C
SURVEY RESULTS
TABLE C-1 Banks That Responded
|
Bank |
State |
|
American Red Cross North Central Blood Services Cord Blood Program |
MN |
|
Carolinas Cord Blood Bank |
NC |
|
Children’s Hospital of Orange County Cord Blood Bank |
CA |
|
Cord Blood Registry |
CA |
|
Cryobanks International, Inc. |
FL |
|
ITxM Cord Blood Services |
IL |
|
JP McCarthy Cord Stem Cell Bank |
MI |
|
Lifebank USA |
NJ |
|
LifeCord |
FL |
|
LifeStor |
VA |
|
Michigan Community Blood Centers Cord Blood Bank |
MI |
|
National Cord Blood Program of the New York Blood Center |
NY |
|
New Jersey Cord Blood Bank |
NJ |
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
WA |
|
Sibling Donor Cord Blood Program Children’s Hospital Oakland Research Institute |
CA |
|
South Texas Blood & Tissue Center |
TX |
|
St. Louis Cord Blood Bank |
MO |
|
StemCyte International Cord Blood Center and Cord Blood Family Trust |
CA |
|
The Elie Katz Umbilical Cord Blood Program |
NJ |
|
University of Colorado Cord Blood Bank |
CO |
|
Viacord |
MA |
TABLE C-2 Type of Bank
|
PUBLIC BANKS American Red Cross North Central Blood Services Cord Blood Program JP McCarthy Cord Stem Cell Bank Michigan Community Blood Centers Cord Blood Bank National Cord Blood Program of the New York Blood Center New Jersey Cord Blood Bank South Texas Blood & Tissue Center St. Louis Cord Blood Bank University of Colorado Cord Blood Bank |
|
PRIVATE BANKS Cord Blood Registry LifeStor Sibling Donor Cord Blood Program Children’s Hospital Oakland Research Institute Viacord |
|
BANKS THAT OFFER BOTH Carolinas Cord Blood Bank Children’s Hospital of Orange County Cord Blood Bank Cryobanks International, Inc. ITxM Cord Blood Services Lifebank USA LifeCord Puget Sound Blood Center and Hawaii Cord Blood Bank StemCyte Inc. and Cord Blood Family Trust The Elie Katz Umbilical Cord Blood Program at Community Blood Services |
|
Totals: 8 Public, 4 Private, 9 Mixed |
TABLE C-3 Self-Reported Accreditationa
|
Bank |
FACT |
AABB |
Otherb |
|
American Red Cross North Central Blood Services Cord Blood Program |
|
X |
|
|
Carolinas Cord Blood Bank |
X |
|
FACT pending, NMDP |
|
Children’s Hospital of Orange County Cord Blood Bank |
X |
|
JCAHO, California Tissue Bank License, Seeking FACT Accreditation |
|
Cord Blood Registry |
|
X |
|
|
Cryobanks International, Inc. |
|
X |
|
|
ITxM Cord Blood Services |
|
|
Will apply for FACT this fall |
|
JP McCarthy Cord Stem Cell Bank |
|
|
NMDP |
|
Lifebank USA |
|
X |
ISO |
|
LifeCord |
X |
X |
NMDP |
|
Michigan Community Blood Centers Cord Blood Bank |
|
X |
FDA Registration |
|
National Cord Blood Program of the New York Blood Center |
X |
|
New York State Board of Health |
|
New Jersey Cord Blood Bank |
|
|
|
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
|
X |
|
|
Sibling Donor Cord Blood Program Children’s Hospital Oakland Research Institute |
X |
|
California State Biologies, California State Tissue Bank (both current) |
|
St. Louis Cord Blood Bank |
|
X |
CAP |
|
StemCyte, Inc. and Cord Blood Family Trust |
|
X |
ASHI, NMDP, CAP, California Biologics License, CLIA, FACT pending |
|
The Elie Katz Umbilical Cord Blood Program |
|
X |
|
|
University of Colorado Cord Blood Bank |
X |
|
|
|
Viacord |
|
X |
CLIA, CAP proficiency, NY State, NJ State, KY State, PA State, registered with FDA and IL State, and MD tissue bank permit |
|
aThe accreditation status of the banks was not independently verified. bThe definitions of the acronyms are provided at the end of this appendix. |
|||
TABLE C-4 Numbers of Collected, Stored, and Transplanted Units
|
Bank |
Consenting to Collect |
Collected |
Banked |
Available |
Searchable |
Used for Transplant |
Note |
|
American Red Cross North Central Blood Services Cord Blood Program |
23,130 |
18,680 |
6,069 |
5,962 |
5,962 |
242 |
8 not transplanted |
|
Carolinas Cord Blood Bank |
17,519 |
17,000 |
8,300 |
7,800 |
7,800 |
320 |
|
|
Children’s Hospital of Orange County Cord Blood Bank |
3390 |
2,900 |
1,550 |
1,375 |
1,350 |
7 |
|
|
Cryobanks International, Inc. |
17,741 |
15,429 |
9,439 |
7,485 |
7,485 |
5 |
4 public, 1 private |
|
ITxM Cord Blood Services |
9,867 |
5,280 |
5,000 |
2,620 |
2,513 |
20 |
Consenting refers to collection kits requested |
|
JP McCarthy Cord Stem Cell Bank |
3323 |
2,486 |
600 |
517 |
445 |
4 |
|
|
Lifebank USA |
17,807 |
17,228 |
16,264 |
3,300 |
3,300 |
1 |
|
|
LifeCord |
1 |
7,429 |
1,975 |
1,474 |
1,474 |
10 |
|
|
LifeStor |
102 |
96 |
|
||||
|
Michigan Community Blood Centers Cord Blood Bank |
6,665 |
5,531 |
1,497 |
1,386 |
1,375 |
6 |
|
|
National Cord Blood Program of the New York Blood Center |
27,328 |
29,525 |
25,989 |
25,989 |
25,989 |
1,765 |
Includes first and second transplantations and multiple-unit grafts |
|
New Jersey Cord Blood Bank |
|
2,621 |
1,984 |
1,796 |
1,796 |
7 |
|
|
Bank Name |
Consenting to Collect |
Collected |
Banked |
Available |
Searchable |
Used for Transplant |
Note |
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
3,877 |
2,358 |
1,097 |
727 |
727 |
5 |
|
|
Sibling Program Children’s Hospital Oakland Research Institute |
1,556 |
1,438 |
1,378 |
1,064 |
0 |
43 |
|
|
St. Louis Cord Blood Bank |
40,000 |
40,000 |
10,000 |
10,000 |
10,000 |
656 |
10,000 means >10,000. Searches include NMDP, CRIR, and direct searches. Transplants are through June 30, 2004 |
|
StemCyte, Inc. and Cord Blood Family Trust |
16,777 |
13,566 |
8,558 |
8,331 |
8,331 |
78 |
|
|
The Elie Katz Umbilical Cord Blood Program |
|
3,635 |
2,473 |
2,473 |
1,511 |
25 |
|
|
University of Colorado Cord Blood Bank |
10,000 |
9,943 |
7,017 |
6,669 |
6,435 |
234 |
Total consenting not known with precision |
|
Viacord |
60,000 |
60,000 |
|
|
0 |
14 |
60,000 means >60,000. Every consent resulted in a collection. |
|
Total |
259,083 |
255,145 |
109,190 |
62,979 |
60,504 |
3,384 |
|
TABLE C-5 Banks That Are Currently Collecting Units
|
Bank |
|
American Red Cross North Central Blood Services Cord Blood Program Carolinas Cord Blood Bank Children’s Hospital of Orange County Cord Blood Bank Cord Blood Registry Cryobanks International, Inc. ITxM Cord Blood Services JP McCarthy Cord Stem Cell Bank Lifebank USA/Anthrogenesis Corp. LifeCord Michigan Community Blood Centers Cord Blood Bank National Cord Blood Program of the New York Blood Center New Jersey Cord Blood Bank Puget Sound Blood Center and Hawaii Cord Blood Bank Sibling Program Children’s Hospital Oakland Research Institute St. Louis Cord Blood Bank StemCyte, Inc. and Cord Blood Family Trust The Elie Katz Umbilical Cord Blood Program Viacord |
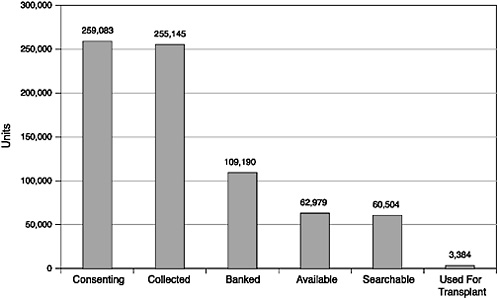
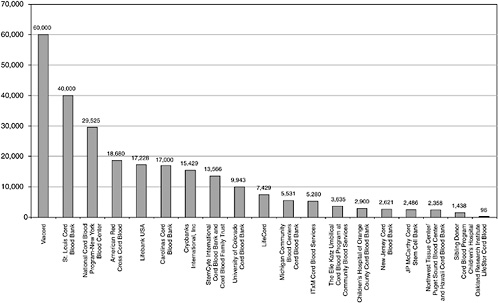
FIGURE C-2 Available units—across all banks.
TABLE C-6 Racial Makeup of Inventory
|
|
Percent Available |
||||||||
|
Asian |
African American |
Hispanic |
Native American |
Middle Eastern |
Caucasian |
Unknown |
Multiracial |
Other |
|
|
Averages by total number of units |
5.79 |
11.60 |
16.81 |
1.02 |
0.11 |
56.08 |
4.89 |
3.01 |
0.69 |
|
BY BANK |
|||||||||
|
American Red Cross North Central Blood Services Cord Blood Program |
2.47 |
6.59 |
12.03 |
0.37 |
|
64.64 |
13.90 |
|
|
|
Carolinas Cord Blood Bank |
2.21 |
19.52 |
9.26 |
0.14 |
0.00 |
59.15 |
0.02 |
9.21 |
0.50 |
|
Children’s Hospital of Orange Country Cord Blood Bank |
17.70 |
0.67 |
35.40 |
2.67 |
|
43.55 |
0.00 |
0.00 |
0.00 |
|
Cryobanks International, Inc. |
1.92 |
8.06 |
11.16 |
1.12 |
0.57 |
67.24 |
0.28 |
9.65 |
|
|
ITxM Cord Blood Services |
3.72 |
2.16 |
5.58 |
1.38 |
0.00 |
86.46 |
|
0.43 |
0.26 |
|
JP McCarthy Cord Stem Cell Bank |
0.45 |
80.67 |
2.25 |
0.22 |
|
6.52 |
|
9.66 |
0.22 |
|
LifeCord |
0.83 |
3.32 |
2.76 |
0.00 |
0.41 |
82.72 |
|
9.95 |
|
|
Michigan Community Blood Centers Cord Blood Bank |
1.17 |
2.20 |
3.03 |
1.38 |
|
64.19 |
28.03 |
|
|
|
National Cord Blood Program of the New York Blood Center |
7.70 |
21.00 |
21.10 |
|
|
45.60 |
4.60 |
|
|
|
New Jersey Cord Blood Bank |
2.17 |
7.24 |
0.50 |
0.89 |
0.39 |
61.41 |
|
|
27.39 |
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
33.01 |
1.79 |
4.54 |
4.26 |
|
56.40 |
|
|
|
|
St. Louis Cord Blood Bank |
0.66 |
1.59 |
0.28 |
4.69 |
|
84.35 |
0.94 |
7.50 |
|
|
StemCyte, Inc. and Cord Blood Family Trust |
14.99 |
4.00 |
51.01 |
0.39 |
|
27.00 |
2.62 |
|
|
|
The Elie Katz Umbilical Cord Blood Program |
2.02 |
2.95 |
10.96 |
0.08 |
1.05 |
42.74 |
40.19 |
0.00 |
0.00 |
|
Averages by bank |
6.50 |
11.55 |
12.13 |
1.35 |
0.41 |
56.57 |
10.06 |
5.80 |
4.73 |
TABLE C-7 Criteria to Determine Which Units Are Suitable for Banking
|
Bank |
Threshold of Nucleated Cell Dose |
Volume |
Bacterial Contamination (Negative) |
|
American Red Cross North Central Blood Services Cord Blood Program |
Preprocessing TNC of 1.2 × 109 |
50 ml |
X |
|
Carolinas Cord Blood Bank |
9 × 108 |
60 ml |
X |
|
Children’s Hospital of Orange County Cord Blood Bank |
9 × 108 |
40 ml minimum |
X |
|
Cryobanks International, Inc. |
Precount >8 × 108 |
60 g |
X |
|
ITxM Cord Blood Services |
|
40 ml collection per NMDP IND |
X |
|
JP McCarthy Cord Stem Cell Bank |
|
|
X |
|
Lifebank USA |
TNC = 5 × 108 |
35 ml |
X |
|
LifeCord |
8 × 108 TNC preprocessing |
≥40 ml |
|
|
Michigan Community Blood Centers Cord Blood Bank |
11 × 108, minority threshold = 9.5 × 108 |
45 ml |
X |
|
National Cord Blood Program of the New York Blood Center |
9 × 108 |
|
X |
|
Level of HLA Resolution |
Other |
|
|
Donation is excluded if:
|
|
Intermediate resolution for Class I HLA, highresolution DRB1 |
Tests negative for homozygous hemoglobinopathies; positive CFU growth Negative infectious disease testing on maternal blood |
|
|
Negative maternal infectious disease markers, processed within 36 hours of collection |
|
|
Maternal and baby health questionnaire eligibility Infectious disease testing, microbiology testing, hemaglobinopathy screen, viability |
|
|
Infectious disease markers, ABO and RH type determination of cord blood unit, newborn screening retention samples, maternal medical health questionnaire, review of maternal and newborn medical records |
|
|
Virology negative (except for CMV) |
|
|
Negative infectious disease test results, lack of family medical history of inherited genetic disorders or diseases, no inappropriate affirmative responses to questions from risk behavior questionnaire |
|
|
Negative infectious disease testing, normal genetic screen “We also apply the same deferral criteria as that of whole blood donors for risk factors” |
|
Class I antigen level (molecular test, SSP) |
No evidence of viral infection for HIV/HILV-1/11, hepatitis B or C virus, CMV by culture or DNA PCR testing. |
|
Bank |
Threshold of Nucleated Cell Dose |
Volume |
Bacterial Contamination (Negative) |
|
New Jersey Cord Blood Bank |
minimum of 1.5 × 108 preprocessing, 1.2 × 108 postprocessing |
>40 ml of cord blood |
X |
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
9 × 108 |
40 ml |
|
|
Sibling Program Children’s ≤ Hospital Oakland Research Institute |
3 × 108 |
≤20 cc |
|
|
St. Louis Cord Blood Bank |
>8.0 × 108 |
45 ml |
X |
|
StemCyte, Inc. and Cord Blood Family Trust |
|
50 ml public, 10 ml private |
X |
|
The Elie Katz Umbilical Cord Blood Program |
4 × 108 TNC for autologous storage and 6 × 108 TNC for allogeneic |
if less than 50 ml then check TNC |
X |
|
University of Colorado Cord Blood Bank |
TNC greater than 109* |
≥95 ml collected* |
X |
|
Viacord |
100 × 106 TNC |
≥10 ml |
|
|
Level of HLA Resolution |
Other |
|
HLA-A HLA-B, and DRB1 intermediate resolution |
Maternal health history, maternal infectious disease screening, 6-month and 1-year follow-up of child’s health |
|
|
Complications and/or congenital abnormalities observed |
|
|
Since units are all related, we bank as many as possible |
|
|
CD34+ and CF4 values, viability |
|
Intermediate-resolution HLA-A and HLA-B, highresolution DRB1 |
Infectious disease markers (only positives banked are CMV and Hepatitis B core), maternal health history and family genetic history |
|
|
Negative viral marker results |
|
HLA-A, HLA-B serological equivalent, HLA-DRB1 high resolution |
Negative infectious disease markers, no risk factors from maternal donor questionnaire, complete consent Criteria with “*” are to be used for qualifying future collections |
|
|
CD34, viability <50 percent, positive HIV status of mother, units with other abnormal infectious disease results stored only after a review by medical director |
TABLE C-8 Criteria Used to Determine Which Units Are for Transplant
|
|
Criteria for a Transplant Center Before Receiving Units from Bank |
||
|
Bank |
NMDP Approved Site |
FACT Accreditation |
Other |
|
American Red Cross North Central Blood Services Cord Blood Program |
|
|
Evidence of IRB approval (or international equivalent); evidence of NMDP, FACT, or equivalent certification; agreement to provide outcome data |
|
Carolinas Cord Blood Bank |
X |
|
Use IRB approved protocols |
|
Children’s Hospital of Orange County Cord Blood Bank |
X |
|
|
|
Cryobanks International, Inc. |
X |
|
Meet WMDA recommendations for transplant center criteria |
|
ITxM Cord Blood Services |
X |
|
|
|
JP McCarthy Cord Stem Cell Bank |
X |
|
|
|
Lifebank USA |
|
|
IRB approved protocol, agreeing to provide post-transplant data |
|
LifeCord |
X |
|
|
|
Michigan Community Blood Centers Cord Blood Bank |
|
|
IRB approved transplant protocol |
|
National Cord Blood Program of the New York Blood Center |
|
|
Must have a recognized track record in BMT, and accreditation by EBMT, JACIE, etc. |
|
New Jersey Cord Blood Bank |
X |
|
Written prescription from transplant center |
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
X |
|
Signed agreement and memorandum of understanding |
|
St. Louis Cord Blood Bank |
|
|
|
|
|
How Bank Is Most Commonly Accessed by Transplant Center |
|||
|
Transplant Center Exceptions |
Direct Contact |
NET-CORD |
NMDP |
Other |
|
Exceptions allowed on basis of approval by medical director |
X |
|
|
|
|
Medical director has personal knowledge of transplant center |
X |
|
X |
parents or referring physician |
|
N/A |
|
|
X |
|
|
Medical director approval |
|
|
|
CRIR |
|
Private family units do not need to go to NMDP centers |
X |
|
X |
|
|
NMDP will permit compassionate-use requests for nonmember transplant centers |
|
|
X |
|
|
|
|
|
|
BMDW |
|
|
|
|
X |
|
|
|
X |
|
|
CRIR |
|
|
X |
X |
|
|
|
|
|
|
X |
|
|
|
|
|
X |
CRIR |
|
|
X |
|
X |
CRIR |
|
|
Criteria for a Transplant Center Before Receiving Units from Bank |
||
|
Bank |
NMDP Approved Site |
FACT Accreditation |
Other |
|
StemCyte, Inc. and Cord Blood Family Trust |
X |
|
For most non-U.S. transplant centers, this does not apply |
|
The Elie Katz Umbilical Cord Blood Program |
|
|
IRB, if available, request form |
|
University of Colorado Cord Blood Bank |
|
|
Must meet WMD recommendations and requirements per Goldman et al., 1994, A special report: Bone marrow transplants using volunteer donors—recommendaions and requirements for a standardized practice throughout the world. Blood 84:2833. |
TABLE C-9 Units That Are Not Usable for Transplant
|
|
Research–External |
|||
|
Bank |
Research/Internal |
Discarded or Destroyed |
Investigator Charged |
No Charge |
|
American Red Cross North Central Blood Services Cord Blood Program |
X |
X |
X |
X |
|
Carolinas Cord Blood Bank |
X |
X |
X |
X |
|
Children’s Hospital of Orange County Cord Blood Bank |
X |
X |
|
|
|
Cryobanks International, Inc. |
X |
X |
|
X |
|
ITxM Cord Blood Services |
X |
X |
|
X |
|
JP McCarthy Cord Stem Cell Bank |
|
X |
|
X |
|
Lifebank USA |
X |
|
|
X |
|
LifeCord |
X |
X |
|
X |
|
Michigan Community Blood Centers Cord Blood Bank |
|
X |
|
|
|
National Cord Blood Program of the New York Blood Center |
X |
X |
X |
X |
|
New Jersey Cord Blood Bank |
X |
X |
|
|
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
X |
X |
|
|
|
St. Louis Cord Blood Bank |
X |
X |
|
X |
|
StemCyte, Inc. and Cord Blood Family Trust |
X |
X |
X |
X |
|
The Elie Katz Umbilical Cord Blood Program |
X |
X |
|
X |
|
University of Colorado Cord Blood Bank |
X |
X |
X |
X |
|
Viacord |
X |
X or continue storage |
|
|
TABLE C-10 Are Units Bar Coded and Tracked Electronically? If Not, How Are They Tracked?
|
Bank |
Tracking of Units |
|
American Red Cross North Central Blood Services Cord Blood Program |
Units are labeled with unique, bar-coded identifiers. All paperwork and corresponding samples are labeled with the unique identifier associated with the unit. A database is used to monitor the location of the unit and inspection and testing status. |
|
Carolinas Cord Blood Bank |
Yes. Units are bar coded with the ISBT 128 system. |
|
Children’s Hospital of Orange County Cord Blood Bank |
Yes, bar coded and tracked |
|
ITxM Cord Blood Services |
Bar coded, tracked in database |
|
LifeCord |
Yes |
|
Michigan Community Blood Centers Cord Blood Bank |
Manual entry into database |
|
National Cord Blood Program of the New York Blood Center |
Yes |
|
New Jersey Cord Blood |
Yes Bank |
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
Yes; bar coded; also, the location is recorded in the unit chart |
|
Sibling Program Children’s Hospital Oakland Research Institute |
Units are bar coded and tracked electronically and manually; a written inventory log is kept |
|
St. Louis Cord Blood Bank |
Yes |
|
StemCyte, Inc. and Cord Blood Family Trust |
Yes |
|
The Elie Katz Umbilical Cord Blood Program |
Yes |
|
University of Colorado Cord Blood Bank |
Yes (for cord blood unit banked after July 1999, 68 percent of searchable inventory) |
|
Viacord |
Bar code from collection of cord blood through transport to receipt at lab. Manual tracking from receipt to storage (and distribution if called on) |
TABLE C-11 Informatics
|
Bank |
Excel Spreadsheet |
Access Database |
Other Database |
|
American Red Cross North Central Blood Services Cord Blood Program |
X |
X |
Oracle Database |
|
Carolinas Cord Blood Bank |
|
X |
EMMES |
|
Children’s Hospital of Orange County Cord Blood Bank |
|
|
CordLink NMDP |
|
Cryobanks International, Inc. |
|
X |
|
|
ITxM Cord Blood Services |
|
X |
NMDP CordLink |
|
JP McCarthy Cord Stem Cell Bank |
|
X |
|
|
Lifebank USA |
X |
|
Stem Lab Software |
|
LifeCord |
|
|
Oracle |
|
Michigan Community Blood Centers Cord Blood Bank |
X |
X |
|
|
National Cord Blood Program of the New York Blood Center |
|
|
Oracle |
|
New Jersey Cord Blood Bank |
|
|
Sybase and CordLink |
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
|
X |
Cordlink |
|
Sibling Program Children’s Hospital Oakland Research Institute |
X |
|
SQL database |
|
St. Louis Cord Blood Bank |
|
X |
SQL server |
|
StemCyte, Inc. and Cord Blood Family Trust |
X |
X |
|
|
The Elie Katz Umbilical Cord Blood Program |
X |
X |
|
|
University of Colorado Cord Blood Bank |
|
X |
|
|
Viacord |
X |
X |
|
TABLE C-12 Collection and Storage: Who Does the Collection
TABLE C-13 Exclusion Criteria
|
Bank |
Multiple Gestations |
Gestational Age of Less Than ______ Weeks |
Chorioamnionitis (Definition at This Center) |
Prolonged Rupture of Membranes (Without Chorioamnionitis) |
Tear in Cord Insertion from Placenta |
|
American Red Cross North Central Blood Services Cord Blood Program |
X |
X |
Evaluation of infection is performed by the cord blood bank through chart review |
|
|
|
Carolinas Cord Blood Bank |
X |
34 |
Inflammation of the chorion and the amnion, the membranes that surround the fetus |
|
|
|
Children’s Hospital of Orange County Cord Blood Bank |
|
36 |
|
|
X |
|
Cryobanks International, Inc. |
|
|
|
|
|
|
ITxM Cord Blood Services |
X |
36 |
Evidence of infection or odor, pus, temperature >38.5.°C |
|
X |
|
JP McCarthy Cord Stem Cell Bank |
|
|
|
|
X |
|
Lifebank USA |
|
|
|
X |
|
|
LifeCord |
|
36 |
|
|
|
|
Michigan Community Blood Centers Cord Blood Bank |
X |
36 |
Per attending physician’s determination |
|
|
|
Positive Group B Strept Carrier |
Active Genital Herpes |
Vulvar or Perineal Condylomata |
Known Fetal Structural Anomalies (e.g., Spinal Bifida) |
Known Fetal Chromosome Abnormalities |
Other |
|
X |
|
X |
|
X |
Reactive for infectious disease markers Less than 70 percent postprocessing and prefreezing viability There is a family history of diseases, conditions, or genetic disorders that may affect the recipient The mother has symptoms of or has been diagnosed with certain infectious diseases including West Nile virus, parasitic blood diseases, syphilis, hepatitis, and HIV/AIDS. The mother has engaged in behaviors or received medical treatments that increase the risk of contracting these diseases |
|
X |
X |
X |
X |
X |
Maternal-fetal shunt |
|
|
|
X |
|
X |
Two vessel cord; temperature over 100.4°F in the infant |
|
|
|
X |
|
X |
|
|
X |
X |
X |
X |
X |
Placenta previa, placental abruption, fetal or maternal distress |
|
X |
|
X |
X |
X |
|
|
|
|
X |
|
X |
|
|
|
|
|
|
X |
Mother’s temperature is 100.4°F or greater |
|
X |
|
|
|
X |
Placental abruption, tears, or infection, eclampsia, HELLP syndrome, stillbirth, maternal fever within 24 hours of delivery |
|
Positive Group B Strept Carrier |
Active Genital Herpes |
Vulvar or Perineal Condylomata |
Known Fetal Structural Anomalies (e.g., Spinal Bifida) |
Known Fetal Chromosome Abnormalities |
Other |
|
X |
|
||||
|
X |
|
X |
|
X |
Stillborn infant |
|
|
|
|
|
|
Known to be affected by a genetic disease |
|
|
|
X |
|
|
Maternal or infant temperature >102°F |
|
X |
|
|
|
X |
Only vaginal deliveries with active genital herpes are deferred; also exclude tumor within or attached to placenta; abruptio placenta |
|
|
|
X |
|
X |
Temperature >38.5°C, maternal eclampsia and/or maternal HELLP syndrome |
|
X |
X |
X |
X |
X |
Caesarean section |
|
|
|
|
X |
|
Customer choice to store or not |
TABLE C-14 Screening Completed Prior to Storage
|
Genetic Screening |
|||||
|
Bank |
Hemoglobinopathy |
ABO Group |
Rh Type |
Health Questionnaire |
Other |
|
Carolinas Cord Blood Bank |
X |
X |
X |
X |
X |
|
Children’s Hospital of Orange County Cord Blood Bank |
X |
X |
X |
X |
|
|
Cryobanks International, Inc. |
|
X |
X |
X |
|
|
ITxM Cord Blood Services |
X |
X |
X |
X |
|
|
JP McCarthy Cord Stem Cell Bank |
X |
X |
X |
X |
|
|
Lifebank USA |
|
|
|
X |
|
|
LifeCord |
X |
X |
X |
X |
X |
|
Michigan Community Blood Centers Cord Blood Bank |
X |
X |
X |
X |
X |
|
National Cord Blood Program of the New York Blood Center |
X |
X |
X |
X |
|
|
New Jersey Cord Blood Bank |
X |
X |
X |
X |
|
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
|
X |
X |
X |
|
|
Sibling Program Children’s Hospital Oakland Research Institute |
X |
X |
X |
X |
|
|
St. Louis Cord Blood Bank |
X |
X |
X |
X |
|
|
StemCyte, Inc. and Cord Blood Family Trust |
|
X |
X |
X |
|
|
The Elie Katz Umbilical Cord Blood Program |
|
X |
X |
X |
|
|
University of Colorado Cord Blood Bank |
X |
X |
X |
X |
|
|
Viacord |
|
X |
X |
X |
|
|
Infectious Disease Testing |
|||||||||||||||||
|
Bank |
Alanine Aminotransferase (ALT) |
Cholesterol |
CMV IgG |
CMV IgM |
CMV Culture/Shell Vial |
Hepatitis B Virus—HBsAg |
Hepatitis B Virus—HB Core Ab |
Hepatitis B Virus—NAT |
Hepatitis C Virus—Ab |
Hepatitis C Virus—NAT |
HIV, Type 1—Ab |
HIV, Type 1—P24 Ag |
HIV, Type 2 |
Human T-cell Lymphotropic Virus, Type I |
Human T-cell Lymphotropic Virus, Type II |
Treponema pallidum (Syphilis) |
West Nile Virus NAT |
|
American Red Cross North Central Blood Services Cord Blood Bank |
X |
|
X |
X |
|
X |
X |
X |
X |
X |
X |
X |
X |
X |
X |
X |
X |
|
Carolinas Cord Blood Bank |
|
X |
X |
X |
X |
X |
X |
|
X |
X |
X |
X |
X |
X |
X |
||
|
Children’s Hospital of Orange County Cord Blood Bank |
X |
X |
X |
X |
|
X |
X |
X |
|
X |
X |
X |
X |
X |
|||
|
Cryobanks International, Inc. |
|
|
X |
|
|
X |
|
X |
|
X |
X |
X |
X |
|
|||
|
ITxM Cord Blood Services |
X |
|
X |
X |
X |
X |
X |
X |
X |
X |
X |
X |
X |
X |
|||
|
JP McCarthy Cord Stem Cell Bank |
X |
X |
X |
|
|
X |
|
X |
|
|
X |
X |
X |
X |
|||
|
Infectious Disease Testing (continued) |
|||||||||||||||||
|
Bank |
Alanine Aminotransferase (ALT) |
Cholesterol |
CMV IgG |
CMV IgM |
CMV Culture/Shell Vial |
Hepatitis B Virus—HBsAg |
Hepatitis B Virus—HB Core Ab |
Hepatitis B Virus—NAT |
Hepatitis C Virus—Ab |
Hepatitis C Virus—NAT |
HIV, Type 1—Ab |
HIV, Type 1—P24 Ag |
HIV, Type 2 |
Human T-cell Lymphotropic Virus, Type I |
Human T-cell Lymphotropic Virus, Type II |
Treponema pallidum (Syphilis) |
West Nile Virus NAT |
|
Lifebank USA |
X |
|
X |
X |
|
X |
X |
X |
X |
X |
X |
|
X |
X |
X |
X |
X |
|
LifeCord |
X |
X |
X |
X |
|
X |
X |
|
X |
X |
X |
|
X |
X |
X |
X |
X |
|
Michigan Community Blood Centers Cord Blood Bank |
|
|
X |
X |
|
X |
X |
|
X |
X |
X |
X |
X |
X |
X |
X |
X |
|
National Cord Blood Program of the New York Blood Center |
X |
|
|
X |
X |
X |
|
|
X |
X |
X |
|
X |
X |
X |
X |
|
|
New Jersey Cord Blood Bank |
|
|
X |
|
|
X |
X |
|
X |
|
X |
|
X |
X |
X |
X |
|
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
|
|
X |
|
|
X |
|
X |
X |
|
X |
|
X |
X |
X |
X |
X |
|
Sibling Program Children’s Hospital Oakland Research Institute |
|
X |
X |
|
|
|
X |
X |
|
|
X |
X |
X |
X |
X |
|
St. Louis Cord Blood Bank |
|
|
|
X |
|
X |
X |
X |
X |
|
X |
X |
X |
X |
X |
|
StemCyte, Inc. and Cord Blood Family Trust |
X |
X |
X |
X |
X |
|
|
X |
X |
|
X |
X |
X |
X |
X |
|
The Elie Katz Umbilical Cord Blood Program |
|
X |
X |
X |
X |
|
X |
X |
X |
|
X |
X |
X |
X |
X |
|
University of Colorado Cord Blood Bank |
X |
X |
X |
X |
X |
|
X |
|
X |
X |
X |
X |
X |
X |
|
|
Viacord |
|
X |
|
X |
X |
|
X |
X |
X |
|
X |
X |
X |
X |
|
|
Other Exclusions: Other Infectious Disease Testing and Travel |
||
|
Bank |
Other |
Current FDA Travel Restrictions |
|
American Red Cross North Central Blood Services Cord Blood Bank |
|
Yes |
|
Carolinas Cord Blood Bank |
|
Yes |
|
Children’s Hospital of Orange County Cord Blood Bank |
HIV 1 and 2 NAT |
Yes |
|
Cryobanks International, Inc. |
HBV Ab and CMV, total by PCR |
Yes |
|
ITxM Cord Blood Services |
|
Yes |
|
JP McCarthy Cord Stem Cell Bank |
HIV and HCV NAT; HBV NAT |
Yes |
|
Lifebank USA |
|
Yes |
|
LifeCord |
HIV NAT |
Yes |
|
Any Other Recommended Travel Restrictions That Would Prevent Inclusion of the Unit? |
|
Mother lived in any area where malaria is endemic within the past 3 years or traveled to those areas in the past 12 months. Mother spent a total time from 1980 to 1996 that adds up to 3 months or more in the United Kingdom (UK) from 1980 to 1996 or she received a blood transfusion in the UK in this time frame. Mother was born or lived in Cameroon, Central African Republic, Chad, Congo Equatorial Guinea, Gabon Niger, Nigeria, or Ivory Coast since 1977 or been the sexual partner of a person who was born in or lived in these countries in this time frame. Note: all time frames relate to date of infant’s delivery. |
|
No |
|
Immigrants, refugees, or citizens coming from a country in which malaria is considered endemic will be deferred for 3 years after departure from the area if they have been free from unexplained symptoms suggestive of malaria. If after their arrival in the United States they visit any region where malaria is endemic, a deferral of 3 years applies. Other donors who have traveled to a country where malaria is considered endemic will be deferred for 12 months after returning from that area, regardless of whether or not they took antimalaria prophylaxis. Also deferral if since 1980 the mother spent 5 years or more in Europe, 3 months or more in the UK from 1980 to 1986; member of military from 1980 to 1996 and spent more than 6 months in Belgium, The Netherlands, or Germany (1980 to 1990), or Spain, Portugal, Turkey, Italy, or Greece (1980 to 1996). |
|
All exclusions applicable to whole-blood donors, e.g. malaria, vCJF, and SARS. |
|
Mothers who have traveled to areas where malaria is endemic are deferred for 1 year after return. Immigrants from countries where malaria is endemic are deferred for 3 years after leaving the country. Persons who have lived for 5 or more years in countries where malaria is endemic are deferred for 3 years after returning to the United States. Travel to Cameroon, Central Africa Republic, Chad, Congo, Equatorial Guinea, Gabon, Niger, Nigeria, and Cote d’Ivoire since 1977 would restrict donation due to possible HIV exposure. Travel to the United Kingdom from 1980 to the present for 3 months or Europe for 6 months would restrict donation due to possible CJF. |
|
Travel to high-risk area |
|
|
Other Exclusions: Other Infectious Disease Testing and Travel (continued) |
||
|
Bank |
Other |
Current FDA Travel Restrictions |
|
Michigan Community Blood Centers Cord Blood Bank |
HIV NAT has replaced p24 Ag since fall 2003. Other genetic tests performed include for CAH, hypothyroidism, galactosemia, maple syrup urine disease, PKU, MCAD, biotinidase deficiency |
Yes |
|
New Jersey Cord Blood Bank |
|
Yes |
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
HIV NAT antibody screen |
Yes |
|
Sibling Program Children’s Hospital Oakland Research Institute |
HIV type 1 NAT |
No |
|
St. Louis Cord Blood Bank |
CMV total Ab, HIV NAT |
|
|
StemCyte, Inc. and Cord Blood Family Trust |
HIV type 1 and type 2 NAT |
Yes |
|
The Elie Katz Umbilical Cord Blood Program |
|
Yes |
|
University of Colorado Cord Blood Bank |
Future collection testing will include HIV and HCV NAT, WNV NAT |
Yes |
|
Viacord |
Antibody screen HIV NAT |
|
|
Any Other Recommended Travel Restrictions That Would Prevent Inclusion of the Unit? |
|
Malarial deferrals will result in quarantine of unit until follow-up phone call and/or other testing is performed after the deferral period. Travel areas affected by SARS would result in exclusion. |
|
The CDC Health Information for International Travel 2003–2004 is used. |
|
Exclusion for travel to malaria risk areas within the past 3 years (is possible to reduce the time restriction to the past 12 months). Exclusion for vCJF risk and SARS as advised by the FDA. |
|
These are evaluated on a case-by-case basis by staff in conjunction with the medical director. |
|
No |
|
Defer for travel areas where malaria and SARS areas are endemic within the last 14 days, extended stay in UK (for CJF). |
|
Collections from 1996 to 2001 utilized the then-current FDA guidelines for donor exclusion based on travel to foreign countries. However, short-term travel did not warrant exclusion. |
|
Questions on travel history were asked and documented on health history questionnaire. |
TABLE C-15 Number of Units Collected and Shipped by Year 1993–2004
|
Bank |
Status |
1993 |
1994 |
1995 |
1996 |
1997 |
|
American Red Cross North Central Blood Services Cord Blood Bank |
Collected |
|
|
|
|
1,350 |
|
Shipped |
|
|
|
|
0 |
|
|
Carolinas Cord Blood Bank |
Collected |
|
||||
|
Shipped |
|
|||||
|
Children’s Hospital of Orange County Cord Blood Bank |
Collected |
0 |
0 |
0 |
0 |
0 |
|
Shipped |
0 |
0 |
0 |
0 |
0 |
|
|
Cryobanks International, Inc. (Pr = private, Pu = public) |
Collected |
Pr: 0 |
Pr: 0 |
Pr: 9 |
Pr: 127 |
Pr: 143 |
|
|
Pu: 0 |
Pu: 0 |
Pu: 23 |
Pu: 397 |
Pu: 268 |
|
|
Shipped |
Pr: 0 |
Pr: 0 |
Pr: 0 |
Pr: 0 |
Pr: 0 |
|
|
|
Pu: 0 |
Pu: 0 |
Pu: 0 |
Pu: 0 |
Pu: 0 |
|
|
ITxM Cord Blood Services |
Collected |
0 |
0 |
3 |
63 |
226 |
|
Shipped |
0 |
0 |
0 |
0 |
0 |
|
|
JP McCarthy Cord Stem Cell Bank |
Collected |
|
||||
|
Shipped |
|
|||||
|
LifeCord |
Collected |
|
|
|
|
122 |
|
Shipped |
|
|
|
|
0 |
|
|
Michigan Community Blood Centers Cord Blood Bank |
Collected |
|
|
|
|
|
|
Shipped |
|
|
|
|
|
|
|
National Cord Blood Program—New York Blood Center |
Collected |
943 |
1,739 |
1,615 |
920 |
763 |
|
Shipped |
2 |
16 |
100 |
214 |
256 |
|
|
New Jersey Cord Blood Bank |
Collected |
|
|
|
|
|
|
Shipped |
|
|
|
|
|
|
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
Collected |
0 |
0 |
0 |
0 |
0 |
|
Shipped |
0 |
0 |
0 |
0 |
0 |
|
|
1998 |
1999 |
2000 |
2001 |
2002 |
2003 |
2004 |
|
2,155 |
175 |
2,579 |
5,062 |
6,511 |
3,092 |
522 |
|
10 |
11 |
9 |
22 |
50 |
81 |
59 |
|
960 |
4,136 |
2,147 |
1,851 |
2,326 |
2,325 |
1,164 |
|
0 |
9 |
36 |
68 |
76 |
75 |
52 |
|
0 |
44 |
370 |
510 |
666 |
528 |
375 |
|
0 |
0 |
0 |
1 |
1 |
1 |
3 |
|
Pr: 251 |
Pr: 156 |
Pr: 126 |
Pr: 159 |
Pr: 220 |
Pr: 241 |
Pr: 75 |
|
Pu: 671 |
Pu: 316 |
Pu: 337 |
Pu: 1,168 |
Pu: 3,983 |
Pu: 5,256 |
Pu: 3,010 |
|
Pr: 0 |
Pr: 0 |
Pr: 0 |
Pr: 0 |
Pr: 0 |
Pr: 1 |
Pr: 0 |
|
Pu: 0 |
Pu: 0 |
Pu: 0 |
Pu: 0 |
Pu: 0 |
Pu: 491 |
Pu: 2 |
|
289 |
1,149 |
1,740 |
1,145 |
388 |
30 |
188 |
|
0 |
0 |
0 |
2 |
9 |
8 |
1 |
|
|
|
|
211 |
922 |
921 |
432 |
|
|
|
|
|
|
2 |
2 |
|
337 |
345 |
287 |
333 |
288 |
168 |
95 |
|
0 |
0 |
0 |
2 |
0 |
6 |
2 |
|
|
124 |
604 |
1,086 |
1,301 |
1,477 |
880 |
|
|
0 |
0 |
1 |
1 |
3 |
1 |
|
255 |
1,395 |
2,530 |
3,301 |
4,322 |
4,333 |
2,884 (as of 8/31/04) |
|
174 |
174 |
200 |
115 |
168 |
214 |
133 (as of 8/31/04) |
|
|
|
12 |
462 |
1,114 |
797 |
237 |
|
|
|
0 |
0 |
1 |
5 |
1 |
|
0 |
154 |
402 |
415 |
477 |
492 |
418 |
|
0 |
0 |
0 |
0 |
0 |
1 |
2 |
|
Bank |
Status |
1993 |
1994 |
1995 |
1996 |
1997 |
|
Sibling Donor Cord Blood Program |
Collected |
|
|
|
|
4 |
|
Shipped |
|
|
|
|
1 |
|
|
St. Louis Cord Blood Bank |
Collected |
|
|
|
1,487 |
4,708 |
|
Banked |
|
|
|
695 |
1,544 |
|
|
Shipped |
|
|
|
0 |
9 |
|
|
StemCyte, Inc. and Cord Blood Family Trust |
Collected |
0 |
0 |
0 |
0 |
0 |
|
Shipped |
0 |
0 |
0 |
0 |
0 |
|
|
The Elie Katz Umbilical Cord Blood Program |
Collected |
0 |
0 |
0 |
4 |
162 |
|
Shipped |
0 |
0 |
0 |
0 |
0 |
|
|
University of Colorado Cord Blood Bank |
Collected |
|
|
|
4 |
569 |
|
Shipped |
|
|||||
|
Viacord |
Collected |
|
|
67 |
524 |
383 |
|
Shipped |
|
|
|
1 |
2 |
|
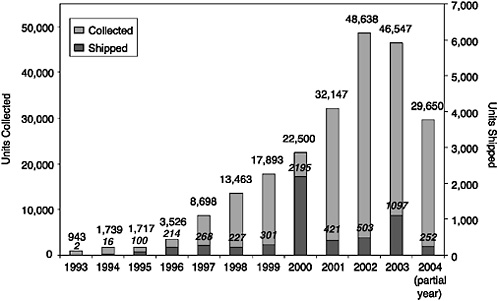
FIGURE C-3 Units collected and shipped, by year.
|
1998 |
1999 |
2000 |
2001 |
2002 |
2003 |
2004 |
|
33 |
160 |
251 |
252 |
307 |
312 |
123 |
|
2 |
2 |
7 |
5 |
11 |
17 |
2 |
|
5,969 |
5,334 |
4,237 |
4,117 |
5,082 |
5,377 |
3,280 |
|
1,447 |
1,188 |
1,059 |
1,023 |
1,286 |
1,546 |
843 |
|
38 |
74 |
96 |
151 |
129 |
110 |
49 |
|
|
|
|
Pu: 4,198 |
Pu: 7,748 |
Pu: 1,519 |
Pu: 101 |
|
0 |
0 |
0 |
|
|
Pr: 33 |
Pr: 147 |
|
0 |
0 |
0 |
Pu: 2 |
Pu: 9 |
Pu: 30 |
Pu: 37 |
|
632 |
1,066 |
916 |
134 |
157 |
220 |
344 |
|
0 |
6 |
3 |
5 |
3 |
6 |
4 |
|
1,161 |
1,779 |
3,935 |
2,495 |
|
|
|
|
3 |
24 |
42 |
44 |
43 |
45 |
33 |
|
750 |
1,560 |
2,027 |
5,248 |
12,826 |
19,426 |
18,259 (as of 10/04) |
|
|
1 |
2 |
3 |
2 |
1 |
2 |
TABLE C-16 Costs
|
|
Private |
||
|
Bank |
Collection |
Storage |
Release |
|
American Red Cross North Central Blood Services Cord Blood Program |
|
||
|
Carolinas Cord Blood Bank |
|
||
|
Children’s Hospital of Orange County Cord Blood Bank |
$1,700 |
$0 |
$0 |
|
ITxM Cord Blood Services |
|
|
$18,000 on release |
|
LifeCord |
$0 |
$0 |
$15,500 |
|
Michigan Community Blood Centers Cord Blood Bank |
|
||
|
National Cord Blood Program of the New York Blood Center |
|
||
|
New Jersey Cord Blood Bank |
|
||
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
$655 (processing and first unit of storage $1,000) |
$210/5 years |
shipping charges only, in addition to any requested testing |
|
Sibling Program Children’s Hospital Oakland Research Institute |
$0 |
$0 |
$19,600 |
|
St. Louis Cord Blood Bank |
|
||
|
StemCyte, Inc. and Cord Blood Family Trust |
$1,450 |
$95/year |
No cost |
|
The Elie Katz Umbilical Cord Blood Program |
$1,350 ($1,500 if paid in installment, $900 at specified hospitals) |
$100/year |
$0 |
|
University of Colorado Cord Blood Bank |
|
||
|
Viacord |
$1,800 |
$125/year |
$0 |
|
Public |
|
Reimbursement |
|
$18,000 |
|
Pending $18,500 |
|
$15,500 |
|
$15,000 per NMDP |
|
$15,500 |
|
$25,000 |
|
$22,490—includes patient CT-negative unit CT-positive Unit DNA for transplant center CT, unit shipment in the United States |
|
NMDP provides $15,500 for a unit when it is transplanted. |
|
$15,000 |
|
$15,000 before 2004, $21,500 after 1/1/04 |
|
$0 |
|
$17,000 |
TABLE C-17 Funding of Public Banks
|
Bank |
When |
NIH |
Private/Philanthropy |
State Funding |
|
American Red Cross North Central Blood Services Cord Blood Program |
Currently |
|
|
|
|
Historically |
|
X |
|
|
|
Carolinas Cord Blood Bank |
Currently |
|
|
|
|
|
Historically |
X |
X |
|
|
Children’s Hospital of Orange County Cord Blood Bank |
Currently |
|
X |
|
|
Historically |
|
X |
|
|
|
ITxM Cord Blood Services |
Currently |
|
X |
|
|
|
Historically |
|
X |
|
|
LifeCord |
Currently |
|
|
|
|
|
Historically |
|
|
|
|
Michigan Community Blood Centers Cord Blood Bank |
Currently |
|
X |
|
|
Historically |
|
X |
|
|
|
National Cord Blood Program of the New York Blood Center |
Currently |
|
X |
|
|
Historically |
X |
X |
|
|
|
New Jersey Cord Blood Bank |
Currently |
|
|
X |
|
|
Historically |
|
X |
X |
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
Currently |
|
X |
|
|
Historically |
|
X |
|
|
|
Sibling Program Children’s Hospital Oakland Research Institute |
Currently |
X |
X |
|
|
Historically |
X |
X |
|
|
|
St. Louis Cord Blood Bank |
Currently |
|
X |
|
|
|
Historically |
|
X |
|
|
StemCyte, Inc and Cord Blood Family Trust |
Currently |
|
X |
|
|
Historically |
|
X |
|
|
|
The Elie Katz Umbilical Cord Blood Program |
Currently |
|
|
|
|
Historically |
|
|||
|
University of Colorado Cord Blood Bank |
Currently |
|
|
|
|
Historically |
|
|
|
|
|
Public Bank |
|
|
Revenue |
Other |
|
X |
|
|
X |
Support From ARC |
|
|
X |
|
|
ARC |
|
|
By participating entities, UF, Shands, LifeSouth |
|
|
By participating entities, UF, Shands, LifeSouth |
|
X |
|
|
X |
|
|
X |
NYBC |
|
X |
NYBC |
|
X |
|
|
X |
|
|
X |
|
|
X |
|
|
X |
Fund raising |
|
X |
Fund raising |
|
|
Private bank revenue |
|
|
Private bank revenue |
|
|
Self-supporting from reimbursement fees |
|
|
University of Colorado Hospital |
TABLE C-18 Neonatal Health Status Follow-Up
|
Bank |
If There Is Follow-Up in Neonatal Health Status, When Is It Done? |
|
American Red Cross North Central Blood Services Cord Blood Bank |
Delivery data are assessed 24 hours or more after delivery. In addition, donors are reminded in a follow-up letter/survey mailed 60 days after delivery and in a birthday card sent at 1 year, to ask the family to report on any changes in infant’s health. |
|
Carolinas Cord Blood Bank |
Yes, 24-hour review of pediatric exam/neonatal history 6 months or more, computer system; mom’s age > 35 years and no amniocentesis follow-up unit at time of request |
|
Children’s Hospital of Orange County Cord Blood Bank |
None |
|
Cryobanks International, Inc. |
No follow-up of neonatal health status is done at present |
|
ITxM Cord Blood Services |
Yes, before release for transplantation; not always able to locate the mother |
|
JP McCarthy Cord Stem Cell Bank |
No follow-up after the baby has been discharged from the hospital |
|
Lifebank USA |
No |
|
LifeCord |
No. However, the mother is asked to call if a health problem arises; the bank receives notification of leukemia in an infant whose cord blood was donated. |
|
Michigan Community Blood Centers Cord Blood Bank |
(1) No formal follow-up. (2) Donor moms are given a card to keep in baby’s book asking for notification if child becomes seriously ill. |
|
National Cord Blood Program of the New York Blood Center |
No |
|
New Jersey Cord Blood Bank |
Follow-up from the donor is requested at 6 months and 1 year |
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
Yes, prior to transplant (when a cord blood unit has been selected for use) |
|
Sibling Program Children’s Hospital Oakland Research Institute |
Hemoglobinopathy screening is performed on the cord blood unit. Clinical status of donor is reviewed before release of the unit for transplantation |
|
Bank |
If There Is Follow-Up in Neonatal Health Status, When Is It done? |
|
St. Louis Cord Blood Bank |
Yes, 2 weeks postdelivery and when confirmatory typing is requested on a cord blood unit |
|
StemCyte, Inc. and Cord Blood Family Trust |
The mother is asked to notify the bank of any change in the health status of the child. A letter with a SASE requesting a response regarding the health of the child is sent between 6 months and 1 year postdelivery. |
|
The Elie Katz Umbilical Cord Blood Program |
No for public units; yes for private units |
|
University of Colorado Cord Blood Bank |
No |
|
Viacord |
Not proactive, at customer request |
TABLE C-19 How the Units Are Stored
|
|
Stored as |
||
|
Bank |
Whole Blood |
Volume Reduction Only |
RRC Depleted/Mononuclear Cell Product |
|
American Red Cross North Central Blood Services Cord Blood Bank |
|
|
X |
|
Carolinas Cord Blood Bank |
|
|
X |
|
Children’s Hospital of Orange County Cord Blood Bank |
|
|
X |
|
Cryobanks International, Inc. |
|
|
X |
|
ITxM Cord Blood Services |
X |
|
X |
|
JP McCarthy Cord Stem Cell Bank |
|
|
X |
|
Lifebank USA |
|
|
X |
|
LifeCord |
X (<50 processed this way) |
|
X |
|
Michigan Community Blood Centers Cord Blood Bank |
|
|
X |
|
National Cord Blood Program of the New York Blood Center |
|
|
X |
|
New Jersey Cord Blood Bank |
|
|
X |
|
Puget Sound Blood Center and Hawaii Cord Blood Bank |
|
|
X |
|
Sibling Program Children’s Hospital Oakland Research Institute |
|
X |
|
|
St. Louis Cord Blood Bank |
|
|
|
|
StemCyte, Inc. and Cord Blood Family Trust |
|
|
|
|
The Elie Katz Umbilical Cord Blood Program |
|
|
|
|
University of Colorado Cord Blood Bank |
|
|
X |
|
Viacell |
|
|
X |
|
Other |
Anticoagulant Used |
Cryoprotectant Used |
|
|
CPD |
DMSO/dextran |
|
|
X |
X |
|
|
X |
X |
|
|
CPD solution |
Stemsol-DMSO, 99% purity, |
|
|
USP |
USP grade |
|
X |
CPD |
DMSO-dextran |
|
|
|
X |
|
|
X |
X |
|
|
CPD-Adenine |
DMSO 10%, HES 1%, dextran40 1% |
|
|
X |
X |
|
|
ACD-A |
DMSO |
|
|
CPD |
DMSO-dextran 40, (unit nos. 668 to present) DSMO-pentastarch-plasma (unit nos 1 to 667) |
|
Buffy coat (RBC and plasma reduced) |
CPD |
DMSO |
|
Plasma depleted, non-redcell-depleted product |
CPD solution |
DMSO-Gentran |
|
Red cell depleted with volume reduction |
CPD |
10% DMSO |
|
|
CPD |
DMSO |
|
|
CPD |
DMSO |
ACRONYMS AND ABBREVIATIONS
AABB
American Association of Blood Banks
ACD-A
Anticoagulant Citrate Dextrose Solution, formula A
ARC
American Red Cross
ASHI
American Society for Histocompatibility and Immunogenetics
BMDW
Bone Marrow Donors Worldwide
BMT
bone marrow transplant
CAH
congenital adrenal hyperplasia
CAP
College of American Pathologists
CBB
cord blood bank
CLIA
Clinical Laboratory Improvement Amendments
CMV
cytomegalovirus
CPD
citrate-phosphate-dextrose
CRIR
Caitlin Raymond International Registry
DMSO
dimethyl sulfoxide
EBMT
European Group for Blood and Marrow Transplantation
FACT
Foundation for the Accreditation of Cellular Therapy
FDA
Food and Drug Administration
g
gram(s)
HbcAb
hepatitis B core antibody
HBV
hepatitis B virus
HCV
hepatitis C virus
HELLP
haemolysis, elevated liver enzymes, low platelets
HES
hetastarch
HIV
human immunodeficiency virus
HLA
human leukocyte antigen
IgG
immunoglobulin G
IND
investigational new drug
IRB
institutional review board
ISBT
International Society of Blood Transfusion
ISCT
International Society for Cellular Therapy
ISO
International Organization for Standardization
JACIE
Joint Accreditation Committee ISCT EBMT
JCAHO
Joint Commission for Accreditation of Healthcare Organizations
MCAD
medium-chain acyl-CoA dehydrogenase
ml
milliliter(s)
NAT
nucleic acid test
NIH
National Institutes of Health
NMDP
National Marrow Donor Program
NYBC
New York Blood Center
PCR
polymerase chain reaction
PKU
phenylketonuria
RBC
red blood cell
SARS
sudden acute respiratory syndrome
SASE
self-addressed stamped envelope
SQL
structured query language
SSP
split spectrum processing
TNC
total nucleated cell count
UCLA
University of California, Los Angeles
UF
University of Florida
USP
United States Pharmacopeia
vCJF
variant Creutzfeldt-Jakob disease
WMDA
World Marrow Donor Association
WNV
West Nile virus