4
Maternal Weight, Gestational Weight Gain, and Maternal Health
Weight patterns (underweight and overweight) and gestational weight gain can influence both short- and long-term consequences for maternal health. Understanding the effects of different weight patterns and gestational weight gain on maternal health outcomes requires close examination of the mechanisms that link gestational weight to later health conditions as well as consideration of risk and protective factors that contribute to or inhibit these effects. As discussed in Chapter 3, the relationship between prepregnancy weight and gestational weight gain is complex; their influence on maternal health outcomes is not easily distinguished, and this is reflected in this summary. In light of concerns about rising rates of obesity, the presenters focused their review on the contributions of excessive gestational weight gain and overweight status on maternal health outcomes. This focus is a reflection of the recent research efforts in this area.
SHORT-TERM MATERNAL HEALTH OUTCOMES
Kathleen Rasmussen provided an overview of the consequences of weight gain for women during pregnancy and immediately afterward. Both prepregnancy body mass index (BMI) and gestational weight gain are predictors of maternal health outcomes. These outcomes arise either through direct effects of BMI (prepregnancy or postpartum) or gestational weight gain, or through their interaction. These two factors may operate through the same mechanism, possibly an excess of fat, or through other mechanisms. In general, higher rates of pregnancy weight gain lead to more
negative health outcomes; this is particularly true for obese and overweight women. Details of these effects are described below.
Preeclampsia
Preeclampsia is an abnormal state of pregnancy characterized by hypertension and fluid retention and albuminuria; it can lead to eclampsia, preeclampsia combined with seizure-like convulsions, if untreated. A recent study of gestational weight gain and preeclampsia by Cedergren (2006) used data on all births registered in Sweden between 1994 and 2002. The data reflect the country’s 96 percent white population. The adjusted odds ratio of developing preeclampsia for women with weight gains of less than 18 lbs. was substantially reduced for all women with a BMI >20 when compared with women who gained between 18 and 35 lbs. Women in all BMI groups who gained more than 35 lbs. were at greater risk of developing preeclampsia than women who gained between 18 and 35 lbs.
Gestational Diabetes Mellitus
Although prepregnant BMI is a well-known predictor of gestational diabetes, the role of gestational weight gain as a separate predictor of this condition has not been demonstrated. An association between total gestational weight gain and diabetes is difficult to interpret, because once a woman is diagnosed with gestational diabetes, her weight gain is often intensely managed. Anna Marie Siega-Riz described a study (Saldana et al., 2005) that examined the relationship between weight gain and glucose intolerance during pregnancy by tracking weight gain up to the time when gestational diabetes was diagnosed. Results showed that the weight gain ratio (observed weight gain over the Institute of Medicine (IOM) recommended weight gain) was higher for women with gestational diabetes when compared statistically with women with normal glucose tolerance. The statistical likelihood of developing gestational diabetes was increased by both prepregnancy overweight and obesity status, while the interaction with weight gain during pregnancy was only marginal. A substantial interaction between prepregnant BMI and gestational weight gain arises when considering impaired glucose tolerance. In this study, women who were classified overweight before pregnancy and who also had excessive gestational weight gain were at a highly increased risk for impaired glucose tolerance when compared with women who had lower gestational weight gain and prepregnancy BMI. In addition, this association was especially strong for black women, suggesting that race/ethnicity may be a moderator of the interaction between prepregnancy BMI and gestational weight gain on glucose intolerance.
Cesarean Delivery
Dietz et al. (2005) used data from the Pregnancy Risk Assessment Monitoring System (PRAMS) to explore the impact of prepregnant BMI and gestational weight gain (independently and in interaction) on cesarean delivery. Statistically, women who were obese or severely obese (BMI >35) before pregnancy had an increased risk of cesarean delivery. In addition, women in the very highest gestational weight gain category (41 lbs. or more) had increased risk of cesarean delivery. There was no interaction between prepregnant BMI and weight gain during pregnancy in this study. In the discussion session, Siega-Riz noted another study with similar results (Vahratian et al., 2005). The possibility of an interaction between prepregnancy BMI and gestational weight gain in predicting the likelihood of cesarean delivery was also studied by Cedergren (2006) using a sample of Swedish women. Among women who gained less than 18 lbs. during pregnancy, those who were overweight, obese, or severely obese had a reduced risk of cesarean delivery compared with those who gained 18 to 35 lbs. during pregnancy. And regardless of a woman’s prepregnant BMI, gaining more than 35 lbs. compared with gaining 18 to 35 lbs. led to an increased risk of having a cesarean delivery.
Duration of Breastfeeding
Research has suggested an association between prepregnant BMI and failure to initiate and sustain breastfeeding among postpartum women. Inasmuch as the biological mechanism for this association could be the accumulation of fat it is possible that gestational weight gain may also play a role.
Data from PRAMS have not demonstrated an interaction between prepregnant BMI and gestational weight gain on the duration of any breastfeeding, but Rasmussen’s recent study of 2,700 women in Cooperstown, New York, reported such an interaction for the duration of exclusive breastfeeding (Hilson et al., 2006). Specifically, gaining more than the IOM recommended weight was associated with shorter breastfeeding duration for underweight women as well as overweight or obese women, when compared with normal-weight women.
Data on U.S. women may reflect the lack of support for breastfeeding in U.S. culture, so it is informative to conduct the same analysis in cultures in which breastfeeding is protected and supported. One such culture is Denmark, where women have 24 weeks of fully paid maternity leave, which encourages longer periods of breastfeeding. Unpublished analyses from 35,000 women in the Danish National Birth Cohort (J.L. Baker and K.M. Rasmussen, unpublished data) provide this counterpoint. In this
sample, the risk of early termination of full breastfeeding went up as BMI increased from underweight to normal to overweight to obese. In each BMI group, women who gained the most weight during pregnancy had the highest risk of early termination, and obese women were at risk of early termination of full breastfeeding regardless of how much weight they gained during pregnancy.
Postpartum Weight Retention
To investigate the association between gestational weight gain and postpartum weight retention, Rasmussen compared data from Greene et al. (1988) and from Muscati et al. (1996). Even though the Greene et al. data examined the amount of weight retained between pregnancies (adjusted for the duration between pregnancies) and those of Muscati et al. reported on weight retention at six weeks postpartum, analyses of both datasets suggest the same conclusion; women who gained more than 15.5 lbs. retained weight at the endpoint measured in each data set.
The only data available on minority women and postpartum weight retention are those drawn from the 1988 National Maternal and Infant Health Survey by Keppel and Taffel (1993). In this study, when women’s total weight gains were less than the IOM guidelines throughout their pregnancies, retention rates for both black and white women were similar at 10 to 18 months postpartum; the proportion of women who retained more than 14 lbs. was less than 10 percent of white women and about 15 percent of black women. At weight gains within the IOM recommendations, 35 percent of black women retained more than 14 lbs. but only 10 percent of white women did so. At weight gains above the IOM recommendations, proportions of both black and white women retained more than 14 lbs. statistically. Parker and Abrams (1993) have also used this data set to provide information about the gestational weight gain of black women. They found that when compared with white women, black women were consistently more likely to retain 20 lbs. at 10 to 18 months postpartum.
The data from the Bassett Mothers’ Health Project allow for a more detailed examination of gestational weight gain and postpartum retention (Olson et al., 2003). In general, in this study heavier women retained more weight if they had gained excessive amounts of weight, although this accounted for relatively little of the variance in postpartum weight retention. In multivariate analysis, Olson et al. demonstrated that a variety of factors predicted postpartum weight retention, including high levels of exercise, low food intake, still breastfeeding at a year postpartum, low gestational weight gain, and extremes of maternal age. One reported statistically significant interaction indicates that lower income women (defined as an annual family income <185 percent of the poverty income ratio) who gained
more than the recommended amount of weight were particularly likely to have retained more than 10 lbs. when compared with women with higher incomes. In addition, lower income women who were obese before pregnancy were also likely to retain more weight than obese women with higher incomes.
Finally, data from the Danish National Birth Cohort have been used to examine whether the duration of breastfeeding modifies the relationships between prepregnant BMI, gestational weight gain, and postpartum weight retention (J.L. Baker and the Postpartum Weight Retention Study Group, unpublished, personal communication). In this study, there was a 29 lbs. difference between those who retained the least at 6 months postpartum (obese women who gained below the IOM guidelines and fully breastfed their babies longer than 16 weeks) and those who retained the most (underweight women who gained above the IOM recommendations and breastfed less than 16 weeks). In other words, breastfeeding behavior modified the effects of both prepregnant BMI and gestational weight gain on postpartum weight retention.
Summary of Short-Term Maternal Health Outcomes
Table 4-1 summarizes the information on immediate and short-term maternal outcomes of gestational weight gain and prepregnancy BMI. Rasmussen observed that while more data were available since the 1990 IOM report, understanding these relationships among key variables re-
TABLE 4-1 Presence of an Association for Immediate and Short-Term Maternal Health Outcomes Attributable to Prepregnant Body Mass Index (BMI) and Gestational Weight Gain
mains limited. Information for minority groups is all but unavailable. The available data, however, consistently show associations between increasing prepregnant BMI, more specifically being overweight or underweight, and a range of negative maternal health outcomes—preeclampsia, gestational diabetes, cesarean delivery, failure to initiate and sustain breastfeeding, and postpartum weight retention at 1 year. The data that demonstrate relationships between gestational weight gain and these same negative outcomes are also consistent: excessive weight gain is associated with these same negative maternal health outcomes. What is currently either unknown or inconsistent in the literature is the nature of the interactions between prepregnant BMI (being overweight or underweight) and gestational weight gain in predicting these varied negative maternal health outcomes.
LONG-TERM MATERNAL HEALTH OUTCOMES
Erica Gunderson reviewed current knowledge about long-term health outcomes (more than 1-year postpartum) for mothers as a function of weight gain during pregnancy. In the 1990 IOM report, limited data were available on the effects of gestational weight gain on maternal health; the main concern was how much weight was gained during pregnancy or retained postpartum.
A growing literature on the influence of childbearing on women’s health examines not only the first few years after delivery but also subsequent decades. Some of the studies presented and reviewed include a nonpregnancy comparison group, which may help distinguish weight gain due to pregnancy from weight change unrelated to pregnancy. Other studies include only women who have had a pregnancy (pregnancy cohort), which may help identify maternal attributes that contribute to weight gain and retention.
Long-Term Weight Gain
The modifiable attributes or predictors of postpartum weight retention addressed in the 1990 IOM report include prepregnancy weight, parity, gestational weight gain, age, and lactation. Since 1990 a number of additional factors have emerged, which include prepregnancy body size (i.e., BMI), interval to first birth, socioeconomic status, and smoking, as well as the fixed attributes of race/ethnicity and age at menarche (see Table 4-2).
Prepregnancy Weight Patterns
Gunderson explained that researchers and clinicians have been trying to estimate long-term maternal weight gain, which is primarily assessed at
TABLE 4-2 Summary of Attributes of Postpartum Obesity
|
Before 1990 |
Since 1990 |
|
Modifiable Attributes Age Gestational weight gain Lactation Prepregnancy weight Parity |
Modifiable Attributes Age Gestational weight gain Lactation Prepregnancy BMI Parity Internal to first birth Socioeconomic status Smoking Fixed Attributes Race/ethnicity Age at menarche |
|
SOURCE: Gunderson (2006). |
|
two years after pregnancy, as a function of weight gain during pregnancy. Gunderson indicated that it has been known since 1957 that the heavier a woman is at the beginning of pregnancy (measured by BMI), the greater her weight is later (McKeown and Record, 1957). This weight includes both retained pregnancy gain and weight gained postpartum due to postpartum behaviors. In a Scottish study, overweight women had the greatest increases in body weight before subsequent pregnancies, and most of this was after the first birth (Billewicz, 1970). In other work with a perinatal cohort (Gunderson et al., 2001), higher long-term weight changes for obese women were found despite their lower pregnancy gain, with no real differences observed in BMI groups across race/ethnicity groups.
Recent pregnancy cohort studies, which primarily have included only primiparous women, have looked at long-term excess weight gain after pregnancy. These studies found that at two or more years postpartum, mean weight gain values compared with preconception weight varied somewhat but are estimated generally at about 6 lbs. (McKeown and Record, 1957), about 1.8 lbs. for women who were underweight, and 5.3 lbs. for women who were overweight (Billewicz, 1970). Harris et al. (1997) reported that 71 percent of primiparous women retained less than 2.2 lbs. In a study with women from multiple racial and ethnic groups, weight gain at 2 years postpartum was around 4.4 lbs. for underweight and normal-weight women and about 8.8 lbs. for overweight and obese women (Gunderson et al., 2001). The key risk factors for the excess weight gain after pregnancy, which were fairly consistent across these studies, were high prepregnancy weight, high prepregnancy BMI, high gestational weight gain, and parity.
Pattern of Postpartum Weight Loss
A study from 1957 looked at the pattern of maternal weight loss during the postpartum period by lactation duration (McKeown and Record, 1957). This is one of the few studies that includes serial weight measurements after delivery, in this case three times during the first year and at 24 months postpartum. The study indicates a potential pattern of postpartum maternal weight loss and suggests a point at which there is a change from a loss to a gain.
As mentioned earlier, work done in the pregnancy cohort looked at whether pregnancy gain varied by prepregnancy BMI (Gunderson et al., 2001). The overweight women gained (net gain) the most weight during pregnancy (30 lbs.). What is striking is that the early net postpartum weight change (first 6 weeks) was almost identical across BMI groups, such that early net weight gain did not vary, but late postpartum net weight change varied tremendously (Figure 4-1). In this study, larger declines occurred in the lighter women than the heavier women. From 6 weeks postpartum to a
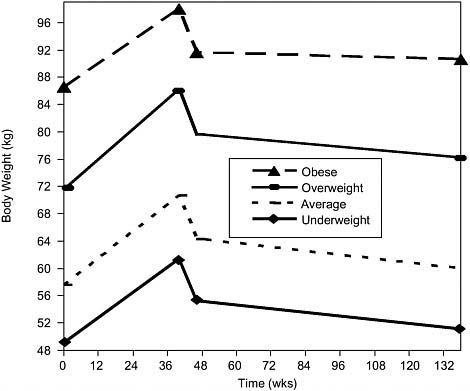
FIGURE 4-1 Patterns of maternal weight changes during gestation and postpartum periods by prepregnancy BMI.
SOURCE: Gunderson et al. (2001). Reprinted by permission from Macmillan Publishers Ltd: International Journal of Obesity.
FIGURE 4-2 Net early and late postpartum weight loss as a percentage of net gestational gain by prepregnancy BMI.
SOURCE: Gunderson et al. (2001). Adapted by permission from Macmillan Publishers Ltd: International Journal of Obesity.
point 2 years later, there was a downward trend for most of the groups, except for the obese women, whose weight remained steady. After adjusting for secular trends as well as underreporting of preconception weight (because it was between two pregnancies), additional weight gain occurred after 6 weeks postpartum among the obese and the overweight women. Gunderson indicated that as a proportion of the total pregnancy gain, obese women gained the least during pregnancy and 56 percent of their early 6-week weight loss postpartum was due to the pregnancy gain. After 6 weeks, however, their weight loss was a much smaller proportion (only 9 percent), and overall they ended up losing a lower proportion of the pregnancy gain over time compared with women with a prepregnant BMI categorized as underweight, normal, and overweight (Figure 4-2).
Incidence and Correlates of Overweight After Pregnancy
Incidence Another important outcome that was addressed is the shift in BMI categories. Using 1998 national data, Parker and Abrams (1993) calculated the risk of retaining more than 20 lbs. at 2 years postpartum. Overall, 14 percent of women retained more than 20 lbs. and a higher proportion of black than white women had excessive weight retention. A study of Swedish women indicated that 8 percent became overweight (Ohlin and Rossner, 1994) within a year after pregnancy, and a study of a pregnancy cohort (Gunderson et al., 2000) found that 6.4 percent became overweight within two years after pregnancy. Of those who became overweight, 69 percent had excessive gestational weight gain. A higher propor-
tion of those who became overweight were Hispanics and blacks, and lower proportions were Asians compared with white women.
Correlates A multivariate model using those same data from the pregnancy cohort looked for associations with excess weight gain postpartum (Gunderson et al., 2001). In this model, excessive gestational weight gain led to a threefold increase in risk of excess weight gain postpartum and a doubling of the risk was associated with increasing age. The researchers also found that women who had short intervals (less than 8 years) between menarche and first birth had a threefold higher risk of excess weight gain postpartum. Age at menarche of less than 12 years, which has been looked at as a predictor of weight change over time, was also independently associated with an almost threefold increase in risk of excess weight gain postpartum. Asian ethnicity was a protective factor for excess weight gain postpartum, with no meaningful increase in risk of excess weight gain postpartum for Hispanics or blacks compared with a white reference group. Gunderson concluded that gestational weight gain is a strong predictor of excess weight gain postpartum, but other biological factors can be strong independent effects.
Gunderson et al. (2004b) and Williamson et al. (1994) have examined childbearing and the risk of becoming overweight. In these studies, they compared women who gave birth to women who did not during a fixed interval. They included preconception measured weights on all the women as well as measured weights during the study follow-up period. Williamson et al. (1994) found that having one birth versus zero doubled the risk of becoming obese. Gunderson et al. (2004b) found an interaction between childbearing and smoking status; for smokers, childbearing reduced the risk of becoming overweight, and for never-smokers childbearing more than doubled the risk of becoming overweight with childbearing (Gunderson et al., 2004b). In addition, in this study, independent of childbearing, black women were 3.5 times more likely to become overweight, whether or not they had a birth (Gunderson et al., 2004b). Parker and Abrams (1993) found overall that women with high gestational weight gain had an increased risk of being overweight postpartum, and this was especially true for black women as compared to white women.
A few studies have looked at whether weight gain during pregnancy might predict weight gain 8 to 15 years later in life (Linne and Rossner, 2003; Rooney and Schauberger, 2002). These studies include only white women and have high attrition rates, but overall their results suggest that high prepregnancy BMI, weight retention at 6 months postpartum, gestational weight gain, and short breastfeeding duration are associated with greater weight gain later in life.
In summary, Gunderson reviewed the incidence and correlates of substantial weight gain after pregnancy (more than 2 years postpartum) based
on pregnancy cohort studies. These correlates include high prepregnancy BMI, gestational weight gain greater than the 1990 IOM recommendations, primiparity, short interval from menarche to first birth, and menarche before 12 years of age.
Long-Term Changes in Adiposity
Two prospective studies with data on long-term weight gain for primiparous women and a nonpregnancy comparison group report conflicting results (Coronary Artery Risk Development in Young Adults [CARDIA] and National Health and Nutrition Examination Study [NHANES] Epidemiologic Follow-up Study). The CARDIA study results (a 5- and 10-year study) suggested that primiparous women have excessive weight gain postpartum, about 4 to 7 lbs. more than nulliparous women (who have no births) (Gunderson et al., 2004a; Smith et al., 1994). The NHANES Epidemiologic Follow-up Study (a 10-year study), which involves a smaller number of primiparous women, found that the multiparous women are the ones with excess gain postpartum (Williamson et al., 1994; Wolfe et al., 1997). The CARDIA study (Gunderson et al., 2004a) examined a large sample of women for 10 years and found that, among normal-weight women, the postpartum excess gain due to childbearing was only about 2 lbs. but women who were overweight before pregnancy gained 7 to 13 lbs. This result occurred only among the primiparous women and was the same for blacks and whites. The absolute weight gain among overweight black women was higher than for overweight white women, and the absolute gain in the black normal-weight group was higher than the white normal-weight group. Looking across number of births, the researchers found that among overweight women, black women show 7 to 11 lbs. long-term postpartum weight gain, while white women show an 11 to 13 lbs. gain due to a single birth.
In a follow-up study of the Black Women’s Health Survey, the researchers found (over a 4-year period) that primiparous women gained more net weight than nulliparous ones, and excess net weight gain associated with one birth was about 7 lbs. for overweight women, who also had some increase in BMI with gestational weight gain (Rosenberg et al., 2003). An increase in BMI was found among women who gained more than 25 lbs. during pregnancy.
Other Long-Term Health Outcomes
Independent of weight gain, other conditions associated with childbearing—such as abdominal obesity, plasma lipid changes, and an increased risk of chronic disease, including coronary heart disease and diabetes—may affect women’s health.
Abdominal Obesity
The CARDIA study measured waist girth to gain insight into the association between childbearing and abdominal obesity (Gunderson et al., 2004a; Smith et al., 1994). Waist girth was measured in this prospective study at preconception for those who bore children and these women were compared with a nongravid (not pregnant) reference group. Primiparous women gained more abdominal girth than nulliparous women (Gunderson et al., 2004a; Smith et al., 1994). There is a similar association with excess gains in waist circumference for normal-weight and overweight women (primiparous and multiparous). Normal-weight women (both black and white) showed about 1 inch excess waist girth gain, and overweight black and white women showed very similar excess gain of 1.5 and 2.5 inches, respectively (Gunderson et al., 2004a).
Plasma Lipid Changes
There also appears to be an association between higher parity and with lower high-density lipoprotein (good cholesterol) levels, and in cross-sectional studies, researchers have found a threshold effect with a high number of births. However, in the best prospective studies of women of childbearing age, having one birth (versus none) contributed to a drop of 3.5 mg/dl on average (Lewis et al., 1996). In the longer term (10 years), there appears to be a 3 to 4 mg/dl difference between primiparous and nulliparous women, with similar changes for black and white women (Gunderson et al., 2004a).
Chronic Diseases
In terms of pregnancy and future risk of chronic disease, a study using a pregnancy cohort found that a high BMI and excess weight gain over 15 years were associated with increased risk of future chronic disease although the researchers did not report a direct association between gestational weight gain and chronic disease (Rooney et al., 2005). Higher lifetime parity has been associated with coronary heart disease risk in women, but the data are conflicting (Colditz et al., 1987; Ness et al., 1994; Rosenberg et al., 1999; Steenland et al., 1996). Diabetes has also been associated with lifetime parity in several early studies, but this association was confounded by age, fertility, socioeconomic status, and BMI.1
Cross-sectional and population-based studies of the association of parity with type II diabetes have produced conflicting results. A single prospective study reported no association between parity and diabetes when age, BMI, and family history of diabetes were controlled (Manson et al., 1992). For women with gestational diabetes, there is some evidence that an additional pregnancy is associated with a threefold higher risk of type II diabetes, independent of weight gain (Peters et al., 1996).
Research Needs
The workshop discussion sessions identified many gaps in the existing body of knowledge. First, understanding the influence of social determinants of health on maternal weight is a major priority. Second, studies should explore the risk of chronic disease (type II diabetes, hypertension, arthritis, cancers) by prepregnancy weight or gestational weight gain and the impact of these factors on not only weight but also abdominal adiposity, body composition, and other risk factors for cardiovascular disease. Third, the field needs more information about preconception measures of the risk factors, gestational weight gain, postpartum weight patterns, whether postpartum weight gain is really retention or secular trends, and what point in time is the most important to intervene. Fourth, research is needed that examines lactation and long-term weight changes in women. Finally, there are few data on Hispanic or Asian women.
Summary of Long-Term Maternal Health Outcomes
In summary, cumulative gains in abdominal obesity, lower high-density lipoprotein levels independent of obesity, and weight gain during midlife are all associated with increased risk of chronic diseases in women. There is some evidence about an increased risk of diabetes after experiencing gestational diabetes. However, the evidence is insufficient about whether childbearing itself is associated with these chronic diseases, or whether the gain over time is due to factors other than childbearing.
The greatest long-term health effects of childbearing (>1-year postpartum) appear to occur after a first birth. These effects occur mostly among women who were already overweight before pregnancy and who have excessive weight gain after pregnancy, higher waist girth, and lower high-density lipoprotein values. Among normal-weight women, there is a modest increase in the risk of postpartum overweight following childbirth as well as a cumulative effect with each birth on waist girth. The long-term health effects of childbearing do not seem to differ by race/ethnicity as long as maternal obesity at the start of pregnancy is taken into account.
SUMMARY
At the workshop, Rasmussen and Gunderson reviewed research on maternal health outcomes of gestational weight gain. Understanding the effects of different weight patterns (overweight or underweight) and gestational weight gain on maternal health outcomes requires further attention to a broad array of factors associated with the status of women’s health in different demographic groups. New data exist on the consequences of insufficient or excessive gestational weight gain for women, but these data are limited in scope. Recent research efforts focus more on being overweight and excessive gestational weight gain as risk factors for poor maternal health outcomes. Information for minority groups primarily includes only non-Hispanic black U.S. women; almost no data are available on Hispanic, Asian, or Native American populations of women.
Although limited, the data available consistently show associations between prepregnant BMI and a range of negative immediate and short-term maternal health outcomes (<1-year postpartum) including preeclampsia, gestational diabetes, cesarean delivery, failure to initiate and sustain breastfeeding, and postpartum weight retention at 1 year. Studies that show a relationship between gestational weight gain and these same immediate and short-term maternal health outcomes are also consistent. However, whether or not there is an interaction between prepregnant BMI (overweight or underweight) and gestational weight gain in predicting immediate and short-term maternal health outcomes is unknown or inconsistent in the literature.
A limited yet growing literature has demonstrated the influence of childbearing on women’s long-term maternal health outcomes (>1-year postpartum). This research base generally uses (1) prospective studies, which may help distinguish weight gain due to pregnancy from weight change unrelated to pregnancy, and (2) pregnancy cohort studies, which may help identify maternal attributes that contribute to weight gain and retention. This body of research has suggested that cumulative gains in abdominal obesity, lower high-density lipoprotein levels independent of the obesity, and weight gain during midlife are all associated with increased risk of chronic diseases in women. However, the evidence is insufficient about whether the childbearing experience is associated with these chronic diseases specifically, or whether the gain over time is due to factors other than childbearing. More research is needed on the impact of childbearing independent from and in combination with additional risk factors on long-term health outcomes. There is some evidence suggesting an increased risk of diabetes after experiencing gestational diabetes. The most serious longer term maternal health outcomes of childbearing appear to occur mostly among women who were already overweight before pregnancy, have excessive gains in weight and waist girth after pregnancy, and have lower high-
density lipoprotein levels. In addition, among women with a normal BMI there is a modest increase in the risk of overweight postpartum, and each birth has a cumulative effect on waist girth.
The high rates of obesity among women of reproductive age suggest that guidelines that are directed solely at changing gestational weight gain should be viewed as only one component of a comprehensive strategy to limit the long-term maternal effects of chronic obesity. Since the ability to predict gestational weight gain is limited and the consequences of inappropriate gestational weight gain are poorly understood, more research is necessary to inform the knowledge base that could guide policy and professional guidance. At a minimum, attention needs to focus on prepregnancy weight status and gestational weight gain, independently and in combination, as strong indicators of subsequent weight gain trends. Additional attention needs to be paid to the adolescent population, since the nature and strength of these relationships may differ from those found among older mothers.
REFERENCES
Billewicz, W.Z. 1970 Body weight in parous women. British Journal of Preventive Social Medicine 24(2): 97–104.
Cedergren, M. 2006 Effects of gestational weight gain and body mass index on obstetric outcome in Sweden. International Journal of Gynaecology and Obstetrics 93(3):269–274.
Colditz, G.A., Willett, W.C., Stampfer, M.J., Rosner, B., Speizer, F.E., and Hennekens, C.H. 1987 A prospective study of age at menarche, parity, age at first birth, and coronary heart disease in women. American Journal of Epidemiology 126(5):861–870.
Dietz, P.M., Callaghan, W.M., Morrow, B., and Cogswell, M.E. 2005 Population-based assessment of the risk of primary cesarean delivery due to excess prepregnancy weight among nulliparous women delivering term infants. Maternal and Child Health Journal 9(3):237–244.
Greene, G.W., Smiciklas-Wright, H., Scholl, T.O., and Karp, R.J. 1988 Postpartum weight change: How much of the weight gained in pregnancy will be lost after delivery? Obstetrics and Gynecology 71(5):701–707.
Gunderson, E.P. 2006 Childbearing, Gestational Gain and Long-term Effects on Women’s Health: Obesity and Chronic Disease. Presentation at the Workshop on the Impact of Pregnancy Weight on Maternal and Child Health, May 30, Washington, DC.
Gunderson, E.P., Abrams, B., and Selvin, S. 2000 The relative importance of gestational gain and maternal characteristics associated with the risk of becoming overweight after pregnancy. International Journal of Obesity Related Metabolic Disorders 24(12):1660–1668.
2001 Does the pattern of postpartum weight change differ according to pregravid body size? International Journal of Obesity Related Metabolic Disorders 25(6):853–862.
Gunderson, E.P., Murtaugh, M.A., Lewis, C.E., Quesenberry, C.P., West, D.S., and Sidney S. 2004a Excess gains in weight and waist circumference associated with childbearing: The Coronary Artery Risk Development in Young Adults study (CARDIA). International Journal of Obesity Related Metabolic Disease 28(4):525–535.
Gunderson, E.P., Quesenberry, C.P., Lewis, C.E., Tsai, A.L., Sternfeld, B., West, D.S., and Signey, S. 2004b Development of overweight associated with childbearing depends on smoking habit: The Coronary Artery Risk Development in Young Adults (CARDIA) study. Obesity Research 12(12):2041–2053.
Harris, H.E., Ellison, G.T., Holliday, M., and Lucassen, E. 1996 The impact of pregnancy on the long-term weight gain of primiparous women in England. International Journal of Obesity and Metabolic Disorders 21(9):747– 755.
Hilson, J.A., Rasmussen, K.M., and Kjolhede, C.L. 2005 Excessive weight gain during pregnancy is associated with earlier termination of breast-feeding among white women. Journal of Nutrition 136(1):140–146.
Institute of Medicine 1990 Nutrition During Pregnancy. Washington, DC: National Academy Press.
Keppel, K.G., and Taffel, S.M. 1993 Pregnancy-related weight gain and retention: Implications of the 1990 Institute of Medicine guidelines. American Journal of Public Health 83(8):1100–1103.
Lewis, C.E., Funkhouser, E., Raczynski, J.M., Signey, S., Bild, D.E., and Howard, B.V. 1996 Adverse effect of pregnancy on high density lipoprotein (HDL) cholesterol in young adult women. The CARDIA study: Coronary Artery Risk Development in Young Adults. American Journal of Epidemiology 144(3):247–254.
Linne, Y., and Rossner, S. 2003 Interrelationships between weight development and weight retention in subsequent pregnancies: The SPAWN study. Acta Obstetrics et Gynecology Scandanavia 82(4): 318–325.
Manson, J.E., Rimm, E.B., Colditz, G.A., Stampfer, M.J., Willett, W.C., Arky, R.A., Rosner, B., Hennekens, C.H., and Speizer, F.E. 1992 Parity and incidence of non-insulin-dependent diabetes mellitus. American Journal of Medicine 93(1):13–18.
McKeown, T., and Record, R.G. 1957 The influence of weight and height on weight changes associated with pregnancy in women. Journal of Endocrinology 15:423–429.
Muscati, S.K., Gray-Donald, K., and Koski, K.G. 1996 Timing of weight gain during pregnancy: Promoting fetal growth and minimizing maternal weight retention. International Journal of Obesity Related Metabolic Diseases 20(6):526–532.
Ness, R.B., Schotland, H.M., Flegal, K.M., and Shofer, F.S. 1994 Reproductive history and coronary heart disease risk in women. Epidemiological Reviews 16(2):298–314.
Ohlin, A., and Rossner, S. 1994 Trends in eating patterns, physical activity and socio-demographic factors in relation to postpartum body weight development. British Journal of Nutrition 71(4): 457–470.
Olson, C.M., Strawderman, M.S., Hinton, P.S., and Pearson, T.A. 2003 Gestational weight gain and postpartum behaviors associated with weight change from early pregnancy to 1 yr postpartum. International Journal of Obesity Related Metabolic Disorders 27(1):117–127.
Parker, J.D., and Abrams, B. 1993 Differences in postpartum weight retention between black and white mothers. Obstetrics and Gynecology 81(5 Pt 1):768–774.
Peters, R.K., Kjos, S.L., Xiang, A., and Buchanan, T.A. 1996 Long-term diabetogenic effect of single pregnancy in women with previous gestational diabetes mellitus. Lancet 347(8996):227–230.
Rasmussen, K.M. 2006 Gestational Weight Gain: Short-term Maternal Health Outcomes. Presentation at the Workshop on the Impact of Pregnancy Weight on Maternal and Child Health, May 30, Washington, DC.
Rooney, B.L., and Schauberger, C.W. 2002 Excess pregnancy weight gain and long-term obesity: One decade later. Obstetrics and Gynecology 100(2):245–252.
Rooney, B.L., Schauberger, C.W., and Mathiason, M.A. 2005 Impact of perinatal weight change on long-term obesity and obesity-related illnesses. Obstetrics and Gynecology 106(6):1349–1356.
Rosenberg, L., Palmer, J.R., Rao, R.S., and Adams-Campbell, L.L. 1999 Risk factors for coronary heart disease in African American women. American Journal of Epidemiology 150(9):904–909.
Rosenberg, L., Palmer, J.R., Wise, L.A., Horton, N.J., Kumanyika, S.K., and Adams-Campbell, L.L. 2003 A prospective study of the effect of childbearing on weight gain in African-American women. Obesity Research 11(12):1526–1535.
Saldana, T.M., Siega-Riz, A.M., Adair, L.S., and Suchindran, C. 2005 The relationship between pregnancy weight gain and glucose tolerance status among black and white women in central North Carolina. American Journal of Obstetrics and Gynecology July 3: E-pub.
Smith, D.E., Lewis, C.E., Caveny, J.L., Perkins, L.L., Burke, G.L., and Bild, D.E. 1994 Longitudinal changes in adiposity associated with pregnancy. Journal of the American Medical Association 271(22):1747–1751.
Steenland, K., Lally, C., and Thun, M. 1996 Parity and coronary heart disease among women in the American Cancer Society CPS II population. Epidemiology 7(6):641–643.
Vahratian, A., Siega-Riz, A.M., Savitz, D.A., and Zhang, J. 2005 Maternal pre-pregnancy overweight and obesity and the risk of primary cesarean delivery in nulliparous women. Annals of Epidemiology 15(7):467–474.
Williamson, D.F., Madams, J., Pamuk, E., Flegal, K.M., Kendrick, J.S., and Serdula M.K. 1994 A prospective study of childbearing and 10-year weight gain in U.S. white women 25 to 45 years of age. International Journal of Obesity Related Metabolic Disorders 18(8):561–569.
Wolfe, W.S., Sobal, J., Olson, C.M., Frongillo, E.A., and Williamson, D.F. 1997 Parity-associated weight gain and its modification by sociodemographic and behavioral factors: A prospective analysis in U.S. women. International Journal of Obesity Related Metabolic Disorders 21(9):802–810.

















