3
Effects of Noise on Marine Mammals
INTRODUCTION
Richardson et al. (1995) provided a comprehensive summary of published and gray literature data on marine mammal responses to specific noise sources. Although the literature continues to expand and many valuable new studies have appeared, most recent publications have tended to provide variations on themes rather than new data at variance with the conclusions summarized by Richardson et al.. A number of factors affect the response of marine mammals to sounds in their environment: the sound level and other properties of the sound, including its novelty; physical and behavioral state of the animal; and prevailing acoustic characteristics and ecological features of the environment in which the animal encounters the sound. Critical issues about what determines effects of and responses to intense transient sounds and what are the effects of long-term anthropogenic sound on individuals and populations remain unanswered (see Box 3-1 for the priority research areas identified by the NRC [2000]). The indirect effects of anthropogenic sound on marine mammals via effects on their predators, prey, and other critical habitat elements are largely uninvestigated.
HEARING CAPABILITIES OF MARINE ORGANISMS
Marine Mammals
Hearing research has traditionally focused on mechanisms of hearing loss in humans. Animal research has therefore emphasized experimental
|
Box 3-1 Priority Research for Whales and Seals Recommended by NRC (2000) To move beyond requiring extensive study of each sound source and each area in which it may be operated, [NRC (2000) recommended that] a coordinated plan should be developed to explore how sound characteristics affect the responses of a representative set of marine mammal species in several biological contexts (e.g., feeding, migrating, and breeding). Research should be focused on studies of representative species using standard signal types, measuring a standard set of biological parameters, based on hearing type (Ketten, 1994), taxonomic group, and behavioral ecology (at least one species per group). This could allow the development of mathematical models that predict the levels and types of noise that pose a risk of injury to marine mammals. Such models could be used to predict in multidimensional space where temporary threshold shift (TTS) is likely (a “TTS potential region”) as a threshold of potential risk and to determine measures of behavioral disruption for different species groups. Observations should include both trained and wild animals. The results of such research could provide the necessary background for future environmental impact statements, regulations, and permitting processes. Groupings of Species Estimated to Have Similar Sensitivity to Sound Research and observations should be conducted on at least one species in each of the following seven groups:
|
work on ears in other species as human analogs. Consequently, researchers have generally investigated either very basic mechanisms of hearing or induced and explored human auditory system diseases and hearing failures through these test species. Ironically, because of this emphasis, remarkably little is known about natural, habitat, and species-specific aspects of hearing in most mammals. Marine mammals represent an extreme example of not only habitat adaptations but also adaptations in ear structure and hearing capabilities.
The same reasons that make marine mammals acoustically and auditorally interesting—that is, that they are a functionally exceptional and an aquatic ear—also make them difficult research subjects. Some issues about marine mammal hearing can be addressed both directly and inferentially from the data at hand. While large gaps remain in our knowledge, progress has been made on some fronts related to sound and potential impacts from noise.
Signal Type Standardized analytic signals should be developed for testing with individuals of the preceding seven species groups. These signals should emulate the signals used for human activities in the ocean, including impulse and continuous sources.
Biological Parameters to Measure When testing representative species, several different biological parameters should be measured as a basis for future regulations and individual permitting decisions. These parameters include the following:
SOURCE: NRC, 2000. |
Marine mammals, and whales in particular, present an interesting hearing paradox. On one hand, marine mammal inner ears physically resemble land mammal inner ears, although the external ears are typically absent and the middle ear extensively modified. Since many forms of hearing loss are based in physical structure of the inner ear, it is likely hearing damage occurs by similar mechanisms in both land and marine mammal ears. On the other hand, the sea is not, nor was it ever, even primordially silent. Whales and dolphins, in particular, evolved ears that function well within this context of natural ambient noise. This may mean they developed “tough” inner ears that are less subject to hearing loss under natural ocean noise conditions. Recent anatomical and behavioral studies do indeed suggest that whales and dolphins may be more resistant than many land mammals to temporary threshold shifts (TTSs), but the data show also that they are subject to disease and aging processes. This means they are not immune to hearing loss, and certainly, increasing ambient noise via human
activities is a reasonable candidate for exacerbating or accelerating such losses.
Unfortunately, existing data are insufficient to predict accurately any but the grossest acoustic impacts on marine mammals. Little information exists to describe how marine mammals respond physically and behaviorally to intense sounds and to long-term increases in ambient noise levels.
The data available show that all marine mammals have a fundamentally mammalian ear, which through adaptation to the marine environment has developed broader hearing ranges (Figure 1-1) than are common to land mammals. Audiograms are available for only 10 species of odontocetes and 11 species of pinnipeds. All are smaller species that were tested as captive animals. However, there are 119 marine mammal species, and the majority are large, wide-ranging animals that are not approachable or testable by normal audiometric methods. Therefore, direct behavioral or physiologic hearing data for nearly 80 percent of the genera and species of concern for coastal and open-ocean sound impacts do not exist. For those species for which no direct measure or audiograms are available, hearing ranges are estimated with mathematical models based on ear anatomy obtained from stranded animals or inferred from emitted sounds and controlled acoustic exposure experiments in the wild.
The combined data from audiograms and models show there is considerable variation among marine mammals in both absolute hearing range and sensitivity. Their composite range is from ultra- to infrasonic. Odontocetes, like bats, are excellent echolocators, capable of producing, perceiving, and analyzing ultrasonic frequencies well above any human hearing. Odontocetes commonly have good functional hearing between 200 and 100,000 Hz, although some species may have functional ultrasonic hearing to nearly 200 kHz. The majority of odontocetes have peak sensitivities (best hearing) in the ultrasonic ranges, although most have moderate sensitivity to sounds from 1 to 20 kHz. No odontocete has been shown audiometrically to have acute, that is, best sensitivity or exceptionally responsive, hearing (<80 dB re 1 µPa) below 500 Hz.
Based on functional models, good lower-frequency hearing appears to be confined to larger species in both the cetaceans and pinnipeds. No mysticete has been directly tested for any hearing ability, but functional models indicate their hearing commonly extends to 20 Hz, with several species, including blue, fin, and bowhead whales, that are predicted to hear at infrasonic frequencies as low as 10–15 Hz. The upper functional range for most mysticetes has been predicted to extend to 20–30 kHz.
Most pinniped species have peak sensitivities between 1 and 20 kHz. Some species, like the harbor seal, have best sensitivities over 10 kHz. Only the northern elephant seal has been shown to have good to moderate hearing below 1 kHz (Kastak and Schusterman, 1999). Some pinniped species are considered to be effectively double-eared in that they hear moderately
well in two domains, air and water, but are not particularly acute in either. Others, however, are clearly best adapted for underwater hearing alone.
To summarize, marine mammals as a group have functional hearing ranges of 10 Hz to 200 kHz. They can be divided into infrasonic balaenids (probable functional ranges of 15 Hz to 20 kHz; good sensitivity from 20 Hz to 2 kHz); sonic to high-frequency species (100 Hz to 100 kHz; widely variable peak spectra), and ultrasonic dominant species (200 Hz to 200 kHz general sensitivity; peak spectra 16-120 kHz) (Wartzok and Ketten, 1999).
Other Marine Organisms
The inner ear of fishes and elasmobranchs (sharks and rays) is very similar to that of terrestrial vertebrates [see Popper and Fay (1999) for review]. While there are data on hearing capabilities for fewer than 100 of the 25,000 extant species, investigations of the auditory system of evolutionarily diverse species support the suggestion that hearing is widespread among virtually all fishes, as well as elasmobranchs.
Most species of fish and elasmobranchs are able to detect sounds from well below 50 Hz (some as low as 10 or 15 Hz) to upward of 500-1,000 Hz (Figure 3-1).1 Moreover, a number of fish species have adaptations in their auditory systems that enhance sound detection and enable them to detect sounds to 3 kHz and above and have better sensitivity than nonspecialist species at lower frequencies. Goldfish and American shad are examples of specialist species, while Atlantic salmon and Atlantic cod are examples of species without specializations.
There are very few data on hearing by marine invertebrates, although a number of species have highly sophisticated structures, called statocysts, that have some resemblance to the ears of fishes (Offutt, 1970; Budelmann, 1988, 1992). The statocysts found in the cephalopods (octopods and squid) may primarily serve for determination of head position in a manner similar to the components of the vertebrate ear that determine head position for vestibular senses. It is possible, but not yet demonstrated, that cephalopods use their statocysts for detection of low-frequency sounds.
There is also some evidence that a number of crustacean species, such as crabs, have statocysts that are somewhat similar to those found in cephalopods, although they have evolved separately. While there are no data for
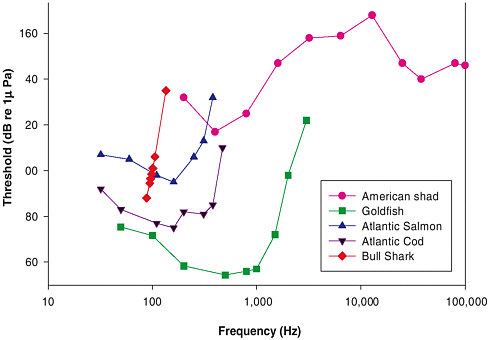
FIGURE 3-1 Fish and shark audiograms. Hearing capabilities in several fish species and a shark showing the lowest sound level that an animal can detect at each frequency. It is important to note that while thresholds here are presented in units of pressure, it is very likely that a number of species, including the sharks, respond best to particle acceleration and had experiments been done in terms of acceleration the shapes of the hearing curves might be somewhat different, though it is likely that the range of detection would not change very much. The stimuli in some of these experiments were in the near field where particle acceleration and pressure are not directly related. SOURCES: American shad: Mann et al. (1997); goldfish: Jacobs and Tavolga (1967); Atlantic salmon: Hawkins and Johnstone (1978); Atlantic cod: Chapman and Hawkins (1973); bull shark: Kritzler and Wood (1961).
hearing by marine crabs, a number of species of semiterrestrial fiddler and ghost crabs are not only able to detect sounds but also use special sounds for communication (reviewed in Popper et al., 2001). In addition, a number of physiological studies of statocysts of marine crabs suggest that some of these species are potentially capable of sound detection (Popper et al., 2001).
Marine reptiles include snakes and turtles. Although marine snakes have auditory systems similar to those of terrestrial snakes, nothing is known about their acoustic abilities. Despite considerable interest in marine turtles, since many species are endangered, very little is known about their hearing. Difficulties in developing methods to successfully train turtles to respond to acoustic stimuli have hindered research in this area. Ears of
turtles are well developed, and there is some evidence that at least a few species of marine turtles can detect sounds below 1 kHz. However, until more data are available, this value must be taken with considerable caution. Bartol et al. (1999) measured the hearing of 35 juvenile loggerhead sea turtles and the results suggested a hearing range from at least 250-750 Hz, with the most sensitive threshold recorded at the lowest frequency tested, 250 Hz. Ridgway et al. (1969) found that green turtles were most sensitive to frequencies between 300 and 400 Hz and sensitivity declined rapidly at frequencies outside of this range. There is some additional evidence from attempts at behavioral studies and from recordings of responses of the inner ear, but no data suggest higher frequencies of hearing.
ACOUSTIC TRAUMA IN MARINE MAMMALS
Recent reports and retrospectively analyzed data show an association between the use of multiple high-energy mid-range sonars and mass strandings of beaked whales (Ziphius cavirostris). Recent mass strandings of beaked whales have occurred in a temporal and spatial association with ongoing military exercises employing multiple high-energy, mid-frequency (1-10 kHz) sonars. Strandings in the Mediterranean (D’Amico and Verboom, 1998), the New Providence Channel in the Bahamas (Evans and England, 2001), and most recently in the Canary Islands (2002) have greatly increase public awareness of the issue of noise in the ocean. In addition, a retrospective review of earlier beaked whale strandings suggests that there is at least an indirect causal relationship between the strandings and the use of multiple, mid-range sonars in military exercises in some nearshore areas. Although the correlation in time between the use of sonars and the strandings is quite compelling, there is no clear demonstration as yet of any causal mechanism. Acoustic trauma is a very explicit form of injury. In the beaked whale cases to date, the traumas that were observed could result from many causes, both directly and indirectly associated with sound, or could have been from other causes. Indeed, similar traumas have been observed in terrestrial mammals under circumstances having no relation to sound exposure. Careful sampling has rarely been possible in beaked whale cases, which has made adequate diagnosis problematic. To date, only six specimens of beaked whale have been rigorously analyzed. The NATO report (D’Amico, 1998) and the joint NOAA-Navy interim report (Evans and England, 2001) have not been discussed in detail by this committee because of the preliminary nature of the findings. However, this is clearly a subject to which much additional research needs to be directed. A program should be instituted to investigate carefully the causal mechanisms that may explain the traumas observed and how the acoustics of high-energy, mid-range sonars directly or indirectly are related to them and to mass stranding events.
EFFECTS OF MARINE NOISE ON MAMMAL BEHAVIOR
Behavioral responses of marine mammals to noise are highly variable and dependent on a suite of internal and external factors. Internal factors include
-
individual hearing sensitivity, activity pattern, and motivational and behavioral state at time of exposure;
-
past exposure of the animal to the noise, which may have led to habituation or sensitization;
-
individual noise tolerance; and
-
demographic factors such as age, sex, and presence of dependent offspring.
External factors include
-
nonacoustic characteristics of the sound source, such as whether it is stationary or moving;
-
environmental factors that influence sound transmission;
-
habitat characteristics, such as being in a confined location; and
-
location, such as proximity to a shoreline.
Behavioral responses range from subtle changes in surfacing and breathing patterns, to cessation of vocalizations, to active avoidance or escape from the region of the highest sound levels.
Typical changes in cetacean response to anthropogenic noise are summarized from several studies of bowhead whales as shorter surfacings, shorter dives, fewer blows per surfacing, and longer intervals between successive blows (Richardson et al., 1995). These subtle changes are often the only observable reaction of whales to reception of anthropogenic stimuli. Although there may be statistically significant changes in some of these subtle behavioral measures, there is no evidence that these changes are biologically significant for the animals. Typical changes in vocalizations are a reduction or cessation in calling as shown in right whales in response to boats (Watkins, 1986); bowhead whales in response to playbacks of industrial sounds (Wartzok et al., 1989); sperm whales in response to short sequences of pulses from acoustic pingers (Watkins and Schevill, 1975); and sperm and pilot whales (Globicephala melaena) in response to the Heard Island Feasibility Test source (Bowles et al., 1994). Humpback whales, which appeared in all other behavioral measures to have habituated to the presence of whale-watching boats, still tended to cease vocalizations when near boats (Watkins, 1986).
Not all cetaceans respond with a decrease or cessation of calls. Sperm whales continued calling when encountering continuous pulsing from echo
sounders (Watkins, 1977) and when exposed to received sound levels of 180 dB re 1 µPa (RMS) from the discharge of a detonator (Madsen and Møhl, 2000); humpback whales moved away from low-frequency (3-kHz range) sonar pulses and sweeps but did not change their calling (Maybaum, 1993); and a fin whale continued to call with no change in rate, level, or frequency components as a container ship went from idle to full power within a kilometer of the whale (Edds, 1988). Sperm whales in the Caribbean became silent in the presence of military sonar signals (3-8-kHz range; Watkins et al., 1985).
In addition to changing the frequency of occurrence of calls in the presence of noise, some species change the source level and output frequency and duration. Beluga whales adjust their echolocation clicks to higher frequencies and to higher source levels in the presence of background noise (Au et al., 1985). Miller et al. (2000) found that humpback whales exposed to low-frequency active (LFA) sonar signals increased the duration of their songs by 29 percent on average, but with a great deal of individual variation.
Given the range of observed reactions in a variety of species, it is likely that a sound that elicits escape behavior on the part of a mother and calf pair could be ignored by feeding juveniles, or actively explored by a reproductively active male. Within a given age and sex class, the cumulative probability of response by the animals is usually assumed to have a sigmoid shape with respect to increasing noise levels. Few studies have actually determined the proportion of animals responding at varying levels of acoustic signal. One study that investigated the probability of response showed that for gray whales (Eschrichtus robustus) the ranges broadside to a seismic gun for 10, 50, and 90 percent probability of avoidance were 3.6, 2.5, and 1.2 km, at which the received sound levels were 164, 170, and 180 dB re 1 µPa, respectively (Malme et al., 1984).
Hearing Sensitivity
Animals will only respond directly to sounds they can detect. The hearing sensitivities of only a few individuals in a select number of species are known. Even less is known about signal detection in the presence of ambient noise. Beluga whales (Delphinapterus leucas) can detect echolocation return signals when they are 1 dB above ambient noise levels (Turl et al., 1987), and gray whales react to playbacks of the vocalizations of a predator, the killer whale (Orcinus orca), when the playback signal is equal to the ambient noise (Malme et al., 1983). In both of these cases the signals have important biological significance for the animal. Anthropogenic signals do not have the same evolutionarily enhanced significance.
Many of the situationally specific responses of marine mammals to sound will be dependent on the loudness of the sound. The loudness of the
sound is a function of the intensity of the sound at the location of the animal and the sensitivity of the animal to the frequencies of the sound. If the audiograms of the marine mammal species of interest are known, the potential effect of the sound can be estimated by weighting the level of the sound at each frequency by the sensitivity of the animal to that frequency, similar to the A-weighting of sound levels for humans hearing in air. Without such knowledge, it will be difficult to develop a predictive model of the impact of novel sounds on marine mammals.
Behavioral State
Animals that are resting are more likely to be disturbed by noise than are animals engaged in social activities. Würsig (personal observation cited in Richardson et al., 1995) summarized the responses of several species of dolphins to boats as “resting dolphins tend to avoid boats, foraging dolphins ignore them, and socializing dolphins may approach.”
Migrating bowhead and gray whales divert around sources of noise, whether actual industrial activities or playbacks of industrial activities (Richardson et al., 1995) with almost all bowheads reacting at received levels of 114 dB re 1 µPa. However, if no other option is available, migrating bowhead whales will pass through an ensonified field to continue their migration. During spring migration, when the only available lead was within 200 m of a projector playing sounds associated with a drilling platform, the bowheads continued through a sound field with received levels of 131 dB re 1 µPa (Richardson et al., 1991).
Age and Sex
Some age and sex classes are more sensitive to noise disturbance, and such disturbance may be more detrimental to young animals. Age and sex classes can be most clearly identified and observed among pinnipeds that are on land or ice, so most of the data come from responses of these pinnipeds. Differences are expected between sexes and age among classes in the way that they respond to underwater sounds. In northern sea lions (Eumetropias jubatus) dominant, territory-holding males and females with young are less likely to leave a haulout site in response to an aircraft overflight than are juveniles and pregnant females (Calkins, 1979). Walrus sometimes stampede into the water in response to aircraft overflights. These stampedes sometimes result in the death of calves (Loughrey, 1959). Vessel approaches to walrus on ice can cause the herd to enter the water and in some cases leave calves stranded in slippery depressions on the ice. These calves are more vulnerable to predation by polar bears (Fay et al., 1984). Mother-calf gray whale pairs appear to be particularly sensitive to disturbance by whale-watching boats (Tilt, 1985). Humpback whale groups
containing at least one calf were more responsive to approaches by small boats on several behavioral measures of respiration, diving, swimming, and aerial behaviors than were groups without a calf (Bauer et al., 1993).
Noise Source Context and Movement
The responses of cetaceans to noise sources are often dependent on the perceived motion of the sound source as well as the nature of the sound itself. For a given source level, fin and right whales are more likely to tolerate a stationary source than they are one that is approaching them (Watkins, 1986). Humpback whales are more likely to respond at lower received levels to a stimulus with a sudden onset than to one that is continuously present (Malme et al., 1985). These startle responses are one reason many seismic surveys are required to “ramp up” the signal so fewer animals will experience the startle reaction and so that animals can vacate the area of loudest signals. There is no evidence, however, that this action reduces the disturbance associated with these activities. The ramp-up of a playback signal or a seismic air-gun array takes place over a short timescale (a few tens of minutes maximum) compared to the changing received levels an animal experiences as it swims toward a stationary signal source. Bowheads react to playback levels of drill ship noise at levels they apparently tolerate quite well when they swim close to operating drill ships. Richardson et al. (1995) provide two explanations for these behavioral differences. First is the speed of ramp-up, as noted earlier. Second, the whales seen near an operating drill ship may be the ones that are more tolerant of noise. The sensitive whales seen responding to the playback levels may have already avoided the actual drill ship at ranges that were undetected by observers near the ship.
Responses of animals also vary depending on where the animals are when they encounter a novel noise source. Pinnipeds generally show reduced reaction distances to ships when the animals are in the water compared to when they are hauled out. Swimming walrus move away from an approaching ship at ranges of tens of meters, whereas walrus hauled out leave the ice at ranges of hundreds of meters (Fay et al., 1984). Similar differences in avoidance ranges have been seen in California sea lions and harbor seals. Sight and smell might also be important cues for hauled-out animals.
Bowhead whales in shallow water are more responsive to the overflights of aircraft than are bowheads in deeper water (Richardson and Malme, 1993). Beluga whales are more sensitive to ship noise when they are confined to open-water leads in the ice in the spring (Burns and Seaman, 1985). Migrating gray whales diverted around a stationary sound source projecting playbacks of LFA sonar when the source was located in the migratory path but seemed to ignore the sound source when it was located
seaward of the migratory path. When the source was in the path, received levels of 140 dB re 1 µPa were sufficient to cause some path deflection. However, when the source was located seaward of the migratory path, the whales ignored source levels of 200 dB re 1 µPa at 1 m and received levels greater than 140 dB re 1 µPa (Tyack and Clark, 1998).
Variability of Responses
The range of variability of responses of marine mammals to anthropogenic noise and other disturbances can be summarized in the responses of beluga whales to ships. One of the most dramatic responses in any species of marine mammal has been observed over several years in beluga whales in the Canadian high arctic during the spring. At distances of up to 50 km from icebreakers, or other ships operating in deep channels, beluga whales respond with a suite of behavioral reactions (LGL and Greeneridge, 1986; Cosens and Dueck, 1988; Finley et al., 1990). The reactions include rapid swimming away from the ship for distances up to 80 km; changes in surfacing, breathing, and diving patterns; changes in group composition; and changes in vocalizations. The initial response occurs when the higher-frequency components of the ship sounds, those to which the beluga whale are most sensitive, are just audible to the whales. Possible explanations for this unique sensitivity to ship sounds are partial confinement of whales by heavy ice, good sound propagation conditions in the arctic deep channels in the spring, and lack of prior exposure to ship noise in that year (LGL and Greeneridge, 1986). Supporting the latter point is the observation that beluga whales that fled icebreaker noise at received levels between 94 and 105 dB re 1 µPa returned in one to two days to the area where received icebreaker noise was 120 dB re 1 µPa (Finley et al., 1990).
Beluga whales in the St. Lawrence River appear more tolerant of larger vessels moving in consistent directions than they are of small boats, fast-moving boats, or two boats approaching from different directions. Older animals were more likely to react than younger ones, and beluga whales feeding or traveling were less likely to react than animals engaged in other activities, but when the feeding or traveling whales did react, they reacted more strongly (Blane and Jaakson, 1994). In contrast to the lower rate of observed reactions of these beluga whales to larger vessels, a study of the response of beluga whale vocalizations to ferries and small boats in the St. Lawrence River showed more persistent reactions to the ferries. The whales reduced calling rate from 3.4 to 10.5 calls per whale per minute to 0.0 or under 1.0 calls per whale per minute while vessels were approaching. Repetition of specific calls increased when vessels were within 1 km, and the mean frequency of vocalizations shifted from 3.6 kHz prior to noise exposure to frequencies of 5.2-8.8 kHz when vessels were close to the whales (Lesage et al., 1999).
In Alaska, beluga whale response to small boats varies depending on the location. Beluga whales feeding on salmon in a river stop feeding and move downstream in response to the noise from outboard motorboats, whereas they are less responsive to the noise from fishing boats to which they may have habituated (Stewart et al., 1982). On the other hand, in Bristol Bay beluga whales continue to feed when surrounded by fishing vessels and resist dispersal even when purposely harassed by motorboats (Fish and Vania, 1971).
Thus, depending on habitat, demography, prior experience, activity, resource availability, sound transmission characteristics, behavioral state, and ever-present individual variability, the response of beluga whales can range from the most sensitive reported for any species to ignoring of intentional harassment. Beluga whales also show the full range of types of behavioral response, including altered headings; fast swimming; changes in dive, surfacing, and respiration patterns; and changes in vocalizations.
Long-Term Responses
Almost all the studies conducted so far have looked at only short-term effects of anthropogenic noise on marine mammals. In most cases the observed responses have been over periods of minutes to hours. Even the dramatic response of beluga whales to icebreakers in the high arctic, in which the whales moved up to 80 km and were out of the area for one to two days, falls into the category of a transient response over the annual activity budget of the animals. The whales habituated and had reduced responses to subsequent icebreakers and ships in a given season.
Multiyear abandonment of a portion of the habitat because of human activity has been reported for Guerrero Negro Lagoon in Baja California, where shipping and dredging associated with an evaporative salt works project caused the whales to abandon the lagoon through most of the 1960s. When the boat traffic declined, the lagoon was reoccupied, first by single whales and subsequently by cow-calf pairs. By the early 1980s the number of cow-calf pairs using the lagoon far exceeded the number prior to the commencement of the commercial shipping (Bryant et al., 1984). Killer whales significantly reduced their use of Broughton Archipelago in British Columbia when high-amplitude acoustic harassment devices (AHDs) were installed to deter harbor seal predation at salmon farms. The AHDs operated between 1993 and 1999, and almost no whales were observed in the archipelago throughout most of this period. However, when the devices were removed in 1999, killer whales repopulated Broughton Archipelago within six months (Morton and Symonds, 2002).
Clearly there are opportunity costs associated with even the transient behavioral changes in response to noise. The movements require energy that might otherwise have been spent in acquiring food or mates or enhanc-
ing reproduction. Repetitive transient behavioral changes have the potential of causing cumulative stress. Even transient behavioral changes have the potential to separate mother-offspring pairs and lead to death of the young, although it has been difficult to confirm the death of the young. On the other hand, pups can be injured or killed when trampled by adults rapidly leaving a haulout in a transitory response to a disturbance.
MASKING OF ACOUSTIC CUES BY MARINE NOISE
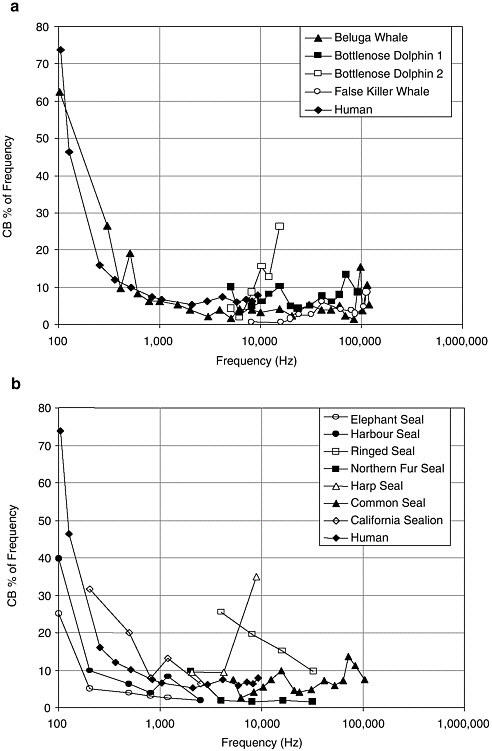
One of the most pervasive and significant effects of a general increase in background noise on most vertebrates, including marine mammals, may be the reduction in an animal’s ability to detect relevant sounds in the presence of other sounds—a phenomenon known as masking. Masking, which might be thought of as acoustic interference, occurs when both the signal and masking noise have similar frequencies and either overlap or occur very close to each other in time. Noise is only effective in masking a signal if it is within a certain “critical band” (CB) around the signal’s frequency. Thus, the extent of an animal’s CB at a signal’s frequency, and the amount of noise energy within this critical frequency band, is fundamentally important for assessing whether or not masking is likely to occur.
CBs have been measured both directly and indirectly in a number of marine mammals. In cases where data are available over a wide range of frequencies, critical bandwidth as a proportion of frequency plotted against frequency shows a steep rise at lower frequency and a less pronounced rise at higher frequencies (Figure 3-2). This pattern is also seen in terrestrial mammals. CBs are narrow for odontocetes at high frequencies (>1 kHz) and increase markedly at lower frequencies. This means that at higher frequencies only the noise energy within a narrow band of a signal will be effective in masking it, while at lower frequencies sound energy in a much wider band will cause masking.
Directional Hearing
When noise and a signal arrive at a receiver from different directions, two mechanisms can function to reduce masking. The first relates to the receiving beam pattern of the animal; that is, the extent to which its auditory system is more sensitive to sound on a particular bearing. Normally the direction of greatest sensitivity is ahead, and an attending animal will typically orient toward a sound source so that the absolute level of the sound at the receiver is increased and (provided the noise and signal are on different bearings) the signal-to-noise ratio is also improved. Animals can also determine the direction from which a sound arrives based on cues, such as differences in arrival times, sound levels, and phases at the two ears. The ability that this provides to resolve the signal and noise to different direc-
tions can further reduce masking. Thus, an animal’s directional hearing capabilities have a bearing on its vulnerability to masking. Odontocetes have good directional hearing above 1 kHz (Renaud and Popper, 1975), but directional hearing at lower frequencies has been less completely studied. The shielding effects of head structures that are important for both the receiver beam and for causing the sound-level differences at the two ears that contribute to directional hearing are both wavelength dependent. This is reflected by a general trend for a less acute directional hearing ability for lower-frequency sounds. The directivity index (DI) is a measure of the effectiveness of an acoustic receiver in reducing the effects of omnidirectional noise and is expressed as the number of dBs above the signal that omnidirectional noise must rise to mask it. Au and Moore (1984) investigated the DI of a bottlenose dolphin for a signal arriving from ahead and found that it ranged from 10.4 dB at 30 kHz to 20.6 dB at 120 kHz. At these frequencies, then, sounds arriving from ahead, such as echolocation return echoes, will be substantially protected from masking. Directional hearing is less acute in pinnipeds and has not been measured formally in any of the great whales.
Masking of Representative Signals by Realistic Noise
Most studies of masking with captive animals have explored the masking of a very simple signal, typically a pure tone, by broadband noise of constant spectral density (i.e., white noise). In the real world both signals and masking noise are more complex spectrally and temporally, and only a few studies have explored these more realistic scenarios.
The masking effects of noise from oil-spill cleanup vessels on killer whale vocalizations were investigated in a series of experiments conducted with two captive killer whales (Bain and Dahlheim, 1994). Three sets of experiments that varied the characteristics and relative position of the interfering noise were conducted. Boat noise masked all tones below 20 kHz. Masking was reduced when signal and noise sources were separated, and this effect was most pronounced at higher frequencies and greater angles of separation, suggesting the directional hearing ability of the whale was reducing masking. In contrast to the pure tone signal results, when the signal was of biological relevance, that is, killer whale vocalizations, there was little evidence of masking by boat noise.
Concern about interference with beluga whales’ communication by icebreaking activity led Erbe and co-workers to explore masking of a beluga call by three different types of icebreaker noise (Erbe, 1997, 2000; Erbe and Farmer, 1998; Erbe et al., 1999). The noise types were ice ramming (primarily propeller cavitation), natural ice cracking, and an icebreaker’s bubbler system (high-pressure air blown into the water to push floating ice away from the ship). Bubbler noise was the most effective masker of beluga
calls with a critical noise-to-signal ratio (CNSR) of 15.4 dB, followed by ramming noise (CNSR of 18 dB), with natural ice-cracking noise being least effective (CNSR 29 dB). Experiments using trained animals are time consuming and expensive to perform, so a series of software models were designed to exhibit the same masking performance as a beluga whale (Erbe et al., 1999). A neural network model showed the best performance. However, the model was trained and tested using only a single vocalization and three samples of masking noise, and thus may not be robust for other signal and noise combinations. Human performance in masking tests was very similar to that of the beluga whale (Erbe et al., 1999).
Zones of Masking
One way of identifying the potential effects of noise is to determine the areas, or zones of influence, over which particular effects might occur. Richardson et al. (1995) identified four concentric zones with decreasing size and increasing intensity of the signal. The largest zone is that of audibility, followed by responsiveness, then masking, and finally the zone of hearing loss, discomfort, or injury. The outer three zones can be essentially coterminous. If marine mammals attend to barely detectable signals, then any increase in noise may contribute to masking. The zone of masking is defined by the range at which sound levels from the noise source are received above threshold within the CB centered on the signal. A ray-tracing propagation model predicted a zone of masking of beluga whale calls by icebreaker ramming noise of 40 km (Erbe and Farmer, 2000).
Møhl (1981) developed an alternate approach for exploring the significance of different levels of masking noise. He used the sonar equation to show that as the noise increases by a set amount, the range for detecting a signal at a given signal to noise would be reduced by a constant proportion called the range reduction factor (RRF). For example, a 6-dB increase in noise would decrease by half the range for signal detection under transmission loss (TL) determined by spherical spreading, given the same signal-to-noise ratio. Under conditions where TL is given by cylindrical spreading, the range is reduced to one-quarter of its original value. (It is worth noting that in some cases the area over which signals can be detected will be a more appropriate measure than the range, in which case reduced effectiveness resulting from masking will scale in relation to RRF2.) One attractive feature of Møhl’s approach is that it does not require assumptions to be made about the signal-to-noise ratio the animal requires to make detections. It follows directly from the sonar equation that the RRFs resulting from an introduced noise are greater when existing levels of background noise are lower. However, it could be argued that in most cases the appropriate measure of the biological cost of masking relates to the absolute level of signal detection efficiency for the animal in the presence of all noise. In
this case, an animal whose auditory efficiency was already reduced by masking from existing higher levels of background noise might be more likely to be adversely affected by an additional masking source than an animal in a quiet environment.
Masking Thresholds
Masking experiments usually measure whether or not any signal can be detected in a particular level of noise. However, detection may not always be the most biologically appropriate measure; in some situations more stringent criteria may apply. Erbe and Farmer (2000) pointed out that relatively low signal-to-noise levels that allow detection might not be sufficient to allow signal recognition. They suggested a higher “recognition threshold” should be considered. An even higher level, an “understanding threshold” may be necessary for an animal to glean all information from complex signals.
Although results from masking experiments are often presented in terms of specific thresholds, it can be more useful to think of masking affecting the probability of correctly detecting a signal. This perspective is particularly appropriate in real-world situations, where levels and spectral characteristics of signal and noise are likely to vary over time.
Strategies to Reduce the Probability of Masking
Marine mammals evolved in an environment containing a wide variety of naturally occurring sounds, and thus they show a variety of strategies to reduce masking. Vocal signals may be designed to be robust to masking effects. Signals can be more easily detected in noise if they are simple, stereotyped, and occur in a distinctive pattern. Signals may also show a high level of redundancy; they may be repeated many times to increase the probability that at least some will be detected. However, these characteristics all minimize the amount of information that a signal can convey. Animals can adapt their behaviors to minimize masking, and it is reasonable to interpret such behavioral changes as an indication that masking has occurred. For example, the vocal output of a beluga whale changed when it was moved to a location with higher levels of continuous background noise (Au et al., 1985). In the noisier environment, the animal increased both the average level and frequency of its vocalizations, as though it were trying to compensate for and avoid the masking effects of, the increased, predominantly low-frequency, background noise levels. Penner et al. (1986) conducted trials in which a beluga whale was required to echolocate on an object placed in front of a source of noise. The animal reduced masking by reflecting its sonar signals off the water surface to ensonify to the object. The strongest echoes from the object returned along a path that was differ-
ent from that of the noise. This animal’s ready application of such complex behavior suggests the existence of many sophisticated strategies to reduce masking effects.
Beluga whales increased call repetition and shifted to higher peak frequencies in response to boat traffic (Lesage et al., 1999). Gray whales increased the amplitude of their vocalizations, changed the timing of vocalizations, and used more frequency-modulated signals in noisy environments (Dahlheim, 1987). Humpback whales exposed to LFA sonar increased the duration of their songs by 29 percent (Miller et al., 2000).
The physiological costs of ameliorating masking effects have not been reported. Although these examples all appear to show animals adapting their vocal behavior to reduce the impact of masking, this does not imply that there were no costs resulting from increased levels of noise. Masking may have been reduced but not eliminated. Costs of the changed behavior, such as increased energetic expenditure on higher-intensity vocalizations and use of vocalizations at suboptimal frequencies cannot be estimated yet.
Critical Research Needs to Understand Effects of Masking
Attempts to assess the masking effects of a particular type of noise in marine mammals are hindered by our poor understanding of how animals make use of the many acoustic cues in the marine environment. Though it is assumed that they attend to, and make use of, each other’s communication vocalizations, it is unclear what received levels are necessary to elicit recognition and response to social calls.
The biological implications of signal masking will depend greatly on the function of the signal and the context. In a healthy animal population in which males compete with each other vocally to attract a female, the introduction of masking noise might have little effect because increased noise would disadvantage all males equally. Even if the females’ ability to make a mating choice were diminished, they would still be likely to find a mate. In the case of a severely depleted population, the ability of males and females to find each other using acoustic cues could become vital for the well-being of the species. If additional noise reduced acoustic range by masking and effective reproduction were compromised, the consequences for individuals and populations could be very significant (Payne and Webb, 1971; Myrberg, 1980).
How marine mammals make use of the myriad acoustic cues in the marine environment, or the “acoustic scene,” is even more poorly understood than masking of communication. Many of these acoustic cues are faint and are thus susceptible to masking by even low levels of noise. While a vocalizing animal may adapt its vocal behavior to compensate for increased levels of masking noise by vocalizing more intensely, changing the emphasized frequency or increasing redundancy, masking of these other
acoustic cues cannot be mitigated. A better understanding of the role of passive listening, that is, investigation of the environment through listening without active generation of echolocation pulses, in the lives of marine mammals may well be the most fundamental research need for assessing masking impacts. Detailed field research involving fine-scale behavioral observations linked to sensitive real-time acoustic monitoring will be required to gain any appreciation of how marine mammals utilize these low-level noises.
To investigate the occurrence of masking in the real world, field projects could be designed to study behavioral changes, thought to be indicative of masking (such as the strategies to avoid masking outlined earlier), and behavioral performance in situations with different levels of background noise could be monitored (see also recommendations in NRC, 2000; Appendix D). Measures of feeding rates and hunting success, mate-searching behavior, and predator avoidance would be necessary to elucidate whether masking effects were likely to affect the survival or reproduction of the individual and ultimately impact populations.
HABITUATION, SENSITIZATION, AND TOLERANCE OF MARINE MAMMALS TO MARINE NOISE
Habituation to repeated presentations of a signal that is not associated with physical discomfort or overt social stress is a common adaptive feature of sensory systems that predates the evolution of mammals. It is not surprising that marine mammals show habituation to many signals that initially cause an overt reaction. To demonstrate habituation, the same signal needs to be presented to the same individual repeatedly and the response of that individual charted over the sequential presentations. Such a demonstration in marine mammals is rare. Instead, habituation is inferred by the changes in the response of animals of the same species in the same area over time. This assumes that although the individuals are unidentified in the group, there is consistency in group composition over the course of the study. A second-order inference of habituation can also be made by comparing the reactions of individuals of the same species from two different areas to the same stimulus, the stimulus being one to which animals in one area have been exposed previously, whereas animals in the other area are assumed naive with respect to this particular stimulus.
Some of the clearest evidence of habituation comes from attempts to use sound sources to keep marine mammals away from an area or a resource (Jefferson and Curry, 1994). Acoustical harassment devices (AHDs) have been used in an attempt to keep pinnipeds away from aquaculture facilities or fishing equipment. AHDs emit tone pulses or pulsed frequency sweeps in the 5-30 kHz range at source levels up to 200 dB re 1 µPa at 1 m. Although initially effective, over time some of the devices became less able
to deter harbor seals (Phoca vitulina), presumably because of habituation (Mate and Harvey, 1987) but also because of a change in seal behavior in which the animals spend more time swimming with their heads out of the water when they are in intense sound fields. Seals and California sea lions (Zalophus californianus) even habituate to “seal bombs” that can have peak sound pressure levels of 220 dB re 1 µPa at 1 m (Mate and Harvey, 1987; Myrick et al., 1990). Harbor porpoises (Phocoena phocoena) habituate to pingers placed on gillnets in an attempt to reduce the porpoise bycatch. The probability of porpoises being within 125 m of a pinger decreased when the pinger was first activated, but within 10-11 days had increased to equal the control (Cox et al., 2001).
Watkins (1986) summarized 25 years of observations of whale responses near Cape Cod to whale-watching boats and other vessels. Minke whales (Balaenoptera acutorostrata) changed from frequent positive interest to generally uninterested reactions. Fin whales (B. physalus) changed from mostly negative to uninterested reactions. Humpbacks (Megaptera novaeangliae) changed dramatically from mixed responses that were often negative to often strongly positive reactions, and right whales continued the same variety of responses with little change. Gray whales wintering in San Ignacio Lagoon are less likely to flee from whale-watching boats later in the season than they are shortly after arriving in the lagoon (Jones and Swartz, 1984). In all these examples, factors in addition to habituation could have contributed to the observed changes.
In contrast to habituation, which results from repeated presentations of an apparently innocuous stimulus, sensitization is the result of prior presentation of a stimulus that either by itself or in conjunction with another action results in a negative experience for the animal. In sensitization, responses at subsequent presentations are more marked than are the responses at the initial presentation. Northern fur seals (Callorhinus ursinus) showed little initial reaction to a ship, but if that ship were subsequently used in seal hunting, the seals avoided it at distances up to a mile (H. Kajimura, in Johnson et al., 1989). Walruses hauled out on land are more tolerant of outboard motorboats in years when they are not hunted from such craft than they are in years when these boats are used in walrus hunts (Malme et al., 1989). Bottlenose dolphins that had previously been captured and released from a 7.3-m boat would flee when that boat was more than 400 m away, whereas bottlenose dolphins that had not been captured by the boat often swam quite close to it (Irvine et al., 1981). All the reported cases of sensitization are the result of conditioning: the pairing of a given stimulus with a significantly negative experience.
Animals will tolerate a stimulus they might otherwise avoid if the benefits in terms of feeding, mating, migrating to traditional habitat, or other factors outweigh the negative aspects of the stimulus. Already noted is the case of bowhead whales on spring migration, where they needed to use the
one available lead in the ice cover to continue on their eastward migration and passed through a sound field with projected drilling ship sounds at levels of 131 dB re 1 µPa (Richardson et al., 1991). Bowheads also return to the same areas of the Canadian Beaufort Sea year after year even though seismic surveys occurring at the same time are an annual feature of these areas (Richardson et al., 1987). Whether there are particularly dense concentrations of prey in these areas or whether the bowheads’ response is simply historical philopatry is unknown.
In at least one case, a source that did not elicit a fleeing response turned out to be capable of causing damage. Humpback whales in Newfoundland remained in a feeding area near where seafloor blasting was occurring. The humpbacks showed no behavioral reaction in terms of general behavior, movements, or residency time. In fact, residency time was greater in the bay closest to the blast site than it was in other bays of equivalent size and productivity nearby. Estimated peak received levels during blasting were approximately 153 dB re 1 µPa with most of the sound energy below 1,000 Hz (Todd et al., 1996). Two humpback whales found dead in fishing nets in the area had experienced significant blast trauma to the temporal bones (Ketten et al., 1993).
ACOUSTICALLY INDUCED STRESS
Acute responses to sounds may be difficult to quantify, but they are much more tractable to investigation than are responses to repeated or chronic sounds. Sounds resulting in one-time acute responses are less likely to have population-level effects than are sounds to which animals are exposed repeatedly over extended periods of time. Long-term population effects will have the greatest impact on marine mammal species.
Long-term effects of ocean sounds can include the transformation of TTS to permanent threshold shift and an increase in occurrence of pathological stress. Stress can be defined as a perturbation to homeostasis. So long as the perturbation is within the range the physiological system is capable of handling, is of short duration, and is not continually encountered, homeostasis is restored through an adaptive stress response. However, when the perturbation is frequent, outside the normal physiological response range, or persistent, the stress response can be pathological.
Stress can induce secretion of corticotrophin releasing factor (CRF) from the hypothalamus. CRF promotes the release of glucocorticoids and catecholamines, which modulate the immune response and can lead to changes in the response to infectious, neoplastic, allergic, inflammatory, and autoimmune diseases (Webster et al., 1977). Chronic stress can also suppress reproduction (Rabin et al., 1988), inhibit growth (Diegez et al., 1988), and alter metabolism (Mizrock, 1995).
Although stress-induced pathologies have been hard to identify in free-
ranging marine mammals, based on work with terrestrial mammals, it is likely that marine mammals would experience the same responses. The stress caused by pursuit and capture activates similar physiological responses in terrestrial mammals (Harlow et al., 1992) and cetaceans (St. Aubin and Geraci, 1992). One of the first recognized effects of chronic stress was the hypertrophy and hyperplasia of the adrenal cortex and medulla (Selye, 1973). Some possibly stress-induced adrenal pathologies have been observed in marine mammals. Harbor porpoises that died of chronic causes were more likely to exhibit adrenocortical hyperplasia than were ones that died of acute causes (Kuiken et al., 1993). Mass-stranded Atlantic white-sided dolphins had adrenal cysts, which were possibly stress related (Geraci et al., 1978). Both adrenocortical hyperplasia and cysts were observed in stranded beluga whales with the incidence and severity of the lesions increasing with age, although the authors could not attribute the adrenocortical changes to chronic stress, in contrast to normal aging (Lair et al., 1997).
Controlled laboratory investigations of the response of cetaceans to noise have shown cardiac responses (Miksis et al., 2001) but have not shown any evidence of physiological effects in any of the blood chemistry parameters measured. Beluga whales exposed for 30 min to 134-153 dB re 1 µPa playbacks of noise with a synthesized spectrum matching that of a semisubmersible oil platform (Thomas et al., 1990) showed no short-term behavioral responses and no changes in standard blood chemistry parameters or in catecholamines. Preliminary results from exposure of a beluga whale and bottlenose dolphin to a seismic watergun with peak pressure of 226 dB re 1 µPa showed no changes in catecholamines, neuroendocrine hormones, serum chemistries, lymphoid cell subsets, or immune function (Romano et al., 2001).
Among terrestrial mammals, a bank of blood indicators is a more reliable measure of stress across species or within species and across time (Hattingh and Petty, 1992). In cetaceans, Southern et al. (2001) and Southern (2000) are attempting to develop microassays to detect in skin samples from free-ranging cetaceans changes in a suite of 40 stress-activated proteins.
Although techniques are being developed to identify indicators of stress in natural populations, determining the contribution of noise exposure to those stress indicators will be very difficult but important to pursue in the future when the techniques are fully refined.
NEW RESEARCH TOOLS TO UNDERSTAND MARINE MAMMAL BEHAVIOR
Any real understanding of long-term and cumulative effects of noise on marine mammals will require the development and refinement of a number
of new research instruments. Ideally, sound pressure level should be recorded as the animal receives it and the vocalizations of the animal also need to be recorded in real time along with as many movement parameters and physiological parameters as possible. Recently several new tags have been developed that incorporate some of these features. Researchers working on northern elephant seals, Mirounga angustirostris, have developed acoustic recording packages that include a hydrophone and temperature and depth sensors (Burgess et al., 1998) or a digital audio recorder with a time-depth recorder and a time-depth-velocity recorder (Fletcher et al., 1996) in a package that can be placed on juvenile seals. The tags record received sound, seal swim strokes, and during quiet intervals at the surface both respiration and heartbeats. Cetacean researchers further developed these concepts into digital sound recording tags that record onto solid-state memory received signal levels, animal vocalizations, pitch roll and orientation, and depth (Burgess, 2001; Johnson et al., 2001; Madsen et al., 2002). Three-dimensional tracks of the whale’s movements can be reconstructed from the recorded data. These tags are typically applied with suction cups so although they provide a lot of data, it is only for a short time period. Another tag places a suction-cup hydrophone on a dolphin to record heartbeats. This has been tested so far on captive animals where the dolphin showed significant heart rate accelerations in response to playbacks of conspecific vocalizations compared to baseline rates or to playbacks of tank noise (Miksis et al., 2001). Finally, radio tags need to be developed that remain attached for several years and transmit only on a programmed cycle or in response to a query signal. For most marine mammal species, the difficulty in identifying individual animals rapidly and reliably makes it very difficult to follow animals for long periods of time to determine cumulative effects. Borggaard et al. (1999) were able to follow individually identified minke whales over four years and noted that this provided a more sensitive means of assessing impacts of industrial activity than did abundance and distribution measures. At a minimum, animals must be identified and observed preexposure, during exposure, and postexposure for a sufficient number of repetitions and for a sufficient period of time to be able to make any reasonable statements on the effect of the exposure on a given animal and potentially on the population. Without these data, we will simply continue to collect disparate observations of transient behavior, which tell us little about the impact of anthropogenic noise on marine mammals.
MARINE ECOSYSTEM IMPACTS OF NOISE
While the focus of the concern regarding the impact of marine ambient sounds is on mammals, mammals make up only a tiny fraction of all marine species. Moreover, other marine organisms, fishes and invertebrates, are critical components of the food chain for marine mammals (and terrestrial
mammals, including humans), and any impact on these organisms, or their eggs and larvae, could have significant impact on mammals.
The data on the impact of sound on fishes are very limited and nonexistent for reptiles and invertebrates. A few studies that suggest that exposure to high-level pure tones for an hour or more will damage the sensory cells of the ears of a few species (one freshwater and one marine; Enger, 1981; Hastings et al., 1996), although the extent of damage is limited and only occurs after several hours of continuous exposure. Moreover, there is evidence that fish will recover from drug (aminoglycoside antibiotic) induced hair cell damage over a period of several weeks (Lombarte and Popper, 1994).2 At the same time, during a recovery period of several weeks, fish are without a full set of sensory cells and so they may not be able to detect predators and prey, and thus have a substantially decreased chance for survival.
There are significant caveats on the fish noise-exposure studies [see Hastings et al. (1996) for a full discussion]. First, the studies were done with just a few species, and only Enger (1981) used a marine species, so it is not clear if these data can be extrapolated to other species. Second, the exposure in all of the studies was for long periods of time and to pure tones. Since most anthropogenic noise is likely to be of short duration, extrapolation from long-term continuous exposure to short-term or pulsed exposure may be inappropriate. Third, the animals in these experiments were confined near the sound source. Since fish are free to move around, it might be expected that they would move away from an intense sound.
Another issue is the sound levels used in the few fish studies. In both studies, sounds were 90-140 dB above threshold (about 180 dB re 1 µPa).
Perhaps a more significant study is one on the impact of air-guns on the ears of a variety of Australian marine fishes. In this study, fish were exposed to the sound of a small air-gun and the ears collected for analysis of inner ear hair cell damage (McCauley et al., 2000, 2003). The results show that exposure to air-guns with a maximum received level of 180 dB re 1 µPa over 20-100 Hz causes major damage to sensory cells of the ear of at least one species. Despite a number of caveats to these results, they suggest air-guns damage sensory hair cells in fishes. While similar studies have not been done with marine mammals, one must question whether these results could also have implications for marine mammals exposed to air-guns, particularly since the hair cells in fishes and marine mammals are so similar to one another.
There are also data that suggest that there may be significant impacts on fish behavior from air-guns, and perhaps from other sound sources. Several studies suggest that intense sounds may result in fish moving from
an area for extended periods of time. For example, Engås et al. (1996) showed a significant catch decrease in a fishing area after use of air-guns, suggesting that fish moved from the ensonified area and only returned days later. There is also some evidence low-frequency noise produced by fishing vessels and their associated gear may cause fish to avoid the vessels (Maniwa, 1971; Konagaya et al., 1980). While all of these data need replication, they do suggest that sounds may change the behavior of fish. Movement of fish from a feeding area of marine mammals (or fishing areas for humans) could have an adverse impact on the higher members of a food chain and therefore have long-term implications despite the fish themselves not being killed or maimed.
Another concern is the impact of high-level anthropogenic sounds on overall behavior. Since many species of fish use sound for attracting mates and for other behaviors, any masking of these sounds could alter behavior. Increased environmental sounds in the vicinity of coral reefs may have a substantial impact on settling of larval fish on the reefs. Larval reef fish of many species spend part of their lives offshore and away from reefs, and then need to find a reef where they will live for the remainders of their lives (Leis et al., 1996). Recent evidence suggests that at least some larval fish are likely to use the reef sounds to find the reefs and that the fish will go to regions of higher-level sounds (Tolimieri et al., 2003). Thus, if there are intense offshore sounds, larval fish may be confused and not be able to find the reef. Alternatively, such sound may mask reef sounds, again preventing larval fish from finding the reef.
Potentially, anthropogenic sounds can have effects on marine life at a number of different levels, from short-term effects on individuals to long-term effects on populations and even species. Effects that can be dramatic, even lethal, at the level of the individual may have negligible consequences at the population level if, for example, small numbers of a large healthy population are affected. Conversely, effects that may seem insignificant for the well-being of individuals could have important conservation consequences for populations that are depleted and under stress. For example, a decrease in feeding rate that might equate to a year’s delay in attaining sexual maturity, a small increase in infant mortality, or a slightly shorter life span may not be overly significant to an individual animal but could mark the difference between extinction and recovery for a critically endangered species. It is important to emphasize that whether or not a particular impact could be of conservation significance will depend on the status of the population; thus, the conservation significance of particular impacts must be assessed on a case-by-case basis. While much legislation and scientific work focuses on conservation goals, it is important to recognize that the well-being and welfare of individual wild animals is also a concern for many members of the public and harassment of any individual marine mammal is prohibited by the Marine Mammal Protection Act.