A3
THE SPRING 2009 INFLUENZA A H1N1 OUTBREAK: A LOCAL PUBLIC HEALTH PERSPECTIVE
Jeffrey S. Duchin, M.D.19
Public Health—Seattle and King County and University of Washington, Seattle, Washington
Epidemiology, Surveillance, and Public Health Emergency Response
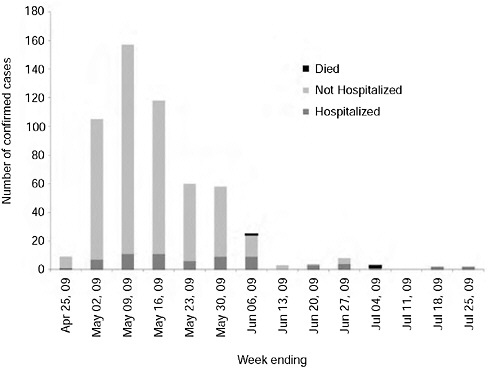
Between April 25, 2009, and July 31, 2009, 565 confirmed cases of 2009-H1N1 influenza A were reported to Public Health–Seattle and King County, WA, including 70 hospitalizations and three deaths (Figure A3-1). Racial and ethnic minorities were also disproportionately represented: 63 percent of cases were nonwhite compared to 24 percent of the county population, and 22 percent of cases were Hispanic compared to 6.8 percent of the county. Twenty-three languages were spoken by the 393 cases for which language data were available and 157 (40 percent) spoke a language other than English as the primary or secondary language at home. Among the foreign language speakers, Spanish was the most common language (48 percent), followed by Somali (13 percent) and Vietnamese (9 percent). Other languages were spoken by less than 5 percent of foreign language speakers (Kwan-Gett et al., 2009).
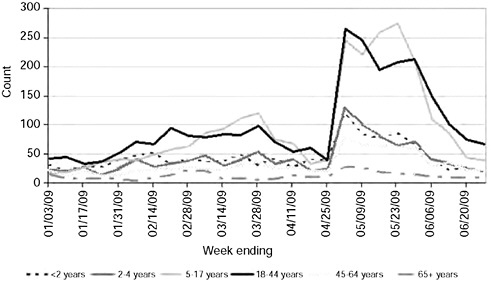
The Centers for Disease Control and Prevention’s (CDC) initial criteria for reporting and testing of suspected cases of 2009-H1N1 influenza A included travel to an area where cases had been confirmed and resulted in a steady increase in the number of reports received locally, accelerating sharply after King County cases were confirmed during the week of April 26, 2009. The peak number of persons with influenza-like illness (ILI) presenting to local emergency departments (EDs) based on syndromic surveillance system data ranged from 200 to 375 percent greater than that during the 2008-2009 seasonal influenza outbreak and averaged 500 percent higher than pre-outbreak levels. Elevated levels of ED visits lasted for over one month, with highest volumes seen among patients in the 5-17 and 18-44 year-old age groups (Figure A3-2). Although the proportion of ED patients with ILI who were admitted to the hospital was comparable to that observed during the seasonal influenza period, an increased number of hospital admissions strained certain facilities, particularly those caring for children and tertiary care center intensive care units.
Syndromic surveillance at 18 of 19 county EDs allowed timely evaluation of the number and proportion of ED visits for ILI stratified by age and hospital,
as well as number and proportion of ILI cases admitted to the hospital. We were also able to evaluate trends over time to compare current ILI activity and age distributions with previous influenza seasons. We observed a good correlation between syndromic ED ILI trends and laboratory-based surveillance data, and between hospitalized confirmed H1N1 cases and syndromic ED admissions among patients with ILI, suggesting that ED ILI data tracked the novel influenza A H1N1 outbreak well. Resources necessary to manage the syndromic surveillance system are provided through federal preparedness funding. Limitations of syndromic surveillance include the inability to do case follow-up easily (because data are deidentified), missing data (e.g., disposition), absence of standardized chief complaint categories or terms resulting in potential misclassification, and variable definitions of ILI used in syndromic systems around the country. Until syndromic ILI and pneumonia data can be validated, their sensitivity and specificity remain unknown.
School absenteeism reporting is a commonly employed indirect indicator of seasonal influenza activity and had special prominence during the spring 2009-H1N1 influenza A outbreak. The increased focus on absenteeism reporting highlighted the limitations of our current system and potential areas for improvement.
Until recently, school absenteeism reporting has been a passive, manual system, in which school personnel are requested to report weekly absenteeism. Participation in this system has been variable and inconsistent across King County’s 19 public school districts (with 525 schools) and 225 private schools. Consequently, there are no reliable historical data to allow analysis of absenteeism trends over time. Absenteeism rates are reported at the school level. Data on grade-level rates are not provided, definitions of absence are not standardized, and the threshold for reporting (traditionally 10 percent) is arbitrary and not adjusted for baseline levels of absenteeism.
To promote increased participation in absenteeism reporting, in 2008 we developed a web-based reporting system that requested the following information: a weekly absenteeism report with peak absenteeism for the week, whether more than half of absenteeism was due to illness, and the predominant reasons for absenteeism. However, many schools did not report because of resource and staff constraints, and consistency of reporting was poor. To further simplify reporting and relieve schools of the associated administrative burden, after the spring 2009-H1N1 influenza A outbreak we implemented district-level automated electronic absenteeism reporting to collect aggregate counts of the daily absences by school and grade level. This system is made possible through collaboration among public health, school districts, and a cooperative of districts and educational service districts that were already collecting electronic data. Strengths of the system include complete data collection from participating school districts, the ability to analyze trends over time, establish baseline levels of absenteeism and define increases above baseline, and inclusion of grade level data. Limitations include
lack of information about the reason for absence (which continues to require manual follow-up), and no long-term historical data. The extent to which local public health should be investing in school absenteeism surveillance systems and the public health value of such systems remain to be determined.
Over 200 staff and 40 volunteers were assigned to the Department’s 2009-H1N1 influenza A response resulting in significant unbudgeted expenses. Our Health and Medical Area Command (HMAC) was fully activated for 19 days beginning on April 27 and many response activities continued after formal closure of the HMAC. HMAC functions included healthcare system situational awareness, regional medical resource management (including management of the strategic national stockpile), mass dispensing, risk communications, public information call center and regional call center coordination, epidemiology and surveillance, business continuity, and volunteer mobilization (including Medical Reserve Corps) for surge response.
The increase in outbreak-associated surveillance and epidemiological response activities overloaded the Department’s Communicable Disease Epidemiology Section. Through the first seven weeks of the 2009-H1N1 influenza A outbreak response, the Section required an extra 10.6 full-time employees (FTE) daily, a total of 41 new surge staff working 2,468 hours. Over the first six weeks of the outbreak, the Section received 1,615 calls from heathcare providers alone—over 100 calls per day initially. Volunteers from the community and department staff reassigned from other programs were recruited to supplement core staff conducting case investigations, follow-up of school absenteeism reports and illness clusters, and surveillance data management. Training surge personnel during the outbreak response was burdensome for the regular staff occupied with response duties. Individual surge staff had different levels of experience and availability and had inconsistent schedules. Because surge staff did not have the experience or training necessary to assume lead roles for surveillance, epidemiological investigation, or outbreak response, the usual Section staff served as leads in our incident command response framework during the entire outbreak response and consequently experienced physical and mental fatigue. Because of the limited number of trained staff, shift work as is practiced in other emergency response activities was not possible.
Sustained media attention and reports of school closures raised public anxiety and likely contributed to a surge in outpatient medical visits for ILI and a large volume of phone calls to both the health department and healthcare facilities. Healthcare providers reported large numbers of patients presenting for care in EDs and primary care settings with mild illnesses. Complying with surveillance guidelines, clinicians requested 2009-H1N1 influenza A laboratory polymerase chain reaction (PCR) testing at the Public Health Laboratory (PHL) on large numbers of patients with nonspecific ILI symptoms. Consequently, public health staff were inundated with processing and investigating case reports, tracking laboratory test requests and results, analysis and reporting of surveillance data,
and responding to questions from healthcare providers and facilities regarding clinical management, infection control considerations, surveillance, and reporting. At times, hospitals did not have adequate staffing to provide timely information on admitted cases and there were not enough public health staff to provide onsite support.
Currently, case reporting is limited to hospitalized cases, which reduces but does not eliminate the burden associated with case reporting and follow-up, and processing, analyzing, and reporting surveillance data. However, identifying and reporting cases continues to require additional resources at healthcare facilities and a proportion of reports are incomplete. More efficient and less labor-intensive surveillance and reporting mechanisms at the local level that take advantage of existing electronic databases would be useful both in routine practice and during health emergencies. Routine collection of information relevant to health disparities including socioeconomic factors such as health insurance coverage, homelessness, primary language spoken, and other social and demographic variables would allow a better understanding of disease risk factors including racial and ethnic data, but requires additional resources.
Healthcare System Response
Local EDs were rapidly overloaded with mildly ill and uncomplicated cases seeking testing and treatment. Outpatient healthcare providers were not prepared to manage cases requiring respiratory (airborne) infection control precautions and some clinics referred cases to EDs regardless of severity of illness. Healthcare providers reported an overwhelming number of phone calls, primarily from concerned patients who did not have serious illness. Standardized triage tools for the public and healthcare providers were not available during the spring 2009-H1N1 influenza A outbreak to help minimize unnecessary use of the healthcare system. Triage tools that are reviewed and endorsed by the appropriate medical professional organizations would be useful in managing future outbreaks. EDs also reported an increase in patients who were uninsured and/or did not have a primary health care provider; in 2008, 12.5 percent (approximately 153,000) of King County adults ages 18-64 years reported no health insurance coverage. The problem of medically underserved populations with no medical home placed additional unnecessary stress on healthcare systems.
Because of the surge in patients tested for 2009-H1N1 influenza A infection, healthcare facilities and laboratories reported shortages of nasopharyngeal swabs and viral transport medium. There was a failure of “just in time” purchasing as hospitals, outpatient facilities, and local vendors reported shortages of surgical masks, N95 respirators, and equipment for fit testing N95 respirators. Recommendations on N95 respirator use (personal protective equipment [PPE]) by healthcare workers by the CDC differed from those of the World Health Organization, the Society for Healthcare Epidemiology of America, the Association
of Professionals in Infection Control, the Infectious Disease Society of America, and other expert professional groups (APIC, 2009; CDC, 2009a,b; IDSA, 2009; SHEA, 2009; WHO, 2009).
Variation in adoption of PPE recommendations by state and local public health agencies and healthcare systems resulted in confusion and hampered development of local infection control guidance. Healthcare providers perceived 2009-H1N1 influenza A to be similar to seasonal influenza with respect to apparent mode of transmission and severity of illness and logistical considerations made widespread adoption of N95 respirator use impractical. Therefore, most healthcare providers and systems reserved N95 masks for high-risk exposures. Differences in respirator policies among healthcare facilities led to inconsistent rates of equipment usage such that a small number of local healthcare facilities requested a large proportion of the regional stockpile of N95 respirators.
Clinicians and healthcare facilities also reported shortages of commercial rapid influenza diagnostic test kits. This was exacerbated by several factors, including the practice of testing cases of ILI for 2009-H1N1 influenza A regardless of severity of illness or need to guide clinical management, and a requirement for a positive rapid test result for subsequent H1N1 testing at the state PHL to reduce the volume of specimens submitted. Case investigations and conversations with local clinicians suggested that some clinicians inappropriately relied on results of 2009-H1N1 influenza A PCR testing at the PHL to guide case management, including infection control measures and antiviral treatment. The lack of widely available rapid, reliable, and affordable point-of-care diagnostic tests for 2009-H1N1 influenza A hampered both the medical and public health response to the evolving outbreak, and their development should be prioritized (IDSA, 2007).
As increasing numbers of persons meeting the 2009-H1N1 influenza A surveillance case definition were identified, the state PHL was inundated with specimens for PCR testing, resulting in delays in testing and reporting of results. For example, influenza subtyping results, needed to help guide empiric treatment during an outbreak in a long-term-care facility, were not available for more than a week. Although clinicians, healthcare facilities, and laboratories were requested to report suspected cases before submitting specimens to the PHL, many specimens were submitted directly to the PHL, bypassing the local health surveillance system and complicating tracking of cases and reporting of results. Pandemic response plans call for integration of clinical laboratories into the public health response (HHS, 2009). Some clinical laboratories, particularly those not affiliated with hospitals, were not well integrated into the public health outbreak response. Examples include not having systems to either receive or act on information from public health authorities and lack of familiarity with processing influenza specimens for submission to the PHL and procedures for reporting results.
Providing information and technical support for healthcare providers was a core component of the public health H1N1 outbreak response and was labor and
time intensive. There was a high demand from clinicians for consultation with public health subject matter experts and for information on diagnosis, treatment, infection control, and community mitigation measures. Frequent written updates were disseminated by email listserv, broadcast fax, and web portals. Daily (and subsequent weekly) conference calls with a variety of healthcare system stakeholders were organized by public health and healthcare coalition staff.
The healthcare system response had several notable successes. HMAC, working through our King County Healthcare Coalition,20 a collaboration between public health and regional healthcare system stakeholders to foster healthcare system emergency preparedness, promptly mobilized hospitals to prepare to implement pandemic response plans and successfully facilitated redistribution of scarce medical supplies among regional hospitals and when necessary through provision from the strategic national stockpile (SNS) and/or the county stockpile. HMAC coordinated communication with area hospitals to provide technical information (e.g., infection control and testing and treatment guidelines) and conducted conference calls with large pharmacy chains across the region to coordinate information regarding available antiviral inventories, protocols to protect pharmacy staff, screening criteria, and dispensing protocols for stockpiled antiviral drugs. HMAC mobilized county antiviral drug stockpiles and organized community dispensing clinics in response to shortages of antiviral drugs. A public call center was opened to provide general health information and medical triage and received 1,199 calls in just under a week of operation. Clinical and nonclinical health department staff and volunteers staffed the call center and a partnership with a regional private after-hours nurse consulting line was established for after-hours operation. Hospitals successfully employed the Hospital Incident Command System during the response.
During the spring outbreak, local intensive care unit (ICU) capacity and capability were stressed but not exceeded. However, hospitalized ICU cases required an unusually high level of medical resources and advanced treatment measures (rescue therapies), consistent with reports from other areas (ANZIC Influenza Investigators, 2009; White and Angus, 2009). The complexity of critical care management required by 2009-H1N1 influenza A cases raises the possibility of exceeding regional ability to care for highly complex cases (surge capability) independent of exceeding ICU bed surge capacity (Knebel and Trabert, 2007). Clinicians reported that caring for 2009-H1N1 influenza A cases was more demanding and stressful compared with cases of seasonal influenza requiring ICU admission. Because ICU occupancy rates are routinely at or near capacity, the potential also exists for exceeding available ICU beds and/or critical life- sustaining medical resources during a larger outbreak, including if ICU admissions for other illnesses are greater during the fall and winter compared to the spring. The spring outbreak also highlighted the pressing need to establish a system for triage and allocation
of scarce medical resources during health emergencies. Although there are few available resources dedicated to this purpose, significant work remains to develop adequate regional critical care surge capacity.
Countermeasures: Antiviral Drugs
In response to large numbers of patients with ILI presenting to healthcare facilities, there was a spike in prescriptions for oseltamivir antiviral treatment. Healthcare providers reported an unusually high demand for treatment by persons with mild and uncomplicated illness and persons with potential exposures to H1N1 cases. There were also reports of healthcare workers filling prescriptions for personal antiviral drug stockpiles that resulted in the issuance of “Dear Doctor” letters urging judicious antiviral drug prescribing from the State Health Officer. Local pharmacies had few courses of oseltamivir available in stores because demand is typically low. In addition, pharmacies had difficulty replenishing their supplies rapidly. At least one healthcare facility did not provide oseltamivir from the hospital formulary to patients discharged from the ED or urgent care clinic, directing patients instead to community pharmacies. Pediatric oseltamivir formulation was in particularly short supply.
The extent to which clinicians from different healthcare systems (both within the private sector and between public and private sectors) complied with guidelines for administration of antiviral drugs varied, resulting in potential inequities in access to antiviral treatment. Another stressor on antiviral drug supplies was the CDC recommendation for postexposure chemoprophylaxis (PEP) for healthcare workers who had close contact with a suspected 2009-H1N1 influenza A case without using an N95 respirator. Because relatively few healthcare workers used N95 respirators, almost all ILI patient encounters met the criteria for PEP. (This recommendation was revised in October 2009.)
Access to antiviral drugs varied across geographic boundaries. Although national pandemic plans call for sufficient antiviral stockpiles to treat 25 percent of the U.S. population, the federal government has purchased a supply adequate to treat approximately 15 percent of the population, relying on subsidized purchases by states and local jurisdictions to provide the remainder (HHS, 2006). Not all local jurisdictions have purchased additional supplies to reach the target. King County previously purchased a local antiviral drug stockpile that together with the federal stockpile is sufficient to treat at least 25 percent of the local population; however, few other local health jurisdictions in Washington did so and no others have met the target for 25 percent population coverage. In order to ensure timely access to treatment before the national pharmaceutical stockpile was available locally, King County provided oseltamivir from the local stockpile to community locations. Neighboring counties were not able to do so, resulting in instances where patients were the referred across county lines for antiviral drug treatment.
CDC activated delivery of the SNS with antiviral drugs and PPE to states on April 27, 2009. On May 4th, the first supplies of SNS antiviral drugs reached our local public health distribution center and it was not until May 14th that the remainder of the initial 25 percent SNS allocation reached the distribution center. In the intervening days, we used oseltamivir supplies from the county stockpile to supply six community-based antiviral drug distribution sites and supplied oseltamivir to local hospitals that could not access commercial supplies.
Current 2009-H1N1 influenza A antiviral treatment guidelines emphasize prompt treatment of all persons in high-risk groups to prevent severe complications and hospitalization. Yet, neither the healthcare system nor the public health system has the capacity to rapidly deliver treatment during a large-scale outbreak. Our experience suggests that the commercial drug distribution system (pharmacies) could play a potentially important role but may not be prepared at this time coordinate such a response with local public health agencies or to respond to unanticipated surge in demand for treatments.
School Closures and Community Mitigation Measures
On April 29th the first probable cases of 2009-H1N1 influenza A were announced in King County, including cases in school-aged children. CDC guidance at that time recommended precautionary school closure for cases of 2009-H1N1 influenza A (swine influenza) in students, and by May 1st, four elementary schools and one middle school in the county were closed by the local health officer. The benefits and potential unintended adverse consequences of school closures during influenza pandemics are not well defined (Cauchemez et al., 2009). National pandemic planning guidance focused on proactive school closures to decrease community transmission; reactive closures were not a focus of the plan (CDC, 2009c).
Notification of immediate reactive school closures were issued after public health officials were informed of laboratory-confirmed cases of 2009-H1N1 influenza A in students, typically late in the day after schools had been dismissed. This complicated communication with both school officials and the public. School officials used flyers, email, and phone messaging in attempts to contact parents. Communication was especially challenging with non-English-speaking families and single working parent households, where telephone contact numbers for some families were not available or current. In some cases, children from closed schools gathered at other locations and continued to participate in organized sports leagues. Following the closure of an elementary school serving diverse immigrant families who reside in a nearby low-income housing project, school children congregated in apartments and common areas of the housing project, frequently without the supervision of parents or other caregivers. Systems to allow education at home for students who were not ill were not in place at the time of the closures but may have been possible with sufficient advance warn-
ing. A minority of public school districts have sufficient resources to include elementary schools on intranet websites used to communicate instructions to students and parents.
In Seattle public schools this school year, 44 percent of children qualify for nutritional support through the National School Lunch and School Breakfast Programs (State of Washington Office of the Superintendent of Public Instruction). During the spring outbreak there was no means to provide meals to children dependent on these programs when schools closed for a health emergency. Subsequently, the United States Department of Agriculture (USDA) authorized provision of reimbursable meals for schools closed for 2009-H1N1 influenza A and not for other health emergencies.21 The provision is only applicable when schools are closed by health officials and not when school administration close schools because of a high level of illness in students or faculty. Significant local capacity and resource issues remain that must be addressed in order to provide meals to low-income children during school closures for 2009-H1N1 influenza A.
Because ILI was widespread in the community and the majority of cases were likely not diagnosed and reported, and because most cases appeared comparable to seasonal influenza, we discontinued the policy of reactive school closures locally on May 5th. After that date, we recommended keeping all students with symptoms of influenza out of school during their period of illness and recuperation, when they are potentially infectious to others. However, there was no practical way to ensure that ill children did not attend school. Schools and child daycare centers did not have the capacity to screen students for illness at entrance each day, and the responsibility fell primarily to parents. There was significant variability in the degree to which parents adhered to, and schools enforced, the recommended 7-day exclusion period for ill students. Some parents brought ill children to school, sometimes after administering fever-reducing medications, and were not available to retrieve the children. School officials speculated that working parents in lower socioeconomic status neighborhoods were concerned about losing wages and/or employment if they had to miss work to be with an ill child. Many parents and healthcare providers felt that the severity of illness did not justify extraordinary exclusion measures, and some healthcare providers provided “doctor’s notes” to students with ILI authorizing return to school before the exclusion period had been completed. Based on this experience, we used resources from a National Association of County and City Health Officials (NACCHO) Advanced Practice Center Grant to develop educational materials for parents on how to recognize flu symptoms, and that encouraged them to develop strategies ahead of time so that children can stay home even if parents must continue to work. The materials created through the grant are made available to health jurisdictions nationally by NACCHO. Our health department designated a full-time liaison to schools to provide guidance and consultation
during the 2009-H1N1 influenza A response, but resources are not available to allow this work to continue.
The school closure experience illustrated the value of incorporating local flexibility in national guidance and this was recognized in subsequent CDC school closure and illness exclusion guidance. Strategies for successful implementation of both proactive and reactive school closures should be incorporated into pandemic and other health emergency plans, including improved coordination and implementation of school closures by public health, school, and local emergency management agencies. Distinguishing between school closure and dismissal of classes should also be considered to allow administrative and other social services functions at schools to continue during health emergencies.
Communication
Intense national and local media coverage of severe cases and school closures contrasted with official statements and the public perception that the pandemic was “mild” and comparable in severity to seasonal influenza, complicating risk communication messaging. Media also showed high interest in the extent and progression of the outbreak, focusing on the latest confirmed case counts. In order to provide timely and accurate information to the local community, frequent press conferences were held that resulted in local media stories and coverage largely reflecting public health messages.
Public messaging regarding when to seek healthcare, however, failed to prevent a surge of mildly ill persons to healthcare facilities or to reduce the demand for testing and treatment of mild cases. Communication strategies and related methods that increase public compliance with recommendations on when to seek healthcare and treatment and community mitigation recommendations would improve local responses to future health emergencies. The use of standardized triage tools for the public should be explored and evaluated to minimize unnecessary use of limited healthcare system resources.
In order to manage the large number of telephone calls from healthcare providers and the public, we operated a public flu telephone hotline. Mobilization of the hotline was difficult because there were not enough expert clinicians available on short notice to rapidly develop phone protocols and train call center staff. In addition, H1N1 topics recorded in English and Spanish were available around the clock; about half of callers used these recordings only.
Coordinating information provided through the public health hotline and by multiple local healthcare systems was challenging and not specifically addressed in pandemic plans. Crafting appropriate messages for persons having ILI but with no regular healthcare provider or health insurance was problematic given the fact that these persons typically use EDs for primary care services.
In Washington State there is no requirement that healthcare providers obtaining or renewing a professional license provide contact information that can be
used for communication during public health emergencies. Consequently, many healthcare providers are “not plugged in” to public health communication networks. Coverage is especially poor for nonprimary care and non-infectious disease practitioners and among clinicians practicing in certain ethnic communities.
An informal survey of clinicians who voluntarily subscribe to the Department’s health alert listserv found approximately half of the respondents complained of “information overload” and of receiving similar (and sometimes conflicting, such as for PPE guidance) messages from national, state, and local public health agencies as well as professional societies and other sources, whereas the other half found the volume of information useful and appropriate. Healthcare providers that had planned for pandemic response reported that it had been useful; however, many practices, particularly smaller ones, had not planned. Communication from CDC to local public health was good, and it was improved over that during the SARS outbreak. Multiple venues were available for local health officials to engage CDC staff on a variety of response topics through regularly scheduled conference calls. On several occasions, it appeared that the release of updated national guidance was delayed for clearance by government agencies. A review of official clearance methods may be useful in order to ensure that the most efficient system is in place to expedite release and dissemination of new and revised guidance, an essential component of effective real-time outbreak management.
H1N1 Vaccination Program
The need to rapidly plan and implement a large-scale vaccine distribution and administration system severely taxed local public health capacity. In King County, 724 health care providers and/or vaccination clinic sites enrolled to provide H1N1 vaccine compared with 240 for our preexisting local Vaccines for Children (VFC) program managed by public health staff. Because there is no national program for adult immunization delivery analogous to the VFC program, healthcare providers that care for adult populations prioritized for 2009-H1N1 influenza A vaccination (including obstetricians, internal medicine specialists, and specialty clinics for high-risk adults such as HIV clinics and dialysis centers) are unfamiliar with procedures for ordering vaccine through the public health system and the associated administrative requirements. Many target groups for 2009-H1N1 influenza A vaccination have low seasonal influenza vaccination coverage rates, creating additional challenges. For example, in order to optimize access to vaccine for pregnant women, we assigned a special liaison to work with local birthing hospitals to ensure that the vaccine is available through either the hospital or the obstetrical practices admitting patients to the facility.
Ongoing uncertainty regarding the timing of availability and expected quantities of specific vaccine formulations, presumably due to the unpredictability of the manufacturing process, presented significant problems for local immunization
planners. Although initial limited doses of live attenuated influenza virus vaccine (LAIV) were targeted to eligible populations of healthcare workers and healthy persons in early October by limiting the number of vaccination sites, expanding vaccine availability to high-risk target populations in the medical home and in the community requires significant supplies of injectable vaccines. Another difficulty hindering vaccine distribution and expanded access through the medical home is that the currently licensed injectable monovalent 2009-H1N1 influenza A vaccine for young children is especially scarce.
Misperceptions about vaccine safety among the public as well as among healthcare professionals have added to the complexity of the 2009-H1N1 influenza A vaccination program and engendered additional work for public health personnel who need to educate both healthcare system stakeholders and the public. Misinformation about the manufacturing process, use of adjuvants, risk of adverse events, and inadequate safety testing circulated for months prior to the release of the first doses of vaccine and before accurate and authoritative public information was made available on the CDC website (Steinhauer, 2009). Many healthcare professionals persist with concerns regarding the safety of LAIV (e.g., FluMist®) in eligible healthcare workers despite clear recommendations for this group from CDC and the vaccine’s excellent safety record. A recent study showing decreased efficacy of LAIV against seasonal influenza in adults exacerbated concerns about the acceptability of LAIV among healthcare workers (Monto, 2009). Anxiety about the mercury (thimerosal) content of vaccines also threatens to decrease public acceptance of 2009-H1N1 influenza A vaccination, particularly among populations that have become accustomed to or favor receiving thimerosal-free injectable formulations that are in short supply. This problem is exacerbated in Washington, where the state legislature passed a law in 2007 prohibiting administration of vaccines containing more than a trace of mercury to pregnant women and children less than three years of age. Paradoxically, when the law was suspended through an emergency order by the state secretary of health because of a shortage of thimerosal-free formulations of 2009-H1N1 influenza A vaccine, it activated a provision in the law requiring healthcare providers to notify the parent or guardian of all children less than 18 years of age and pregnant or lactating women that they are receiving a vaccine with “more mercury than is usually allowed.” Few local obstetrical practices are willing to offer thimerosal-containing injectable vaccine to pregnant patients despite having no other available option, and virtually all healthcare providers have expressed a preference for thimerosal-free vaccine. Sustained efforts to educate and increase the public’s trust in immunizations and vaccine safety would be useful during health emergencies due to vaccine-preventable diseases.
Vaccine supply shortfalls further complicated local vaccine delivery and public messaging. Optimistic national forecasts for vaccine availability raised expectations among the public and influenced public health vaccination program strategy. Early media reports heralded the onset of the national 2009-H1N1
influenza A vaccination program and the arrival locally of the first doses of 2009-H1N1 influenza A (LAIV) vaccine. This further raised interest among healthcare providers and the public, including target populations with underlying medical or age-related risks for severe illness and pregnant women who are not eligible for LAIV (which was the predominant formulation initially available). Local vaccination program implementation plans did not emphasize strategies for supply shortages because initial national supply projections were robust. For example, national forcasts predicted that by the end of October, vaccine supply would be adequate to cover 75 percent of the local target population recommended for vaccination by the CDC’s Advisory Committee on Immunization Practices (ACIP; CDC, 2009d). However, by the time the second wave of 2009-H1N1 influenza A peaked locally in late October, King County had only received enough vaccine to immunize approximately 17 percent of the ACIP target population. Variable practices across jurisdictions occurred related to restriction of eligibility for vaccination to one or more subsets of the target population as described in the ACIP guidance and in the use of sequential versus simultaneous vaccination of risk groups within the target population and/or target population subsets. Most health care systems offered vaccine to all their healthcare workers, not just those in direct patient contact, before making it available to patients.
The ability to implement the 2009-H1N1 influenza A vaccination program vastly exceeded our routine public health immunization program capacity. Our Department’s immunization program includes a total of 10.5 FTEs, including both public health nurses and administrative staff engaged in the administration of the VFC program and associated activities. In order to manage enrollment of new vaccination providers; provide technical support to participating healthcare providers; conduct necessary oversight and management of vaccine ordering, delivery, and utilization; and continue routine VFC program operations for all other recommended vaccines that healthcare providers must continue to administer during this time, more than 16 new full-time staff were required. As with other public health surge staffing, finding appropriately trained and experienced staff to supplement the small core of public health program experts is difficult if not impossible on short notice.
The primary strategy for administering 2009-H1N1 influenza A vaccine locally is to use the existing healthcare system or “medical home,” including safety-net clinics, supplemented by regional pharmacies and a relatively small number of special community vaccination clinics. Due to repeated funding reductions in recent years, the department has minimal capacity to provide clinical immunization services. Immunization clinic nurse capacity was reduced by 70 percent from 2004 to 2009, from 10.3 to 3.0 FTEs. As a consequence, the department does not have the ability to staff community school-based or other vaccination clinics. A recent survey in Washington State found that, on a typical day, 31 of the state’s 36 local health jurisdictions had one vaccinator available to provide routine immunizations and only two jurisdictions had four vaccinators available.
In addition to diminishing immunization nurse capacity, a number of other positions relevant to pandemic and other health emergency response activities were reduced or eliminated due to budget cuts in 2009: the Department’s Child Care Health program including six public health nurses, a nutritionist, an educator, and administrative support; our Children with Special Health Care Needs program with 3.5 public health nurse positions; and the loss of three death investigator positions in the Medical Examiner’s Office. This predicament is not unique among local health jurisdictions. NACCHO reports that, in 2008, 27 percent of local health departments nationally had budget cuts and 53 percent had layoffs resulting in 7,000 jobs lost; in 2009, 44 percent of local health departments had budget cuts and 32 percent had layoffs with approximately 8,000 jobs lost and an additional 12,000 local health department (LHD) employees subjected to reduced hours or mandatory furloughs (NACCHO, 2009). Federal “flu allocation” stimulus funding helps mitigate additional unbudgeted expenses but does not provide real-time capacity or sustainable resources.
Summary
Local public health capacity to respond to the 2009-H1N1 influenza A outbreak and other large-scale health emergencies is tenuous and unstable, waxing and waning with availability of year-to-year grants and other short-term funding supplements. We have used CDC preparedness funds, one-time pandemic flu allocations, and NACCHO grants to develop, enhance, and test a wide range of response capabilities for influenza pandemics and other public health emergencies. With these resources, we were able to develop capacity in a number of areas critical to effective outbreak and health emergency response that were essential to our 2009-H1N1 influenza A outbreak response. Examples include creation and staffing of a regional Healthcare Coalition to coordinate public health and healthcare system preparedness planning and develop community-wide medical surge capabilities; the capability to activate and manage alternate care facilities; establishing a toll-free public hotline incorporating nurses on-site as well as an external nurse consultation line; and integration of public health preparedness activities with those of organizations serving vulnerable populations (including development of a communication tool to rapidly connect with these critical partners during disasters). In addition, key disease surveillance, investigation, and response positions (including for the development and operation of syndromic and school absenteeism surveillance and epidemiologic response), our communications team’s emergency preparedness and response activities, and emergency preparedness planning for vulnerable populations are all dependent on federal grants funds and other limited, competitive awards.
In addition to local budget cuts that reduced the core public health workforce, our response to the 2009-H1N1 influenza A outbreak has been hampered by significant reductions over the past three years in federal public health preparedness
funding. CDC preparedness grants for building and sustaining core capabilities have been reduced by 37 percent since 2006. Training funds to prepare staff for their emergency roles has dropped by 30 percent. One-time federal pandemic flu preparedness funds, which accounted for nearly 20 percent of our preparedness resources in 2006, have not been restored. Consequently, key staff positions responsible for disease surveillance, vulnerable populations planning, risk communications, medical surge planning, and other core preparedness capabilities were recently eliminated. As a result of these cuts, surge capacity planning with key community organizations such as long-term-care facilities, nursing homes, and ambulatory care facilities has been suspended. Similar reductions in funding for hospital emergency preparedness activities threaten the future viability of our regional healthcare coalition and the medical response capacity for future large-scale public health emergencies. Unlike certain material resources, public health personnel trained in emergency response are not a just-in-time commodity.
Conclusion
The spring 2009-H1N1 influenza A outbreak highlighted accomplishments, gaps, and challenges in the local public health and medical response to large-scale health emergencies. It also provided opportunities for improvement across the health emergency response spectrum that should be addressed before the next inevitable outbreak or natural disaster. Inadequate long-term sustainable funding for both core public health and health emergency preparedness undermines the ability of local communities to adequately prepare for and respond to large-scale health emergencies of any type.
Acknowledgments
The author gratefully acknowledges the ongoing work of many colleagues at Public Health–Seattle and King County responding to the 2009-H1N1 influenza A outbreak, and all who reviewed this manuscript and/or provided valuable comments, including James Apa, Atar Baer, Julia Byrd, Joe Cropley, Cynthia Dold, Carina Elsenboss, Paul Etkind, David Fleming, Tao-Sheng Kwan Gett, Michael Loehr, Jennifer Lloyd, Pegi McEvoy, Shelly McKeirnan, Margaret Neff, Betsy Pesek, David Ralphs, Krista Rietberg, Anne Shields, Mark Tonelli, and Diane Young.
References
ANZIC Influenza Investigators, S. A. Webb, V. Pettilä, I. Seppelt, R. Bellomo, M. Bailey, D. J. Cooper, M. Cretikos, A. R. Davies, S. Finfer, P. W. Harrigan, G. K. Hart, B. Howe, J. R. Iredell, C. McArthur, I. Mitchell, S. Morrison, A. D. Nichol, D. L. Paterson, S. Peake, B. Richards, D. Stephens, A. Turner, and M. Yung. 2009. Critical care services and 2009 H1N1 influenza in Australia and New Zealand. New England Journal of Medicine 361(20):1925-1934.
APIC (Association of Professionals in Infection Control). 2009. SHEA, APIC, ACOEM, and IDSA joint position statement: healthcare personnel at high-risk for severe influenza illness: care of patients with suspected or confirmed novel H1N1 influenza A, August 11, 2009, http://www.apic.org/Content/NavigationMenu/GovernmentAdvocacy/IssuesInitiatives/PersonalProtectiveEquipmenttoProtectHealthcareWorkersfromNovelH1N1Influenza/Personal_Protective_.htm (accessed October 29, 2009).
Cauchemez, S., N. M. Ferguson, C. Wachtel, A. Tegnell, G. Saour, B. Duncan, and A. Nicoll. 2009. Closure of schools during an influenza pandemic. Lancet Infectious Diseases 9(8):473-481.
CDC (Centers for Disease Control and Prevention). 2009a. Interim guidance on infection control measures for 2009 H1N1 influenza in healthcare settings, including protection of healthcare personnel, http://www.cdc.gov/h1n1flu/guidelines_infection_control.htm (accessed October 29, 2009).
———. 2009b. Summary of HICPAC recommendations for care of patients with confirmed or suspected 2009 H1N1 influenza infection in healthcare settings, July 23, 2009, http://www.cdc.gov/ncidod/dhqp/hicpac_h1n1.html (accessed October 29, 2009).
———. 2009c. Interim pre-pandemic planning guidance: community strategy for pandemic influenza mitigation in the United States, http://www.flu.gov/professional/community/index.html (accessed October 30, 2009).
———. 2009d. Use of influenza A (H1N1) 2009 monovalent vaccine: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2009. Morbidity and Mortality Weekly Report 58(RR-10):1-8.
HHS (Department of Health and Human Services). 2006. Pandemic influenza implementation plan, part I of II. Washington, DC: HHS.
———. 2009. HHS pandemic influenza plan supplement 2 laboratory diagnostics, http://www.hhs.gov/pandemicflu/plan/sup2.html#s2-II (accessed October 29, 2009).
IDSA (Infectious Disease Society of America). 2007. Pandemic and seasonal influenza: principles for U.S. action. Alexandria, VA: IDSA, http://www.idsociety.org/influenza.htm (accessed October 29, 2009).
———. 2009. IDSA endorses SHEA position statement on infection control, June 11, 2009, http://www.idsociety.org/pandemicinfluenzapolicy.htm (accessed October 29, 2009).
Knebel, A., and E. Trabert, eds. 2007. Medical surge capacity and capability handbook, second edition. Washington, DC: HHS, http://www.hhs.gov/disasters/discussion/planners/mscc/ (accessed October 29, 2009).
Kwan-Gett, T., A. Baer, and J. Duchin. 2009. Spring 2009 H1N1 influenza outbreak in King County, Washington. Disaster Medicine and Public Health Preparedness 3:S109-S116.
Monto, A. S. 2009. Comparative efficacy of inactivated and live attenuated influenza vaccines. New England Journal of Medicine 361(13):1260-1267.
NACCHO (National Association of County and City Health Officials). 2009. Survey of local health department job losses and program cuts. Washington, DC: NACCHO, http://www.naccho.org/advocacy/upload/LHD-job-and-service-cuts-2009.pdf (accessed December 17, 2009).
SHEA (Society for Healthcare Epidemiology of America). 2009. SHEA position statement: interim guidance on infection control precautions for novel swine-origin influenza A H1N1 in healthcare facilities, http://www.shea-online.org/Assets/files/policy/061209_H1N1_Statement.pdf (accessed October 29, 2009).
Steinhauer, J. 2009 (October 16). Swine flu shots revive debate about vaccines. New York Times, http://www.nytimes.com/2009/10/16/health/16vaccine.html (accessed November 30, 2009).
White, D. B., and D. C. Angus. 2009. Preparing for the sikest patients with 2009 influenza A H1N1. Journal of the American Medical Association 302(17):1905-1906.
WHO (World Health Organization). 2009. Infection prevention and control in health care for confirmed or suspected cases of pandemic (H1N1) 2009 and influenza-like illnesses, June 25, 2009, http://www.who.int/csr/resources/publications/swineflu/swineinfinfcont/en/index.html (accessed October 29, 2009).