3
Materials and Processes
As described in chapter 2, the primary purposes of high-temperature structural coatings are to enable high temperature components to operate at even higher temperatures, to improve component durability, and to allow use of a broader variety of fuels in land-based and marine-based engines. Although high-temperature coatings protect the substrate, the demarcation between coating and substrate (either metal or nonmetal) is becoming increasingly blurred. The demanding requirements of high-temperature service in both isothermal and cyclic modes have recast the way researchers think about coated structures. These structures can be considered part of a continuum; at the limit the coating will be a progressive modification of the substrate and therefore must be concurrently designed with the substrate. Table 3-1 summarizes the relationship between coating functions and coating characteristics.
There are essentially two types of high-temperature coatings. The first type is a diffusion (or conversion) coating in which the deposited mass is diffused and/or reacted with the substrate to form a somewhat continuous gradation in composition. The second type is an overlay coating in which material is deposited at the surface of the substrate. This chapter discusses current and potential coatings for superalloys, ceramics, refractory metals, intermetallic materials, and metal-matrix composites. The processes used to apply these coatings to substrate materials are also reviewed.
TABLE 3-1 Coating Functions and Coating Materials Characteristics
|
Functions |
Materials Characteristics |
|
Reduction in surface temperature |
Low thermal conduction Low radiative heat transfer High emittance |
|
Reduction in rate of oxidation |
Thermodynamically stable oxide formers with slow growth rates |
|
Reduction in rate of hot corrosion |
Chemically stable and impervious oxide scale |
|
Resistance to particulate erosion |
Hard, dense material |
|
Increased abradability (sacrificial wear) |
Rub tolerance via plastic deformation (densification) Energy transformation via fracture (material loss) |
|
Increased abrasiveness |
Inclusion of hard particles to induce cutting of seal |
COATINGS FOR HIGH-TEMPERATURE STRUCTURES
Coatings must maintain their performance on a continued and reliable basis, or the performance of the turbine system could be compromised. Advanced coatings that show excessive variability and unpredictability at the required engine operating conditions will not be employed, regardless of their potential benefits to the system. For example, thermal barrier coatings (TBCs) are currently used to improve the performance of several engines, including GE's CF6-80C2 and CFM56-5a and Pratt & Whitney's PW2000 and PW4000 series engines (Bose and DeMasi-Marcin, 1995). TBCs are currently relied on for thermal insulation to improve component durability at elevated temperatures (i.e., 40°C [72°F] higher than uncoated parts) but still below the incipient melting temperature of the substrate material. In addition, TBC patches are used to minimize thermomechanical fatigue cracking by reducing substrate temperatures at component hot spots. However, TBCs will not be widely accepted for use at temperatures that will result in the rapid degradation of substrate materials in order to provide the performance benefits of which they are capable until they can be produced with predictable, reliable properties, as discussed in chapter 1 (see figure 1-1).
The majority of hot-section components are currently made from superalloys. To achieve the aggressive performance goals of the advanced engine programs (e.g., High-Speed Civil Transport, Integrated High-Performance Turbine Engine Technology, and Advanced Turbine Systems programs) discussed in chapter 2, material systems with higher inherent temperature capabilities and better oxidation and corrosion resistance are required. These systems may require the use of advanced substrates, such as intermetallic compounds, ceramics, or refractory metals. TBCs deposited on current alloys may allow components to operate at higher temperatures.
TABLE 3-2 Types of Coatings Used in Hot-Section Components
Coatings for Superalloys
In the past, coatings for superalloys formed a metallic-aluminide layer on the gas-path surfaces of high-pressure turbine airfoils. As alloys with improved temperature and cyclic capability were synthesized, suitable coatings were developed in parallel. These more advanced coatings were often tailored for expected environmental conditions.
Metallic coatings (i.e., aluminide, chromide, and MCrAlY) protect superalloys against aggressive environmental factors. Ceramic oxide coatings insulate the substrate from the maximum gas-path temperatures. Table 3-2 provides an overview of the types of coatings currently used for hot-section structures. Basic service use and application process information for each main coating type is summarized in table 3-3. In figure 3-1, Novak (1994) schematically depicts the tradeoffs in selecting coating compositions for an environment that requires resistance to both oxidation and corrosion.
In most applications, TBCs contain yttria-stabilized zirconia. TBCs are applied to an oxidation-resistant bondcoat, typically a MCrAlY designed for high-temperature oxidation resistance or an aluminide modified by a platinum addition (Smith and Boone, 1990). The platinum in an aluminide diffusion coating has been shown to improve the protective properties of the alumina scale by (1) increasing the purity of the alumina scale by prohibiting the diffusion of the refractory metals present in the substrate (e.g., tungsten and molybdenum) to the oxide scale; (2) increasing the diffusivity of aluminum and promoting alumina formation; (3) improving the selective oxidation of aluminum; and (4) decreasing the activity coefficient of aluminum in the coating. The bondcoat acts to retard a principal cause of TBC failure: oxidation of the interface between the coating and the substrate. However, alloys currently under development have shown improved oxidation resistance and may not require a bondcoat (Miller and Brindley, 1992; Ulion and Anderson, 1993). Furthermore, because of its additional weight, the bondcoat can be detrimental to the creep life of the component, particularly in the case of rotating parts. Research indicates that adherent, durable, plasma-sprayed zirconia-yttria TBCs can be deposited onto smooth substrates (without a metallic bondcoat) if an initial layer of the TBC is applied by low-pressure plasma spraying (Miller and Brindley, 1992). Current familiarity with TBCs will lead to improved design criteria and manufacturing experience that will allow continual improvement and additional benefits.
Coatings for Ceramics
Current approaches for coating the silicon-based ceramic materials include the use of mullite coatings that have an excellent expansion match with silicon carbide (Van Roode et al., 1992; Lee et al., 1994). While mullite contains silica, the aluminum in mullite promotes formation of a solid rather than a liquid oxidation product. This cuts the weight loss in half for
TABLE 3-3 Generic Information on Coating Types Used in Superalloy Hot-Section Components
|
|
Coating Type |
|||
|
Information |
Aluminide |
Chromide |
MCrAlY |
Ceramic Oxide |
|
Primary Protection Function |
Oxidation |
Hot corrosion |
Oxidation |
Thermal barrier |
|
Thickness (µm) |
||||
|
Nominal |
65 |
25 |
125 |
250 |
|
Range |
25-100 |
25-50 |
125-500 |
> 125 |
|
Service Temperature |
||||
|
°C |
815-1150 |
600-925 |
815-1150 |
980-1200 |
|
°F |
1500-2100 |
1100-1700 |
1500-2100 |
1800-2200 |
|
Application Process |
Vapor phase |
Vapor phase |
Thermal spray |
Thermal spray |
|
|
reaction |
reaction |
|
|
|
|
|
|
air/vacuum |
air/vacuum |
|
|
pack cementation |
pack cementation |
plasma |
plasma |
|
|
above the pack |
above the pack |
combustion |
|
|
|
chemical vapor |
|
|
Vapor deposition |
|
|
deposition |
|
Vapor deposition |
electron beam |
|
|
slurry |
|
electron beam |
sputtering |
|
|
|
|
sputtering |
|
|
|
|
|
Laser deposition |
|
|
|
|
|
powder cladding |
|
|
|
|
|
Reaction sinteringa |
|
|
|
|
|
controlled |
|
|
|
|
|
composition |
|
|
Relative Cost |
1x |
1x |
2x-4x |
3x-5x |
|
Commercial Availability |
Widely available |
Widely available |
Widely available |
Selectively available |
|
a Hsu et al. (1979). |
||||
silicon carbide materials under active oxidation conditions Multilayer coatings that incorporate mullite are currently under investigation (K. Lee, personal communication, 1996)
Coatings for Refractory Metals
Efforts to develop coatings for refractory metals have been hampered by their high reactivity. The aluminides (Stoloff and Sims, 1986) and silicides (Mueller et al., 1991) have produced coatings that reduce environmental-induced degradation. A new (molybdenum/tungsten)(silicon/germanium)2 diffusion coating on niobium has shown excellent cyclic oxidation resistance (Mueller et al., 1991) but has yet to be commercially developed. Use of even excellent coatings is unlikely, however, since flaws in the coating could severely curtail component life.
Coatings for Intermetallic Materials
In coating both the low-and high-aluminum-content titanium aluminides (i.e., Ti3Al and TiAl), researchers may have
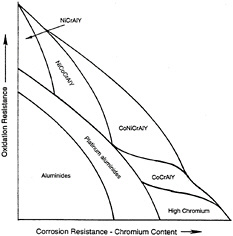
Figure 3-1 Coating compositions as related to oxidation and corrosion resistance. Source: Novak (1994).
to contend with coating/component interdiffusion and the potential for formation of brittle intermetallic compounds at the coating/substrate interface (Brindley et al., 1992; McKee, 1993). The high reactivity of the titanium-base alloys may require reaction barriers between the coating and the substrate to reduce the possibility of degradation (McKee, 1993). Flaws in either the coating or the reaction barrier could severely curtail component life.
The mechanical properties of nickel aluminide (NiAl) substrates are sensitive to compositional changes. Thus, the potential for diffusion between the coating and the component must be considered in the use of this alloy in the coated condition.
COATING PROCESSES
In general, the development of manufacturing processes for high-temperature coatings has paralleled the evolution of gas-turbine materials and component design. Early turbine blades for Allison and Curtiss Wright engines in 1952 were given a protective coating by simply dipping them into molten aluminum (Goward and Cannon, 1988). The first practical use of a diffusion coating for turbine airfoils was in 1957 (Goward and Cannon, 1988).
As interest surged during the 1970s, processes were developed for the application of overlay coatings that offered greater compositional flexibility than was possible with diffusion coatings. Most overlay coatings for gas-turbine applications are currently applied using electron-beam physical vapor deposition (EB-PVD), which is an atomistic deposition method, and low-pressure plasma spraying, which is a particulate deposition method. The benefits and limitations of the atomistic and particulate deposition methods are summarized in table 3-4.
The features, applications, advantages, and disadvantages of the coating methods most relevant to high-temperature structural materials are summarized in the remainder of this section. Appendix C cites a number of nondestructive evaluation methods that could be used for process control monitoring. Appendix E reviews examples of industrial manufacturing technologies for selected processes.
TABLE 3-4 Summary of the Benefits and Limitations of the Atomistic and Particulate Deposition Methods
|
|
Processing |
||||
|
Features |
Evaporation |
Sputtering Deposition |
CVD |
Electrodeposition |
Thermal Spraying |
|
Mechanism to produce depositing species |
Thermal energy |
Momentum transfer |
Chemical reaction |
Solution |
Flames or plasmas |
|
Deposition rate |
Moderate (up to 750,000 Å/min.) |
Low |
Moderate |
Low to high |
Very high |
|
Deposition species |
Atoms |
Atoms/ions |
Atms/ions |
Ions |
Droplets |
|
Complex shapes |
Poor line of sight |
Good but nonuniform |
Good |
Good |
Poor resolution |
|
Deposits in small, blind holes |
Poor |
Poor |
Limited |
Limited |
Very limited |
|
Metal/alloy deposition |
Yes |
Yes |
Yes |
Yes |
Yes |
|
Refractory compounds and ceramics |
Yes |
Yes |
Yes |
Limited |
Yes |
|
Energy of deposit-species |
Low |
Can be high |
Can be high |
Can be high |
Can be high |
|
Growth interface perturbation |
Not normally |
Yes |
Yes |
No |
No |
|
Substrate heating |
Yes, normally |
Not generally |
Yes |
No |
Not normally |
Physical Vapor Deposition
The physical vapor deposition process is an atomistic deposition method that involves the vaporization and subsequent deposition of coating species. It has the advantage of being able to deposit coatings of metal, alloys, and ceramics on most materials and a wide range of shapes. Because application requires clear line-of-sight, complete coating coverage is achieved by manipulating the part during the coating cycle with a complex mechanical system.
Electron-beam guns for EB-PVD are favored for supplying the energy necessary for evaporation because they can achieve higher energy densities than other methods of heating. EB-PVD can successfully deposit mixed oxide coatings that are currently of greatest benefit to high-temperature structural materials (e.g., yttria-stabilized zirconia TBCs). In this case, the process parameters are adjusted so that the deposit has a columnar structure that is perpendicular to the interface. This morphology maximizes resistance to strains that stem from differences in thermal expansion coefficients. Because TBCs are porous, they allow gases and fused condensates to penetrate and, therefore, are deposited onto an underlying bondcoat resistant to oxidation and hot corrosion (usually MCrAlY).
Most models for EB-PVD processes are primarily empirical. EB-PVD equipment is computer controlled, and regulation of the process can often be devised by trial and error. Plasma and ion control of the vapor allows formation of the desired coatings. The committee was unable to learn much of the processing details of EB-PVD because this information is largely proprietary. Basic scientific understanding of vapor formation and deposition is known for simple systems but not for the more complex, multicomponent systems that generate superalloy coatings. Particularly troublesome are components with constituents that have vastly different vapor pressures. Deposition on parts with widely varying curvatures, such as airfoils, also presents difficulties in processing. The committee is not aware of any mathematical models for these situations that are available in the public domain. Additional information about this process is summarized in appendix E.
Sputtering
The primary advantage of sputtering is its ability to deposit a wide variety of materials (e.g., alloys, oxide solutions, and intermetallics). These compositions can be derived from many types of targets. A reactive gas can be introduced with the heavy inert gas, so that reactive sputtering occurs. Often the deposit has a columnar microstructure, with elongated grains normal to the interface. Such a structure would be ideal for oxide TBCs. Sputtering is not yet a production coating method for turbine hardware because current equipment deposits the coating too slowly. Appendix E provides additional information about this process.
Thermal Spray Processes
Thermal spray processes are particulate deposition methodologies that involve the deposition of molten droplets of material on a substrate. The coating typically begins as a powder. It is then injected into a hot carrier gas and sprayed on the target substrate with a gun. This process can quickly deposit a wide variety of coatings. A shortcoming of traditional thermal spray technologies has been a limitation on the thinness and smoothness of the deposit.
A wide variety of thermal spray techniques, as discussed in appendix E, can be employed to deposit metallic corrosion and oxidation-resistant overlay coatings, bondcoats, and ceramic TBCs. Plasma spraying is the most widely used thermal spray technique for gas-turbine component overlay coating.
The microstructure of a plasma-sprayed coating depends on the starting material and its particle size distribution, as well as processing parameters. New and improved powder processing methods can produce starting materials with predictable and controllable compositions and well-delineated particle size. Key plasma-spray process parameters include plasma power, plasma gas composition, pressures and flow rates, powder injection details and carrier flow, and torch/substrate distance. These parameters may be linked in complex ways, making process control difficult. A clear goal is to achieve on-line feedback control of the process. This will require a much more detailed understanding of the process parameterization.
Diffusion Coating Methods
Diffusion coating is a surface modification process wherein the coating species is diffused into the substrate surface to form a protective layer. Diffusion coatings are the most used method for providing improved hot-corrosion and oxidation resistance for nickel-or cobalt-base superalloys. Diffusion coatings have been produced with aluminum, chromium, silicon, hafnium, zirconium, and yttrium alloys. The
aluminum, chromium, aluminum-chromium duplex, aluminum-silicon duplex, and platinum-modified aluminide coatings are the most commercially significant. The surface of the superalloy is typically modified to a depth of 0.5 to 5.0 mils (thousandths of an inch) depending on the type of coating and the process parameters selected. These methods are sometimes classified as chemical vapor deposition methods. The committee considers three primary methods for producing diffusion coatings in this report: pack cementation, out-of-pack cementation, and chemical vapor deposition. Appendix E describes these processes.
COATING PROCESS CONTROL
Many of the recent advancements in coating technology have been the result of improved process control. For example, thermal spray technology has grown from an empirical art with highly variable results to a well-controlled process that gives consistently reliable results. Reliability has been achieved through improved control of carrier gas and power, control of feedstock material, and close monitoring of key process steps.
Process modeling, process monitoring, and real-time control must become more common if the manufacture of improved coatings is to be realized. Process control can only be achieved by understanding the empirical relationship between the coating process and the resulting coating. The choice of coating process should depend on a balanced approach among the technical attributes, the coatability issues, and the performance requirements demanded from the coated component. Developing the relationship of the process to product performance must be a priority, near-term endeavor for advanced coating systems. Such parametric data, guided by an understanding of coating behavior and failure modes, can be quickly and reliably acquired through rig testing, but the variability of the rig test must also be known. Rig-test modeling, parameter sensing, and feedback control will be an evolutionary and continuing near-term activity. The NRC (1989) study of On-Line Control of Metal Processing predicted this process evolution.
The current generation of metallic coatings, as well as the emerging TBC technologies, would benefit significantly from advances in process control. Both types of coatings are deposited by the same basic processes (i.e., plasma spray and physical vapor deposition), and process improvements will enhance the performance of both coating types. Improved on-line control must be developed to help ensure that the resultant behavior of the coated structure is highly reproducible and within the performance limits needed for the service requirements. For example, reducing the large variability in TBC performance and extending the service life are priorities. Many emerging nondestructive evaluation methods have yet to be effectively exploited (Murphy et al., 1993). A representative selection of methods are described in appendix C. The most promising nondestructive evaluation methods should be further developed and applied for manufacturing process control applications.
Accurate and comprehensive in situ measurements of coating processes are often difficult and sometimes impossible, leading to the need for process modeling. For example, in the typical direct-current plasma-spray system, powder particles are injected at approximately right angles into a high-velocity gas flame. The powder carrier gas increases turbulence within the flame and also disturbs its temperature distribution. Ideally, each particle becomes entrained in the flame, rapidly increases in temperature, melts (without excessive vaporization), and impacts the substrate where rapid solidification occurs. In practice, to formulate useful models, simplifying assumptions based on the underlying physical phenomena must be made and validated.
Within the manufacturing domain, the committee believes that important data do not currently exist in an appropriate form. The conceptual Taguchi Loss Function, outlined in figure 3-2, provides a starting point for developing a conceptual framework for identifying and classifying the data needed for a robust manufacturing process. The Taguchi Loss Function is based on the ideas of statistical process control and associates a deviation from the design-specified mean value of a parameter as a deviation from the minimum-cost performance product (Taguchi, 1993). A first step in applying this concept to coatings would be to chart the distribution of the parameters thought to be important to coating performance. Their frequency of deviations from the mean could then be determined. This would guide the search for those process parameters that must be controlled (i.e., those parameters that deviated most from the mean). Once a set of parameters needing monitoring and control are identified, process sensors (NRC, 1995b) and control systems can narrow the deviations from the mean. Appendix E contains more details of this approach.
SUMMARY
The majority of hot-section coatings are applied to protect superalloy components from degradation caused by the turbine engine environment. Since current hot-section structures are produced from nickel-and cobalt-base superalloys, substrate coatings in current engines have been optimized for superalloys. These coatings are primarily designed to protect the superalloys from oxidation and hot corrosion. Most recently, TBCs are being designed to reduce the effects of high temperatures and temperature gradients. Current and future generations of superalloys have extremely complex chemistries and microstructures, carefully tailored to meet the high demand of turbine
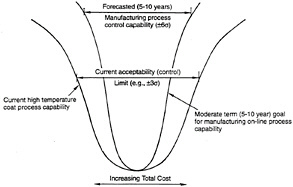
Figure 3-2 Schematic showing the benefit of development and deployment of manufacturing process control for high-temperature coatings.
engines. These complex substrate chemistries may be disturbed through interdiffusion with a coating, and their precisely crafted microstructures can be thrown into disarray as a result of coating process conditions. Therefore, coating/component combinations will have to be engineered together as a system to achieve optimum performance.
Thus the primary considerations in selecting a coating should be to protect the component and to ensure that the coating retains its protective qualities in the engine environment. Failure modes are discussed in chapter 4. Other considerations arise from various engineering factors, such as the requirement that the coating must also be compatible with the bulk material of the component under the demanding conditions in a turbine engine. These factors are further discussed in chapter 5.
Materials under development for use in future advanced components will also depend on coatings to protect against oxidation, hot corrosion, and embrittlement. Most of the new materials differ significantly from superalloys in physical, chemical, and mechanical properties. Also, many of these emerging materials, in their current form, are much less tolerant of flaws and failure in their coatings than the superalloys. Therefore, coating these materials presents significant challenges. Development of new coating materials and processes should be done concurrently with the development of the emerging materials.







