2
Overview
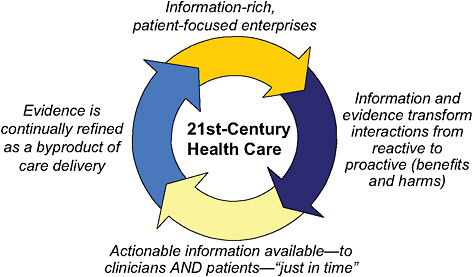
Dr. Amy Abernethy of Duke Comprehensive Cancer Center and Lynn Etheredge of George Washington University began the workshop by giving an overview of what an ideal RLHS entails, how a RLHS might improve care, and how it differs from what is currently standard practice in cancer care. The originator of the concept of a learning healthcare system, Etheredge defined a cancer RLHS as one that generates as rapidly as possible the evidence needed to deliver the best care for each cancer patient. Such a system bridges the gap between clinical research and clinical practice “to learn as much as possible as soon as possible” by enabling the collection of data at the point of care that can then be used to inform clinical, payer, and policy decisions. “For each cancer, each stage, all the way down to each patient, we want to be able to do what physicians and patients really need, which is to be able to tell them the available treatments, their comparative effectiveness, and how to personalize their decision making. That is a long way from where we are now in terms of results,” he said.
RLHS is both patient centered and system centered, a point underscored by both Etheredge and Abernethy. Data collected at the individual patient level not only inform care for that person, but also contribute to evidence development and systemic improvements, along with other data collected system-wide using the experience of all cancer patients, as well as results of clinical trials, systematic reviews, and other relevant aggregated information. In addition, patient data can be used for large-scale evidence
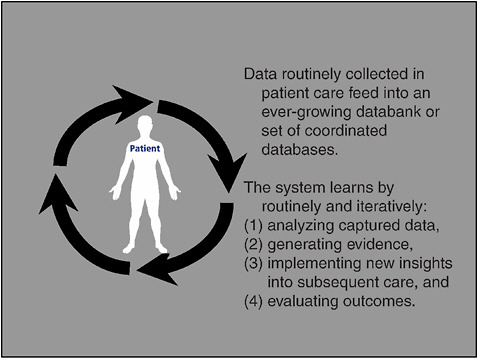
FIGURE 2-1 A patient-centered model of a RLHS.
SOURCE: Etheredge and Abernethy, 2009.
synthesis, comparative effectiveness research (CER), discovery, and evidence implementation on the health system and national levels (see Figure 2-1). Such a system is built at the patient level and scaled to the societal level.
Dr. Abernethy views the critical elements of a RLHS as being linked information, motivated individuals, and systems that are engaged to provide reliable integrated information. She expanded on the patient-centered perspective of a RLHS. “In order to have rapid-learning health care, we have to have rapid-learning health care around patients,” she stressed. “The care of the individual patient is informed by the care of people coming before, and his or her care also informs the care of people in the future in a circuitous way.” (See Box 2-1.)
For purposes of illustrating the way cancer care is currently delivered (i.e., in a data-poor and slow-learning environment), Dr. Abernethy discussed the care she recently gave one of her patients. This 37-year-old woman had newly diagnosed Stage IIIB melanoma. She was considering starting a family and wanted to know what adjuvant treatments might reduce her risk of death at five years, and how such treatment would affect her quality of life and ability to become pregnant. Although the National
|
BOX 2-1 A Rapid Learning Healthcare System A rapid learning healthcare system (RLHS) is one that uses advances in information technology to continually and automatically collect and compile from clinical practice, disease registries, clinical trials, and other sources of information, the evidence needed to deliver the best, most up-to-date care that is personalized for each patient. That evidence is made available as rapidly as possible to users of a RLHS, which include patients, physicians, academic institutions, hospitals, insurers, and public health agencies. A RLHS ensures that this data-rich system learns routinely and iteratively by analyzing captured data, generating evidence, and implementing new insights into subsequent care. SOURCES: Adapted from Etheredge, 2007; IOM, 2007. |
Comprehensive Cancer Network (NCCN) guidelines offered three treatment options, a lack of comparative data prevented delineation of the best option for her, in terms of improving survival and quality of life. The guidelines also lacked evidence from a recently published study indicating that shorter-dose interferon therapy might be just as effective as longer-term treatment. There also was no evidence provided on how such treatments might affect fertility. As Dr. Abernethy pointed out, for this patient, “I can roughly predict her odds of surviving, but I cannot really refine that or personalize it using data from recently treated patients like her. I cannot determine which is the right adjuvant program for her, and I do not have any clue about the risk of infertility. Her mother died of melanoma, and she worries that what happens for her does not really [clinically] impact people like her in the future at all, and that was really very distressing for her.”
In contrast, if Dr. Abernethy were part of a functioning RLHS, she would continually be collecting information about how various treatment options are affecting her patients. That information would be added to such point-of-care data collected on other patients throughout the country, or even globally, as well as to data collected from clinical trials and other sources, and the aggregate information would be used for real-time analyses to determine the best treatment for her individual patients at the time care
is provided. Dr. Abernethy currently is able to access comparative clinical data only from the hospital in which she practices, and then only through laborious and slow manual methods.
Explaining the system-centered aspects of a RLHS, Dr. Carolyn Clancy, director of the Agency for Healthcare Research and Quality (AHRQ), stressed that such a system ideally would be able to produce evidence on the comparative effectiveness of cancer care options. This comparative effectiveness research would be facilitated by the data collected on each treated patient in a RLHS. Another critical aspect of a cancer RLHS is that it engages system-wide learning, using the experience of all cancer patients, not just those enrolled in clinical trials.
“Only a very small percentage of cancer patients today have their key clinical data captured for research purposes. We need to think about the potential, with our new computers and EHRs, of capturing the key data from virtually every patient and feeding that into a learning system to try to learn as much as possible,” Etheredge said. He added that such a system-wide approach would engage virtually all oncologists and cancer clinics in an active research enterprise, as opposed to the small percentage who currently participates in clinical cancer research. This data-rich system of the future would learn routinely and iteratively by linking and analyzing captured patient data, linking patient data to clinical trials and other research data, generating evidence, and implementing new insights into subsequent care (see Figure 2-2).
This RLHS is in contrast to the current system of clinical learning, in which it often takes more than five years to develop a large Phase III cancer clinical trial, accrue patients, and generate evidence (Dilts et al., 2008) and an additional 10 years before that evidence substantially changes clinical practice. Although randomized clinical trials (RCTs) are the gold standard for the development of clinical guidelines and are an essential component in expanding the knowledge base in oncology, in a RLHS the results of RCTs would be complemented by data collected internally within the healthcare system as well as external linked information, such as genomic and molecular data.
Enabling RLHS are advances in in silico research, which Etheredge defined as research on computerized databases. Such research brings together the extraordinary power of high-speed computing with the data-rich capabilities of large computerized databases and distributed database networks. Internet capability also enables data to be accessed by researchers around the globe. In addition, computer advances enable the complex
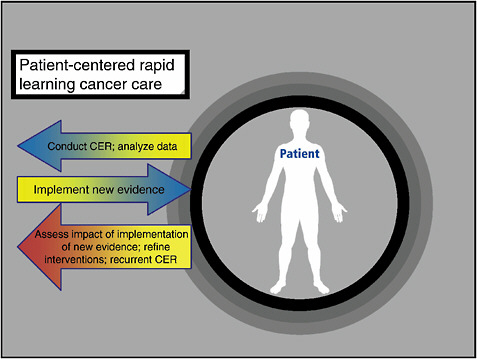
FIGURE 2-2 A rapid learning patient-centered system for cancer care encompasses information and data gleaned from patient care, continuously analyzing and implementing new evidence.
SOURCE: Etheredge and Abernethy, 2009.
genetic analyses that underlie efforts to personalize medicine. “Researchers plus high-speed computers plus great databases equal rapid learning,” Etheredge said.
Dr. Abernethy stressed that a RLHS “provides a means through which we can achieve a number of important national goals, including comparative effectiveness research, improved healthcare quality, personalized medicine, and patient-centered care.” Dr. Clancy expanded on some of these goals and added other challenges that a RLHS might help address, including concerns about healthcare spending, pervasive problems with quality of care, and uncertainty about best practices. Data collected by her agency and the Dartmouth Atlas of Health Care revealed that the quality of health care delivered in this country varied considerably by region, literally all over the map, with some regions outperforming others, and that furthermore there was a lack of correlation between the amount of Medicare spending and the quality of care received (Fisher et al., 2003a, 2003b; Hossain, 2009).
“We speculate a lot, but we urgently need to learn why that is. We are not going to achieve and sustain health reform without getting some of these answers and bringing it right back to the level of clinicians and patients,” Dr. Clancy said.
Dr. Clancy suspects that much of the variability in care stems from clinical uncertainty about best practices and treatments—knowledge gaps on the comparative effectiveness of various options that could be ameliorated by gathering more information from patients. “There is a big gap between best possible care and care that is routinely delivered just about everywhere,” she said. “We do not reward or create a space to learn from systems that are way ahead.” In theory, a RLHS should improve the uniformity and quality of care by providing such a learning space and by promulgating appropriate standards of care. “We can use this learning healthcare system approach to figure out how to make sure that we more rapidly translate those very promising advances and make sure that they get to the patients likely to benefit. At a system level we want to know with precision how well we are getting what is learned into practice,” Dr. Clancy said.
Dr. Clancy referred to the six elements of good care—care that is safe, timely, effective, efficient, equitable, and patient centered—criteria previously delineated by the IOM (2001). Quality measures for various healthcare practices and health systems can be privately or publicly reported so as to provide an impetus for improving care and a means to link payment to performance. Yet such collection and reporting of information are not consistently done because “we do not have good systems in place” to do so, she said. A system-wide approach is also vitally needed to coordinate the care that individual patients receive from a variety of specialists, Dr. Clancy added. “We are completely blind because our information systems do not connect,” she said. More collaboration and trust among providers, purchasers, and consumers will be needed to achieve more effortless and transparent information sharing with health IT, ultimately leading to transformation of the healthcare system (see Figure 2-3).