3
A Class Approach to Evaluating Organohalogen Flame Retardants: Case Studies
In Chapter 2, the committee described a scoping plan for evaluating nonpolymeric, additive organohalogen flame retardants (OFRs) as a single class for the purpose of hazard assessment.1 This chapter first discusses creation of an inventory of OFRs and then provides analyses that show that chemicals in the inventory do not represent a single class for a regulatory hazard assessment that uses Consumer Product Safety Commission (CPSC) guidance. However, a science-based approach to creating OFR subclasses is described and recommended. The chapter then provides an example of a literature survey and identifies two case studies that are used to illustrate several steps described in the scoping plan (see Chapter 2, Figure 2-1). Because the case studies are illustrative, not all steps of the scoping plan are conducted nor are any hazard assessments performed. Furthermore, the committee does not provide a comprehensive list of all databases or tools that might be needed to assist with the analyses. The chapter concludes by discussing options for handling discordant data on subclass members and addressing a projected timeline and cost for a class approach to hazard assessment of OFRs.
CAN ORGANOHALOGEN FLAME RETARDANTS BE DEFINED AS A SINGLE CLASS?
The committee used several approaches to answer the question of whether the OFRs can be treated as a single class for hazard assessment. Details of the approaches are provided in Appendix B. As noted in Chapter 2, the main steps are as follows:
- Identify and characterize a “seed” set of chemicals as a working inventory of the class.
- Generate an “expanded” set of chemical analogues of the seed set on the basis of combined functional, structural, and predicted bioactivity information.
- Evaluate the similarity of the seed set to the analogues to evaluate whether the OFRs are distinguishable as a single class.
Box 3-1 presents an overview of the steps used by the committee to develop an OFR inventory. Additional details are provided in Appendix B.
After defining the OFR inventory, the committee identified chemically related analogues that are similar to the seed chemicals (Box 3-2). Analogues can serve several purposes other than comparison with seed chemicals, including identification of chemicals that might be used as OFRs in the future and of chemicals that might have toxicology and other data to support a class-based hazard assessment. Further details are provided in Appendix B.
When an expanded set of chemical analogues had been created, the committee analyzed the chemical space represented by the seed and analogue chemicals to determine whether the OFRs could be distinguished as a single class. The committee’s analyses included the following:
- Prediction of structure–activity relationships (SARs) by using an open-source application called OPEn structure–activity/property Relationship App (OPERA v2.0) (Mansouri et al. 2016a,b; 2018a,b; Kleinstreuer et al. 2018).2 The OPERA SAR predictions showed that the seed chemicals exhibited heterogeneity in physicochemical properties, environmental fate, and toxicity end points, including estrogen and androgen receptor activities and acute oral toxicity.
- Principal component analysis (PCA) on OPERA physicochemical properties. The PCA analysis
___________________
1 The abbreviation OFRs in this report refers specifically to nonpolymeric, additive organohalogen flame retardants.
2 See prediction file: pred_OPERA_OFR.csv and list of OPERA models: OPERA2.0_models.xlsx and OFRs.xlsx. Files available at www.nap.edu/25412.
showed that the seed chemicals have similar physicochemical, toxicological and environmental fate properties that were not distinguishable from those of the expanded set of chemical analogues.3
When the analyses were considered collectively (see Appendix B for a detailed discussion), the committee concluded that the seed chemicals do not have a common chemical structure or predicted biologic activity. That conclusion was supported by compiling and reviewing data from the US Environmental Protection Agency (EPA) Dashboard (Williams et al. 2017) and ToxCast and Tox21 databases (Richard et al. 2016), which showed that the OFR seed chemicals had a wide array of biologic activities that varied from chemical to chemical.4 Thus, the broad class needs to be divided into subclasses to support a regulatory hazard assessment.
DEFINITION OF SUBCLASSES OF ORGANOHALOGEN FLAME RETARDANTS
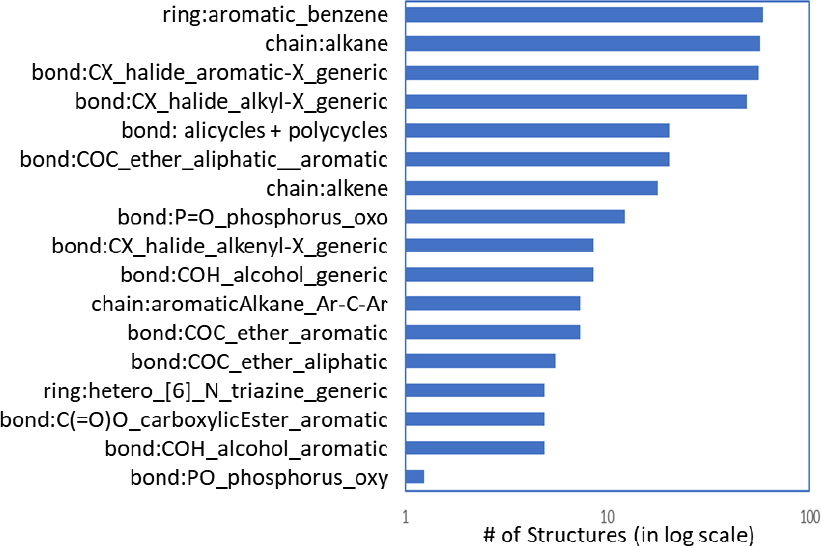
The committee used cheminformatic approaches to create OFR subclasses. A public set of chemotypes5 and methods that have been developed by Yang et al. (2015) and Richard et al. (2016) were used to identify the chemotypes present in the seed chemicals, which are listed in Figure 3-1. Using the chemotypes, the committee was able to identify several generic classes that represented the entirety of the OFR seed set (Table 3-1). Merging the biology-informed groups with the chemotypes listed in Figure 3-1 led to the formulation of 14 OFR categories for the inventory of 161 OFR chemicals (Table 3-2). Appendix B provides additional details on how the subclasses were formed and evaluated. The committee recommends that CPSC use the subclasses in Table 3-2 at least as a starting point for the class-based hazard assessment of OFRs.
___________________
3 See Appendix B for additional details.
4 See OFR_ChemistryDashboard-Batch-Search_2018-12-03_17 _03_39.xls and Appendix C for additional details. File available at www.nap.edu/25412.
5 ToxPrint, available at https://chemotyper.org, provides coverage of EPA and Food and Drug Administration inventories and captures chemical features important for chemical safety assessments.
SURVEY OF THE LITERATURE
When the subclasses have been formed, the committee recommends conducting a literature survey (see Chapter 2, Figure 2-1). As discussed in Chapter 2, the goal of the literature survey is to determine the extent, range, and nature of toxicity data (human, animal, in vitro, and other relevant studies) and to identify end points that deserve investigation.6 It is distinct from the literature search performed later in the scoping plan in that it is meant to provide a broad understanding of the literature, not to identify every relevant article that might need to be retrieved and evaluated for the hazard assessment of a specific subclass.
To identify subclasses that could serve as case studies, the committee surveyed the literature in several steps. It first conducted an initial mapping exercise to evaluate the types of toxicity data available on each subgroup and then conducted several literature surveys to identify end points of possible interest. Specifically, the committee initially surveyed the literature by using bioinformatically mappable databases, which contain biologic data that can be systematically retrieved by using batch software queries for the chemicals of interest. The committee searched the following databases to determine whether they contained data for the subclass members: Comparative Toxicogenomics Database (CTD), EPA Chemical Dashboard, Hazardous Substances Data Bank (HSDB), Integrated Risk Information System (IRIS), ToxCast/Tox21, Toxicity Reference Database (ToxRefDB), PubChem, and ChEMBL. The committee notes that other databases might also be surveyed, and it simply used the ones listed for illustrative purposes. Results for the OFR subclasses and analogues are shown in Figures 3-2 and 3-3 (see also Appendix C: Figures C-1, C-2, C-3, C-4, and C-5). The survey provided an indication of the amounts and types of information (data coverage) that might be available on each OFR. The mapping exercise, however, did not identify end points of interest. The details of the exercise are provided in Appendix C.
To gain an understanding of possible end points of interest, the committee next used CAS numbers to explore PubChem for toxicity data on each seed chemical. The query focused on chronic toxicity, reproductive and developmental toxicity, mutagenicity, and cancer, which were end points that were specifically listed in the committee’s task statement. Other end points could be considered. The survey results were used to identify subclasses that contained members on which there was relevant toxicity information, including epidemiology, traditional mammalian toxicity, and new approach methodologies (NAM) studies.7 One objective in the selection of the two case studies was to illustrate how high-throughput, alternative species, and other NAM data could be used to inform a hazard assessment (Box 3-3). Two relatively
___________________
6 As noted in Chapter 2, the term literature is used broadly to refer to scientific literature and databases.
7 Completion of the scoping plan does not require that a subclass have each of the data streams noted.
TABLE 3-1 Chemotypes Identified in OFR Seed Chemicals That Have Been Associated with Predicted Biologic Activitya
| Chemotype | Biologic Activity | Reference |
|---|---|---|
| Chain: aliphatic cycles | GABA receptor antagonist; AhR enzyme in steroidogenesis | Eager et al. 1999 |
| Ring: polycycles | GABA receptor modulator; ER/AR modulator | Babot et al. 2007 |
| Bond: ether aromatic diphenyl | Aromatase enzymes in steroidogenesis; aromatase inhibition | Peters et al. 2006 |
| Bond: alcohol aromatic phenols | Aromatase enzymes in steroidogenesis Bisphenol A: proposed for estrogen-mediated pathways by binding estrogen receptors; proposed for oxidative stress for ROS formation leading to early embryonic damage |
Guo et al. 2017 |
| Ring: aromatic_benzenes | AhR signaling | Abiko et al. 2016 |
| Ring: hetero_triazines | Effects in steroidogenesis | Forgacs et al. 2013 |
| Bond: aromatic carboxylic acid and derivatives | Nuclear receptor pathways (such as PPAR alpha) | Eveillard et al. 2009 |
| Bond: P=O or P-O | Acetylcholinesterase inhibitors | Abou-Donia et al. 2016 |
aIndividual OFRs that have a given chemotype might or might not demonstrate the biologic activity indicated here.
Abbreviations: AhR, aryl hydrocarbon receptor; ER/AR, estrogen receptor/androgen receptors; GABA, gamma aminobutyric acid; PPAR, peroxisome proliferator activated receptor; ROS, reactive oxygen species.
TABLE 3-2 OFR Subclasses Formulated by Using Chemotypes and Predicted Biologic Activity
| OFR Subclass | No. Chemicals in Subclassa |
|---|---|
| Polyhalogenated alicycles | 17 |
| Polyhalogenated aliphatic carboxylate | 4 |
| Polyhalogenated aliphatic chains | 12 |
| Polyhalogenated benzene alicycles | 4 |
| Polyhalogenated benzene aliphatics and functionalized | 19 |
| Polyhalogenated benzenes | 19 |
| Polyhalogenated bisphenol aliphatics and functionalized | 11 |
| Polyhalogenated carbocycles | 15 |
| Polyhalogenated diphenyl ethers | 12 |
| Polyhalogenated organophosphates (OPs) | 22 |
| Polyhalogenated phenol derivatives | 7 |
| Polyhalogenated phenol–aliphatic ether | 9 |
| Polyhalogenated phthalates/benzoates/imides | 11 |
| Polyhalogenated triazines | 6 |
aSeven chemicals were categorized by using two chemotypes and included in two subclasses. This analysis was performed by using the chemicals in the OFR inventory.

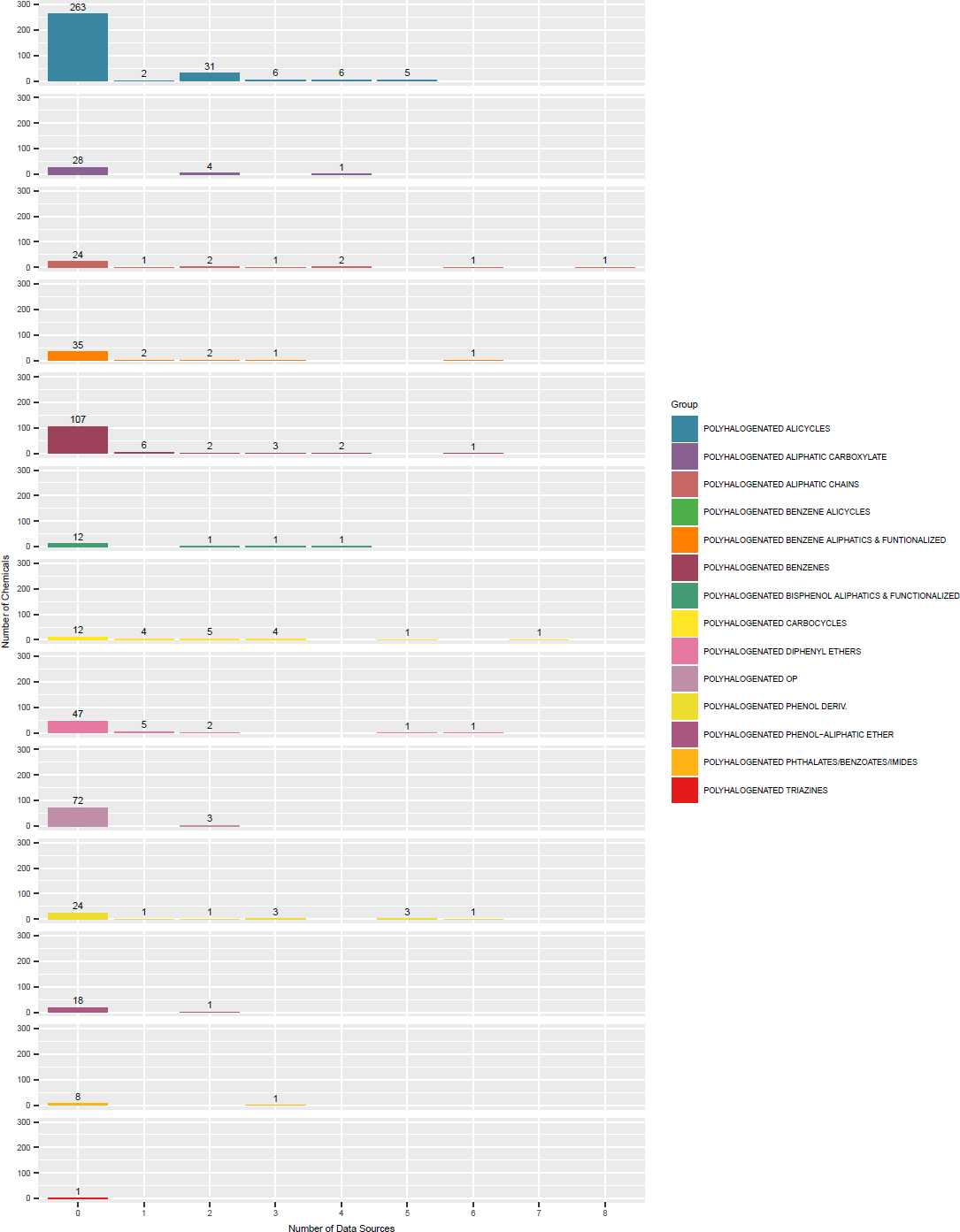
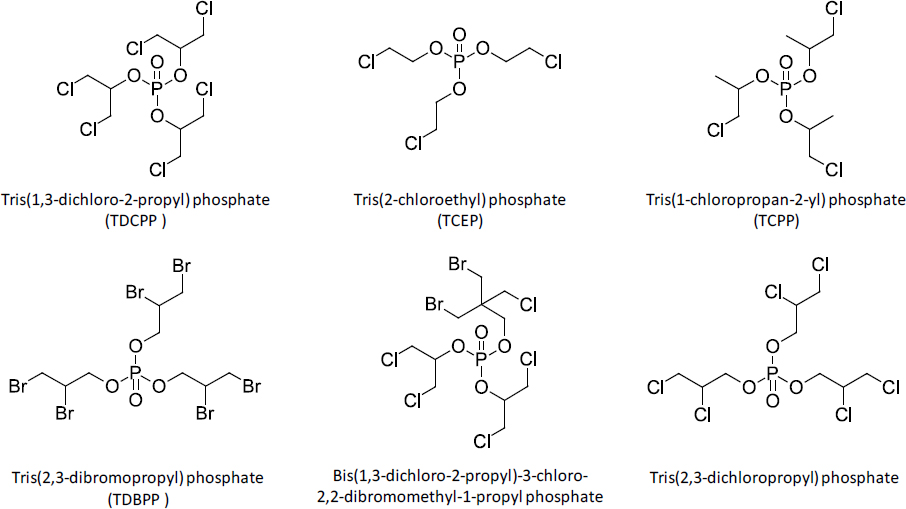
TABLE 3-3 Members of the Polyhalogenated Organophosphate Subclass
| Chemical Name | Abbreviation | CAS No. |
|---|---|---|
| Tris(1,3-dichloro-2-propyl) phosphate | TDCPP | 13674-87-8 |
| Tris(2-chloroethyl) phosphate | TCEP | 115-96-8 |
| Tris(1-chloropropan-2-yl) phosphate | TCPP | 13674-84-5 |
| Tris(2,3-dibromopropyl) phosphate | TDBPP | 126-72-7 |
| Bis(1,3-dichloro-2-propyl)-3-chloro-2,2-dibromomethyl-1-propyl phosphate | 66108-37-0 | |
| Tris(tribromo-neopentyl) phosphate | TTBNPP | 19186-97-1 |
| 2,2-Bis(chloromethyl)-1,3-propanediyl bis(bis(2-chloroethyl) phosphate) | 38051-10-4 | |
| Tris(2,3-dichloropropyl) phosphate | 78-43-3 | |
| Tris(2-chloropropyl) phosphate | 6145-73-9 | |
| Tetrakis(2-chloroethyl) ethane-1,2-diyl bis(phosphate) | 33125-86-9 | |
| Bis(2-chloroethyl) vinylphosphonate | 115-98-0 | |
| Bis(2,3-dibromopropyl) phosphate | 5412-25-9 | |
| Tris(2-chloroethyl) phosphite | 140-08-9 | |
| Phosphonic acid, (1-(((2-chloroethoxy)(2-chloroethyl)phosphinyl)oxy)ethyl)-, 1-(bis(2-chloroethoxy)phosphinyl)ethyl 2-chloroethyl ester | 4351-70-6 | |
| Bis(2-chloroethyl) (2-chloroethyl)phosphonate | 6294-34-4 | |
| Tris(1,3-dichloropropan-2-yl) phosphite | 6749-73-1 | |
| Oxydiethylene tetrakis(2-chloroethyl) bisphosphate | 53461-82-8 | |
| ,4,8,10-Tetraoxa-3,9-diphosphaspiro[5.5]undecane, 3,9-bis[3-bromo-2,2-bis(bromomethyl)propoxy]-, 3,9-dioxide | 61090-89-9 | |
| Tris(2,4-dibromophenyl) phosphate | 49690-63-3 | |
| Phenol, 2,4,6-tribromo-, phosphate | 7046-64-2 | |
| Polyhalogenated OP + triazines (phosphonic acid, P-[[(4,6-dichloro-1,3,5-triazin-2-yl)oxy]methyl]-, dimethyl ester) | TPN1 | 114955-21-4* |
| Polyhalogenated OP + triazines (phosphonic acid, (4,6-dichloro-1,3,5-triazin-2-yl)-, diethyl ester (9CI)) | TPN2 | 1373346-90-7* |
*CAS no. from SciFinder.
data-rich OFR subclasses—polyhalogenated organophosphates (OPs) and polyhalogenated bisphenol aliphatics—were selected to serve as case studies to illustrate various aspects of the hazard-assessment scoping plan.
SEARCH OF THE LITERATURE
After the literature survey and the creation of an analysis plan, the literature is searched to identify all relevant data associated with the subclass members and the end points of interest.8 As noted at the beginning of this chapter, the committee did not attempt to complete all steps of its scoping plan and so did not create an analysis plan. It also did not conduct comprehensive, systematic literature searches; the searches were conducted so that the committee could illustrate steps of its scoping plan, and relevant literature could have been missed. The committee did, however, search multiple databases for toxicity data in vertebrates on each member of the subclasses chosen for the case studies.9 The searches were intended to gather the available literature on the two subclasses and to illustrate the process that could be used (see Appendix C for details). For the two selected subclasses, the committee searched for English-language, peer-reviewed articles in PubChem and PubMed by CAS number, chemical name and synonyms, and outcomes of interest by using the following search terms:
- Toxicity OR reproductive toxicity OR developmental toxicity.
___________________
8 As noted in Chapter 2, the term literature is used broadly to refer to scientific literature and databases.
9 The committee recognizes that OFRs have also been evaluated in invertebrate animal models. For example, the effect of flame retardants on feeding, larval development, reproduction, and motor activity have been evaluated in the nematode Caenorhabditis elegans (Behl et al 2016; Xu et al 2016).
TABLE 3-4 Genotoxicity Data on the Polyhalogenated Organophosphate Subclass
| Chemical | Experimental Results |
|---|---|
| TDCPP | Mostly negative in in vitro gene-mutation assays in bacteria and yeasts; positive in some Salmonella typhimurium strains when tested with metabolic activation; negative in assays of point mutations, sister chromatid exchange, and unscheduled DNA synthesis; mixed results in mouse lymphoma assay, chromosomal-aberration assay, and transformation assay; and negative in in vivo tests of sex-linked recessive lethal mutations in Drosophila, unscheduled DNA synthesis in rats, formation of micronuclei in polychromatic erythrocytes of mice, and chromosomal aberrations in mice (Environment Canada and Health Canada 2016). |
| TCEP | Not mutagenic in bacteria in absence of metabolic activation; equivocal results with metabolic activation (IARC1999a). EPA (2009) found overall evidence to be negative. |
| TCPP | Negative in Ames assays; equivocal results in mouse lymphoma assays, unscheduled DNA synthesis assays, and comet assays; positive in in vitro mouse lymphoma assay when tested with metabolic activation; negative in in vivo micronucleus tests and comet assays; equivocal or negative in in vivo assays of unscheduled DNA synthesis and chromosomal aberrations; and in vivo micronucleus tests in mice and rats had positive results only in male mice (Environment Canada and Health Canada 2016). |
| TDBPP | Mutagenic (van Beerendonk et al. 1994; de Boer et al. 1996, 2000). Mutagenic in S. typhimurium and in V79 Chinese hamster lung cells; positive in assays of sister chromatid exchanges and morphologic transformation in mouse and hamster embryo cells; binds covalently to proteins and DNA and causes DNA strand breaks in mammalian cells in vitro and in vivo; mutagenic, clastogenic, and recombinogenic in Drosophila melanogaster; induces bone-marrow micronuclei in mice and hamsters, liver micronuclei in rats, and gene mutations in mouse kidney in vivo (IARC 1999b). |
| Bis(1,3-dichloro-2-propyl)-3-chloro-2,2-dibromomethyl-1-propyl phosphate | Mutagenic in rat dominant lethal assay (Litton Bionetics, Inc. 1977). |
| Bis(2,3-dibromopropyl) phosphate | Evidence of mutagenicity (IPCS 1995a). |
TABLE 3-5 Polyhalogenated Organophosphate Chronic Toxicity Studies
| Chemical | Experimental Results |
|---|---|
| TDCPP | In a 2-year carcinogenicity study in Sprague-Dawley rats (Stauffer Chemical Co. 1981): male rats, significant increase in renal cortical adenomas, testicular interstitial cell tumors, and hepatocellular adenomas; female rats, significant increase in renal cortical adenomas, hepatocellular adenomas, and adrenal cortical adenomas. |
| TCEP | Benign and malignant tumors occurred in various organs in rats and mice (NTP 1991); tumors were found in kidneys, liver, forestomach, and hematopoietic system of mice (Takada et al. 1989). |
| TCPP | 90-day and 2-year oral-carcinogenicity studies are being conducted by NTP. |
| TDBPPa | Reasonably anticipated to be a human carcinogen (NTP 2016); probably carcinogenic to humans (IARC 1999b). |
| Bis(2,3-dibromopropyl) phosphate | Rats developed papillomas and adenocarcinomas of the tongue, esophagus, and forestomach; adenocarcinomas of the intestine; and hepatocellular adenomas [neoplastic nodules] and carcinomas (Takada et al. 1991). |
aThe TDBPP metabolite 2,3-dibromo-1-propanol is carcinogenic in rodents (Eustis et al. 1995).
- Genotoxicity OR mutagenicity.
- Cancer OR carcinogenicity.
The search found a few well-studied chemicals in both classes that could help to anchor the hazard assessment of other subclass members. Each subclass also contained chemicals on which there were few or no relevant traditional (mammalian) toxicity data. An additional PubMed literature search conducted in November 2018 used the search terms “zebrafish” and “flame retardants” and retrieved 118 publications. The committee also considered two publications that reported results of zebrafish assays that were used to screen the large ToxCast chemical library, which included polyhalogenated OPs (Truong et al. 2014; Reif et al. 2016). The committee illustrates the use of zebrafish data in the following case studies because such data were available on several OFRs. Their use is not a statement that zebrafish data will be the best or most useful NAM data to use in all cases.
CASE STUDY 1: POLYHALOGENATED ORGANOPHOSPHATES
The committee identified 22 chemicals in the polyhalogenated OP subclass, which are listed in Table 3-3. Chemical structures for several subclass members are shown in Figure 3-4.
As noted above, literature searches were performed by using each chemical name, CAS number, and relevant toxicity search terms. Studies of genotoxicity, carcinogenicity, and reproductive and developmental toxicity were also sought from authoritative compilations from the European Chemicals Agency (ECHA), EPA, Environment Canada and Health Canada, the World Health Organization’s International Programme on Chemical Safety, and the National Toxicology Program (NTP). The committee found that Tox21 or ToxCast data, rodent reproductive or developmental toxicity data, or zebrafish data on several members of the polyhalogenated OP subclass were available. Some epidemiologic data on several subclass members were available. Genotoxicity data are summarized in Table 3-4. As a subclass, these chemicals have discordant data; that is, some members have been negative in the Ames assay, and others have been positive.
The results of chronic mammalian bioassays are presented in Table 3-5. A metabolite of TDBPP, 2,3-dibromo-1-propanol, has been shown to be carcinogenic in rodents (Eustis et al. 1995). TDBPP has been identified as probably carcinogenic in humans (Group 2A) by the International Agency for Research on Cancer (IARC 1999b) and reasonably expected to be a human carcinogen by NTP (2016) on the basis of animal carcinogenicity data (Reznik et al. 1981). TCEP causes benign and malignant tumors at various organ sites in rats and mice (NTP 1991). A carcinogenicity study with TCPP is underway at NTP.
A third outcome, developmental toxicity, was also identified as an end point of interest in this case study, and the committee chose this end point to illustrate evaluation and integration of data in the scoping plan (Figure 2-1). The committee emphasizes that a hazard assessment of the OPs would consider mutagenicity, carcinogenicity, and developmental toxicity and might also consider neurotoxicity, hepatotoxicity, and other systemic effects; that is, the overall class-based hazard assessment would consider multiple end points of interest. The following subsections discuss the various data streams for developmental toxicity and overall classification for this specific end point.
Developmental Toxicity Data on Polyhalogenated Organophosphates
The committee’s analysis considered three main data streams: epidemiologic, traditional mammalian, and zebrafish studies. Exposures were considered relevant if they occurred during development. Additional toxicity data could also be considered. For example, Farhat et al. (2013) indicated that exposure of chicken embryos to TDCPP lowered free thyroxine (T4) concentrations in the blood and resulted in malformations, including changes in head-to-bill lengths. Similarly, frog (Xenopus tropicalis) embryos exposed to TCPP or TDCPP developed multiple malformations (Zhang et al. 2016).
Epidemiologic Data
Despite their relatively short half-lives, polyhalogenated OPs likely contribute to continuous exposure concentrations in humans (Dishaw et al. 2014; He et al. 2018; Phillips et al. 2018). Polyhalogenated OPs are hydrolyzed to diester metabolites that undergo urinary excretion (Cequier et al. 2015), and the metabolites have been used as biomarkers of OP flame-retardant exposure of people (Dodson et al. 2014; Hoffman et al. 2014; Cequier et al. 2015; Romano et al. 2017). However, not all OP metabolites can be attributed to the polyhalogenated OP flame retardants; some nonhalogenated OP flame retardants, such as triphenyl phosphate (TPHP), are metabolized to some of the same metabolite diesters as the polyhalogenated OPs (Cequier et al. 2015; Wei et al. 2018).
Some OP flame-retardant metabolites—including diphenyl phosphate (DPHP), bis(1,3-dichloro-2-propyl) phosphate (BDCIPP), isopropylphenyl phenyl phosphate (ip-PPP), and 1-hydroxy-2-propyl bis(1-chloro-2-propyl) phosphate (BCIPHIPP)—are nearly ubiquitous in human samples (Ospina et al. 2018; Doherty et al. 2019; Wang et al. 2019), but few epidemiologic studies have examined associated health outcomes. A few small studies of reproductive-age cohorts have supported the potential of some metabolites to be associated with changes in sex and
TABLE 3-6 Available Human Epidemiologic Data on Organophosphate Flame Retardantsa
| Study Design | Sample, Exposure Period,b Chemical | Outcome | Results | Reference |
|---|---|---|---|---|
| Neurodevelopment | ||||
| Prospective cohort (n = 227) | Prenatal urine 2001-2005 DPHP ip-PPP BDCIPP BCIPHIPP |
Cognition at age 3 years | ip-PPP associated with lower cognition, fine motor, and expressive language. | Doherty et al. 2019 |
| Prospective cohort (n = 310) | Prenatal urine 1999-2000 DPHP ip-PPP BDCIPP BCIPHIPP |
Cognition, behavior age 7 years | No association between ip-PPP and BDCIPP metabolite (DPHP) and working memory, IQ deficits. | Castorina et al. 2017 |
| Cross-sectional (n = 72) | Age 3-5 years wristband 2012-2013 Sum OPFR |
Behavior, social skills | Sum OPFR associated with externalizing problems (aggression). | Lipscomb et al. 2017 |
| Reproduction | ||||
| Cross-sectional (n = 50) | Dust 2002-2007 TDCPP TPP |
Semen quality | TDCPP and TPP associated with alterations in thyroid, sex hormones. | Meeker and Stapleton 2010 |
| Cross-sectional (n= 33) | Urine 2003-2004 DPHP BDCIPP |
Semen quality, sex hormone, thyroid hormone | BDCIPP and DPHP associated with altered sperm quality and higher total T3. BDCIPP associated with higher TSH. | Meeker et al. 2013 |
| Prospective cohort (n = 201) | Preconception urine 2005-2015 Sum OPFR |
Semen quality, fertility | Decreased oocyte fertilization, embryo quality. | Carignan et al. 2018 |
| Prospective cohort (n = 211) | Preconception urine 2004-2015 Sum OPFR DPHP ip-PPP BDCIPP |
Pregnancy, implantation, live birth | Sum OPFR, DPHP, ip-PPP associated with decreased implantation, pregnancy. | Carignan et al. 2017 |
| Retrospective cohort (n = 220) | Preconception urine 2005-2015 Sum OPFR DPHP ip-PPP BDCIPP |
Semen quality | OPFR not associated with semen measures. | Ingle et al. 2018 |
| Prospective cohort (n = 179) | Preconception urine 2005-2015 Sum OPFR DPHP ip-PPP BDCIPP |
Pregnancy loss | DPHP associated with pregnancy loss; ip-PPP and BDCIPP not associated with loss. | Messerlian et al. 2018 |
| Prospective cohort (n = 349) | Prenatal urine 2002-2005 DPHP ip-PPP BDCIPP BCIPHIPP |
Gestational age, birthweight | BDCIPP, ip-PPP associated with early delivery, especially of female infants; DPHP and BCIPHIPP not associated with reproductive outcomes. | Hoffman et al. 2018 |
| Prospective cohort (n = 23) | Prenatal urine 2015 DPHP BDCIPP |
Miscarriage, birthweight | DPHP, BDCIPP not associated with miscarriage, birthweight, but study was small. | Feng et al. 2016 |
| Thyroid | ||||
| Cross-sectional (n = 51) | Urine 2010-2011 DPHP |
Thyroid hormone concentrations | DPHP associated with increase in total TT4, especially among women, but not associated with other thyroid hormones. | Preston et al. 2017 |
a Polyhalogenated OPs are identified by boldface, and halogenated metabolites are identified by italic type.
bExposure period for prospective studies of perinatal exposures in relation to later reproductive-developmental outcomes. No study period listed for cross-sectional studies concurrently measuring exposure and outcome.
Abbreviations: DPHP, diphenyl phosphate; ip-PPP, isopropylphenyl phenyl phosphate; BDCIPP, bis(1,3-dichloro-2-propyl) phosphate; BCIPHIPP, 1-hydroxy-2-propyl bis(1-chloro-2-propyl) phosphate; OPFR, organophosphate flame retardant; TDCPP, tris(1,3-dichloro-2propyl) phosphate; TPP, triphenyl phosphate; TSH, thyrotropin; TT4, total thyroxine.
TABLE 3-7 Developmental Toxicity of Polyhalogenated Organophosphates
| Chemical | Experimental Results |
|---|---|
| TDCPP | No developmental toxicity or thyrotoxicity in rats exposed during gestation and weaning (Moser et al. 2015). |
| Suppression of sexual behavior and reduced testes in male rats exposed neonatally (Kamishima et al. 2018). | |
| NOAEL of 100 mg/kg-day in rats (significant increase in rate of resorption, significant decrease in fetal viability index, retarded skeletal development); maternal toxicity observed (NOAEL, 25 mg/kg-day) (Stauffer Chemical Co. 1978). | |
| NOAEL of 200 mg/kg-day in rats (based on fetal mortality); no effects on neurodevelopment at 200 mg/kg-day or below; maternal toxicity observed (NOAEL, 100 mg/kg-day) (Tanaka et al. 1981). | |
| TCEP | No developmental toxicity or thyrotoxicity in rats exposed during gestation and weaning (Moser et al. 2015). |
| NOAEL of 100 mg/kg-day in rats for maternal toxicity and developmental toxicity (Kawashima et al. 1983); teratogenic and neurobehavioral effects were evaluated. | |
| TCPP | Two-generation reproductive toxicity study in rats found LOAEL of 1,500 mg/k-day based on an increased number of runts (TNO Quality of Life 2007); preliminary range-finding study had similar results. |
| Study in Wistar rats found NOAEL of 1,000 mg/kg-day; a few cases of missing 13th ribs and cervical ribs; no maternal toxicity (Kawasaki et al. 1982). | |
| TDBPP | No teratogenic effect in rats (IPCS 1995a; IARC 1999b). |
Abbreviations: LOAEL, lowest observed-adverse-effect level; NOAEL, no-observed-adverse-effect level.
aMalformations associated with increased mortality.
bEffects associated with systemic toxicity.
| Chemical | Mammalian | Zebrafish | ||
|---|---|---|---|---|
| Teratogenic | Developmental Neurotoxicity | Teratogenic | Developmental Neurotoxicity or Altered Locomotor Activity | |
| TDCPP | – | – | + | + |
| TCEP | – | – | + | + |
| TCPP | – | Not determined | – | + |
| TDBPP | – | Not determined | – | + |
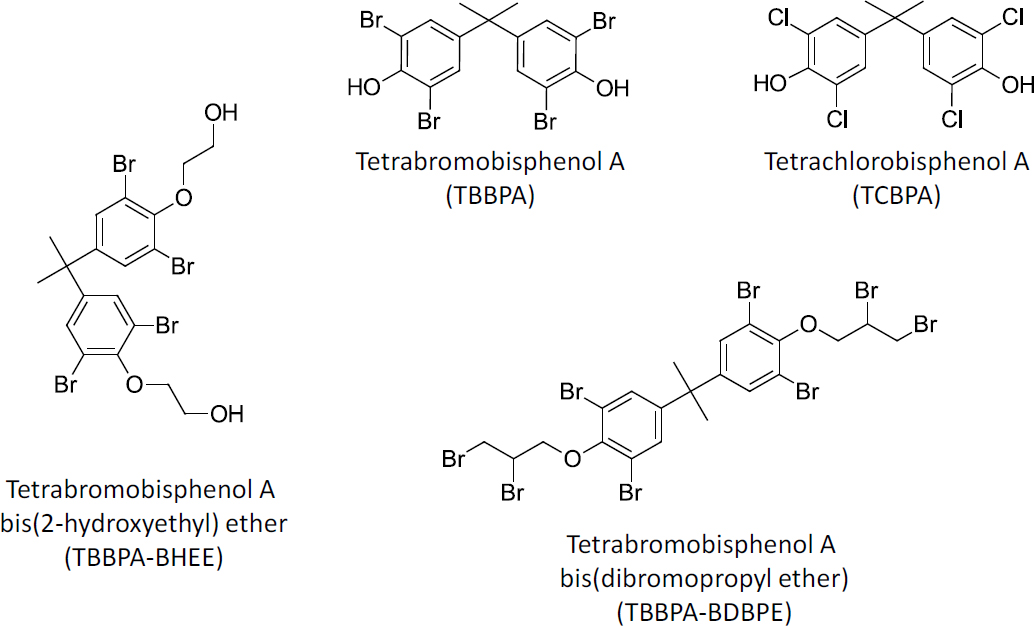
TABLE 3-10 Polyhalogenated Bisphenol Aliphatics
| Chemical Name | Abbreviations | CAS No. |
|---|---|---|
| Tetrabromobisphenol A (phenol, 4,4’-(-methylethylidene) bis[2,6-dibromo-]) | TBBPA | 79-94-7 |
| Tetrachlorobisphenol A | TCBPA | 79-95-8 |
| Tetrabromobisphenol A bis(2-hydroxyethyl) ether (ethanol, 2,2'-[(1-methylethylidene) bis[(2,6-dibromo-4,1-phenylene)oxy]]bis-) | TBBPA-BHEE TBBPA-OHEE | 4162-45-2 |
| Tetrabromobisphenol A bis(dibromopropyl ether) (benzene, 1,1'-(1-methylethylidene) bis[3,5-dibromo-4-(2,3-dibromopropoxy)) | TBBPA-BDBPE TBBPA-DBPE | 21850-44-2 |
| Tetrabromobisphenol A BME (benzene, 1,1'-(1-methylethylidene) bis[3,5-dibromo-4-methoxy) | TBBPA-BME TBBPA-DME | 37853-61-5 |
| 3,3',5,5'-Tetrabromobisphenol A bispropionate (phenol, 4,4'-(1-methylethylidene) bis[2,6-dibromo-, dipropanoate (9CI)) | TBBPA-BP | 37419-42-4 |
| Tetrabromobisphenol A bis(2-hydroxyethyl) ether bis(acrylate) (2-propenoic acid, 1,1'-[(1-methylethylidene)bis[(2,6-dibromo-4,1-phenylene)oxy-2,1-ethanediyl]] ester) | TBBPA-BHEEBA | 66710-97-2 |
| Tetrabromobisphenol A diglycidyl ether (oxirane, 2,2'-[(1-methylethylidene) bis[(2,6-dibromo-4,1-phenylene)oxymethylene]]bis-) | TBBPA-BGE | 3072-84-2 |
| Phenol, 4,4'-(1-methylethylidene)bis[2,6-dibromo-, 1,1'-diacetate | TBBPA-BOAc | 33798-02-6 |
| Tetrabromobisphenol A diallyl ether (benzene, 1,1'-(1-methylethylidene)bis[3,5-dibromo-4-(2-propen-1-yloxy)-) | TBBPA-BAE | 25327-89-3 |
| 2,2,6,6-Tetrabromo bisphenol A diacrylate (2-propenoic acid, 1,1'-[(1-methylethylidene)bis(2,6-dibromo-4,1-phenylene)] ester) | TBBPA-BA | 55205-38-4 |
thyroid hormones (Meeker and Stapleton 2010; Preston et al. 2017), but results were imprecise and inconsistent. Disruption of sex and thyroid hormones can adversely affect fertility, birth outcomes (Carignan et al. 2017, 2018; Messerlian et al. 2018), and neurodevelopment (Sarles et al. 1987; Castorina et al. 2017). In two prospective studies of exposure during pregnancy, OP flame retardants were associated with reduced language, cognition, and working memory in the children (Castorina et al. 2017; Doherty et al. 2019), but the specific implicated metabolites differed between the two studies. Table 3-6 provides a summary of the findings of the epidemiologic studies.
Mammalian Toxicity Studies
Developmental toxicity data were available on four of the polyhalogenated OPs (see Table 3-7). Although the four do not appear to be teratogenic, one study of TDCPP reported effects on sexual behavior in male rats exposed during development. Oral exposure (28 consecutive days after birth) of neonatal male rats to TDCPP suppressed male sexual behavior and reduced testis size (Kamishima et al. 2018). In contrast, oral exposure of pregnant Long-Evans rats to TDCPP or TCEP from gestational day 10 to weaning was not associated with thyrotoxicity or developmental neurotoxicity in offspring (Moser et al. 2015).
Zebrafish Studies
Toxicity data on four of the polyhalogenated OPs (TDCPP, TCEP, TCPP, and TDBPP) were available from the zebrafish model system. Depending on the polyhalogenated OP, chemical exposure of zebrafish resulted in malformations or behavioral changes, although contradictory (negative) studies have also been reported (Table 3-8). Study details and key findings from the studies are presented in Appendix D (Table D-1). Other studies of zebrafish in which teratogenic or developmental neurotoxic effects were not assessed were also reviewed and are summarized in Appendix D (Table D-2).
Several laboratories conducted zebrafish studies with multiple polyhalogenated OPs that allowed more direct comparisons between subclass members. Exposure concentrations of polyhalogenated OPs varied among chemicals. Noyes et al. (2015) reported that TDBPP (3.3 µM) and TDCPP (10 µM) had overt toxicity thresholds, and none was found for TCEP or TCPP up to 100 µM. A similar trend was observed for neurotoxicity with effect thresholds at 0.56 µM for TDBPP, 3.14 µM for TDCPP, 31.4 µM for TCEP, and 100 µM for TCPP (Noyes et al. 2015). McGee et al. (2012), Du et al. (2015), and Alzualde et al. (2018) likewise noted greater toxicity of TDCPP than of TCEP or TCPP. Differences in responses were also seen among the four chemicals. Dishaw et
al. (2014) found that TCEP, TCPP, and TDBPP were not teratogenic, whereas TDCPP was teratogenic. Noyes et al. (2015) concluded that TCEP and TCPP exhibited similar neurotoxicity, but the neurotoxicity of TDCPP differed from that of TCEP and TCPP. Dach et al. (2019) observed hypoactivity as an effect of TCPP and TCEP at 30 µM 96 and 120 h after fertilization and detected no malformations up to this test concentration. In adult fish, TDCPP and TCEP were found to undergo metabolism through a dechlorination pathway, but TDCPP had a longer half-life in tissues than TCEP (Wang et al. 2017). The data also indicate that differences among the chemicals regarding neurotoxic, teratogenic, and developmental toxicity depend on the exposure period.
Hazard Assessment of the Polyhalogenated Organophosphates
After data evaluation, the next step in the scoping plan is integration of the analyses for each end point of interest, for example, mutagenicity, carcinogenicity, and developmental toxicity. Regarding developmental toxicity, the committee considered the best-studied chemicals in the polyhalogenated OP subclass (Table 3-9). Oral exposure of pregnant rats to TDCPP, TCEP, TCPP, or TDBPP did not produce teratogenic or developmental neurotoxicity in their offspring. However, studies of the same chemicals in zebrafish found positive results for teratogenic effects of TDCPP and TCEP and developmental neurotoxicity for all four; this suggests developmental effects. A TDCPP metabolite, BDCIPP (Hoffman et al. 2017), has not been associated with changes in working memory or IQ deficits in children (Castorina et al. 2017).
The developmental toxicity data on the four chemicals are discordant. Several options for handling the discordant data are discussed after the next case study.
CASE STUDY 2: POLYHALOGENATED BISPHENOL ALIPHATICS
The committee identified 11 chemicals in the subclass of polyhalogenated bisphenol aliphatics (Table 3-10). The subclass members have bisphenol A as the core structure.
TBBPA is the base structure of this subclass, and TCBPA is the chlorinated analogue (Figure 3-5). Ten of the bisphenol A-based OFRs contain bromines in the 3,3’,5,5’ positions. There is a comprehensive traditional toxicology database on TBBPA. TBBPA and TCBPA are active against several biological targets in in vitro assay systems. Other members of the subclass have various R group substitutions on the phenolic carbons, and these substitutions might alter targets of activity in in vitro studies in contrast with TBBPA. In mammals and other vertebrates, conjugation with glucuronic acid or sulfate on
TABLE 3-11 Genotoxicity Data on Polyhalogenated Bisphenol Aliphatics
| Chemical | Experimental Results | Alertsa | |
|---|---|---|---|
| Ames | In Vivo Clastogenicity | ||
| TBBPA | Ames (-)b In vivo cytogenicity (-)b In vitro DNA damage (+/-)c In vivo DNA damage (-)c In vivo mutation (+)c |
No | No |
| TCBPA | No data | No | No |
| TBBPA-BHEE | Ames (-)d | No | Yes |
| TBBPA-DBPE | Ames (+/-)e Clastogenicity (-)e |
Yes | Yes (aliphatic halogen) |
| TBBPA-BME | No data | No | No |
| TBBPA-BP | No data | No | No |
| TBBPA-BHEEBA | No data | No | Yes |
| TBBPA-BGE | Ames (-)f Clastogenicity (-)f |
Yes | Yes (epoxide) |
| TBBPA-BOAc | No data | No | No |
| TBBPA-BAE | Ames (-)e | No | No |
| TBBPA-BA | No data | No | No |
Note: (+), positive results; (-), negative results; (+/-), conflicting results.
aToxtree, http://toxtree.sourceforge.net/ames.html; http://toxtree.sourceforge.net/mic.html.
TABLE 3-12 Subchronic and Chronic Toxicity Studies of Polyhalogenated Bisphenol Aliphatics
| Chemical | Experimental Results |
|---|---|
| Subchronic Studies | |
| TBBPA | Rats: NOAEL, 100 mg/kg-day; ↓T4 (no change in T3 or TSH), ↑liver weight; ↓spleen weight (NTP 2014). Mice: NOAEL, 100 mg/kg-day; ↑liver weight, ↓spleen weight, ↓kidney weight, histopathologic changes in kidney (NTP 2014). |
| TBBPA-BHEE | Non-GLP 28-day study: NOAEL, 1,000 mg/kg-day (IPCS 1995b). |
| TBBPA-DBPE | 90-day GLP gavage study in F344N rats and B6C3F1 mice: no toxicity reported at doses up to 1,000 mg/kg-day in rats or 2,000 mg/kg-day in mice (highest doses tested) (NTP 2017). |
| TBBPA-BGE | 28-day GLP/guideline oral study in Wistar rats: NOAEL, 300 mg/kg-day; at 1,000 mg/kg-day, rats had ↓body weight, ↑liver weight, centrilobular hypertrophy, ↑alanine aminotransferase, aspartate aminotransferase, bile acids (considered treatment-related but not adverse) (ECHA 2018a). |
| Chronic Studies | |
| TBBPA | Gavage studies in B6C3F1/N mice and Wistar Han [Crl:WI(Han)] rats (NTP 2014). Male mice: significant increases in incidence of hepatoblastoma. Female mice: no significant increases in tumor incidence. Male rats: no significant increases in tumor incidence. Female rats: significant increase in incidence of adenocarcinoma of the uterus; several rats had malignant mixed Müllerian tumor, a rare uterine tumor; significant increase in incidence of adenoma, adenocarcinoma, or malignant mixed Müllerian tumor (combined) of the uterus. |
Abbreviations: NOAEL, no-observed-adverse-effect level; GLP, good laboratory practices; T4, thyroxine; T3, triiodothyronine; TSH, thyroid-stimulating hormone.
TABLE 3-13 Summary of Human Epidemiologic Studies of Polyhalogenated Bisphenol Aliphatics
| Study Design | Chemical | Outcome Measured | Results | Reference |
|---|---|---|---|---|
| Human infants (n = 38, 26 with congenital hypothyroidism and 12 typical) |
TBBPA | Thyroid | TBBPA higher in infants than mothers, but the two correlated; TBBPA exposure was associated with higher maternal free thyroxine, higher thyroid peroxidase antibody micro AB, and thyroid-stimulating immunoglobulin; interpretation of study results difficult due to small, hypothyroid sample. | Kim and Oh 2014 |
| Cross-sectional study of adolescents (age 13–17 years) (n = 515) |
TBBPA | Attention, motor development, thyroid | Contemporary TBBPA was not associated with attention, working memory, motor function, or thyroid hormone concentrations. | Kiciński et al. 2012 |
TABLE 3-14 Mammalian Developmental Toxicity Studies of Polyhalogenated Bisphenol Aliphatics
| Chemical | Experimental Results |
|---|---|
| TBBPA | Prenatal studies in rodents: no effects in standard studies at doses up to 10,000 mg/kg-day (ECB 2006a). |
| Two-generation rat study: NOAEL for fertility, fecundity, and developmental changes was 1,000 mg/kg-day; ↓T4 at LOAEL of 100 mg/kg-day; and ↓T3 at 1,000 mg/kg-day (reported in NTP 2014b). | |
| Negative in uterotrophic assay by both oral and subcutaneous routes in mice (Ohta et al. 2012). Positive in mice by intraperitoneal injection (Kitamura et al. 2005). | |
| EPA (2015c): moderate evidence of developmental effects.c | |
| Developmental toxicology studies in rats found no significant developmental effects; NOAEL of 1,000 mg/kg-day (Cope et al. 2015). | |
| TBBPA-DBPE | Prenatal developmental toxicity study (rats, oral, according to OECD Guideline 414): fetal NOAEL of 1,000 mg/kg-day; maternal NOAEL of 300 mg/kg-day (↓food consumption, ↓body weight) (ECHA 2018b). |
aPrimary references are MPI Research (2001), Noda et al. (1985), and Velsicol Corporation (1978).
bPrimary reference is ACC (2002).
cPrimary references are Goldenthal et al. (1978), Noda et al. (1985), MPI Research (2001), ACC (2002), Darnerud (2003), Tada et al. (2006), and Saegusa et al. (2009).
the TBBPA and TCBPA phenolic rings leads to (or is predicted to lead to) relatively rapid excretion. Metabolism of other subclass members might occur on the phenolic substitutions and possibly lead to metabolic and toxicologic divergence.
TBBPA and related subclass members have been assessed by several authoritative bodies (IPCS 1995b; ECB 2006; Environment Canada and Health Canada 2013; EFSA 2011a; NTP 2014; EPA 2015b). As noted above, the committee searched for additional traditional toxicology data and NAM data that might be incorporated to facilitate bridging from the well-studied chemicals to the poorly studied ones. PubChem, SciFinder, and ChemID plus were searched along with the European Food Safety Authority, ECHA, EPA, Environment Canada and Health Canada, and International Programme on Chemical Safety Web sites. Primary searches were restricted to sources indexed by CAS number. Additional focused searches were conducted for identified biologic targets of subclass members to locate comparative data on at least two subclass members. Genotoxicity data are provided in Table 3-11. As a subclass, these chemicals have been negative in the Ames assay and generally lack structural alerts that suggest a mutagenic potential. Data on the subclass from mammalian subchronic and chronic studies are presented in Table 3-12. Only one subclass member, TBBPA, has been tested for carcinogenicity.
As with the first case study, the hazard assessment of the polyhalogenated bisphenol aliphatics would consider multiple end points of interest. To provide another example of data evaluation and integration, the committee focused its evaluation of this OFR subclass on its ability to
alter thyroid hormone homeostasis in adults or neonates and to produce morphologic or behavioral developmental effects. The committee considered epidemiologic, mammalian toxicity, and zebrafish data and considered exposures relevant if they occurred during development. The following subsections discuss the various data streams and their evaluation and integration for the specific end points noted.
Epidemiologic Studies
Two studies have investigated the polyhalogenated bisphenol aliphatics in relation to health outcomes in human samples (Table 3-13). One small study of mother–infant pairs at delivery suggested that TBBPA might be associated with higher free T4 in serum but not other endocrine markers in peripartum women; the effect was not observed in their infants (Kim and Oh 2014). A cross-sectional study of adolescents did not find contemporary TBBPA concentrations to be associated with attention, working memory, or thyroid hormone concentrations; however, studies of neurodevelopment ideally investigate exposure during earlier windows of brain development, such as the prenatal period or early childhood, rather than contemporary exposure during adolescence (Kiciński et al. 2012). The current epidemiology literature contributes little to hazard assessment, but assays of TBBPA and other subclass members could complement cohort studies with stored prenatal or early life samples.
Mammalian Toxicity Studies
In subchronic studies, TBBPA has been shown to decrease circulating T4 concentrations with no change in either triiodothyronine (T3) or thyroid-stimulating hormone (TSH) concentrations, increase liver weight, and decrease spleen weight in rats and mice (NTP 2014). Additional evidence from animal studies suggests that exposure to TBBPA can alter thyroid homeostasis in rodents (Lilienthal et al. 2008; Van der Ven et al. 2008; Cope et al. 2015). Lai et al. (2015) proposed that the thyroid effects are mediated by induction of UGT1A that results in increased T4 catabolism. Sanders et al. (2016) reported decreased serum T4 concentration and increased hepatic and uterine expression of Thra gene that encodes TRα in rats 24 h after 5 days of oral administration of TBBPA. They also noted UGT1A was upregulated in the liver and uterus after TBBPA administration; however, they concluded that the mechanism for changes in T4 is uncertain. TBBPA can displace T4 from its plasma transport protein (Hamers et al. 2006) and inhibits binding of T3 to the thyroid receptor TRβ1 and binding of T4 to transthyretin
TABLE 3-15 Summary of Evidence on TBBPA and Changes in Thyroid Homeostasis
| Species | Finding | Reference |
|---|---|---|
| Human | TBBPA exposure was associated with higher maternal free T4, thyroid peroxidase antibody micro AB and thyroid-stimulating immunoglobulin. | Kim and Oh (2014) |
| Human | TBBPA was not associated with attention, working memory, motor function, or thyroid hormone concentrations. | Kiciński et al. (2012) |
| Rat | ↓T4 (no change in T3 or TSH) | NTP (2014) |
| Rat | Two-generation rat study: NOAEL for fertility, fecundity, and developmental changes was 1,000 mg/kg-day; ↓T4 at LOAEL of 100 mg/kg-day; and ↓T3 at 1,000 mg/kg-day. | NTP (2014) |
| Zebrafish | ↑ T4; ↓ T3. | Zhu et al. (2018) |
| Zebrafish | Interference in thyroid homeostasis. | De Wit et al. (2008) |
| Mammalsa | Zebrafishb | ||
|---|---|---|---|
| Chemical | Developmental toxicity | Teratogenesis | Locomotor Activity |
| TBBPA | +/− | + | +/− |
| TBBPA-DBPE | +/− | − | − |
| TBBPA-BHEE | Not determined | + | + |
| TBBPA-BME | Not determined | − | Not determined |
aSee Table 3-14.
bSee Appendix D, Table D-4.
(Meerts et al. 2000; Sun et al. 2009). Meerts et al. (2000) found that TCBPA inhibits T4 binding to transthyretin but with less potency than TBBPA. Despite effects on thyroid metabolism, the polyhalogenated bisphenol aliphatic subclass, however, has minimal developmental effects in mammals (Table 3-14).
Zebrafish Studies
TBBPA is toxic to zebrafish embryos, and there is a database on TBBPA-related effects on thyroid hormone concentrations and actions in zebrafish that suggest that the zebrafish assay might be a useful model for assessing similarities and differences within the structural class. For example, Chen et al. (2016) reported an increase in the expression of UGT genes after TBBPA exposure, which they note could affect T4 metabolism and then lead to neurobehavioral changes. Zebrafish studies of TBBPA, TCBPA, TBBPA-BHEE, TBBPA-BDBPE, and TBBPA-BME are available. Effects of this subclass on thyroid and development (including behavior) are summarized in Appendix D (Tables D-3 and D-4). Additional zebrafish studies are summarized in Table D-5.
Class Hazard Assessment Based on Effects on Thyroid Homeostasis
The committee’s initial evaluation considered TBBPA, which is the best-studied chemical in the polyhalogenated bisphenol aliphatic subclass (Table 3-15). TBBPA can alter thyroid homeostasis so as to result in inconsistent changes in T3 and T4 concentrations. The data are discordant between rodent and zebrafish. Additional in vitro, zebrafish, and other thyroid-homeostasis studies of the polyhalogenated bisphenol aliphatics could provide confidence that thyroid function can serve as a key end point for hazard classification.
Class Hazard Assessment Based on Developmental Toxicity
The committee’s evaluation initially considered the best-studied chemicals in the polyhalogenated bisphenol aliphatic subclass (Table 3-16). Each chemical has been studied in zebrafish, and the results are discordant. The rodent developmental toxicity studies of two subclass members have also resulted in mixed findings. Overall, the data on the four best-studied chemicals in the subclass are discordant for this specific end point. Analysis of other end points might support a hazard assessment of the subclass. The next section discusses the committee’s recommendations for dealing with discordant data.
ADDRESSING DISCORDANT DATA
Both case studies had discordant data on developmental toxicity between species (for example, rodent vs zebrafish), within a species, or both. Table 3-17 presents the options that could be considered when discordant data are identified: performing analyses, collecting new data, reclassifying the subclass, or making policy decisions. The approaches listed in Table 3-17 for the various options are offered as examples and are not meant to constitute a comprehensive list of all approaches that might be useful or appropriate.
TABLE 3-17 Options and Approaches for Handling Discordant Data
| Options | Approach | Advantages | Potential Pitfalls |
|---|---|---|---|
| Perform analyses | Evaluate data quality, pharmacokinetics, or specific physicochemical properties, such as log P; investigate doses, metabolites, exposure windows, or other features that could explain discordance. | Potential to understand discordance and move forward. | Might not reveal clear reason for discordance. |
| Generate new data | Conduct NAM studies, targeted animal studies, or new evaluations of epidemiologic samples. | Can increase clarity and scientific basis of decision. | Is time-consuming, is expensive, and could still be discordant. |
| Reclassify | Divide or merge classes or identify individual “outlier” chemicals. | Refines the subclass. | Presents a potential for repeated reclassifications and could result in individual chemical assessments. |
| Make policy decisions | Impute any hazard identified within the class to chemicals on which there are no data. | Provides a health-protective default and incentivizes data generation. | Could encounter statutory and regulatory limitations. |
Analyses
One possible explanation for discordant data within an OFR subclass might be differences in pharmacokinetics. For example, an analysis of the pharmacokinetic data on two members of the polyhalogenated bisphenol aliphatic subclass (TBBPA and TBBPA-DBPE) showed different bioavailabilty in rodents after oral exposure. Unlike TBBPA, TBBPA-DBPE is poorly absorbed from the gastrointestinal tract (Hakk et al. 2000; Kuester et al. 2007; NTP 2017). That observation might partly account for the lack of effects seen in rodent developmental toxicity studies. Metabolism data could also help to identify chemicals within a subgroup that share metabolic intermediates and, by extension, toxicity profiles.
Another approach to resolving discordant data is to explore the mechanisms of action of subclass members. One method of collecting mechanistic data is to collect NAM data on subclass members (Box 3-3). Such data can help to identify OFR biologic targets of interest. Mechanistic data have many possible applications, including the following:
- Inform additional approaches to grouping the chemicals. For example, several members of the polyhalogenated OP subclass are structurally similar to some nonpolyhalogenated OP counterparts used as agrochemicals. The nonhalogenated OPs are associated with diverse toxicity mechanisms, including cholinesterase inhibition, endocrine disruption, and neurotoxicity. In vitro assays and other NAM data could be used to evaluate whether some polyhalogenated OPs inhibit acetylcholinesterase and could therefore be associated with cholinergic neurotoxicity.
- Determine the most appropriate animal models for human risk assessment.
- Identify analogues, including nonhalogenated chemicals, that have a common mechanism of action. Data on those chemicals can help to inform the risk assessment of the subclass.
It is not uncommon for different responses to occur among studies that use different species (such as rats, mice, and zebrafish), different strains within a species (such as strains of rats or mice), or different exposure windows. Assessing the human relevance of different species or strains can be useful for understanding which data would be expected to be more predictive of a selected effect, such as changes in thyroid hormones or mutagenicity. No single strain or species is expected to be predictive of all human health effects of interest, and nonmammalian species are increasingly recognized as providing reasonable predictions of selected effects in humans. However, using nonmammalian species can pose additional challenges. For example, both case studies used zebrafish data, which often were discordant. One possible explanation of discordance between zebrafish data and rodent data is that ex vivo exposure of zebrafish embryos or larvae to the parent OFR might not reflect in vivo metabolism that might occur in the rodent species. Further testing with OFR metabolites might be necessary for appropriate comparisons and yield concordance of data among species. A possible explanation for discordance among zebrafish data might be that different study protocols were used to expose the zebrafish to OFRs. Although a standardized OECD guideline (Test No. 236: Fish Embryo Acute Toxicity Test) was published in 2013, many laboratories do not use it. As a result, there are variations in developmental toxicity study designs, especially regarding the exposure window. There are also challenges in using zebrafish data, including extrapolation of water concentrations to doses that are suitable for human health assessment, but there has been progress in addressing this challenge in recent studies in which total chemical mass (based on exposure concentration and volume) relative to the mass of embryo immersed in the exposure solution is calculated. Those values can then be compared with human exposure values to determine whether a hazard would be expected to be associated with an environmentally relevant exposure.
In assessing discordant data, it is important to consider study design and implementation, which could influence observed results and might explain the discordance. For example, studies can have different powers to detect outcomes depending on the numbers of subjects or the exposure periods in reproductive-developmental studies. The doses or concentrations used in studies also can influence outcomes, and assessment of chemical purity can be important to ensure that effects do not arise from a contaminant.
Collection of New Data
Chapter 2 (Box 2-3) outlined a tiered approach for the collection of new data when there are no relevant data on any member of a subclass. That tiered approach could also be applied to the discordant-data scenario. However, the assessment that resulted in the conclusion of discordant data should have illuminated data gaps that should be useful in directing the data-collection effort.
Reclassification of the Subgroup
As mentioned above, mechanistic and pharmacokinetic data might suggest that a subgroup formed primarily on the basis of chemical structure needs to be parsed into two or more smaller subgroups. In some cases, chemicals that have multiple chemotypes are included in more than one subclass, and these chemicals might be the source of discordant data in one of the subclasses. Removing the
chemical from the subclass might be appropriate then. As mentioned in Chapter 2, however, the committee advises that reclassification should be performed judiciously to obviate a never-ending cycle of reclassification.
The committee also recognizes that alternative approaches have been used to generate OFR subclasses. For example, a study conducted by the Danish EPA (2016) grouped 67 brominated flame retardants on the basis of their chemical structures and then made QSAR predictions of selected environmental and health effects. Despite the existence of alternative classification schemes, the committee recommends that CPSC begin its analyses with the subclasses described earlier in this chapter.
Policy Decisions
The class approach relies on using data on tested chemicals to draw inferences about the potential hazard associated with class members that have not been tested. That approach is scientifically supported most strongly when many of the available data support a single conclusion (for example, when a specific toxicity is observed). When class members on which there are data appear to yield discordant findings on an end point, a key question is how to evaluate class members on which there are no data. Several inferences would be possible from the discordant findings, although they will have greater uncertainty than if the findings were concordant. Inferences would include the idea that the class members on which there are no data are similar in toxicity to class members on which there are data—for example, similar to the most toxic chemical or similar to a distribution of observed findings. Each inference could be considered in developing a hazard assessment of the class that would use policy choices to provide appropriate protection of public health. Ideally, the approach would create incentives for stakeholders to collect additional data and reduce the uncertainty in analyses.
PROJECTED TIMELINE AND COSTS
Traditional hazard-assessment methods take years and are too expensive to cover all chemicals under production. The committee has proposed a class-based hazard assessment of OFRs as an efficient alternative to traditional approaches. The proposed class-based process conveys time and cost savings in that one is no longer conducting separate hazard assessments of each individual chemical, and traditional hazard-assessment methods can be complemented by new approaches that take advantage of existing data (read-across) and require less de novo testing.
The next steps in completing class-based hazard assessments of the OFR subclasses will involve literature surveys and data-mapping for relevant toxicity end points. That process will likely require months for a research team to complete. The goal of the initial step is to evaluate whether well-studied subclass members can be used to anchor the assessment. When no such anchor chemical can be identified, it will be necessary to obtain minimal datasets on some subclass members. The committee’s initial literature survey exercise suggests that this could be the case for several OFR subclasses. For subclasses on which there are adequate data, there might be a need to create one or more chronic hazard advisory panels (CHAPs) that have specific chemical and end-point expertise. On the basis of CPSC’s experience with the CHAP that was formed to address phthalates, completion of that step could take several years.
CONCLUSIONS
This report proposes the use of emerging cheminformatic approaches to systematically expanding and refining the chemical and toxicologic information traditionally considered in assessing possible health hazards posed by a structurally or functionally related chemical class. The larger information base might reveal biologic activities of concern, such as effects on thyroid hormone homeostasis or neurodevelopment at one or more biologic organizational levels (for example, cells, zebrafish, laboratory mammals, and humans). The choices of which health end points are most important, how choices are made in the presence of uncertainty, and the relative importance of health end points require value judgments in the hazard-assessment process. Therefore, the committee recommends that CPSC consider providing both philosophic and regulatory-policy guidance to any CHAP that is charged with carrying out a class-based hazard assessment that uses the approaches described in this document.
REFERENCES
Abiko, Y., F.Y. Lin, H. Lee, A. Puga, and Y. Kumagai. 2016. Quinone-mediated induction of cytochrome P450 1A1 in HepG2 cells through increased interaction of aryl hydrocarbon receptor with aryl hydrocarbon receptor nuclear translocator. J. Toxicol. Sci. 41(6):775-781.
Abou-Donia, M.B., M. Salama, M. Elgalmal, I. Elkholi, and Q. Wang. 2016. Organophosphorus flame retardants (OPFR): Neurotoxicity. J. Environ. Health Sci. 2(1):1-30.
ACC (American Chemistry Council). 2002. An oral two generation reproductive, fertility, and developmental neurobehavioral study of tetrabromobisphenol A in rats. Submitted to the U.S. Environmental Protection Agency under TSCA Section 8E. [Secondary reference from EPA 2015a.]
Alzualde, A., M. Behl, N.S. Sipes, J.H. Hsieh, A. Alday, R.R. Tice, R.S. Paules, A. Muriana, and C. Quevedo. 2018. Toxicity profiling of flame retardants in zebrafish embry-
os using a battery of assays for developmental toxicity, neurotoxicity, cardiotoxicity and hepatotoxicity toward human relevance. Neurotoxicol. Teratol. 70:40-50.
Babot, Z., M.T. Vilaró, and C. Suñol. 2007. Long-term exposure to dieldrin reduces gamma-aminobutyric acid type A and N-methyl-D-aspartate receptor function in primary cultures of mouse cerebellar grandule cells. J. Neurosci. Res. 85(16):3687-3695.
Behl, M., J.H. Hsieh, T.J. Shafer, W.R. Mundy, J.R. Rice, W.A. Boyd, J.H. Freedman, E.S. Hunter, 3rd, K.A. Jarema, S. Padilla, and R.R. Tice. 2015. Use of alternative assays to identify and prioritize organophosphorus flame retardants for potential developmental and neurotoxicity. Neurotoxicol. Teratol. 52(Pt. B):181-193.
Behl, M., J.R. Rice, M.V. Smith, C.A. Co, M.F. Bridge, J.H. Hsieh, J.H. Freedman, and W.A. Boyd. 2016. Editor’s highlight: Comparative toxicity of organophosphate flame retardants and polybrominated diphenyl ethers to Caenorhabditis elegans. Toxicol Sci. 154(2):241-252.
Carignan, C.C., L. Mínguez-Alarcón, C.M. Butt, P.L. Williams, J.D. Meeker, H.M. Stapleton, T.L. Toth, J.B. Ford, and R. Hauser. 2017. Urinary concentrations of organophosphate flame retardant metabolites and pregnancy outcomes among women undergoing in vitro fertilization. Environ. Health Perspect. 125(8):087018.
Carignan, C.C., L. Mínguez-Alarcón, P.L. Williams, J.D. Meeker, H.M. Stapleton, C.M. Butt, T.L. Toth, J.B. Ford, and R. Hauser. 2018. Paternal urinary concentrations of organophosphate flame retardant metabolites, fertility measures, and pregnancy outcomes among couples undergoing in vitro fertilization. Environ. Int. 111:232-238.
Castorina, R., A. Bradman, H.M. Stapleton, C. Butt, D. Avery, K.G. Harley, R.B. Gunier, N. Holland, and B. Eskenazi. 2017. Current-use flame retardants: Maternal exposure and neurodevelopment in children of the CHAMACOS cohort. Chemosphere 189:574-580.
Cequier, E., A.K. Sakhi, R.M. Marcé, G. Becher, and C. Thomsen. 2015. Human exposure pathways to organophosphate triesters—a biomonitoring study of mother-child pairs. Environ. Int. 75:159-165.
Chen, J., R.L. Tanguay, Y. Xiao, D.E. Haggard, X. Ge, Y. Jia, Y. Zheng, Q. Dong, C. Huang, and K. Lin. 2016. TBBPA exposure during a sensitive developmental window produces neurobehavioral changes in larval zebrafish. Environ. Pollut. 216:53-63.
Cheng, R, Y. Jia, L. Dai, C. Liu, J. Wang, G. Li, and L. Yu. 2017. Tris(1,3-dichloro-2-propyl) phosphate disrupts axonal growth, cholinergic system and motor behavior in early life zebrafish. Aquat. Toxicol. 192:7-15.
Cope, R.B., S. Kacew, and M. Dourson. 2015. A reproductive, developmental and neurobehavioral study following oral exposure of tetrabromobisphenol A on Sprague-Dawley rats. Toxicology 329:49-59.
Dach, K., B. Yaghoobi, M.R. Schmuck, D.R. Carty, K.M. Morales, and P.J. Lein. 2019. Teratological and behavioral screening of the National Toxicology Program 91-compound library in zebrafish (Danio rerio). Toxicol. Sci. 167(1):77-91.
Danish EPA (Environmental Protection Agency). 2016. Category approach for selected brominated flame retardants: Preliminary structural grouping of brominated flame retardants. Environmental project No. 1872. Copenhagen, Denmark: Danish Environmental Protection Agency [online]. Available: https://www2.mst.dk/Udgiv/publications/2016/07/978-87-93435-90-2.pdf [accessed September 25, 2018].
Darnerud, P.O. 2003. Toxic effects of brominated flame retardants in man and in wildlife. Environ. Int. 29(6):841-853. [Secondary reference from EPA 2015a.]
de Boer, J.G., J.C. Mirsalis, G.S. Provost, K.R. Tindall, and B.W. Glickman. 1996. Spectrum of mutations in kidney, stomach, and liver from lacI transgenic mice recovered after treatment with tris(2,3-dibromopropyl)phosphate. Environ. Mol. Mutagen. 28(4):418-423.
de Boer, J.G., J. Holcroft, M.L. Cunningham, and B.W. Glickman. 2000. Tris(2,3-dibromopropyl)phosphate causes a gradient of mutations in the cortex and outer and inner medullas of the kidney of lacI transgenic rats. Environ. Mol. Mutagen. 36(1):1-4.
De Wit, M., D. Keil, N. Remmerie, K. van der Ven, E.J. van den Brandhof, D. Knapen, E. Witters, and W. De Coen. 2008. Molecular targets of TBBPA in zebrafish analysed through integration of genomic and proteomic approaches. Chemosphere 74(1):96-105.
Dishaw, L.V., L.J. Macaulay, S.C. Roberts, and H.M. Stapleton. 2014. Exposures, mechanisms, and impacts of endocrine-active flame retardants. Curr. Opin. Pharmacol. 19:125-133.
Dodson, R.E., N. Van den Eede, A. Covaci, L.J. Perovich, J.G. Brody, and R.A. Rudel. 2014. Urinary biomonitoring of phosphate flame retardants: Levels in California adults and recommendations for future studies. Environ. Sci. Technol. 48(23):13625-13633.
Doherty, B.T., K. Hoffman, A.P. Keil, S.M. Engel, H.M. Stapleton, B.D. Goldman, A.F. Olshan, and J.L. Daniels. 2019. Prenatal exposure to organophosphate esters and cognitive development in young children in the Pregnancy, Infection, and Nutrition Study. Environ. Res. 169:33-40.
Du, Z., G. Wang, S. Gao, and Z. Wang. 2015. Aryl organophosphate flame retardants induced cardiotoxicity during zebrafish embryogenesis: by disturbing expression of the transcriptional regulators. Aquat. Toxicol. 161:25-32.
Eager, E.I., 2nd., D.D. Koblin, J. Sonner, D. Gong, M.J. Laster, P. Ionescu, M.J. Halsey, and T. Hudlicky. 1999. Nonimmobilizers and transitional compounds may produce convulsions by two mechanisms. Anesth. Analg. 88(4):884-892.
Eastmond, D.A. 2015. Letter to Consumer Product Safety Commission (CPSC). Exhibit C in CPSC staff briefing package in response to petition HP15-1, requesting rulemaking on certain products containing organohalogen flame retardants. May 24, 2017. Available: https://www.cpsc.gov/content/ballot-vote-petition-hp-15-1-requesting-rulemaking-on-certain-products-containing [accessed July 18, 2018].
ECB (European Chemicals Bureau). 2006. European Union risk assessment report on 2,2’, 6,6’-tetrabromo-4,4’-isopropylidenediphenol (tetrabromobisphenol-A or TBBP-A) Part II – Human. European Commission, Joint Research Centre, European Chemicals Bureau, 63:EUR22161E.
ECHA (European Chemicals Agency). 2016. New approach methodologies in regulatory science. Proceedings of a scientific workshop, Helsinki, 19-20 April 2016. Helsinki, Finland: European Chemicals Agency [online]. Available: https://echa.europa.eu/documents/10162/22816069/scientific_ws_proceedingsen.pdf [accessed January 8, 2019].
ECHA. 2018a. Dossier 2,2’-[(1-methylethylidene)bis[(2,6-dibromo-4,1-phenylene)oxymethylene]]bisoxirane [online]. Available: https://echa.europa.eu/registration-dossier/-/registered-dossier/19397 [accessed December 27, 2018].
ECHA. 2018b. Dossier 1,1’-(isopropylidene)bis[3,5-dibro mo-4-(2,3-dibromopropoxy)benzene] [online]. Available: https://echa.europa.eu/registration-dossier/-/registereddossier/13876 [accessed December 27, 2018].
EFSA (European Food Safety Authority). 2010. Scientific opinion on polybrominated biphenyls (PBBs) in food. EFSA Journal 8(10):1789 [online]. Available: www.efsa.europa.eu/efsajournal [accessed September 25, 2018].
EFSA. 2011a. Scientific opinion on tetrabromobisphenol A (TBBPA) and its derivatives in food. EFSA Journal 9(12):2477 (updated 2013) [online]. Available: www.efsa.europa.eu/efsajournal [accessed September 25, 2018].
EFSA. 2011b. Scientific opinion on polybrominated diphenyl ethers (PBDEs) in food. EFSA Journal 9(5):2156 [online]. Available: www.efsa.europa.eu/efsajournal [accessed September 25, 2018].
EFSA. 2011c. Scientific opinion on hexabromocyclododecanes (HBCDDs) in food. EFSA Journal 9(7):2296 [online]. Available: www.efsa.europa.eu/efsajournal [accessed September 25, 2018].
EFSA. 2012a. Scientific opinion on brominated flame retardants (BFRs) in food: Brominated phenols and their derivatives. EFSA Journal 10(4):2634 [online]. Available: www.efsa.europa.eu/efsajournal [accessed September 25, 2018].
EFSA. 2012b. Scientific opinion on emerging and novel brominated flame retardants (BFRs) in food. EFSA Journal 10(10):2908 [online]. Available: www.efsa.europa.eu/efsajournal [accessed September 25, 2018].
Environment Agency. 2003. Prioritisation of flame retardants for environmental risk assessment. United Kingdom: Environment Agency [online]. Available: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/291681/scho1008bote-e-e.pdf [accessed October 5, 2018].
Environment Canada and Health Canada. 2013. Screening Assessment Report: Phenol, 4,4’-(1-methylethy-lidene) bis[2,6-dibromo- Chemical Abstracts Service Registry Number 79-94-7; Ethanol, 2,2’-[(1-methy-lethylidene)bis[(2,6-dibromo-4,1-phenylene)oxy]]bis Chemical Abstracts Service Registry Number 4162-45-2; Benzene, 1,1’-(1-methylethylidene)bis[3,5-dibromo-4-(2-propenyloxy)- Chemical Abstracts Service Registry Number 25327-89-3. Minister of the Environment [online]. Available: http://www.ec.gc.ca/ese-ees/default.asp?lang=En&n=BEE093E4-1 [accessed January 7, 2019].
Environment Canada and Health Canada. 2016. Draft Screening Assessment, Certain Organic Flame Retardants Substance Grouping: 2-Propanol, 1-chloro, phosphate (3:1) (TCPP), Chemical Abstracts Service Registry Number 13674-84-5, 2-Propanol, 1,3-dichloro-, phosphate (3:1) (TDCPP), Chemical Abstracts Service Registry Number 13674-87-8 [online]. Available: http://www.ec.gc.ca/ese-ees/B4374491-04B7-4586-8D3B4482F4141408/DSAR_Flame%20Retardants%20%28TDCPP-TCPP%29_EN_Final.pdf [accessed January 7, 2019].
EPA. 2009. Provisional peer-reviewed toxicity values for tris(2-chloroethyl)phosphate (TCEP) (CASRN 115-96-8). EPA/690/R-09/069F. Cincinnati, OH: EPA [online]. Available: https://cfpub.epa.gov/ncea/pprtv/documents/Tris2chloroethylphosphate.pdf [accessed January 10, 2019].
EPA. 2015a. Flame retardants used in flexible polyurethane foam: An alternatives assessment update. EPA 744-R-15-002. Design for the Environment, US EPA. Available: https://www.epa.gov/saferchoice/2015-update-report-flame-retardants-used-flexible-polyurethane-foam-publications [accessed February 21, 2019].
EPA. 2015b. TSCA work plan chemical problem formulation and initial assessment, tetrabromobisphenol A and related chemicals cluster flame retardants. EPA Document # 740-R1-4004. Office of Chemical Safety and Pollution Prevention, EPA [online]. Available: https://www.epa.gov/sites/production/files/2015-09/documents/tbbpa_problem_formulation_august_2015.pdf [accessed December 28, 2018].
EPA. 2015c. Flame retardants in printed circuit boards. EPA Publication 744-R-15-001. Cincinnati, OH: EPA [online]. Available: https://www.epa.gov/sites/production/files/2015-08/documents/pcb_final_report.pdf [accessed December 27, 2018].
Eustis, S.L., J.K. Haseman, W.F. Mackenzie, and K.M. Abdo. 1995. Toxicity and carcinogenicity of 2,3-dibro-mo-1-propanol in F344/N rats and B6C3F1 mice. Fun-dam. Appl. Toxicol. 26(1):41-50.
Eveillard, A., L. Mselli-Lakhal, A. Mogha, F. Lasserre, A. Polizzi, J.M. Pascussi, H. Guillou, P.G. Martin, and T. Pineau. 2009. Di-(2-ethylhexyl)-phthalate (DEHP) activates the constitutive androstane receptor (CAR): a novel signalling pathway sensitive to phthalates. Biochem. Pharmacol. 77(11):1735-1746.
Farhat, A., D. Crump, S. Chiu, K.L. Williams, R.J. Letcher, L.T. Gauthier, and S.W. Kennedy. 2013. In ovo effects of two organophosphate flame retardants–TCPP and TDCPP–on pipping success, development, mRNA expression, and thyroid hormone levels in chicken embryos. Toxicol. Sci. 134(1):92-102.
Feng, L., F. Ouyang, L. Liu, X. Wang, X. Wang, Y.J. Li, A. Murtha, H. Shen, J. Zhang, and J.J. Zhang. 2016. Levels of urinary metabolites of organophosphate flame retardants, TDCIPP, and TPHP, in pregnant women in Shanghai. J. Environ. Public Health 2016:9416054. doi: 10.1155/2016/9416054.
Forgacs, A.L., M.L. D’Souza, I.T. Huhtaniemi, N.A. Rah-man, and T.R. Zacharewski. 2013. Triazine herbicides and their chlorometabolites alter steroidogenesis in BLTK1 murine Leydig cells. Toxicol Sci. 134(1):155-167.
Fu, J., J. Han, B. Zhou, Z. Gong, E.M. Santos, X. Huo, W. Zheng, H. Liu, H. Yu, and C. Liu. 2013. Toxicogenomic responses of zebrafish embryos/larvae to tris(1,3-dichloro-2-propyl) phosphate (TDCPP) reveal possible molecular mechanisms of developmental toxicity. Environ. Sci. Technol. 47(18):10574-10582.
Godfrey, A., B. Hooser, A. Abdelmoneim, K.A. Horzmann, J.L. Freeman, and M.S. Sepúlveda. 2017. Thyroid disrupting effects of halogenated and next generation chemicals on the swim bladder development of zebrafish. Aquat. Toxicol. 193:228-235.
Goldenthal, E.I., D.C. Jessup, and D.E. Rodwell. 1978. Pilot teratogenicity study in rats. International Research and Development Corporation for Velsicol Chemical Corporation. Great Lakes Chemical Corporation. Submitted to the U.S. Environmental Protection Agency under TSCA Section 8D. [Secondary reference from EPA 2015b.]
Guo, J., M.H. Zhao, K.T. Shin, Y.J. Niu, Y.D. Ahn, N.H. Kim, and X.S. Cui. 2017. The possible molecular mechanisms of bisphenol-A action on porcine early embryonic development. Sci. Rep. 7(1):8632.
Hakk, H., G. Larsen, A. Bergman, and U. Orn. 2000. Metabolism, excretion and distribution of the flame retardant tetrabromobisphenol A in conventional and bile-duct cannulated rats. Xenobiotica 30(9):881-890.
Hamers, T., J.H. Kamstra, E. Sonneveld, A.J. Murk, M.H.A. Kester, P.L. Andersson, J. Legler, and A. Brouwer. 2006. In vitro profiling of the endocrine-disrupting potency of brominated flame retardants. Toxicol. Sci. 92(1):157-173.
He, C., K. English, C. Baduel, P. Thai, P. Jagals, R.S. Ware, Y. Li, X. Wang, P.D. Sly, and J.F. Mueller. 2018. Concentrations of organophosphate flame retardants and plasticizers in urine from young children in Queensland, Australia and associations with environmental and behavioural factors. Environ. Res. 164:262-270.
Hoffman, K., M. Fang, B. Horman, H.B. Patisaul, S. Garantziotis, L.S. Birnbaum, and H.M. Stapleton. 2014. Urinary tetrabromobenzoic acid (TBBA) as a biomarker of exposure to the flame retardant mixture Firemaster® 550. Environ. Health Perspect. 122(9):963-969.
Hoffman, K., L. Gearhart-Serna, M. Lorber, T.F. Webster, and H.M. Stapleton. 2017. Estimated tris(1,3-dichloro2-propyl) phosphate exposure levels for U.S. infants suggest potential health risks. Environ. Sci. Technol. Lett. 4(8):334-338.
Hoffman, K., H.M. Stapleton, A. Lorenzo, C.M. Butt, L. Adair, A.H. Herring, and J.L. Daniels. 2018. Prenatal exposure to organophosphates and associations with birthweight and gestational length. Environ. Int. 116:248-254.
IARC (International Agency for Research on Cancer). 1999a. Tris(2-chloroethyl) phosphate. Pp. 1543-1548 in IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Re-evaluation of Some Organic Chemicals, Hydrazine and Hydrogen Peroxide. Volume 71. Lyon, France: IARC [online]. Available: https://monographs.iarc.fr/wp-content/uploads/2018/06/mono71-114.pdf [accessed January 10, 2019].
IARC. 1999b. Tris(2,3-dibromopropyl) phosphate. Pp. 905-921 in IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Re-evaluation of Some Organic Chemicals, Hydrazine and Hydrogen Peroxide. Volume 71. Lyon, France: IARC [online]. Available: https://monographs.iarc.fr/wp-content/uploads/2018/06/mono71-114.pdf [accessed January 10, 2019].
IARC. 2018. Tetrabromobisphenol A. Pp. 247-289 in IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Some Industrial Chemicals, Volume 115. Lyon, France: IARC [online]. Available: https://monographs.iarc.fr/iarc-monographs-on-the-evaluation-of-carcinogenic-risks-to-humans/ [accessed December 27, 2018].
Ingle, M.E., L. Mínguez-Alarcón, C.C. Carignan, C.M. Butt, H.M. Stapleton, P.L. Williams, J.B. Ford, R. Hauser, and J.D. Meeker. 2018. The association between urinary concentrations of phosphorous-containing flame retardant metabolites and semen parameters among men from a fertility clinic. Int. J. Hyg. Environ. Health 221(5):809-815.
IPCS (International Programme on Chemical Safety). 1995a. Tris (2,3-dibromopropyl) phosphate and bis(2,3dibromopropyl) phosphate. Environmental Health Criteria 173. Geneva, Switzerland: World Health Organization [online]. Available: http://www.inchem.org/documents/ehc/ehc/ehc173.htm [accessed January 7, 2019].
IPCS. 1995b. Tetrabromobisphenol A and derivatives. Environmental Health Criteria 172. Geneva, Switzerland: World Health Organization [online]. http://www.inchem.org/documents/ehc/ehc/ehc172.htm [accessed January 7, 2019].
IPCS. 1997. Flame retardants: A general introduction. Environmental Health Criteria 192. Geneva, Switzerland: World Health Organization [online]. Available: http://www.inchem.org/documents/ehc/ehc/ehc192.htm [accessed October 5, 2018].
Jarema, K.A., D.L. Hunter, R.M. Shaffer, M. Behl, and S. Padilla. 2015. Acute and developmental behavioral effects of flame retardants and related chemicals in zebrafish. Neurotoxicol. Teratol. 52(Pt. B):194-209.
Kamishima, M., T. Hattori, G. Suzuki, H. Matsukami, C. Komine, Y. Horii, G. Watanabe, T. Oti, H. Sakamoto, T. Soga, I.S. Parhar, Y. Kondo, H. Takigami, and M. Kawaguchi. 2018. Early-life exposure to tris(1,3-dichloroisopropyl) phosphate induces dose-dependent suppression of sexual behavior in male rats. J. Appl. Toxicol. 38(5):649-655.
Kawasaki, H., T. Murai, and S. Kanoh. 1982. Studies on the toxicity of insecticides and food additives in pregnant rats. Foetal toxicity of tris-(chloropropyl) phosphate. Oyo Yakuri 24(5):697-702. [Secondary reference from ECHA. 2018. Dossier tris(2-chloro-1-methylethyl) phosphate. Available: https://echa.europa.eu/registrationdossier/-/registered-dossier/1355 (accessed December 27, 2018)].
Kawashima, K., S. Tanaka, S. Nakaura, S. Nagao, T. Endo, K. Onoda, A. Takanaka, and Y. Omori. 1983. [Effect of oral administration of tris(2-chloroethyl) phosphate to pregnant rats on prenatal and postnatal development]. Eisei Shikenjo Hokoku (101):55-61. [Secondary reference from EPA 2009.]
Kiciński, M., M.K. Viaene, E. Den Hond, G. Schoeters, A. Covaci, A.C. Dirtu, V. Nelen, L. Bruckers, K. Croes, I. Sioen, W. Baeyens, N. Van Larebeke, and T.S. Nawrot. 2012. Neurobehavioral function and low-level exposure to brominated flame retardants in adolescents: a cross-sectional study. Environ. Health. 11:86.
Kim, U.J., and J.E. Oh. 2014. Tetrabromobisphenol A and hexabromocyclododecane flame retardants in infant-mother paired serum samples, and their relationships with thyroid hormones and environmental factors. Environ. Pollut. 184:193-200.
Kitamura, S., T. Suzuki, S. Sanoh, R. Kohta, N. Jinno, K. Sugihara, S. Yoshihara, N. Fujimoto, H. Watanabe, and S. Ohta. 2005. Comparative study of the endocrine-disrupting activity of bisphenol A and 19 related compounds. Toxicol. Sci. 84(2):249-259.
Kleinstreuer, N.C., A. Karmaus, K. Mansouri, D.G. Allen, J.M. Fitzpatrick, and G. Patlewicz. 2018. Predictive models for acute oral systemic toxicity: A workshop to bridge the gap from research to regulation. Comput. Toxicol. 8(11):21-24.
Kuester, R.K., A.M. Sólyom, V.P. Rodriguez, and I.G. Sipes. 2007. The effects of dose, route and repeated dosing on the disposition and kinetics of tetraboromobisphenol A in male F-344 rats. Toxciol. Sci. 96(2):237-245.
Lai, D.Y., S. Kacew, and W. Dekant. 2015. Tetrabromobisphenol A (TBBPA): Possible modes of action of toxicity and carcinogenicity in rodents. Food Chem. Toxicol. 80:206-214.
Li, J., M. Ma, and Z. Wang. 2010. In vitro profiling of endocrine disrupting effects of phenols. Toxicol. In Vitro 24(1):201-207.
Li, R., L. Zhang, Q. Shi, Y. Guo, W. Zhang, and B. Zhou. 2018. A protective role of autophagy in TDCIPP-induced developmental neurotoxicity in zebrafish larvae. Aquat. Toxicol.199:46-54.
Lieschke, G.J., and P.D. Currie. 2007. Animal models of human disease: zebrafish swim into view. Nat. Rev. Genet. 8(5):353-367.
Lilienthal, H., C.M. Verwer, L.T. van der Ven, A.H. Piersma, and J.G. Vos. 2008. Exposure to tetrabromobisphenol A (TBBPA) in Wistar rats: Neurobehavioral effects in offspring from a one-generation reproduction study. Toxicology 246(1):45-54.
Lipscomb, S.T., M.M. McClelland, M. MacDonald, A. Cardenas, K.A. Anderson, and M.L. Kile. 2017. Cross-sectional study of social behaviors in preschool children and exposure to flame retardants. Environ. Health 16(1):23.
Litton Bionetics, Inc. 1977. Mutagenicity Evaluation of MC984 in the Rat Dominant Lethal Assay Final Report. EPA Document No. 8EHQ-0278-0107, Fiche No. OTS0200498. [Secondary reference from PubChem, https://pubchem.ncbi.nlm.nih.gov.]
Mansouri, K., C.M. Grulke, A.M. Richard, R.S. Judson, and A.J. Williams. 2016a. An automated curation procedure for addressing chemical errors and inconsistencies in public datasets used in QSAR modelling. SAR QSAR Environ. Res. 27(11):939-965.
Mansouri, K., A. Abdelaziz, A. Rybacka, A. Roncaglioni, A. Tropsha, A. Varnek, A. Zakharov, A. Worth, A.M. Richard, C.M. Grulke, D. Trisciuzzi, D. Fourches, D. Horvath, E. Benfenati, E. Muratov, E.B. Wedebye, F. Grisoni, G.F. Mangiatordi, G.M. Incisivo, H. Hong, H.W. Ng,
I.V. Tetko, I. Balabin, J. Kancherla, J. Shen, J. Burton, M. Nicklaus, M. Cassotti, N.G. Nikolov, O. Nicolotti, P.L. Andersson, Q. Zang, R. Politi, R.D. Beger, R. Todeschini, R. Huang, S. Farag, S.A. Rosenberg, S. Slavov, X. Hu, and R.S. Judson. 2016b. CERAPP: Collaborative Estrogen Receptor Activity Prediction Project. Environ. Health Perspect. 24(7):1023-1033.
Mansouri, K., C.M. Grulke, R.S. Judson, and A.J. Williams. 2018a. OPERA models for predicting physicochemical properties and environmental fate endpoints. J. Cheminform. 10(1):10.
Mansouri, K., C.M. Grulke, R.S. Judson, and A.J. Williams. 2018b. Virtual screening of chemicals for endocrine disrupting activity through CERAPP and CoMPARA projects. Poster presented at Society of Toxicology Annual Meeting, San Antonio, TX, March 11-15, 2018. doi: 10.13140/RG.2.2.19612.80009.
McGee, S.P., E.M. Cooper, H.M. Stapleton, and D.C. Volz. 2012. Early zebrafish embryogenesis is susceptible to developmental TDCPP exposure. Environ. Health Perspect. 120(11):1585-1591.
Meeker, J.D., and H.M. Stapleton. 2010. House dust concentrations of organophosphate flame retardants in relation to hormone levels and semen quality parameters. Environ. Health Perspect. 118(3):318-323.
Meeker, J.D., E.M. Cooper, H.M. Stapleton, and R. Hauser. 2013. Exploratory analysis of urinary metabolites of phosphorus-containing flame retardants in relation to markers of male reproductive health. Endocr. Disruptors (Austin). 1(1):e26306.
Meerts, I.A., J.J. van Zanden, E.A. Luijks, I. van Leeuwen-Bol, G. Marsh, E. Jakobsson, A. Bergman, and A. Brouwer. 2000. Potent competitive interactions of some brominated flame retardants and related compounds with human transthyretin in vitro. Toxicol. Sci. 56(1):95-104.
Messerlian, C., P.L. Williams, L. Mínguez-Alarcón, C.C. Carignan, J.B. Ford, C.M. Butt, J.D. Meeker, H.M. Stapleton, I. Souter, and R. Hauser. 2018. Organophosphate flame-retardant metabolite concentrations and pregnancy loss among women conceiving with assisted reproductive technology. Fertil. Steril. 110(6):1137-1144.
Moser, V.C., P.M. Phillips, J.M. Hedge, and K.L. McDaniel. 2015. Neurotoxicological and thyroid evaluations of rats developmentally exposed to tris(1,3-dichloro-2-propyl) phosphate (TDCIPP) and tris(2-chloro-2-ethyl)phosphate (TCEP). Neurotoxicol. Teratol. 52(Pt. B):236-247.
MPI Research. 2001. An oral prenatal developmental toxicity study with tetrabromobisphenol A in rats. Initial submission: Ltr from ACC to USEPA Submitting health effects & physical chemistry studies of tetrabromobisphenol A, with attachments & dated 121101. Prepared by MPI Research for American Chemistry Council. Submitted to the U.S. Environmental Protection Agency under TSCA Section 8E. [Secondary reference from EPA 2015b.]
NASEM (National Academies of Sciences, Engineering, and Medicine). 2017. Using 21st Century Science to Improve Risk-Related Evaluations. Washington, DC: The National Academies Press.
Noda, T., S. Morita, and S. Ohgaki, et al. 1985. Safety evaluation of chemicals for use in household products (VII) teratological studies on tetrabromobisphenol-A in rats. Annual report of the Osaka Institute of Public Health and Environmental Sciences 48, 106-112.NOTOX (2000) Determination of the water solubility of tetrabromobisphenol A. Hertogenbosch: NOTOX B. V. [Secondary reference from EPA 2015a.]
Noyes, P.D., D.E. Haggard, G.D. Gonnerman, and R.L. Tanguay. 2015. Advanced morphological - behavioral test platform reveals neurodevelopmental defects in embryonic zebrafish exposed to comprehensive suite of halogenated and organophosphate flame retardants. Toxicol. Sci. 145(1):177-195.
NRC (National Research Council). 2014. A Framework to Guide Selection of Chemical Alternatives. Washington, DC: The National Academies Press.
NTP (National Toxicology Program). 1991. Toxicology and carcinogenesis studies of tris(2-chloroethyl) phosphate (CAS No. 115-96-8) in F344/N rats and B6C3F1 mice (gavage studies). NTP TR 391, NIH Publication No. 91-2846. Research Triangle Park, NC: National Toxicology Program, National Institutes of Health, Public Health Service, US Department of Health and Human Services [online]. Available: https://ntp.niehs.nih.gov/ntp/htdocs/lt_rpts/tr391.pdf [accessed January 3, 2019.]
NTP. 2014. NTP technical report on the toxicology studies of tetrabromobisphenol A (CAS No. 79-94-7) in F344/NTac rats and B6C3F1/N mice and toxicology and carcinogenesis studies of tetrabromobisphenol A in Wistar Han [Crl:WI(Han)] rats and B6C3F1/N mice (gavage studies). NTP TR 587, NIH Publication No. 14-5929. Research Triangle Park, NC: National Toxicology Program, National Institutes of Health, Public Health Service, US Department of Health and Human Services [online]. Available: https://ntp.niehs.nih.gov/ntp/htdocs/lt_rpts/tr587_508.pdf [accessed December 27, 2018].
NTP. 2016. Tris(2,3-dibromopropyl) phosphate (CAS No. 126-72-7). Report on Carcinogens, Fourteenth Edition. Research Triangle Park, NC: US Department of Health and Human Services., Public Health Service [online]. Available: https://ntp.niehs.nih.gov/pubhealth/roc/index-1.html [accessed January 3, 2019].
NTP. 2017. NTP technical report on the toxicity studies of tetrabromobisphenol A-bis(2,3-dibromopropyl ether) (CAS No. 21850-44-2) administered by gavage to F344/NTac rats and B6C3F1/N mice. NTP Toxicity Report Series, No. 85. Research Triangle Park, NC: National Toxicology Program, National Institutes of Health, Pub-
lic Health Service, US Department of Health and Human Services [online]. Available: https://ntp.niehs.nih.gov/ntp/htdocs/st_rpts/tox085_508.pdf [accessed December 27, 2018].
Ohta, R., A. Takagi, H. Ohmukai, H. Marumo, A. Ono, Y. Matsushima, T. Inoue, H. Ono, and J. Kanno. 2012. Ovariectomized mouse uterotrophic assay of 36 chemicals. J. Toxicol. Sci. 37(5):879-889.
Oliveri, A.N., J.M. Bailey, and E.D. Levin. 2015. Developmental exposure to organophosphate flame retardants causes behavioral effects in larval and adult zebrafish. Neurotoxicol. Teratol. 52(Pt. B):220-227.
Oliveri, A.N., E. Ortiz, and E.D. Levin. 2018. Developmental exposure to an organophosphate flame retardant alters later behavioral responses to dopamine antagonism in zebrafish larvae. Neurotoxicol. Teratol. 67:25-30.
Ospina, M., N.K. Jayatilaka, L.Y. Wong, P. Restrepo, and A.M. Calafat. 2018. Exposure to organophosphate flame retardant chemicals in the U.S. general population: Data from the 2013-2014 National Health and Nutrition Examination Survey. Environ. Int. 110:32-41.
Peters, A.K., S. Nijmeijer, K. Gradin, M. Backlund, A. Bergman, L. Poellinger, M.S. Denison, and M. Van den Berg. 2006. Interactions of polybrominated diphenyl ethers with the aryl hydrocarbon receptor pathway. Toxicol. Sci. 92(1):133-142.
Phillips, A.L., S.C. Hammel, K. Hoffman, A.M. Lorenzo, A. Chen, T.F. Webster, and H.M. Stapleton. 2018. Children’s residential exposure to organophosphate ester flame retardants and plasticizers: Investigating exposure pathways in the TESIE study. Environ. Int. 116:176-185.
Planchart, A., C.J. Mattingly, D. Allen, P. Ceger, W. Casey, D. Hinton, J. Kanungo, S.W. Kullman, T. Tal, M. Bondesson, S.M Burgess, C. Sullivan, C. Kim, M. Behl, S. Padilla, D.M. Reif, R.L. Tanguay, and J. Hamm. 2016. Advancing toxicology research using in vivo high throughput toxicology with small fish models. ALTEX 33(4):435-452.
Preston, E.V., M.D. McClean, B. Claus Henn, H.M. Stapleton, L.E. Braverman, E.N. Pearce, C.M. Makey, and T.F. Webster. 2017. Associations between urinary diphenyl phosphate and thyroid function. Environ. Int. 101:158-164.
Reif, D.M., L. Truong , D. Mandrell, S. Marvel, G. Zhang, and R.L. Tanguay. 2016. High-throughput characterization of chemical-associated embryonic behavioral changes predicts teratogenic outcomes. Arch. Toxicol. 90(6):1459-1470.
Reznik, G., H.M. Reznik-Schüller, J.M. Rice, and B.F. Hague, Jr. 1981. Pathogenesis of toxic and neoplastic renal lesions induced by the flame retardant tris(2,3-dibromopropyl)phosphate in F344 rats, and development of colonic adenomas after prolonged oral administration. Lab. Invest. 44(1):74-83.
Richard, A.M. 2004. DSSTox website launch: improving public access to databases for building structure-toxicity prediction models. Preclinica 2:103-108.
Richard, A.M., L.S. Gold, and M.C. Nicklaus. 2006. Chemical structure indexing of toxicity data on the internet: moving toward a flat world. Curr. Opin. Drug Discov. Devel. 9(3):314-325.
Richard, A.M., R.S. Judson, K.A. Houck, C.M. Grulke, P. Volarath, I. Thillainadarajah, C. Yang, J. Rathman, M.T. Martin, J.F. Wambaugh, T.B. Knudsen, J. Kancherla, K. Mansouri, G. Patlewicz, A.J. Williams, S.B. Little, K.M. Crofton, and R.S. Thomas. 2016. ToxCast chemical landscape: Paving the road to 21st century toxicology. Chem. Res. Toxicol. 29(8):1225-1251.
Riu, A., M. Grimaldi, A. le Maire, G. Bey, K. Phillips, A. Boulahtouf, E. Perdu, D. Zalko, W. Bourguet, and P. Balaguer. 2011. Peroxisome proliferator-activated receptor γ is a target for halogenated analogs of bisphenol A. Environ. Health Perspect. 119(9):1227-1232.
Romano, M.E., N.L. Hawley, M. Eliot, A.M. Calafat, N.K. Jayatilaka, K. Kelsey, S. McGarvey, M.G. Phipps, D.A. Savitz, E.F. Werner, and J.M. Braun. 2017. Variability and predictors of urinary concentrations of organophosphate flame retardant metabolites among pregnant women in Rhode Island. Environ. Health 16(1):40.
Saegusa, Y., H. Fujimoto, G.H. Woo, K. Inoue, M. Takahashi, K. Mitsumori, M. Hirose, A. Nishikawa, and M. Shibutani. 2009. Developmental toxicity of brominated flame retardants, tetrabromobisphenol A and 1,2,5,6,9,10-hexabromocyclododecane, in rat offspring after maternal exposure from mid-gestation through lactation. Reprod. Toxicol. 28(4):456-467. [Secondary reference from EPA 2015b.]
Sanders, J.M., S.J. Coulter, G.A. Knudsen, J.K. Dunnick, G.E. Kissling, and L.S. Birnbaum, 2016. Disruption of estrogen homeostasis as a mechanism for uterine toxicity in Wistar Han rats treated with tetrabromobisphenol A. Toxicol. Appl. Pharmacol. 298:31-39.
Sarles, J.C., B. Consentino, R. Léandri, A.M. Dor, and P.H. Navarro. 1987. Mixed familial polyposis syndromes. Int. J. Colorectal Dis. 2(2):96-99.
Stauffer Chemical Co. 1978. Teratology study in rats: FR-2 (Fyrol). (Unpublished report). [Secondary reference from Environment Canada and Health Canada 2016].
Stauffer Chemical Co. 1981. A two-year oral toxicity/carcinogenicity study of Fyrol FR-2 in rats (volume I-IV) (final reports) with attachments, cover sheets and letter dated 093081. USEPA TSCAT document OTS0204911. [Secondary reference from Environment Canada and Health Canada 2016.]
Steinbeck, C., Y. Han, S. Kuhn, O. Horlacher, E. Luttmann, and E. Willighagen. 2003. The Chemistry Development Kit (CDK): An open-source Java library for chemo- and bioinformatics. J. Chem. Inf. Comput. Sci. 43:493-500.
Steinbeck, C., C. Hoppe, S. Kuhn, M. Floris, R. Guha, and E.L. Willighagen. 2006. Recent developments of the chemistry development kit (CDK) - an open-source java library for chemo- and bioinformatics. Curr. Pharm. Des. 12(17):2111-2120.
Sun, H., O.X. Shen, X.R, Wang, L. Zhou, S.Q. Zhen, and X.D. Chen. 2009. Anti-thyroid hormone activity of bisphenol A, tetrabromobisphenol A and tetrachlorobisphenol A in an improved reporter gene assay. Toxicol. In Vitro 23(5):950-954.
Sun, L., W. Xu, T. Peng, H. Chen, L. Ren, H. Tan, D. Xiao, H. Qian, and Z. Fu. 2016. Developmental exposure of zebrafish larvae to organophosphate flame retardants causes neurotoxicity. Neurotoxicol. Teratol. 55:16-22.
Tada, Y., T. Fujitani, N. Yano, H. Takahashi, K. Yuzawa, H. Ando, Y. Kubo, A. Nagasawa, A. Ogata, and H. Kamimura. 2006. Effects of tetrabromobisphenol A, a brominated flame retardant, in ICR mice after prenatal and postnatal exposure. Food Chem. Toxicol. 44(8):1408-1413. [Secondary reference from EPA 2015a.]
Takada, K., K. Yasuhara, Y. Nakaji, H. Yoshimoto, J. Momma, Y. Kurokawa, Y. Aida, and M. Tobe. 1989. Carcinogenicity study of tris(2-chloroethyl) phosphate in ddY mice. J. Toxicol. Pathol. 2(2):213-222. [Secondary reference from EPA 2009.]
Takada, K., K. Naito, K. Kobayashi, M. Tobe, Y. Kurokawa, and M. Fukuoka. 1991. Carcinogenic effect of bis(2,3-dibromopropyl) phosphate in Wistar rats. J. Appl. Toxicol. 11(5): 323-331. [Secondary reference in IPCS 1995a.]
Tanaka, S., S. Nakaura, K. Kawashima, S. Nagao, T. Endo, K.I. Onoda, Y. Kasuya, and Y. Omori. 1981. [Effect of oral administration of tris(1,3-dichloroisopropyl) phosphate to pregnant rats on prenatal and postnatal developments]. Eisei Shikenjo Hokoku 99:50-55. [Secondary reference from Environment Canada and Health Canada 2016].
TERA (Toxicology Excellence for Risk Assessment). 2016. Flame retardant exposure assessment database. Conducted in conjunction with Life Line Group under a contract with the US Consumer Project Safety Commission, Task Order 15, Contract Number CPSC-D-12-0001 [online]. Available: https://www.cpsc.gov/s3fs-public/FR-exposure-assessment-contractor-report-18-09282016-with-cover.pdf?miFGGrplONrzWVCDOIQSOY1lTeGzQvWi [accessed September 25, 2018.]
TNO Quality of Life. 2007. Oral two-generation reproduction toxicity study (including a dose range finding study) with Tris(2-chloro-1-methylethyl) -phosphate in rats (unpublished report). [Secondary reference from Environment Canada and Health Canada 2016].
Truong, L., D.M. Reif, L. St. Mary, M.C. Geier, H.D. Truong, and R.L. Tanguay. 2014. Multidimensional in vivo hazard assessment using zebrafish. Toxicol. Sci. 137(1):212-233.
van Beerendonk, G.J., S.D. Nelson, and J.H. Meerman. 1994. Metabolism and genotoxicity of the halogenated alkyl compound tris(2,3-dibromopropyl)phosphate. Hum. Exp. Toxicol. 13(12):861-865.
Van der Ven, L.T., T. Van de Kuil, A. Verhoef, C.M. Verwer, H. Lilienthal, P.E. Leonards, U.M. Schauer, R.F. Cantón, S. Litens, F.H. De Jong, T.J. Visser, W. Dekant, N. Stern, H. Håkansson, W. Slob, M. Van den Berg, J.G. Vos, and A.H. Piersma. 2008. Endocrine effects of tetrabromobisphenol-A (TBBPA) in Wistar rats as tested in a one-generation reproduction study and a subacute toxicity study. Toxicology 245(1-2):76-89.
Velsicol Chemical Corporation. 1978. To determine the rates of absorption, distribution and excretion of tetrabromobisphenol A in rats (unpublished). [Secondary reference from ECB 2006.]
Wang, Q., J.C. Lam, Y.C. Man, N.L. Lai, K.Y. Kwok, Y. Guo, P.K. Lam, and B. Zhou. 2015a. Bioconcentration, metabolism and neurotoxicity of the organophorous flame retardant 1,3-dichloro 2-propyl phosphate (TDCPP) to zebrafish. Aquat. Toxicol. 158:108-115.
Wang, Q., J.C. Lam, J. Han, X. Wang, Y. Guo, P.K. Lam, and B. Zhou. 2015b. Developmental exposure to the organophosphorus flame retardant tris(1,3-dichloro-2-propyl) phosphate: estrogenic activity, endocrine disruption and reproductive effects on zebrafish. Aquat. Toxicol. 160:163-171.
Wang, Q., N.L. Lai, X. Wang, Y. Guo, P.K. Lam, J.C. Lam, and B. Zhou. 2015c. Bioconcentration and transfer of the organophorous flame retardant 1,3-dichloro-2-propyl phosphate causes thyroid endocrine disruption and developmental neurotoxicity in zebrafish larvae. Environ. Sci. Technol. 49(8):5123-5132.
Wang, G., H. Chen, Z. Du, J. Li, Z. Wang, and S. Gao. 2017. In vivo metabolism of organophosphate flame retardants and distribution of their main metabolites in adult zebrafish. Sci. Total Environ. 590-591:50-59.
Wang, Y., W. Li, M.P. Martínez-Moral, H. Sun, and K. Kan-nan. 2019. Metabolites of organophosphate esters in urine from the United States: Concentrations, temporal variability, and exposure assessment. Environ. Int. 122:213-221.
Wei, B., M.L. Goniewicz, R.J. O’Connor, M.J. Travers, and A.J. Hyland. 2018. Urinary metabolite levels of flame retardants in electronic cigarette users: A study using the data from NHANES 2013-2014. Int. J. Environ. Res. Public Health 15(2):201.
Williams, A.J., C.M. Grulke, J. Edwards, A.D. McEachran, K. Mansouri, N.C. Baker, G. Patlewicz, I. Shah, J.F. Wambaugh, R.S. Judson, and A.M. Richard. 2017. The CompTox Chemistry Dashboard: a community data resource for environmental chemistry. J. Cheminform. 9(1):61.
Wu, Y., G. Su, S. Tang, W. Liu, Z. Ma, X. Zheng, H. Liu, and H. Yu. 2017. The combination of in silico and in vivo approaches for the investigation of disrupting effects of tris (2-chloroethyl) phosphate (TCEP) toward core receptors of zebrafish. Chemosphere 168:122-130.
Xu, T., P. Li, S. Wu, L. Lei, and D. He. 2016. Tris(2-chloroethyl) phosphate (TCEP) and tris (2-chloropropyl) phosphate (TCPP) induce locomotor deficits and dopaminergic degeneration in Caenorhabditis elegans. Toxicol. Res. (Camb.) 6(1):63-71.
Yang, C., A. Tarkhov, J. Marusczyk, B. Bienfait, J. Gasteiger, T. Kleinoder, T. Magdziarz, O. Sacher, C.H. Schwab, J. Schwoebel, L. Terfloth, K. Arvidson, A. Richard, A. Worth, and J. Rathman. 2015. New publicly available chemical query language, CSRML, to support chemotype representations for application to data mining and modeling. J. Chem. Inf. Model 55(3):510-528.
Yu, L., Y. Jia, G. Su, Y. Sun, R.J. Letcher, J.P. Giesy, H. Yu, Z. Han, and C. Liu. 2017. Parental transfer of tris(1,3dichloro-2-propyl) phosphate and transgenerational inhibition of growth of zebrafish exposed to environmentally relevant concentrations. Environ. Pollut. 220(Pt. A):196-203.
Zhang, Q., C. Ji, X. Yin, L. Yan, M. Lu, and M. Zhao. 2016. Thyroid hormone-disrupting activity and ecological risk assessment of phosphorus-containing flame retardants by in vitro, in vivo and in silico approaches. Environ. Pollut. 210:27-33.
Zhu, Y., D. Lin, D. Yang, Y. Jia, and C. Liu. 2018. Environmentally relevant concentrations of the flame retardant tris(1,3-dichloro-2-propyl) phosphate change morphology of female zebrafish. Chemosphere 212:358-364.