4
Screening for Thyroid Dysfunction
This chapter discusses the conceptual issues surrounding screening for thyroid dysfunction and evaluates the evidence base for the value of screening, using the basic methods of the United States Preventive Services Task Force. The chapter concludes with a recommendation on the value of screening in the Medicare population. We take advantage of the evidence review, which was conducted concurrently by Dr. Mark Helfand of the Oregon Health & Science University Evidence-based Practice Center for the Task Force; the review is contained in Appendix B of this volume.
PRINCIPLES OF DISEASE PREVENTION
The intent of preventive interventions is to eliminate totally or to defer as long as possible the clinical onset of overt disease or, if a disease has become clinically apparent in an individual, to defer the progression and secondary consequences of that disease. Preventive interventions, by conventional definitions, comprise three kinds of activities: primary, secondary, and tertiary prevention.
Types of Prevention
Primary prevention generally refers to interventions that avoid the biological onset of a disease or condition within an individual, either by avoiding exposure to disease-causing agents or situations or by protecting the individual from the harmful effects of those exposures. Examples include all immunization against infectious agents, pasteurization of milk and other foods, avoidance of alcohol
when operating a motor vehicle, and condom use to prevent sexually transmitted diseases. These interventions may not work perfectly, nor are they necessarily free of adverse effects: A vaccine may protect most but not all of those immunized and occasionally may cause a clinical illness itself. In general, society demands that primary preventive interventions have relatively few adverse effects because they are administered to healthy individuals with varying levels of disease risk.
Secondary prevention refers to the early and asymptomatic detection of existing but subclinical diseases in individuals; this is usually referred to as disease screening. The term “subclinical” is paramount here because it indicates that the disease shows no apparent manifestations that are identifiable either to the individual or to the health professional. Examples of screening tests include cervical cytology (the “Pap smear”) for early detection of cervical cancer; a blood cholesterol test to detect blood lipid abnormalities that may put someone at risk of heart attack, stroke, and other conditions; and mammography, which may detect early breast cancers that are more curable than when they become palpable to the patient or the professional. As a rule, society may tolerate less than perfect accuracy in a screening test and, if unavoidable, a higher level of adverse effects due to the screening test or its later clinical consequences than in primary prevention because of the high likelihood of emerging disease being present.
It is important to note that tests that are used for screening purposes usually have different characteristics from tests used for diagnostic purposes. Usually, a positive screening test only indicates that a disease or condition has a higher likelihood of being present; additional definitive diagnostic testing is indicated to confirm that likelihood. Screening tests emphasize sensitivity over specificity. In order to achieve their purpose, they generally produce a large proportion of false-positive results. Thus, a positive screening test usually leads to further diagnostic testing before the condition is assumed to be present. In fact, for some conditions, various levels of staged screening tests may be applied. Like screening tests, diagnostic tests may not always be perfectly accurate; thus persons being screened for a condition should be counseled about the levels of accuracy of both types of tests and the possibility that a negative screening test does not always guarantee that disease is not present or will not occur in the future. As will be discussed, this is directly relevant to the issue of screening for thyroid dysfunction.
Screening tests are generally only applied if they meet a set of standard criteria (Cuckle and Wald, 1984), which are briefly summarized here:
-
The natural history of the condition being screened must be understood, so that there is good evidence that the disease outcome will be favorably influenced by further diagnosis and treatment.
-
The screening test must be suitably reliable and valid so that most of those tested are accurately classified as to the current presence or absence of the disease in question.
-
The screening test must be acceptable when applied to most persons for whom it is indicated. If not, the test will not effectively reach its intended target population and will fail as a disease prevention measure.
-
If a screening test indicates the possibility of a disease being present, there must be suitable, definitive tests to make a formal diagnosis of that condition.
-
Suitable professional resources should be available to explain the results of the screening test to those with both negative and positive findings.
-
Proven, effective treatments must exist for the conditions screened for and diagnosed—treatments that lead to increased survival, function, and quality of life. It is important that these treatments be widely available and accessible, both geographically and fiscally. Raising patient concern may have little value if the requisite medical care cannot be delivered.
-
Early intervention must have value. Diagnosis as a result of screening must either provide a better chance of cure, less disability, a reduction in the development of pain or other significant symptoms, or enable treatment that is less arduous or expensive. If early diagnosis only extends the period of treatment of chronic illness or results in expenditures for expensive treatment occurring sooner rather than later, it may only increase costs without commensurate benefit.
Tertiary prevention usually refers to the prevention of the secondary adverse consequences of existing diseases and conditions. Examples include the administration of physical therapy to prevent freezing of arthritic joints or the use of medications that prevent rhythm disorders of the heart in those who have had a heart attack. Thus, tertiary prevention merges with the normal medical management of diseases, an activity by and large covered by Medicare as “reasonable and necessary for the diagnosis or treatment of illness,” and therefore not considered to be a preventive service excluded by Section 1862(a)(1)(A) of Title XVIII of the Social Security Act.
Issues in Screening for Thyroid Dysfunction
When considering screening for thyroid dysfunction, several issues concerning the application of the screening principles require discussion. First, as noted elsewhere in this volume, thyroid dysfunction is more than one condition. Thus, screening applications and outcomes may be different for hypothyroidism than for hyperthyroidism, and this issue will be discussed. This is true of other screening applications. For example, mammography screens for several types of histologically distinct breast tumors, some with varied natural histories and clinical trajectories.
Second, there is the issue of whether persons whose thyroid dysfunction is identified by screening are truly asymptomatic. In general, among diseases for which screening is known to be effective, a symptomatic patient with that condition, ceteris paribus, is likely to be in a more advanced stage than an individual
without symptoms. When this is the case, the impact and improved outcomes from screening may be lessened or eliminated. As noted in Chapter 3, the Committee discussed the definition of subclinical thyroid dysfunction and concluded that it was not possible to conclude that an individual with the biochemical diagnosis was or was not truly free of symptoms because the symptoms are general, common, and have a high background occurrence in biochemically normal persons. Thus, the Committee concluded that screening would be considered when thyroid dysfunction-compatible symptoms, if present, are unrecognized by the patient or the health professional. Otherwise, if a clinician has a strong suspicion that clinically manifest thyroid dysfunction is present, then any testing is a diagnostic process already covered by Medicare. This is the position that Helfand (Appendix B) takes on this matter. This situation is made more complex by the fact that many older persons have substantial co-morbidity (Kaplan et al., 1999). Thus, many thyroid dysfunction-compatible symptoms (e.g., fatigue) may in fact be related to other conditions.
Helfand (Appendix B) also points out that the scientific literature usually applies the term “subclinical hypothyroidism” on purely biochemical terms as an elevated serum thyroid stimulating hormone (TSH) level coincident with a normal serum free T4. This definition would apply to many persons who are not candidates for screening: Because they are known to or are likely to have thyroid disease by their history, they have abnormal results stemming from the consumption of an incorrect amount of levothyroxine in the treatment of hypothyroidism, they have thyroid failure after treatment of hyperthyroidism; or they have thyroid failure already recognized by its symptoms. There is also the clinical situation in which a patient is tested for thyroid dysfunction because it is a known cause or aggravating factor for some other condition, such as atrial fibrillation or hyperlipidemia. For the purposes of this Committee’s analysis anyone with recognized symptoms or a history of any kind of thyroid disease or exposure to an agent known to be thyrotoxic will not be considered a potential screening subject, and the search for thyroid dysfunction as a cause or complicating factor in patients with other conditions will not be considered a screening procedure.
Third, as for many other disorders that are first diagnosed biochemically, thyroid dysfunction is characterized by blood measures that possess continuous distributions, as are blood total cholesterol and hemoglobin levels. In this instance, assigning clinical or biochemical cut-points is in many ways arbitrary, if necessary; a particular categorical definition may lead to a certain amount of mis-classification of disease occurrence or future risk of disease. Thus, not everyone assigned by biochemical criteria to have or not have a particular condition will be classified correctly on measurement grounds alone. In the case of subclinical thyroid dysfunction, there are conceptual arguments as to whether biochemical criteria reflect physiological abnormalities; for the same reasons of measurement error and miscategorization, the likelihood that a biochemical abnormality indi
cates an abnormal clinical or functional state may depend on the degree of that abnormality.
A similar difficulty occurs with the division of thyroid dysfunction into “subclinical” or “overt” on the basis of serum T4 measurements. In the entire population of persons with thyroid dysfunction defined by abnormal serum TSH concentrations, those who also have abnormal serum T4 will be much more likely to have clinically recognizable morbidity and potential to benefit from treatment. The screening population, however, is a much more homogeneous group; subjects, by definition, lack recognized morbidity. The distinction between biochemically defined “subclinical” or “overt” thyroid dysfunction in the screening population is less likely to be meaningful in terms of either burden of disease or potential to benefit from treatment.
EVIDENCE OF EFFICACY OF SCREENING FOR THYROID DYSFUNCTION
The Committee considered the evidence concerning the efficacy of biochemical screening for thyroid dysfunction using serum TSH levels in several ways: (1) reviewing and considering the evidence summarized in the systematic evidence review by Helfand (Appendix B); this was prepared for the Task Force, which has presented screening recommendations for thyroid conditions in the past (United States Preventive Services Task Force, 1996); (2) reviewing other peer-reviewed literature relevant to the Committee’s mission; and (3) conducting a workshop on thyroid function screening, held in Irvine, CA, in October 2002. See Appendix A for the workshop agenda.
Analytical Framework for Interpretation of the Evidence
Although the paper (Appendix B) by Helfand contains an analytical framework for evaluating the evidence on thyroid dysfunction screening, the Committee chose to create its own framework, shown in Figure 4-1. This framework is similar to the Helfand approach in several ways but differs in certain areas of organization and complexity. The Committee’s framework portrays the theoretical benefits to health from TSH screening and looks for evidence to confirm or reject the theory. This approach begins with TSH screening of an adult population with no recognized symptoms related to thyroid dysfunction (see earlier discussion). While not noted, a repeat TSH level may be performed at this point for confirmatory reasons. If an abnormal TSH value is found, a free thyroxine (free T4) level is determined (Paths A-D). If the TSH is high and the free T4 is low, then a biochemical diagnosis of overt hypothyroidism is made (Path A). Effective treatment could then reduce the risk of this condition’s secondary disorders and metabolic consequences (Effect A1), which in turn could reduce the morbidity from these consequences. In an analogous manner, if the TSH level is low, and
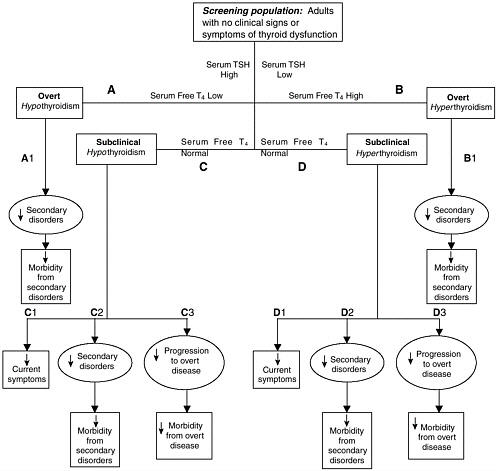
FIGURE 4-1 Theoretical benefits to health from screening for thyroid dysfunction
the free T4 level is elevated (Path B), a diagnosis of biochemical overt hyperthyroidism is made, and with effective treatment could lead to amelioration of the consequent secondary disorders (Effect B1) and morbid outcomes.
If the TSH level is high, but the free T4 level is normal (Path C), then biochemical subclinical hypothyroidism is designated. This condition, in turn, may be treated leading in the short or long term to fewer current if unrecognized symptoms (Effect C1), a decrease in secondary disorders and consequent morbidity (Effect C2), or prevention of progression to overt hypothyroidism, with any associated morbid consequences (Effect C3). If the screening TSH level, however, is abnormally low and the free T4 is normal, then a designation of subclinical hyperthyroidism is made (Path D). Effective treatment could theoretically lead to fewer symptoms if present (Effect D1), the management of secondary
disorders and their morbid consequences (Effect D2), or the prevention of progression to overt hyperthyroidism, with its morbid consequences (Effect D3).
However, as emphasized, the evidence for effective screening and treatment may not be fully present; and, using this framework for discussion, the following addresses the evidence related to each of these pathways. It should be noted at the outset that the Committee could find no randomized trial of thyroid dysfunction screening among adults with unrecognized thyroid disease and symptoms potentially related to thyroid dysfunction in which long-term clinical morbid outcomes were determined or compared.
Clinical Consequences of Subclinical Thyroid Dysfunction
Overt hypothyroidism and hyperthyroidism, when identified in a clinical as opposed to a screening context, are well-described clinical syndromes and will not be recounted further here. As was explained in the previous chapter and earlier in this chapter, subjects with biochemically overt thyroid dysfunction discovered in a screening context should more closely resemble persons with subclinical dysfunction than persons whose overt thyroid dysfunction is diagnosed on the basis of clinical suspicion. This discussion is based on the evidence-based review by Helfand (Appendix B), which will be cited by name and not further referenced. All paths noted refer to Figure 4-1 unless otherwise noted.
Evidence of the Clinical Consequences of Subclinical Hyperthyroidism
Symptoms Important to understanding the value of screening for subclinical thyroid dysfunction is the determination of concurrent symptoms that may be relieved and subsequent morbidity and mortality that could be prevented through diagnosis and treatment. As Helfand notes, subclinical hyperthyroidism has been associated with cognitive abnormalities, and in several studies with abnormal myocardial contractility, sometimes associated with exercise intolerance. However, the frequency of these cardiac abnormalities in screening of persons with unrecognized symptoms is not well studied, and no study has linked these physiological abnormalities to a greater risk of developing overt congestive heart failure (Effect D1).
Morbidity With respect to subclinical hyperthyroidism in persons not known to have thyroid disease, Helfand found one good-quality observational study showing that a low TSH level was associated with a substantially increased risk of atrial fibrillation. Although the clinical consequences of atrial fibrillation in such patients have not been studied longitudinally, this condition is generally associated with increased risk of stroke and other conditions and with a higher risk of death (Effect D2).
In one prospective cohort study of older persons from the Netherlands, low TSH levels were associated with an increased risk of dementia after 2 years of
follow-up, after adjusting for age and sex. With respect to osteoporosis, Helfand found one good-quality study suggesting there were similar amounts of age-related bone loss among women with normal, low, or undetectable serum TSH levels. In that same cohort, there was an increased rate of hip fracture among those being treated for hyperthyroidism. Other studies on the risk of osteoporosis were in small numbers of patients with nodular thyroid disease or Graves’ disease.
Mortality With regard to the impact of subclinical hyperthyroidism on total mortality associated with subclinical hyperthyroidism, Helfand found one study where mortality was higher among those with low serum TSH levels. However, there was no adjustment for co-morbidity, which, as noted in Chapter 2, may cause low TSH levels in the absence of thyroid dysfunction and therefore create an association between low TSH levels and mortality that would be neither causal nor related to thyroid disease.
Evidence of the Clinical Consequences of Subclinical Hypothyroidism
As documented in Chapter 3, the incidence and prevalence of subclinical hypothyroidism is substantially greater than of subclinical hyperthyroidism, and thus the former is in some dimensions more thoroughly studied. Particular attention has been paid to symptoms and well-being, blood lipid abnormalities, and atherosclerotic cardiovascular disease. Issues of thyroid disease in pregnancy will not be considered here.
Symptoms and quality of life As Helfand notes, a 1998 review by the American College of Physicians concluded there were no clear differences in prevalence and severity of symptoms or quality of life between euthyroid individuals in the general population and those with untreated subclinical hypothyroidism. Two more recent cross-sectional studies came to the same conclusion, although one found a higher prevalence of “changed” symptoms in those with subclinical dysfunction. Thus, overall, it appears there is little evidence that the subclinical condition is associated with a higher prevalence of symptoms in a population naïve for thyroid disease (Effect C1).
Hyperlipidemia Overt hypothyroidism has long been known to be associated with elevated levels of blood total cholesterol and LDL cholesterol. Most of these studies were performed in patients with severe hypothyroidism. As Helfand notes, studies of blood lipid abnormalities in subclinical hypothyroidism have yielded inconsistent results; they have been mostly in women, with sparse and inconsistent data in men (Effect C2). Most but not all studies, generally cross-sectional in nature, do not find significantly altered blood cholesterol or other lipid levels associated with subclinical hypothyroidism; there are exceptions where increased levels of 2-15 mg/dl are found. Some of the studies, both with null and positive findings, do not always adequately control for possible confounders such as co-morbidity, socioeconomic status, diet, other drugs that may
alter lipid levels such as estrogens, or history of known thyroid disease, with or without treatment.
Atherosclerosis Helfand states the relation between subclinical hypothyroidism and clinical atherosclerotic conditions is likewise unclear (Effect C2). In an English cohort (the Whickham study), no relation was found. However, in a Dutch cohort (the Rotterdam study) using mainly a cross-sectional analysis, myocardial infarction and abdominal atherosclerosis rates were significantly elevated among women with subclinical hypothyroidism, even after adjusting for major cardiovascular risk factors, including blood cholesterol levels. Helfand reviewed other cross-sectional studies and concluded that the causal direction was uncertain and that few adequately controlled for the range of cardiovascular risk factors.
EVIDENCE OF EFFICACY OF TREATMENT FOR THYROID DYSFUNCTION
This section draws on the work of Helfand in considering the evidence that treatment of thyroid dysfunction affects the long-term clinical outcomes of individuals with these conditions. Details of the strategy for literature search, inclusion criteria for studies and the grading of study quality, are detailed within the Helfand report. (Appendix B)
Evidence of Efficacy of Treatment of Overt Thyroid Dysfunction
While it has been accepted without randomized trials that treatment will benefit patients with overt thyroid dysfunction who present with significant symptoms or complications, there cannot be as much confidence of net long-term benefit for treatment of persons with biochemically overt thyroid dysfunction who are detected by screening. The latter group has a lower burden of disease, on average (the more symptomatic patients are more likely to be discovered without screening), but is subject to the same degree of adverse effects from treatment. No studies were found that examined the benefits of treating overt thyroid dysfunction identified by screening. Some patients found by screening to have overt hypothyroidism had symptoms of which the treating physicians were unaware, and in uncontrolled “pre/post” studies, reductions in high blood cholesterol levels were reported.
Evidence of Efficacy of Treatment for Subclinical Hyperthyroidism
Helfand found no controlled trials of the treatment for subclinical hyperthyroidism. Small observational treatment studies of patients with thyroid nodular disease reported improvements in bone metabolism and hemodynamic measures.
Evidence of Efficacy of Treatment for Subclinical Hypothyroidism
Helfand found no clinical trials of treatment of persons who were ascertained as having subclinical hypothyroidism through TSH screening. However, 15 randomized clinical trials of treatment with levothyroxine were found. Seven of these were excluded from analysis for a variety of reasons, such as not reporting clinical outcomes, including patients with overt disease, or being conducted to test alternative thyroxine preparations. The remaining eight trials are discussed at length and presented in Helfand’s Tables B-3 and B-4.
Six of the eight trials were deemed to have several important methodological or conceptual problems, including small numbers of subjects; treatment of persons with previous hyperthyroidism, with or without Graves’ disease; lack of reports on important methodological details; follow-up durations too short to assess long-term benefits or adverse effects on morbidity or mortality; and absence of blinding. Only one of the studies was rated as high as “good” quality. Where symptomatic outcomes were evaluated, the evidence was decidedly mixed, with more studies showing no improvement than showing benefit. However, the methods of evaluating symptoms and outcomes were diverse. Many of the patients in these treatment trials were below the usual age of Medicare beneficiaries.
Patients in the remaining two trials began TSH levels in the normal range and thus were deemed not relevant to questions at issue here. In one, blood LDL cholesterol levels, which were elevated at baseline, were modestly decreased by treatment. In the other, patients with symptoms of hypothyroidism (but normal blood parameters) found a decrement in vitality associated with the active drug. Most of the observational studies of levothyroxine treatment were primarily to evaluate blood lipid-lowering effects. In general, small effects were seen in some studies, but not in others.
Adverse Effects of Levothyroxine Treatment
A clear description of the adverse effects of levothyroxine is available in textbooks and the drug package insert (Bartalena et al., 1996). These include nervousness, palpitations, atrial fibrillation, and exacerbation of angina pectoris. The randomized trials reviewed by Helfand did not systematically report adverse events, but some were reported incidentally and were of the types noted earlier. A systematic review of observational studies published from 1966 to 1997 found that replacement doses of levothyroxine were not associated with osteoporosis or any other long-term adverse effects. However, Helfand notes that suppressive therapy for thyroid cancer, goiter, or nodules has been reported to be associated with increased osteoporosis risk. Helfand also notes that “overtreatment” with levothyroxine is a potential problem, as about one-fourth of patients on replacement therapy have been reported to be on doses sufficient to suppress the serum TSH level to below normal. Observational data from the Framingham study
suggest that replacement therapy can increase the risk of atrial fibrillation in those with suppressed TSH levels. In the observational Study of Osteoporotic Fractures, low TSH levels in subjects taking levothyroxine were associated with increased osteoporotic fracture risk.
SUMMARY AND CONCLUSIONS
In assessing the balance of benefits and harms for both hyperthyroidism and hypothyroidism, the key uncertainties are the following questions: (1) Without screening, how long would thyroid dysfunction be undetected? (2) How much morbidity would undiagnosed thyroid dysfunction cause while undetected? (3) What are the harms of treatment in those who do not progress?
Based on the review by Helfand of the evidence base for screening for thyroid dysfunction, the Committee reached the following conclusions.
-
There is suitable evidence that individuals in the community with no history of thyroid disease can be identified through serum TSH screening and systematic medical evaluation with hypothyroidism and hyperthyroidism as defined biochemically. The prevalence of thyroid dysfunction is higher in persons over age 65 years than among younger adults.
-
Individuals with subclinical hyperthyroidism appear to be at greater risk of atrial fibrillation and possibly to its downstream consequences, such as stroke. Also, individuals with subclinical hypothyroidism are more likely to have functional abnormalities in cardiac contraction, although whether this translates into long-term risk of congestive heart failure or other cardiac conditions is uncertain. The evidence that persons with subclinical hypothyroidism are clinically different from persons who have biochemically normal thyroid function—either with respect to symptoms or blood lipid levels—is inconclusive.
-
No randomized trials examining the treatment of overt or subclinical hyperthyroidism (Figure 1, Effects B1 and D1-D3) could be identified. As noted in Chapter 3, subclinical hyperthyroidism will frequently resolve spontaneously. While treatment of subclinical hyperthyroidism effectively eliminates the biochemical condition, it usually leads to a hypothyroid state and to other adverse effects. Properly designed clinical studies are needed to determine whether treatment reduces the risks associated with hyperthyroidism, such as atrial fibrillation, and whether benefits outweigh adverse effects.
-
There are no trials of the treatment of subclinical or overt hypothyroidism in patients who were identified by serum TSH screening programs. Although thyroxine treatment manifestly eliminates the biochemical characteristics of subclinical hypothyroidism, evidence from controlled trials suggests that overall short-term (i.e., 6-24 months) treatment does not lead to any important clinical improvements. No long-term controlled trials of thyroid hormone replacement have been published; the long-term clinical benefits and adverse effects are un
known. There is evidence that about one-fourth of patients on levothyroxine are overtreated, and these patients may be at increased risk of cardiac complications and osteoporotic fractures.
REFERENCES
Bartalena L, Bogazzi F, Martino E. 1996. Adverse effects of thyroid hormone preparations and antithyroid drugs. [Review] [98 refs]. Drug Safety 15(1):53–63.
Cuckle HS, Wald NJ. 1984. Principles of screening. In: Antenatal and Neonatal Screening. Oxford: Oxford University Press.
Kaplan GA, Haan MN, Wallace RB. 1999. Understanding changing risk factor associations with increasing age in adults. [Review] [62 refs]. Annual Review of Public Health 20:89–108.
United States Preventive Services Task Force. 1996. Screening for Thyroid Disease. Guide to Clinical Preventive Services, 2nd edition. [Online]. Available: http://hstat.nlm.nih.gov/hq/Hquest/fws/T/db/local.gcps.cps/screen/Browse/s/50802/action/GetText/linek/285.












