2
The Present State of Knowledge About Mars and Possible Life
ENVIRONMENTAL REQUIREMENTS FOR LIFE
Life requires energy sources, the nutrients necessary for building structures and synthesizing catalysts, and access to environments in which biosynthesis and maintenance of biostructures are possible. Potential energy sources for life on Mars include the direct and indirect utilization of solar radiation, lightning, ionizing radiation, geothermal heat, and various redox couples involving carbon or inorganic compounds. As discussed by Price and Sowers,1 the energy requirements for survival are likely a million times less than the energy required for growth. The concept of survival energy, that which is required for repair of macromolecules, maintenance of ion gradients, and so on,2 implies that life could survive, although not multiply, in a much wider range of environments than was formerly thought possible.
The availability of nutrient elements may also impose limits to life. As mentioned in Chapter 1, it is reasonable to assume that martian life, if any, would be based on carbon (C), hydrogen (H), nitrogen (N), oxygen (O), phosphorus (P), and sulfur (S). Mars was built from the same carbonaceous chondritic material as Earth. The same elements would accordingly be available to be utilized by life on Mars as on Earth unless processes on Mars caused their depletion. C, H, N, O, P, and S have all been detected on Mars. But the amount of nitrogen is a potential problem.3 The martian atmosphere contains only 160 microbars (mbar) of N2, and nitrates have not been detected in the soils. Much of the original nitrogen inventory could have been lost by impact erosion of the atmosphere during heavy bombardment, subsequent sputtering by the solar wind, or photochemical processes. Access to organic carbon is another potential problem. While CO2 is the dominant species in the atmosphere, reduced carbon compounds may be rare at the surface, despite continual delivery of organic compounds to the martian surface in meteorites and interplanetary dust particles. Nevertheless, reduced carbon compounds have been detected in the martian meteorite ALH 84001. There is little reason to assume that the availability of other elements on Mars is significantly different from their availability on Earth.
Since no martian life has been detected, it is not possible to determine what its environmental requirements are. However, current understanding of the limits of terran life continue to expand as living forms are found in
ever greater extremes of temperature, pressure, pH, salinity, and so forth.4 One environmental factor that is almost universally accepted as necessary for life is at least episodic access to liquid water. This observation has implications for temperature but does not necessarily imply temperatures between 273 K and 373 K. Although there is theoretical evidence that metabolism can continue at temperatures at or below 233 K, there is no evidence of active cells below 253 K and no direct observation of cell replication below 248 K.5,6 At the high-temperature end, life has been cultured in the laboratory at 394 K,7 although evidence from deep-sea hydrothermal vents indicates that the upper temperature limit for life may be much higher than this.8 Apart from temperature, two other parameters critical to the survival of terran life are worthy of note—water activity and radiation resistance. The availability of liquid water to an organism is critical for its survival. Many organisms are resistant to desiccation; however, currently no organisms are known to survive at water activities lower than aW = 0.61.9 Therefore, temperature and water activity have recently been used to constrain regions on Mars associated with special planetary protection considerations.10
DOES MARS MEET HABITABILITY REQUIREMENTS?
The following discussion focuses on the availability of water. The committee assumes that solar energy, geothermal energy, and chemical energy, as well as nutrients, are available on Mars. Sources of energy and nutrients are present on Mars as they would be on any geologically active planet, although questions about the availability of nitrogen have not been resolved.11 A major unknown is where and when liquid water was available to enable the assets present to be used for a possible origin or maintenance of life.
Present Environmental Conditions
Conditions on the surface of Mars today are very inhospitable for life, but geological evidence suggests that conditions were more hospitable in the past, particularly the distant past. Liquid water is believed to be essential for life. With mean annual surface temperatures close to 215 K at the equator and 160 K at the poles, the ground is frozen on average to a depth of several kilometers to form a thick cryosphere. Any water present in this zone would be frozen. The cryosphere might be thinner locally in areas of anomalously high heat flow, but no such areas have been identified. The atmosphere is thin, with an average surface pressure of 5.6 mbar, and composed largely of CO2. Because the atmosphere is so thin, the Sun’s ultraviolet radiation passes almost unattenuated through it to the surface. Surface temperatures fluctuate widely during the day. On a clear summer day they may exceed 273 K close to noon. However, the fluctuations damp out rapidly at depth to converge on the average daily temperature, which is everywhere well below freezing, so that temperatures above freezing are restricted to the upper few centimeters. The ground is permanently frozen down to a kilometer or so below these depths.
Under present conditions, and probably under conditions that have prevailed for the last few billion years, weathering rates have been extremely low. Rocks in the Gusev crater have a millimeter-thick, oxidized rind, rich in volatiles such as sulfur, chlorine, and bromine.12,13 Soils have highly variable volatile contents and may contain an oxidizing agent. The fraction of organics in the soil is unknown. Although it was anticipated that the Viking gas chromatograph-mass spectrometer would detect some complex organic compounds, none were detected at the parts per billion level, and the Mars Pathfinder APXS measurements of soil could not detect carbon. The nitrogen content of the soils is also unknown.
Both poles have a residual water ice cap that is exposed when the CO2 cap recedes in the summer (see Figures 3.1 and 6.1), although in the south only small areas are exposed even at midsummer.14 Because of the cold polar temperatures, only minute amounts of water vapor are present in the atmosphere.15 Observations of seasonal frost and water fog in some areas on Mars demonstrate that the water content of the atmosphere varies both spatially and seasonally. However, if all the water vapor were precipitated out, it would form a global layer only about 10 μm thick. Abundant ground ice, however, may be present and available to interact with the atmosphere, and to enhance its water content should conditions change.16 Under present conditions, at depths greater than a few tens of centimeters below the surface at latitudes in excess of 50° north and south, water ice is stable. Consistent with these conditions, large fractions of ice have been detected just below the surface at these latitudes by orbital
gamma-ray and neutron spectroscopy.17 At lower latitudes ground ice is unstable at all depths because temperatures exceed the 200 K frost-point temperature. The cause of the several percent water detected at these low latitudes is still being debated. It could be mineralogically bound water or ice inherited from an earlier era when stability relationships were different.
Parts of the surface are at elevations where the atmospheric pressure exceeds 6.1 mbar, the pressure at the triple point of water. At most locations, heating of ice-containing soils or surface frosts would result in sublimation without the intervening liquid phase, but where the pressure is in excess of the triple point, liquid could form transiently. Because of the low diurnal mean temperature, any such liquid would be very short lived. It would rapidly freeze and sublimate.
Although the present-day average climate is not conducive to the occurrence of liquid water, the possibility exists that liquid water can occur at the surface as a transient phase. Gullies appear to have released water to the surface, for example, and recent observations (Figure 2.2) suggest that this is happening in the present epoch (i.e., within the last 5 years or so).18 Similarly, transient melting of snow also can occur under very specific conditions.19 While the ramifications of transient liquid water are very different from those for a steady-state occurrence of liquid water, both have potential implications for possible life.
In the recent geological past, stability relationships may have been somewhat different. Mars undergoes large changes in its obliquity (i.e., the tilt of its polar axis).20 At present the obliquity ranges from 23 to 27° about a mean of 25°, but during the last 10 million years obliquities have been at least as high as 46°. At higher obliquities, the water content of the atmosphere is likely higher, ground ice is stable closer to the equator, and surface ice may be transferred from the poles to lower latitudes. In addition, during the summer at high latitudes, pole-facing slopes are continually illuminated by the Sun. One possibility for the formation of young gullies (see Figures 2.7 and 8.1) on steep, pole-facing slopes is that they form during periods of high obliquity as a result of liquid water produced during the summer by melting of snow that accumulated on the slopes during the long cold winter.21 High obliquities may also be implicated in the formation of some of the younger valley networks.
On present-day Mars, the subsurface may be more hospitable to life than the cold, oxidizing surface with its high ultraviolet (UV)-radiation fluxes. As indicated above, the cryosphere is on average several kilometers thick, and liquid water is unlikely within kilometers of the surface. However, the young crystallization ages of most martian meteorites22 and the apparent youthfulness of some volcanic features suggest that Mars today is volcanically active, at least intermittently. Heat flow under volcanic regions such as Tharsis may be significantly larger than the average, and the cryosphere correspondingly thinner. Moreover, given the presence of extensive ground ice, hydrothermal activity is likely in volcanically active areas, although none has been detected, and such activity (or even the background geothermal heat flux) could drive water to the surface.
In summary, present conditions at the surface of Mars are inhospitable to life, mainly because of the high UV flux, the presence of oxidants, and the scarcity of organic compounds, and because the low temperatures inhibit the presence of liquid water. However, liquid water may exist near the surface transiently in anomalous situations, ground water may be present at shallow depths in areas of anomalously high heat flow, and hydrothermal systems may be present in volcanic regions. Furthermore, at depths below a couple of kilometers, temperatures will be warm enough to allow liquid water in the pore space in rock, such that a deep-subsurface biosphere is possible, provided that appropriate nutrients are accessible and water can circulate.
Past Environmental Conditions
Noachian
Conditions in the geological past, particularly the distant past, were likely, at least at times, to have been very different from present-day conditions. The best evidence for different conditions is for the Noachian, the period of heavy meteorite bombardment that ended around 3.8 billion years ago (Box 2.1). Most surfaces that date from this era are heavily dissected by networks of valleys a few kilometers wide but up to a few thousand kilometers long. Relatively high drainage densities suggest surface runoff that would require either rainfall or melting snow, which in turn implies significantly warmer and wetter conditions than those that prevail today (Figure 2.3). The

FIGURE 2.2 A gully (see arrow) in the wall of an unnamed crater (A) in Terra Sirenum (36.6° S, 161.8° W) provides tantalizing evidence that water might have flowed on the martian surface in recent times. Close-up images (B) clearly indicate that sometime between December 2001 (left) and August/September 2005 (right) a new, light-toned deposit filled the gully. The thinness of the deposit and its multilobed appearance at its downhill end (C) suggest that material of some sort flowed in a fluid-like manner down the crater wall and then splayed out when it reached the relatively flat crater floor. These characteristics can be interpreted as suggesting that a mixture of sediment and a fluid with the properties of liquid water emerged from the crater wall and ran down through a preexisting gully channel within the last 5 years. These images were taken with the Mars Orbiter Camera on the Mars Global Surveyor spacecraft and are courtesy of NASA/JPL/Malin Space Science Systems.
belief that Mars was warmer in the Noachian than it is today is also supported by the finding of evaporites at the Mars Exploration Rover landing site on Meridiani Planum and elsewhere, by evidence for fluctuation of the water table at Meridiani, by detection of clay minerals from orbit in Noachian terrains, by the presence of hydrothermally altered rocks in the Columbia Hills at the Mars Exploration Rover landing site in Gusev crater, and by surface erosion rates that were 4 to 5 orders of magnitude higher than they were subsequently.23 These observations, in combination, suggest an Earth-like, active hydrological cycle with large lakes or oceans that acted as evaporative sources, sinks, and base levels for erosion. Given the likely large inventory of water at the surface, if Mars did have periods with an active hydrological cycle, oceans could have been present in lows such as Hellas and the northern plains (see Figure 7.1). However, although shorelines have been tentatively identified around both these lows, the observational evidence for the postulated oceans is weak.24 In contrast, there is abundant evidence, such as deltas, of lakes in local lows within the uplands (Figure 2.4).
Although dissection of Noachian terrain is widespread, several morphological characteristics of the drainage basins suggest that, compared with those of Earth, the drainage system is immature.25 The morphology of the Noachian terrains is dominated by primary terrain-building processes, such as impacts, volcanism, and deformation, rather than by fluvial processes. Even for the Noachian, for which researchers have the best evidence of abundant liquid water at the surface, the conditions necessary for fluvial erosion may have been achieved only episodically.
The Noachian was also characterized by high rates of volcanism and high rates of impact. The formation of large impact basins such as Hellas and Argyre would have had devastating effects on any emerging life.26 Large fractions of any oceans present would have boiled away, and the planet would have been enveloped in hot-rock vapor that would have condensed and rained hot rock back onto the surface. Such global catastrophes would, however, have been separated by millions of years of relative quiet even in the era of heavy bombardment.
Although Mars appears to have had benign periods during the Noachian, when water flowed freely across the surface, the cause of the warmer conditions remains unknown. A thick CO2-H2O atmosphere may be incapable of warming the surface to above freezing without additional forms of heating such as infrared absorption by dust in the atmosphere.27 Moreover, thick carbonate deposits that would contain CO2 from a thick, early atmosphere have not been found despite intensive searches using orbital spectroscopy, although the CO2 may have been lost to space instead. Another possibility is that large impacts or large volcanic eruptions episodically altered surface conditions temporarily,28 thereby briefly stabilizing liquid water at the surface. At the end of the Noachian, the rate of formation of valley networks declined rapidly, although not to zero, erosion rates fell precipitously, and clay minerals appear to have stopped forming.29 There can be little doubt that a major change in surface conditions occurred at the end of the Noachian.
Post-Noachian
The post-Noachian period, which encompasses roughly the last 3.8 billion years, is characterized by very low rates of weathering and erosion. The most characteristic fluvial feature of the post-Noachian era is the outflow channel, formed by large floods, rather than the valley networks that characterize the preceding era (Figure 2.5). Nevertheless, young valley networks are found in places, such as on young volcanoes, indicating that conditions necessary for slow erosion by running water were occasionally and locally met.
Large flood channels are readily recognizable by the scoured floors, streamlined walls, and tear-drop-shaped islands.30 The largest are around the Chryse basin, into which several enormous channels converge. Peak discharges may have ranged as high as 108 m3s–1, as compared with 104 m3s–1 for the Mississippi River. The Chryse channels emerge from local rubble-filled depressions, or from the Valles Marineris. Elsewhere channels may start at faults. The Chryse channels are mostly Hesperian in age (some 3.0 to 3.7 billion years ago), but crater dating of some channels elsewhere suggests that they can be as young as a few tens of millions of years.31 If so, then floods could form today. The flood channels appear to have formed by eruptions of groundwater from below a thick cryosphere, or in the case of those adjacent to Valles Marineris, by the drainage of large lakes. Eruptions may have been triggered by a variety of causes such as large impacts, tectonic forces, or dike injection.
The best morphological evidence for volcano-ice interactions, and hence hydrothermal systems, is in the
|
BOX 2.1 Martian Geological Eras and the Age of Surface Features The superposition of one surface feature (e.g., an impact crater) upon another (e.g., a lava flow) enables their relative ages to be determined. Thus, the impact crater is younger than the lava, which, in turn, is younger than the underlying surface of the planet. When such stratagraphic relationships are examined planet-wide, the history of Mars can be divided into three major geological eras, the Noachian, the Hesperian, and the Amazonian (Figure 2.1.1).1 
FIGURE 2.1.1 Major events in the geological histories of Earth and Mars over the last 4.5 billion years. The timing of the boundaries between Mars’s three major named geological eras is highly uncertain because of the absence of an absolute calibration of the ages of martian surface features. Moreover, the geological record of the earliest events in martian history, those of the so-called pre-Noachian era, has been largely erased by subsequent events, including the heavy bombardment that took place during the Noachian era. Diagram courtesy of Michael H. Carr, U.S. Geological Survey. |
Elysium region where several large fluvial channels emerge from graben radial to the volcanos there.32 One possibility is that release of groundwater was caused by propagation of dikes radial to the volcanos through the local cryosphere and hydrosphere. Large bodies of water must have been left behind after the floods.33 A thick cryosphere was probably present when the floods formed. Any terminal lakes or seas would thus have frozen, and ultimately the ice would have sublimated away or been buried. Observational evidence for any such oceans is sparse, although there is good evidence in the northern plains for burial of pre-flood craters and ridges by sediments,34,35 and several features suggest the former presence of ice in the low areas at the ends of the channels (Figure 2.6).36 It has been suggested that formation of a large flood would have temporarily changed global climates by injecting large amounts of H2O and CO2 into the atmosphere, but failure to detect carbonates is troubling. However, carbonate could be distributed uniformly throughout a crust, emplaced by circulation of water, and could hold a several-bar CO2 atmosphere without being detectable spectroscopically. Also, thermal-emission spectrometer results have
|
The oldest geological features recognizable on Mars belong to the Noachian era, named after Noachis Terra in the southern highlands. In fact, most of Mars’s rugged southern highlands are of Noachian age. This era was characterized by very high cratering rates and the formation of the major impact basins (e.g., Hellas and Argyre); major episodes of volcanic activity; and the formation of the oldest valley networks. Lying immediately atop Noachian geological units are features of the Hesperian era. Much of the northern lowlands and, in particular, the ridged plains are of Hesperian age. The youngest major surface features on Mars belong to the Amazonian era, so named because they are typified by the plains and volcanic materials of Amazonis Planitia. The surfaces of the prominent volcanos of Elysium and Tharsis Montes are of Amazonian age. The absolute ages of martian features and thus the time history of the planet’s evolution are currently uncertain. Converting the relative chronology implied by stratigraphy relationships requires that the absolute ages of key surface features be determined, and this will almost certainly require a sample-return mission. In the meantime, observations of the density of craters in a region can be used as a means of estimating that region’s absolute age.2 In other words, if a surface has a greater density of craters than an adjacent region and the rate at which impacts have occurred over time is known, then the ages of the surfaces can be estimated. Unfortunately, this technique is dependent on imperfect models of the cratering rate at Mars through time, which are, in turn, extrapolated from the known absolute chronology of the Moon. Despite the very great uncertainties, particularly in the dating of the boundary between the Hesperian and the Amazonian, researchers estimate that the age limits of the major geological eras on Mars are as follows:
Features older than the Noachian are usually referred to by the informal designation of pre-Noachian. Virtually nothing is known about this important period of martian history because the geological record of this time has been erased by later events. Unfortunately, this lost era includes the time in martian history when conditions might have most closely resembled those on Earth. By the end of the Noachian era, Mars was firmly established on a global evolutionary track that was significantly different from that followed by Earth over the subsequent 3.7 billion years. |
detected carbonates in the dust, and the martian meteorite ALH 84001 has several percent carbonates by weight, indicating that carbonates are present although the total quantity is unknown.
The martian canyons (e.g., Valles Marineris) are among the least understood features of the planet. Their relevance to liquid water and biology is that they may have once contained large lakes that ultimately drained catastrophically to the east to form outflow channels that connect to the canyons.37 They also provide access via spectroscopy to the deep subsurface, where liquid water might have been present. Several large channels also start in box canyons to the north of the main canyons, indicating that liquid water was present locally at elevations well above the floor of the main canyons. The central and eastern sections of the canyons contain thick stacks of layered sediments, rich in sulfates, which could have been deposited subaqueously. However, even if the canyons did once contain lakes, as appears likely, the source of the sediments, their mode of deposition, and the lifetime of the lakes all remain undetermined.
Although the rate of valley formation tailed off at the end of the Noachian, valleys continued to form at a low rate.38 Some of the most prominent valleys such as Nirgal Vallis and Nanedi Vallis are Hesperian in age, but both these valleys have characteristics that suggest that they formed mainly by groundwater sapping rather than by surface runoff as is the case for most of the Noachian valleys (Figure 2.7).39 Nevertheless, post-Noachian valley networks with runoff characteristics are found, as adjacent to Echus Chasma. In addition, several post-Noachian volcanos have surfaces that are highly dissected. In fact some of the most dissected surfaces anywhere are on volcanos. Several suggestions have been made to explain the young valleys on volcanos: that they formed by nuées ardentes or lava, that they formed during temporary warm periods caused by floods or large impacts, that they formed as a result of local conditions associated with volcanic eruptions, or that they resulted from the melting of ice deposited on the volcanos during periods of high obliquity, or after large floods. Whatever the cause, the presence of the valleys strongly supports the occasional temporary availability of liquid water on the volcano surfaces (Figure 2.8).
In summary, the present-day surface of Mars, with its cold temperatures, high ultraviolet flux, oxidizing conditions, and scarcity of liquid water and organics, is inhospitable to life as we know it. If life is present today, it likely is below the surface, protected from the harsh surface environment, or perhaps in exceedingly rare, localized environments driven by recent volcanic activity. The surface has experienced more benign conditions in the past, particularly the distant past. During the Noachian period, which ended around 3.7 billion years ago, liquid water was abundant at the surface, lakes were common, oceans may have been present, and the planet, like Earth, experienced high weathering rates with the production of clay minerals, and high rates of erosion and deposition, all consistent with warm, wet, habitable conditions.
At the end of the Noachian, conditions changed. Weathering and erosion rates declined rapidly to very low rates, which resulted in dominantly cold surface conditions and development of a thick cryosphere. Large floods episodically flowed across the surface leaving behind temporary lakes or seas, which could have proved temporary refuges. In addition, the planet intermittently experienced high obliquities that may have allowed accumulation of ice and snow at low latitudes, which on melting by sunlight or volcanic heat may have provided moist conditions in local areas. In all epochs, the combination of volcanism and water-rich conditions must have inevitably led to hydrothermal systems in which life could have thrived. Finally, accompanying these changes in physical processes were chemical changes.40 Weathering in the Noachian (>3.7 billion years ago) produced clay minerals, which have not been detected in the younger (3.0 billion to 3.7 billion years old) Hesperian rocks. Instead, many Hesperian deposits are rich in sulfates, many of which may have formed in highly acidic waters. Alteration of the younger (<3.0 billion years ago) Amazonian rocks is mainly by oxidation.
LESSONS LEARNED FROM PRIOR INVESTIGATIONS ABOUT MARS AND POSSIBLE LIFE
There have been two prior detailed investigations into possible martian life. The lessons learned from these investigations have an important bearing on the search for life on another planet. In addition, recent measurements of possible methane in the martian atmosphere are important for the same reason—their potential relevance to searching for martian life.
The Viking Mission
Life detection was one of the major goals of the two Viking spacecraft that successfully landed on the martian surface in 1976. Each spacecraft carried three life-detection experiments designed to detect metabolism and, in addition, a gas chromatograph-mass spectrometer (GC-MS) to detect and identify organic compounds.41 Overall, the results from the experiments were negative. The GC-MS detected no organic matter, and the results from the biology experiment all have plausible abiotic explanations.42,43
The GC-MS was, in principle, capable of detecting most organic compounds, except for highly polymerized, kerogen-like matter. Sensitivities were at the parts per billion level for compounds containing three or more carbon atoms and at the parts per million range for compounds containing one or two carbon atoms. Prior to the landings, it was thought that the soils would have detectable levels of organics from meteorite infall alone, and that photo-

FIGURE 2.4 Delta in the Crater Holden at 27°S, 326°E. A stream has cut through the south rim of the crater, just visible at the bottom of the picture, and deposited its sediment load to form a fan within the crater. The branching ridges on the delta surface are former water courses left higher than their surroundings because of greater resistance to erosion. Image from the Thermal Emission Imaging System on the Mars Odyssey spacecraft courtesy of NASA/JPL/Arizona State University.

FIGURE 2.5 Mangala Vallis. The source of the outflow channel Mangala Vallis at 18°S, 210°E. The channel starts at a 7-km gap in a graben wall (bottom center) and then extends hundreds of kilometers northward. Faulting appears to have triggered massive release of groundwater. Image from the Thermal Emission Imaging System on the Mars Odyssey spacecraft courtesy of NASA/JPL/Arizona State University.

FIGURE 2.6 Ice-rich debris flows in the fretted terrain at 40°N, 25°E. At 30° to 50° latitudes in both hemispheres, material shed from slopes commonly shows indications of having flowed like glaciers. Here, what is probably an ice-rich debris flow has been deflected through a gap in an obstructing ridge. Image from the Thermal Emission Imaging System on the Mars Odyssey spacecraft courtesy of NASA/JPL/Arizona State University.

FIGURE 2.7 Gullies on the wall of Nirgal Vallis at 30°S, 321°E. Their origin is still being debated, but they formed recently and liquid water may have been involved in their formation. Image from the Mars Orbiter Camera on the Mars Global Surveyor spacecraft courtesy of NASA/JPL/ Malin Space Science Systems.
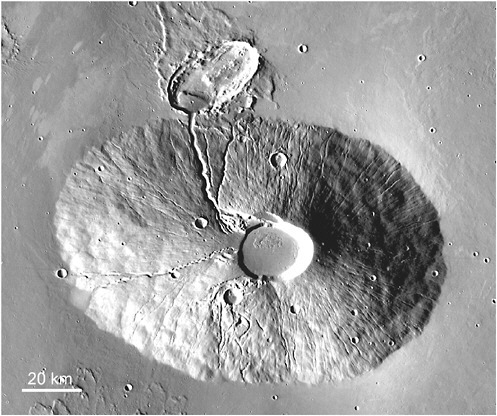
FIGURE 2.8 Ceraunius Tholus. Some volcanos, such as this, are densely dissected, possibly the result of melting of surface or subsurface ice by volcanic heat. Formation of the valleys may have been accompanied by hydrothermal activity. Image from the Thermal Emission Imaging System on the Mars Odyssey spacecraft courtesy of NASA/JPL/Arizona State University.
chemical fixation of CO into organics would be an additional contributor. Failure to detect organics implied that they are destroyed at the martian surface. One possible cause of the absence of organics is that they are destroyed by UV-stimulated reactions with metal oxides in the soils.44 Another possibility is that they are destroyed by reactions with labile oxidants, since the biology experiments showed that in the presence of water, the soil releases tens to hundreds of nanomoles of O2 per cubic centimeter.45 The oxidants may be produced by photodissociation of water in the atmosphere. Another possibility is that organics were present but that oxidants in the soil destroyed them during sample processing by the GC-MS.46
In the pyrolytic-release experiment, isotopically labeled CO and CO2 were added to the soil.47 After a suitable incubation time, the gases were flushed out, and the soil was heated to see if any of the labeled carbon had been incorporated into less volatile species. Small amounts of carbon were fixed into organics, which the experimenters
attributed to inorganic synthesis catalyzed by the martian soil. In a second gas-exchange experiment, the soil was humidified and nutrients added.48 The resulting rapid release of oxygen was attributed to the presence of oxidants in the soil. In the third experiment, simple organic compounds labeled with radioactive tracers were added to the sample.49 This resulted in rapid release of labeled gas followed by slow release. Subsequent experiments show that addition of the nutrients to soil containing Fe2O3 and H2O2 simulated the Viking results. The consensus is that all the results from the biology experiments have plausible inorganic explanations. A few researchers, however, maintain a contrary view.50
In retrospect, the Viking mission could be criticized as reaching too far too soon. In the late 1960s when the mission was conceived, knowledge of conditions on the martian surface was rudimentary. Speculations on the prospects for life were based largely on telescopic observations. Little information was available on the surface conditions and on where best to land in order to look for life. It could also be argued that the experiments were narrowly designed to detect a limited spectrum of terran life. Despite the negative results from the life-detection investigations, the Viking mission returned invaluable information for future biological experiments, such as data on the oxidizing nature of the surface and the possible scarcity of organics. In hindsight, the Viking missions constitute a compelling argument in favor of the kind of systematic approach advocated in this report and in NASA’s 1995 report An Exobiological Strategy for Mars Exploration.51
The Search for Life on Early Earth and in the Martian Meteorite ALH 84001
The lack of a conclusive set of criteria for life detection and preservation has been illustrated recently by two debates: the search for the oldest evidence of life on Earth and the raging debate on the claims for life in ALH 84001. The scientific controversies over the former debate, that of the earliest evidence of life on Earth, have recently intensified but are still unresolved.52–60 The common denominator in both of these debates is the underlying difficulty, or inability, to demonstrate conclusively the biological origin of the respective evidence.
The Earliest Life on Earth
Various claims and counterclaims have been published in the scientific literature in recent years concerning the earliest evidence for life on Earth.61,62 The conflicting results of these studies illustrate the potential pitfalls in collecting and interpreting data from the ancient geological record.
Our planet’s earliest known microfossils have been ascribed to 3.5-billion-year-old cyanobacteria identified by Schopf in samples of the Apex chert of Western Australia.63 The morphological identification of these tiny dark clumps was always controversial, given that the range of bacterial morphologies at their simplest are ambiguous to interpret; bacteria have little morphology to begin with. Furthermore, the earliest evidence of biomarkers specific to cyanobacteria is 2.7 billion years ago, roughly consistent with molecular clock estimations of the emergence of cyanobacteria.64 Such organisms would have produced photosynthetic oxygen, which does not appear as a significant atmospheric component until much later.65,66
The identification of these “microfossils” has been called into question by Brasier at al.,67 based on reexamination of the chert sections of the original study. Many of the “microfossils”were observed to have branched morphologies inconsistent with filamentous bacteria. Schopf countered that the specimens are not branched, but folded by later deformations. In revisiting the collection site, Brasier et al. also determined that the Apex chert itself was not a sedimentary deposit, but instead was a vein formed by hydrothermal activity. Brasier et al. argued that the “microfossils” are merely bits of carbonaceous matter, unrelated to life, and squeezed out of forming quartz crystals and wrapped around them to resemble microfossils. To date no studies have satisfactorily determined the abiogenic or biogenic nature of the carbon forming the “pseudofossils.”
To support his original claim, Schopf teamed with other scientists utilizing laser-Raman spectra to determine that carbon is present within the “microfossils.”68 However, other experts in this technique have criticized this work,69 noting that there is nothing diagnostic in the spectra that indicates that the analyzed carbon-bearing clumps are the remains of organisms rather than abiotic organic matter.
The isotopic composition of carbon has been used as a biomarker, because photosynthetic organisms pref-
erentially incorporate the lighter isotopes. Tiny bits of carbon (now graphite) in a 3.85-billion-year-old gneiss in Greenland have been determined to be depleted in 13C.70,71 The authors of this study suggested that the host rock was a sedimentary, banded iron formation. They hypothesized that biogenic matter collected at the bottom of the ocean and was incorporated into sediments; later metamorphism transformed the organic matter into graphite, but its carbon isotopic composition was preserved. They interpreted this finding as strong evidence for life, some 400 million years earlier than previously thought. In mapping the outcrop from which the samples were collected, other scientists have found that it is not a banded iron formation, but instead represents a volcanic rock into which metamorphic fluids were injected.72 These fluids precipitated quartz to form the observed banding. This appears to be a highly unlikely site for the preservation of organisms. Moreover, it has now been shown that isotopically light hydrocarbons can be produced via several abiotic pathways.73
Although argument continues on both these controversies, most scientists appear to have sided with the skeptics. Several lessons can be drawn from these controversies about Earth’s oldest life. Morphology alone is not a sufficient criterion for the identification of simple life forms. Understanding of the geological context of the sample is of prime importance, because it provides information on the environment in which the putative organism lived or was preserved. And, finally, the interpretation of geochemical analyses of extremely small samples is fraught with difficulty and sometimes ambiguity. In short, it is not enough to show that some chemical property is consistent with life, but it must also be inconsistent with abiotic formation.
ALH 84001
In August 1996, McKay et al. announced that they had found evidence for life in the martian meteorite ALH 84001,74 a coarse-grained igneous rock (an orthopyroxenite) that crystallized 4.5 billion years ago. In support of their claim they listed the presence of the following:
-
Objects shaped like bacteria in scanning electron microscope imagery,
-
Polycyclic aromatic hydrocarbons (PAHs),
-
Disequilibrium mineral assemblages, and
-
Magnetic particles similar to magnetofossils produced by terran bacteria.
The claim of finding evidence for life in ALH 84001 is now viewed with skepticism.75 The suggestion that some of the objects viewed in the meteorite at high resolution could be bacteria was immediately doubted because of the extremely small size of the objects. The bacteria-like forms are 100 nm long and as little as 20 to 30 nm wide, much smaller than the generally accepted size for the smallest bacteria. Although there are scattered reports in the biomedical literature of “organisms” of comparable size,76 a panel convened by the National Research Council to assess how large an organism must be to enclose all the metabolic and genetic machinery that modern terran life requires concluded that independent free-living bacteria must be at least 200 to 300 nm in diameter.77 Nevertheless, more recent results based on the use of genomic techniques have identified the so-called Archaeal Richmond Mine Acidophilic Nanoorganism, whose size is at or below the lower limit quoted in the NRC report.78
If the objects described by McKay et al. are organisms, they must have had a much simpler chemistry than even the simplest modern terran organism. Bradley et al.79 alternatively suggested that the objects might be artifacts caused by the Au-Pd conductive coatings used by McKay et al. in the scanning electron microscope work. Similar objects have not been found when other coatings are used. There are reports of terran nanobacteria of comparable size to those in the martian meteorite,80 but these claims are not widely accepted. Shortly after the publication by McKay et al., Anders pointed out that production of PAHs does not require biology.81 Abiotic origins have been proposed for PAHs found in carbonaceous chondrites. Moreover, some phases (e.g., magnetite and clay minerals) associated with the carbonates in ALH 84001 are known to catalyze Fischer-Tropsch reactions, which convert simple carbon compounds such as CO into more complex ones, including PAHs. Anders also pointed out that the disequilibrium mineral assemblages described by McKay et al. could be produced abiotically. Finally, while the size, purity, morphology, and mineral structure of some of the magnetite grains in ALH 84001 do resemble those produced by terran magnetotactic bacteria, similar magnetites have been produced experimentally without the
intervention of biology.82 Indeed, some of the magnetites have growth textures suggesting that vapor condensation and shock vaporization of iron carbonate may account for these grains.
Lessons from Early Earth and ALH 84001
A review of the salient arguments for and against life both on early Earth and in martian meteorites reveals several positive points of note that must be addressed when looking for life on Mars:
-
The demand for multiple lines of evidence, especially chemistry in addition to any morphological data. An integrated strategy relying on multiple instruments and measurements by independent investigators is necessary but not sufficient to build confidence in any positive claim for life detection.
-
Both in situ and bulk inorganic and organic chemical analysis are needed to build up a fuller picture of the context of any interesting morphological features.
-
The need to distinguish among disparate pools of carbon compounds, such as distinguishing compounds having an abiotic origin from those with a biotic or terrestrial contaminant origin. The potential pools of carbon contributing to the total organic carbon of a martian sample are shown in Table 2.1.
-
The spatial and geological context and history from which the sample is taken are essential parameters in understanding the source of a potential biosignature.
The detailed mission objectives described by the Astrobiology Field Laboratory Science Steering Group provides one example of how these lessons have become an essential part of spacecraft mission planning.83
Methane
Mars has an atmosphere dominated by CO2 with minor components of N2, CO, O2, H2O, and Ar. Atmospheric investigations regarding the potential for life on Mars have focused on trace gases that could represent biomarkers. Examples include CH4, H2S, methyl mercaptans (CH3SH), and N2O. Recently three groups reported detection of methane with mixing ratios ranging up to 250 ppb,84–86 with two of the reports, those of Formisano et al. and Mumma et al., suggesting spatial variations; however, these measurements are at the limit of detectability. If found to be correct, these observations would indicate that methane is being continuously released into the atmosphere since its calculated photochemical lifetime (<300 years) necessitates a continuing resupply. Verification of these findings awaits improvement in both precision and detection limits.
While initial excitement focused on the significance of methane as a potential biomarker, what is known about the planet is consistent with a variety of possible origins for methane.87 Geological sources of methane include volcanic emissions and production of methane via low-temperature rock-water reactions as well as thermogenic gas from recycling of buried organic remnants from putative past life. The timing of methanogenesis is also complex, because spatial variation in methane in the atmosphere could result from ongoing methanogenesis as well as from intermittent release of gas stored over time in hydrates or subsurface fractures. Although an essential measurement, the isotopic composition of methane alone will not unambiguously distinguish between these biological and geological sources.88 Resolution of the origin of methane requires integration of multiple lines of evidence that include the following:
-
Verification of reported methane abundance in the atmosphere with higher sensitivity;
-
Constraints on the spatial and temporal variations of methane abundances on the planet’s surface; and
-
Integration of chemical and isotopic analyses with data from a range of possible co-generated species to weigh the overall probability of origin.
A finding that might suggest methane production via ongoing or past biological processes would have obvious significance. Equally, indications of a geological origin for methane due to volcanic venting or significant water-rock alteration would significantly impact further investigation and exploration since such sites would have
TABLE 2.1 Possible Sources of Organic Carbon That Have to Be Distinguished in Martian Samples
|
Source of Carbon |
Carbon Compounds, Examples, Comments |
|
Abiotic molecules from meteoritic/cometary influx |
Amino acids, purines and pyrimidines, polycyclic aromatic hydrocarbons, chain hydrocarbons, fatty acids, and sugars and sugar derivatives |
|
Prebiotic/abiotic molecules from synthesis reaction process on Mars |
Amino acids, purines and pyrimidines, polycyclic aromatic hydrocarbons, chain hydrocarbons, fatty acids, and sugars and sugar derivatives |
|
Organic contamination from Earth |
Condensation products derived from rocket exhaust, lubricants, plasticizers, atmospheric contaminants |
|
Contaminating organisms from Earth |
Whole cells, cell components (LPS, DNA, proteins, cytochromes). |
|
Terran organisms, from Earth |
Organisms not present on the craft measuring them, but previously transferred from Earth by either meteorite impact or contamination of previous spacecraft; target molecules could include individual genes, membrane constituents, specific enzymes, and co-enzymes that would be expected to be overexpressed or adapted in martian conditions |
|
Terran organisms, evolved on Mars |
Organisms that utilize terran biochemistries and have evolved on Mars; target molecules could include individual genes, membrane constituents, specific enzymes, and co-enzymes that would be expected to be overexpressed or adapted in martian conditions or organisms using metabolisms that would not be present on a spacecraft contaminant such as methanogens, or psychrophiles with endolithic survival mechanisms |
|
Non-terran organisms |
Organisms that utilize an array of molecules for information storage, information transfer, compartmentalization, and enzymatic activity that differ from those used by extant terran life; examples include the use of novel amino acids and nucleotides or the use of novel nitrogen utilization strategies |
|
Fossil biomarkers |
Established terrestrial fossil biomarkers such as hopanes, archaeal lipids, and steranes for detection of the diagenetic remains of terran life; characterization of potential breakdown products that can be reasonably extrapolated from the detection of molecules constituting an extant martian life form; detection of the diagenesis products of fossil martian organisms based on carbon compositions consistent with biological fractionation of a narrow range of abiotic precursors |
many of the characteristics potentially conducive for life. Either way, a confirmation of the mechanism for origin of methane in the Mars atmosphere would have important implications for astrobiology.
REFERENCES
1. P.B. Price and T. Sowers, “Temperature Dependence of Metabolic Rates for Microbial Growth, Maintenance, and Survival,” Proceedings of the National Academy of Sciences 101(13):4631-4636, doi:10.1073/pnas.0400522101, 2004.
2. R.Y. Morita, Bacteria in Oligotrophic Environments, Chapman and Hall, New York, 1997.
3. D.G. Capone, R. Popa, B. Flood, and K.H. Nealson, “Follow the Nitrogen,” Science 312:708-709, 2006.
4. See, for example, Table 5.1 in National Research Council, Preventing the Forward Contamination of Mars, National Academies Press, Washington, D.C., 2006, p. 71.
5. D. Beaty, K. Buxbaum, M. Meyer, N. Barlow, W. Boynton, B. Clark, J. Deming, P.T. Doran, K. Edgett, S. Hancock, J. Head, M. Hecht, V. Hipkin, T. Kieft, R. Mancinelli, E. McDonald, C. McKay, M. Mellon, H. Newsom, G. Ori, D. Paige, A.C. Schuerger, M. Sogin, J.A. Spry, A. Steele, K. Tanaka, and M. Voytek, “Findings of the Mars Special Region Science Analysis Group,” Astrobiology 6(5):677-732, 2006.
6. P.B. Price and T. Sowers, “Temperature Dependence of Metabolic Rates for Microbial Growth, Maintenance, and Survival,” Proceedings of the National Academy of Sciences 101(13):4631-4636, doi:10.1073/pnas.0400522101, 2004.
7. K. Kashefi and D.R. Lovley, “Extending the Upper Limits of Life,” Science 301:93, 2003.
8. M.O. Schrenk, D.S. Kelley, J.R. Delaney, and J.A. Baross, “Incidence and Diversity of Microorganisms Within the Walls of an Active Deep-Sea Sulfide Chimney,” Applied and Environmental Microbiology 69(6):3580-3592, 2003.
9. W.D. Grant, “Life at Low Water Activity,” Phil. Trans. R. Soc. Lond. B 359:1249-1267, doi:10.1098/rstb.2004.1502, 2004.
10. D. Beaty, K. Buxbaum, M. Meyer, N. Barlow, W. Boynton, B. Clark, J. Deming, P.T. Doran, K. Edgett, S. Hancock, J. Head, M. Hecht, V. Hipkin, T. Kieft, R. Mancinelli, E. McDonald, C. McKay, M. Mellon, H. Newsom, G. Ori, D. Paige, A.C. Schuerger, M. Sogin, J.A. Spry, A. Steele, K. Tanaka, and M. Voytek, “Findings of the Mars Special Region Science Analysis Group,” Astrobiology 6(5):677-732, 2006.
11. D.G. Capone, R. Popa, B. Flood, and K.H. Nealson, “Follow the Nitrogen,” Science 312:708-709, 2006.
12. L.A. Haskins, A. Wang, H.Y. McSween, B.C. Clark, D.J. Des Marais, S.M. McLennan, N.J. Tosca, J.A. Hurowitz, J.D. Farmer, A. Yen, S.W. Squyres, R.E. Arvidson, G. Klingelhöfer, C. Schröder, P.A. de Souza, Jr., D.W. Ming, R. Gellert, J. Zipfel, J. Brückner, J.F. Bell, III, K. Herkenhoff, P.R. Christensen, S. Ruff, D. Blaney, S. Gorevan, N.A. Cabrol, L. Crumpler, J. Grant, and L. Soderblom, “Water Alteration of Rocks and Soils on Mars and the Spirit Rover Site in Gusev Crater,” Nature 436:66-69, 2005.
13. H.P. Klein, “The Viking Mission and the Search for Life on Mars,” Reviews of Geophysics 17:1655-1662, 1979.
14. See, for example, P.B. James, H.H. Kieffer, and D.A. Paige, “The Seasonal Cycle of Carbon Dioxide on Mars,” pp. 934-968 in H.H. Kieffer, B.M. Jakosky, C.W. Snyder, and M.S. Matthews (eds.), Mars, University of Arizona Press, Tucson, Arizona, 1992.
15. See, for example, B.M. Jakosky and M.T. Mellon, “Water on Mars,” Physics Today 57(4):71-76, 2004.
16. See, for example, H.H. Kieffer and A.P. Zent, “Quasi-periodic Climate Change on Mars,” pp. 1180-1218 in H.H. Kieffer, B.M. Jakosky, C.W. Snyder, and M.S. Matthews (eds.), Mars, University of Arizona Press, Tucson, Arizona, 1992.
17. W.C. Feldman, T.H. Prettyman, S. Maurice, J.L. Plaut, D.L. Bish, D.T. Vaniman, M.T. Mellon, A.E. Metzger, S.W. Squyres, S. Karunatillake, W.V. Boynton, R.C. Elphic, H.O. Funsten, D.J. Lawrence, and R.L. Tokar, “The Global Distribution of Near Surface Hydrogen on Mars,” Journal of Geophysical Research 109(E9):E09006, doi:10.1029/2003JE02160, 2004.
18. M.C. Malin, K.S. Edgett, L.V. Posiolova, S.M. McColley, and E.Z. Noe Dobrea, “Present-Day Impact Cratering Rate and Contemporary Gully Activity on Mars,” Science 314:1573-1577, 2006.
19. P.R. Christensen, “Formation of Recent Martian Gullies Through Melting of Extensive Water-rich Snow Deposits,” Nature 422:45-48, 2003.
20. J. Laskar, A.C.M Correia, M. Gastineau, F. Joutel, B. Levrard, and P. Robutel, “Long Term Evolution and Chaotic Diffusion of the Insolation Quantities of Mars,” Icarus 170:343-364, 2004.
21. F. Costard, F. Forget, N. Mangold, and J.P. Peulvast, “Formation of Recent Martian Debris Flows by Melting of Near-surface Ground Ice at High Obliquity,” Science 295:110-113, 2002.
22. See, for example, L. Borg and M.J. Drake, “A Review of Meteorite Evidence for the Timing of Magmatism and of Surface or Near-surface Liquid Water on Mars,” Journal of Geophysical Research 110:E12S03, doi:1029/2005JE002402, 2005.
23. M.H. Carr, The Surface of Mars, Cambridge University Press, Cambridge, 2006.
24. M.H. Carr and J.E. Head, “Oceans on Mars: An Assessment of Observational Evidence and Possible Fate,” Journal of Geophysical Research 108(E5):5042-5070, 2003.
25. See, for example, M.H. Carr, The Surface of Mars, Cambridge University Press, New York, N.Y. 2006; and references therein.
26. N.H. Sleep and K. Zahnle, “Refugia from Asteroid Impact on Early Mars and the Early Earth,” Journal of Geophysical Research 103(E12):28529-28544, 1998.
27. R.M. Haberle, “Early Climate Models,” Journal of Geophysical Research 103(E12):28467-28479, 1998.
28. T.L. Segura, O.B. Toon, A. Colaprete, and K. Zahnle, “Environmental Effects of Large Impacts,” Science 298:1977-1980, 2002.
29. J. Bibring, Y. Langevin, J.F. Mustard, F. Poulet, R. Arvidson, A. Gendrin, B. Gondet, N. Mangold, P. Pinet, F. Forget, and the OMEGA team M. Berthé, J. Bibring, A. Gendrin, C. Gomez, B. Gondet, D. Jouglet, F. Poulet, A. Soufflot, M. Vincendon, M. Combes, P. Drossart, T. Encrenaz, T. Fouchet, R. Merchiorri, G. Belluci, F. Altieri, V. Formisano, F. Capaccioni, P. Cerroni, A. Coradini, S. Fonti, O. Korablev, V. Kottsov, N. Ignatiev, V. Moroz, D. Titov, L. Zasova, D. Loiseau, N. Mangold, P. Pinet, S. Douté, B. Schmitt, C. Sotin, E. Hauber, H. Hoffmann, R. Jaumann, U. Keller, R. Arvidson, J.F. Mustard, T. Duxbury, F. Forget, and G. Neukum, “Global Mineralogical and Aqueous Mars History Derived from OMEGA/Mars Express Data,” Science 312:400-404, 2006.
30. See, for example, M.H. Carr, The Surface of Mars, Cambridge University Press, New York, N.Y. 2006; and references therein.
31. D.M. Burr, A.S. McEwen, and S.E. Sakimoto, “Recent Aqueous Floods from the Cerberus Fossae, Mars,” Geophysical Research Letters 29:1, 2002. doi:10.1029/2001Gl013345.
32. See, for example, M.H. Carr, The Surface of Mars, Cambridge University Press, New York, N.Y. 2006; and references therein.
33. See, for example, M.H. Carr, The Surface of Mars, Cambridge University Press, New York, N.Y. 2006; and references therein.
34. M.A. Kreslavsky, and J.W. Head, “Fate of Outflow Channel Effluents in the Northern Lowlands of Mars: The Vastitas Borealis Formation as a Sublimation Residue from Frozen, Ponded Bodies of Water,” Journal of Geophysical Research 107(E12):5121, doi:10.1029/2001JE001831, 2002.
35. J. Laskar, A.C.M Correia, M. Gastineau, F. Joutel, B. Levrard, and P. Robutel, “Long Term Evolution and Chaotic Diffusion of the Insolation Quantities of Mars,” Icarus 170:343-364, 2004.
36. J.S. Kargel, V.R. Baker, J.E. Beget, J.E. Lockwood, T.L. Péwé, J.S. Shaw, and R.G. Strom, “Evidence for Ancient Continental Glaciation in the Martian Northern Plains,” Journal of Geophysical Research 100:5351-5368, 1995.
37. See, for example, M.H. Carr, The Surface of Mars, Cambridge University Press, New York, N.Y. 2006; and references therein.
38. M.H. Carr, The Surface of Mars, Cambridge University Press, New York, N.Y., 2006.
39. See, for example, M.H. Carr, The Surface of Mars, Cambridge University Press, New York, N.Y. 2006; and references therein.
40. J. Bibring, Y. Langevin, J.F. Mustard, F. Poulet, R. Arvidson, A. Gendrin, B. Gondet, N. Mangold, P. Pinet, F. Forget, and the OMEGA team M. Berthé, J. Bibring, A. Gendrin, C. Gomez, B. Gondet, D. Jouglet, F. Poulet, A. Soufflot, M. Vincendon, M. Combes, P. Drossart, T. Encrenaz, T. Fouchet, R. Merchiorri, G. Belluci, F. Altieri, V. Formisano, F. Capaccioni, P. Cerroni, A. Coradini, S. Fonti, O. Korablev, V. Kottsov, N. Ignatiev, V. Moroz, D. Titov, L. Zasova, D. Loiseau, N. Mangold, P. Pinet, S. Douté, B. Schmitt, C. Sotin, E. Hauber, H. Hoffmann, R. Jaumann, U. Keller, R. Arvidson, J.F. Mustard, T. Duxbury, F. Forget, and G. Neukum, “Global Mineralogical and Aqueous Mars History Derived from OMEGA/Mars Express Data,” Science 312:400-404, 2006.
41. See, for example, H.P. Klein, “The Viking Biological Investigations: General Aspects,” Journal of Geophysical Research 82:4677-4680, 1977.
42. K. Biemann, J. Oro, P. Toulmin III, L.E. Orgel, A.O. Nier, D.M. Anderson, D. Flory, A.V. Diaz, D.R. Rushneck, and P.G. Simmonds, “The Search for Organic Substances and Inorganic Volatile Compounds in the Surface of Mars,” Journal of Geophysical Research 82:4641-4662, 1977.
43. H.P. Klein, “The Search for Life on Mars,” Review Geophysics and Space Physics 17:1655-1662, 1979.
44. D.Y. Sumner, “Poor Preservation Potential of Organics in Meridiani Planum Hematite-bearing Sedimentary Rocks,” Journal of Geophysical Research 109:E12007, doi:10.1029/2004JE002321, 2004.
45. K. Biemann, J. Oro, P. Toulmin III, L.E. Orgel, A.O. Nier, D.M. Anderson, D. Flory, A.V. Diaz, D.R. Rushneck, and P.G. Simmonds, “The Search for Organic Substances and Inorganic Compounds in the Surface of Mars,” Journal of Geophysical Research 82:4641-4658, 1977.
46. R. Navarro-Gonzalez, K.F. Navarro, J. de la Rosa, E. Iñiguez, P. Molina, L.D. Miranda, P. Morales, E. Cienfuegos, P. Coll, F. Raulin, R. Amils, and C.P. McKay, “The Limitations on Organic Detection in Mars-like Soils by Thermal Volatilization–Gas Chromatography–MS and their Implications for the Viking Results,” Proceedings of the National Academy of Sciences 103:16089-16094, 2006.
47. N.H. Horowitz, G.L. Hobby, and J.S. Hubbard, “Viking on Mars: The Carbon Assimilation Experiments,” Journal of Geophysical Research 82:4659-4662, 1977.
48. V.I. Oyama and B.J. Berdahl, “The Viking Gas Exchange Experiment Results from Chryse and Utopia Surface Samples,” Journal of Geophysical Research 82:4669-4676, 1977.
49. V.V. Levin and P.A. Straat, “Recent Results from the Viking Labeled Release Experiment on Mars,” Journal of Geophysical Research 82:4663-4667, 1977.
50. See, for example, G.V. Levin, “The Viking Labeled Release Experiment and Life on Mars,” SPIE Proceedings 3111:146-161, 1997.
51. National Aeronautics and Space Administration, An Exobiology Strategy for Mars Exploration, National Aeronautics and Space Administration, Washington, D.C., 1995.
52. M. Schidlowski, “A 3,800 Million Year Isotopic Record of Life from Carbon in Sedimentary Rocks,” Nature 333:313-318, 1988.
53. J.W. Schopf “Microfossils of the Early Archaean Apex Chert: New Evidence of the Antiquity of Life,” Science 260:640-646, 1993.
54. S.J. Mojzsis, G. Arrhenius, K.D. McKeegan, T.M. Harrison, A.P. Nutman, and C.R. Friend, “Evidence for Life on Earth Before 3,800 Million Years Ago,” Nature 384:55-59, 1996.
55. M.T. Rosing, “13C-depleted Carbon Microparticles in >3700 Ma Sea-floor Sedimentary Rocks from West Greenland,” Science 283:674-676, 1999.
56. S.J. Mojzsis and T.M. Harrison, “Vestiges of a Beginning: Clues to the Emergent Biosphere Recorded in the Oldest Known Sedimentary Rocks,” Geological Society of America Today 10:1-7, 2000.
57. M.D. Brasier, O.R. Green, A.P. Jephcoat, A.K. Kleppe, M.J. Van Kranendonk, J.F. Lindsay, A. Steele, and N.V. Grassineau, “Questioning the Evidence for Earth’s Oldest Fossils,” Nature 416:76-81, 2002.
58. C.M. Fedo and M.J. Whitehouse, “Metasomatic Origin of Quartz-Pyroxene Rock, Akilia, Greenland, and Implications for Earth’s Earliest Life,” Science 296:1448-1452, 2002.
59. J.D. Pasteris and B. Wopenka, “Laser Raman Spectroscopy: Images of the Earth’s Earliest Fossils?,” Nature 420:476-477, 2002.
60. H. Furnes, N.R. Banerjee, K. Muehlenbachs, H. Staudigel, and M. de Wit, “Life Recorded in Archean Pillow Lavas,” Science 304:578, 2004.
61. See, for example, R.A. Kerr, “Reversals Reveal Pitfalls in Spotting Ancient and E.T. Life,” Science 296:1384-1385, 2002.
62. See, for example, R. Dalton, “Microfossils: Squaring Up Over Ancient Life,” Nature 417:782-784, 2002.
63. J.W. Schopf “Microfossils of the Early Archaean Apex Chert: New Evidence of the Antiquity of Life,” Science 260:640-646, 1993.
64. J.J. Brocks, G.A. Logan, R. Buick, and R.E. Summons, “Archean Molecular Fossils and the Early Rise of Eukaryotes,” Science 285:1033-1036, 1999.
65. J.J. Brocks, G.A. Logan, R. Buick, and R.E. Summons, “Archean Molecular Fossils and the Early Rise of Eukaryotes,” Science 285:1033-1036, 1999.
66. S.B. Hedges, J.E. Blair, M.L. Venturi, and J.L. Shoe, “A Genomic Timescale for the Origin of Eukaryotes and the Rise of Complex Multicellular Life,” BMC Evolutionary Biology 1:4, 2001.
67. M.D. Brasier, O.R. Green, A.P. Jephcoat, A.K. Kleppe, M.J. Van Kranendonk, J.F. Lindsay, A. Steele, and N.V. Grassineau, “Questioning the Evidence for Earth’s Oldest Fossils,” Nature 416:76-81, 2002.
68. J.W. Schopf, A.B. Kudryavtsev, D.G. Agresti, T.J. Wdowiak, and A.D. Czaja, “Laser-Raman Imagery of Earth’s Oldest Fossils,” Nature 416:73-76, 2002.
69. See, for example, J.D. Pasteris and B. Wopenka, “Laser Raman Spectroscopy: Images of the Earth’s Earliest Fossils?” Nature 420:476-477, 2002.
70. S.J. Mojzsis, G. Arrhenius, K.D. McKeegan, T.M. Harrison, A.P. Nutman, and C.R. Friend, “Evidence for Life on Earth Before 3,800 Million Years Ago,” Nature 384:55-59, 1996.
71. S.J. Mojzsis and T.M. Harrison, “Vestiges of a Beginning: Clues to the Emergent Biosphere Recorded in the Oldest Known Sedimentary Rocks,” Geological Society of America Today 10:1-7, 2000.
72. C.M. Fedo and M.J. Whitehouse, “Metasomatic Origin of Quartz-Pyroxene Rock, Akilia, Greenland, and Implications for Earth’s Earliest Life,” Science 296:1448-1452, 2002.
73. For a summary, see, for example, C.M. Fedo, M.J. Whitehouse, and B.S. Kamber, “Geological Constraints on Detecting the Earliest Life on Earth: A perspective from the Early Archaean (older than 3.7 Gyr) of Southwest Greenland,” Philosophical Transactions of the Royal Society B 361:851-867, 2006.
74. D.S. McKay, E.K. Gibson, Jr., K.L. Thomas-Keprt, H. Vali, C.S. Romanek, S.J. Clemett, X.D.F. Chillier, C.R. Maechling, and R.N. Zare, “Search for Past Life on Mars: Possible Relic Biogenic Activity in Martian Meteorite ALH 84001,” Science 273:924-930, 1996.
75. See, for example, D.J. DesMarais, B.M. Jakosky, and B.M. Hynek, “Astrobiological Implications of Mars Surface Composition and Properties,” in The Martian Surface: Composition, Mineralogy, and Physical Properties, J.F. Bell III, ed., Cambridge University Press, Cambridge, U.K., in press.
76. See, for example, E.O. Kajander, M. Bjorklund, and N. Ciftcioglu, “Suggestions from Observations on Nanobacteria Isolated from Blood,” pp. 50-55, and references therein, in National Research Council, Size Limits of Very Small Microorganisms: Proceedings of a Workshop, National Academy Press, Washington, D.C., 1999.
77. National Research Council, Size Limits of Very Small Microorganisms: Proceedings of a Workshop, National Academies Press, Washington D.C., 1999.
78. B.J. Baker, G.W. Tyson, R.I. Webb, J. Flanagan, P. Hugenholtz, E.E. Allen, and J.F. Banfield, “Lineages of Acidophilic Archaea Revealed by Community Genomic Analysis,” Science 314:1933-1935, 2006.
79. J.P. Bradley, R.P. Harvey, and H.Y. McSween, “No ‘Nanofossils’ in Martian Meteorite,” Nature 390:454, 1997.
80. See for example R.L. Folk, “SEM Imaging of Bacteria and Nanobacterial in Carbonate Sediments and Rocks,” Journal of Sedimentary Petrology 63:990-999, 1993.
81. E. Anders, “Evaluating the Evidence for Past Life on Mars,” Science 274:2119-2120, 1996.
82. D.C. Golden, D.W. Ming, R.V. Morris, A. Brearley, H.V. Lauer, A.H. Treiman, M.E. Zolensky, C.S. Schwandt, G.E. Lofgren, and G.A. McKay, “Evidence for Exclusively Inorganic Formation of Magnetite in Martian Meteorite ALH 84001,” American Mineralogist 89:681-695, 2004.
83. A. Steele, D.W. Beaty, J. Amend, R. Anderson, L. Beegle, L. Benning, J. Bhattacharya, D. Blake, W. Brinckerhoff, J. Biddle, S. Cady, P. Conrad, J. Lindsay, R. Mancinelli, G. Mungas, J. Mustard, K. Oxnevad, J. Toporski, and H. Waite, “The Astrobiology Field Laboratory,” Unpublished white paper, 72 p, posted Dec., 2005 by the Mars Exploration Program Analysis Group (MEPAG) at http://mepag.jpl.nasa.gov/reports/index.html.
84. V. Formisano, S. Atreva, T. Encrenaz, N. Ignatiev, and M. Giuranna, “Detection of Methane in the Atmosphere of Mars,” Science 306:1758-1761, 2004.
85. V.A. Krasnopolsky, J.P. Maillard, and T.C. Owen, “Detection of Methane in the Martian Atmosphere: Evidence for life?” Icarus 172:537-547, 2004.
86. M.J. Mumma, R.E. Novak, M.A. DiSanti, B.P. Bonev, and N. Dello Russo, “Detection and Mapping of Methane and Water on Mars,” Bulletin of the American Astronomical Society 36:1127, 2004.
87. M. Allen, B. Sherwood Lollar, B. Runnegar, D.Z. Oehler, J.R. Lyons, C.E. Manning, and M.E. Summers, “Is Mars Alive?,” Eos 87(41):433-448, 2006.
88. M. Allen, B. Sherwood Lollar, B. Runnegar, D.Z. Oehler, J.R. Lyons, C.E. Manning, and M.E. Summers, “Is Mars Alive?,” Eos 87(41):433-448, 2006.
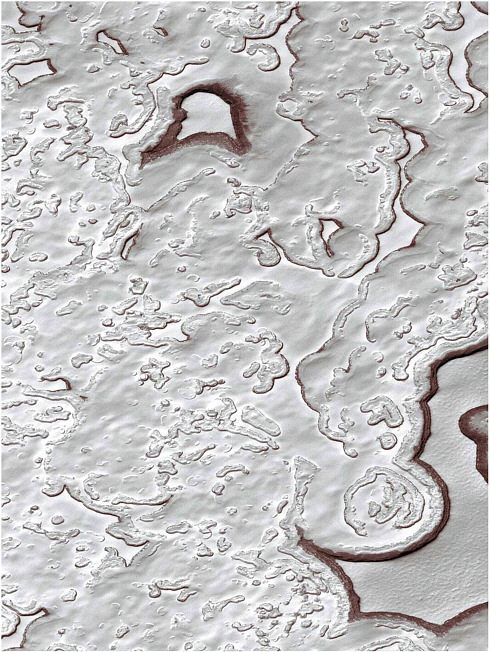
FIGURE 3.1 This image shows a portion of the residual southern polar cap. The carbon dioxide has sublimated to expose underlying water ice. Changes from year to year suggest that in the present epoch the amount of carbon dioxide in the residual cap is decreasing. This image from the Mars Orbiter Camera on the Mars Global Surveyor spacecraft is courtesy of NASA/ JPL/Malin Space Science Systems. The image covers an area of 2.9 by 4.8 km.























