sheet, CVM. (http://www.fda.gov/cvm/aboutcvm/aboutosc.htm; accessed 2/27/04) |
|
201 |
FDA Bovine Spongiform Encephalopathy, Emergency Response Plan, December 24, 2003. (http://www.fda.gov/oc/opacom/hottopics/bseplan/bseplan.html; accessed 4/2/04). |
|
202 |
CRS Issue Brief for Congress, Meat and Poultry Inspection Issues, Updated June 6, 2003. IB10082 |
|
203 |
Ibid. |