5
Maternal Weight, Gestational Weight Gain, and Children’s Health
A major session of the workshop addressed the role of maternal weight and gestational weight gain as direct predictors and moderators of children’s growth and health. Matthew Gillman introduced the session by observing that the 1990 Institute of Medicine (IOM) report placed an emphasis on birth outcomes that are chiefly related to low birth weight, which was a particular concern at that time. However, in this current era of epidemic obesity, attention has expanded the range of birth weight outcomes of interest as well as other salient short- and long-term outcomes for which birth weight is a proxy. Gillman also discussed fetal and developmental origins of chronic disease, and the life-course approach, which postulates that perturbations during the earliest stages of human development can have lifelong impact on chronic disease. Gillman expressed the view that the human development model may be an important key in understanding the effects of maternal weight and weight patterns during pregnancy on infants’ and children’s growth and health.
The following sections review the available research on short-term infant health outcomes and the long-term child health outcomes of maternal and gestational weight gain. In general, short-term health outcomes refer to infants up to 1 year of age. Long-term health outcomes refer to children over 1 year. Although the charge to the committee is a focus on infant health outcomes (up to 12 months), several presenters also reviewed literature on older child health outcomes of maternal and gestational weight gain, and this research is reflected in this chapter. The primary focus of this
review is the influence of excessive gestational weight gain and overweight pregnancies on child health outcomes, reflecting recent research trends.
SHORT-TERM INFANT HEALTH OUTCOMES
Patrick Catalano provided an overview of the impact of prepregnancy maternal weight and gestational weight gain on the fetus. Specifically, he discussed preterm delivery, intrauterine growth restriction, macrosomia, and body composition. As mentioned in Chapter 2, statistical increases have occurred in prepregnancy weight in women of childbearing age. The prevalence of obesity has nearly doubled from the 1980s to early 2000, to close to 30 percent. In addition, data from MetroHealth Medical Center in Cleveland show the mean maternal weight at the time of delivery has increased to 190 lbs. in 2003, up from 170 lbs. in 1987.
Preterm Delivery
Maternal prepregnancy weight and gestational weight gain are related to preterm delivery.1 A recent meta-analysis of 13 studies published from 1980 through 1996 concluded that inadequate weight gain is associated with an increased risk of prematurity, with a possible indication specifically on inadequate gain late in pregnancy (Carmichael and Abrams, 1997). Overall, about 75 percent of preterm deliveries are not medically indicated but occur from spontaneous labor due to premature rupture of the membranes. In the past 10 years, an increased number of multiple births has also occurred, which are frequently associated with preterm deliveries. Approximately one-quarter of all preterm births are indicated on the basis of maternal complications, such as hypertension, diabetes, and preeclampsia. Women at the greatest risk for having a preterm birth are those with a history of preterm birth, with about three to four times the baseline population risk.
Analyses using the Pregnancy Nutrition Surveillance System show that the risk of preterm delivery varied by both maternal prepregnancy body mass index (BMI) and gestational weight gain (Schieve et al., 1999). The lowest risk of preterm delivery was found in women gaining between 0.6 and 1 lb per week, and the highest preterm delivery rate was to women who gained below or above these parameters. The greatest risk was to those women with a low prepregnancy BMI and a gestational weight gain of less than 0.2 lb/week. Similar results were found using the National Maternal
and Infant Health Survey excluding medically indicated preterm deliveries (Schieve et al., 2000), and analyzing data from the Pregnancy Risk Assessment Monitoring System (Dietz et al., 2006).
Preterm risk associations with gestational weight gain may be related to maternal adipose tissue and cytokine production, because preterm delivery is related to a maternal or fetal inflammatory response. In addition, recent studies indicate that inadequate gestational weight gain and low prepregnancy BMI are associated with an increased risk of premature deliveries; the highest risk was found in underweight women who gained little weight during pregnancy. Although a substantial increase in BMI has occurred in women of reproductive age, the preterm delivery rates in the United States have steadily increased in the past 20 years from about 8 to about 12 percent (Mercer et al., 2006). This trend suggests that other factors are influencing preterm birth in addition to prepregnancy BMI and gestational weight gain, especially since an increase in prepregnancy BMI should be protective against preterm birth.
Mercer conducted a secondary analysis of the preterm prediction study, a component of the Maternal Fetal Medicine Network Units of the National Institute on Child Health and Human Development, looking at factors related to preterm delivery (Mercer et al., 2006). He compared four groups: (1) women who had recurrent term births only (two or three term births), (2) women with no history of any preterms, (3) women who had an isolated spontaneous preterm birth and a term birth in between, and (4) women who were at the highest risk of recurrent spontaneous preterm births, with two or three spontaneous preterm births and no interval term pregnancy. He further divided women who had isolated spontaneous preterm births based on whether this delivery was prior to or was itself the index pregnancy for the study. Results showed that 38 percent of women with low prepregnancy BMI (less than 19.8) and recurrent spontaneous preterm births delivered preterm. Compared with women with either recurrent term births or isolated spontaneous preterm births, those women with a low prepregnancy BMI had a greater risk of another spontaneous preterm delivery. The women who had recurrent spontaneous preterm births had a greater risk of low BMI and low weight gain in the early part of pregnancy. The women who had the recurrent spontaneous preterm births also had the shortest cervixes compared with the other groups, a characteristic related to their BMI, not to their height. Levels of maternal plasma cytokines, in this particular instance IL-6, were lower in the women who had recurrent spontaneous preterm births; women who were lean and had recurrent spontaneous preterm births had lower values. In conclusion, women with recurrent spontaneous preterm births weigh less and are leaner before and during the pregnancy, have shorter cervixes and an advanced Bishop score (an indication of readiness to induce birth) by 22 to 24 weeks related to the pre-
pregnancy weight but not their height, and they deliver at an earlier gestational age than those who did not have an isolated spontaneous preterm birth in their current pregnancy.
Intrauterine Growth Restriction
Maternal prepregnancy weight and gestational weight gain are also related to intrauterine growth restriction. Most people define intrauterine growth restriction as less than 10 percent weight for gestational age, adjusting for gender, race, and geography, primarily altitude. Many different factors contribute to intrauterine growth restriction, including fetal placental factors, such as chromosomes, genetic syndromes, congenital malformations, infectious disease, and placental pathologies. Other factors include such medical problems as maternal hypertension, diabetes, particularly involving vasculopathies, low maternal prepregnancy weight, premature labor, hypoxia, and substance abuse.
Catalano stated that poor maternal weight gain in pregnancy has not been found to be directly related to the risk of intrauterine growth restriction.2 For example, in the Dutch winter famine of 1945, women in the latter part of pregnancy were severely restricted in their calories, but the average birth weight decreased by only 240 grams. In data published in 2002, the number of term small-for-gestational-age births that occurred from 1985 through 1998 decreased in both the white and the black populations by 11 to 12 percent in the United States; in Canada, the numbers decreased by 27 percent (Ananth and Wen, 2002).
Macrosomia
On the other extreme are babies born with extremely high birth weights, or macrosomia, defined as birth weight greater than 90th percentile, and also referred to as large for gestational age. A commonly used criterion is 4,000 grams (about 9 lbs.), but this confounds some of the important predictors of macrosomia, notably gestational age, the most important factor, as well as altitude, socioeconomic status, and race/ethnicity. International data suggest an increasing incidence of macrosomic births. In Denmark in the past 10 years the mean birth weight has increased 45 grams, and the number of babies greater than 9 lbs. has increased to 20 percent (Orskou et al., 2001). In Sweden during the same time period there has
been a 23 percent increase in the incidence of large babies, which is related to an increase in maternal BMI and a decrease in smoking (Surkan et al., 2004). The incidence of term babies large for gestational age has increased between 5 and 9 percent in the United States during the same time period, and in Canada up to 24 percent (Ananth and Wen, 2002). The epidemiological data show the number of large-for-gestational-age babies seems to be increasing in the population.
Finally, Catalano looked at data from Cleveland’s MetroHealth Medical Center dating back to 1975, which shows a mean increase in birth weight of 116 grams. The rate of birth of large babies, variously defined, is increasing, a trend that is primarily related to maternal prepregnancy weight.
Infant Body Composition
Catalano examined an emerging area of research on body composition at birth in humans and its possible relationship to maternal weight and gestational weight gain. Humans, of all the mammalian species, have the greatest amount of body fat at the time of delivery, around 12 to 15 percent. Studies have considered lean body mass to have a genetic component, and fat mass may be reflecting the intrauterine environment (Sparks, 1984).
One study of women with normal glucose tolerance and women with gestational diabetes looked at the factors relating to fetal growth as indexed by body composition measures (see Box 5-1) (Catalano and Ehrenberg, 2006). Attributable variance of the infant’s birth weight, lean body mass, fat mass, and percentage body fat were calculated. Gestational age is the strongest predictor of infant birth weight, followed by maternal prepregnancy weight, maternal gestational weight gain, smoking (as a negative factor), and parity. These factors account for 24 percent of the total variance in birth weight. Infant lean body mass at birth was predicted by gestational age (the strongest predictor), smoking, maternal prepregnancy weight and maternal weight gain during pregnancy, parity, maternal height, and genetic influence. These factors account for a total of about 25 percent of the variance. Maternal prepregnancy BMI has the strongest correlation with infant fat mass at the time of delivery (among term singleton infants). Gestational age is an important predictor of infant fat mass, but maternal gestational weight gain accounts for only about half of the variance of infant fat mass compared with variance accounted for by maternal prepregnancy BMI. Even though close to half of the women had gestational diabetes with a known risk factor for macrosomia, it accounted for only about 1.6 percent of the variance of about 19 percent explained. The data for percentage body fat were similar to fat mass for the infant, maternal
prepregnancy BMI being the strongest correlate, gestational age followed by maternal weight gain during pregnancy, and gestational diabetes.
One study compared the body composition of neonates born to lean normal-weight women (prepregnancy BMI less than 25) and women who were overweight or obese (BMI greater than or equal to 25) prior to their pregnancy (Sewell et al., 2006). In this one study, infant birth weight was slightly greater (not statistically significant) for the prepregnancy overweight and obese women compared with the lean normal-weight women. Among other body composition variables, there was no difference in infant lean body mass between the two groups of mothers. However, the women who were overweight or obese before pregnancy had infants who had a greater the amount of fat mass and percentage body fat than the lean normal-weight women. Prepregnancy BMI was not predictive of infant lean body mass, but gestational age was the best predictor of infant lean body mass in women who were lean and of average weight before pregnancy. Percentage body fat for the infant was predicted by gestational age plus the fetal gender (female) for the lean average weight. Considering the women who were
overweight or obese before they got pregnant, gestational age plus fetal gender (male) correlated with infant lean body mass. In women who were overweight or obese prior to pregnancy, maternal weight gain in pregnancy, the 1-hour glucose screen, and gestational age contributed to the infant’s percent body fat.
Summary of the Short-Term Infant Health Outcomes
The review by Catalano at the workshop indicated that lower rates of maternal prepregnancy weight and gestational weight gain increase the risk of preterm delivery.3 There has been a substantial increase in infant birth weight concomitant with the increase in maternal weight over the past decade. Gestational age is the strongest predictor of fetal fat mass. In addition, fetal fat mass has a stronger correlation with maternal prepregnancy BMI than gestational weight gain. Finally, for women who are overweight and obese prior to conception, an increase in maternal weight gain during pregnancy is correlated with an increase in fetal adiposity.
LONG-TERM CHILD HEALTH OUTCOMES
Emily Oken presented an overview of the literature on longer term (>1 year) health outcomes for children based on maternal weight and weight gain during pregnancy. Child weight patterns (based on BMI) are the primary outcome of interest in this literature. Limited information is available about fat and lean body proportions in offspring or about the central distribution of body fat, which is predictive of disease risk. Just a handful of studies have looked at cardiovascular risk factors, including blood pressure, lipids, glucose intolerance, and insulin resistance, as well as type II diabetes and cardiovascular disease. Finally although higher weight at birth has been linked with increased risk for cancer, such as breast cancer in particular, and lower weight at birth is associated with increased risk for osteoporosis and schizophrenia, the existing literature does not link these conditions to maternal weight. Therefore, a number of intermediate steps occur in the pathway linking maternal weight and gestational weight gain to child health outcomes.
This section reviews what is known about the effect of maternal weight and weight gain during pregnancy on child health outcomes.
Early Life Factors and Other Considerations
Several factors may influence the association of maternal weight and weight gain during pregnancy with long-term child health outcomes. These include glucose tolerance during pregnancy, birth weight, fetal growth, smoking during pregnancy, infant feeding practices, and maternal and paternal BMI.
Higher maternal weight is a risk factor for gestational diabetes or glucose tolerance during pregnancy, as noted in Chapter 4. A fairly robust body of evidence now suggests that children born to mothers who had diabetes during pregnancy are themselves at higher risk for overweight, gestational diabetes, and type II diabetes. These risks appear to be independent of the maternal prepregnancy weight.
As discussed in the previous section, many studies have demonstrated a direct association between birth weight and higher BMI in childhood and adulthood. There is also an extensive body of evidence suggesting an association between lower fetal growth and increased risk for obesity, glucose intolerance, and cardiovascular disease risk, even after adjusting for offspring BMI. Finally, more rapid growth in the first months, and even perhaps the first days of postnatal life, are associated with increased risk for child overweight.
Maternal and paternal BMI reflect the shared genes in the environment between parents and child, so adjusting for parental size is important for isolating the role of the prenatal environment. Paternal weight data have generally not been as readily available as maternal weight data. When present, paternal weight tends to be less strongly linked with child weight, which may suggest a persistent influence of the intrauterine environment. Oken and others have shown that smoking during pregnancy is associated with increased risk for child overweight.
Mothers who gain more weight and who are more overweight may be less likely to breastfeed, and breastfeeding protects against later child obesity. Overweight mothers may also be more likely to introduce solid foods earlier, and early introduction of solids is also a risk factor for later development of obesity.
The pathways linking maternal weight and weight gain with child outcomes are somewhat complex (Figure 5-1). It is important to consider the extent to which maternal weight and weight gain during pregnancy act independent of these pathways, because they may help to clarify the population impact of greater pregnancy obesity and higher gestational weight gains.
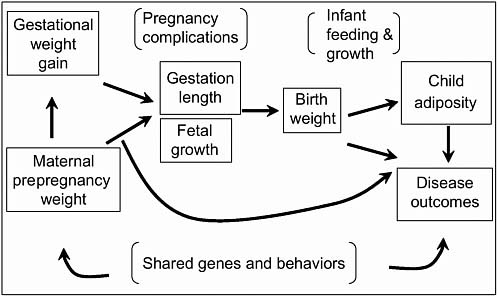
FIGURE 5-1 Relationship of maternal weight and pregnancy weight gain to child outcomes.
SOURCE: Oken (2006).
Maternal Weight
In a study of low-income children in the Special Supplemental Food Program for Women, Infants, and Children (WIC), Whitaker (2004) found a linear association between maternal BMI (in the first trimester) and the odds of overweight in 2-, 3-, and 4-year-old children. A second study in Denmark found a direct association between maternal BMI and offspring BMI and also reported the strengthening of this association with increasing offspring age into adulthood (Schack-Nielsen et al., 2005).
A study of men and women in China suggests that, after adjustment for offspring BMI, lower maternal BMI was associated with increased glucose, increased insulin, and increased low-density lipoprotein (bad cholesterol) levels in the offspring (Mi et al., 2000). It is important to note that, in this study, maternal BMI was measured at 15 weeks gestation and is low compared with current U.S. BMI levels.
A study of about 3,000 adult men with data from birth in Finland found an indirect association between size at birth and cardiovascular disease mortality (Forsen et al., 1997). Offspring of mothers who had higher BMI at the end of their pregnancies were at elevated risk for cardiovascular disease. This association was limited to mothers who were below average height in the population. The authors conclude that since height is an
indicator of early life nutrition, a mother who was undernourished in early life but then became overnourished in later life confers elevated cardiovascular disease risk for her offspring. However, these risks were not adjusted for offspring BMI, so it is not clear whether they were mediated through higher obesity in the offspring or an independent association of maternal BMI on child cardiovascular disease risk.
In summary, a linear relationship exists between maternal weight and child weight. This relationship is in part mediated through fetal growth, may become stronger with increasing offspring age, and certainly reflects shared genes and behaviors between mother and child but also perhaps demonstrates a persistent effect of the fetal intrauterine environment. There is limited information about child health outcomes other than weight in relation to maternal weight.
Gestational Weight Gain
Very few published studies examine gestational weight gain and child outcomes. Studies that have included gestational weight gain use it as a covariate but not as a primary predictor, so it is not clear that interactions and associations were completely explored.
Fisch et al. (1975) suggest an association between gestational weight gain and child weight. Data from the Collaborative Perinatal Project showed that mothers of babies who were above the 95th percentile (weight for height) at birth had a higher mean gestational weight gain than mothers of babies who were below the 5th percentile (weight for height) at birth. These associations persisted into childhood, although they were less strong and no longer statistically significant (not adjusted for maternal weight, birth weight, or any other factors). Another study, using data from a different subpopulation of the Collaborative Perinatal Project, reported that the association of gestational weight gain with child weight was null (Stettler et al., 2000).
The Danish cohort study discussed earlier reported a linear association of maternal gestational weight gain with offspring BMI that appears to be fairly consistent across ages (Schack-Nielsen et al., 2005). It is also associated with child gender, fetal growth, maternal age, sociodemographics and smoking. The authors report that they saw no interaction between maternal prepregnancy BMI and gestational weight gain. In a larger study of Israelis who were born in the 1970s, Seidman et al. (1996) indicated higher gestational weight gain was associated with an elevated odds ratio of offspring overweight. A study of over 1,000 children in Italy born in the 1970s and early 1980s found no association between gestational weight gain and child weight, although gestational weight gain was assessed retrospectively and by recall only (Maffeis et al., 1994). This is the only study to show an
inverse association of gestational weight gain with BMI and percentage body fat.
A more recent study by Whitaker (2004) with a large population of children enrolled in the WIC program shows no obvious linear association of gestational weight gain with child overweight risk, and in fact suggests a J shape with a higher risk in the first quartile, lower in the second, and then perhaps increasing. Gestational weight gain here was obtained from birth certificates and was reported as net weight gain, not total weight gain, and was adjusted for maternal but not paternal BMI, smoking, sociodemographics, and fetal growth. The authors reported no interaction between BMI and gestational weight gain.
Oken described initial data analyzed from Project Viva, a long-term pregnancy and child cohort study with information on over 1,000 children, followed to age 3. In the study, maternal and child weight are very similar to current national estimates. These data suggest a linear association between maternal gestational weight gain and the risk of child overweight at age 3, child BMI score, and the sum of subscapular and triceps skinfolds. There was no association between gestational weight gain and the ratio of subscapular to triceps skinfolds, which is a measure of central obesity. Increased gestational weight gain was also associated with increase of systolic blood pressure, adjusted for maternal prepregnancy BMI, smoking, race/ethnicity, income, marital status, glucose tolerance during pregnancy, paternal BMI, child gender, gestation length, and breastfeeding duration. Final adjustment estimates remain statistically meaningful, suggesting that gestational weight gain has an independent effect on child overweight and child BMI at age 3. Looking at maternal weight gain (using IOM categories), compared with mothers who gained inadequate weight, mothers who gained both adequate and excessive weight had similar elevations in the child’s BMI score.
Finally, Oken described one study that suggests the relationship between maternal gestational weight gain and child overweight risk seems to vary within different categories of maternal prepregnancy weight; it assumes a J shape among underweight mothers, a U shape among mothers with gestational weight gain less than 45 lbs., and a linear pattern at the highest weight gains (personal communication, A.J. Sharma, Centers for Disease Control and Prevention). The lowest risk of child overweight was seen in mothers who were underweight before pregnancy and gained less than 45 lbs. and the highest absolute risk of overweight was seen in mothers who were obese before pregnancy and gained 30 lbs. or more.
Research Needs
Potential areas for future research include studies to examine cohorts using the recommended IOM prepregnancy BMI categories and gestational
weight gain, measures of body fat distribution and disease risk as well as just BMI, and longer follow-up of infants born in this current era of obesity. One way of overcoming the limitations of the observational studies would be to follow children whose mothers were enrolled in trials targeting gestational weight gain, to see whether those children whose mothers receive interventions to reduce their gestational weight gain have lower attained weight. Nearly all of the data presented here are from developed populations, with no real presence of nutritionally compromised populations and few minorities. Finally, there is little direct information about mechanisms by which weight and weight gain might influence offspring weight, although it is possible to extrapolate from studies of gestational diabetes and also from animal studies. Some animal models that might be informative include experimental induction of gestational diabetes in rats, injection of insulin or glucocorticoids into the pregnant mother to examine effects on offspring, and neonatal overfeeding of rats.
Summary of Long-Term Infant and Child Health Outcomes
In summary, Oken’s review of the literature suggests a direct association of maternal weight and gestational weight gain with offspring overweight, but some exceptions to these findings deserve notice. The associations seem to be in part mediated through fetal growth. The shape of the association varies among women with differing profiles of prepregnancy weight and gestational weight gain, so it is not entirely clear, and not many studies have sufficient data to look at the broad range of gestational weight gain. The interaction between maternal BMI and gestational weight gain is not consistent across published studies. However, many of these data come from previous generations with different prevalence of obesity and gestational weight gain; many were not adjusted for important potential co-variates and pathway variables, and all of them are observational studies.
SUMMARY
In summary, maternal BMI and gestational weight gain are higher than in the past. These trends are associated with the increasing prevalence of macrosomia (large-for-gestational-age) infants as well as decreasing rates of small-for-gestational-age infants. Low prepregnancy BMI and inadequate gestational weight gain, in combination, are associated with preterm birth, but maternal BMI is also independently related. A few studies describe the relationship of gestational weight gain and infant body composition. Maternal BMI and gestational weight gain have been shown to predict fat mass and percentage fat in newborns and maternal BMI and gestational weight gain can also predict infant lean body mass. It is unknown what body composition in the newborn predicts for the child weight composition in
the long term. It is also unknown if maternal fat gain, not just gestational weight gain, predicts child health outcomes.
In long-term child outcomes, the literature suggests that both BMI and gestational weight gain independently predict risk of overweight in children. There are limited data on obesity-related physiological or morbid outcomes, such as metabolic and cardiovascular risk factors.
Research is limited on body composition, which is difficult to measure in a clinical setting. In addition, the role of the placenta may give insight into the complex mechanism and relationship between maternal weight and child health outcomes. In studying complex interactions among maternal weight, gestational weight gain, and infant and child health outcomes, investigations need to use statistical models that build on conceptual theories. There is a compelling need to examine these relationships in a more diverse population.4
Finally, these findings need to be interpreted and referenced through the application of a theoretical model that can lend both coherence and additional research. A life-course approach to chronic diseases offers important promise. This intervention framework includes prenatal, postnatal, biological, environmental, and behavioral characteristics that may occur at many dynamic stages of the mother’s and her child’s lives. It also incorporates different causal models, including the critical (or sensitive period) model, as well as an accumulation of risk model. In addition, investigations need to consider the relative merits of a public health approach compared with individualized approaches in addressing weight gain and body composition before, during, and after pregnancy.
REFERENCES
Ananth, C.V., and Wen, S.W. 2002 Trends in fetal growth among singleton gestations in the United States and Canada, 1985 through 1998. Seminars in Perinatology 26(4):260–267.
Carmichael, S.L., and Abrams, B. 1997 A critical review of the relationship between gestational weight gain and preterm delivery. Obstetrics and Gynecology 89(5 Part 2):865–873.
Catalano, P., and Ehrenberg, H. 2006 The short- and long-term implications of maternal obesity on the mother and her offspring. BJOG: An International Journal of Obstetrics and Gynecology July 7: E-pub DOI: 10.1111/j.1471-0528.2006.00989.x.
Dietz, P.M., Callaghan, W.M., Cogswell, M.E., Morrow, B., Ferre, C., and Schieve, L.A. 2006 Combined effects of prepregnancy body mass index and weight gain during pregnancy on the risk of preterm delivery. Epidemiology 17:170–177.
Fisch, R.O., Bilek, M.K., and Ulstrom, R. 1975 Obesity and leanness at birth and their relationship to body habits in later childhood. Pediatrics 56(4):521–528.
Forsen, T., Eriksson, J.G., Tuomilehto, J., Teramo, K., Osmond, C., and Barker, D.J. 1997 Mother’s weight in pregnancy and coronary heart disease in a cohort of Finnish men: Follow up study. British Medical Journal 315(7112):837–840.
Institute of Medicine 1990 Nutrition During Pregnancy. Washington, DC: National Academy Press.
2007 Preterm Birth: Causes, Consequences, and Prevention. Washington, DC: The National Academies Press.
Maffeis, C., Micciolo, R., Must, A., Zaffanello, M., and Pinelli, L. 1994 Parental and perinatal factors associated with childhood obesity in north-east Italy. International Journal of Obesity Related Metabolic Disorders 18(5):301–305.
Mercer, B.M., Macpherson, C.A., Goldenberg, R.L., Goepfert, A.R., Haugel-De Mouzon, S., Varner, M.W., Iams, J.D., Meis, P.J., Moawad, A.H., Miodovnik, M., Caritis, S.N., Van Dorsten, J.P., Sorokin, Y., Thurnau, G.R., and Spong, C.Y. 2006 Are women with recurrent spontaneous preterm births different from those without such history? American Journal of Obstetrics and Gynecology 194(4):1176–1184.
Mi, J., Law, C., Zhang, K.L., Osmond, C., Stein, C., and Barker, D. 2000 Effects of infant birthweight and maternal body mass index in pregnancy on components of the insulin resistance syndrome in China. Annals of Internal Medicine 132(4):253–260.
Oken, E. 2006 Maternal Weight and Gestational Weight Gain as Predictors of Long-Term Offspring Growth and Health. Presentation at the Workshop on the Impact of Pregnancy Weight on Maternal and Child Health, May 30, Washington, DC.
Orskou, J., Kesmodel, U., Henriksen, T.B., and Secher, N.J. 2001 An increasing proportion of infants weight more than 4000 grams at birth. Acta Obstetrics and Gynecology Scandanavia 80(10):931–936.
Schack-Nielsen, L., Mortensen, E.L., and Sorensen, T.I.A. 2005 High maternal pregnancy weight gain is associated with an increased risk of obesity in childhood adulthood independent of maternal BMI. Pediatric Research 58(5): 1020.
Schieve, L.A., Cogswell, M.E., and Scanlon, K.S. 1999 Maternal weight gain and preterm delivery: Differential effects by body mass index. Epidemiology 10(2):141–147.
Schieve, L.A., Cogswell, M.E., Scanlon, K.S., Perry, G., Ferre, C., Blackmore-Prince, C., Yu, S.M., and Rosenberg, D. 2000 Prepregnancy body mass index and pregnancy weight gain: Associations with preterm delivery. The NMIHS Collaborative Study Group. Obstetrics and Gynecology 96(2):194–200.
Seidman, D.S., Laor, A., Shemer, J., Gale, R., and Stevensen, D.K. 1996 Excessive maternal weight gain during pregnancy and being overweight at 17 years of age. Pediatric Research 39:112A.
Sewell, M.F., Huston-Presley, L., Super, D.M., and Catalano, P. 2006 Increased neonatal fat mass, not lean body mass, is associated with maternal obesity. American Journal of Obstetrics and Gynecology 195(4):1100–1103.
Sparks, J.W. 1984 Human intrauterine growth and nutrient accretion. Seminars in Perinatology 8(2): 74–93.
Stettler, N., Tershakovec, A.M., Zemel, B.S., Leonard, M.B., Boston, R.C., Katz, S.H., and Stallings, V.A. 2000 Early risk factors for increased adiposity: A cohort study of African American subjects followed from birth to young adulthood. American Journal of Clinical Nutrition 72(2):378–383.
Surkan, P.J., Hsieh, C.C., Johansson, A.L., Dickman, P.W., and Cnattingius, S. 2004 Reasons for increasing trends in large for gestational age births. Obstetrics and Gynecology 104(4):720–726.
Whitaker, R.C. 2004 Predicting preschooler obesity at birth: The role of maternal obesity in early pregnancy. Pediatrics 114(1):e29–e36.















