Housing Standards: Development of Guidelines and the Process for Change
William J. White
The 1996 Guide for the Care and Use of Laboratory Animals (Guide) is the most recent version of seven editions of the document beginning with the 1963 edition. The 1963 Guide was developed by a committee of seven members and consisted of 33 pages divided into three sections, whereas the 1996 Guide was developed by a committee of 16 members and spanned 125 pages divided into five sections. The charge to the 1996 Guide committee was to develop a guidance document for laboratory animal care and use—not to develop regulations.
COMMITTEE FOR REVISION OF THE GUIDE
The writing of the 1996 Guide spanned five committee meetings and involved seven major drafts with numerous minor drafts developed over a 2-year period. The major sections of the Guide were selected based on the principal components of an animal care and use program. Sub-committees of the parent committee prepared drafts of sections for full committee review and discussion. Literature searches were provided by the National Agricultural Library. The final document underwent two rounds of external review before being published.
The charge to the committee and the committee’s approach set the tone for the document. It was thought that previous guidelines were accepted and generally were serving well; hence the committee was charged with updating and improving, as well as addressing any short-
comings. It was believed that radical departure would probably be difficult to justify, and the committee wanted to be certain that the guidance was compatible with existing regulations in the United States. It was clear to the committee that a broad vision was necessary because the document would have to be applied to diverse units, settings, and situations. The recommendations made in the Guide had to be balanced between science, animal well-being, ethics, and resource requirements. The committee believed it was essential to reaffirm the institutional animal care and use committee (IACUC) as the principal local oversight mechanism and empower it to administer a performance-based approach.
COMMUNICATING THE PERFORMANCE-BASED APPROACH
The performance-based approach specifies the desired outcome and provides criteria for assessing the outcome, but it does not specify how to achieve the outcome. In constructing the Guide, it was appropriate in some instances to provide examples, but to emphasize clearly that multiple methods could be acceptable and the choice of method had to be adjusted, based on circumstances. Given the lack of data on which to base detailed guidance in many aspects of animal care and use, the performance-based approach used in the Guide encouraged the development of data that could be used to improve animal care and use.
The committee began the process by a careful review of the existing guidance and regulations. It considered whether there were problems with existing guidance and whether new science or technology was available that needed to be addressed. It examined the literature, especially that generated since the last Guide was published, to determine whether new peer-reviewed literature provided significant information that required inclusion. The committee thought it was important to use all sources of information in constructing the Guide. In particular, opinion and data from the public, as well as the affected community, were actively sought through public meetings and written comments. Every committee member attended the public meetings and read all written materials submitted.
The quality of information available to the committee varied, from statements of opinion, to unpublished reports, to comprehensive peerreviewed scientific studies. The committee analyzed the peer-reviewed literature, which varied considerably in adequacy and approach. In some cases, only a single study under a defined set of conditions using limited measures was available. In other cases, a series of studies with multiple measures exploring a range of conditions or practices could be found, whereas in other cases, published reports could be found that surveyed large numbers of animals with limited measures and limited control.
Guidance developed by the committee had to be qualified, based on the quality of the information available. Of particular concern was the general applicability of findings across multiple strains, age, sex, and species. In addition, the magnitude of the changes found and their potential impact on study variation and animal well-being had to be weighed carefully. Some studies used insensitive measures that could be confounded by other variables. Data from such studies were interpreted to have some value, but recommendations had to be tempered. Often there were only a limited number of studies that directly addressed a particular topic, and these studies often were unlinked or in some cases were conflicting. In a number of cases, studies could be classified as proof of principle, but with no exploration of mechanism or their general applicability, and they seldom could be verified independently. In a number of instances, professional judgment had to fill in gaps where studies were lacking. Overall, however, if there was little evidence available, the committee avoided the temptation to make assumptions and to extend conclusions beyond available data. Of concern to the committee was that any guidance had to fit practically and logically into existing animal care programs and had to be of sufficient impact/importance to suggest action.
The committee spent time analyzing how new guidance would apply to different animal care and use situations. It was important to assure that the guidance made sense and, if there were exceptions, to indicate how common they were and why they occurred. The committee also tried to determine whether the proposed changes in guidance would generate unintended consequences. Thus, if a guidance recommendation improved one aspect of animal well-being at the expense of another, such as causing a potential increase in animal usage by the application of some forms of environmental enrichment, it was necessary for the committee to determine whether the trade-off was appropriate.
IMPACT OF RECOMMENDATIONS
The committee was also concerned about the impact of recommendations on the conduct of research or testing including a potentially increased risk of microbiological contamination. Implementation of recommendations that might contribute to microbiological contamination could invalidate many types of research and thus result in greater animal usage. The guidance provided in the Guide had to be achievable, such that the intended benefit would be proportional to the resources required. The committee felt that any changes proposed in the Guide had to be defensible at least in proportion to their specificity. For example, if cage space allocated per rat of a given weight was currently 23 in2 and it was suggested to change it to 30 in2, why was 28 or 35 in2 not chosen? It was clear
that more than good intent was required to make changes—in fact, good data were needed to be specific in making such changes.
The committee believed that the guidance had to be very clear. Any recommendations had to use terms that were clearly defined either within the document or within appropriate references. In many cases, the committee provided examples to clarify intent and to expand the meaning of terms. The committee also thought it appropriate to ensure that the characteristics of intended outcomes of the recommendations were clearly described. Overall, the tone of the document was an explanatory one in which the rationale behind the recommendations was given in some detail. The committee believed that wording was critical in developing the document. It avoided poorly defined or emotional terms. It used words to indicate importance as well as limits of application or knowledge. It reserved the word must for programmatic issues for which there was no other interpretation or method. Must was also used where there was overwhelming scientific information or ethical considerations. There are very few musts in the 1996 Guide.
The word should was used as a strong recommendation for achieving a goal, but it was clearly recognized that some individual circumstances justified an alternative strategy. Words such as can, might, could, recommend, and encourage were considered alternative verbs to indicate that the recommendations may have to be modified, multiple methods of achieving the outcome were possible, only limited information was available, or that the guidance may only apply to certain circumstances.
PRACTICAL ISSUES IN THE DEVELOPMENT OF GUIDANCE FOR HOUSING STANDARDS
One of the most difficult sections to construct in the Guide was the animal environment section, which included housing needs of laboratory animals. Although it was intuitive that the housing environment of laboratory animals somehow affects their performance and well-being, there was not a great deal of information available to provide very specific recommendations. In most cases, only unrelated proof of principle studies that demonstrated housing/environmental effects on animals or research results were available. In a number of studies, there were clear design flaws, which included the use of small numbers of animals, the use of a single species, stock, or strain, and the failure to dissect out confounding variables such as the effects of group size and animal density with respect to cage space effects.
Temperature and Humidity
A review of housing guidelines and published data pertaining to temperature and humidity reveals the gaps in our knowledge that make it difficult to specify precise environmental conditions. Most common laboratory animals are adaptive homeotherms and as such make anatomic, metabolic, and physiologic adjustments in response to their environment to maintain well-being. Environmental adaptation in both wild and laboratory animals suggests that consistency in environment may not be “normal” or perhaps even desirable. To demonstrate effects caused by temperature or relative humidity, it may be necessary to have a complex/ unique set of conditions present unless extreme and clearly unacceptable conditions are utilized. For example, testicular degeneration/infertility in mice has been shown to occur when temperatures exceed 83°F within the secondary enclosure.
It would appear that the rationale for specifying any temperature or relative humidity conditions within the laboratory, other than avoiding extreme conditions that clearly could cause harm to the animals, would be to control research variation caused by unpredictable adaptation to housing conditions. The adaptive processes may, in fact, serve the animals quite well, but those are the processes that may interfere with research results. The question then remains as to how much variation due to adaptive processes is acceptable and, hence, what limitations must be placed on temperature and relative humidity. It also begs the question of why such changes would not be sorted out by the use of appropriate controls. Clearly, a number of variables would affect these adaptive processes, including the type of housing (e.g., pen, run, open cage, microisolation cage, isolator), the type of ventilation system used within the primary and secondary enclosures, the specifics of cage/room coupling of ventilation, as well as stratification of temperature and relative humidity within the room itself.
Interaction of other environmental factors with the thermal regulatory behavior of rodents is also an important consideration. Substantial existing data demonstrate that singly housed mice prefer ambient temperatures between 28°C and 30°C whereas group-housed animals prefer temperatures between 24°C and 27°C. The ability of group-housed animals to share metabolically generated heat by huddling together explains the differences in selected ambient temperatures between group- and singly housed animals. The resultant effective ambient temperatures in both group- and singly housed mice are compatible with the estimates of thermal neutral zones for mice of 28°C to 30°C. This range is at variance with the human comfort zone of 22°C (± 2°C), which appears to correspond more closely to temperatures recommended within guidance documents.
Bedding
Recent studies have shown that bedding types that allow burrowing or nesting allow operating ambient temperatures to be increased from an ambient of 22°C to an effective ambient of 29°C. By contrast, bedding that allowed only resting on its surface, but not burrowing, increased temperatures by 2 to 4°C depending on the bedding type. These findings beg the question of whether rodents are using bedding for thermal regulation rather than for psychological enrichment, as suggested in some guidance documents. It is possible that we are recommending the correct thing in terms of the use of bedding or nesting materials for the wrong reasons.
Cage Space
Cage space is another environmental parameter for which there are very few specific data. Providing an adequate amount of cage space is generally thought to be important for animal well-being. Unfortunately, guidelines have been established based on consensus, surveys, some data, and appearance to observers. Although it is intuitive that relationships must exist between cage space and parameters thought to indicate overcrowding, these relationships have never been studied well and are prone to be influenced by a wide range of variables. Most studies have not separated out the effects of group size (number of animals in an enclosure) from density (space provided to each animal regardless of the number in the group). These effects are independent of each other and may be affected by sex and the age of the animals in the enclosure. In general, single housing appears to be the most likely to elicit negative effects on the animals. Recommendations for cage space were developed in the early 1960s and published in the first ILAR Guide as suggestions, not standards. In 1969, a density-based set of recommendations using six weight categories was developed in all likelihood from a survey of common practices coupled with limited unpublished data. Although this guidance was modified slightly in ensuing years, principally by adjusting weight ranges and providing for very large animals, the body weight/space relationship remains almost linear, which is suspicious. European regulations currently in effect express this same relationship simply, and often use a continuous rather than a discontinuous format. Newly proposed European regulations deviate significantly from this space allocation, but the changes do not appear to be based on peer-reviewed literature specific to species and weight.
Significant data exist to establish that rodents are thigmotaxic, i.e., they prefer to be along the edges of cages rather than in the exposed center of the cage. Moreover, they appear to utilize hiding places/shelters.
Specifying cage space requirements by virtue of density guidelines appears to be contrary to these natural behaviors because it disproportionately increases the unused center portion of the cage versus the used perimeters of the cage. It is quite probable that cage space, like temperature, cannot be specified very tightly and that a range of available floor space or usable surface is acceptable depending on the type and quality of the space. There is clearly a need to explore requirements of rodents and other animals with respect to group housing because it appears that there are complex relationships that differ with group size. Sufficient evidence exists to suggest that enclosure design or complexity can alter space requirements.
NEED FOR ADDITIONAL SCIENTIFIC DATA
Clearly, much more information needs to be generated before any changes are made in guidance. Complex interactions need to be more clearly understood and considered across a wide range of applications of these guidance documents. It is unlikely that a very defined amount of critical space can be shown to be an absolute requirement, and it is unlikely that relationships are going to be linear. Key information is not available on topics as simple as occupied floor area versus body weight. It is difficult to conceive how guidance documents can be updated without a great deal more peer-reviewed information. At the very least, in generating such information, more than proof of principle studies alone need to be done, and it is imperative that multiple parameters be measured and that both positive and negative controls be provided. It is also essential that there be consideration of confounding variables and confirmation of findings under field conditions. There should be some ranking of physiologic/metabolic and behavioral significance because simply describing that a condition exists and is statistically significant may not be adequate justification for providing for all eventualities in animal housing.
Revision of Appendix A to the European Convention ETS 123: The Participants, the Process, and the Outcome
Derek Forbes
Papers presented by earlier speakers have emphasized that the “Guidelines for accommodation and care of animals” presented in Appendix A of the Convention have proven to be very useful and have been widely applied. However, since 1976 when the Convention was first applied, scientific knowledge and experience have advanced considerably. Moreover, there has been an increased public interest in and awareness of animal usage in experimentation and a regard for their welfare. The author as a representative of a nongovernmental organization (NGO; see below) recognizes and acknowledges the importance of the participation and input from the wide spectrum of interested parties who have contributed to the process in satisfying, to the extent possible, the needs of animals used in research, those who work with them, and the public on whose behalf the work is done.
The participants who are involved in the Working Party preparing for Multilateral Consultation of Parties to the Convention include the following:
-
Parties: Belgium, Netherlands, Cyprus, Norway, Czech Republic, Spain, Denmark, Sweden, Finland, Switzerland, France, United Kingdom, Germany, European Community, and Greece.
-
Signatory States: Bulgaria, Ireland, Portugal, Slovenia, and Turkey.
-
Observers—Member States: Austria, Croatia, Hungary, Italy, and Malta.
-
Observers—Nonmember States: Australia, Canada, Holy See, Japan, New Zealand, and the United States of America.
-
Participants Who Are Experts from International Organizations: Canadian Council on Animal Care, European Biomedical Research Association, European Federation of Animal Technologists, European Federation for Primatology, European Federation of Pharmaceutical Industries and Associations, European Science Foundation, Eurogroup, Federation of European Laboratory Animal Breeders Associations, Federation of European Laboratory Animal Science Associations, Federation of Veterinarians of Europe, Institute for Laboratory Animal Research, International Council for Laboratory Animal Science, International Society for Applied Ethology, and World Society for Protection of Animals.
An examination of the list reveals first that all the organizations represented were nongovernmental, pan-European, or internationally recognized. Second, the organizations included those whose primary concerns were research based, or who had expert knowledge of the science and care of animals used in research, as well as those who concentrated on the protection of animals used in the laboratory and the ethological restrictions that such use imposed on the animals. Although the interests may appear diverse, experience gained from working alongside persons from such groups has shown that everyone involved was very aware of the paramount need to satisfy the animal’s welfare and ensure its well-being.
During the course of the revision, the process has developed and evolved in content from that which was included in the original Appendix A. The strategy was determined within the working party by the member states. Initially, the general part of the appendix was updated. Thereafter the sections dealing with species of animals most commonly used in research and that had been included in the original convention were revised. However, it was realized that other animals were used in research for which there were no agreed-upon standards within Europe. This realization led to the decision to include all of the following groups within the remit of the revision. The full list of species now includes rodents and rabbits; dogs, cats, and ferrets; nonhuman primates; birds; farm animals (sheep, goats, cattle, horses, (mini)pigs); fishes; and amphibians and reptiles.
A group of experts drawn from the NGOs considered each of the groups listed. The constitution of each group included individuals representing the diverse interests of the experts as described above. The format inevitably was conducive to change because it was necessary to reach some consensus within each of the working groups. The overriding prerogative was to try to achieve an enriched environment that satisfied the ethological needs of the animal, with special attention being given to the
most appropriate implementation for the species concerned. Because most of the species are social animals, it was agreed that interaction with conspecifics by group housing was of paramount importance. Although some form of containment in accommodation is usually inevitable, the best possible utilization of space in relation to the animal’s natural activities was a second important criterion. Some variety within the environment was also crucial to provide stimulation.
Each group of experts produced a report on one or more species that was then presented to the next meeting of the Working Party. There it was debated and amended, or additional information was requested, before it was submitted back to the expert group for their further consideration. In some cases, this cycle has been repeated several times, often with each stage reversing or revising issues discussed at the previous meeting. It should be noted that during the protracted process, there has been a war of attrition between interest groups, the outcome of which has been an eventual meeting of minds in consensus and the adoption of standards that are reasonable and accepted.
Faster progress was made after the introduction of a Drafting Group. This group was composed of a small number of representatives of the national authorities together with the Secretariat. The group met between meetings of the Working Party and rationalized the output of the previous meeting. The groups of experts produced reports with varying formats that the Drafting Group standardized, which greatly facilitated progress. For example, all of the environmental standards that were common to all species were described in the general part of the Appendix, leaving only those specific to a particular group to be mentioned in the species-specific text.
During the course of its several meetings, the Working Party has progressively “finalized” most of the species-specific reports as well as the General Part. Such finalization denotes that the document concerned will not be open for any further discussion. This discipline has been essential because the discussions could have been endlessly iterative, considering the breadths of opinions represented. Everyone involved in the process has been aware of the paucity of good scientifically based data that could be used to optimize the environment of animals used in research. They also recognize the difficulty of producing such data. Therefore, the outcome of the process will be a consensus of those with a genuine interest and knowledge in the subject. Although it will result in changes that will have a financial cost, the process overall should be seen as the best way to satisfy the needs of science and politics in improving the welfare of animals used in essential research.
Breakout Session: Rats and Mice
Leader: Axel Kornerup-Hansen Rapporteur: Rosemary Elliott
The purpose of this session was to elicit different points of view, based on the following set of questions and issues introduced by the breakout leader. Brief comments of participants during the general discussion are provided below. Two of the participants in this session, Abigail Smith and Jan Ottesen, were invited by the breakout leader to present the results of studies conducted in their laboratories. The summaries of these presentations also appear in this section.
Why change the guidelines?
What is the basis for creating new guidelines?
-
Scientific proof
-
Experience
-
Good/best practice
Define good/best practice.
-
The European guidelines deal with these issues.
-
Space for mice changes only for young ones, which have increased space.
-
Space for rats shows more change, increasing 35%, especially during experiments, primarily as an increase in average height. The rationale for this observation was not that more space creates better welfare, but allows for enrichment.
Is there a need to change existing guidelines for rats and mice?
The most important issues to be considered are enrichment, solid flooring, and social housing.
COMMENTS FROM AUDIENCE
Several members of the audience were rather skeptical about several aspects of enrichment. It was suggested that many enrichment items, such as houses and tubes, can be used without increasing the space. Because these objects also can provide extra space for exploration of the mice or rats, they themselves increased the useable space. The assumption that enrichment increases an animal’s well-being was questioned. It was suggested by participants that scientific data should be sought in order to avoid assumptions. Along these lines it was suggested that animals be observed for a 24-hour period to determine the use made of the enriching items and to attempt to assess their value. Another suggestion was to rearrange the space, by adding structures. One cage designer indicated that he keeps floors clear, but adds a resting shelf or feeding structure.
One speaker expressed concern about how the meeting was progressing and would like to see more science-based knowledge on all issues. Another speaker introduced the issue of the impact on the science in which the animals are used, and asked whether scientists had had input on the decisions. There was discussion on defining an optimal enrichment device and whether commercial breeders were expected to use enrichment and whether additions to bedding such as nestlets were acceptable.
Several speakers expressed concern about what happens to animals during data collection, in contrast to animals in breeding and stock colonies. For instance, what would be the effect of the lack of enrichment objects in metabolic cages, particularly for animals used to enrichment? Others wished to know the effect of modifying cages on experimental results and whether one should be focusing on the total environment for the animal.
The Chairman indicated that the revised Council of Europe Convention represented minimal guidelines rather than regulations. He also indicated that justifications for not following the guidelines could be presented.
Examples of Issues/Questions Requiring Scientific Evidence
-
Rodents dislike wire floor.
-
Do rats have a need for gnawing, or is it an escape mechanism?
-
Is there an effect of noise (e.g., music) on animals?
Good Practice
-
It is difficult to base some aspects of good practice on specific factors other than experience. If we do not have enough data to change guidelines, we should ask the following:
-
Is it acceptable to use good practice?
-
Should the guidelines then remain the same?
COMMENTS FROM AUDIENCE
One participant indicated that we have not discussed work of experimenters who say we need to improve on the barren cages. Others indicated that, again, we need more data. We need to identify knowledge gaps and develop funding for experiments to address these gaps. In the meantime, there should be a moratorium on changes to the Guide. The idea “Let’s do something” is a bad idea when there are no data to decide what to do. One person suggested that we continue using the current cage as we try to improve the environment.
After the two presentations that appear below (by Dr. Smith of work of Drs. Mabus and colleagues, and by Dr. Ottesen), the Breakout Group summarized as follows:
-
The diversity of guidelines, regulations, and traditions in different countries must be acknowledged. In publications, the set of guidelines/ regulations under which the animal experimentation was conducted should be specified.
-
The treatment and conditions for laboratory animals should be evaluated regularly as new information becomes available. Animals have the same needs, wherever they live, and efforts must be made to conduct research in the best interest of the science and the animals.
-
It is premature to consider global standardization in the absence of scientific data.
-
Standards for maintaining animals should be posted, to eliminate the need for constant monitoring of research performed with animals. Flexibility to allow for professional judgment is critical.
-
There is a recognized element of fear of being forced into global standards, which implies a push toward increased regulation. Many feel the Canadian system is a better oversight model to emulate because of its flexibility.
-
There is concern about the acceptability of studies, particularly those that are based on animal preferences. Animal preferences do not always reflect the best welfare and scientific needs.
Effects of Housing Density and Cage Type on Young Adult C57BL/6J Mice
Sarah L. Mabus, Abigail L. Smith, Jason D. Stockwell, and Cameron Muir
(Presenter: Abigail Smith)
The Guide for the Care and Use of Laboratory Animals (the Guide) (NRC 1996) specifies floor space requirements for laboratory mice of different weights. All cages must be at least 5 inches high. Floor space requirements (per mouse) are designated as at least 6 in2 for mice less than 10 g, 8 in2 for mice up to 15 g, 12 in2 for mice up to 25 g, and more than 15 in2 for mice weighing more than 25 g. The few peer-reviewed publications that address floor space needs of laboratory mice suggest that mice can be housed at densities higher than those recommended in the Guide and that mice housed at higher densities are healthier and less aggressive than mice housed at lower densities (Fullwood and others 1998; McGlone and others 2001; Van Loo and others 2001).
Rodent population densities have been shown to alter a number of normal and experimental parameters. In general, provision of less floor space either had no effect or was beneficial, resulting in enhanced immune responses and reduced mortality and aggression. Our study was designed to reveal how floor space and cage type might influence several parameters in young adult C57BL/6J (B6) male and female mice. The indices we studied were survival, aggressive behavior or injuries, body weight, food and water consumption, cage microenvironment (in-cage ammonia and CO2 levels, temperature, and relative humidity), hair loss (a commonly observed characteristic in B6 mice, particularly females), urinary testosterone concentrations, and microscopic evidence of ammonia damage to nasal passages and eyeballs. We housed the mice in three
readily available cage types that had different amounts of floor space. Populations of 4-week-old B6 mice were housed for 8 weeks in each cage type at four different densities—one compatible with recommendations in the Guide (approximately 12 in2 per mouse) and three higher densities. A second, 4-week study was performed to determine whether recently weaned B6 male and female mice could be housed with even less floor space (reduced to 3.2 in2 per mouse). We conclude that male and female B6 mice between the ages of 4 and 12 weeks can be housed with 5.6 in2 of floor space per mouse without ill effect. This is approximately half the floor space recommended in the Guide.
METHODS
The cages we used were Thoren #1 (“shoebox,” area = 67.6 in2) (Thoren Caging Systems, Inc., Hazleton, PA), Thoren #2 (weaning cage, area = 112.9 in2), and Thoren #3 (duplex, area = 51.7 in2 per side). A total of 540 mice of each sex were included in the 8-week study, and 660 mice of each sex were included in the 4-week study. The densities are coded as follows: (1) = 12.9 in2 per mouse; (2) = 8.6 in2 per mouse; (3) = 6.6 in2 per mouse; (4) = 5.6 in2 per mouse; (5) = 4.5 in2 per mouse; (6) = 3.8 in2 per mouse; and (7) = 3.2 in2 per mouse.
RESULTS
Eight-Week Study: C57BL/6J Mice House in Three Cage Types
Animal Health. All 1080 B6 mice that began the study survived, and we did not observe any aggressive behavior or injured mice. The mean weights of mice at the termination of this experiment were (± standard error [SE]) 20.4 ± 0.6 g and 29.8 ± 0.8 g for females and males, respectively. The incidence of alopecia among B6 female mice used in this study was relatively low, varying from 0 to 6% per treatment group, and was unrelated to cage type or housing density.
Microenvironment in Cages Housing C57BL/6J Male Mice. Ammonia levels were significantly affected by density. The levels at densities 3 and 4 significantly exceeded those in the two lowest densities and there was not a cage effect. In general, carbon dioxide concentrations increased with increasing densities. Mean CO2 levels varied two-fold, ranging from 2733 to 5349 ppm, and were not affected by cage type. Temperature increased with increasing density, but mean temperatures varied 4oC and did not exceed the recommendation in the Guide. Mean relative humidity was reasonably constant across densities for each cage type.
Urinary Testosterone Concentrations for Male and Female C57BL/6J Mice.
Housing density had no effect on urinary testosterone levels of mice housed in any of the three cage types.
51.7-in2 duplex cages. Mean female urinary testosterone levels (+ SE) significantly increased from 1.82 + 1.10 (baseline) to 4.35 + 2.37 ng/mg of creatinine by the end of the 8-week study, and male testosterone levels decreased (not significantly) from 2.86 + 1.16 to 1.87 + 0.91 ng/mg of creatinine by 8 weeks.
67.6-in2 shoebox cages. Female mean urinary testosterone levels (± SE) increased from 1.29 + 0.86 ng/mg of creatinine (baseline) to 3.96 ± 1.22 ng/mg of creatinine (8 weeks), whereas male testosterone levels decreased from 3.19 ± 1.06 ng/mg creatinine (baseline) to 1.54 ± 0.71 ng/mg of creatinine (8 weeks). Both changes were statistically significant.
112.9-in2 weaning cages. A single regression described the testosterone data from the weaning cages for all densities and both sexes. There was no significant difference in male or female mean urinary testosterone output between the baseline and 8-week samples. Unlike the results for males in 51.7 in2 duplex or 67.6 in2 shoebox cages, urinary testosterone levels increased for males between baseline and 8 weeks, although not significantly.
Four-Week Study: C57BL/6J Mice Housed at Higher Densities
Because we observed no deleterious effects of housing 20 C57BL/6J mice in 112.9 in2 weaning cages for 8 weeks with 5.6 in2 per mouse, we followed up with a 4-week study that evaluated the same parameters for mice provided with even less floor space—3.2 in2 per mouse. We monitored the microenvironments of both male and female mice twice weekly in this study, and we also assessed both the noses and the eyeballs of selected mice microscopically at study termination.
Animal Health. Of the 1,320 mice that began this study, two mice were culled and one was found dead within the first 10 days of the study. These mice were replaced. No effect of density on the rate of weight gain was observed, and food and water consumption was not different among the densities. Density did affect the incidence of alopecia in female mice, which developed hair loss in one of six cages at density 4, two of six cages at densities 5 and 6, and five of six cages at density 7. Male B6 mice developed hair loss in one of six cages at each of the four densities.
Microenvironment in Cages Housing C57BL/6J Male or Female Mice. Mean ammonia concentrations (± SE) increased significantly with each increase in housing density (12.6 ± 1.1 ppm, 20.7 ± 1.1 ppm, 43.4 ± 1.1ppm, and 139.8 ± 1.1 ppm at densities 4, 5, 6, and 7, respectively). Nasal passages from selected mice in each density were examined microscopically for ammonia damage and were found to be normal. Eyes from 13 randomly
chosen male mice were examined. The range of ammonia concentrations to which they had been subjected was 23 ppm to 410 ppm, including five mice from cages containing 198 to 399 ppm of ammonia. All of the examined eyes were histologically normal.
Carbon dioxide concentrations among the densities were not significantly different. Least squares means were greater for males (4,335 ppm) than females (3,103 ppm; p < 0.05). Least squares mean temperatures (± SE) were significantly higher for densities 5, 6, and 7 (28.3°C ± 0.2°C, 28.8°C ± 0.2°C, 28.4°C ± 0.2°C, respectively) compared with density 4 (26.6°C ± 0.2°C). The least squares mean relative humidity value (± SE) for the highest density (density 7:57.1% ± 1.0%) was significantly higher than the two middle densities 5 and 6 (density 5:53.0% ± 1.0%; density 6:52.9% ± 1.0%) but not different from the lowest density (density 4:56.3% ± 1.0%).
Urinary Testosterone Concentrations for Male and Female C57BL/6J Mice. Mean urinary testosterone levels were unrelated to housing density and were higher for males than females (p = 0.002). The mean baseline concentration for males was 14.4 + 4.1 ng/mg of creatinine, which increased to 26.8 + 12.6 ng/mg of creatinine by the end of the fourth week. For females, the baseline was 10.7 + 3.9 ng/mg of creatinine, which increased to 15.2 + 5.2 ng/mg of creatinine by week 4.
DISCUSSION
Based on gross measures, the health and well-being of the mice used in these studies were not affected by cage type or housing density. There were no significant differences among mice housed in three cage types, at any of the seven densities, in growth rates or food and water consumption. We did not observe aggressive or injurious behavior, and all mice survived the 8-week period of the first study. The incidence of alopecia among B6 female mice ranged from 0 to 6% in the 8-week study and was not associated with a particular cage type or housing density. The incidence of alopecia in the 4-week study was density dependent, with five of six cages containing affected female mice at the highest density.
In the 8-week study, in-cage CO2 levels generally increased with density, and there were no apparent differences among the cage types—all reached maximum levels of approximately 5,000 ppm, the maximum allowable US workplace exposure limit during an 8-hour shifta or a 10-hour shift.b Increases in CO2 concentration would be expected at higher
densities because CO2 reflects the amount of respiration occurring within the cage. Mean temperatures generally increased with density, and on average, the difference between the mean high and low temperatures in each cage type was 2.5°C. The Guide indicates that temperatures ranging from 18 to 26°C are recommended for housing laboratory rodents. Exposure to a temperature higher than 29.4°C in animals not adapted to the high temperature could produce adverse clinical effects (NRC 1996). We have repeatedly measured in-cage temperatures at or exceeding 29°C and have not observed any adverse effects on multiple strains of mice. In this study, in-cage temperatures did not exceed 29°C. In-cage relative humidity was generally unaffected by cage density. The Guide indicates that relative humidity can vary widely, from 30 to 70%, and our results were well within that range.
In the 4-week study, average in-cage ammonia concentrations significantly exceeded 25 ppm at densities 6 and 7, reaching 43.4 and 139.8 ppm, respectively. The maximum allowable workplace ammonia exposure over an 8-hour period is 25 ppm.c,d Cages housing male mice had higher concentrations than those housing females. In-cage CO2 concentrations were independent of density in the 4-week study. Temperatures were higher in cages housing the three highest densities but did not exceed 29°C. Humidity levels were variable and there was no clear relation to housing density.
Male urinary testosterone levels either remained relatively constant or decreased slightly over 8 weeks in the primary study. For female B6 mice housed in any of the three cage types, urinary testosterone levels increased over the course of the 8-week study. Irrespective of cage type, housing density did not influence urinary testosterone output of male or female B6 mice, although week and/or gender did. In the 4-week study, male urinary testosterone concentrations were uniformly higher than female concentrations. As found in the 8-week study, neither housing density nor cage type influenced hormone concentrations. However, in contrast to the 8-week study, male hormone levels increased over the 4-week period. Female hormone levels increased in both studies. It may be noted that the testosterone concentrations in the 4-week study were substantially higher than those in the 8-week study. This interassay variation is expected and makes it essential that values to be directly compared must be the result of simultaneous assays.
Although the OSHA and NIOSH standards cited above indicate that workplace exposure to ammonia should not exceed 25 ppm over 8 hours
or 35 ppm over a 15-minute period, two factors can substantially reduce human exposure in animal facilities. First, when filter tops are removed from rodent cages, there is an immediate dilution effect by mixing with ambient air. Second, as is the case with Mus m 1 allergen exposure in mouse rooms (Schweitzer and others 2003), exposure can be greatly reduced by husbanding rodents on ventilated tables. In addition, the type of bedding that is used to house the mice can have a significant impact on in-cage ammonia concentrations (E. Smith and others, submitted). The finding that rabbits exposed continuously to > 400 ppm had opacities greater than one-fourth to one-half of their corneas (Coon and others 1970) was the basis for our microscopic examination of mouse eyes in the 4-week study, and lesions were not observed.
Results of our 8-week study indicate that only the least squares mean ammonia level (49 ppm) in density 4 in the 112.9-in2 weaning cages exceeded the concentration considered unhealthy for humans. This result may have been anomalous because the mean concentration for that density in the second study was 20.1 ppm. However, in the 4-week study, ammonia levels were very high (43.4 and 139.8 ppm) in cages housing mice with less than 4.5 in2 of floor space. None of the mice in either of the two studies showed evidence of ammonia toxicity, despite exposure to > 200 ppm in some individual cages in the 4-week study. Nonetheless, given the OSHA workplace standards for humans of 25 ppmc,d (Vigliani and Zurlo 1956), the use of ventilated changing tables should be encouraged in mouse rooms.
The floor space recommended in the Guide was based on best professional judgment at a time when there was very little peer-reviewed literature on the topic. We are attempting to apply scientific methods to learn the real floor space needs of mice. Thus, we recommend housing C57BL/ 6J male and female mice, aged 4 to 12 weeks, in cages that provide not less than 5.6 in2 of floor space per mouse. This housing translates to nine mice per side of duplex (51.7 in2) cages, 12 mice per shoebox (67.6 in2) cage, and 20 mice per weaning (112.9 in2) cage.
Our results and those of others (Fulwood and others 1998; McGlone and others 2001; Van Loo and others 2001) have consistently pointed to the same conclusion: Mice that are housed at higher densities tend to be healthier and less aggressive toward their cage mates. For this reason, it is necessary to re-evaluate the current guidelines in the context of what is known about this social species. Animal care should not be dictated by the anthropomorphic perceptions of animal caretakers and regulatory
bodies. Animal care staff almost always want to do what is best for the animals, but they may need to be educated in the area of rodent housing density. The role of the Guide is to ensure that laboratory animals are well treated and housed in a species-appropriate manner.
This housing density study and others reported in the literature have included only a few inbred mouse strains. Universal provision of the floor space needs of mice may be difficult, and strain variation is to be expected. We are currently using the same protocols described herein to evaluate the needs of young adult BALB/cJ, NOD/LtJ, and FVB/NJ mice; and we have data indicating that there are, indeed, differences (A.L. Smith, manuscript in preparation).
ACKNOWLEDGMENTS
We thank Drs. Richard Smith and Ralph Bunte for microscopic evaluation of eyes and nasal passages, respectively, and Drs. Beverly Paigen and David Threadgill for helpful comments during manuscript preparation. This work was supported by grant RR12552 from the National Center for Research Resources, National Institutes of Health; by a Henry and Lois Foster Foundation grant (ACLAM Foundation); and by funds from the Jackson Laboratory.
REFERENCES
Coon, R.A., Jones, R.A., Jenkins, L.J., Siegel, J. 1970. Animal inhalation studies on ammonia, ethylene glycol, formaldehyde, dimethylamine and ethanol. Toxicol Appl Pharmacol 16:646-655.
Fullwood, S., Hicks, T.A., Brown, J.C., Norman, R.L., McGlone, J.J. 1998. Floor space needs for laboratory mice: C57BL/6 males in solid-bottom cages with bedding. ILAR J 39:29-36.
McGlone, J.J., Anderson, D.L., Norman, R.L. 2001. Floor space needs for laboratory mice: BALB/cJ males or females in solid-bottom cages with bedding. Contemp Top Lab Anim Sci 40:21-25.
NRC [National Research Council]. 1996. Guide for the Care and Use of Laboratory Animals. Washington, DC: National Academy Press.
Schweitzer, I.B., Smith, E., Harrison, D.J., Myers, D.D., Eggleston, P.A., Stockwell, J.D., Paigen, B., Smith, A.L. 2003. Reducing exposure to laboratory animal allergens. Comp Med 53:487-492.
Van Loo, P.L.P., Mol, J.A., Koolhaas, J.M., Van Zutphen, B.F.M., Baumans, V. 2001. Modulation of aggression in male mice: Influence of group size and cage size. Physiol Behav 72:675-683.
Vigliani, E.C., and Zurlo, N. 1956. Experiences of the Clinical Del Lavoro with maximum allowable concentrations of industrial poisons. AMA Arch Ind Hlth 13:403.
New Housing Standards for Rats and Mice Developed with Focus on the Needs of the Animals
Jan L. Ottesen
INTRODUCTION
The Expert Group on Rodents and Rabbits of the Council of Europe has clearly stated that its objective is to meet the needs of animals. As part of the extensive background information in the Preamble to their proposal for revision of Appendix A of the European Convention ETS 123, the following statements appear (Stauffacher et al. 2002):
The exact numeric values for minimum cage sizes and heights as well as for maximum stocking densities can never be scientifically evaluated and “proved.” Working out minimum requirements with respect to animal welfare and to supposed well-being of laboratory animals is a political question. Nevertheless, the decision-making process should be based first and foremost on sound arguments on the biology of species and strains in question. During discussion it should be carefully distinguished between biological facts, scientific evidence and practical experience on one side and ethical principles of animal protection and the assessment of economical and political reason on the other side.
It is important to bear this position in mind during any discussion on “Science-based Guidelines for Laboratory Animal Care.”
In 1999, at the Third World Congress on Alternatives and Animal Use in Bologna, Coenraad Hendriksen (2000) proposed the “Three C Principles”—Common sense, Commitment, and Communication. Dr. Hendriksen
described these principles as drivers toward implementation of the principle of the “Three Rs”—Refinement, Reduction, and Replacement (Russell and Burch 1959).
Of course, optimal guidelines for laboratory animal care should be science based. However, scientific proof is often not possible to obtain (e.g., determination of the exact numeric values for minimum cage sizes and heights). Furthermore, scientific proof is often used defensively as a prerequisite before introducing new environmental enrichment ideas, which unfortunately often stops further progress. At Novo Nordisk, we have tried to use common sense when we develop new housing facilities for our experimental animals, based on expert views on animal needs. We do not yet have scientific proof that all of our initiatives have resulted in increased animal welfare, yet one could argue that neither do we have proof that demonstrates the opposite. In the absence of proof, we provide what common sense tells us is good animal welfare.
Environmental enrichment is one of the major ways of trying to improve the welfare of laboratory animals in our care. Freedom of movement and a structured environment that allows natural behavioral patterns of the animals are considered an enrichment of the environment. However, rats in particular are highly adaptive, and it is difficult to prove that environmental enrichment does in fact increase the welfare of these animals. It seems obvious from studies both of rats in captivity and of laboratory rats released to semi-natural conditions (Berdoy 2002) that they rear to perform grooming and to look out, as part of their natural behavior. In our new rat cage system, which has been implemented during the testing period, we have increased the height of the cage from 18 to 30 cm. In the future, we plan to study the potential benefit to the rats of the new housing conditions (see below). In the meantime, we believe that the welfare of the rats is not jeopardized under current housing conditions.
FROM GUIDELINES TO LAW
In 1998, while revising the 1986 European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (ETS 123 Guidelines), the Council of Europe established a number of working groups to review the different animal species used as experimental animals. Based on proposals from these expert groups, species-specific sections have been prepared. In addition, extensive background information containing scientific evidence as well as practical experience has been compiled to support the expert groups’ proposals. For most of the species, the proposals for species-specific provisions have been finalized, although officially, they are still considered draft versions, not currently in effect (www.coe.int/animalwelfare).
In Denmark, the government decided not to await the final revision of ETS 123. In August 2003, the Ministry of Justice issued a government order that authorized all of the above-mentioned “finalized” draft versions, thereby changing the content from guidelines to law. It should be noted, however, that any necessary major changes in building constructions have until 2007 to be in place.
NEEDS OF RATS AND MICE
At the end of 1999, in an effort to identify and establish the most important needs of mice, rats, guinea pigs, rabbits, and dogs in their natural habitat, Novo Nordisk A/S and the Danish Animal Welfare Society invited internationally recognized animal welfare experts to participate in several workshops. Based on the results from those expert workshops, new laboratory animal housing prototypes have been developed that take the needs of animals into consideration much more than in prior years (Ottesen et al. 2004).
Mice
Mice are social animals that prefer to be with conspecifics. They have a need to live in stable, harmonious groups, although it might be necessary to separate adult male mice to avoid their aggressiveness. Mice need to be able to rest, hide, and build nests. They also have a need for complex and challenging surroundings. Mice are nocturnal animals, and therefore need darkness.
At Novo Nordisk, we believe that the needs of the mice can be accommodated for the most part in types III and IV macrolon cages (800 and 1800 cm2, respectively). The optimal cage size will depend on the weight of the mice, the group size, and the extent of environmental enrichment. Further improvement of the cage may be accomplished by using a commercially available lid that is 7 cm higher.
Rats
Rats are social animals that need a structured and enriched environment with access to both hiding and viewing places. They need space for rearing, climbing, gnawing, digging, and grooming.
A few years ago, Novo Nordisk implemented the European type IV macrolon cage system (a cage with 1800 cm2 of floor space) as the standard cage system for rats. This cage has been enriched further in the replacement of traditional lids with specially designed “elevated lids,” increasing the height of the cages from 18 to 30 cm. This replacement
affords the rats the possibility of visual control of the environment and space for rearing and grooming. A shelf in the cage provides the rats with additional possibilities for exploring, exercising, jumping, and looking out, as well as improved hiding possibilities (Figure 1).
DISCUSSION
Economy is sometimes used as an argument against larger cage sizes for experimental animals, and various calculations are used to support the argument. The annual total budget for the animal facility is often used as reference. Even though the necessary investment in new cage systems is 25, 50, or 100% of the facility’s annual budget, it should be noted that this expense is a one-time investment. Compared with the annual budget of the company or institution, that expense most likely totals a percentage that is less than one digit.
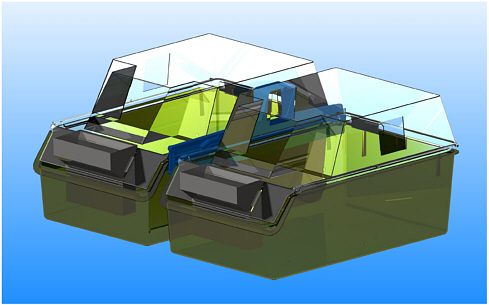
FIGURE 1 (A) The European type IV macrolon cage (1800 cm2) is used for housing rats. A new, high lid increases the total height of the cage from 18 cm to approximately 30 cm, which allows the rats to rear and perform grooming. The possibility of connecting two or more cages with a tunnel has been developed and will allow larger group sizes or more room for the rats. (Figure 1A: Courtesy of Scanbur BK.)

FIGURE 1 (B) Provision of a shelf gives the rats more choices and provides both hiding and nesting opportunities (nesting material partly removed on photograph).
At Novo Nordisk, we believe that our new cage systems for mice and rats will cover their basic needs much better than the smaller, traditional cage systems. Beyond ensuring the conditions we believe greatly improve the welfare of the animals, the new system appears much more pleasant and inviting and should therefore contribute to a broader acceptance of experimental animal use by the public.
It will be necessary for laboratory animal care guidelines to deal with minimum requirements. It is acknowledged that for some rodent studies (e.g., in research on brain development), less enriched cages may be required. For other studies (e.g., some feeding studies), single housing of the animals may be necessary. Nevertheless, using as examples the studies that require less enrichment is not a valid argument for housing all animals under minimal conditions.
If possible, housing standards should be science based; however if many parameters need to be scientifically proven to implement new housing standards, common sense should also be applied.
ACKNOWLEDGMENTS
Thanks are due the animal technicians in the Novo Nordisk Environmental Enrichment Committees for the commitment demonstrated in the excellent translation of expert advice from M. Stauffacher, R. Hubrecht, R. Murison, P. Hawkins, A. Kornerup Hansen, V. Baumans, M. Ritskes-Hoitinga, I.M. Jegstrup, and L. Lidfors into new housing systems for laboratory animals.
REFERENCES
Berdoy, M. 2002. The Laboratory Rat: A Natural History (Video/DVD). London: Oxford University.
Hendriksen, C.F.M. 2000. Replacement, reduction and refinement and biologicals: About facts, fiction and frustration. In: Balls, M., van Zeller, A.-M., Halder, M.E., eds. Progress in the Reduction, Refinement and Replacement of Animal Experimentation. Amsterdam: Elsevier Science B.V. p. 51-63.
Ottesen, J.L., Weber, A., Gürtler, H., Friis Mikkelsen, L. 2004. New housing conditions: Improving the welfare of experimental animals. ATLA 32(Suppl 1) (In Press).
Russell, W.M.S., and Burch, R.L. 1959. The Principles of Humane Experimental Technique. London: Methuen & Co. Ltd. p. 1-238.
Stauffacher, M., Peters, A., Jennings, M., Hubrecht, R.C., Holgate, B., Francis, R., Elliott, H., Baumans, V., Hansen, A.K. 2002. Future principles for housing and care of laboratory rodents and rabbits. Report for the Revision of the Council of Europe Convention ETS 123 Appendix A for Rodents and Rabbits. PART B. (http://www.coe.int/T/E/Legal_affairs/Legal_co-operation/Biological_safety,_use_of_animals/Laboratory_animals/GT123(2001)4%20Final%20PART%20B%20Rodents.pdf). Strasbourg: Council of Europe.
Breakout Session: Approaches for Implementing Current US and European Guidelines for Housing Standards for Dogs and Cats
Leader: Robert Hubrecht
Rapporteur: Thomas Wolfle
The session began with a review of dog cage or pen size guidelines, policies, and regulations in the United Kingdom and the Council of Europe (CoE 2001), in Canada (according to pertinent documents of the Canadian Council on Animal Care [CCAC; www://ccac.ca]), and in the United States (according to the Guide for the Care and Use of Laboratory Animals [NRC 1996]). Video examples were shown of stereotypies in dogs (e.g., cage chewing) and remediation with cage enrichment. A time-budget chart was presented showing that dogs spend large amounts of time on elevated resting platforms. Early socialization, habituation, and training were reviewed, and a behavioral technician playing with dogs in an enriched play environment was presented to set the stage for discussing space requirements.
Participants expressed the concern that too little space restricts group size and associated social interaction. They felt that the size of cages or pens should be judged adequate only when the following needs are accommodated: (1) species-specific activity and interaction; (2) enrichment, such that the animals are able to manipulate and control particular aspects of the environment; and (3) essential space for resting, temperature control, sanitation, and noise control. To discuss these concerns, the leader posed the following questions:
What factors led to the CoE’s large cage size?
Participants noted that smaller cages are required in the United States compared with those described in the CoE documents, yet few abnormalities are noted in the US cages when adequate socialization is provided. Participants seemed to agree that science-driven cage size recommendations are needed, and that ever-larger cage size mandates without supportable science are not justified. Focusing on cage size, rather than on behavior, forces the use of engineering, rather than on performance, standards.
How should environments be designed?
Participants indicated that performance-oriented approaches to cage size and environments are the most appropriate. The definition of these performance goals revolves around behavioral assessment and includes the considerations described below.
Some participants felt that spatial requirements for dogs in research should take into account the whole experimental program as well as species needs (although dogs are not different from other species in this respect). In addition, they felt that acceptable environments should allow the following: positive interaction with humans, harmonious conspecific social housing and exercise, and opportunities for reasonable species-specific behavior such as play and gnawing. Moreover, it was felt that the occurrence of stereotypies and other abnormal behavior should be minimized. The life-to-death experience was considered by some participants to be critical in ensuring high standards of welfare and high-quality science. One participant felt that standards at supply sources (i.e., breeders) are important, and there should be good communication between suppliers and users. Consideration should be given to the animals’ use in acute versus long-term studies, or survival studies in which the animal might be adoptable. Some participants felt that housing standards should take into account the adaptability of the species (although it is not clear that this factor is any greater than for some other species commonly used) and the variability of different breeds.
Should the length of time that a dog spends in a facility be a factor in the standards provided?
The focus of the question above was whether additional attention should be paid to addressing the needs of dogs used in longer-term studies. Some participants argued that dog housing should meet the needs of the species regardless of the length of time the animals might need to be housed in it. In addition, some opined that a multiplicity of standards for studies of different length might lead to unnecessary bureaucracy and confusion. The participants did not reach any consensus on this question.
Should standards be based on engineering, performance, or a mixture of the two?
Participants expressed the belief that a mixture of the two standards is best. There is a need to concentrate on performance standards because they often indicate the true success or nonsuccess of the dog enclosure. However, engineering standards are also useful to ensure the fulfillment of minimum standards.
How should changes be implemented?
When developing standards, participants indicated that it is necessary to begin with the requirements of the dog, and then move on to regulatory issues. More guidance is needed to evaluate the adequacy of environments in order to assess performance standards. It is important that any new standards be phased in, and that anticipated costs of implementing new standards be included in applications for funding. Many felt that more training of personnel is needed to recognize normal and abnormal animal behavior. Participants emphasized the need for consideration of the research mission in making changes. Involvement of the Principal Investigator in planning prospective changes in housing and enrichment is essential to ensure the high quality of ongoing data.
What arguments should be used in the process?
Many participants felt that changes should be made with due caution and based upon scientific evidence, professional judgment, and widely accepted “Best Practice.” To avoid reinventing the wheel, and to help harmonize international standards, it is advisable to refer to existing standards (e.g., NRC 1996, CoE 2001, and other nations’ codes of practice).
What does the public expect?
The view was expressed that science is carried out on the public’s behalf and indirectly with their consent in general terms. Therefore, the public has a particular concern for the welfare of species commonly used as companion animals, and standards used in the laboratory should reflect that fact.
How should economic arguments be weighed against biological arguments?
This question, stated another way, asks how the cost-benefit of animal research should be established. Some expressed the view that political decisions mandating engineering standards are likely to be unduly expensive without concomitant benefit to the animals.
Good science and good welfare go together, and ongoing assessments provide valuable answers to the cost-benefit question. Central to the
assessment is an understanding and application of science-driven performance standards. While many felt that global harmonization of animal care and use practices offers many potential benefits both to humans and to animals, some expressed the notion that harmonization would be unlikely if the engineering standards are politically motivated.
Is further science needed? How should it be directed?
Participants readily endorsed the need for additional research (such as is listed below) for better planning of research. However, some felt that greater use of sound experimental design and statistics is necessary to accommodate any new science-derived changes in the use of animals in research. Participants also recommended expanded use of biologic telemetry.
What are the needs for housing- and welfare-related research?
-
Areas described above.
-
Further investigation/research is needed regarding economical and practical ways of enriching the pen environment and of taking into account the needs and sensory modalities of dogs.
-
Relation between pen size, contents/structures/other enrichment, number of individuals, and behavior, preferably under carefully controlled experimental conditions.
-
Ways to ameliorate the negative effects of single housing. Do exercise plans for single-housed dogs actually make a difference?
-
Ways to prevent and manage aggression.
-
Determination of the effects of sound on dogs
-
Cost versus benefit of different toys and chews (no such studies have been attempted in canids).
-
Comparison of different methods of presentation of toys and chews, and determination of the effectiveness of various types of enrichment in single housing and in larger groups.
-
Influence of breeding for the selection of desirable characteristics.
-
Psychological and physiological effects of transport.
-
Design of metabolism cages to reduce their impact on dog welfare.
REFERENCES
CoE [Council of Europe]. 2001. European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (ETS 123). Future Principles for Housing and Care of Laboratory Rodents and Rabbits. Strasbourg: Council of Europe.
NRC [National Research Council]. 1996. Guide for the Care and Use of Laboratory Animals. 7th ed. Washington, DC: National Academy Press.
Breakout Session: Nonhuman Primates
Leader: David Whittaker
Rapporteur: Randall J. Nelson
Participants discussed the questions that appear below, in general consideration of the guideline revision process—Who, How, and Outcomes:
How should the next revision of the Guide for the Care and Use of Laboratory Animals (the Guide) (NRC 1996) be conducted?
Participants believed that expert groups chosen to address specific issues should conduct the revision of the Guide.
Is the lack of scientific knowledge in an area sufficient reason not to move forward with revision?
Session Leader David Whittaker believed not.
The Council of Europe (CoE) formed expert groups to deal with formulated guidelines, but eventually legislative recommendations for acceptance or nonacceptance of guidelines developed. The CoE participants believed that competent authorities (i.e., ministries) who implemented laws protected against conflict of interest.
Is a smaller group of experts more efficient in developing guidelines?
Participants affirmed small-group efficiency in contrast to the inertia of larger groups. They also opined that competent authorities should agree in advance to abide by the recommendations of the expert groups unless they vary radically from socially accepted norms. Moreover, they felt that: (1) expert groups should provide technical information early in
the process because subsequent change is difficult to implement; (2) large groups are more costly and difficult to manage; and (3) industry should be involved from the beginning, as should all of the stakeholders.
To what extent has social housing of nonhuman primates (NHPs) been accomplished in Europe?
Individuals in the group indicated that only about 1% of NHPs in the United Kingdom are singly housed and in those cases only for scientific reasons. Many felt that positive reinforcement training is beneficial in facilitating the handling of socially housed NHPs. Some also felt that regulations should influence, rather than require, compliance with factors such as social housing and that influence should be exerted to achieve “best/good practices.”
How does one determine best/good practices?
There was no consensus among the participants on this question. It was pointed out that the United Kingdom maintains a central clearing-house for best/good practices However, the UK does not promote the blanket utilization of justification of exceptions because doing so discourages the consideration of alternatives and refinements. Nevertheless, participants felt that there should be ready access to information about best/ good practices so that refinements can be made with a minimum of regulatory burden.
Further discussion elicited the following opinions from the participants:
-
Consistency in the guidelines and the authority to impose them is lacking in instances in which few scientific studies are available to sub-stantiate expert opinion and professional judgment.
-
The scientific and animal care communities need to convince competent authorities to be supportive of the need to gain more scientific data on factors such as cage sizes. They also need to convince society that such studies are worth the initial investment, because it may be perceived that funds are being redirected from health-related research.
-
The fundamental issues are economics and politics.
-
In studies that will have an impact on welfare issues, expert groups should agree on the range of experimental variables before the studies are performed, to avoid instances in which the scientific validity of the results is called into question.
How do we legitimize the science needed to fill gaps in the literature related to welfare issues?
Participants provided perspectives and outlined the following potential strategies for change:
-
Scientists and veterinarians need to be proactive with legislators from the very beginning to effect the change in societal attitudes needed to make funding of these studies more likely. The Medical Research Council gives monies for appropriate changes in approaches to be made, but if they give monies, the resultant changes are required. Thus, they have the “force of law” behind their support.
-
Data mining may be beneficial in obtaining needed scientific data with little or no cost. The data may already be available in some instances.
-
Veterinary outreach to investigators and to the community is an important way to educate others about best/good practices.
-
Qualified experts should attempt to identify the “bad science” in extant guideline documents, thus increasing the validity and applicability of the documents before refining them or rewriting them.
-
Participants recommend that a list of perceived gaps in the scientific basis for welfare decisions should be maintained. Although not all gaps may be filled at the time of any revision of guidelines, maintenance of these lists will facilitate their consideration at a later date. These lists act as bellwethers for areas where additional guidance may be needed. Additional indicators may come from indirect observations. For example, in instances in which guidance is less than adequate, interinstitutional variation in the implementations of guidelines due to professional judgment may indicate areas where additional guidelines are necessary to promote consistency in welfare and care.
-
Minimum acceptable standards may need to be established to facilitate consistency in enforcement. Without minimum standards, enforcement may be perceived as arbitrary.
-
Difficulties may arise when members of expert groups are included for political reasons. All stakeholders should be included, but representation should be balanced to ensure efficiency.
-
Some participants asked whether the questions being asked are the right ones. It was suggested by some that the goal should be the maximum improvement in welfare relative to the amount of effort generated to reach that goal.
What concerns exist relative to the way the Guide deals with NHPs?
Participants identified four concerns:
-
Occupational Health—Occupational health is fraught with variability across institutions as a result of vagaries in guidelines. Personal protective equipment in laboratories and proximity issues were discussed. Exposure as a function of proximity to NHPs and duration of exposure should be dealt with more specifically because investigators are looking for guidance.
-
Positive Reinforcement Training—Positive reinforcement training is not dealt with in detail but could facilitate welfare in instances of social housing.
-
Social Housing—Species-specific considerations are not extensive in the Guide. Social contact without social housing (by touch windows) may allow animals to withdraw when necessary, which achieves welfare goals. However, it may also reduce the vulnerability of individuals to injury, which is of concern to those who question the utility of social housing as a default condition.
-
Animal Welfare—There is a need to think about welfare from the “standpoint of the animal.”
CONCLUSION
The participants stressed that “one size does not fit all,” especially with respect to NHPs. Individuals of the same species often behave quite differently under the same environmental and behavioral situations. Participants felt that the “Redbook” (NRC 2003) successfully maintains this philosophy throughout discussions of individual experimental situations and other documents should be continued in this stance. It was felt that guidelines should include the consideration of an individual’s needs, experimental contingencies, and ethical responsibilities.
REFERENCES
NRC [National Research Council]. 1996. Guide for the Care and Use of Laboratory Animals. 7th Ed. Washington, DC. National Academy Press.
NRC. 2003. Guidelines for the Care and Use of Mammals in Neuroscience and Behavioral Research. Washington, DC. The National Academies Press.
Breakout Session: Rabbit Housing
Leader: Vera Baumans
Rapporteur: Jennifer Obernier
Guidelines for this breakout session encouraged its leader and participants to debate current research on rabbit housing standards and guidelines in light of current scientific information. In particular, the participants discussed the pros and cons of group housing, the standards and guidelines that govern minimum caging size, and the climate of the housing environment.
GROUP HOUSING
Both the Council of Europe (CoE 1986) and the Guide for the Care and Use of Laboratory Animals (the Guide) (NRC 1996) recognize that rabbits are social creatures and should be housed, when possible, in social groups to maximize species-specific behaviors and minimize stress-induced behaviors. The participants readily agreed that in some cases, for scientific or veterinary reasons, rabbits should not be group housed. However in most cases, social housing is an excellent idea, provided there is complexity in the caging. Complexity may include providing visual barriers and hiding places to minimize aggressive encounters and to allow animals to avoid contact by withdrawal. Social housing does have drawbacks, such as fighting, which can cause injury, and the need for improved animal husbandry and housing, to ensure the adequacy of food and water and the hygiene of the cage.
CAGE SIZE
Participants discussed the Council of Europe proposal to revise Appendix A of the European Convention ETS 123. This revision establishes a new minimum cage size standard based on weight, such that one or two compatabile rabbits of less than 4 kg should be housed in a cage with a floor area of 42 cm2 and a height of 45 cm, including a raised area of approximately 55 × 30 cm. Without a raised floor space, the floor area for one rabbit should be 5600 cm2 and 6700 cm2 for two rabbits. Existing standards and guidelines for rabbit caging size also base minimum cage size on weight, including (1) the European Convention ETS 123, which sets a standard of 2500 cm2 floor area and 35 cm of vertical space for a rabbit of less than 4 kg, and (2) the Guide, which recommends allocating a rabbit less than 4 kg approximately 2800 cm2 of floor space and approximately 35.5 cm of vertical space. During the discussion, participants agreed that basing cage size on weight is not optimal because it does not take into consideration that young rabbits are active and need more space in relation to their body weight than adults.
CLIMATE
The Council of Europe has proposed specific standards for the climate of rabbit housing, including temperature and humidity standards. However, participants pointed out that these standards were arbitrarily set and not based on science. Furthermore, it is unclear whether fluctuations above and below these standards during cleaning and scientific manipulation have any impact on animal well-being. Some participants felt that additional research is needed to understand seasonal fluctuations and the effects of extending the boundaries of the temperature and humidity standards proposed by the Council of Europe.
REFERENCES
CoE [Council of Europe]. 1986. European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (ETS 123). Strasbourg: Council of Europe.
NRC [National Research Council]. 1996. Guide for the Care and Use of Laboratory Animals. 7th Ed. Washington, DC: National Academy Press.








































